Selected Abiotic and Biotic Environmental Stress Factors Affecting Two Economically Important Sugarcane Stalk Boring Pests in the United States
Abstract
:1. The Plant
2. The Stalk Borers
3. Abiotic Environmental Stress Factors
3.1. Water Deficit or Drought Stress

| Free Amino Acids a | Treatment b | |||
|---|---|---|---|---|
| L 97-128 W | CP 70-321 W | L 97-128 D | CP 70-321 D | |
| Alanine | 3560 ± 193 b | 5836 ± 1324 b | 12,257 ± 1423 a | 7857 ± 2277 ab |
| Arginine | 1436 ± 185 a | 677 ± 52 b | 1084 ±93 a | 855 ± 35 a |
| Aspartic acid | 2074 ± 158 | 2827 ± 233 | 2516 ± 61 | 2118 ± 311 |
| Glutamic acid | 18 ± 18 b | 7 ± 7 b | 126 ± 43 a | 860 ± 8 a |
| Glycine | 396 ± 52 b | 505 ± 37 b | 623 ± 83 ab | 653 ± 173 a |
| Histidine | 598 ± 85 b | 577 ± 77 b | 1642 ± 218 a | 1995 ± 273 a |
| Isoleucine | 605 ± 57 b | 565 ± 54 b | 1293 ± 204 a | 2858 ± 47 a |
| Leucine | 441 ± 79 b | 510 ± 52 b | 776 ± 23 a | 2639 ± 88 a |
| Lysine | 367 ± 94 b | 375 ± 57 ab | 650 ± 80 ab | 514 ± 65 a |
| Methionine | 225 ± 20 b | 239 ± 64 b | 771 ± 88 a | 1241 ± 168 a |
| Phenylanaline | 258 ± 43 b | 226 ± 49 b | 908 ± 88 a | 1008 ± 88 a |
| Proline | 558 ± 36 b | 518 ± 99 b | 1198 ± 89 a | 4062 ± 51 a |
| Serine | 2306 ± 285 c | 4315 ± 623 b | 4468 ± 913 b | 8875 ± 931 a |
| Threonine | 1490 ± 95 b | 2258 ± 192 ab | 2954 ± 444 a | 3887 ± 510 a |
| Tyrosine | 209 ± 24 | 186 ± 26 | 357 ± 84 | 515 ± 60 |
| Valine | 1193 ± 260 b | 1563 ± 30 ab | 2797 ± 502 a | 5584 ± 259 a |
| Essential amino acids c | 6616 ± 145 b | 6992 ± 68 b | 12,878 ± 1472 a | 20,585 ± 983 a |
| Total | 15,749 ± 540 c | 21,189 ± 2108 bc | 24,424 ± 2837 b | 45,528 ± 2656 a |
3.2. Soil Nitrogen
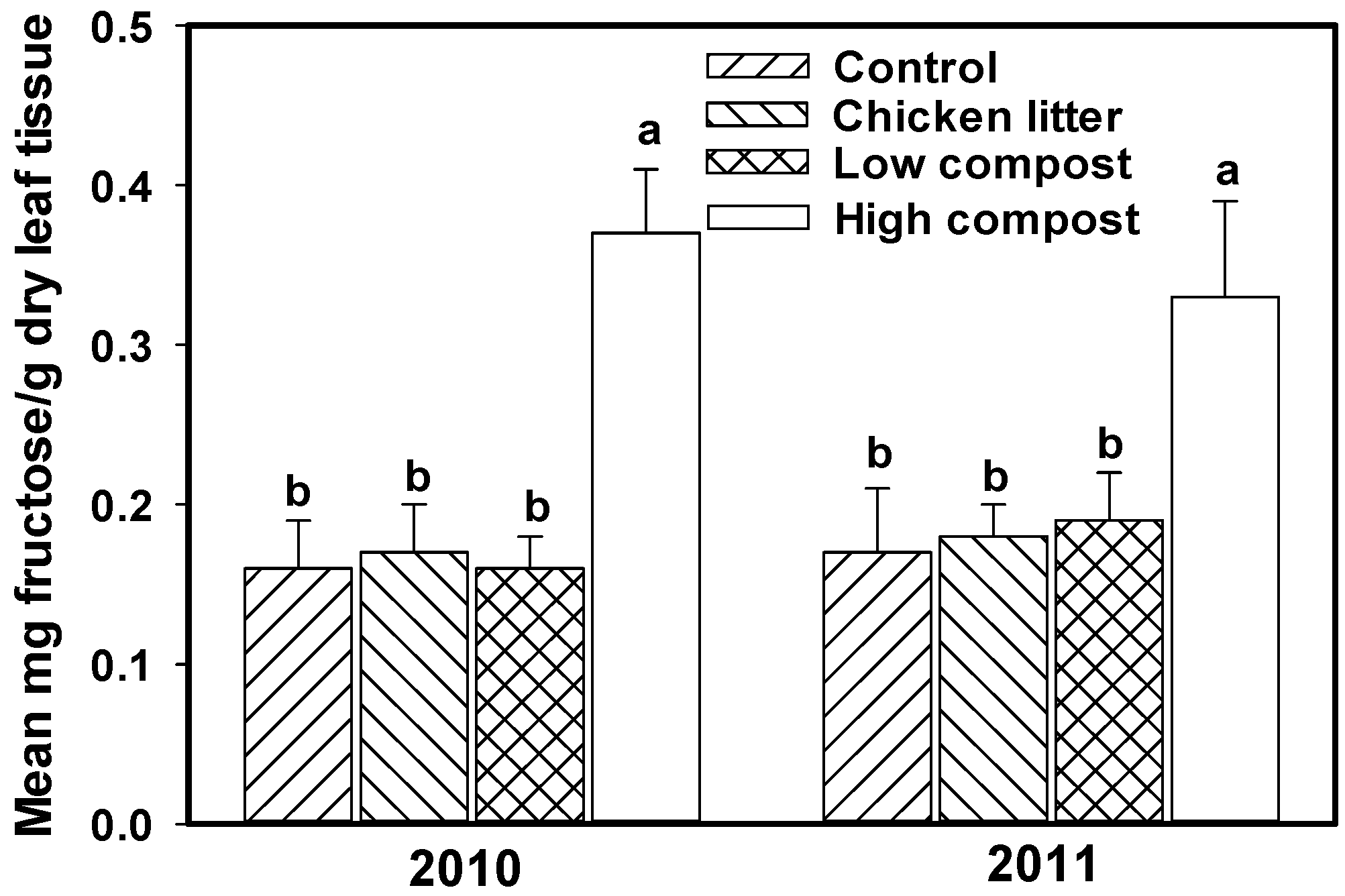
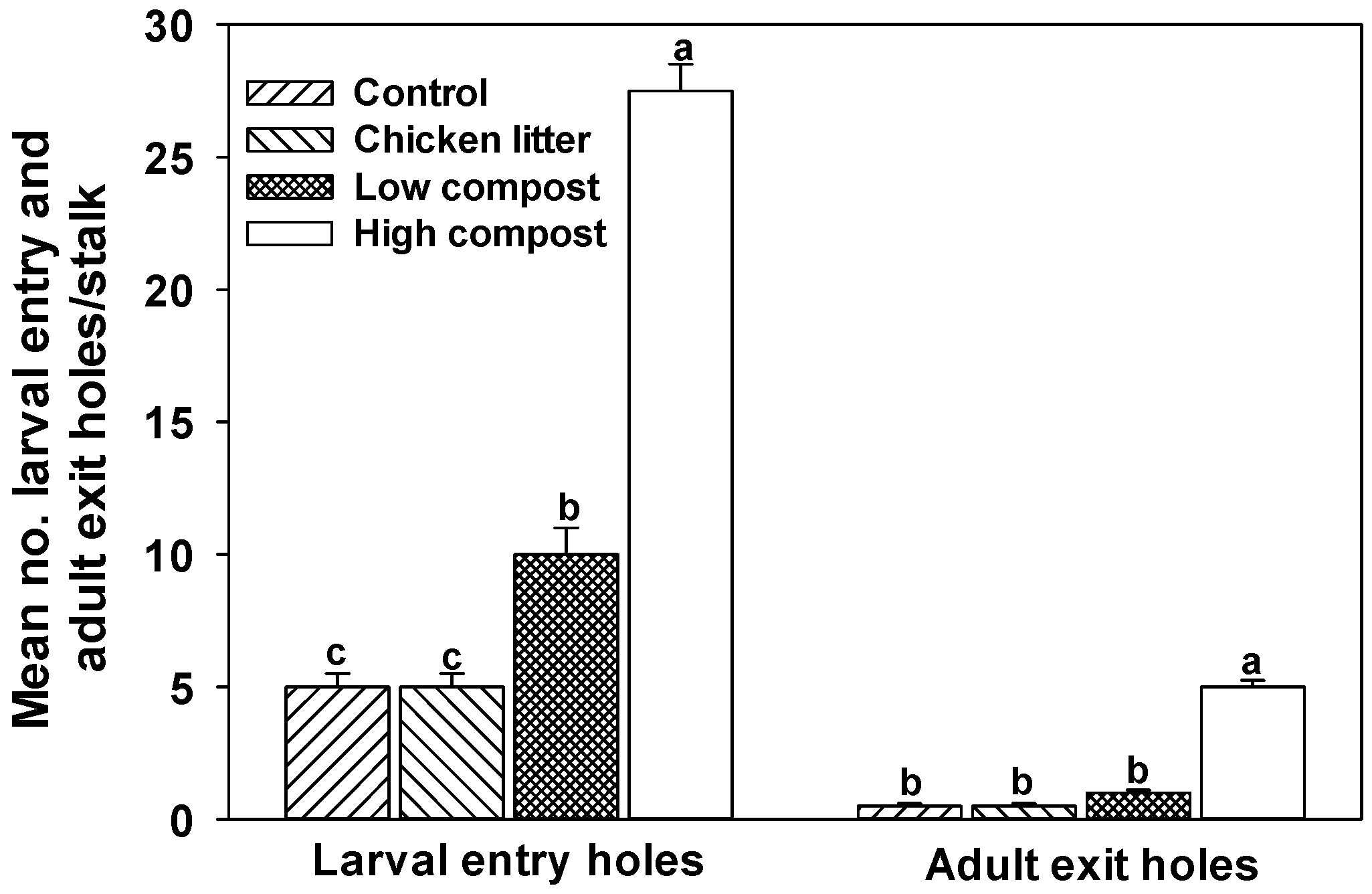
4. Biotic Environmental Stress Factors
4.1. Weed Growth
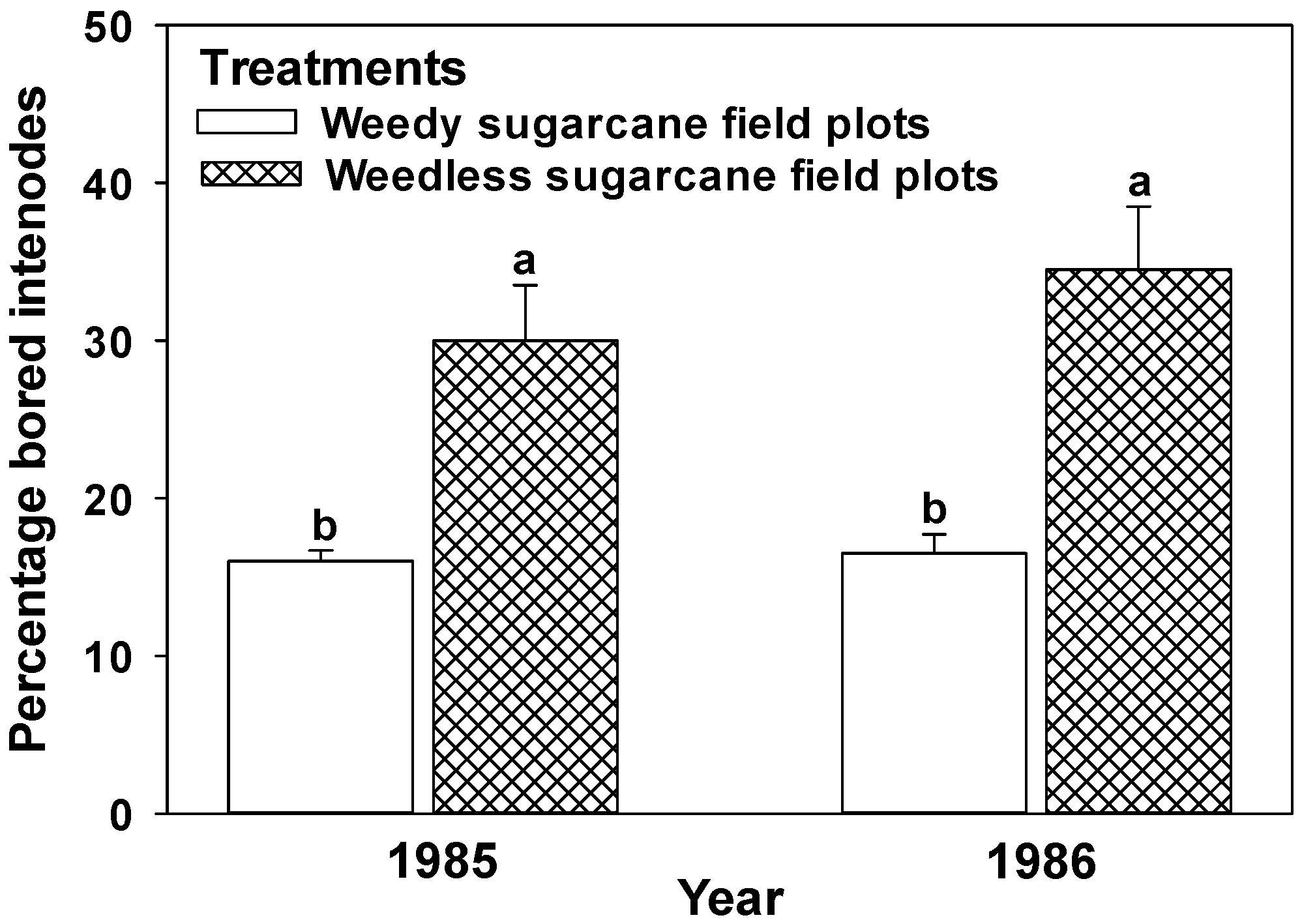
4.2. Greenchop Leaf Residue
4.3. Proximity to Susceptible Maize
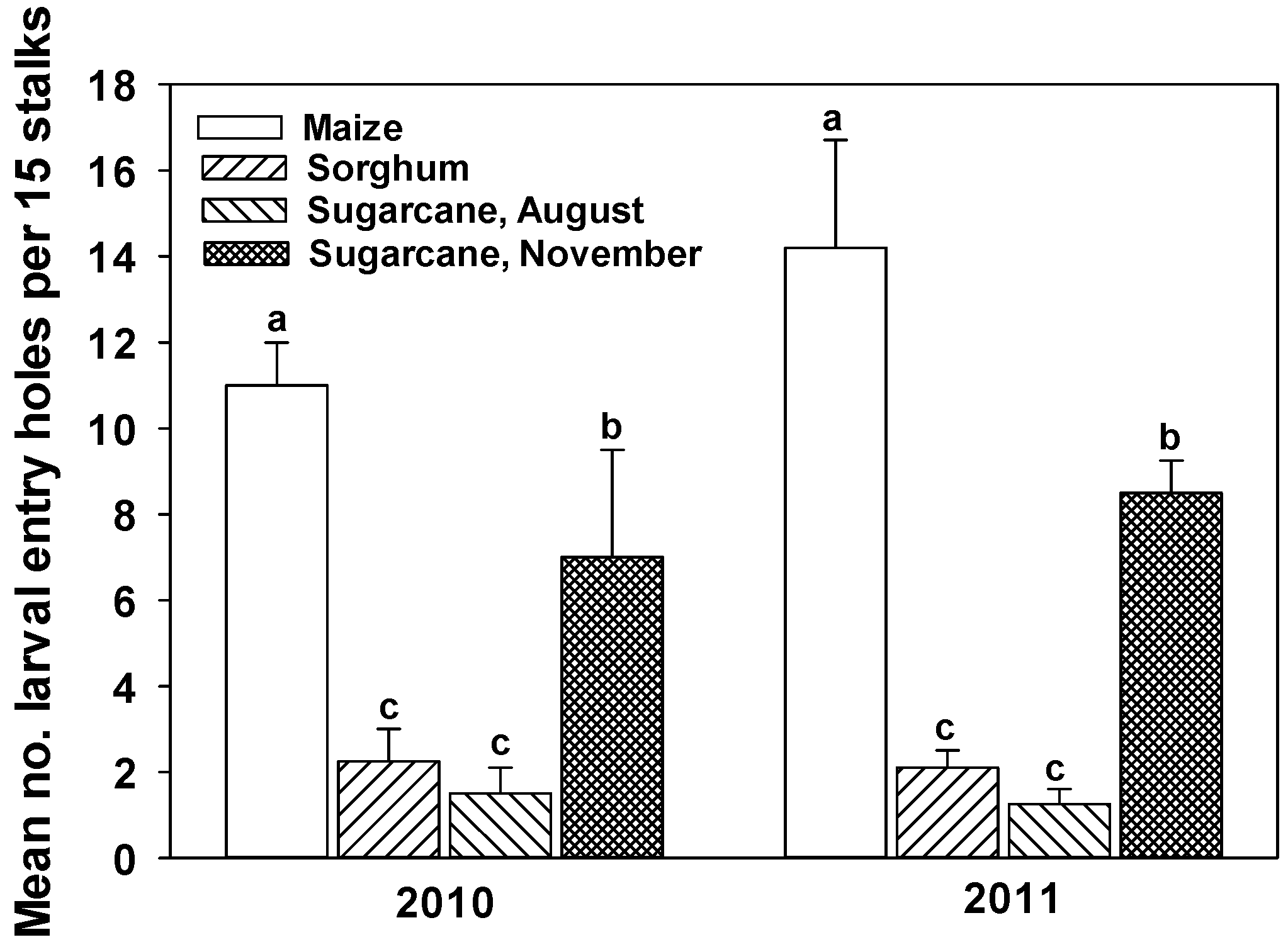
| Pests | ||
|---|---|---|
| Variety and Active Gene | Common Name | Scientific Name |
| Pioneer 31G71 HX1 gene | black cutworm | Agrotis ipsilon Hufnagel |
| corn earworm a | Heliothis zea (Boddie) | |
| European corn borer | Ostrinia nubilalis Hübner | |
| fall armyworm | Spodoptera frugiperda J.E. Smith | |
| lesser corn stalk borer | Elasmopalpus lignosellus (Zeller) | |
| southern corn stalk borer | Diatraea crambidioides (Grote) | |
| southwestern corn borer | Diatraea grandiosella Dyar | |
| sugarcane borer | Diatraea saccharalis (F.) | |
| western bean cutworm | Loxagrotis albicosta Smith | |
| Golden Acres 28V81 VT3Pro gene | corn earworm a | Heliothis zea (Boddie) |
| corn rootworm | Diabrotica spp. | |
| European corn borer | Ostrinia nubilalis Hübner | |
| fall armyworm | Spodoptera frugiperda J.E. Smith | |
| DKC 69-72 (non-Bt) | none | |
| BH Genetics 9050 (non-Bt) | none |
| Cultivar a | No. Internodes Per Stalk | No. Bored Internodes Per Stalk | No. Larval Entry Holes Per Stalk | No. Adult Exit Holes Per Stalk |
|---|---|---|---|---|
| 9050 | 13.0 ± 0.1 | 2.28 ± 0.25 | 5.12 ± 0.34 a | 1.40 ± 0.21 a |
| 69-72 | 13.0 ± 0.01 | 2.15 ± 0.21 | 4.82 ± 0.78 a | 1.38 ± 0.17 a |
| 31G71 | 13.1 ± 0.1 | 1.30 ± 0.44 | 2.52 ± 0.89 ab | 0.45 ± 0.20 b |
| 28V81 | 13.0 ± 0.1 | 0.02 ± 0.02 | 0.05 ± 0.05 b | 0 b |
| F b | 0.47 | 12.61 | 11.96 | 14.41 |
| P | 0.7094 | 0.0001 | 0.0001 | <0.0001 |
5. Conclusions
Conflicts of Interest
References
- FAO (Food and Agriculture Organization of the United Nations). Major Food and Agricultural Commodities and Producers. Available online: http://www.fao.org/ess/top/commodity.html? lang+en&item=156&year=2005 (accessed on 1 July 2015).
- Marsh, J. Sugar—The political economy of a product. J. Sci. Food Agric. 2006, 86, 2516–2522. [Google Scholar] [CrossRef]
- GMO Compass. Sugarcane. Available online: http://www.gmo-compass.org/eng/database/plants/76.sugarcane.html (accessed on 1 July 2015).
- Sharpe, P. Sugar Cane: Past and Present; Southern Illinois University: Carbondale, IL, USA, 1998. [Google Scholar]
- Nass, L.L.; Pereira, P.A.A.; Ellis, D. Biofuels in Brazil: An overview. Crop Sci. 2007, 47, 2228–2237. [Google Scholar] [CrossRef]
- Outlaw, J.L.; Ribera, L.A.; Richardson, J.W.; da Silva, J.; Bryant, H.; Kloss, S.L. Economics of sugar-based ethanol production and related policy issues. J. Agric. Appl. Econ. 2007, 39, 357–363. [Google Scholar]
- Salassi, M.E. Economics of sugarcane production: What does it take for this industry to survive? La. Agric. 2008, 51, 20–24. [Google Scholar]
- NASS (National Agricultural Statistics Service). Sugarcane area harvested, yield, and production—States and United States: 2012–2104. In Crop Production 2014 Summary; NASS, USDA: Washington, DC, USA, 2015; p. 55. [Google Scholar]
- USDA-ERS (U.S. Department of Agriculture-Economic Research Service). U.S. Sugar Production. Available online: http:www.ers.usda.gov/topics/crops/sugar-sweeteners/background.aspx (accessed on 30 July 2015).
- Blackburn, F. Sugar Cane; Longman: New York, NY, USA, 1984. [Google Scholar]
- Edgerton, C.W. Stubble deterioration. Proc. Int. Soc. Sugar Cane Technol. 1939, 6, 334–341. [Google Scholar]
- Hoy, J.W.; Schneider, R.W. Role of Pythium in sugarcane stubble decline: Effects on plant growth in field soil. Phytopathology 1988, 78, 1692–1696. [Google Scholar] [CrossRef]
- Hoy, J.W.; Schneider, R.W. Role of Pythium in sugarcane stubble decline: Pathogenicity and virulence of Pythium species. Phytopathology 1988, 78, 1688–1692. [Google Scholar] [CrossRef]
- Showler, A.T.; Reagan, T.E. Ecology and tactics for control of three sugarcane stalk-boring species in the Western Hemisphere and Africa. In Sugarcane: Production and Uses; Goncalves, J.F., Correia, K.D., Eds.; Nova: Hauppauge, NY, USA, 2012; pp. 1–15. [Google Scholar]
- Fuller, B.W.; Reagan, T.E. The relationship of sweet sorghum plant fiber and survival of the sugarcane borer, Diatraea saccharalis (F.) (Lepidoptera: Pyralidae). J. Agric. Entomol. 1989, 6, 113–118. [Google Scholar]
- Sosa, O. Oviposition preference by the sugarcane borer (Lepidoptera: Pyralidae). J. Econ. Entomol. 1990, 83, 866–868. [Google Scholar] [CrossRef]
- More, M.; Trumper, E.V.; Prola, M.J. Influence of corn, Zea mays, phenological stages in Diatraea saccharalis F. (Lep. Crambidae) oviposition. J. Appl. Entomol. 2003, 127, 512–515. [Google Scholar] [CrossRef]
- Lv, J.; Wilson, L.T.; Longnecker, M.T. Tolerance and compensatory response of rice to sugarcane borer (Lepidoptera: Crambidae) injury. Environ. Entomol. 2008, 37, 796–807. [Google Scholar] [CrossRef] [PubMed]
- Reagan, T.E.; Martin, F.A. Breeding for resistance to Diatraea saccharalis (F.). In Sugarcane Varietal Improvement; Naidu, K.M., Sreenivasan, T.C., Premachandran, M.N., Eds.; Sugarcane Breeding Institute: Coimbatore, India, 1989; pp. 313–331. [Google Scholar]
- Rodriguez-del-Bosque, L.A.; Smith, J.W., Jr.; Martinez, A.J. Winter mortality and spring emergence of corn stalkborers (Lepidoptera: Pyralidae) in subtropical Mexico. J. Econ. Entomol. 1995, 88, 628–634. [Google Scholar] [CrossRef]
- Fuchs, T.W.; Harding, J.A. Seasonal abundance of the sugarcane borer, Diatraea saccharalis, on sugarcane and other hosts in the Lower Rio Grande Valley of Texas. Southwest Entomol. 1979, 4, 125–131. [Google Scholar]
- Showler, A.T.; Reagan, T.E. Effects of sugarcane borer, weed, and nematode control strategies in Louisiana sugarcane. Environ. Entomol. 1991, 20, 358–370. [Google Scholar] [CrossRef]
- Bessin, R.T.; Reagan, T.E. Fecundity of sugarcane borer (Lepidoptera: Pyralidae), as affected by larval development on graminous host plants. Environ. Entomol. 1990, 19, 635–639. [Google Scholar] [CrossRef]
- White, W.H. Movement and establishment of sugarcane borer (Lepidoptera: Pyralidae) larvae on resistant and susceptible sugarcane. Fla. Entomol. 1993, 76, 465–473. [Google Scholar] [CrossRef]
- Capinera, J.L. Sugarcane Borer, Diatraea saccharalis (Fabricius) (Insecta: Lepidoptera: Pyralidae); Entomology and Nematology Department, Florida Cooperative Extension Service, University of Florida: Gainesville, FL, USA, 2001. [Google Scholar]
- Van Zwaluenberg, R.H. Insect enemies of sugarcane in western Mexico. J. Econ. Entomol. 1926, 19, 664–669. [Google Scholar] [CrossRef]
- Osborn, H.T.; Phillips, G.R. Chilo loftini in California, Arizona, and Mexico. J. Econ. Entomol. 1946, 39, 755–759. [Google Scholar] [CrossRef]
- Ogunwolu, E.O.; Reagan, T.E.; Flynn, J.L.; Hensley, S.D. Effects of Diatraea saccharalis (F.) (Lepidoptera: Pyralidae) damage and stalk rot fungi on sugarcane yield in Louisiana. Crop Prot. 1991, 10, 57–61. [Google Scholar] [CrossRef]
- Johnson, K.J.R. Seasonal occurrence and insecticidal suppression of Eoreuma loftini (Lepidoptera: Pyralidae) in sugarcane. J. Econ. Entomol. 1985, 78, 960–966. [Google Scholar] [CrossRef]
- Ulloa, M.; Bell, M.G.; Miller, J.D. Losses caused by Diatraea saccharalis in Florida. Am. Soc. Sugar Cane Technol. 1982, 1, 8–10. [Google Scholar]
- Charpentier, L.J.; McCormick, W.J.; Mathes, R. Beneficial arthropods inhabiting sugarcane fields and their effects on borer infestations. Sugar Bull. 1967, 45, 276–277. [Google Scholar]
- Pollet, D.K.; Reagan, T.E.; White, W.H.; Rester, D.C. Pest Management of Sugarcane Insects; Louisiana State University and Agricultural and Mechanical College, Cooperative Extension Service: Baton Rouge, LA, USA, 1978. [Google Scholar]
- White, W.H.; Reagan, T.E.; Smith, J.W., Jr.; Salazar, J.A. Refuge releases of Cotesia flavipes (Hymenoptera: Braconidae) into the Louisiana sugarcane ecosystem. Environ. Entomol. 2004, 33, 627–632. [Google Scholar] [CrossRef]
- Legaspi, J.C.; Legaspi, B.C., Jr.; Irvine, J.E.; Saldana, R.R. Mexican rice borer, Eoreuma loftini (Lepidoptera: Pyralidae) in the Lower Rio Grande Valley of Texas: Its history and control. Subtrop. Plant Sci. 1997, 49, 53–64. [Google Scholar]
- Reay-Jones, F.P.F.; Wilson, L.T.; Reagan, T.E.; Legendre, B.L.; Way, M.O. Predicting economic losses from the continued spread of the Mexican rice borer (Lepidoptera: Crambidae). J. Econ. Entomol. 2008, 101, 237–250. [Google Scholar] [CrossRef] [PubMed]
- Wilson, B.E.; Hardy, T.N.; Beuzelin, J.M.; VanWeelden, M.T.; Reagan, T.E.; Miller, R.; Meaux, J.; Stout, M.J.; Carlton, C.E. Expansion of the Mexican rice borer (Lepidoptera: Crambidae) into rice and sugarcane in Louisiana. Environ. Entomol. 2015, 44, 757–766. [Google Scholar] [CrossRef] [PubMed]
- Hall, S.G.; Schellinger, D.A.; Carney, W.A. Enhancing sugarcane field residue biodegradation by grinding and use of compost tea. Compost Sci. Util. 2006, 14, 32–39. [Google Scholar] [CrossRef]
- Alves, S.B.; Risco, S.H.; Neto, R.M. Pathogenicity of nine isolates of Metarhizium anisopliae (Metsch.) Sorok. To Diatraea saccharalis (Fabr.). J. Appl. Entomol. 1984, 97, 403–406. [Google Scholar] [CrossRef]
- Legaspi, B.C., Jr.; Legaspi, J.C.; Lauziere, I.; Jones, W.A.; Saldana, R.R. Jalisco fly as a parasitoid of the Mexican rice borer on different host plants. Southwest Entomol. 2000, 25, 77–79. [Google Scholar]
- Rosas-Garcia, N.M. Laboratory and field tests of spray-dried and granular formulations of a Bacillus thuringiensis strain with insecticidal activity against the sugarcane borer. Pest Manag. Sci. 2006, 62, 855–861. [Google Scholar] [CrossRef] [PubMed]
- Morrill, A.W. Commercial entomology on the west coast of Mexico. J. Econ. Entomol. 1925, 18, 707–716. [Google Scholar] [CrossRef]
- Johnson, K.J.R. Acigona loftini (Lepidoptera: Pyralidae) in the Lower Rio Grande Valley of Texas, 1980–1981. In 2nd Inter-American Sugar Cane Seminar (Insect and Rodent Pests); Miami, FL, USA, 1981; pp. 166–171. [Google Scholar]
- Johnson, K.J.R.; van Leerdam, M.B. Range extension of Acigona loftini into the Lower Rio Grande Valley of Texas. Sugar Azucar 1981, 76, 119. [Google Scholar]
- Johnson, K.J.R. Identification of Eoreuma loftini (Dyar) (Lepidoptera: Pyralidae) in Texas, 1980: Forerunner for other sugarcane boring pest immigrants from Mexico? Bull. Entomol. Soc. Am. 1984, 30, 47–52. [Google Scholar] [CrossRef]
- Browning, H.W.; Way, M.O.; Drees, B.M. Managing the Mexican rice borer in Texas. B Texas Agric. Ext. Serv. Stn. 1989, B-1620. [Google Scholar]
- Reay-Jones, F.P.F.; Wilson, L.T.; Way, M.O.; Reagan, T.E.; Carlton, C.F. Movement of the Mexican rice borer (Lepidoptera: Crambidae) through Texas rice belt. J. Econ. Entomol. 2007, 100, 54–68. [Google Scholar] [CrossRef] [PubMed]
- Hummel, N.A.; Reagan, T.E.; Pollet, D.; Akbar, W.; Beuzelin, J.M.; Carlton, C.; Saichuk, J.; Hardy, T.; Way, M.O. Mexican Rice Borer, Eoreuma loftini (Dyar); Louisiana State University AgCenter Pub. 3098: Baton Rouge, LA, USA, 2008. [Google Scholar]
- Hummel, N.A.; Hardy, T.; Reagan, T.E.; Pollet, D.; Carlton, C.; Stout, M.J.; Beuzelin, J.M.; Akbar, W.; White, W.H. Monitoring and first discovery of the Mexican rice borer, Eoreuma loftini (Lepidoptera: Crambidae) in Louisiana. Fla. Entomol. 2010, 93, 123–124. [Google Scholar] [CrossRef]
- Hayden, J.E. Mexican Rice Borer, Eoreuma loftini (Dyar) (Lepidoptera: Crambidae: Crambinae) in Florida. Pest Alert, Florida Department of Agric. and Consumer Serv. DACS-P-01827. Available online: http://www.freshfrom florida.com/content/download/ 23854/eoreuma-loftini.pdf (accessed on 7 July 2015).
- University of Florida. Mexican Rice Borer: An Introduction. Available online: http://erec.ifas.ufl. edu/mexican_rice_borer_introduction.pdf (accessed on 30 June 2015).
- Showler, A.T.; Beuzelin, J.M.; Reagan, T.E. Alternate crop and weed host plant oviposition preferences by the Mexican rice borer (Lepidoptera: Crambidae). Crop Prot. 2011, 30, 895–901. [Google Scholar] [CrossRef]
- Showler, A.T.; Wilson, B.E.; Reagan, T.E. Mexican rice borer (Lepidoptera: Crambidae) injury to corn greater than to sorghum and sugarcane under field conditions. J. Econ. Entomol. 2012, 105, 1597–1602. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Showler, A.T.; Reagan, T.E. Mexican rice borer, Eoreuma loftini (Dyar) (Lepidoptera: Crambidae), biology, range expansion, ecology, and control in United States sugarcane. Am. Entomol. 2016. submitted. [Google Scholar]
- Showler, A.T.; Castro, B.A. Mexican rice borer (Lepidoptera: Crambidae) oviposition site selection stimuli on sugarcane, and potential field applications. J. Econ. Entomol. 2010, 103, 1180–1186. [Google Scholar] [CrossRef] [PubMed]
- Van Leerdam, M.B.; Johnson, K.J.R.; Smith, J.W., Jr. Ovipositional sites of Eoreuma loftini (Lepidoptera: Pyralidae) in sugarcane. Environ. Entomol. 1986, 15, 75–78. [Google Scholar] [CrossRef]
- Wilson, B.E.; Showler, A.T.; Reagan, T.E.; Beuzelin, J.M. Improved chemical control for the Mexican rice borer (Lepidoptera: Crambidae) in sugarcane: Larval exposure, a novel scouting method, and efficacy of a single aerial insecticide application. J. Econ. Entomol. 2012, 105, 1998–2006. [Google Scholar] [CrossRef] [PubMed]
- Ring, D.R.; Browning, H.W.; Johnson, K.J.R.; Smith, J.W., Jr.; Gates, C.E. Age-specific susceptibility of sugarcane internodes to attack by the Mexican rice borer (Lepidoptera: Pyralidae). J. Econ. Entomol. 1991, 84, 1001–1009. [Google Scholar] [CrossRef]
- Beuzelin, J.M.; Reagan, T.E.; Mészáros, A.; Wilson, L.T.; Way, M.O.; Blouin, D.; Showler, A.T. Seasonal infestations of two stem borers (Lepidoptera: Crambidae) in non-crop grasses of Gulf Coast rice agroecosystems. Environ. Entomol. 2011, 40, 1036–1050. [Google Scholar] [CrossRef] [PubMed]
- Van Leerdam, M.B.; Johnson, K.J.R.; Smith, J.W., Jr. Effects of substrate physical characteristics and orientation on oviposition by Eoreuma loftini (Lepidoptera: Pyralidae). Environ. Entomol. 1984, 13, 800–802. [Google Scholar] [CrossRef]
- Meagher, R.L., Jr.; Smith, J.W., Jr.; Johnson, K.J.R. Insecticidal management of Eoreuma loftini (Lepidoptera: Pyralidae) on Texas sugarcane: A critical review. J. Econ. Entomol. 1994, 87, 1332–1344. [Google Scholar] [CrossRef]
- Legaspi, J.C.; Legaspi, B.C., Jr.; Irvine, J.E.; Meagher, R.L., Jr.; Rozeff, N. Stalkborer damage on yield and quality of sugarcane in the Lower Rio Grande Valley of Texas. J. Econ. Entomol. 1999, 92, 228–234. [Google Scholar] [CrossRef]
- Meagher, R.L., Jr.; Wilson, L.T.; Pfannenstiel, R.S. Sampling Eoreuma loftini (Lepidoptera: Pyralidae) on Texas sugarcane. Environ. Entomol. 1996, 25, 7–16. [Google Scholar] [CrossRef]
- Wilson, B.E.; VanWeelden, M.T.; Beuzelin, J.M.; Reagan, T.E.; Way, M.O.; White, W.H.; Wilson, L.T.; Showler, A.T. A relative resistance ratio for evaluation of Mexican rice borer (Lepidoptera: Crambidae) susceptibility among sugarcane cultivars. J. Econ. Entomol. 2015, 108. [Google Scholar] [CrossRef] [PubMed]
- Reay-Jones, F.P.F.; Way, M.O.; Setamou, M.; Legendre, B.L.; Reagan, T.E. Resistance to the Mexican rice borer (Lepidoptera: Crambidae) among Louisiana and Texas sugarcane cultivars. J. Econ. Entomol. 2003, 96, 1929–1934. [Google Scholar] [CrossRef] [PubMed]
- Akbar, W.; Beuzelin, J.M.; Reagan, T.E.; Showler, A.T.; Way, M.O. Small plot assessment of insecticides against the Mexican rice borer, Raymondville, TX 2008. In Mexican Rice Borer and Sugarcane Borer Sugarcane and Rice Research Report; Louisiana State University AgCenter: Baton Rouge, LA, USA, 2009. [Google Scholar]
- Showler, A.T. Drought and arthropod pests of crops. In Droughts: New Research; Neves, D.F., Sanz, J.D., Eds.; Nova: Hauppauge, NY, USA, 2012; pp. 131–156. [Google Scholar]
- Mattson, W.L.; Haack, R.A. Role of drought in outbreaks of plant-eating insects. Bioscience 1987, 37, 110–118. [Google Scholar] [CrossRef]
- Reay-Jones, F.P.F.; Wilson, L.T.; Showler, A.T.; Reagan, T.E.; Way, M.O. Role of oviposition on preference in an invasive crambid impacting two graminaceous host crops. Environ. Entomol. 2007, 36, 938–951. [Google Scholar] [CrossRef] [PubMed]
- Showler, A.T.; Castro, B.A. Influence of drought stress on Mexican rice borer (Lepidoptera: Crambidae) oviposition preference and development to adulthood in sugarcane. Crop Prot. 2010, 29, 722–727. [Google Scholar] [CrossRef]
- Reay-Jones, F.P.F.; Showler, A.T.; Reagan, T.E.; Legendre, B.L.; Way, M.O.; Moser, E.B. Integrated tactics for managing the Mexican rice borer (Lepidoptera: Crambidae) in sugarcane. Environ. Entomol. 2005, 34, 1558–1565. [Google Scholar] [CrossRef]
- Jordan, W.R.; Ritchie, J.T. Influence of soil water stress on evaporation, root absorption, and internal water status of cotton. Plant Physiol. 1971, 48, 783–788. [Google Scholar] [CrossRef] [PubMed]
- Janagouar, B.S.; Venkatasubbaiah, D.; Janardhan, K.V.; Panchal, Y.C. Effect of short term stress on free proline accumulation, relative water content and potassium content in different plant parts of three cotton genotypes. Ind. J. Plant Physiol. 1983, 26, 82–87. [Google Scholar]
- Ingram, J.; Bartels, D. The molecular basis of dehydration tolerance in plants. Annu. Rev. Plant Biol. 1996, 47, 377–403. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.-K. Salt and drought stress signal transduction in plants. Annu. Rev. Plant Biol. 2002, 53, 247–273. [Google Scholar] [CrossRef] [PubMed]
- Seki, M.; Narusaka, M.; Abe, H.; Ksuga, K.; Yamaguchi-Shinozaki, K.; Carninci, P.; Hayashizaki, P.; Shinozaki, K. Monitoring the expression pattern of 1300 Arabidopsis genes under drought and cold stress by using a full-length cDNA microarray. Plant Cell 2001, 13, 61–72. [Google Scholar] [CrossRef] [PubMed]
- Showler, A.T.; Cavazos, C.O.; Moran, P.J. Dynamics of free amino acid accumulations in cotton leaves measured on different timelines after irrigation. Subtrop. Plant Sci. 2007, 59, 38–55. [Google Scholar]
- Labanauskas, C.K.; Stolzy, L.H.; Handy, M.F. Protein and free amino acids in field-grown cowpea seeds as affected by water stress at various growth stages. Plant Soil 1981, 63, 355–368. [Google Scholar] [CrossRef]
- Golan-Goldhirsch, A.N.; Samish, S.; Agami, M.; Lips, H. The relationship between some perennial desert plants originated in different phytogeographical regions and proline concentration. J. Arid Environ. 1989, 17, 327–333. [Google Scholar]
- Bussis, D.; Heineke, D. Acclimation of potato plants to polyethylene glycol-induced water deficit II. Contents and subcellular distribution of organic solutes. J. Exp. Bot. 1998, 49, 1361–1370. [Google Scholar] [CrossRef]
- Becana, M.; Moran, J.F.; Iturbe-Ormaetxe, I. Iron-dependent oxygen free radical generation in plants subjected to environmental stress: Toxicity and antioxidant protection. Plant Soil 1998, 201, 137–147. [Google Scholar] [CrossRef]
- Knight, H.; Knight, M.R. Abiotic stress signaling pathways: Specificity and cross-talk. Trends Plant Sci. 2001, 6, 262–267. [Google Scholar] [CrossRef]
- Jones, C.G. Plant stress and insect herbivory: Toward an integrated perspective. In Responses of Plants to Multiple Stresses; Mooney, H.A., Winner, W.E., Pell, E.J., Eds.; Academic Press: New York, NY, USA, 1991; pp. 249–280. [Google Scholar]
- Schur, K.; Holdaway, F.G. Olfactory responses of female Ostrinia nubilalis (Lepidoptera: Pyraustinae). Entomol. Exp. Appl. 1970, 13, 455–461. [Google Scholar] [CrossRef]
- Fenemore, P.G. Oviposition of potato tuber moth, Phthorimaea operculella Zell. (Lepidoptera: Gelechiidae); identification of host-plant factors influencing oviposition responses. N. Z. J. Zool. 1980, 7, 435–439. [Google Scholar] [CrossRef]
- Waladde, S.M. Chemoreceptors of adult stem borers: Tarsal and ovipositor sensilla on Chilo partellus and Eldana saccharina. Int. J. Trop. Insect Sci. 1983, 4, 159–165. [Google Scholar] [CrossRef]
- Burton, R.L.; Schuster, D.J. Oviposition stimulant for tomato pinworms from surfaces of tomato plants. J. Econ. Enomol. 1981, 74, 512–515. [Google Scholar] [CrossRef]
- Ramaswamy, S.B. Host finding by moths: Sensory modalities and behaviors. J. Insect Physiol. 1988, 34, 235–249. [Google Scholar] [CrossRef]
- Salama, H.S.; Rizk, A.F.; Sharaby, A. Chemical stimuli in flowers and leaves of cotton that affect behavior in the cotton moth, Spodoptera littoralis (Lepidoptera: Noctuidae). Entomologia 1984, 10, 27–34. [Google Scholar]
- Udayagiri, S.; Mason, C.E. Host plant constituents as oviposition stimulants for a generalist herbivore: European corn borer. Entomol. Exp. Appl. 1995, 76, 59–65. [Google Scholar] [CrossRef]
- Showler, A.T. Water deficit stress, host plant nutrient accumulations and associations with phytophagous arthropods. In Abiotic Stress; Vahdata, K., Ed.; InTech: Rijeka, Croatia, 2013; pp. 387–410. [Google Scholar]
- Reagan, T.E.; Way, M.O.; Beuzelin, J.M.; Akbar, W. Assessment of Varietal Resistance to the Sugarcane Borer and Mexican Rice Borer; Sugarcane Research: Annual Progress Report; Louisiana State University AgCenter, Louisiana State University: Baton Rouge, LA, USA, 2008. [Google Scholar]
- Showler, A.T.; Moran, P.J. Associations between host plant concentrations of selected biochemical nutrients and Mexican rice borer, Eoreuma loftini (Dyar) (Lepidoptera: Crambidae), infestation. Entomol. Exp. Appl. 2014, 151, 135–143. [Google Scholar] [CrossRef]
- Showler, A.T. Effects of compost and chicken litter on soil nutrition, and sugarcane physiochemistry, yield, and injury caused by Mexican rice borer, Eoreuma loftini (Dyar) (Lepidoptera: Crambidae). Crop Prot. 2015, 71, 1–11. [Google Scholar] [CrossRef]
- Showler, A.T. Beneficial and detrimental interactions between weeds and other pests of sugarcane. In Weeds and Their Ecological Functions; Taub, A., Ed.; Nova: Hauppauge, NY, USA, 2013; pp. 153–188. [Google Scholar]
- Showler, A.T. Plant-arthropod interactions affected by water deficit stress through association with changes in plant free amino acid accumulations. In Molecular Approaches to Plant Abiotic Stress; Gaur, A.K., Ed.; CABI: Silwood Park, UK, 2013; pp. 339–352. [Google Scholar]
- Atkinson, P.R. On the biology, distribution and natural host plants of Eldana saccharina Walker. J. Entomol. Soc. S. Afr. 1980, 43, 171–194. [Google Scholar]
- Cochereau, P. Observations on the borer Eldana saccharina Walker (Lep., Pyralidae) in maize and sugarcane in Ivory Coast. Proc. S. Afr. Sugarcane Technol. Assoc. 1982, 49, 82–84. [Google Scholar]
- Kaufmann, T. Behavioral biology, feeding habits, and ecology of three species of maize stem-borers: Eldana saccharina (Lepidoptera: Pyralidae), Sesamia calamistis and Busseola fusca (Noctuidae) in Ibadan, Nigeria, West Africa. J. Ga. Entomol. Soc. 1983, 18, 255–259. [Google Scholar]
- Moyal, P. Borer infestation and damage in relation to maize stand density and water stress in the Ivory Coast. Int. J. Pest Manag. 1995, 41, 114–121. [Google Scholar] [CrossRef]
- Atkinson, P.R.; Nuss, K.J. Associations between host-plant nitrogen and infestations of the sugarcane borer, Eldana saccharina Walker (Lepidoptera: Pyralidae). Bull. Entomol. Res. 1989, 79, 489–506. [Google Scholar] [CrossRef]
- Mazodze, R.; Nyanthete, C.; Chidoma, S. First outbreak of Eldana saccharina (Lepidoptera: Pyralidae) in sugarcane in the south-east lowland of Zimbabwe. Proc. S. Afr. Sugarcane Technol. Assoc. 1999, 73, 107–111. [Google Scholar]
- Mazodze, R.; Conlong, D.E. Eldana saccharina (Lepidoptera: Pyralidae) in sugarcane (Saccharum hybrids), sedge (Cyperus digitatus) and bulrush (Typha latifolia) in south-eastern Zimbabwe. Proc. S. Afr. Sugarcane Technol. Assoc. 2003, 77, 266–274. [Google Scholar]
- Carnegie, A.J.M.; Leslie, G.W. Eldana saccharina (Lepidoptera: Pyralidae): Ten years of light trapping. Proc. S. Afr. Sugarcane Technol. Assoc. 1990, 57, 107–110. [Google Scholar]
- Carnegie, A.J.M. Combating Eldana saccharina Walker, a progress report. Proc. S. Afr. Sugarcane Technol. Assoc. 1981, 48, 107–110. [Google Scholar]
- Keeping, M.G.; Meyer, J.H. Calcium silicate enhances resistance of sugarcane to the African stalk borer Eldana saccharina Walker (Lepidoptera: Pyralidae). Agric. For. Entomol. 2002, 4, 265–274. [Google Scholar] [CrossRef]
- Bryant, J.P.; Clausen, T.P.; Reichardt, P.B.; McCarthy, M.C.; Werner, R.A. Effect of nitrogen fertilization upon the secondary chemistry and nutritional value of quaking aspen (Populus tremuloides Michx.) leaves for the large aspen tortrix (Choristoneura conflicana (Walker)). Oecologia 1987, 73, 513–517. [Google Scholar] [CrossRef]
- Chau, L.M.; Heong, K.L. Effects of organic fertilizers on insect pest and diseases of rice. Omonrice 2005, 13, 26–33. [Google Scholar]
- Ahmed, S.H.; Saba, S.; Ali, C.M. Effect of different doses of nitrogen fertilizer on sucking insect pests of cotton, Gossypium hirsutum. J. Agric. Res. 2007, 45, 43–48. [Google Scholar]
- Ramzan, M.; Hussain, S.; Akhter, M. Incidence of insect pests on rice crop under various nitrogen doses. J. Anim. Plant Sci. 2007, 17, 67–69. [Google Scholar]
- Bottrell, D.G.; Schoenly, K.G. Resurrecting the ghost of green revolutions past: The brown planthopper as a recurring threat to high-yielding rice production in tropical Asia. J. Asia-Pac. Entomol. 2012, 15, 122–140. [Google Scholar] [CrossRef]
- SASA (South African Sugarcane Association). Eldana Borer Control Measures; Information Sheet: SASA Mount Edgecombe, Natal, Republic of South Africa, 1994. [Google Scholar]
- McWhorter, C.G. Factors affecting johnsongrass rhizome production and germination. Weed Sci. 1972, 20, 41–45. [Google Scholar]
- Ali, A.D.; Reagan, T.E.; Flynn, J.L. Effects of johnsongrass (Sorghum halepense) density on sugarcane (Saccharum officinarum) yield. Weed Sci. 1986, 14, 381–383. [Google Scholar]
- Ogunwolu, E.O.; Reagan, T.E.; Damman, K.E. Efficacy of pest control strategies in Louisiana sugar cane: A preliminary survey. J. Agric. Sci. 1987, 108, 661–665. [Google Scholar] [CrossRef]
- Arevelo, R.A.; Cerrizuela, E.A.; Olea, I.L. Competition from specific weeds in sugarcane II. Sorghum halepense (L.) Pers. Revu. Agron. Noroeste Argent 1977, 14, 39–51. [Google Scholar]
- Peng, S.Y. The Biology and Control of Weeds in Sugarcane; Elsevier: Amsterdam, The Netherlands, 1984. [Google Scholar]
- Ali, A.D.; Reagan, T.E. Vegetation manipulation impact on predator and prey populations in Louisiana sugarcane ecosystems. J. Econ. Entomol. 1985, 78, 1409–1414. [Google Scholar] [CrossRef]
- Showler, A.T. Effects of water deficit stress, shade, weed competition, and kaolin particle film on selected foliar free amino acid accumulations in cotton, Gossypium hirsutum L. J. Chem. Ecol. 2002, 28, 631–651. [Google Scholar] [CrossRef] [PubMed]
- Showler, A.T.; Reagan, T.E.; Shao, K.P. Nematode interactions with weeds and sugarcane mosaic virus in Louisiana sugarcane. J. Nematol. 1990, 22, 31–38. [Google Scholar] [PubMed]
- Showler, A.T.; Knaus, R.M.; Reagan, T.E. Studies of the territorial dynamics of the red imported fire ant (Hymenoptera: Formicidae). Agric. Ecosyst. Environ. 1991, 30, 97–105. [Google Scholar] [CrossRef]
- Ali, A.D.; Reagan, T.E.; Flynn, J.L. Influence of selected weedy and weed-free sugarcane habitats on diet composition and foraging activity of the imported fire ant (Hymenopera: Formicidae). Environ. Entomol. 1984, 13, 1037–1041. [Google Scholar] [CrossRef]
- Showler, A.T.; Knaus, R.M.; Reagan, T.E. Foraging territoriality of the imported fire ant, Solenopsis invicta Buren, in sugarcane as determined by neutron activation analysis. Insectes Soc. 1989, 36, 235–239. [Google Scholar] [CrossRef]
- Showler, A.T.; Reagan, T.E. Ecological interactions of the red imported fire ant in the southeastern United States. J. Entomol. Sci. 1987, 1, 52–64. [Google Scholar]
- Negm, A.A.; Hensley, S.D. Evaluation of certain biological control agents of the sugarcane borer in Louisiana. J. Econ. Entomol 1969, 62, 1003–1013. [Google Scholar] [CrossRef]
- Ali, A.D.; Reagan, T.E. Spider inhabitants of sugarcane ecosystems in Louisiana: An update. Proc. La. Acad. Sci. 1985, 48, 18–22. [Google Scholar]
- Hatley, C.L.; McMahon, J.A. Spider community organization: Seasonal variation and the role of vegetation architecture. Environ. Entomol. 1980, 9, 632–639. [Google Scholar] [CrossRef]
- Riechert, S.E.; Lockley, T. Spiders as biocontrol agents. Annu. Rev. Entomol. 1984, 29, 299–320. [Google Scholar] [CrossRef]
- Ali, A.D.; Reagan, T.E. Influence of selected weed control practices on araneid faunal composition and abundance in sugarcane. Environ. Entomol. 1896, 15, 527–531. [Google Scholar] [CrossRef]
- Topham, M.; Beardsley, J.W. Influence of nectar source plants on the New Guinea weevil parasite, Lixophaga sphenophori (Villeneuve). Proc. Hawaii Entomol. Soc. 1975, 22, 145–154. [Google Scholar]
- VanWeelden, M.T.; Beuzelin, J.M.; Wilson, B.E.; Reagan, T.E.; Way, M.O. Impact of red imported fire ant on Mexican rice borer in sugarcane and non-crop hosts. Import. Fire Ant Conf. Proc. 2012, 17–19. [Google Scholar]
- Showler, A.T.; Greenberg, S.M. Effects of weeds on selected arthropod herbivore and natural enemy populations, and on cotton growth and yield. Environ. Entomol. 2003, 32, 39–50. [Google Scholar] [CrossRef]
- Lara, L.L.; Artaxo, P.; Martinelli, L.A.; Camargo, P.B.; Victoria, R.L.; Ferraz, E.S.B. Properties of aerosols from sugar-cane burning emissions in southeastern Brazil. Atmos. Environ. 2005, 39, 4627–4637. [Google Scholar] [CrossRef]
- Cançado, J.E.D.; Saldiva, P.H.N.; Pereira, L.A.A.; Lara, L.B.L.S.; Artaxo, P.; Martinelli, L.A.; Arbex, M.A.; Zanobetti, A.; Braga, A.L.F. The impact of sugar-cane burning emissions on the respiratory system of children and the elderly. Environ. Health Perspect. 2006, 114, 725–729. [Google Scholar] [CrossRef] [PubMed]
- Arbex, M.A.; Martins, L.C.; de Oliveira, R.C.; Pereira, L.A.A.; Arbex, F.A.; Cançado, J.E.D.; Saldiva, P.H.N.; Braga, A.L.F. Air pollution from biomass burning and asthma hospital admissions in a sugar cane plantation area in Brazil. J. Epidemiol. Community Health 2007, 61, 395–400. [Google Scholar] [CrossRef] [PubMed]
- Juo, A.S.R.; Lal, R. The effect of fallow and continuous cultivation on the chemical and physical properties of an Alfisol in Western Nigeria. Plant Soil 1997, 47, 567–584. [Google Scholar] [CrossRef]
- Biederbeck, V.O.; Campbell, C.A.; Bowren, K.E.; Schnitzer, M.; McIver, R.N. Effect of burning cereal straw on soil properties and grain yields in Saskatchewan. Soil Sci. Soc. Am. J. 1980, 44, 103–111. [Google Scholar] [CrossRef]
- Wood, A.W. Movement of crop residues following green harvesting of sugar cane in northern Queensland. Soil Tillage Res. 1991, 20, 69–85. [Google Scholar] [CrossRef]
- Basanta, M.V.; Dourado-Neto, D.; Reichardt, K.; Bacchi, O.O.S.; Oliveira, J.C.M.; Trivelin, P.C.O.; Timm, L.C.; Tominaga, T.T.; Correchel, V.; Cassaro, F.M.; et al. Management effects on nitrogen recovery in a sugarcane crop grown in Brazil. Geoderma 2003, 1998, 1–14. [Google Scholar] [CrossRef]
- Cumberbatch, E.R. Soil properties and the effect of organic mulches on reclamation of Scotland clays, Barbados. Trop. Agric. 1969, 46, 17–24. [Google Scholar]
- Eavis, B.W.; Cumberbatch, E.R.S.J. Sugar cane growth in response to mulch and fertilizer on saline-alkali subsoils. Agron. J. 1977, 69, 839–842. [Google Scholar] [CrossRef]
- Sandhu, B.S.; Prihar, S.S.; Khera, H.L. Sugarcane response to irrigation and straw mulch in a subtropical region. Agric. Water Manag. 1980, 3, 35–44. [Google Scholar] [CrossRef]
- Yadav, R.L.; Prasad, S.R.; Singh, S.R.K. Effect of potassium and trash mulch on yield and quality of sugarcane under limited water supply. J. Potassium Res. 1986, 2, 136–139. [Google Scholar]
- Ball-Coelho, B.R.; Tiessen, H.; Stewart, J.W.B.; Salcedo, I.H.; Sampiao, E.V.S.B. Residue management effects on sugarcane yield and soil properties in northwestern Brazil. Agron. J. 1993, 85, 1004–1008. [Google Scholar] [CrossRef]
- Lima, M.A. Nitrogen Nutrition of Sugar Cane in NE Brazil. Ph.D. Thesis, Department of Soil Science, University of Saskatchewan, Saskatoon, SK, Canada, 1982. [Google Scholar]
- White, R.E.; Ayoub, A.T. Composition of plant residues of variable C/P ratio and the effect on soil phosphate availability. Plant Soil 1983, 74, 163–173. [Google Scholar] [CrossRef]
- Kwong, K.F.N.K.; Deville, J.; Cavalot, P.C.; Rivière, V. Value of cane trash in nitrogen nutrition of sugarcane. Plant Soil 1987, 102, 79–83. [Google Scholar] [CrossRef]
- Yadav, D.V.; Singh, T.; Srivastava, A.K. Recycling of nutrients in trash with N for higher cane yield. Biol. Wastes 1987, 20, 133–141. [Google Scholar] [CrossRef]
- Monzon, F.A. Queima da cana. Solo 1956, 48, 53–58. [Google Scholar]
- Veiga, F.M.; Dobereinker, J.; Pinto, R.S.; Gondim, G.S. Influencia do tratamento do palchiço da cana-de-açúcar da matéria orgânica do solo. Divulg. Agron. 1962, 10–13. [Google Scholar]
- Showler, A.T. unpublished data.
- Showler, A.T.; Cook, S.C.; Abrigo, V. Transgenic Bt corn varietal resistance against the Mexican rice borer, Eoreuma loftini (Dyar) (Lepidoptera: Crambidae) and implications to sugarcane. Crop Prot. 2013, 48, 57–62. [Google Scholar] [CrossRef]
- Gould, F. Bt-resistance management—Theory meets data. Nat. Biotechnol. 2003, 21, 1450–1451. [Google Scholar] [CrossRef] [PubMed]
© 2016 by the author; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Showler, A.T. Selected Abiotic and Biotic Environmental Stress Factors Affecting Two Economically Important Sugarcane Stalk Boring Pests in the United States. Agronomy 2016, 6, 10. https://doi.org/10.3390/agronomy6010010
Showler AT. Selected Abiotic and Biotic Environmental Stress Factors Affecting Two Economically Important Sugarcane Stalk Boring Pests in the United States. Agronomy. 2016; 6(1):10. https://doi.org/10.3390/agronomy6010010
Chicago/Turabian StyleShowler, Allan T. 2016. "Selected Abiotic and Biotic Environmental Stress Factors Affecting Two Economically Important Sugarcane Stalk Boring Pests in the United States" Agronomy 6, no. 1: 10. https://doi.org/10.3390/agronomy6010010
APA StyleShowler, A. T. (2016). Selected Abiotic and Biotic Environmental Stress Factors Affecting Two Economically Important Sugarcane Stalk Boring Pests in the United States. Agronomy, 6(1), 10. https://doi.org/10.3390/agronomy6010010





