Reducing the Sodium Adsorption Ratio Promotes Cotton Growth and Development by Enhancing Antioxidant Enzyme Activities and the Plant’s Potassium–Sodium Ratio Under Brackish-Water Irrigation
Abstract
1. Introduction
2. Materials and Methods
2.1. Overview of Experimental Area
2.2. Experimental Design
2.3. Observation Indicators and Methods
2.3.1. Relative Water Content and Electrical Conductivity of Soil
2.3.2. Photosynthetic Indexes
- (1)
- Net photosynthetic rate: A portable photosynthesizer (LI-6400, LICOR, Lincoln, NE, USA) was used to measure the net photosynthetic rate at 13:00–14:00 a.m. during the bolling stage, and the fully expanded leaf at the top of the plant was selected for each treatment.
- (2)
- Chlorophyll SPAD value: After each measurement of photosynthetic parameters, the SPAD value was determined on the same leaf using a SPAD meter (Konica Minolta SPAD-502 chlorophyll meter, Chiyoda, Tokyo, Japan).
2.3.3. Antioxidant Enzyme Activities of Leaf
2.3.4. Measurement of Cotton Dry Matter Quality
2.3.5. Determination of Nutrient Ions in Cotton Plants
2.4. Statistical Analysis
3. Results
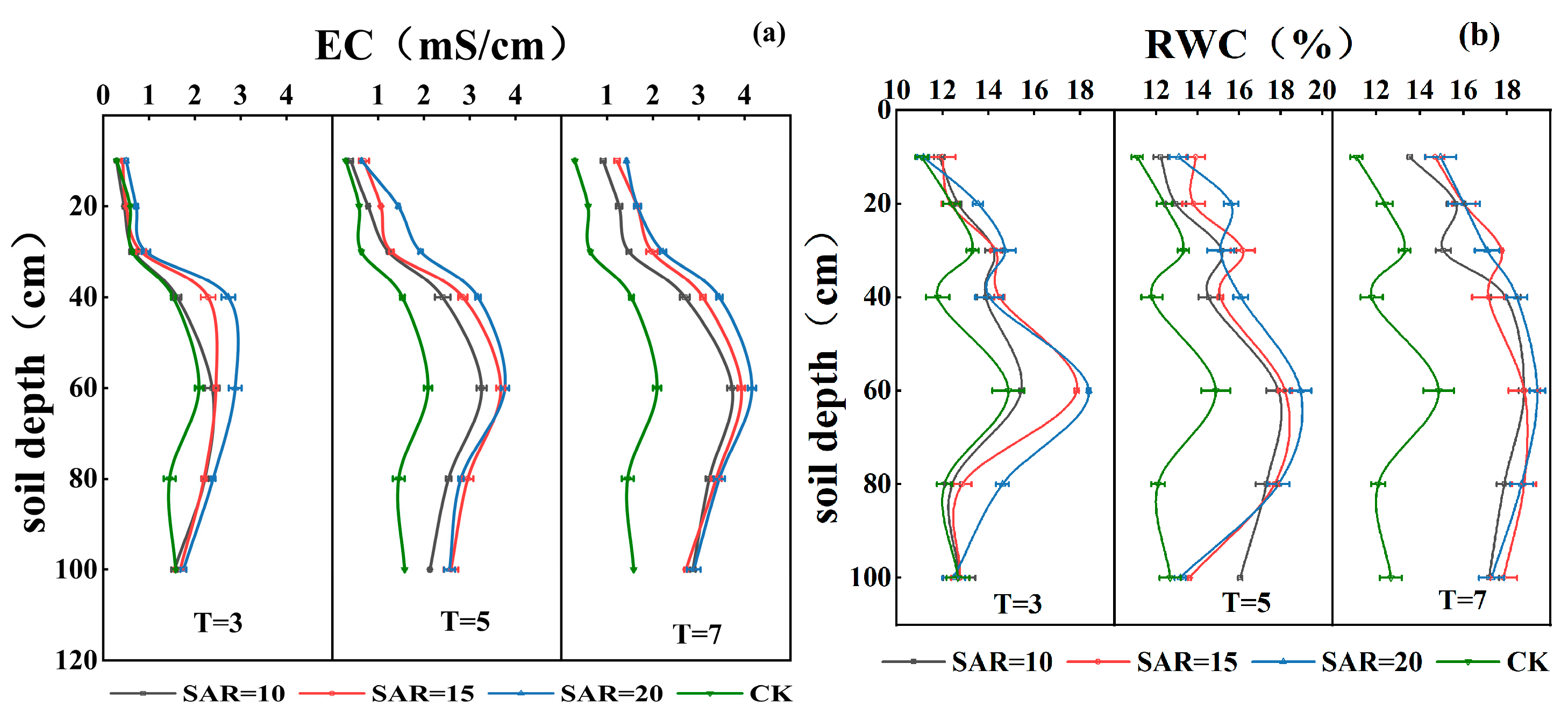
3.1. Effects of Irrigation Water Salinity and SAR on Soil Water Content and Electrical Conductivity
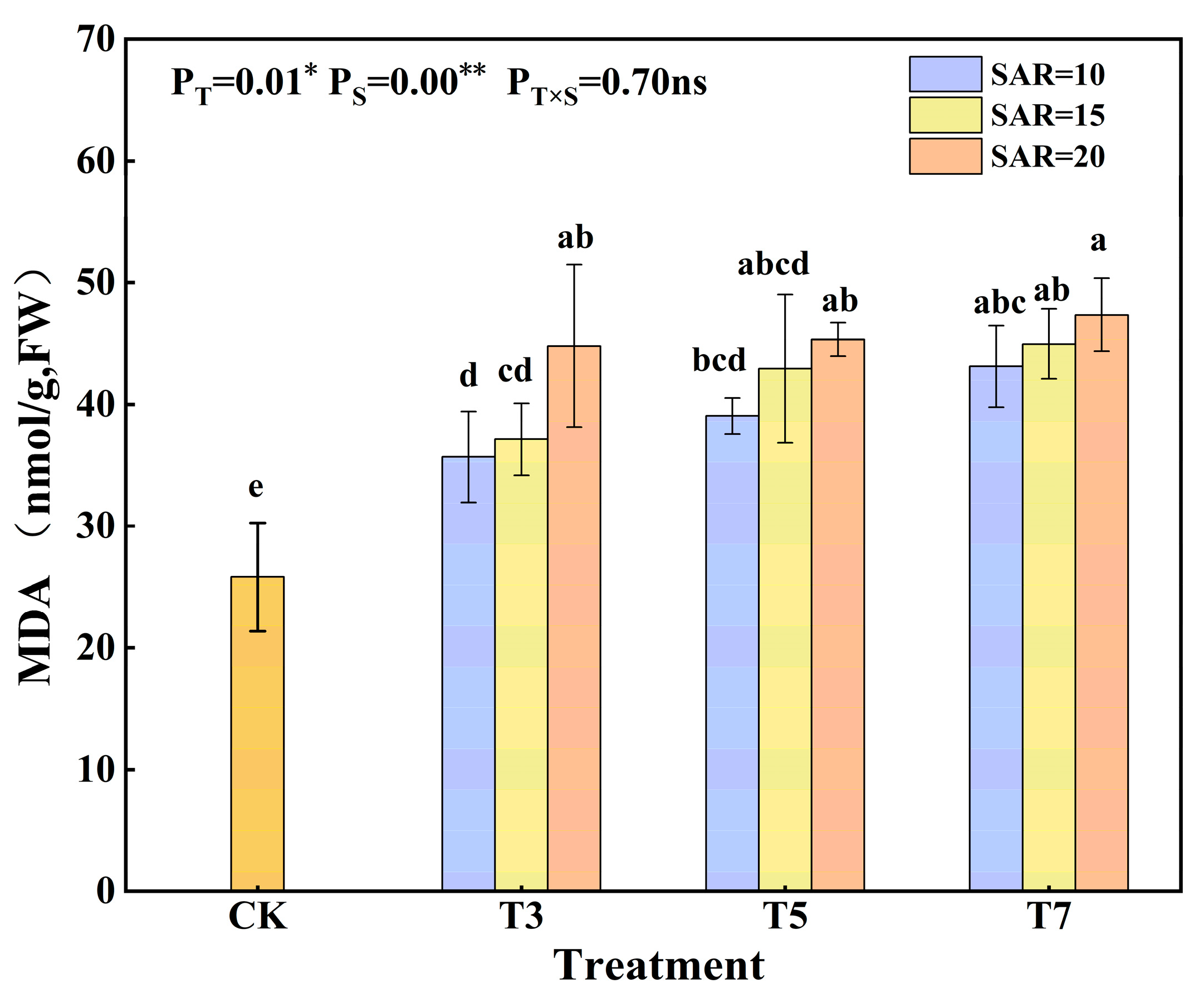
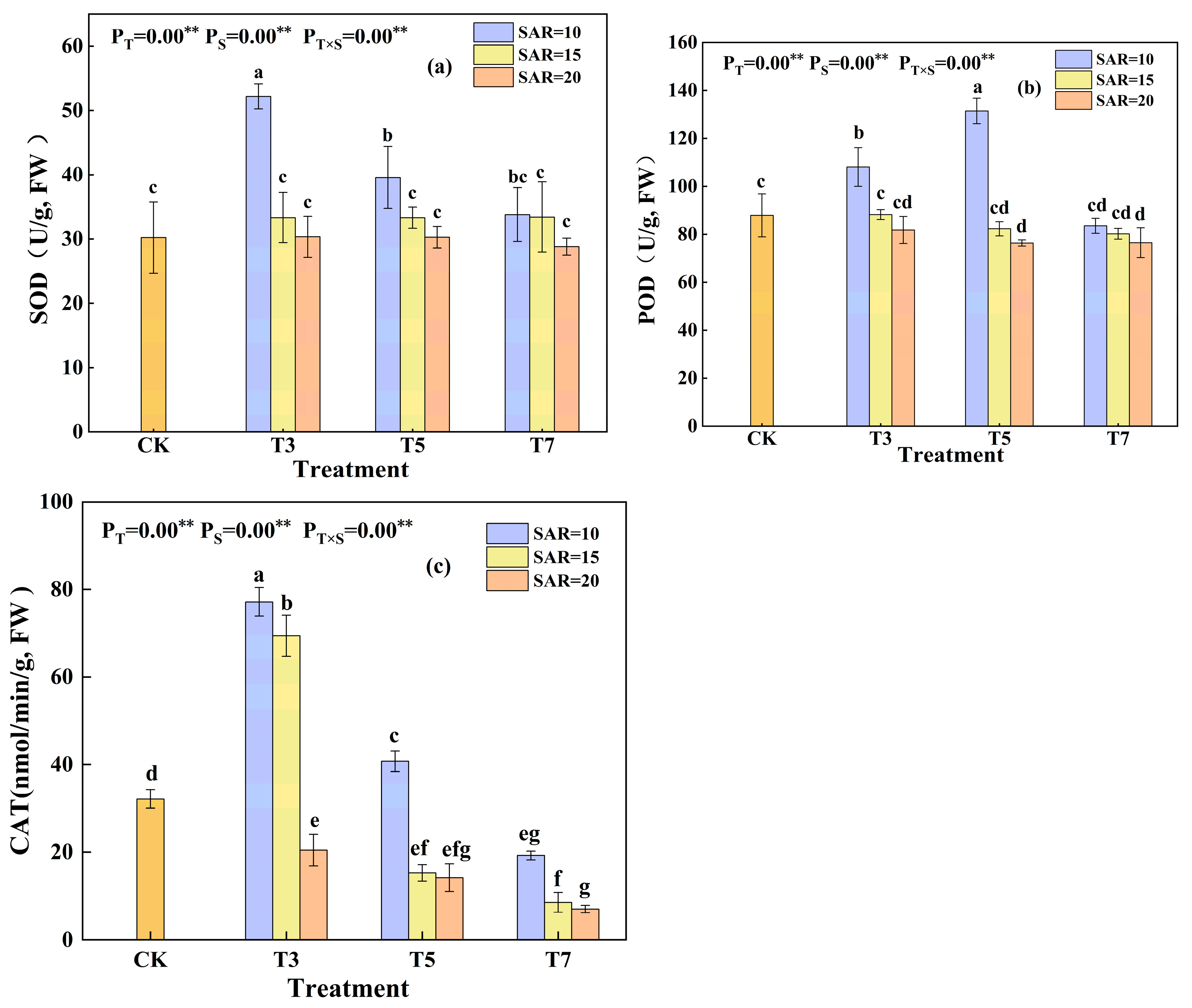
3.2. Effects of Irrigation Water Salinity and SAR on MDA Content and Antioxidant Activity in Cotton Leaves
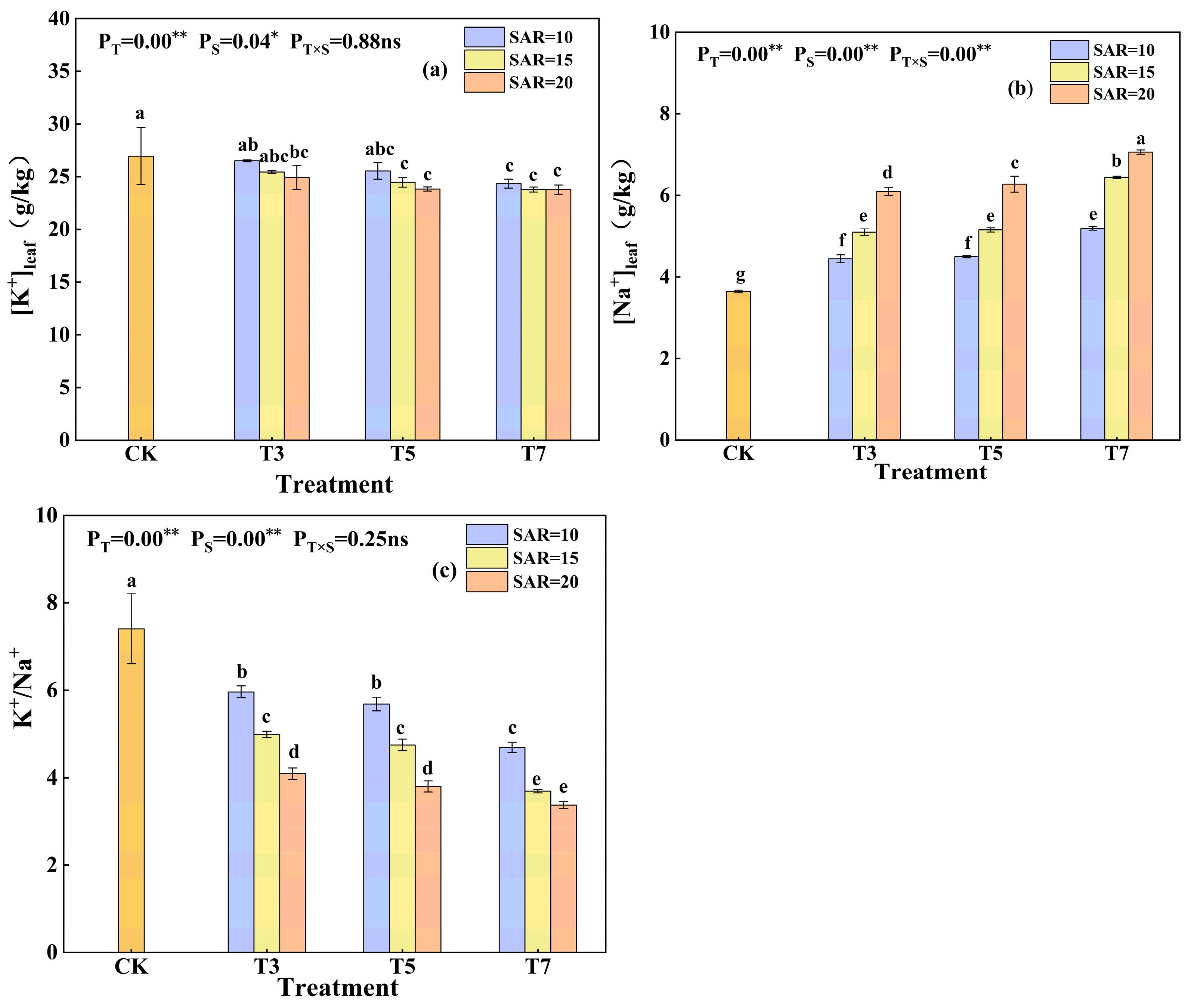
3.3. Effects of Irrigation Water Salinity and SAR on K+ and Na+ Uptake in Cotton Leaves
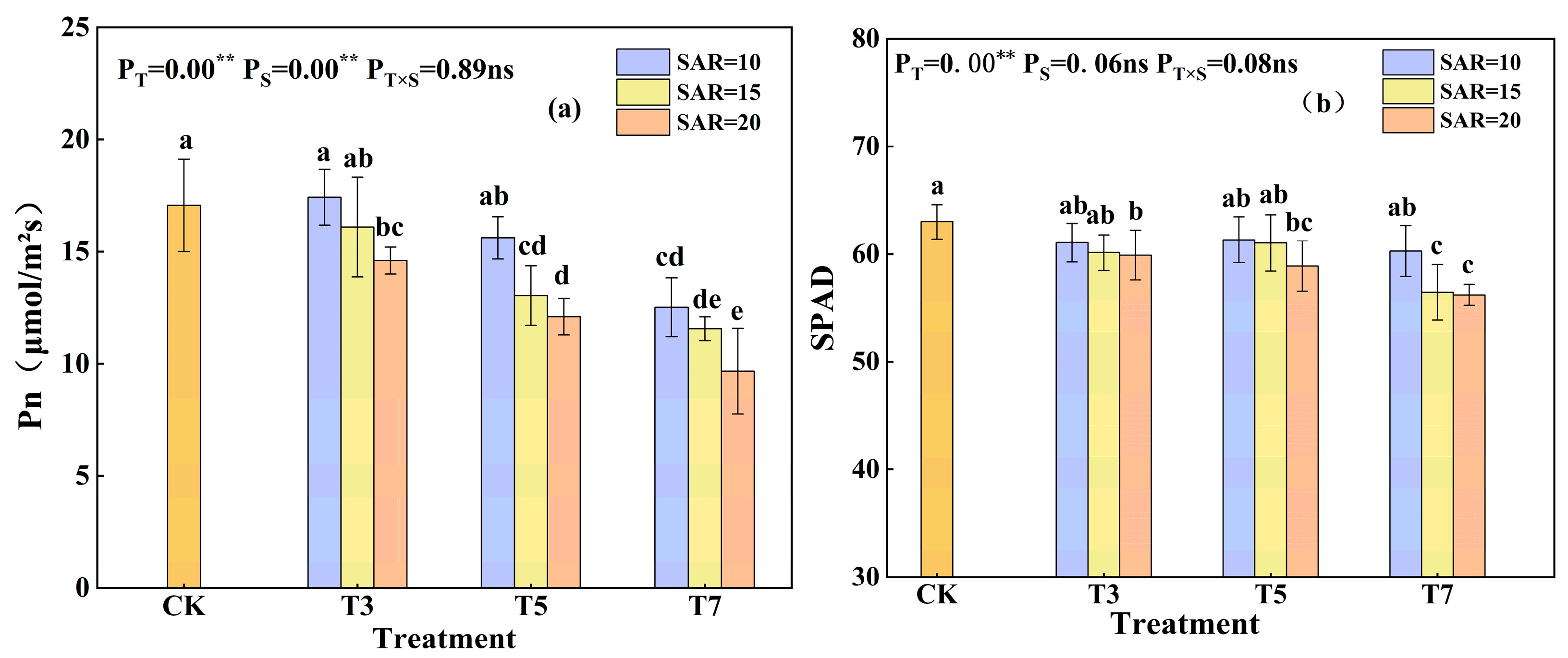
3.4. Effects of Irrigation Water Salinity and SAR on Net Photosynthetic Rate and SPAD Value in Cotton
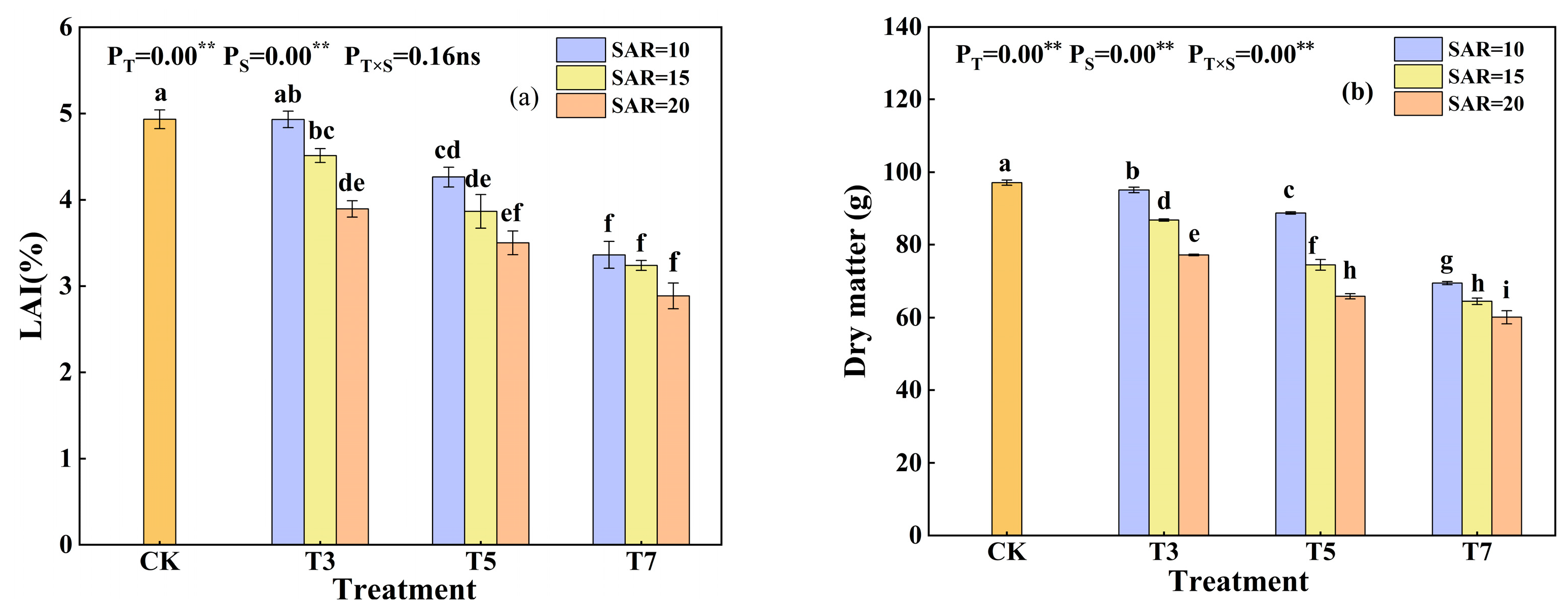
3.5. Effects of Irrigation Water Salinity and SAR on Cotton Growth
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Chen, W.; Hou, Z.; Wu, L.; Liang, Y.; Wei, C. Evaluating salinity distribution in soil irrigated with saline water in arid regions of northwest China. Agric. Water Manag. 2010, 97, 2001–2008. [Google Scholar] [CrossRef]
- Kang, S.; Hao, X.; Du, T.; Tong, L.; Su, X.; Lu, H.; Li, X.; Huo, Z.; Li, S.; Ding, R. Improving agricultural water productivity to ensure food security in China under changing environment: From research to practice. Agric. Water Manag. 2017, 179, 5–17. [Google Scholar] [CrossRef]
- Chen, W.; Jin, M.; Ferré, T.P.A.; Liu, Y.; Xian, Y.; Shan, T.; Ping, X. Spatial distribution of soil moisture, soil salinity, and root density beneath a cotton field under mulched drip irrigation with brackish and fresh water. Field Crops Res. 2018, 215, 207–221. [Google Scholar] [CrossRef]
- Guo, H.; Hu, Z.; Zhang, H.; Hou, Z.; Min, W. Soil Microbial Metabolic Activity and Community Structure in Drip-Irrigated Calcareous Soil as Affected by Irrigation Water Salinity. Water Air Soil Pollut. 2019, 230, 44. [Google Scholar] [CrossRef]
- Li, J.; Gao, Y.; Zhang, X.; Tian, P.; Li, J.; Tian, Y. Comprehensive comparison of different saline water irrigation strategies for tomato production: Soil properties, plant growth, fruit yield and fruit quality. Agric. Water Manag. 2019, 213, 521–533. [Google Scholar] [CrossRef]
- Ma, Z.; Liu, J.; Wen, Y.; Zhang, J.; Yin, F.; Guo, L.; Li, W.; He, J.; Ma, J.; Liang, Y.; et al. Optimizing cotton yield through appropriate irrigation water salinity: Coordinating above- and below-ground growth and enhancing photosynthetic capacity. Eur. J. Agron. 2024, 154, 127095. [Google Scholar] [CrossRef]
- Chen, W.; Jin, M.; Ferré, T.P.A.; Liu, Y.; Huang, J.; Xian, Y. Soil conditions affect cotton root distribution and cotton yield under mulched drip irrigation. Field Crops Res. 2020, 249, 107743. [Google Scholar] [CrossRef]
- Baath, G.S.; Shukla, M.K.; Bosland, P.W.; Steiner, R.L.; Walker, S.J. Irrigation water salinity influences at various growth stages of Capsicum annuum. Agric. Water Manag. 2017, 179, 246–253. [Google Scholar] [CrossRef]
- Alharby, H.F.; Colmer, T.D.; Barrett-Lennard, E.G. Salinization of the soil solution decreases the further accumulation of salt in the root zone of the halophyte Atriplex nummularia Lindl. growing above shallow saline groundwater. Plant Cell Environ. 2017, 41, 99–110. [Google Scholar] [CrossRef]
- Li, W.; Gao, Y.; Tian, Y.; Li, J. Double-root-grafting enhances irrigation water efficiency and reduces the adverse effects of saline water on tomato yields under alternate partial root-zone irrigation. Agric. Water Manag. 2022, 264, 107488. [Google Scholar] [CrossRef]
- Liu, S.; Dong, Y.; Xu, L.; Kong, J. Effects of foliar applications of nitric oxide and salicylic acid on salt-induced changes in photosynthesis and antioxidative metabolism of cotton seedlings. Plant Growth Regul. 2013, 73, 67–78. [Google Scholar] [CrossRef]
- Anjum, S.A.; Ashraf, U.; Zohaib, A.; Tanveer, M.; Naeem, M.; Ali, I.; Tabassum, T.; Nazir, U. Growth and developmental responses of crop plants under drought stress: A review. Zemdirb.-Agric. 2017, 104, 267–276. [Google Scholar] [CrossRef]
- Li KeJiang, L.K.; Ma JunYong, M.J.; Cao CaiYun, C.C.; Zheng ChunLian, Z.C.; Zhang CangGen, Z.C.; Niu YingJie, N.Y. Effect of irrigation water salt content on cotton growth and yield. Zhongguo Shengtai Nongye Xuebao/Chin. J. Eco-Agric. 2011, 19, 312–317. [Google Scholar] [CrossRef]
- Jiao, Y.; Pan, Z.; Zhang, Y.; Niu, L.; Zheng, C. Effect of Saline Water Irrigation on Soil Salt and Cotton in Hebei Low Plain. J. Irrig. Drain. 2012, 31, 31–34, 39. [Google Scholar]
- Wang, C.X.; Wang, Q.J.; Liu, J.J.; Su, L.J.; Shan, Y.Y.; Zhuang, L. Effects of mineralization of irrigation water and soil salinity on cotton emergence rate in Southern Xinjiang Uygur Autonomous Region of China. Trans. Chin. Soc. Agric. Eng. 2010, 26, 28–33. [Google Scholar]
- Ma, Z.; Zhu, Y.; Liu, J.; Li, Y.; Zhang, J.; Wen, Y.; Song, L.; Liang, Y.; Wang, Z. Multi-objective optimization of saline water irrigation in arid oasis regions: Integrating water-saving, salinity control, yield enhancement, and CO2 emission reduction for sustainable cotton production. Sci. Total Environ. 2023, 912, 169672. [Google Scholar] [CrossRef]
- Yang, G.; Li, F.; Tian, L.; He, X.; Gao, Y.; Wang, Z.; Ren, F. Soil physicochemical properties and cotton (Gossypium hirsutum L.) yield under brackish water mulched drip irrigation. Soil Tillage Res. 2020, 199, 104592. [Google Scholar] [CrossRef]
- Zhang, A.; Zheng, C.; Li, K.; Dang, H.; Cao, C.; Rahma, A.E.; Zhang, J.; Feng, D. Responses of soil water-salt variation and cotton growth to drip irrigation with saline water in the low plain near the bohai sea. Irrig. Drain. 2020, 69, 448–459. [Google Scholar] [CrossRef]
- Li, J.; Chen, J.; He, P.; Chen, D.; Dai, X.; Jin, Q.; Su, X. The optimal irrigation water salinity and salt component for high-yield and good-quality of tomato in Ningxia. Agric. Water Manag. 2022, 274, 107940. [Google Scholar] [CrossRef]
- Sevik, H.; Cetin, M. Effects of Water Stress on Seed Germination for Select Landscape Plants. Pol. J. Environ. Stud. 2015, 24, 689–693. [Google Scholar] [CrossRef]
- Shabala, S. Ionic and osmotic components of salt stress specifically modulate net ion fluxes from bean leaf mesophyll. Plant Cell Environ. 2000, 23, 825–837. [Google Scholar] [CrossRef]
- Read, J.J.; Reddy, K.R.; Jenkins, J.N. Yield and fiber quality of Upland cotton as influenced by nitrogen and potassium nutrition. Eur. J. Agron. 2006, 24, 282–290. [Google Scholar] [CrossRef]
- Flores, P.; Hernández, V.; Hellín, P.; Fenoll, J.; Cava, J.; Mestre, T.; Martínez, V. Metabolite profile of the tomato dwarf cultivar Micro-Tom and comparative response to saline and nutritional stresses with regard to a commercial cultivar. J. Sci. Food Agric. 2015, 96, 1562–1570. [Google Scholar] [CrossRef] [PubMed]
- Quanen, G.; Lili, N.; Baoguo, L.; Shiyu, C. Effect of salt ion composition of irrigation water on parameters of soil water and salt movement. J. Agric. Eng. 2017, 33, 123–128. [Google Scholar]
- Gao, F.; Wang, L.; Xie, Y.; Sun, J.; Ning, H.; Han, Q.; Kanneh, J.E.; Liu, H. Optimizing canopy structure through equal row spacing and appropriate irrigation enhances machine-harvested seed cotton yield and quality. Ind. Crops Prod. 2024, 216, 118799. [Google Scholar] [CrossRef]
- Yucai, X.; Huifeng, N.; Xianbo, Z.; Wang, Z.; Peiwen, X.; Yinping, S.; Nanfang, L.; Xingpeng, W.; Hao, L. Reducing the Sodium Adsorption Ratio Improves the Soil Aggregates and Organic Matter in Brackish-Water-Irrigated Cotton Fields. Agronomy 2024, 14, 2169. [Google Scholar] [CrossRef]
- Song, J.; Xin, L.; Gao, F.; Liu, H.; Wang, X. Effects of Foliar Selenium Application on Oxidative Damage and Photosynthetic Properties of Greenhouse Tomato under Drought Stress. Plants 2024, 13, 302. [Google Scholar] [CrossRef]
- Zahedi, S.M.; Moharrami, F.; Sarikhani, S.; Padervand, M. Selenium and silica nanostructure-based recovery of strawberry plants subjected to drought stress. Sci. Rep. 2020, 10, 17672. [Google Scholar] [CrossRef]
- Rady, M.M.; Belal, H.E.E.; Gadallah, F.M.; Semida, W.M. Selenium application in two methods promotes drought tolerance in Solanum lycopersicum plant by inducing the antioxidant defense system. Sci. Hortic. 2020, 266, 109290. [Google Scholar] [CrossRef]
- Heitkemper, D.T.; Vela, N.P.; Stewart, K.R.; Westphal, C.S. Determination of total and speciated arsenic in rice by ion chromatography and inductively coupled plasma mass spectrometry. J. Anal. At. Spectrom. 2001, 16, 299–306. [Google Scholar] [CrossRef]
- Ben Ahmed, C.; Magdich, S.; Ben Rouina, B.; Boukhris, M.; Ben Abdullah, F. Saline water irrigation effects on soil salinity distribution and some physiological responses of field grown Chemlali olive. J. Environ. Manag. 2012, 113, 538–544. [Google Scholar] [CrossRef] [PubMed]
- Hong, C.-Y.; Chao, Y.-Y.; Yang, M.-Y.; Cho, S.-C.; Huei Kao, C. Na+ but not Cl− or osmotic stress is involved in NaCl-induced expression of Glutathione reductase in roots of rice seedlings. J. Plant Physiol. 2009, 166, 1598–1606. [Google Scholar] [CrossRef] [PubMed]
- Yamane, K.; Mitsuya, S.; Kawasaki, M.; Taniguchi, M.; Miyake, H. Antioxidant Capacity and Damages Caused by Salinity Stress in Apical and Basal Regions of Rice Leaf. Plant Prod. Sci. 2009, 12, 319–326. [Google Scholar] [CrossRef]
- Sarkar, R.K.; Mahata, K.R.; Singh, D.P. Differential responses of antioxidant system and photosynthetic characteristics in four rice cultivars differing in sensitivity to sodium chloride stress. Acta Physiol. Plant. 2013, 35, 2915–2926. [Google Scholar] [CrossRef]
- Li, J.; Chen, J.; Qu, Z.; Wang, S.; He, P.; Zhang, N. Effects of Alternating Irrigation with Fresh and Saline Water on the Soil Salt, Soil Nutrients, and Yield of Tomatoes. Water 2019, 11, 1693. [Google Scholar] [CrossRef]
- Chunthaburee, S.; Dongsansuk, A.; Sanitchon, J.; Pattanagul, W.; Theerakulpisut, P. Physiological and biochemical parameters for evaluation and clustering of rice cultivars differing in salt tolerance at seedling stage. Saudi J. Biol. Sci. 2015, 23, 467–477. [Google Scholar] [CrossRef]
- Kenji, U. Accumulation of some nitrogen compounds in response to salt stress and their relationships with salt tolerance in rice (Oryza sativa L.) seedlings. Plant Growth Regul. 2003, 41, 159–164. [Google Scholar] [CrossRef]
- Ali, R.; Gul, H.; Hamayun, M.; Rauf, M.; Iqbal, A.; Shah, M.; Hussain, A.; Bibi, H.; Lee, I.-J. Aspergillus awamori ameliorates the physicochemical characteristics and mineral profile of mung bean under salt stress. Chem. Biol. Technol. Agric. 2021, 8, 9. [Google Scholar] [CrossRef]
- Sun, J.; Chen, S.-L.; Dai, S.-X.; Wang, R.-G.; Li, N.-Y.; Shen, X.; Zhou, X.-Y.; Lu, C.-F.; Zheng, X.-J.; Hu, Z.-M.; et al. Ion flux profiles and plant ion homeostasis control under salt stress. Plant Signal. Behav. 2009, 4, 261–264. [Google Scholar] [CrossRef]
- Li, L. Screening of Kidney Bean Cultivars forSaline–alkaline Tolerance and Study ofPhysiological and Biochemical ResponseMechanism. Master’s Thesis, Heilongjiang Bayi Agricultural University, Daqing, China, June 2016. [Google Scholar]
- Sun, J. Effects of nitrogen on the growth and photosynthesis of industrial hemp(Cannabis sativa L.) under salt stress. Master’s Thesis, Yunnan University, Kunming, China, 15 January 2022. [Google Scholar]
- Ashraf, M.; Ahmad, A.; McNeilly, T. Growth and Photosynthetic Characteristics in Pearl Millet under Water Stress and Different Potassium Supply. Photosynthetica 2001, 39, 389–394. [Google Scholar] [CrossRef]
- Turan, S.; Tripathy, B.C. Salt-stress induced modulation of chlorophyll biosynthesis during de-etiolation of rice seedlings. Physiol. Plant. 2014, 153, 477–491. [Google Scholar] [CrossRef] [PubMed]
- Hu, L.; Xiang, L.; Li, S.; Zou, Z.; Hu, X.H. Beneficial role of spermidine in chlorophyll metabolism and D1 protein content in tomato seedlings under salinity-alkalinity stress. Physiol. Plant. 2015, 156, 468–477. [Google Scholar] [CrossRef] [PubMed]
- Xie, Y.; Liu, H.; Zhao, F.; Zhang, L.; Zhao, X.; Shi, Z.; Wang, X. Effects of irrigation water mineralization and sodium adsorption ratio on the growth and yield of drip-irrigated cotton under film. Arid. Zone Res. 2024, 41, 2071–2082. [Google Scholar] [CrossRef]

| Treatments | SAR (mmol/L)1/2 | Salinity (g/L) | Major Ion Content (mg/L) | |||||
|---|---|---|---|---|---|---|---|---|
| Ca2+ | Mg2+ | Na+ | K+ | Cl− | SO42− | |||
| CK | 8.74 | 1.44 | 103.60 | 42.07 | 419.09 | 6.45 | 648.10 | 87.54 |
| T3S10 | 10.10 | 3.00 | 356.69 | 42.07 | 759.08 | 6.45 | 1616.02 | 87.54 |
| T3S15 | 15.10 | 3.00 | 209.40 | 42.07 | 918.27 | 6.45 | 1604.10 | 87.54 |
| T3S20 | 20.11 | 3.00 | 121.78 | 42.07 | 1013.00 | 6.45 | 1597.01 | 87.54 |
| T5S10 | 10.11 | 5.00 | 789.42 | 42.07 | 1077.46 | 6.45 | 2864.91 | 87.54 |
| T5S15 | 15.08 | 5.00 | 536.00 | 42.07 | 1350.00 | 6.45 | 2844.47 | 87.54 |
| T5S20 | 20.11 | 5.00 | 368.69 | 42.07 | 1532.23 | 6.45 | 2830.87 | 87.54 |
| T7S10 | 10.10 | 7.00 | 1269.42 | 42.07 | 1344.74 | 6.45 | 4117.63 | 87.54 |
| T7S15 | 15.14 | 7.00 | 914.87 | 42.07 | 1727.97 | 6.45 | 4088.95 | 87.54 |
| T7S20 | 20.11 | 7.00 | 671.24 | 42.07 | 1991.32 | 6.45 | 4069.24 | 87.54 |
| Irrigation and Fertilizer Rates | Irrigation Quota/mm | Fertilizing Amount (kg·hm−2) | ||
|---|---|---|---|---|
| N | P2O5 | K2O | ||
| 10 June 2024 | 37.50 | 45 | 0 | 0 |
| 18 June 2024 | 34.87 | 30 | 30 | 15 |
| 25 June 2024 | 32.26 | 45 | 45 | 30 |
| 2 July 2024 | 43.21 | 75 | 75 | 30 |
| 9 July 2024 | 41.68 | 60 | 60 | 30 |
| 16 July 2024 | 42.49 | 60 | 45 | 75 |
| 22 July 2024 | 40.77 | 45 | 30 | 75 |
| 29 July 2024 | 47.41 | 30 | 30 | 60 |
| 5 August 2024 | 36.93 | 30 | 30 | 45 |
| 12 August 2024 | 39.58 | 30 | 30 | 15 |
| 20 August 2024 | 42.57 | 0 | 0 | 0 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Song, Y.; Xie, Y.; Zhang, C.; Ning, H.; Zhang, X.; Yang, G.; Liu, H. Reducing the Sodium Adsorption Ratio Promotes Cotton Growth and Development by Enhancing Antioxidant Enzyme Activities and the Plant’s Potassium–Sodium Ratio Under Brackish-Water Irrigation. Agronomy 2025, 15, 2092. https://doi.org/10.3390/agronomy15092092
Song Y, Xie Y, Zhang C, Ning H, Zhang X, Yang G, Liu H. Reducing the Sodium Adsorption Ratio Promotes Cotton Growth and Development by Enhancing Antioxidant Enzyme Activities and the Plant’s Potassium–Sodium Ratio Under Brackish-Water Irrigation. Agronomy. 2025; 15(9):2092. https://doi.org/10.3390/agronomy15092092
Chicago/Turabian StyleSong, Yinping, Yucai Xie, Chenfan Zhang, Huifeng Ning, Xianbo Zhang, Guang Yang, and Hao Liu. 2025. "Reducing the Sodium Adsorption Ratio Promotes Cotton Growth and Development by Enhancing Antioxidant Enzyme Activities and the Plant’s Potassium–Sodium Ratio Under Brackish-Water Irrigation" Agronomy 15, no. 9: 2092. https://doi.org/10.3390/agronomy15092092
APA StyleSong, Y., Xie, Y., Zhang, C., Ning, H., Zhang, X., Yang, G., & Liu, H. (2025). Reducing the Sodium Adsorption Ratio Promotes Cotton Growth and Development by Enhancing Antioxidant Enzyme Activities and the Plant’s Potassium–Sodium Ratio Under Brackish-Water Irrigation. Agronomy, 15(9), 2092. https://doi.org/10.3390/agronomy15092092







