Biochar Type, Ratio, and Nutrient Levels in Growing Media Affects Seedling Production and Plant Performance
Abstract
1. Introduction
2. Materials and Methods
2.1. Biochars and Plant Material
2.2. Preparation of Growing Media
2.3. Seed Emergence
2.4. Plant Growth and Nutrient Content
2.5. Total Phenols Content and Antioxidant Capacity
2.6. Lipid Peroxidation, Hydrogen Peroxide, and Enzyme Antioxidant Activity
2.7. Statistical Analysis
3. Results
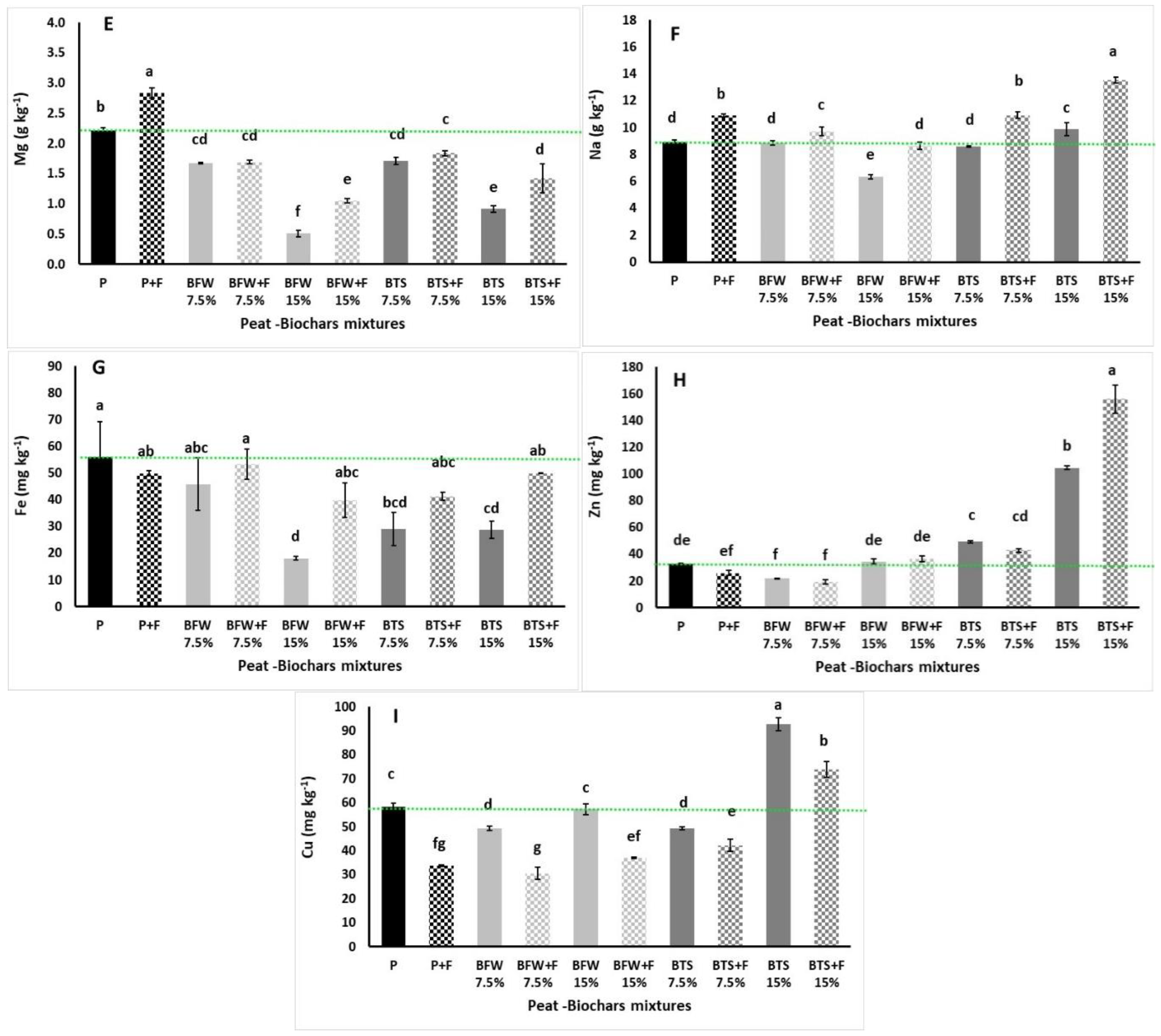
3.1. Growing Media Properties
3.2. Experiment I
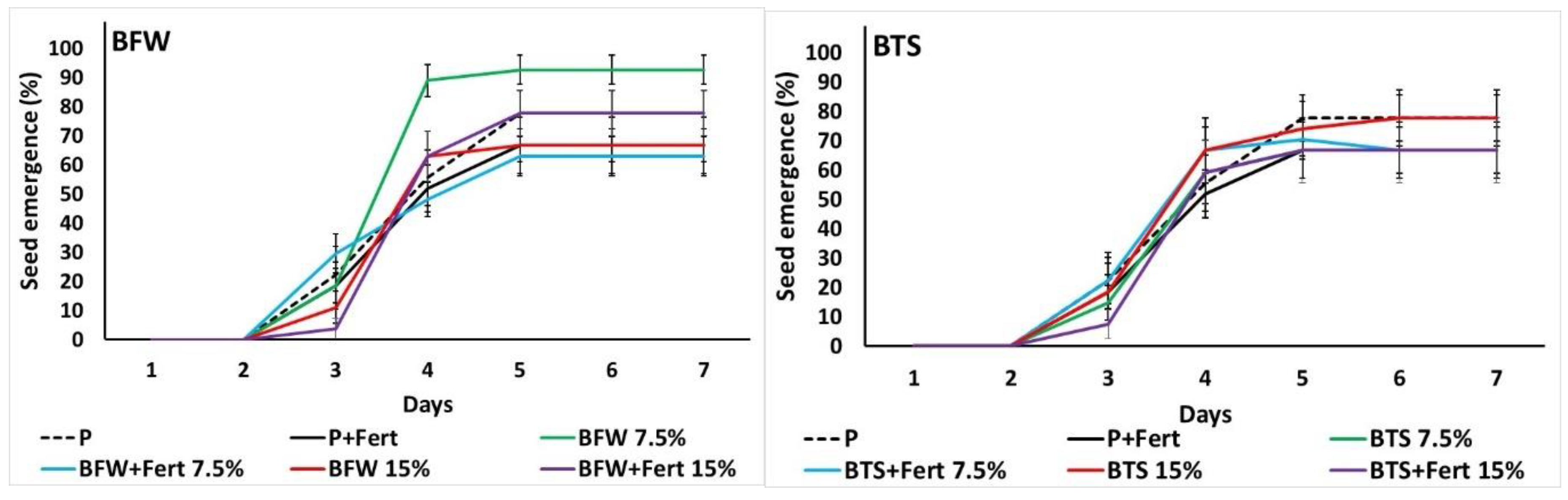
3.2.1. Seed Emergence
3.2.2. Plant Growth and Physiology
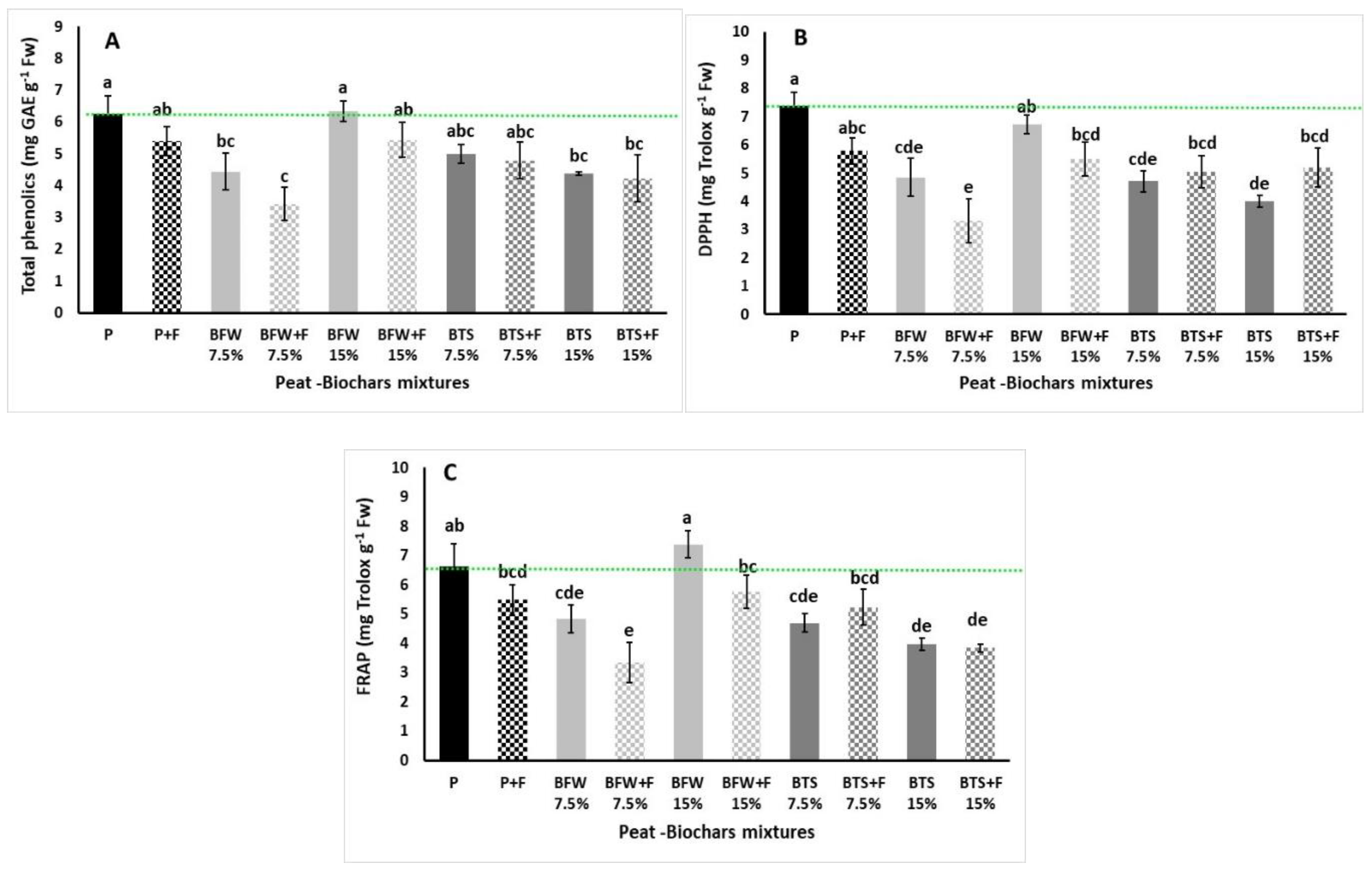
3.2.3. Total Phenol Content and Antioxidant Activity
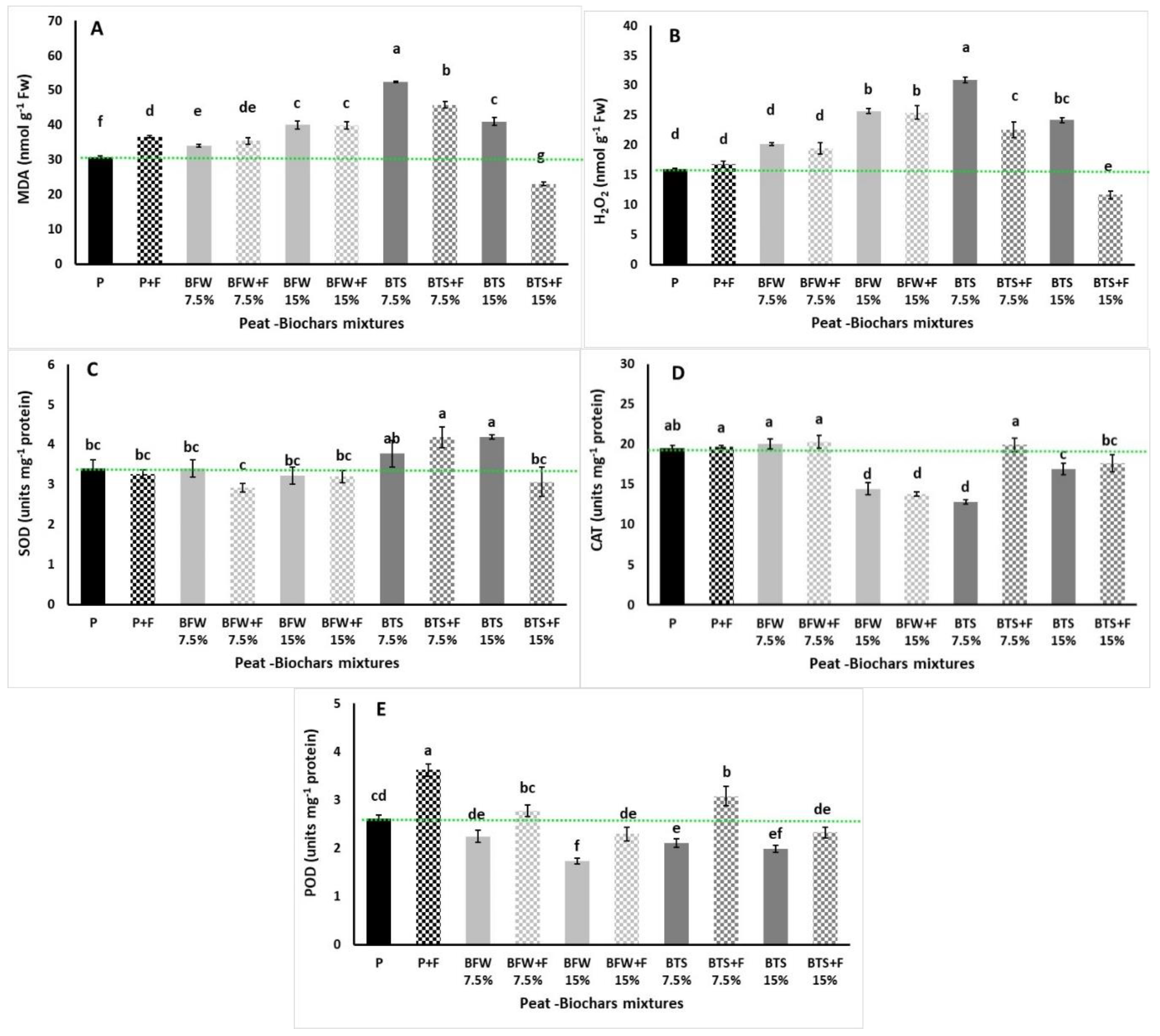
3.2.4. Lipid Peroxidation, Hydrogen Peroxide, and Enzyme Antioxidant Activity
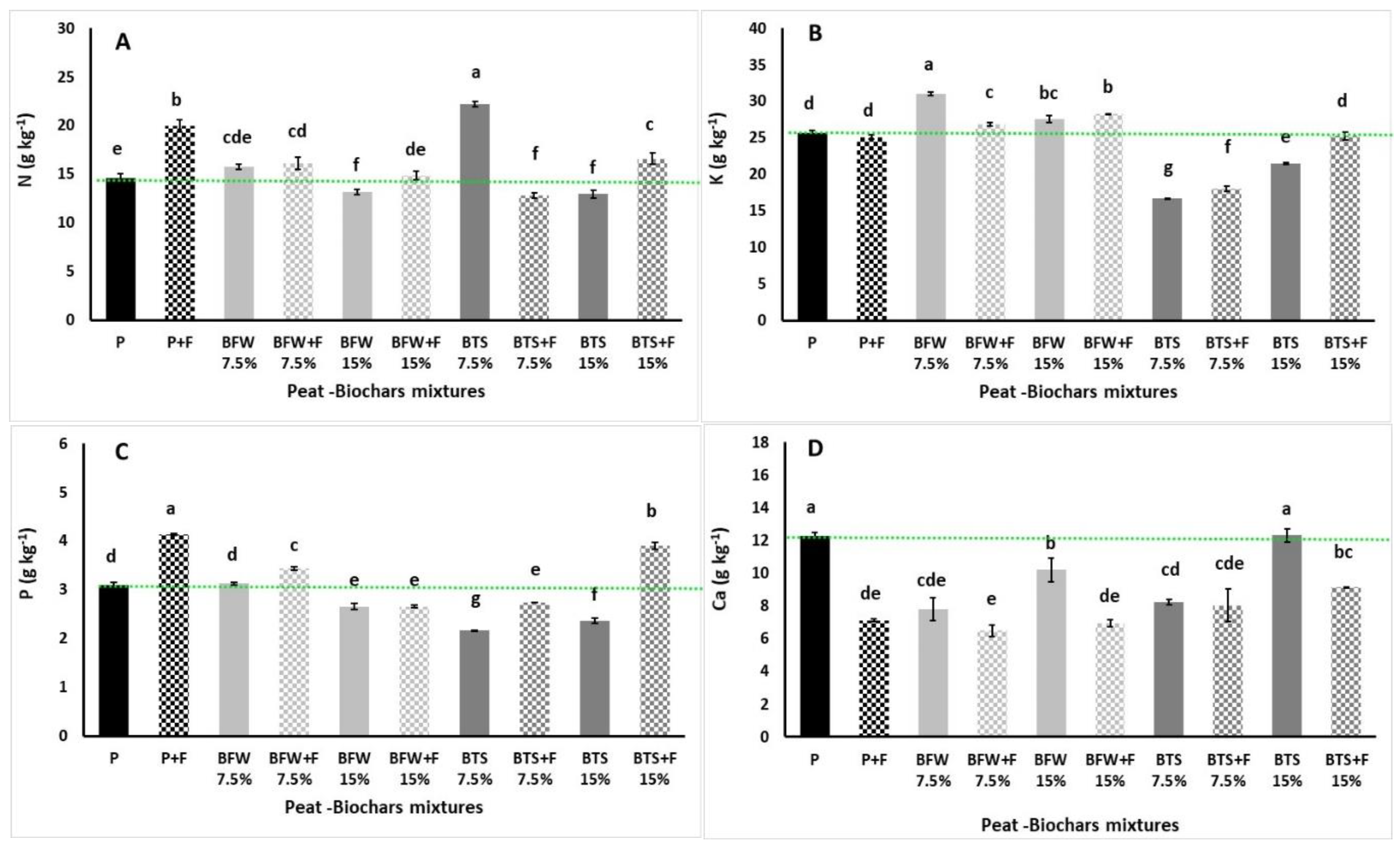
3.2.5. Nutrient Content
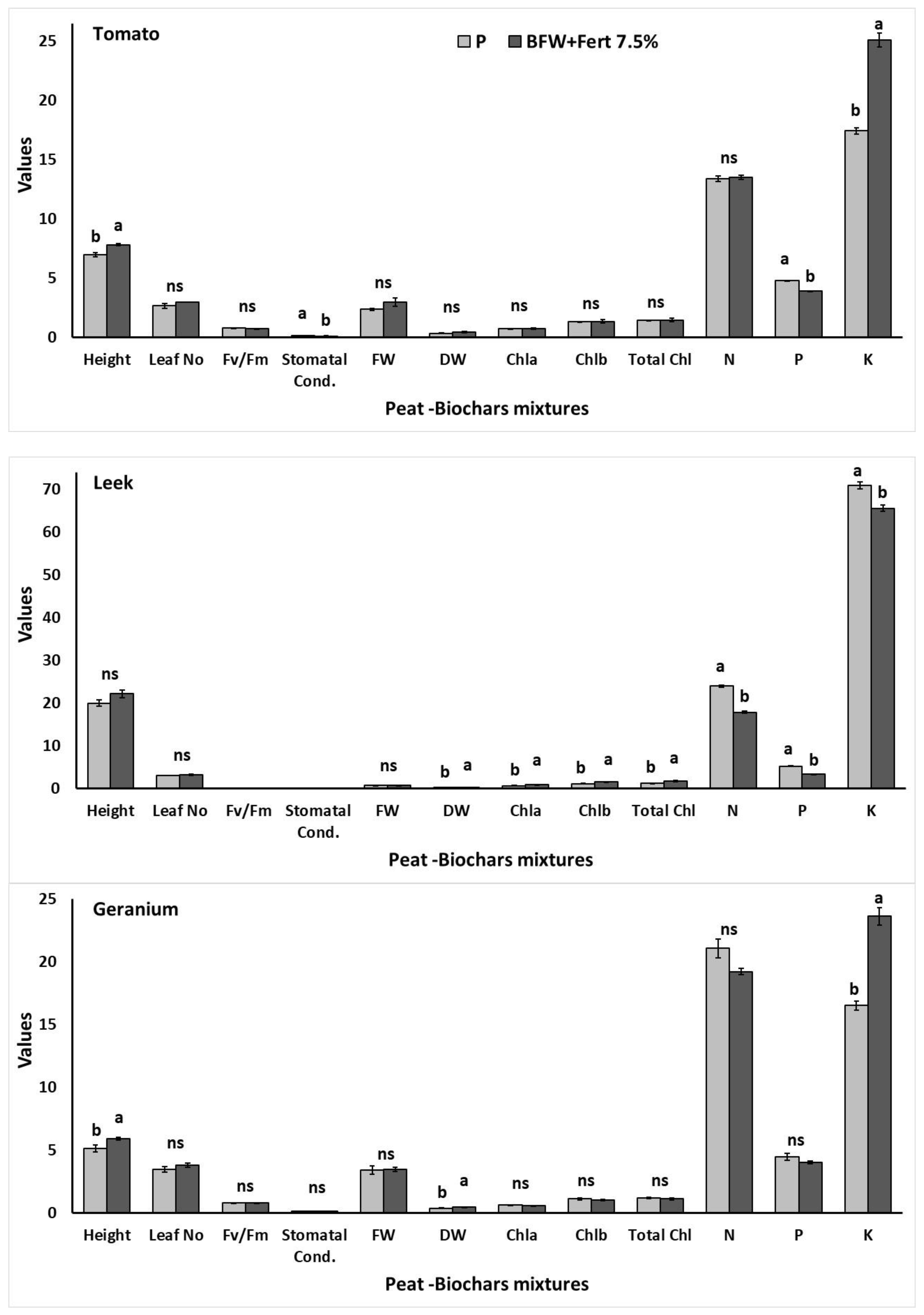
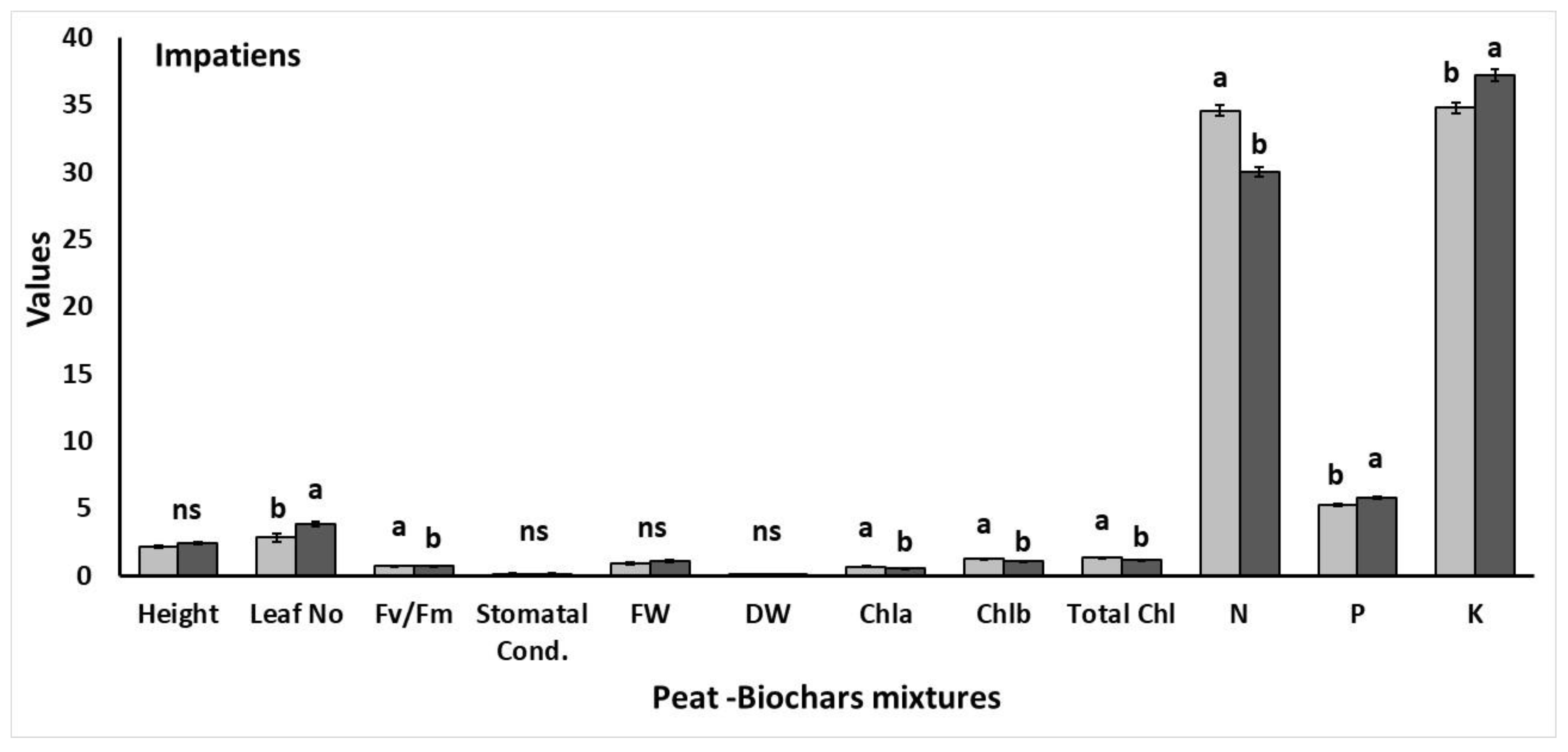
3.3. Experiment II
3.3.1. Seed Emergence
3.3.2. Effects on Plant Growth, Physiology, and Nutrient Content
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Solaiman, Z.M.; Murphy, D.V.; Abbott, L.K. Biochars influence seed germination and early growth of seedlings. Plant Soil 2012, 353, 273–287. [Google Scholar] [CrossRef]
- Laghari, M.; Hu, Z.; Mirjat, M.S.; Xiao, B.; Tagar, A.A.; Hu, M. Fast pyrolysis biochar from sawdust improves the quality of desert soils and enhances plant growth. J. Sci. Food Agric. 2016, 96, 199–206. [Google Scholar] [CrossRef]
- Méndez, A.; Paz-Ferreiro, J.; Gil, E.; Gascó, G. The effect of paper sludge and biochar addition on brown peat and coir based growing media properties. Sci. Hortic. Amst. 2015, 193, 225–230. [Google Scholar] [CrossRef]
- Agegnehu, G.; Bass, A.M.A.M.; Nelson, P.N.P.N.; Bird, M.I.M.I. Benefits of biochar, compost and biochar-compost for soil quality, maize yield and greenhouse gas emissions in a tropical agricultural soil. Sci. Total Environ. 2016, 543, 295–306. [Google Scholar] [CrossRef]
- Belda, R.M.; Lidón, A.; Fornes, F. Biochars and hydrochars as substrate constituents for soilless growth of myrtle and mastic. Ind. Crop. Prod. 2016, 94, 132–142. [Google Scholar] [CrossRef]
- Calamai, A.; Palchetti, E.; Masoni, A.; Marini, L.; Chiaramonti, D.; Dibari, C.; Brilli, L. The influence of biochar and solid digestate on rose-scented geranium (Pelargonium graveolens L’Hér.) productivity and essential oil quality. Agronomy 2019, 9, 260. [Google Scholar] [CrossRef]
- Saha, A.; Basak, B.B.; Gajbhiye, N.A.; Kalariya, K.A.; Manivel, P. Sustainable fertilization through co-application of biochar and chemical fertilizers improves yield, quality of Andrographis paniculata and soil health. Ind. Crop. Prod. 2019, 140, 111607. [Google Scholar] [CrossRef]
- Huang, L.; Niu, G.; Feagley, S.E.; Gu, M. Evaluation of a hardwood biochar and two composts mixes as replacements for a peat-based commercial substrate. Ind. Crop. Prod. 2019, 129, 549–560. [Google Scholar] [CrossRef]
- Lashari, M.S.; Ye, Y.; Ji, H.; Li, L.; Kibue, G.W.; Lu, H.; Zheng, J.; Pan, G. Biochar-manure compost in conjunction with pyroligneous solution alleviated salt stress and improved leaf bioactivity of maize in a saline soil from central China: A 2-year field experiment. J. Sci. Food Agric. 2015, 95, 1321–1327. [Google Scholar] [CrossRef]
- Prasad, M.; Tzortzakis, N.; McDaniel, N. Chemical characterization of biochar and assessment of the nutrient dynamics by means of preliminary plant growth tests. J. Environ. Manag. 2018, 216, 89–95. [Google Scholar] [CrossRef]
- Nadeem, S.M.; Imran, M.; Naveed, M.; Khan, M.Y.; Ahmad, M.; Zahir, Z.A.; Crowley, D.E. Synergistic use of biochar, compost and plant growth-promoting rhizobacteria for enhancing cucumber growth under water deficit conditions. J. Sci. Food Agric. 2017, 97, 5139–5145. [Google Scholar] [CrossRef]
- Alvarez, J.M.; Pasian, C.; Lal, R.; Lopez, R.; Fernandez, M. Vermicompost and Biochar as growing media replacement for ornamental plant production. J. Appl. Hortic. 2017, 19, 205–214. [Google Scholar] [CrossRef]
- Alvarez, J.M.; Pasian, C.; Lal, R.; Lopez-Nuñez, R.; Fernández, M. A biotic strategy to sequester carbon in the ornamental containerized bedding plant production: A review. Span. J. Agric. Res. 2018, 16, 4. [Google Scholar] [CrossRef]
- Zulfiqar, F.; Younis, A.; Chen, J. Biochar or Biochar-Compost Amendment to a Peat-Based Substrate Improves Growth of Syngonium podophyllum. Agronomy 2019, 9, 460. [Google Scholar] [CrossRef]
- Blok, C.; Van Der Salm, C.; Hofland-Zijlstra, J.; Streminska, M.; Eveleens, B.; Regelink, I.; Fryda, L.; Visser, R. Biochar for horticultural rooting media improvement: Evaluation of Biochar from gasification and slow pyrolysis. Agronomy 2017, 7, 6. [Google Scholar] [CrossRef]
- Gu, M.; Li, Q.; Steele, P.H.; Niu, G.; Yu, F. Growth of “Fireworks” gomphrena grown in substrates amended with biochar. J. Food Agric. Environ. 2013, 11, 819–821. [Google Scholar]
- Fryda, L.; Visser, R.; Schmidt, J. Biochar Replaces Peat in Horticulture: Environmental Impact Assessment of Combined Biochar & Bioenergy Production. Detritus 2019, 5, 132–149. [Google Scholar]
- Huang, L.; Gu, M. Effects of biochar on container substrate properties and growth of plants—A review. Horticulturae 2019, 5, 14. [Google Scholar] [CrossRef]
- Margenot, A.J.; Griffin, D.E.; Alves, B.S.Q.; Rippner, D.A.; Li, C.; Parikh, S.J. Substitution of peat moss with softwood biochar for soil-free marigold growth. Ind. Crop. Prod. 2018, 112, 160–169. [Google Scholar] [CrossRef]
- Guo, Y.; Niu, G.; Starman, T.; Volder, A.; Gu, M. Poinsettia Growth and Development Response to Container Root Substrate with Biochar. Horticulturae 2018, 4, 1. [Google Scholar] [CrossRef]
- Steiner, C.; Harttung, T. Biochar as a growing media additive and peat substitute. Solid Earth 2014, 5, 995–999. [Google Scholar] [CrossRef]
- Vaughn, S.F.; Kenar, J.A.; Thompson, A.R.; Peterson, S.C. Comparison of biochars derived from wood pellets and pelletized wheat straw as replacements for peat in potting substrates. Ind. Crop. Prod. 2013, 51, 437–443. [Google Scholar] [CrossRef]
- Dispenza, V.; De Pasquale, C.; Fascella, G.; Mammano, M.M.; Alonzo, G. Use of biochar as peat substitute for growing substrates of Euphorbia × lomi potted plants. Span. J. Agric. Res. 2016, 14, 21. [Google Scholar] [CrossRef]
- Choi, H.; Son, H.; Kim, C. Predicting financial distress of contractors in the construction industry using ensemble learning. Expert Syst. Appl. 2018, 110, 1–10. [Google Scholar] [CrossRef]
- Shackley, S.; Hammond, J.; Gaunt, J.; Ibarrola, R. The feasibility and costs of biochar deployment in the UK. Carbon Manag. 2011, 2, 335–356. [Google Scholar] [CrossRef]
- Gul, S.; Whalen, J.K. Biochemical cycling of nitrogen and phosphorus in biochar-amended soils. Soil Biol. Biochem. 2016, 103, 1–15. [Google Scholar] [CrossRef]
- Altland, J.E.; Locke, J.C. Biochar affects macronutrient leaching from a soilless substrate. HortScience 2012, 47, 1136–1140. [Google Scholar] [CrossRef]
- Viger, M.; Hancock, R.D.; Miglietta, F.; Taylor, G. More plant growth but less plant defence? First global gene expression data for plants grown in soil amended with biochar. GCB Bioenergy 2015, 7, 658–672. [Google Scholar] [CrossRef]
- French, E.; Iyer-Pascuzzi, A.S. A role for the gibberellin pathway in biochar-mediated growth promotion. Sci. Rep. 2018, 8, 1–10. [Google Scholar] [CrossRef]
- Prendergast-Miller, M.T.; Duvall, M.; Sohi, S.P. Biochar-root interactions are mediated by biochar nutrient content and impacts on soil nutrient availability. Eur. J. Soil Sci. 2014, 65, 173–185. [Google Scholar] [CrossRef]
- Abiven, S.; Hund, A.; Martinsen, V.; Cornelissen, G. Biochar amendment increases maize root surface areas and branching: A shovelomics study in Zambia. Plant Soil 2015, 395, 45–55. [Google Scholar] [CrossRef]
- Hilioti, Z.; Michailof, C.M.; Valasiadis, D.; Iliopoulou, E.F.; Koidou, V.; Lappas, A.A. Characterization of castor plant-derived biochars and their effects as soil amendments on seedlings. Biomass Bioenergy 2017, 105, 96–106. [Google Scholar] [CrossRef]
- Bachmann, R.T.; Adawiyah, S.; Krishnan, T.; Khoo, B.; Sian, T.S.; Richards, T. Partial substitution of peat moss with biochar for sustainable cultivation of Durio zibethinus L. in nurseries. Arab. J. Geosci. 2018, 11, 426. [Google Scholar] [CrossRef]
- Chrysargyris, A.; Prasad, M.; Kavanagh, A.; Tzortzakis, N. Biochar type and ratio as a peat additive/partial peat replacement in growing media for cabbage seedling production. Agronomy 2019, 9, 693. [Google Scholar] [CrossRef]
- Kloss, S.; Zehetner, F.; Dellantonio, A.; Hamid, R.; Ottner, F.; Liedtke, V.; Schwanninger, M.; Gerzabek, M.H.M.H.; Soja, G. Characterization of Slow Pyrolysis Biochars: Effects of Feedstocks and Pyrolysis Temperature on Biochar Properties. J. Environ. Qual. 2012, 41, 990–1000. [Google Scholar] [CrossRef]
- Yu, P.; Li, Q.; Huang, L.; Niu, G.; Gu, M. Mixed hardwood and sugarcane bagasse biochar as potting mix components for container tomato and basil seedling production. Appl. Sci. 2019, 9, 4713. [Google Scholar] [CrossRef]
- Judd, L.A.; Jackson, B.E.; Fonteno, W.C. Advancements in root growth measurement technologies and observation capabilities for container-grown plants. Plants 2015, 4, 369–392. [Google Scholar] [CrossRef]
- Chrysargyris, A.; Antoniou, O.; Athinodorou, F.; Vassiliou, R.; Papadaki, A.; Tzortzakis, N. Deployment of olive-stone waste as a substitute growing medium component for Brassica seedling production in nurseries. Environ. Sci. Pollut. Res. 2019, 26, 35461–35472. [Google Scholar] [CrossRef]
- Chrysargyris, A.; Stavrinides, M.; Moustakas, K.; Tzortzakis, N. Utilization of paper waste as growing media for potted ornamental plants. Clean Technol. Environ. Policy 2019, 21, 1937–1948. [Google Scholar] [CrossRef]
- Busch, D.; Stark, A.; Kammann, C.I.; Glaser, B. Genotoxic and phytotoxic risk assessment of fresh and treated hydrochar from hydrothermal carbonization compared to biochar from pyrolysis. Ecotoxicol. Environ. Saf. 2013, 97, 59–66. [Google Scholar] [CrossRef]
- Liao, N.; Li, Q.; Zhang, W.; Zhou, G.; Ma, L.; Min, W.; Ye, J.; Hou, Z. Effects of biochar on soil microbial community composition and activity in drip-irrigated desert soil. Eur. J. Soil Biol. 2016, 72, 27–34. [Google Scholar] [CrossRef]
- Gomez, J.D.; Denef, K.; Stewart, C.E.; Zheng, J.; Cotrufo, M.F. Biochar addition rate influences soil microbial abundance and activity in temperate soils. Eur. J. Soil Sci. 2014, 65, 28–39. [Google Scholar] [CrossRef]
- Ameloot, N.; Sleutel, S.; Case, S.D.C.; Alberti, G.; McNamara, N.P.; Zavalloni, C.; Vervisch, B.; delle Vedove, G.; De Neve, S. C mineralization and microbial activity in four biochar field experiments several years after incorporation. Soil Biol. Biochem. 2014, 78, 195–203. [Google Scholar] [CrossRef]
- Gorovtsov, A.V.; Minkina, T.M.; Mandzhieva, S.S.; Perelomov, L.V.; Soja, G.; Zamulina, I.V.; Rajput, V.D.; Sushkova, S.N.; Mohan, D.; Yao, J. The mechanisms of biochar interactions with microorganisms in soil. Environ. Geochem. Health 2019, 42, 2495–2518. [Google Scholar] [CrossRef]
- EN 13037-2002, 2002 Soil Improvers and Growing Media-Determination of pH.
- EN 13038-2002, 2002 Soil Improvers and Growing Media-Determination of Electrical Conductivity.
- EN 13651-2002, 2002. Soil Improvers and Growing Media-Extraction of Calcium Chloride/DTPA (CAT).
- Sinha, P.; Datar, A.; Jeong, C.; Deng, X.; Chung, Y.G.; Lin, L.C. Surface Area Determination of Porous Materials Using the Brunauer-Emmett-Teller (BET) Method: Limitations and Improvements. J. Phys. Chem. C 2019, 123, 20195–20209. [Google Scholar] [CrossRef]
- Kelepesi, S.; Tzortzakis, N.G. Olive mill wastesA growing medium component for seedling and crop production of lettuce and chicory. Int. J. Veg. Sci. 2009, 15, 325–339. [Google Scholar] [CrossRef]
- Chrysargyris, A.; Panayiotou, C.; Tzortzakis, N. Nitrogen and phosphorus levels affected plant growth, essential oil composition and antioxidant status of lavender plant (Lavandula angustifolia Mill.). Ind. Crop. Prod. 2016, 83, 577–586. [Google Scholar] [CrossRef]
- Tzortzakis, N.G.; Tzanakaki, K.; Economakis, C.D.C.D. Effect of origanum oil and vinegar on the maintenance of postharvest quality of tomato. Food Nutr. Sci. 2011, 2, 974–982. [Google Scholar] [CrossRef][Green Version]
- Chrysargyris, A.; Xylia, P.; Botsaris, G.; Tzortzakis, N. Antioxidant and antibacterial activities, mineral and essential oil composition of spearmint (Mentha spicata L.) affected by the potassium levels. Ind. Crop. Prod. 2017, 103, 202–212. [Google Scholar] [CrossRef]
- Loreto, F.; Velikova, V. Isoprene produced by leaves protects the photosynthetic apparatus against ozone damage, quenches ozone products, and reduces lipid peroxidation of cellular membranes. Plant Physiol. 2001, 127, 1781–1787. [Google Scholar] [CrossRef]
- De Azevedo Neto, A.D.; Prisco, J.T.; Enéas-Filho, J.; De Abreu, C.E.B.; Gomes-Filho, E. Effect of salt stress on antioxidative enzymes and lipid peroxidation in leaves and roots of salt-tolerant and salt-sensitive maize genotypes. Environ. Exp. Bot. 2006, 56, 87–94. [Google Scholar] [CrossRef]
- Fornes, F.; Belda, R.M.; Fernández de Córdova, P.; Cebolla-Cornejo, J. Assessment of biochar and hydrochar as minor to major constituents of growing media for containerized tomato production. J. Sci. Food Agric. 2017, 97, 3675–3684. [Google Scholar] [CrossRef] [PubMed]
- Shackley, S.; Ruysschaert, G.; Zwart, K.; Glaser, B. Biochar in European Soils and Agriculture: Science and Practice; Routledge: London, UK, 2016. [Google Scholar]
- Ronga, D.; Francia, E.; Allesina, G.; Pedrazzi, S.; Zaccardelli, M.; Pane, C.; Tava, A.; Bignami, C. Valorization of Vineyard By-Products to Obtain Composted Digestate and Biochar Suitable for Nursery Grapevine (Vitis vinifera L.) Production. Agronomy 2019, 9, 420. [Google Scholar] [CrossRef]
- Dunlop, S.J.; Arbestain, M.C.; Bishop, P.A.; Wargent, J.J. Closing the loop: Use of biochar produced from tomato crop green waste as a substrate for soilless, hydroponic tomato production. HortScience 2015, 50, 1572–1581. [Google Scholar] [CrossRef]
- Nair, A.; Carpenter, B. Biochar rate and transplant tray cell number have implications on pepper growth during transplant production. Horttechnology 2016, 26, 713–719. [Google Scholar] [CrossRef]
- Sun, L.; Li, L.; Chen, Z.; Wang, J.; Xiong, Z. Combined effects of nitrogen deposition and biochar application on emissions of N2O, CO2 and NH3 from agricultural and forest soils. Soil Sci. Plant Nutr. 2014, 60, 254–265. [Google Scholar] [CrossRef]
- Bedussi, F.; Zaccheo, P.; Crippa, L. Pattern of pore water nutrients in planted and non-planted soilless substrates as affected by the addition of biochars from wood gasification. Biol. Fertil. Soils 2015, 51, 625–635. [Google Scholar] [CrossRef]
- Prasad, M.; Chrysargyris, A.; McDaniel, N.; Kavanagh, A.; Gruda, N.S.; Tzortzakis, N. Plant nutrient availability and pH of biochars and their fractions, with the possible use as a component in a growing media. Agronomy 2020, 10, 10. [Google Scholar] [CrossRef]
- Bunt, A. Media and Mixes for Container-Grown Plants: A Manual on the Preparation and Use of Growing Media for Pot Plants, 1st ed.; Unwin Hyman Ltd.: London, UK, 1988. [Google Scholar]
- Reyes-Torres, M.; Oviedo-Ocaña, E.R.; Dominguez, I.; Komilis, D.; Sánchez, A. A systematic review on the composting of green waste: Feedstock quality and optimization strategies. Waste Manag. 2018, 77, 486–499. [Google Scholar] [CrossRef]
- Wang, S.; Gao, B.; Zimmerman, A.R.; Li, Y.; Ma, L.; Harris, W.G.; Migliaccio, K.W. Physicochemical and sorptive properties of biochars derived from woody and herbaceous biomass. Chemosphere 2015, 134, 257–262. [Google Scholar] [CrossRef]
- Altland, J.E.; Locke, J.C. Effect of biochar type on macronutrient retention and release from soilless substrate. HortScience 2013, 48, 1397–1402. [Google Scholar] [CrossRef]
- Liang, B.; Lehmann, J.; Solomon, D.; Kinyangi, J.; Grossman, J.; O’Neill, B.; Skjemstad, J.O.; Thies, J.; Luizão, F.J.; Petersen, J.; et al. Black Carbon Increases Cation Exchange Capacity in Soils. Soil Sci. Soc. Am. J. 2006, 70, 1719–1730. [Google Scholar] [CrossRef]
- Maroušek, J.; Kolář, L.; Vochozka, M.; Stehel, V.; Maroušková, A. Biochar reduces nitrate level in red beet. Environ. Sci. Pollut. Res. 2018, 25, 18200–18203. [Google Scholar] [CrossRef] [PubMed]
- Takaya, C.A.; Fletcher, L.A.; Singh, S.; Anyikude, K.U.; Ross, A.B. Phosphate and ammonium sorption capacity of biochar and hydrochar from different wastes. Chemosphere 2016, 145, 518–527. [Google Scholar] [CrossRef] [PubMed]
- Chintala, R.; Mollinedo, J.; Schumacher, T.E.; Papiernik, S.K.; Malo, D.D.; Clay, D.E.; Kumar, S.; Gulbrandson, D.W. Nitrate sorption and desorption in biochars from fast pyrolysis. Microporous Mesoporous Mater. 2013, 179, 250–257. [Google Scholar] [CrossRef]
- Kammann, C.I.; Schmidt, H.P.; Messerschmidt, N.; Linsel, S.; Steffens, D.; Müller, C.; Koyro, H.W.; Conte, P.; Stephen, J. Plant growth improvement mediated by nitrate capture in co-composted biochar. Sci. Rep. 2015, 5, 11080. [Google Scholar] [CrossRef]
- Yan, M. Seed priming stimulate germination and early seedling growth of Chinese cabbage under drought stress. S. Afr. J. Bot. 2015, 99, 88–92. [Google Scholar] [CrossRef]
- Méndez, A.; Cárdenas-Aguiar, E.; Paz-Ferreiro, J.; Plaza, C.; Gascó, G. The effect of sewage sludge biochar on peat-based growing media. Biol. Agric. Hortic. 2017, 33, 40–51. [Google Scholar] [CrossRef]
- Graber, E.R.; Harel, Y.M.; Kolton, M.; Cytryn, E.; Silber, A.; David, D.R.; Tsechansky, L.; Borenshtein, M.; Elad, Y. Biochar impact on development and productivity of pepper and tomato grown in fertigated soilless media. Plant Soil 2010, 337, 481–496. [Google Scholar] [CrossRef]
- Kim, H.S.; Kim, K.R.; Yang, J.E.; Ok, Y.S.; Kim, W., Il; Kunhikrishnan, A.; Kim, K.H. Amelioration of Horticultural Growing Media Properties Through Rice Hull Biochar Incorporation. Waste Biomass Valorization 2017, 8, 483–492. [Google Scholar] [CrossRef]
- Chrysargyris, A.; Nikolaidou, E.; Stamatakis, A.; Tzortzakis, N. Vegetative, physiological, nutritional and antioxidant behaivor of spearmint (Mentha spicata L.) in response to differnet nitrogen supply in hydroponics. J. Appl. Res. Med. Aroma. 2017, 6, 52–61. [Google Scholar]
- Gupta, D.D.; Palma, J.M.; Corpas, F.J. Reactive Oxygen Species and Oxidative Damage in Plants under Stress; Springer: Berlin/Heidelberg, Germany, 2015; ISBN 9783319204208. [Google Scholar]
- Savvas, D.; Gruda, N. Application of soilless culture technologies in the modern greenhouse industry—A review. Eur. J. Hortic. Sci. 2018, 83, 280–293. [Google Scholar] [CrossRef]
| Peat | BWF | BTS | |
|---|---|---|---|
| pH | 3.13 | 9.57 | 9.55 |
| EC (mS cm−1) | 0.034 | 0.613 | 0.410 |
| NH4-N (mg L−1) | 17.0 | <1.0 | <1.0 |
| NO3-N (mg L−1) | 3.0 | <1.0 | <1.0 |
| K (mg L−1) | 8.0 | 1087.0 | 745.0 |
| P (mg L−1) | 6.0 | 2.0 | 3.0 |
| Oxygen Uptake Rate (mmol O2 kg−1 organic matter per hour) | 5.5 | 2.3 | 4.3 |
| Particle size of < 1 mm (%) | 51.0 | 63.9 | 60.5 |
| Particle size of 1–5 mm (%) | 46.0 | 35.6 | 37.0 |
| Particle size of > 5 mm (%) | 3.0 | <1.0 | 3.0 |
| Surface area (m2 g−1) | 2.4 | 243.2 | 62.3 |
| Biochar Type (T) | Biochar Ratio (R) | Fertilizers (F) | T × R | T × F | R × F | T × R × F | |
|---|---|---|---|---|---|---|---|
| Growing media | |||||||
| EC (mS cm−1) | *** | *** | *** | * | ** | ns | ns |
| pH | *** | ns | ns | *** | ns | ns | * |
| Organic matter (%) | *** | *** | * | *** | ns | ns | ns |
| Organic C (%) | *** | *** | * | *** | ns | ns | ns |
| N (mg L−1) | ** | *** | *** | *** | ns | ns | ns |
| K (mg L−1) | *** | *** | *** | *** | ns | ** | ** |
| P (mg L−1) | ns | ** | *** | * | ** | ns | *** |
| Ca (mg L−1) | *** | ns | * | ns | ns | ns | * |
| Mg (mg L−1) | ns | *** | ns | * | ns | ns | ns |
| Na (mg L−1) | *** | *** | ns | *** | ns | ns | ns |
| SO4-S (mg L−1) | *** | *** | *** | *** | *** | *** | *** |
| Fe (mg L−1) | *** | *** | * | *** | *** | *** | *** |
| Cu (mg L1) | *** | ns | *** | ns | *** | *** | *** |
| Zn (mg L−1) | *** | *** | ns | *** | ns | ns | ns |
| Mn (mg L−1) | *** | *** | ns | *** | ns | ns | ns |
| B (mg L−1) | *** | ** | ns | ** | ns | ns | ns |
| Lettuce plant | |||||||
| Plant height (cm) | * | ns | *** | * | ns | ** | ** |
| Leaf Number | ns | ns | ** | ns | ns | ns | * |
| Chlorophyll fluorescence (Fv/Fm) | *** | *** | ns | ns | ** | ns | *** |
| Stomatal conductance (cm s−1) | ns | ns | ns | * | * | ** | ns |
| Fresh weight (g) | ns | ns | *** | ns | ns | ns | ns |
| Dry weight (g) | ns | ns | *** | ns | ns | * | ns |
| Chlorophyll a (mg g−1 Fw) | * | ns | * | * | * | ns | ns |
| Chlorophyll b (mg g−1 Fw) | * | ns | ** | * | * | ns | ns |
| Total Chlorophylls (mg g−1 Fw) | * | ns | * | * | * | ns | ns |
| Total phenolics (mg GAE g−1 Fw) | ns | ns | ns | * | ns | ns | ns |
| FRAP (mg trolox g−1 Fw) | * | ns | ns | ** | ns | ns | ns |
| DPPH (mg trolox g−1 Fw) | ns | ns | ns | * | ns | ns | ns |
| H2O2 (nmol g−1 Fw) | ns | ns | *** | *** | *** | ns | ns |
| MDA (nmol g−1 Fw) | *** | *** | ** | *** | *** | *** | ** |
| SOD (units mg−1 protein) | * | ns | ns | ns | ns | ns | * |
| CAT (units mg−1 protein) | ns | *** | * | *** | ** | ** | * |
| POD (units mg−1 protein) | ns | *** | *** | ns | ns | ns | ns |
| N (g kg−1) | *** | *** | ** | ns | *** | *** | *** |
| K (g kg−1) | *** | *** | ns | *** | *** | *** | ** |
| P (g kg−1) | *** | ns | *** | *** | *** | *** | *** |
| Ca (g kg−1) | *** | *** | *** | ns | ns | ** | ns |
| Mg (g kg−1) | *** | *** | *** | * | ns | ** | ns |
| Na (g kg−1) | *** | ns | *** | *** | *** | *** | ns |
| Fe (mg kg−1) | ns | ns | * | * | ns | ns | ns |
| Cu (mg kg−1) | *** | *** | *** | *** | * | * | ns |
| Zn (mg kg−1) | *** | *** | * | *** | *** | *** | *** |
| P | P + Fert | BFW 7.5% | BFW + Fert 7.5% | BFW 15% | BFW + Fert 15% | BTS 7.5% | BTS + Fert 7.5% | BTS 15% | BTS + Fert 15% | |
|---|---|---|---|---|---|---|---|---|---|---|
| EC (μS cm−1) | 417.14 ± 16.34 c | 576.02 ± 25.98 a | 329.85 ± 17.45 d | 478.90 ± 20.90 b | 209.53 ± 4.27 e | 236.15 ± 13.65 e | 318.90 ± 15.41 d | 502.45 ± 22.35 b | 252.66 ± 11.16 e | 361.15 ± 02.18 d |
| pH | 4.79 ± 0.01 c | 4.89 ± 0.05 c | 5.08 ± 0.06 b | 4.86 ± 0.6 c | 5.42 ± 0.03 a | 5.41 ± 0.05 a | 4.86 ± 0.06 c | 4.96 ± 0.10 bc | 4.57 ± 0.02 d | 4.38 ± 0.07 e |
| Organic matter (%) | 96.32 ± 0.18 a | 94.84 ± 0.02 c | 95.71 ± 0.11 b | 95.76 ± 0.17 ab | 95.57 ± 0.10 b | 96.01 ± 0.02 ab | 94.74 ± 0.09 c | 94.76 ± 0.12 c | 93.43 ± 0.34 d | 93.51 ± 0.26 d |
| Organic C (%) | 55.87 ± 0.10 a | 55.01 ± 0.01 c | 55.51 ± 0.06 b | 55.55 ± 0.10 ab | 55.43 ± 0.06 b | 55.69 ± 0.01 ab | 54.95 ± 0.05 c | 54.96 ± 0.07 c | 54.20 ± 0.20 d | 54.24 ± 0.15 d |
| N (mg L−1) | 85.16 ± 2.66 e | 157.93 ± 4.93 a | 87.10 ± 2.72 e | 126.44 ± 3.94 b | 71.74 ± 2.24 f | 113.60 ± 3.61 c | 85.16 ± 2.66 e | 131.61 ± 4.11 b | 46.45 ± 1.45 g | 101.21 ± 3.20 d |
| K (mg L−1) | 118.70 ± 3.70 f | 180.64 ± 5.64 d | 232.25 ± 7.25 c | 304.23 ±9.51 b | 381.92 ± 9.54 a | 372.96 ± 11.24 a | 103.22 ± 3.22 f | 154.83 ± 4.64 e | 103.21 ± 3.16 f | 147.42 ± 4.74 e |
| P (mg L−1) | 36.13 ± 1.13 e | 66.58 ± 2.08 b | 44.85 ± 1.40 d | 71.74 ± 2.24 ab | 40.77 ± 1.27 de | 58.35 ± 1.85 c | 44.78 ± 1.28 d | 69.16 ± 2.16 ab | 37.16 ± 1.16 e | 74.87 ± 2.17 a |
| Ca (mg L−1) | 701.91 ± 21.91 a | 717.54 ± 22.41 a | 701.84 ± 21.73 a | 728.23 ± 22.73 a | 725.65 ± 22.65 a | 719.42 ± 21.53 a | 521.79 ± 16.29 c | 524.88 ± 16.38 c | 486.17 ± 15.17 c | 615.12 ± 11.87 b |
| Mg (mg L−1) | 486.85 ± 15.21 a | 479.46 ± 14.96 a | 329.74 ± 10.29 b | 330.93 ± 9.56 b | 240.04 ± 7.49 d | 236.59 ± 7.49 d | 310.56 ± 9.69 b | 317.72 ± 9.92 b | 241.54 ± 7.54 d | 277.54 ± 8.79 c |
| Na (mg L−1) | 45.93 ± 1.43 c | 46.45 ± 1.45 c | 41.80 ± 1.28 c | 45.93 ± 1.43 c | 42.83 ± 1.33 c | 41.31 ± 1.31 c | 64.51 ± 2.01 b | 63.48 ± 1.98 b | 89.28 ± 2.78 a | 85.71 ± 1.95 a |
| SO4-S (mg L−1) | 8.77 ± 0.27 b | 8.26 ± 0.26 b | 6.71 ± 0.21 c | 6.71 ± 0.21 c | 6.70 ± 0.20 c | 6.72 ± 0.22 c | 6.71 ± 0.21 c | 6.71 ± 0.20 c | 6.19 ± 0.19 c | 13.94 ± 0.44 a |
| Fe (mg L−1) | 9.25± 0.29 bc | 8.26 ± 0.21 cde | 9.54 ± 0.30 b | 8.51 ± 0.23 cde | 8.15 ± 0.25 de | 7.79 ± 0.21 e | 8.36 ± 0.26 cde | 8.64 ± 0.26 bcde | 9.03 ± 0.28 bcd | 13.98 ± 0.45 a |
| Cu (mg L−1) | 0.11 ± 0.00 e | 0.10 ± 0.00 e | 0.04 ± 0.00 f | 0.04 ± 0.00 f | 0.04 ± 0.00 f | 0.04 ± 0.00 f | 0.21 ± 0.01 c | 0.26 ± 0.01 b | 0.15 ± 0.00d | 0.31 ± 0.01 a |
| Zn (mg L−1) | 1.08 ± 0.03 cd | 0.98 ± 0.03 d | 1.03 ± 0.03 d | 1.18 ± 0.04 c | 1.03 ± 0.03 d | 0.98 ± 0.03 d | 1.55 ± 0.05 b | 1.55 ± 0.05 b | 2.11 ± 0.06 a | 2.22 ± 0.07 a |
| Mn (mg L−1) | 2.42 ± 0.07 d | 2.43 ± 0.07 d | 8.98 ± 0.28 b | 8.97 ± 0.027 b | 12.95 ± 0.39 a | 12.75 ± 0.37 a | 2.63 ± 0.08 d | 2.68 ± 0.08 cd | 2.99 ± 0.09 cd | 3.41 ± 0.11 c |
| B (mg L−1) | 0.62 ± 0.02 c | 0.62 ± 0.02 c | 0.67 ± 0.02 c | 0.66 ± 0.02 c | 0.67 ± 0.02 c | 0.67 ± 0.02 c | 0.83 ± 0.03 b | 0.83 ± 0.02 b | 0.98 ± 0.03 a | 0.88 ± 0.03 b |
| Height | Leaf Number | Upper Fresh Weight | Upper Dry Weight | |
|---|---|---|---|---|
| P | 9.11 ± 0.71 abc | 5.33 ± 0.21 ab | 0.908 ± 0.153 bcd | 0.090 ± 0.026 bc |
| P + Fert | 9.61 ± 0.55 ab | 5.50 ± 0.42 a | 1.588 ± 0.241 a | 0.207 ± 0.054 a |
| BFW 7.5% | 6.50 ± 0.53 de | 4.16 ± 0.31 b | 0.504 ± 0.053 d | 0.046 ± 0.002 c |
| BFW + Fert 7.5% | 8.30 ± 0.58 bcd | 5.16 ± 0.47 ab | 1.061 ± 0.047 bc | 0.240 ± 0.051 a |
| BFW 15% | 6.06 ± 0.42 e | 4.66 ± 0.21 ab | 0.603 ± 0.015 cd | 0.085 ± 0.002 bc |
| BFW + Fert 15% | 8.01 ± 0.49 bcde | 5.00 ± 0.25 ab | 0.889 ± 0.083 cd | 0.093 ± 0.018 bc |
| BTS 7.5% | 6.42 ± 0.41 de | 4.50 ± 0.42 ab | 0.570 ± 0.009 d | 0.091 ± 0.000 bc |
| BTS + Fert 7.5% | 10.83 ± 1.06 a | 5.50 ± 0.62 a | 1.341 ± 0.274 ab | 0.159 ± 0.011 ab |
| BTS 15% | 7.01 ± 0.59 de | 4.83 ± 0.40 ab | 0.625 ± 0.063 cd | 0.089 ± 0.009 bc |
| BTS + Fert 15% | 7.56 ± 0.47 cde | 4.66 ± 0.33 ab | 0.853 ± 0.186 cd | 0.101 ± 0.018 bc |
| Stomatal Conductance | Chlorophyll Fluorescence | Chl a | Chl b | Total Chls | |
|---|---|---|---|---|---|
| P | 13.64 ± 0.60 ab | 0.903 ± 0.006 b | 0.621 ± 0.003 d | 0.161 ± 0.001 c | 0.782 ± 0.001 c |
| P + Fert | 11.41 ± 1.28 bc | 0.878 ± 0.007 c | 0.695 ± 0.035 cd | 0.184 ± 0.008 c | 0.879 ± 0.044 c |
| BFW 7.5% | 13.70 ± 2.12 ab | 0.871 ± 0.005 c | 0.638 ± 0.093 cd | 0.165 ± 0.027 c | 0.804 ± 0.121 c |
| BFW + Fert 7.5% | 8.41 ± 0.68 c | 0.919 ± 0.002 a | 0.681 ± 0.055 cd | 0.177 ± 0.018 c | 0.858 ± 0.074 c |
| BFW 15% | 11.01 ± 0.91 bc | 0.865 ± 0.003 c | 0.795 ± 0.032 bcd | 0.202 ± 0.007 bc | 0.997 ± 0.040 bc |
| BFW + Fert 15% | 17.26 ± 1.03 a | 0.868 ± 0.001 c | 0.822 ± 0.086 bcd | 0.214 ± 0.022 bc | 1.037 ± 0.109 bc |
| BTS 7.5% | 16.13 ± 2.29 a | 0.865 ± 0.003 c | 0.830 ± 0.045 bc | 0.207 ± 0.009 bc | 1.036 ± 0.054 bc |
| BTS + Fert 7.5% | 7.13 ± 1.70 c | 0.838 ± 0.001 d | 1.139 ± 0.108 a | 0.308 ± 0.030 a | 1.446 ± 0.138 a |
| BTS 15% | 9.33 ± 1.67 bc | 0.825 ± 0.007 d | 0.661 ± 0.037 cd | 0.168 ± 0.008 c | 0.830 ± 0.045 c |
| BTS + Fert 15% | 7.33 ± 1.19 c | 0.838 ± 0.002 d | 0.939 ± 0.048 b | 0.246 ± 0.013 b | 1.185 ± 0.061 b |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chrysargyris, A.; Prasad, M.; Kavanagh, A.; Tzortzakis, N. Biochar Type, Ratio, and Nutrient Levels in Growing Media Affects Seedling Production and Plant Performance. Agronomy 2020, 10, 1421. https://doi.org/10.3390/agronomy10091421
Chrysargyris A, Prasad M, Kavanagh A, Tzortzakis N. Biochar Type, Ratio, and Nutrient Levels in Growing Media Affects Seedling Production and Plant Performance. Agronomy. 2020; 10(9):1421. https://doi.org/10.3390/agronomy10091421
Chicago/Turabian StyleChrysargyris, Antonios, Munoo Prasad, Anna Kavanagh, and Nikos Tzortzakis. 2020. "Biochar Type, Ratio, and Nutrient Levels in Growing Media Affects Seedling Production and Plant Performance" Agronomy 10, no. 9: 1421. https://doi.org/10.3390/agronomy10091421
APA StyleChrysargyris, A., Prasad, M., Kavanagh, A., & Tzortzakis, N. (2020). Biochar Type, Ratio, and Nutrient Levels in Growing Media Affects Seedling Production and Plant Performance. Agronomy, 10(9), 1421. https://doi.org/10.3390/agronomy10091421







