Comparative Study of Three Low-Tech Soilless Systems for the Cultivation of Geranium (Pelargonium zonale): A Commercial Quality Assessment
Abstract
1. Introduction
2. Materials and Methods
2.1. Location
2.2. Treatments and Experimental Design
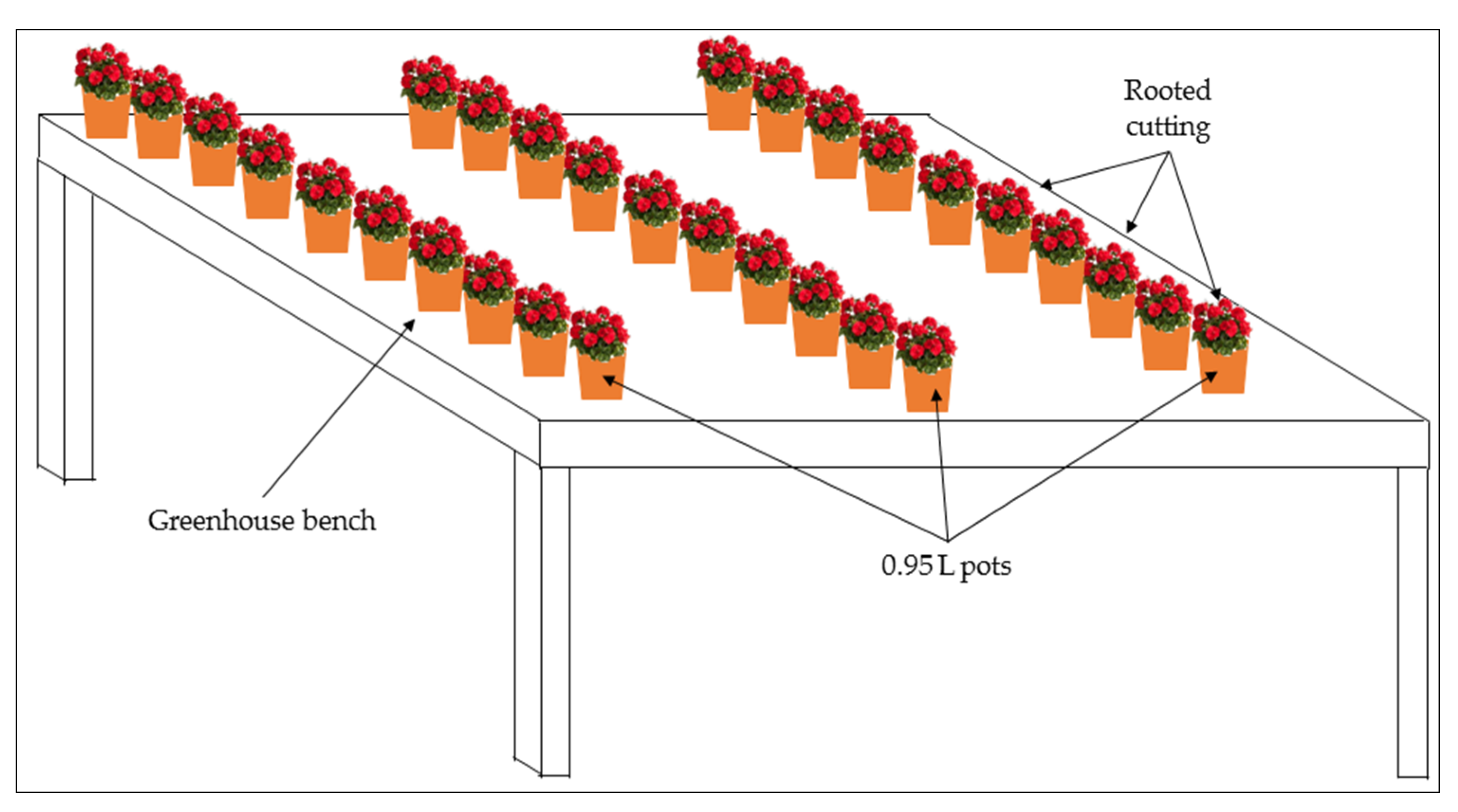
- T-1 (farm system with substrate, Figure 1): 30 rooted cuttings were grown on 0.95 L pot (Ø1 9 cm, Ø2 13 cm, h 10.8 cm), each featuring eight bottom holes, filled with a mixture of two different peats with a 1:1 volume ratio mixture (Peat A: Geotec srl, Adria, Italy, dry bulk density = 0.15 g·cm−3, total porosity = 92%; Peat B: Tercomposti S.p.A., Calvisano, Italy, dry bulk density = 0.10 g·cm−3, total porosity = 95%). The 30 pots were placed on a greenhouse bench, arranged in 3 rows with 10 pots each. Plants were manually watered using a 15 L watering can, as usually done in the local farms, daily supplying 100% of water evapotranspiration. The leaching fraction of water was tending to 0%. The rooted cuttings were fertilized only three times in total with granular fertilizer solubilized in water to have each time a concentration of 2.08 g·L−1.
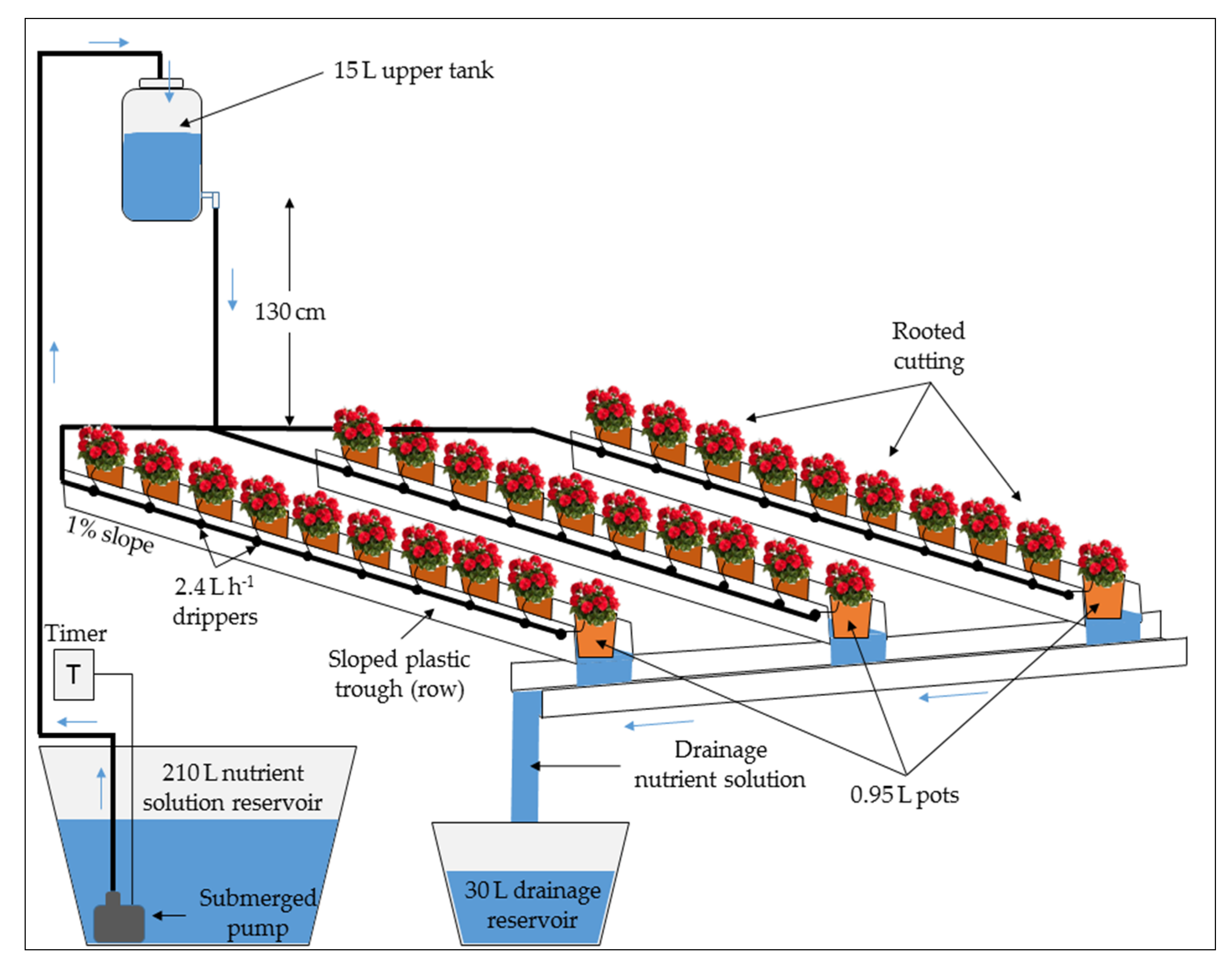
- T-2 (open-cycle drip system with substrate, Figure 2): 30 rooted cuttings were grown on 0.95 L pot (Ø1 9 cm, Ø2 13 cm, h 10.8 cm), with eight bottom holes, filled with a mixture of two different peats with a 1:1 volume ratio mixture (Peat A: Geotec srl, Adria, Italy, dry bulk density = 0.15 g·cm−3, total porosity = 92%; Peat B: Tercomposti S.p.A., Calvisano, Italy, dry bulk density = 0.10 g·cm−3, total porosity = 95%). The 30 pots were arranged in three rows with 10 pots each. Rows consisted of three plastic troughs measuring 1.50 m in length, 12 cm in width, and 6 cm in depth with a rectangular section and displaying a slope of about 1% to allow collecting the drained NS. The pots were placed inside the plastic troughs. The rooted cuttings were watered only with the NS by the drip irrigation system, daily supplying 130% of daily water requirement (leaching fraction of about 30%). The system was further integrated with a 210 L·NS reservoir tank located at the bottom of the plastic troughs, a submerged pump (Comet Elegance, Germany) with a flow rate of 10 L·min−1 and a pressure of 0.5 bar, a 15 L upper tank located at 130 cm high to receive the pumped NS, a drip irrigation system equipped with non-self-compensating emitters (2.4 L·h−1 nominal flow rate, one for each pot), and a 30 L reservoir tank for collecting the drainage. The leaching fraction was not reused, and T-2 was managed as an open system. Given that from the upper tank to the drippers the NS descended only by gravity, the actual flow rate of the drippers (0.66 L·h−1, as measured before the experiment started) was lower than their nominal flow rate. Accordingly, the correct amount of NS to be introduced in the system was determined through a programmable electronic timer that activated the pump.
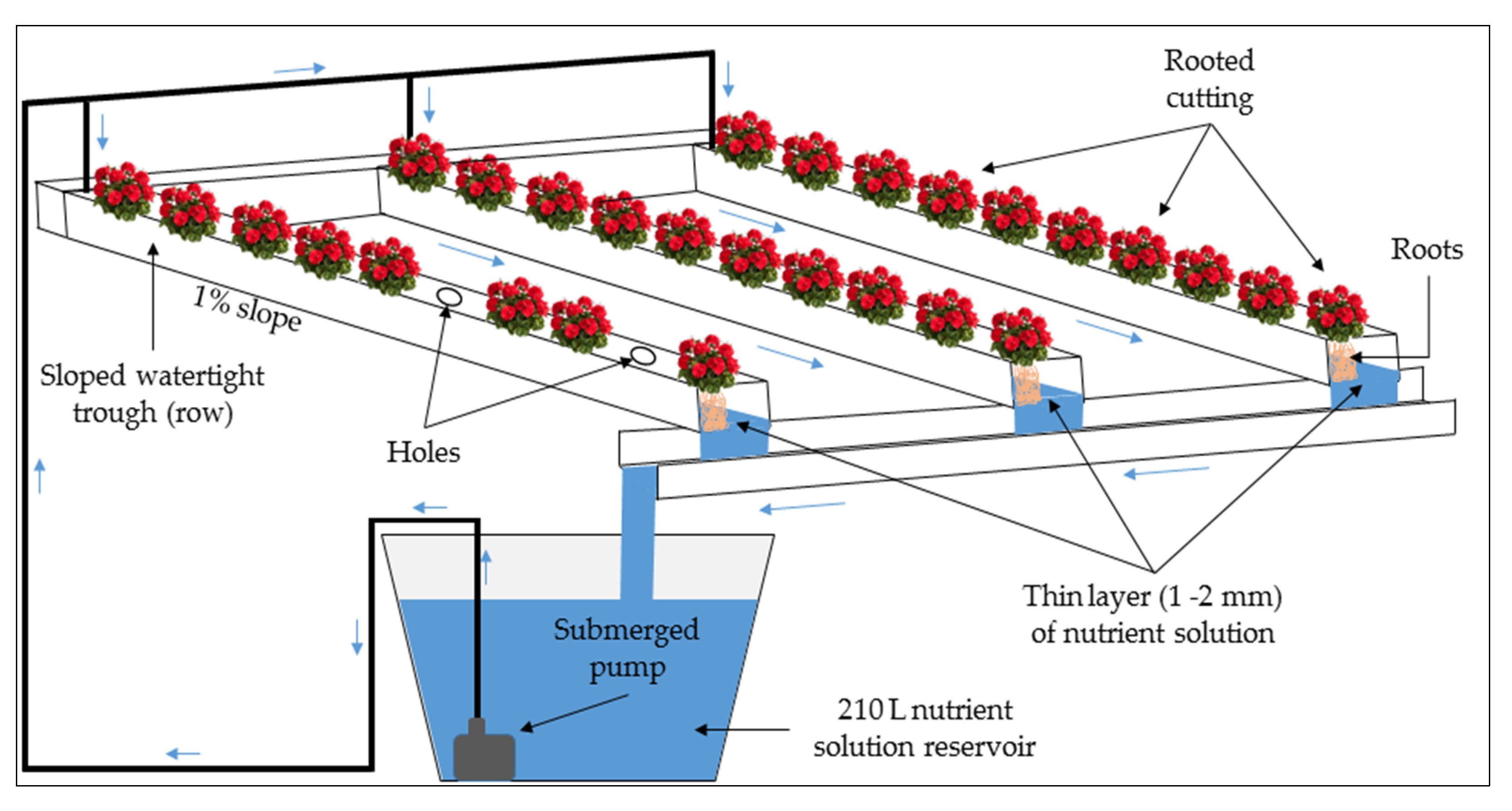
- T-3 (Nutrient Film Technique system, Figure 3): the system adopted the Nutrient Film Technique (NFT) and featured a closed soilless system with a thin layer of around 1–2 mm of NS flowing through sloped watertight troughs that hosted the plant roots. Thirty rooted cuttings were arranged in 3 rows consisting of 3 plastic troughs measuring 1.50 m in length, 12 cm in width, and 6 cm in depth with a rectangular section and a slope of about 1%. The plastic covers featured holes, where the rooted cuttings were placed. T-3 was also composed of a 210 L·NS reservoir located at the bottom of the plastic troughs and a submerged adjustable flow pump (Newa Jet, Italy) that pumped the NS in the plastic troughs.
2.3. Plant Material and Crop Management
2.4. Sampling and Analysis
2.5. Statistical Analysis
3. Results
3.1. Climate and Nutrient Solution Monitoring during the Experiment
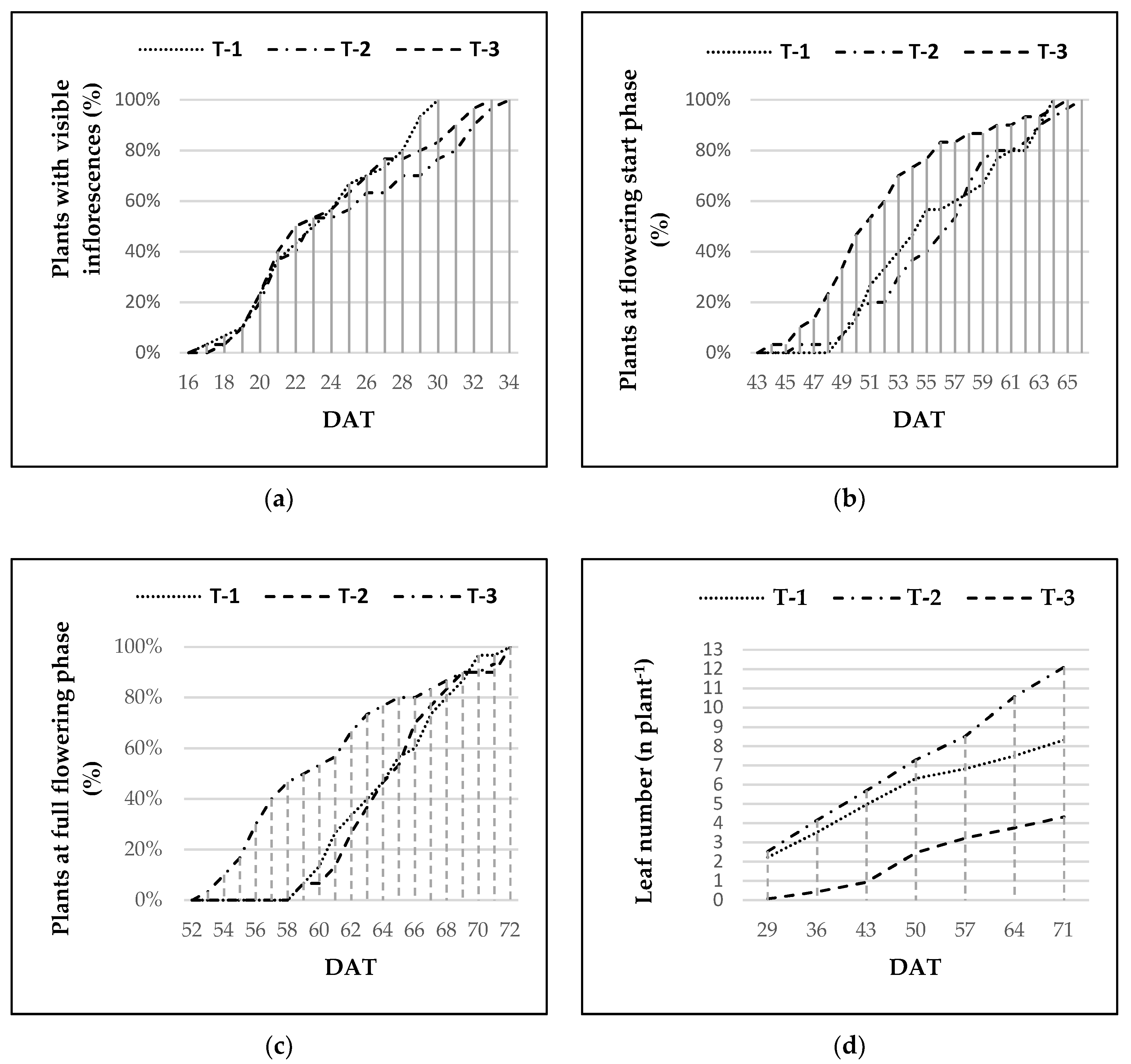
3.2. Date of the Appearance of Inflorescences
3.3. Date of Flowering Start
3.4. Date of Full Flowering
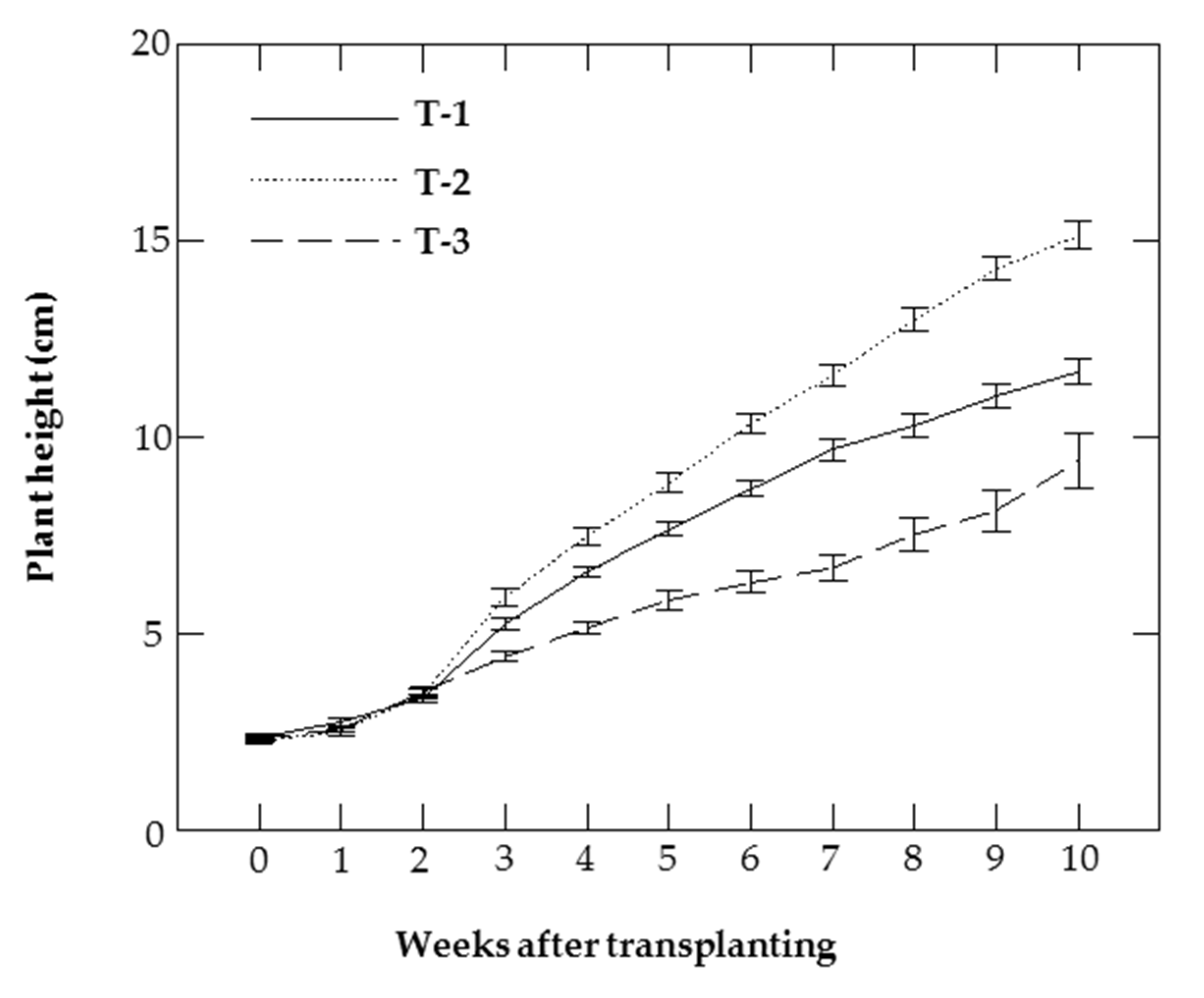
3.5. Biometrical Parameters
3.6. Number of Leaves
3.7. Leaf Chlorophyll
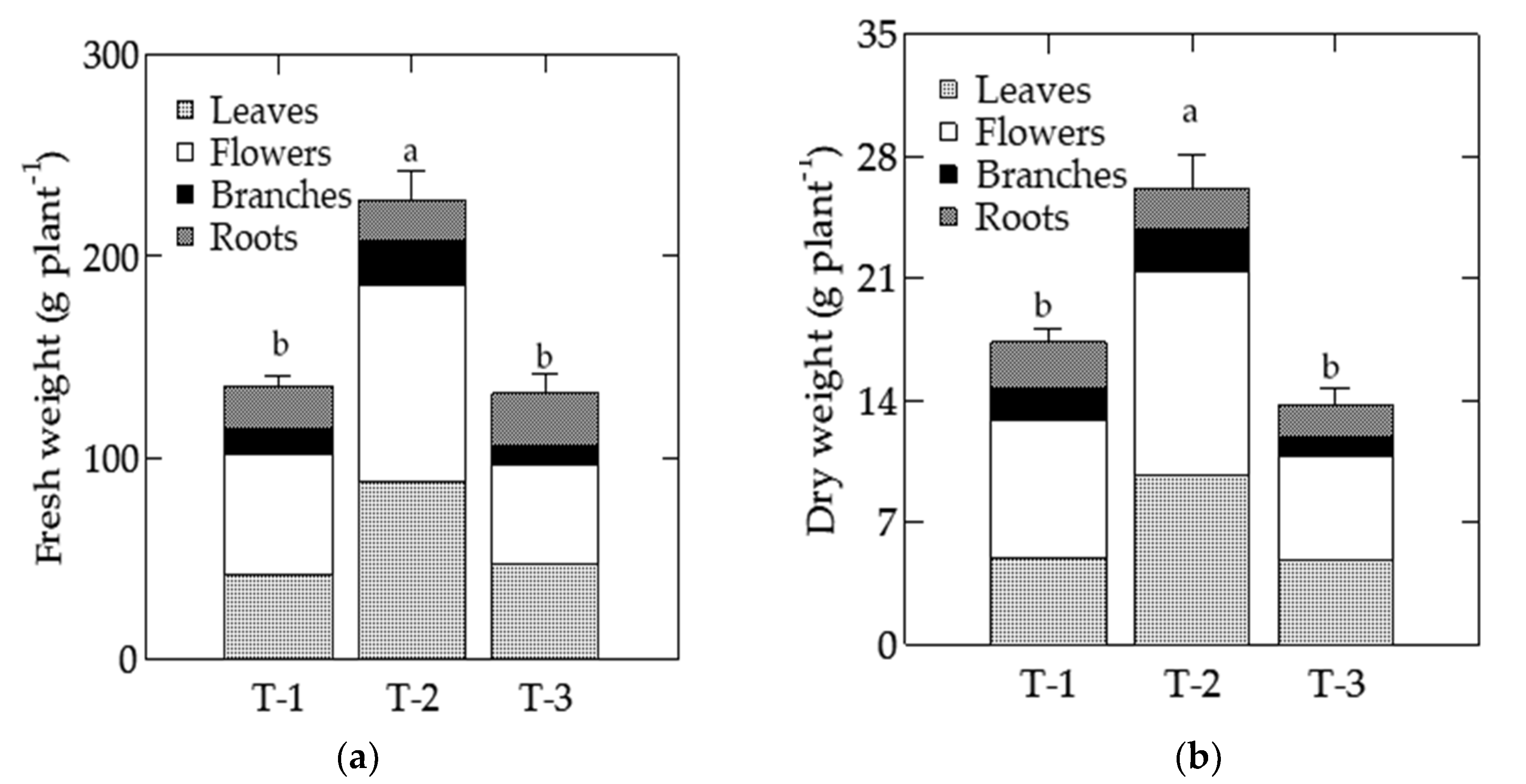
3.8. Fresh and Dry Weight
3.9. Aesthetic-Commercial Assessment
3.10. Water Consumption and WUE
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Size, E.O.M. Share & Trends Analysis Report by Application (Cleaning & Home, Medical, Food & Beverages, Spa & Relaxation), By Product, By Sales Channel, And Segment Forecasts, 2019–2025. In Market Analysis Report; Grand View Research: San Francisco, CA, USA, 2020; 978-1. [Google Scholar]
- MiPAAF. Available online: https://www.politicheagricole.it/flex/cm/pages/ServeBLOB.php/L/IT/IDPagina/10906 (accessed on 9 February 2017).
- Pardossi, A.; Incrocci, L.; Incrocci, G.; Tognoni, F. What limits and how to improve water use efficiency in outdoor container cultivation of ornamental nursery stocks. Acta Hortic. 2009, 843, 73–80. [Google Scholar] [CrossRef]
- Pardossi, A.; Marzialetti, P. La concimazione delle piante in contenitore. In Uso Razionale delle Risorse nel Forovivaismo: L’acqua; ARSIA: Florence, Italy, 2004; pp. 183–192. [Google Scholar]
- Pardossi, A.; Tognoni, F.; Incrocci, L. Mediterranean greenhouse technology. Chron. Hortic. 2004, 44, 28–34. [Google Scholar]
- Meric, M.K.; Tuzel, I.H.; Tuzel, Y.; Oztekin, G.B. Effects of nutrition system and irrigation programs on tomato in soilless culture. Agric. Water Manag. 2011, 99, 19–25. [Google Scholar] [CrossRef]
- Fernández, J.A.; Orsini, F.; Baeza, E.; Oztekin, G.B.; Muñoz, P.; Contreras, J.; Montero, J.I. Current trends in protected cultivation in Mediterranean climates. Eur. J. Hortic. Sci. 2018, 83, 294–305. [Google Scholar] [CrossRef]
- Van Os, E.A.; Gieling, T.H.; Lieth, J.H. Technical equipment in soilless production systems. In Soilless Culture: Theory and Practice; Raviv, M., Lieth, J.H., Eds.; Elsevier Publications: London, UK, 2008; pp. 157–207. [Google Scholar]
- Savvas, D.; Gianquinto, G.; Tüzel, Y.; Gruda, N. Soilless culture. In Good Agricultural Practices Principles for Greenhouse Vegetable Production in the Mediterranean Region; FAO Paper, AGP series n° 217; FAO: Rome, Italy, 2013; pp. 303–354. [Google Scholar]
- Incrocci, L.; Leonardi, C.I. Sistemi di coltivazione fuori suolo a ciclo chiuso. In Proceedings of the La produzione in Serra dopo l’Era del Bromuro di Metile International Workshop, Comiso, Italy, 1–3 April 2004. [Google Scholar]
- Hardgrave, M.R. Recirculation system for greenhouse vegetables. Acta Hortic. 1993, 342, 85–92. [Google Scholar] [CrossRef]
- Van Os, E.A. Closed growing system for more efficient and environmental friendly production. Acta Hortic. 1994, 361, 194–200. [Google Scholar] [CrossRef]
- Böhme, M. Influence of Closed Systems on the Development of Cucumber. Proceedings of the ISOSC Proceedings, Berlin, Germany, January 1996. pp. 75–87. Available online: https://www.library.wur.nl/WebQuery/titel/898177 (accessed on 20 September 2020).
- Tüzel, I.H.; Tüzel, Y.; Gül, A.; Meric, M.K.; Yavuz, Ö.; Eltez, R.Z. Comparison of open and closed systems on yield, water and nutrient consumption and their environmental impact. Acta Hortic. 2000, 554, 221–228. [Google Scholar] [CrossRef]
- Giuffrida, F.; Lipari, V.; Leonardi, C. A simplified management of closed soilless cultivation systems. Acta Hortic. 2003, 614, 155–160. [Google Scholar] [CrossRef]
- Lopez, J.; Perez, J.S.; Trejo, S.L.; Urrestarazu, M. Mineral nutrition and productivity of hydroponically grown tomatoes in relation to nutrient solution recycling. Acta Hortic. 2003, 609, 219–223. [Google Scholar] [CrossRef]
- Malorgio, F. Le colture fuori suolo per le produzioni floricole in serra. In Uso Razionale Delle Risorse nel Florovivaismo: L’acqua; ARSIA: Florence, Italy, 2004; pp. 49–57. [Google Scholar]
- Wortman, S.E. Crop physiological response to nutrient solution electrical conductivity and pH in an ebb-and-flow hydroponic system. Sci. Hortic. 2015, 194, 34–42. [Google Scholar] [CrossRef]
- Eccel, E.; Cordano, E. Progetto Indiclima: Elaborazione di indici climatici per il Trentino: Relazione finale. 2015. Fondazione Edmind Mach, San Michele all’Adige, Italy. Available online: http://www.hdl.handle.net/10449/34308 (accessed on 19 September 2020).
- Rouphael, Y.; Colla, G. The influence of drip-irrigation or subirrigation on zucchini squash grown in closed-loop substrate culture with high and low nutrient solution concentrations. HortScience. 2009, 44, 306–311. [Google Scholar] [CrossRef]
- Cardarelli, M.; Rouphael, Y.; Salerno, A.; Rea, E.; Colla, G. Fertilizer concentration and irrigation method affects growth and flowering of two bedding plants. Acta Hortic. 2012, 937, 689–696. [Google Scholar] [CrossRef]
- Riga, P. Avoiding the use of plant growth regulator in geranium production by application of a cyclic deficit irrigation strategy. JAH 2012, 14, 7–12. [Google Scholar] [CrossRef]
- Wang, Y.; Dunn, B.L.; Arnall, D.B.; Mao, P. Use of an active canopy sensor and SPAD chlorophyll meter to quantify geranium nitrogen satus. HortScience. 2012, 47, 45–50. [Google Scholar] [CrossRef]
- Smith, B.R.; Fisher, P.R.; Argo, W.R. Water-soluble fertilizer concentration and pH of a peat-based medium affect growth, nutrient uptake, and chlorosis of container-grown seed geraniums (Pelargonium x hortorum L.H.Bail). J. Plant. Nutr. 2004, 27, 497–524. [Google Scholar] [CrossRef]
- Enzo, M.; Gianquinto, G.; Lazzarin, R.; Pimpini, F.; Sambo, P. Principi Tecnico-Agronomici Della Fertirrigazione e del Fuori Suolo; Veneto Agricoltura, ARSAFA: Legnaro (PD), Italy, 2001; pp. 160–181. [Google Scholar]
- Voogt, W.; Sonneveld, C. Nutrient management in closed growing system for greenhouse production. In Plant Production in Closed Ecosystems; Goto, E., Kurata, K., Hayashi, M., Sase, S., Eds.; Kluwer Academic Publishers: Dordrecht, The Netherlands, 1997; pp. 83–102. [Google Scholar]
- Van Os, E.A. Closed soilless growing systems: A sustainable solution for Dutch greenhouse horticulture. Water Sci. Technol. 1999, 39, 105–112. [Google Scholar] [CrossRef]
- Rouphael, Y.; Colla, G.; Battistelli, A.; Moscatello, S.; Rea, E. Yield, water requirement, nutrient uptake and fruit quality of zucchini squash grown in soil and soilless culture. J. Hort. Sci. Biotech. 2004, 79, 423–430. [Google Scholar] [CrossRef]
- Van Os, E.A.; Ruijs, M.N.A.; van Weel, P.A. Closed business system for less pollution from greenhouses. Acta Hortic. 1991, 294, 49–57. [Google Scholar] [CrossRef]
| Treatment | Inflorescences (n·Plant−1) | Flowers (n·Inflorescence−1) | Branches (n·Plant−1) | Plant Height (cm) |
|---|---|---|---|---|
| T-1 | 10.63 b | 96.33 b | 5.33 b | 11.66 b |
| T-2 | 13.67 a | 143.11 a | 8.33 a | 15.13 a |
| T-3 | 6.00 c | 64.67 b | 2.78 c | 9.41 c |
| Mean | *** | *** | *** | *** |
| Treatment | SPAD Value |
|---|---|
| T-1 | 47.16 c |
| T-2 | 54.88 b |
| T-3 | 65.63 a |
| Mean | *** |
| Treatment | Leaves (%) | Flowers (%) | Branches (%) | Roots (%) |
|---|---|---|---|---|
| T-1 | 88.28 b | 86.90 b | 84.76 b | 87.55 b |
| T-2 | 89.06 ab | 87.76 a | 89.46 a | 87.93 b |
| T-3 | 89.68 a | 87.58 ab | 89.26 a | 93.22 a |
| Mean | *** | * | *** | *** |
| Treatment | MV | Vegetative Growth | Foliage Compactness | General Aspect |
|---|---|---|---|---|
| T-1 | 4.02 a | 4.17 a | 3.80 b | 4.10 a |
| T-2 | 4.27 a | 4.13 a | 4.53 a | 4.13 a |
| T-3 | 3.30 b | 2.98 b | 3.67 b | 3.28 b |
| Mean | *** | *** | *** | *** |
| Treatment | Plant FW (g·Plant−1) | TWC (L·Plant−1) | WUE (g·FW·L−1·H2O) |
|---|---|---|---|
| T-1 | 132.22 b | 7.50 c | 17.63 a |
| T-2 | 220.95 a | 10.11 a | 21.85 a |
| T-3 | 113.34 b | 8.68 b | 13.07 b |
| Mean | *** | *** | *** |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Brentari, L.; Michelon, N.; Gianquinto, G.; Orsini, F.; Zamboni, F.; Porro, D. Comparative Study of Three Low-Tech Soilless Systems for the Cultivation of Geranium (Pelargonium zonale): A Commercial Quality Assessment. Agronomy 2020, 10, 1430. https://doi.org/10.3390/agronomy10091430
Brentari L, Michelon N, Gianquinto G, Orsini F, Zamboni F, Porro D. Comparative Study of Three Low-Tech Soilless Systems for the Cultivation of Geranium (Pelargonium zonale): A Commercial Quality Assessment. Agronomy. 2020; 10(9):1430. https://doi.org/10.3390/agronomy10091430
Chicago/Turabian StyleBrentari, Luca, Nicola Michelon, Giorgio Gianquinto, Francesco Orsini, Federico Zamboni, and Duilio Porro. 2020. "Comparative Study of Three Low-Tech Soilless Systems for the Cultivation of Geranium (Pelargonium zonale): A Commercial Quality Assessment" Agronomy 10, no. 9: 1430. https://doi.org/10.3390/agronomy10091430
APA StyleBrentari, L., Michelon, N., Gianquinto, G., Orsini, F., Zamboni, F., & Porro, D. (2020). Comparative Study of Three Low-Tech Soilless Systems for the Cultivation of Geranium (Pelargonium zonale): A Commercial Quality Assessment. Agronomy, 10(9), 1430. https://doi.org/10.3390/agronomy10091430






