Analysis of Inclusions in the Entire Smelting Process of High-Grade Rare Earth Non-Oriented Silicon Steel
Abstract
1. Introduction
2. Materials and Methods
3. Results and Discussion
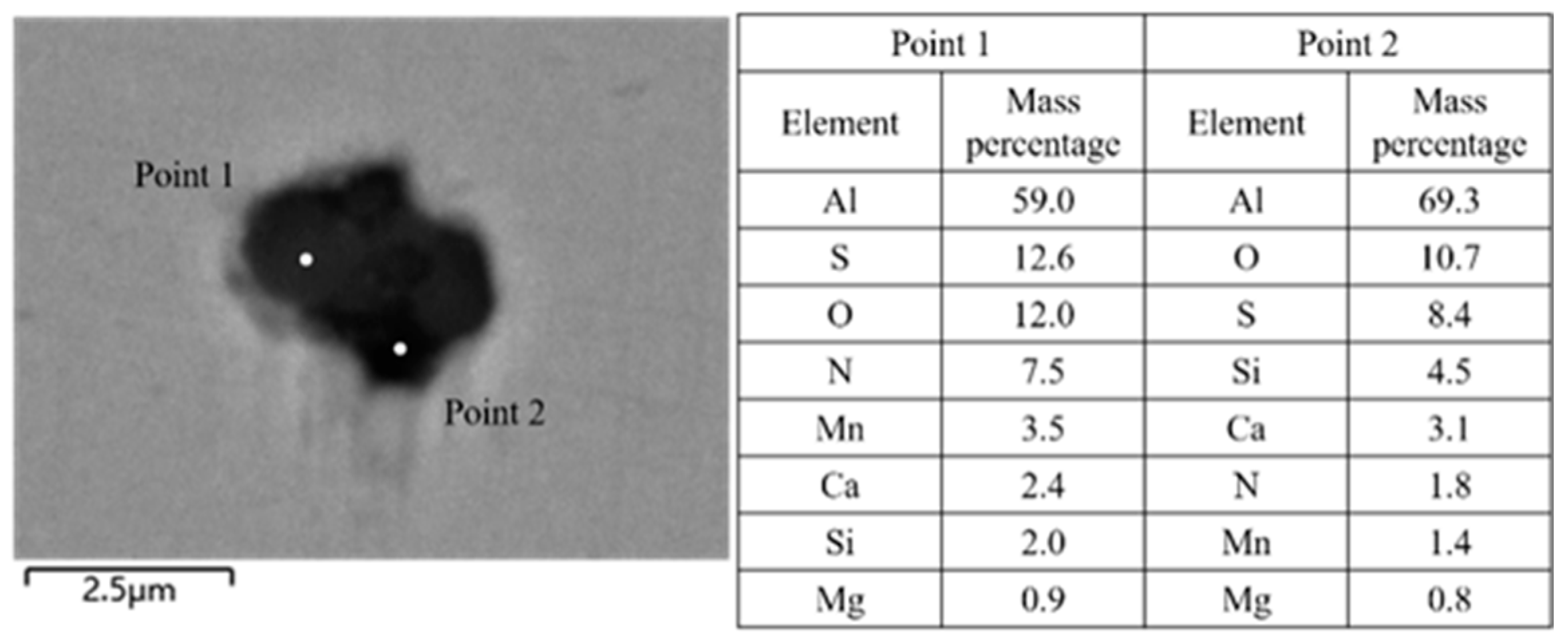
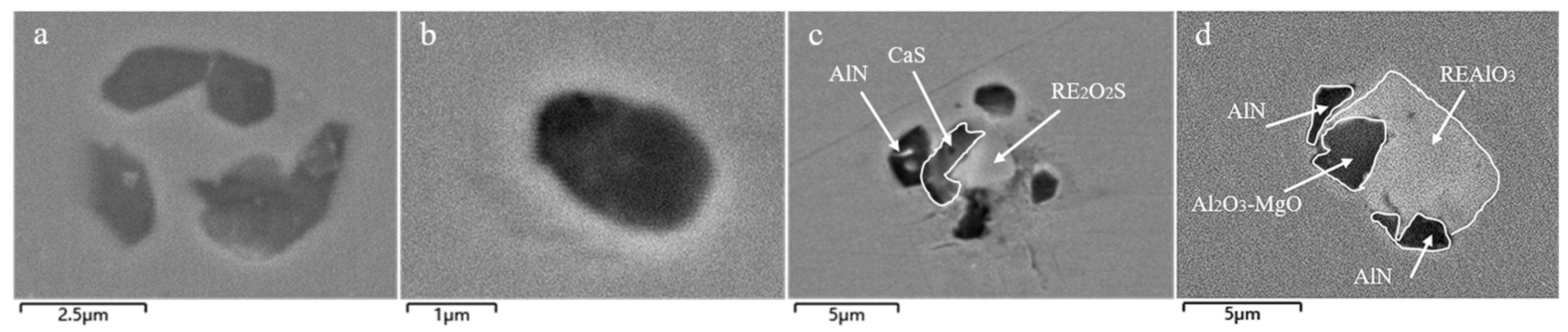
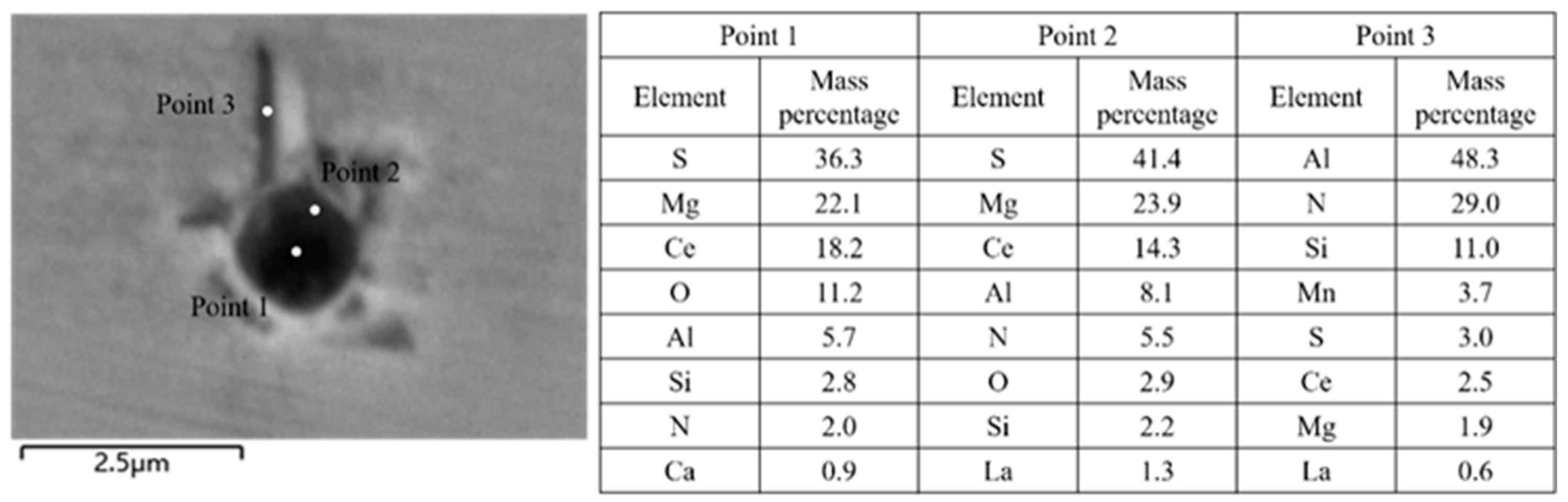
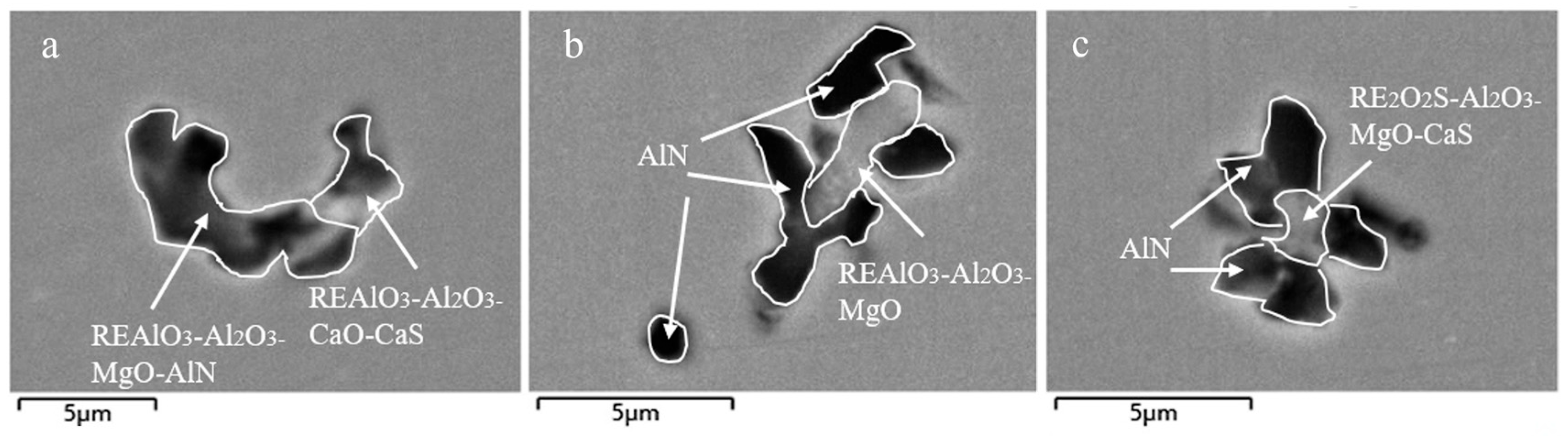
3.1. Analysis of Inclusions in the RH Process
3.2. Analysis of Inclusions in the Tundish
3.3. Analysis of Inclusions in Slab
3.4. Analysis of the Evolution of Inclusions Throughout the Entire Process
3.5. Analysis of Inclusion Size
3.6. Thermodynamic Calculation
3.6.1. Thermodynamic Calculation of Phase Stability Zone Diagram
3.6.2. Thermodynamic Calculation of Second Phase Precipitation in Molten Steel
3.7. Discussions
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Abbreviations
| RH | Ruhrstahl-Heraeus |
| RE | Rare earth |
| EDS | Energy Dispersive X-ray Spectroscopy |
References
- Irfan, M. Assessing Consumers’ Behavioral Intention and Willingness to Pay for Electric Vehicles: An Evidence from China. J. Compr. Bus. Adm. Res. 2024, 1, 2–11. [Google Scholar] [CrossRef]
- Li, W.; Cui, K.; Wu, L.; Zheng, B. Users’ Perceived Value of Electric Vehicles in China: A Latent Class Model-Based Analysis. World Electr. Veh. J. 2025, 16, 461. [Google Scholar] [CrossRef]
- Zuo, L.; Zhou, S.; Wang, M.; Zhang, S. Research on the Impact of Fiscal Subsidies on High-Quality Development of New Energy Vehicle Companies Based on Threshold Effects. J. Compr. Bus. Adm. Res. 2025, 1–13. [Google Scholar] [CrossRef]
- Kubota, T. Recent Progress on Non-oriented Silicon Steel. Steel Res. Int. 2005, 76, 464–470. [Google Scholar] [CrossRef]
- Oda, Y.; Kohno, M.; Honda, A. Recent development of non-oriented electrical steel sheet for automobile electrical devices. J. Mag. Mag. Mater. 2008, 320, 2430–2435. [Google Scholar] [CrossRef]
- Ouyang, G.Y.; Chen, X.; Liang, Y.F.; Macziewski, C.; Cui, J. Review of Fe-6.5 wt% Si high silicon steel-A promising soft magnetic material for sub-kHz application. J. Mag. Mag. Mater. 2019, 481, 234–250. [Google Scholar] [CrossRef]
- Xu, N.; Liang, Y.F.; Zhang, C.Y.; Wang, Z.; Wang, Y.L.; Ye, F.; Lin, J.P. Development of strong Goss texture in ultra-thin high silicon steel with excellent magnetic properties fabricated by two-stage rolling. Int. J. Miner. Metall. Mater. 2025, 32, 1595–1606. [Google Scholar] [CrossRef]
- Wang, J.Y.; Ren, Q.; Luo, Y.; Zhang, L.L. Effect of non-metallic precipitates and grain size on core loss of non-oriented electrical silicon steels. J. Mag. Mag. Mater. 2018, 451, 454–462. [Google Scholar] [CrossRef]
- Dijkstra, L.J.; Wert, C. Effect of inclusions on coercive force of iron. Phys. Rev. 1950, 79, 979–985. [Google Scholar] [CrossRef]
- Wang, H.J.; Xu, R.; Ling, H.T.; Zhong, W.; Chang, L.Z.; Qiu, S.T. Numerical simulation of fluid flow and alloy melting in RH process for electrical steels. J. Iron Steel Res. Int. 2022, 29, 1423–1433. [Google Scholar] [CrossRef]
- Wang, J.H.; Ni, P.Y.; Chen, C.; Ersson, M.; Li, Y. Effect of gas blowing nozzle angle on multiphase flow and mass transfer during RH refining process. Int. J. Miner. Metall. Mater. 2023, 30, 844–856. [Google Scholar] [CrossRef]
- Xue, L.Q.; Zhao, Y.S.; Wang, J.; Wang, H.; Miao, Z.; Xiong, R.X.; Xu, Z.B.; Lin, W.M.; Niu, X.F.; Chen, C. The flow performance of combined gas blowing at the vacuum chamber and up-snorkel in the ruhrstahl–heraeus (RH) degasser for electrical steel production. Processes 2025, 13, 448. [Google Scholar] [CrossRef]
- Xu, Z.B.; Ouyang, X.; Chen, C.; Li, Y.H.; Wang, T.Y.; Ren, R.J.; Yang, M.M.; Zhao, Y.S.; Xue, L.Q.; Wang, J. Numerical simulation of the density effect on the macroscopic transport process of tracer in the ruhrstahl–heraeus (RH) vacuum degasser. Sustainability 2024, 16, 3923. [Google Scholar] [CrossRef]
- Liu, X.J.; Yang, J.C.; Ren, H.P.; Jia, X.B.; Zhang, M.Y.; Yang, C.Q. Effect of solute Ce, Mn, and Si on mechanical properties of silicon steel: Insights from DFT calculations. J. Iron Steel Res. Int. 2024, 31, 700–709. [Google Scholar] [CrossRef]
- Ren, Q.; Zhang, L.F.; Hu, Z.Y.; Cheng, L. Transient influence of cerium on inclusions in an Al-killed non-oriented electrical steel. Ironmak. Steelmak. 2020, 48, 191–199. [Google Scholar] [CrossRef]
- Ren, Q.; Hu, Z.Y.; Liu, Y.X.; Zhang, W.C.; Gao, Z.Q.; Zhang, L.F. Effect of lanthanum on inclusions in non-oriented electrical steel slabs. J. Iron Steel Res. Int. 2024, 31, 1680–1691. [Google Scholar] [CrossRef]
- Cao, J.Q.; Chen, C.; Xu, L.Q.; Li, Z.; Zhao, J.; Lin, W.M. Analysis of the inclusions in the whole smelting process of non-oriented silicon steel DG47A. Iron Steel 2023, 58, 61–71. [Google Scholar] [CrossRef]
- Wang, T.; Chen, C.; Mu, W.Z.; Xue, L.Q.; Cao, J.Q.; Lin, W.M. Analysis of inclusions in whole smelting process of non-oriented silicon steel 23W1700 by rare earth treatment. Iron Steel 2024, 59, 92–103. [Google Scholar] [CrossRef]
- Kong, W.; Chen, Y.F.; Cang, D.Q. A statistical study of inclusions in medium-grade non-oriented silicon steel. Meter. Res. Technol. 2019, 116, 207. [Google Scholar] [CrossRef]
- Zhang, F.; Li, G.Q.; Miao, L.D. Effects of chemical composition systems on non-metallic inclusions in non-oriented silicon steel sheets. J. Iron Steel Res. 2012, 24, 40–44. [Google Scholar]
- Zhu, C.Y.; Liu, Y.L.; Xiao, Y.; Yan, W.; Li, G.Q. A new review on inclusion and precipitate control in grain-oriented silicon steels. JOM 2022, 74, 3141–3161. [Google Scholar] [CrossRef]
- Pindar, S.; Pande, M.M. Assessment of Si-O equilibria and nonmetallic inclusion characteristics in high silicon steels. Steel Res. Int. 2023, 94, 2300115. [Google Scholar] [CrossRef]
- Pindar, S.; Pande, M.M. Investigation of Inclusion Characteristics in Ferrosilicon Killed High Silicon Steels. Steel Res. Int. 2024, 95, 2400331. [Google Scholar] [CrossRef]
- Li, X.A.; Wang, N.; Chen, M.; Ma, T.Y. Tracking Large-size Inclusions in Al Deoxidated Tinplate Steel in Industrial Practice. ISIJ Int. 2021, 61, 2074–2082. [Google Scholar] [CrossRef]
- Ueshima, Y.; Sawada, Y.; Mizoguchi, S.; Kajioka, H. Precipitation behavior of MnS during δ/γ transformation in Fe−Si alloys. Metall Mater. Trans. A 1989, 20, 1375–1383. [Google Scholar] [CrossRef]
- Ito, Y.M.; Masumitsu, N.; Matsubara, K.; Komatsubra, M. Formation of MnS-type inclusion in steel. Tetsu Hagane 1980, 66, 647–656. [Google Scholar] [CrossRef] [PubMed]
- Zhou, B.; Zhu, C.Y.; Li, G.Q.; Luo, Z.; Gao, Y.Z.; Bao, S.Q.; Schneider, J. Effect of sulphur and acid soluble aluminium content on grain oriented silicon steel. Mater. Sci. Technol. 2015, 31, 1809–1817. [Google Scholar] [CrossRef]
- Takamiya, T.; Obare, T.; Muraki, M.; Komatsubara, M. Influence of sulfur content and deformation temperature on precipitation behavior of MnS in 3% Si steel. Tetsu Hagane 2003, 89, 518–523. [Google Scholar] [CrossRef][Green Version]
- Wu, S.J.; Wang, W.L.; Yue, C.X.; Qian, H.W.; Li, H.L. Effect of Annealing Stress on the Magnetic Properties of High-Grade Non-oriented Electrical Steel. Metall. Mater. Trans. A. 2024, 55, 2602–2608. [Google Scholar] [CrossRef]
- Ren, Q.; Hu, Z.Y.; Cheng, L.; Zhang, L.F. Effect of rare earth elements on magnetic properties of non-oriented electrical steels. J. Magn. Magn. Mater. 2022, 560, 169624. [Google Scholar] [CrossRef]
- Song, J.T.; Chen, C.; Guo, Z.J.; Geng, M.J.; Rong, Z.R.; Wang, T.Y.; Wang, J.; E, D.; Fan, J.P.; Sun, Y.H. Analysis of Flow Field, Temperature, and Inclusion Evolution in a 12-Strand Tundish During Ladle Changeover Process. Metall. Mater. Trans. B 2025, 56, 2700–2714. [Google Scholar] [CrossRef]
- Liu, P.; Cai, Z.Z.; Zhu, M.Y. Effect of La Treatment and Al–La Composite Treatment on Sulfur-Containing Peritectic Steel. Metall. Mater. Trans. B 2025, 56, 4114–4124. [Google Scholar] [CrossRef]
- Zhang, L.F.; Wei, H.; Duan, S.C. Effect of Gadolinium on Inclusions in a High-Sulfur Free-Cutting Steel and Converting Two-Dimensional to Three-Dimensional Morphology of Inclusions Through Matlab Programming. Metall. Mater. Trans. B 2024, 55, 4759–4774. [Google Scholar] [CrossRef]
- Ren, Q.; Zhang, L.F. Effect of cerium content on inclusions in an ultra-low-carbon aluminum-killed steel. Metall. Mater. Trans. B 2020, 51, 589–600. [Google Scholar] [CrossRef]
- Cao, J.Q.; Li, Y.; Lin, W.M.; Che, J.L.; Zhou, F.; Tan, Y.F.; Li, D.L.; Dang, J.; Chen, C. Assessment of inclusion removal ability in refining slags containing Ce2O3. Crystals 2023, 13, 202. [Google Scholar] [CrossRef]
- Ren, P.; Wang, H.; Zhao, Y.; Zhu, X.; Shi, C.B.; Lan, P. Effect of Cerium on Solidification Structure and Eutectic Carbides in Electroslag Remelted S136 Tool Steel. Metall. Mater. Trans B 2024, 55, 5080–5092. [Google Scholar] [CrossRef]
- Zhao, Y.; Wang, H.; Liu, J.; Zhu, X.; Zhang, H.; Shi, C.B. Effect of Cerium on Solidification Structure and Eutectic Precipitates in Electroslag Remelted Cr–W–Co Martensitic Heat-Resistant Steel. Metall. Mater. Trans. B 2024, 55, 4915–4928. [Google Scholar] [CrossRef]
- Liang, Y.Y.; Ni, P.Y.; Liu, Q.L.; Li, Y. Effect of Rare Earth Metal Alloying on Inclusion Evolution in High-Strength Oil Casing Steel. Metall. Mater. Trans. B 2024, 55, 3158–3173. [Google Scholar] [CrossRef]
- Yu, Z.; Liu, C.J. Modification mechanism of spinel inclusions in medium manganese steel with rare earth treatment. Metals 2019, 9, 804. [Google Scholar] [CrossRef]
- Liu, J.R.; Cui, H.; Wang, Z.D.; Fan, L.F. Evolution of Inclusions in Low-Alloy Wear-Resistant Steel with Ce-Ca Combined Treatment. Metall. Mater. Trans. B 2025. [Google Scholar] [CrossRef]
- Wang, L.C.; Tian, J.L.; Ren, J.; Jiang, C.G.; Jiang, Z.H.; You, Z.M. Effect of Mg–Ce Treatment on the Inclusion Evolution of Al Pre-deoxidized M50 Aerospace Bearing Steel During Vacuum Induction Melting Process. Metall. Mater. Trans. B 2025, 56, 3997–4009. [Google Scholar] [CrossRef]
- Liu, T.; Ma, H.; Liu, Y.; Li, D.Z. Effect of Aluminum-Oxygen (Al-O) Enrichment on Inclusion Evolution During Cerium Treatment. Metall. Mater. Trans. B 2025. [CrossRef]
- Kang, J.; Yu, Y.C.; Zhang, J.L.; Chen, C.; Wang, S.B. Effect of rare earth on inclusion evolution in industrial production of HRB500E steel. Metall. Res. Technol. 2021, 118, 220. [Google Scholar] [CrossRef]
- Zhao, L.; Yang, J.C.; Fu, X.Y. Effect of Ce content on modification behavior of inclusions and corrosion resistance of 316L stainless steel. Materials 2025, 18, 69. [Google Scholar] [CrossRef] [PubMed]
- Zhuo, C.; Liu, R.; Zhao, Z.R.; Zhang, Y.L.; Hao, X.S.; Wu, H.J.; Sun, Y.H. Effect of rare earth cerium content on manganese sulfide in U75V heavy rail steel. Metals 2022, 12, 1012. [Google Scholar] [CrossRef]
- Takashima, M.; Ono, S.M.; Nishimura, K. Low iron loss non-oriented electrical steel for high-efficiency Motor “RAM series”. Kawasaki Steel Gihou 1997, 29, 185. [Google Scholar]
- Kono, M.; Ozaki, Y.; Honda, A. Non-Oriented Silicon Steel Sheet Excellent in Magnetic Property after Stress Relieving Annealing and Its Production. Patent Abstracts of Japan JPH10212556A, 28 July 2003. [Google Scholar]
- Takashima, M.; Kono, M. Method for Producing Semiprocess Non-Oriented Magnetic Steel Sheet with Excellent Iron-Loss Characteristic. Patent Abstracts of Japan JP2006169577A, 29 June 2006. [Google Scholar]
- Li, X.Y.; Chen, C.; Wang, T.; Guo, Z.J.; Wang, H.Y.; Wang, H.Z.; Xue, L.Q.; Tian, J.; Sun, Y.H. Inclusions and phases in Fe-RE-Si steelmaking alloys and their effect on evolution of inclusions in non-oriented silicon steels. J. Rare Earths 2025. [Google Scholar] [CrossRef]
- Wang, H.J.; Niu, Y.; Ling, H.T.; Qiao, J.L.; Zhang, Y.L.; Zhong, W.; Qiu, S.T. Effects of rare earth La–Ce alloying treatment on modification of inclusions and magnetic properties of W350 non-oriented silicon steel. Metals 2023, 13, 626. [Google Scholar] [CrossRef]
- Li, N.; Wang, Y.Q.; Qiu, S.T.; Li, X. Effect of Ce on the evolution of recrystallization texture in a 1.2%Si-0.4%Al non-oriented electrical steel. ISIJ Int. 2016, 56, 1256–1261. [Google Scholar] [CrossRef]
- Song, C.; Xiang, L.; Shi, C.; Qiao, J.L.; Liu, J.F.; Qiu, S.T. Effect of rare earth La-Ce on solidification structure of 3.2%Si-0.9%Al non-oriented silicon steel. ISIJ Int. 2024, 64, 1347–5460. [Google Scholar] [CrossRef]
- Qin, J.; Zhao, H.B.; Wang, D.S.; Wang, S.L.; Yang, Y.W. Effect of Y on recrystallization behavior in non-oriented 4.5 wt% Si steel sheets. Materials 2022, 15, 4227. [Google Scholar] [CrossRef]
- Fan, L.F.; Zhu, R.; He, J.Z.; Lu, B. Effect of rare earth element La on texture and inclusion of non-oriented electrical steel produced by thin slab casting and rolling process. ISIJ Int. 2018, 58, 2348–2353. [Google Scholar] [CrossRef]
- Zhao, L.; Yang, J.C.; Hu, H.G.; Fu, X.Y. Effect of rare-earth Ce on the texture of non-oriented silicon steels. High Temp. Mater. Process. 2024, 43, 20220321. [Google Scholar] [CrossRef]
- Hou, C.K.; Liao, C.C. Effect of cerium content on the magnetic properties of non-oriented electrical steels. ISIJ Int. 2008, 48, 531–539. [Google Scholar] [CrossRef]
- Wan, Y.; Chen, W.Q.; Wu, S.J. Effect of lanthanum content on microstructure and magnetic properties of non-oriented electrical steels. J. Rare Earths 2013, 3, 727–733. [Google Scholar] [CrossRef]
- Wang, H.J.; Niu, Y.H.; Ling, H.T.; Qiao, J.L.; Zhang, Y.L.; Zhong, W.; Qiu, S.T. Modification of rare earth Ce on inclusions in W350 non-oriented silicon steel. Metals 2023, 13, 453. [Google Scholar] [CrossRef]
- Cui, L.X.; Liu, Y.B.; Ren, Q.; Hu, Z.Y.; Zhang, L.F. Effect of La-Ce mixed rare earth on inclusions in high-grade non-oriented silicon steel. Steelmaking 2025, 41, 53–64. [Google Scholar]
- Ma, F.X.; Gu, Q.; Liu, G.Q.; Zhang, Y.; Li, H.X. A systematic study of carbon-free oxide-based lining for preventing submerged entry nozzle clogging in continuous casting of rare earth steel. J. Iron Steel Res. Int. 2025, 32, 1584–1595. [Google Scholar] [CrossRef]





















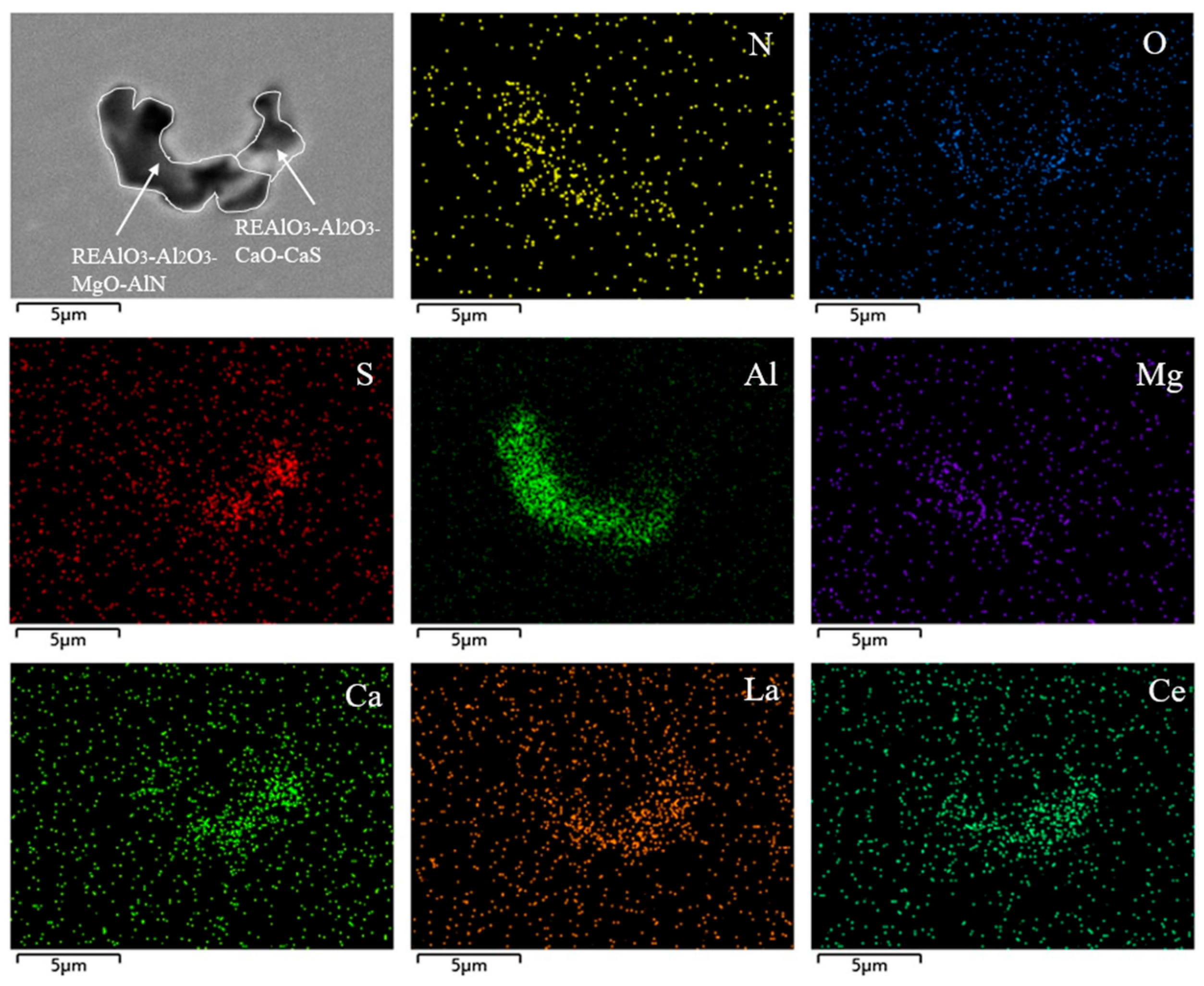


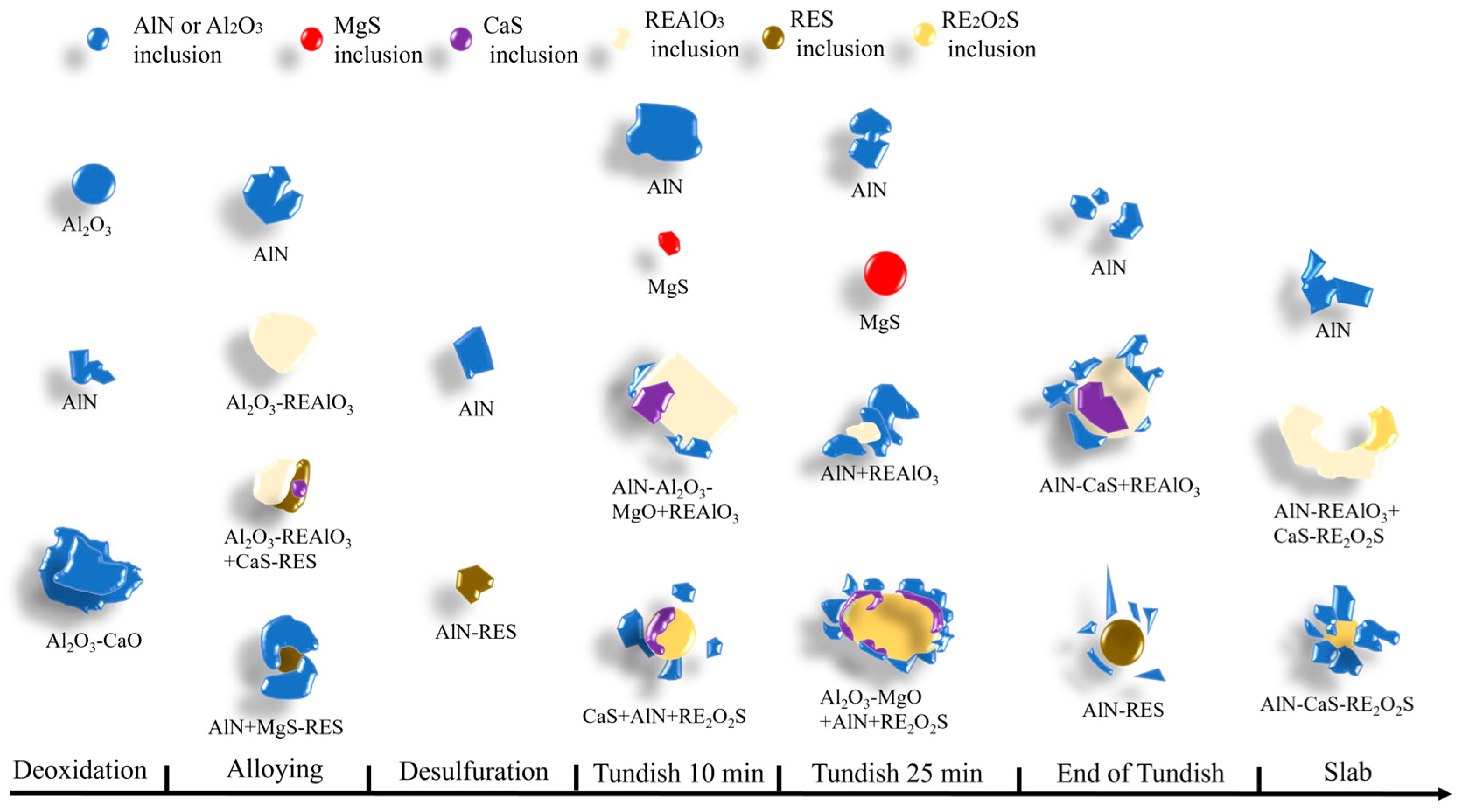
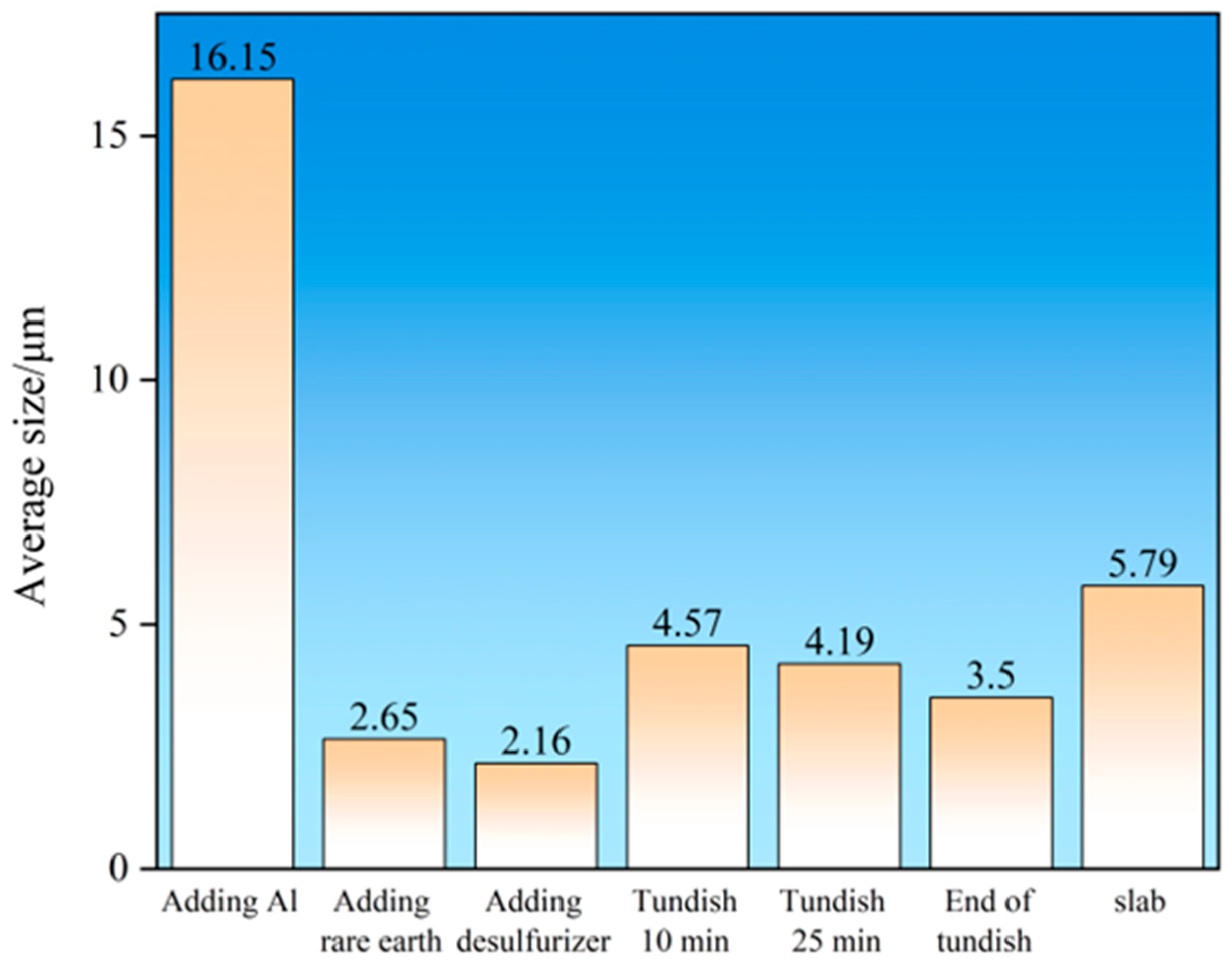
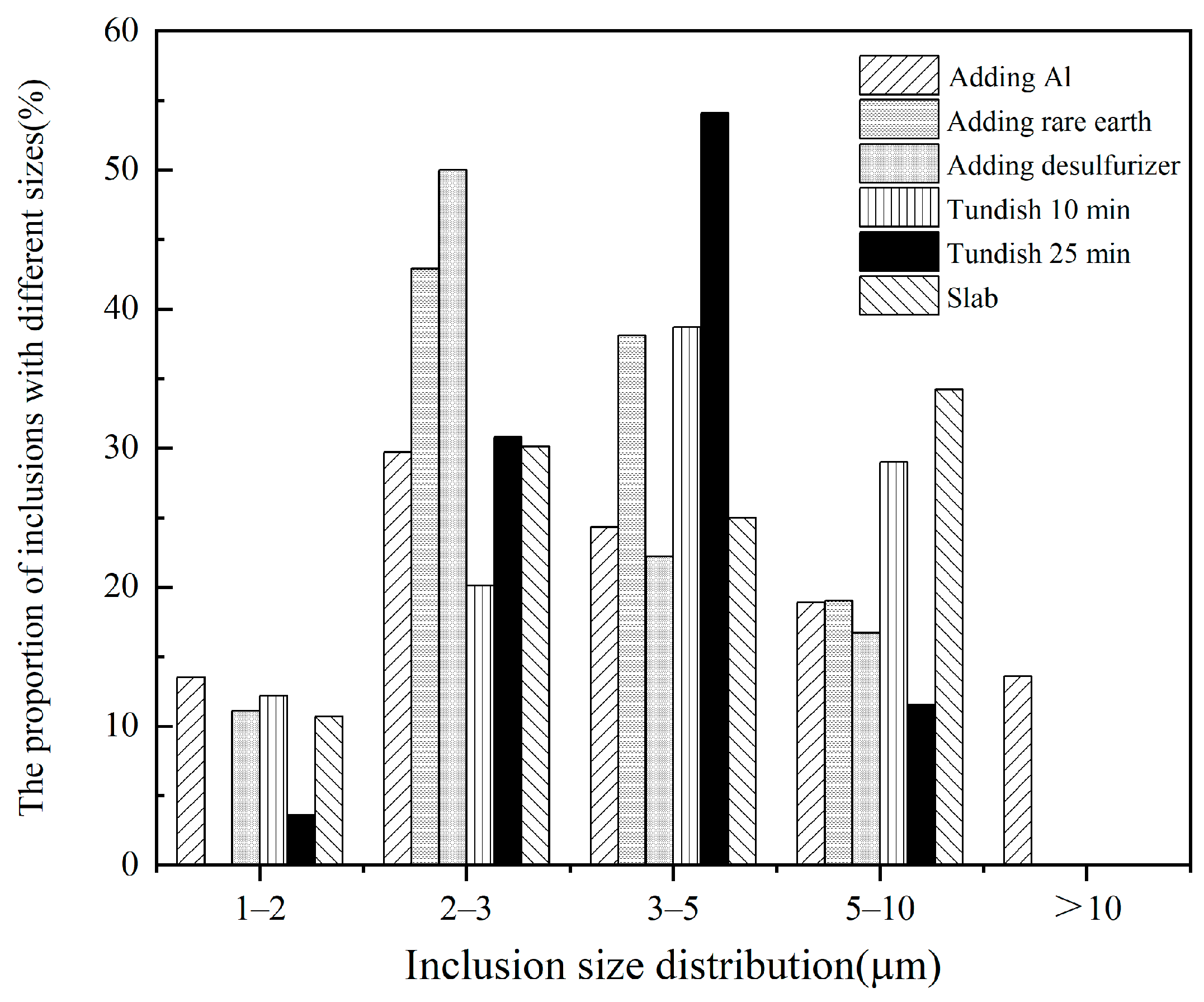
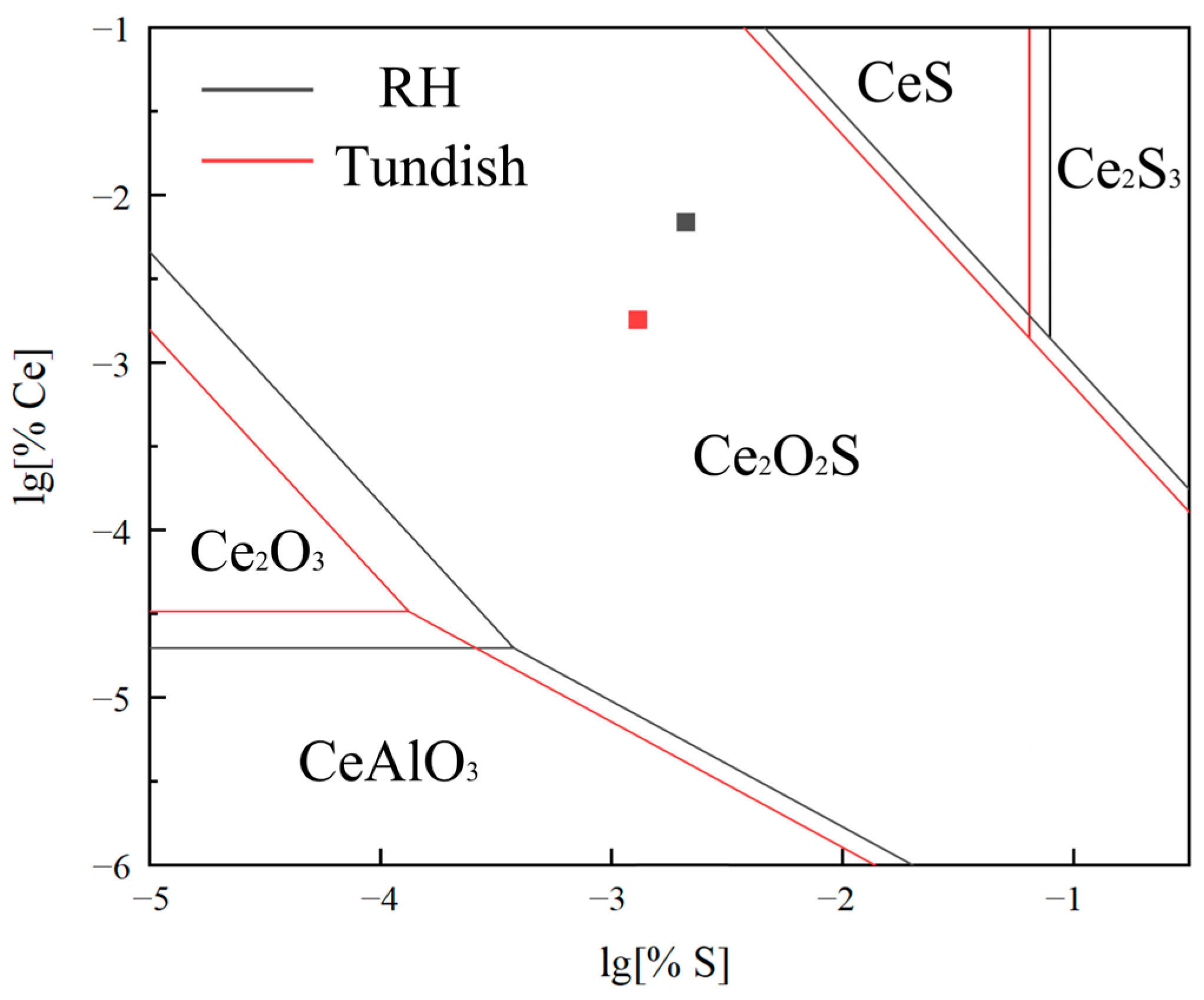
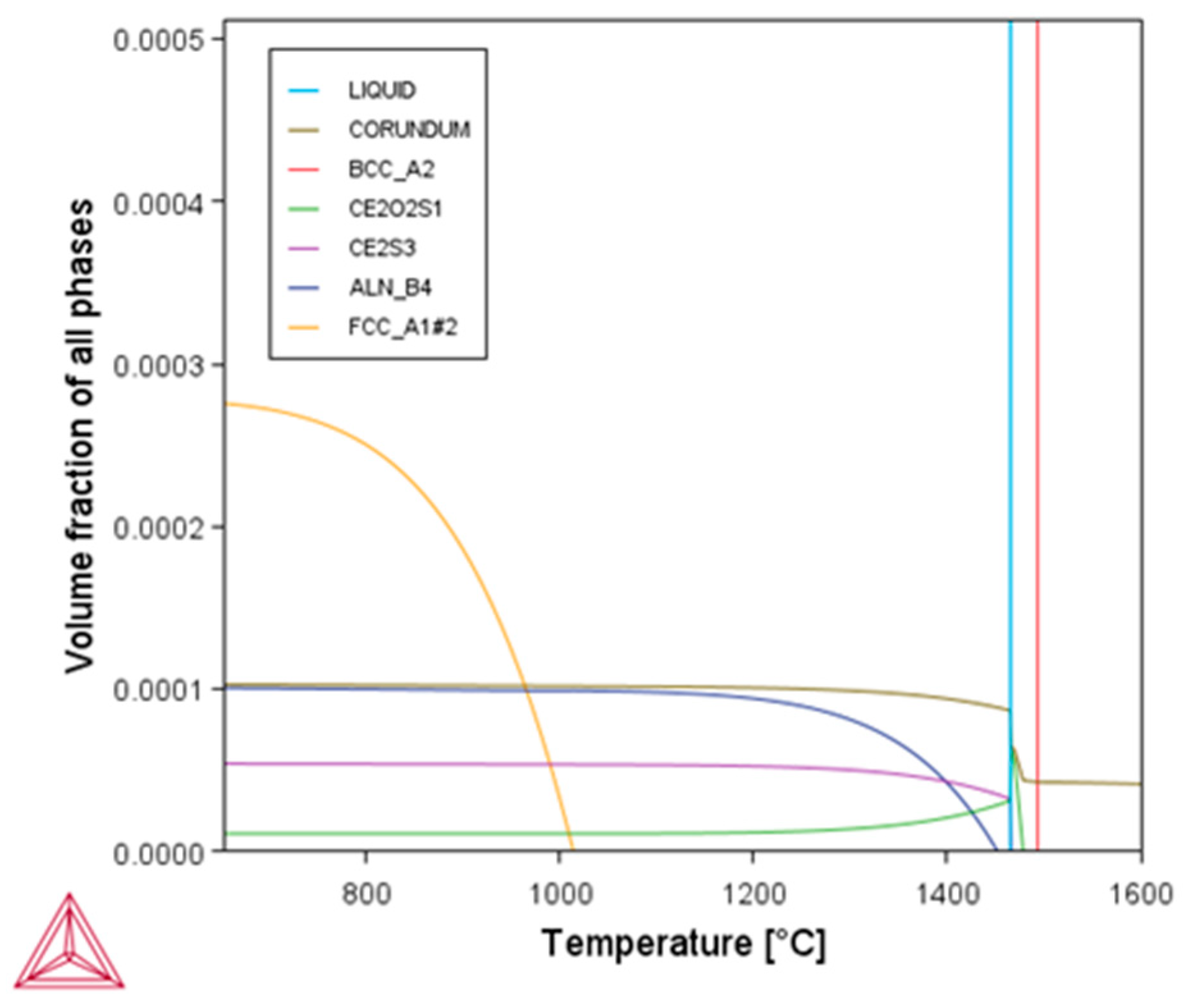
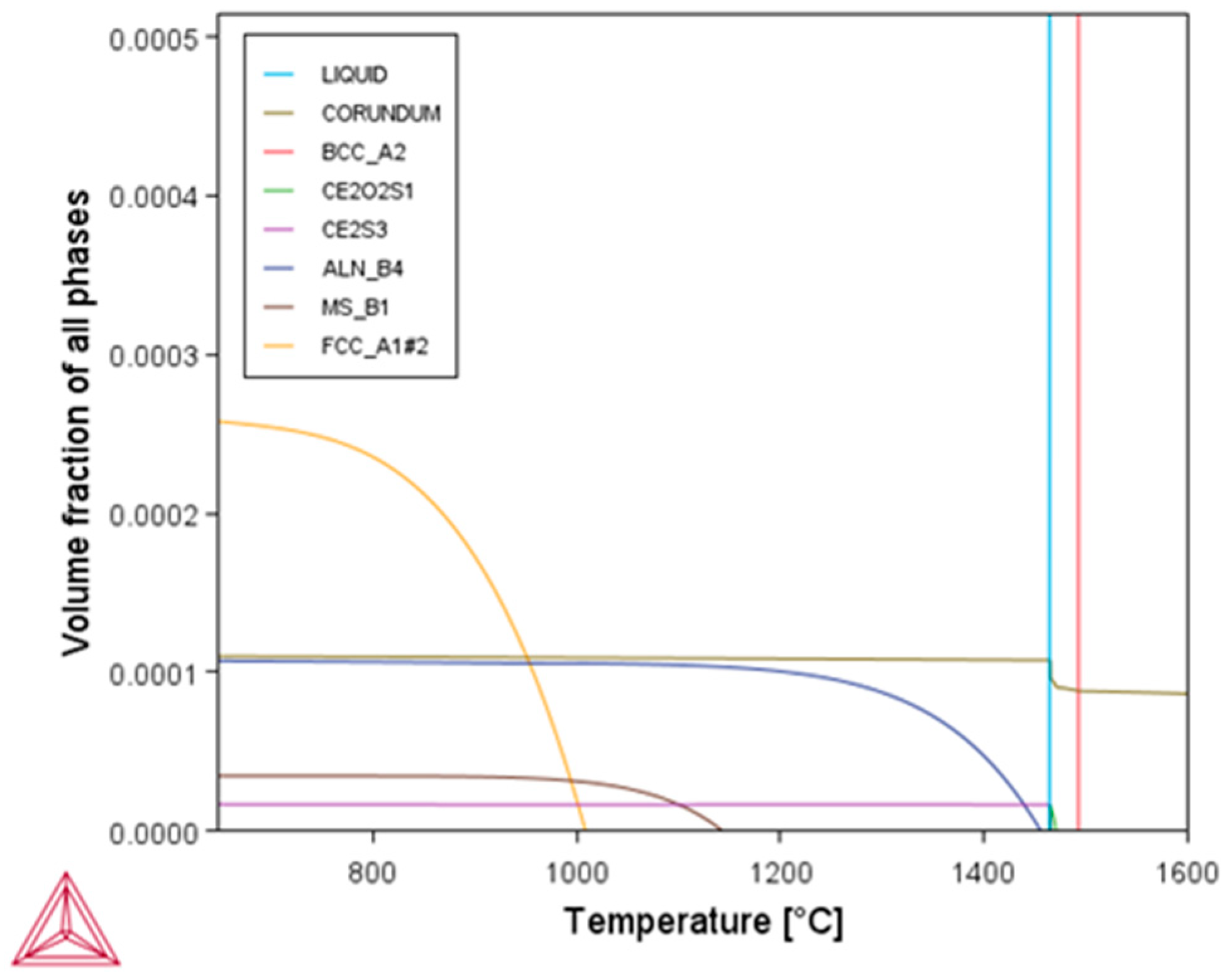
| Element | C | Si | Mn | P | S | Al | O | Ce | La | N | Cr |
|---|---|---|---|---|---|---|---|---|---|---|---|
| RH | 0.0032 | 3.144 | 0.281 | 0.0128 | 0.0021 | 0.8797 | 0.0025 | 0.0047 | 0.0022 | 0.0016 | 0.0147 |
| Tundish | 0.0029 | 3.114 | 0.281 | 0.0125 | 0.0013 | 0.8644 | 0.0007 | 0.0011 | 0.0005 | 0.0016 | 0.0147 |
| Rare Earth Inclusion | Gibbs Free Energy |
|---|---|
| Chemical Equation | RH | Tundish |
|---|---|---|
| CeAlO3-Ce2O2S | ||
| Ce2O2S-CeS | ||
| CeS-Ce2S3 | ||
| Ce2O2S-Ce2O3 | ||
| CeAlO3-Ce2O3 |
| Name | The Type of Rare Earth | The Content of Rare Earth (wt%) | Research Conclusions |
|---|---|---|---|
| Song, C. et al. [52] | La-Ce | 21, 34, 58 ppm | As the content of RE increases, the inclusions transform from Al2O3 to REAlO3 and RE2O2S, and the number density of inclusions first decreases and then increases with the increase of RE content. |
| Ren, Q. et al. [15] | SiMgCe | 23 ppm | Ce modifies MgO · Al2O3 inclusions into Ce2O2S inclusions and Ce-Al-Mg-Ca-S-O inclusions. As the Ce content in the molten steel decreases, the inclusions in the tundish are Ce2O2S and CeAlO3. |
| Wang, H.J. et al. [58] | La-Ce | 25 ppm | After the addition of rare earths, the main rare earth inclusions in the RH and tundish are REAlO3. Rare earths promote the agglomeration of inclusions, resulting in an increase in the number of inclusions with a size of 1.0~3.5 μm, and the average size of the inclusions is 2.66 μm. |
| Cui, L.X et al. [59] | La-Ce | 25 ppm | Rare earths slightly reduce the sulfur content and modify the original Al2O3·MgO inclusions into RES and RE2O2S. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xue, L.; Li, X.; Wang, T.; Zhao, Q.; Wang, H.; Wang, J.; Lin, W.; Niu, X.; Mu, W.; Chen, C. Analysis of Inclusions in the Entire Smelting Process of High-Grade Rare Earth Non-Oriented Silicon Steel. Crystals 2025, 15, 779. https://doi.org/10.3390/cryst15090779
Xue L, Li X, Wang T, Zhao Q, Wang H, Wang J, Lin W, Niu X, Mu W, Chen C. Analysis of Inclusions in the Entire Smelting Process of High-Grade Rare Earth Non-Oriented Silicon Steel. Crystals. 2025; 15(9):779. https://doi.org/10.3390/cryst15090779
Chicago/Turabian StyleXue, Liqiang, Xiangyu Li, Tao Wang, Qi Zhao, Haozheng Wang, Jia Wang, Wanming Lin, Xiaofeng Niu, Wangzhong Mu, and Chao Chen. 2025. "Analysis of Inclusions in the Entire Smelting Process of High-Grade Rare Earth Non-Oriented Silicon Steel" Crystals 15, no. 9: 779. https://doi.org/10.3390/cryst15090779
APA StyleXue, L., Li, X., Wang, T., Zhao, Q., Wang, H., Wang, J., Lin, W., Niu, X., Mu, W., & Chen, C. (2025). Analysis of Inclusions in the Entire Smelting Process of High-Grade Rare Earth Non-Oriented Silicon Steel. Crystals, 15(9), 779. https://doi.org/10.3390/cryst15090779








