Selective Hydrogenation of 2-Methyl-3-butyn-2-ol in Microcapillary Reactor on Supported Intermetallic PdZn Catalyst, Effect of Support Doping on Stability and Kinetic Parameters
Abstract
1. Introduction
2. Results and Discussion
2.1. Structural and Composition Analysis of Catalysts and Coatings
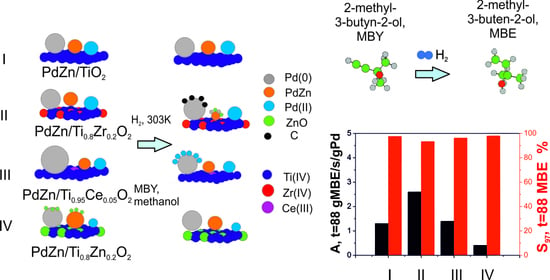
2.2. Hydrogenation of 2-Methyl-3-butyn-2-ol on PdZn/TixM1−xOy Coatings in a Microcapillary Reactor
2.3. Hydrogenation of 2-Methyl-3-butyn-2-ol on PdZn/TixM1−xOy Coatings after Different Pretreatments
3. Materials and Methods
3.1. Synthesis of PdZn/TixM1−xO2 (M = Zr, Ce, Zn) Coatings
3.2. Investigation of the Physicochemical Properties of TixM1−xO2 Composites (M = Zr, Ce, Zn) and PdZn/Tix Zr1−xO2 Catalysts and Coatings
3.3. Catalytic Tests
3.4. Calculations of Kinetic Parameters
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Renken, A.; Kiwi-Minsker, L. Microstructured Catalytic Reactors. Adv. Catal. 2010, 53, 47–122. [Google Scholar] [CrossRef]
- Fanelli, F.; Parisi, G.; Degennaro, L.; Luisi, R. Contribution of Microreactor Technology and Flow Chemistry to the Development of Green and Sustainable Synthesis. Beilstein J. Org. Chem. 2017, 13, 520–542. [Google Scholar] [CrossRef] [PubMed]
- Rebrov, E.V.; Berenguer-Murcia, A.; Skelton, H.E.; Johnson, B.F.G.; Wheatley, A.E.H.; Schouten, J.C. Capillary Microreactors Wall-Coated with Mesoporous Titania Thin Film Catalyst Supports. Lab Chip 2009, 9, 503–506. [Google Scholar] [CrossRef]
- Rebrov, E.V.; Klinger, E.A.; Berenguer-Murcia, A.; Sulman, E.M.; Schouten, J.C. Selective Hydrogenation of 2-Methyl-3-Butyne-2-Ol in a Wall-Coated Capillary Microreactor with a Pd25Zn75/TiO2 Catalyst. Org. Process Res. Dev. 2009, 13, 991–998. [Google Scholar] [CrossRef]
- Cherkasov, N.; Ibhadon, A.O.; Rebrov, E.V. Novel Synthesis of Thick Wall Coatings of Titania Supported Bi Poisoned Pd Catalysts and Application in Selective Hydrogenation of Acetylene Alcohols in Capillary Microreactors. Lab Chip 2015, 15, 1952–1960. [Google Scholar] [CrossRef]
- Okhlopkova, L.B.; Kerzhentsev, M.A.; Ismagilov, Z.R. Capillary Microreactor with a Catalytic Coating Based on Mesoporous Titanium Dioxide for the Selective Hydrogenation of 2-Methyl-3-Butyn-2-Ol. Kinet. Catal. 2016, 57, 501–507. [Google Scholar] [CrossRef]
- Okhlopkova, L.B.; Cherepanova, S.V.; Prosvirin, I.P.; Kerzhentsev, M.A.; Ismagilov, Z.R. Semi-Hydrogenation of 2-Methyl-3-Butyn-2-Ol on Pd-Zn Nanoalloys Prepared by Polyol Method: Effect of Composition and Heterogenization. Appl. Catal. A 2018, 549, 245–253. [Google Scholar] [CrossRef]
- Mashkovsky, I.S.; Markov, P.V.; Bragina, G.O.; Baeva, G.N.; Bukhtiyarov, A.V.; Prosvirin, I.P.; Bukhtiyarov, V.I.; Yu Stakheev, A. Formation of Supported Intermetallic Nanoparticles in the Pd–Zn/α-Al2O3 Catalyst. Kinet. Catal. 2017, 58, 499–507. [Google Scholar] [CrossRef]
- Markov, P.V.; Bragina, G.O.; Rassolov, A.V.; Baeva, G.N.; Mashkovsky, I.S.; Murzin, V.Y.; Zubavichus, Y.V.; Stakheev, A.Y. Pd–Cu Catalyst Prepared from Heterobimetallic PdCu2(OAc)6: An XRD-EXAFS Study and Activity/Selectivity in the Liquid-Phase Hydrogenation of a C≡C Bond. Mendeleev Commun. 2016, 26, 502–504. [Google Scholar] [CrossRef]
- Markov, P.V.; Bragina, G.O.; Rassolov, A.V.; Mashkovsky, I.S.; Baeva, G.N.; Tkachenko, O.P.; Yakushev, I.A.; Vargaftik, M.N.; Stakheev, A.Y. Performance of a Bimetallic Pd–In Catalyst in the Selective Liquid-Phase Hydrogenation of Internal and Terminal Alkynes. Mendeleev Commun. 2016, 26, 494–496. [Google Scholar] [CrossRef]
- Khan, N.A.; Shaikhutdinov, S.; Freund, H.J. Acetylene and Ethylene Hydrogenation on Alumina Supported Pd-Ag Model Catalysts. Catal. Letters 2006, 108, 159–164. [Google Scholar] [CrossRef]
- Johnston, S.K.; Cherkasov, N.; Pérez-Barrado, E.; Aho, A.; Murzin, D.Y.; Ibhadon, A.O.; Francesconi, M.G. Pd 3 Sn Nanoparticles on TiO 2 and ZnO Supports as Catalysts for Semi-Hydrogenation: Synthesis and Catalytic Performance. Appl. Catal. A Gen. 2017, 544, 40–45. [Google Scholar] [CrossRef]
- Niu, W.; Gao, Y.; Zhang, W.; Yan, N.; Lu, X. Pd-Pb Alloy Nanocrystals with Tailored Composition for Semihydrogenation: Taking Advantage of Catalyst Poisoning. Angew. Chemie - Int. Ed. 2015, 54, 8271–8274. [Google Scholar] [CrossRef]
- Crespo-Quesada, M.; Yarulin, A.; Jin, M.; Xia, Y.; Kiwi-Minsker, L. Structure Sensitivity of Alkynol Hydrogenation on Shape- and Size-Controlled Palladium Nanocrystals: Which Sites Are Most Active and Selective? J. Am. Chem. Soc. 2011, 133, 12787–12794. [Google Scholar] [CrossRef]
- Yarulin, A.E.; Crespo-Quesada, R.M.; Egorova, E.V.; Kiwi-Minsker, L.L. Structure Sensitivity of Selective Acetylene Hydrogenation over the Catalysts with Shape-Controlled Palladium Nanoparticles. Kinet. Catal. 2012, 53, 253–261. [Google Scholar] [CrossRef]
- Protasova, L.N.; Rebrov, E.V.; Glazneva, T.S.; Berenguer-Murcia, A.; Ismagilov, Z.R.; Schouten, J.C. Control of the Thickness of Mesoporous Titania Films for Application in Multiphase Catalytic Microreactors. J. Catal. 2010, 271, 161–169. [Google Scholar] [CrossRef]
- Muraza, O.; Rebrov, E.V.; de Croon, M.H.J.M.; Schouten, J.C. Enhancement of the Stability of Microporous Silica Films in Non-Aqueous Solvents at Elevated Temperature. Microporous Mesoporous Mater. 2009, 124, 20–29. [Google Scholar] [CrossRef]
- Muraza, O.; Rebrov, E.V.; Berenguer-Murcia, A.; de Croon, M.H.J.M.; Schouten, J.C. Selectivity Control in Hydrogenation Reactions by Nanoconfinement of Polymetallic Nanoparticles in Mesoporous Thin Films. Appl. Catal. A Gen. 2009, 368, 87–96. [Google Scholar] [CrossRef]
- Protasova, L.N.; Rebrov, E.V.; Skelton, H.E.; Wheatley, A.E.H.; Schouten, J.C. A Kinetic Study of the Liquid-Phase Hydrogenation of Citral on Au/TiO2 and Pt–Sn/TiO2 Thin Films in Capillary Microreactors. Appl. Catal. A Gen. 2011, 399, 12–21. [Google Scholar] [CrossRef]
- Okhlopkova, L.B.; Kerzhentsev, M.A.; Ismagilov, Z.R. Improved Thermal Stability of PdZn/TiO2coating by Ce Doping. Surf. Eng. 2015, 31, 78–83. [Google Scholar] [CrossRef]
- Okhlopkova, L.B.; Kerzhentsev, M.A.; Ismagilov, Z.R. Development, Synthesis, and Study of Sanomaterials of Titania Doped by Zirconium for Selective Hydrogenation of 2-Methyl-3-Butyn-2-Ol in a Microcapillary Reactor. Kinet. Catal. 2019, 60, 474–483. [Google Scholar] [CrossRef]
- Ye, C.; Chen, X.; Li, S.; Feng, B.; Fu, Y.; Zhang, F.; Chen, D.L.; Zhu, W. PdZn Intermetallic Compound Stabilized on ZnO/Nitrogen-Decorated Carbon Hollow Spheres for Catalytic Semihydrogenation of Alkynols. Nano Res. 2022, 15, 3090–3098. [Google Scholar] [CrossRef]
- Vernuccio, S.; Goy, R.; Rudolf Von Rohr, P.; Medlock, J.; Bonrath, W. Hydrogenation of 2-Methyl-3-Butyn-2-Ol over a Pd/ZnO Catalyst: Kinetic Model and Selectivity Study. React. Chem. Eng. 2016, 1, 445–453. [Google Scholar] [CrossRef]
- Vernuccio, S.; Goy, R.; Meier, A.; Rudolf von Rohr, P.; Medlock, J. Kinetics and Mass Transfer of the Hydrogenation of 2-Methyl-3-Butyn-2-Ol in a Structured Pd/ZnO/Al2O3 Reactor. Chem. Eng. J. 2017. [Google Scholar] [CrossRef]
- Chen, X.; Shi, C.; Wang, X.B.; Li, W.Y.; Liang, C. Intermetallic PdZn Nanoparticles Catalyze the Continuous-Flow Hydrogenation of Alkynols to Cis-Enols. Commun. Chem. 2021, 4, 175. [Google Scholar] [CrossRef]
- González-Fernández, A.; Pischetola, C.; Kiwi-Minsker, L.; Cárdenas-Lizana, F. Partial Hydrogenation of 2-Methyl-3-Butyn-2-Ol over Pd/ZnO: Effect of Reduction Temperature on Alloy Formation and Catalytic Response. J. Phys. Chem. C 2020, 124, 3681–3691. [Google Scholar] [CrossRef]
- Tew, M.W.; Emerich, H.; Van Bokhoven, J.A. Formation and Characterization of PdZn Alloy: A Very Selective Catalyst for Alkyne Semihydrogenation. J. Phys. Chem. C 2011, 115, 8457–8465. [Google Scholar] [CrossRef]
- Cherkasov, N.; Denissenko, P.; Deshmukh, S.; Rebrov, E.V. Gas-Liquid Hydrogenation in Continuous Flow – The Effect of Mass Transfer and Residence Time in Powder Packed-Bed and Catalyst-Coated Reactors. Chem. Eng. J. 2020, 379, 122292. [Google Scholar] [CrossRef]
- Asano, S.; Adams, S.J.; Tsuji, Y.; Yoshizawa, K.; Tahara, A.; Hayashi, J.I.; Cherkasov, N. Homogeneous Catalyst Modifier for Alkyne Semi-Hydrogenation: Systematic Screening in an Automated Flow Reactor and Computational Study on Mechanisms. React. Chem. Eng. 2022, 7, 1818–1826. [Google Scholar] [CrossRef]
- Cherkasov, N.; Bai, Y.; Rebrov, E. Process Intensification of Alkynol Semihydrogenation in a Tube Reactor Coated with a Pd/ZnO Catalyst. Catalyst. 2017, 7, 358. [Google Scholar] [CrossRef]
- Cherkasov, N.; Al-Rawashdeh, M.; Ibhadon, A.O.; Rebrov, E.V. Scale up Study of Capillary Microreactors in Solvent-Free Semihydrogenation of 2-Methyl-3-Butyn-2-Ol. Catal. Today 2016, 273, 205–212. [Google Scholar] [CrossRef]
- Semagina, N.; Grasemann, M.; Xanthopoulos, N.; Renken, A.; Kiwi-Minsker, L. Structured Catalyst of Pd/ZnO on Sintered Metal Fibers for 2-Methyl-3-Butyn-2-Ol Selective Hydrogenation. J. Catal. 2007, 251, 213–222. [Google Scholar] [CrossRef]
- Okhlopkova, L.B.; Kerzhentsev, M.A.; Ismagilov, Z.R. Coating the Internal Surface of a Capillary Microreactor for the Selective Hydrogenation of 2-Methyl-3-Butyn-2-Ol by PdxZn1 – x/TiO2 Catalysts. A Kinetic Study. Kinet. Catal. 2018, 59, 450–458. [Google Scholar] [CrossRef]
- Okhlopkova, L.B.; Kerzhentsev, M.A.; Tuzikov, F.V.; Larichev, Y.V.; Ismagilov, Z.R. Palladium-Zinc Catalysts on Mesoporous Titania Prepared by Colloid Synthesis. II. Synthesis and Characterization of PdZn/TiO2 Coating on Inner Surface of Fused Silica Capillary. J. Nanoparticle Res. 2012, 14, 1088. [Google Scholar] [CrossRef]
- Carter, J.H.; Shah, P.M.; Nowicka, E.; Freakley, S.J.; Morgan, D.J.; Golunski, S.; Hutchings, G.J. Enhanced Activity and Stability of Gold/Ceria-Titania for the Low-Temperature Water-Gas Shift Reaction. Front. Chem. 2019, 7, 443. [Google Scholar] [CrossRef] [PubMed]
- Cao, Y.; Ran, R.; Wu, X.; Si, Z.; Kang, F.; Weng, D. Progress on Metal-Support Interactions in Pd-Based Catalysts for Automobile Emission Control. J. Environ. Sci. (China) 2023, 125, 401–426. [Google Scholar] [CrossRef]
- Wessels, K.; Minnermann, M.; Rathousky, J.; Wark, M.; Oekermann, T. Influence of Calcination Temperature on the Photoelectrochemical and Photocatarytic Properties of Porous TiO2 Films Electrodeposited from Ti(IV)-Alkoxide Solution. J. Phys. Chem. C 2008, 112, 15122–15128. [Google Scholar] [CrossRef]
- Juliet, S.S.; Ramalingom, S.; Ravidhas, C.; Raj, A.M.E. Effect of Calcination Temperature on Titanium Oxide Nanocrystallites in the Anatase Phase Synthesized By Sol-Gel Route. IOSR J. Appl. Phys. 2017, 9, 32–39. [Google Scholar] [CrossRef]
- Nikoshvili, L.Z.; Makarova, A.S.; Lyubimova, N.A.; Bykov, A.V.; Sidorov, A.I.; Tyamina, I.Y.; Matveeva, V.G.; Sulman, E.M. Kinetic Study of Selective Hydrogenation of 2-Methyl-3-Butyn-2-Ol over Pd-Containing Hypercrosslinked Polystyrene. Catal. Today 2015, 256, 231–240. [Google Scholar] [CrossRef]
- Okhlopkova, L.B.; Prosvirin, I.P.; Kerzhentsev, M.A.; Ismagilov, Z.R. Capillary Microreactor with PdZn/(Ti, Ce)O2 Coating for Selective Hydrogenation of 2-Methyl-3-Butyn-2-Ol. Chem. Eng. Process. - Process Intensif. 2021, 159, 108240. [Google Scholar] [CrossRef]
- Okhlopkova, L.B.; Kerzhentsev, M.A.; Ismagilov, Z.R. Internal Surface Coating of a Capillary Microreactor for the Selective Hydrogenation of 2-Methyl-3-Butyn-2-Ol Using a PdZn/TiO2 Catalyst. The Effect of the Catalyst’s Activation Conditions on Its Catalytic Properties. Kinet. Catal. 2018, 59, 347–356. [Google Scholar] [CrossRef]
- Naganuma, T.; Traversa, E. Stability of the Ce 3+ Valence State in Cerium Oxide Nanoparticle Layers. Nanoscale 2012, 4, 4950–4953. [Google Scholar] [CrossRef] [PubMed]
- Rameshan, C.; Stadlmayr, W.; Weilach, C.; Penner, S.; Lorenz, H.; Hävecker, M.; Blume, R.; Rocha, T.; Teschner, D.; Knop-Gericke, A.; et al. Subsurface-Controlled CO2 Selectivity of PdZn near-Surface Alloys in H2 Generation by Methanol Steam Reforming. Angew. Chemie - Int. Ed. 2010, 49, 3224–3227. [Google Scholar] [CrossRef] [PubMed]
- Engels, V.; Jefferson, D.; Benaskar, F.; Thune, P.; Berenguer-Murcia, A.; Johnson, B.; Wheatley, A. Nanoparticulate PdZn - Pathways towards the Synthetic Control of Nanosurface Properties. Nanotechnology 2011, 22, 205701. [Google Scholar] [CrossRef]
- Holzapfel, H.H.; Wolfbeisser, A.; Rameshan, C.; Weilach, C.; Rupprechter, G. PdZn Surface Alloys as Models of Methanol Steam Reforming Catalysts: Molecular Studies by LEED, XPS, TPD and PM-IRAS. Top. Catal. 2014, 57, 1218–1228. [Google Scholar] [CrossRef]
- Doyle, A.M.; Shaikhutdinov, S.K.; Jackson, S.D.; Freund, H.J. Hydrogenation on Metal Surfaces: Why Are Nanoparticles More Active than Single Crystals? Angew. Chemie Int. Ed. 2003, 42, 5240–5243. [Google Scholar] [CrossRef]
- Crespo-Quesada, M.; Andanson, J.M.; Yarulin, A.; Lim, B.; Xia, Y.; Kiwi-Minsker, L. UV-Ozone Cleaning of Supported Poly(Vinylpyrrolidone)-Stabilized Palladium Nanocubes: Effect of Stabilizer Removal on Morphology and Catalytic Behavior. Langmuir 2011, 27, 7909–7916. [Google Scholar] [CrossRef]
- Okhlopkova, L.B.; Matus, E.V.; Prosvirin, I.P.; Kerzhentsev, M.A.; Ismagilov, Z.R. Selective Hydrogenation of 2-Methyl-3-Butyn-2-Ol Catalyzed by Embedded Polymer-Protected PdZn Nanoparticles. J. Nanoparticle Res. 2015, 17, 475. [Google Scholar] [CrossRef]
- Wowsnick, G.; Teschner, D.; Armbrüster, M.; Kasatkin, I.; Girgsdies, F.; Grin, Y.; Schlögl, R.; Behrens, M. Surface Dynamics of the Intermetallic Catalyst Pd2Ga, Part II - Reactivity and Stability in Liquid-Phase Hydrogenation of Phenylacetylene. J. Catal. 2014, 309, 221–230. [Google Scholar] [CrossRef]
- Shen, L.; Mao, S.; Li, J.; Li, M.; Chen, P.; Li, H.; Chen, Z.; Wang, Y. PdZn Intermetallic on a CN@ZnO Hybrid as an Efficient Catalyst for the Semihydrogenation of Alkynols. J. Catal. 2017, 350, 13–20. [Google Scholar] [CrossRef]
- Armbruster, M.; Behrens, M.; Cinquini, F.; Fottinger, K.; Grin, Y.; Haghofer, A.; Klotzer, B.; Knop-Gericke, A.; Lorenz, H.; Ota, A.; et al. How to Control the Selectivity of Palladium-Based Catalysts in Hydrogenation Reactions: The Role of Subsurface Chemistry. ChemCatChem 2012, 4, 1048–1063. [Google Scholar] [CrossRef]
- Jackson, S.D.; Shaw, L.A. The Liquid-Phase Hydrogenation of Phenyl Acetylene and Styrene on a Salladium /Carbon Catalyst. Appl. Catal. A, Gen. 1996, 134, 91–99. [Google Scholar] [CrossRef]
- Jackson, S.D.; Casey, N.J.; Huntington, I.J.; Ts, C. Characterization of the Carboneceous Deposit Formed during the Selective Hydrogenation of Propyne over Palladium/Zirconia. Stud. Surf. Sci. Catal. 1994, 88, 313–318. [Google Scholar] [CrossRef]
- Dominguez-Dominguez, S.; Berenguer-Murcia, A.; Cazorla-Amoros, D.; Linares-Solano, A. Semihydrogenation of Phenylacetylene Catalyzed by Metallic Nanoparticles Containing Noble Metals. J. Catal. 2006, 243, 74–81. [Google Scholar] [CrossRef]
- AGILENT. Available online: https://www.agilent.com/search/?Ntt=160-2530-10 (accessed on 15 January 2020).
- Scofield, J.H. Hartree-Slater Subshell Photoionization Cross-Sections at 1254 and 1487 EV. J. Electron Spectros. Relat. Phenomena 1976, 8, 129–137. [Google Scholar] [CrossRef]
- Semagina, N.; Joannet, E.; Parra, S.; Sulman, E.; Renken, A.; Kiwi-Minsker, L. Palladium Nanoparticles Stabilized in Block-Copolymer Micelles for Highly Selective 2-Butyne-1,4-Diol Partial Hydrogenation. Appl. Catal. A Gen. 2005, 280, 141–147. [Google Scholar] [CrossRef]
- Kashid, M.N.; Renken, A.; Kiwi-Minsker, L. (Eds.) Microstructured Devices for Chemical Processing; Wiley-VCH Verlag GmbH & Co. KGaA: Weinheim, Germany, 2014; ISBN 9783527685226. [Google Scholar]
- Duca, D.; Liotta, L.F.; Deganello, G. Selective Hydrogenation of Phenylacetylene on Pumice-Supported Palladium Catalysts. J. Catal. 1995, 154, 69–79. [Google Scholar] [CrossRef]
- Cherkasov, N.; Ibhadon, A.O.; McCue, A.J.; Anderson, J.A.; Johnston, S.K. Palladium-Bismuth Intermetallic and Surface-Poisoned Catalysts for the Semi-Hydrogenation of 2-Methyl-3-Butyn-2-Ol. Appl. Catal. A Gen. 2015, 497, 22–30. [Google Scholar] [CrossRef]
- Singh, U.K.; Albert Vannice, M. Liquid-Phase Hydrogenation of Citral over Pt/SiO2 Catalysts: I. Temperature Effects on Activity and Selectivity. J. Catal. 2000, 191, 165–180. [Google Scholar] [CrossRef]









| Supports | Calcination Temperature, K | Specific Surface Area, m2/g | Pore Size, nm | Pore Volume, cm3/g | Crystallite Size, nm |
|---|---|---|---|---|---|
| TiO2 | 673 | 151.8 | 3.8 | 0.132 | 6 |
| TiO2 | 873 | 23.4 | 10.2 | 0.061 | 22 |
| Ti0.95Ce0.05O2 | 673 | 261.4 | 4.0 | 0.22 | amorphous |
| Ti0.95Ce0.05O2 | 873 | 57.5 | 8.9 | 0.144 | 9 |
| Ti0.80Zr0.20O2 Ti0.80Zr0.20O2 | 673 | 185.5 | 3.6 | 0.084 | 9 |
| 873 | 128.6 | 5.9 | 0.236 | 15 | |
| Ti0.80Zn0.20O1.8 Ti0.80Zn0.20O1.8 | 673 | 250.9 | 4.2 | 0.253 | amorphous |
| 873 | 51 | 13.5 | 0.217 | 25 |
| Sample | MBY Concentration, t=28, mol/L | Q, t = 28, gMBE/day | A, t = 28, gMBE/s/gPd | S97, t = 28 % | MBY Concentration, t = 88, mol/L | Q, t = 88, gMBE/day | A, t = 88, gMBE/s/gPd | S97, t = 88 % |
|---|---|---|---|---|---|---|---|---|
| PdZn/TiO2 | 0.8 | 3.6 | 1.5 | 96.7 | 2.2 | 4.1 | 1.3 | 97.6 |
| PdZn/Ti0.8Zr0.2O2 | 1.0 | 6.3 | 1.4 | 96.8 | 2.0 | 12.0 | 2.6 | 93.4 |
| PdZn/Ti0.95Ce0.05O2 | 1.0 | 3.6 | 1.8 | 93.7 | 2.0 | 2.9 | 1.4 | 96.3 |
| PdZn/Ti0.8Zn0.2O2 | 0.4 | 0.43 | 0.12 | 97.6 | 1.0 | 1.56 | 0.41 | 98.0 |
| Parameter | PdZn/TiO2 | PdZn/Ti0.8Zr0.2O2 | PdZn/Ti0.95Ce0.05O2.08 | PdZn/Ti0.8Zn0.2O1.8 |
|---|---|---|---|---|
| k1’/mol/L/s/gPd | 714 | 788 | 1258 | 186 |
| k2’mol/L/s/gPd | 132 | 176 | 941 | 14 |
| k3’/mol/L/s/gPd | 20 | 41 | 0.0001 | 5 |
| KMBY/L/mol | 61 | 43 | 30 | 56 |
| KMBE/L/mol | 0.8 | 0.6 | 1 | 0.3 |
| KMBA/L/mol | 8 | 0.01 | 0.001 | 1 |
| KMBE/KMBY | 0.013 | 0.014 | 0.031 | 0.005 |
| KMBA/KMBE | 10 | 0.017 | 0.001 | 3.3 |
| KMBA/KMBY | 0.13 | 2.3 × 10−4 | 3.3 × 10−5 | 0.02 |
| σMBY, % | 25.0 | 7.5 | 24.0 | 3.3 |
| σMBE, % | 5.3 | 3.7 | 3.9 | 0.8 |
| σMBA, % | 13.0 | 8.0 | 11.6 | 20.7 |
| Q, gMBE/day | 3.6 | 6.3 | 3.6 | 0.42 |
| S97,% | 96.7 | 96.8 | 93.7 | 97.5 |
| Catalyst | Pretreatment | Pd3d5/2 | Zn2p3/2 | Ce3d5/2 | Pd/Ti | Pd/Zn | At. Conc. C, % | At. Conc. Zn,% |
|---|---|---|---|---|---|---|---|---|
| PdZn/TiO2 | H2/Ar, 573 K, 2 h | 335.1 | 1022 | - | 0.005 | 0.17 | 52.4 | 0.31 |
| PdZn/TiO2 | Air, 573 K, 2 h, H2/Ar, 573 K, 2 h | 335.3 | 1022.3 | - | 0.006 | 0.21 | 26.1 | 0.53 |
| PdZn/Ti0.95Ce0.05O2 | H2/Ar, 573 K, 2 h | 335.1 | 1022 | 882.3 | 0.006 | 2.0 | 50.9 | 0.04 |
| PdZn/Ti0.95Ce0.05O2 | Air, 573 K, 2 h, H2/Ar, 573 K, 2 h | 335.3 | 1022.2 | 882.2 | 0.009 | 2.3 | 32.1 | 0.07 |
| PdZn/Ti0.8Zn0.2O1.8 | H2/Ar, 573 K, 2 h | 335.1 | 1022.3 | - | 0.003 | 0.005 | 43.3 | 5.84 |
| PdZn/Ti0.8Zn0.2O1.8 | Air, 573 K, 2 h, H2/Ar, 573 K, 2 h | 335.2 | 1022.3 | - | 0.004 | 0.007 | 26.3 | 7.2 |
| Catalyst | Pretreatment | Pd(0) (%) | PdZn (%) | Pd(II) (%) |
|---|---|---|---|---|
| PdZn/TiO2 | H2/Ar, 573 K, 2 h | 48.9 | 23.6 | 27.5 |
| PdZn/TiO2 | Air, 573 K, 2 h, H2/Ar, 573 K, 2 h | 50.5 | 27.6 | 21.9 |
| PdZn/Ti0.95Ce0.05O2 | H2/Ar, 573 K, 2 h | 60.3 | 19.5 | 20.2 |
| PdZn/Ti0.95Ce0.05O2 | Air, 573 K, 2 h, H2/Ar, 573 K, 2 h | 46.1 | 18.1 | 35.8 |
| PdZn/Ti0.8Zn0.2O1.8 | H2/Ar, 573 K, 2 h | 52.6 | 36.0 | 11.4 |
| PdZn/Ti0.8Zn0.2O1.8 | Air, 573 K, 2 h, H2/Ar, 573 K, 2 h | 58.1 | 31.8 | 10.1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Okhlopkova, L.; Prosvirin, I.; Kerzhentsev, M.; Ismagilov, Z. Selective Hydrogenation of 2-Methyl-3-butyn-2-ol in Microcapillary Reactor on Supported Intermetallic PdZn Catalyst, Effect of Support Doping on Stability and Kinetic Parameters. Catalysts 2022, 12, 1660. https://doi.org/10.3390/catal12121660
Okhlopkova L, Prosvirin I, Kerzhentsev M, Ismagilov Z. Selective Hydrogenation of 2-Methyl-3-butyn-2-ol in Microcapillary Reactor on Supported Intermetallic PdZn Catalyst, Effect of Support Doping on Stability and Kinetic Parameters. Catalysts. 2022; 12(12):1660. https://doi.org/10.3390/catal12121660
Chicago/Turabian StyleOkhlopkova, Lyudmila, Igor Prosvirin, Mikhail Kerzhentsev, and Zinfer Ismagilov. 2022. "Selective Hydrogenation of 2-Methyl-3-butyn-2-ol in Microcapillary Reactor on Supported Intermetallic PdZn Catalyst, Effect of Support Doping on Stability and Kinetic Parameters" Catalysts 12, no. 12: 1660. https://doi.org/10.3390/catal12121660
APA StyleOkhlopkova, L., Prosvirin, I., Kerzhentsev, M., & Ismagilov, Z. (2022). Selective Hydrogenation of 2-Methyl-3-butyn-2-ol in Microcapillary Reactor on Supported Intermetallic PdZn Catalyst, Effect of Support Doping on Stability and Kinetic Parameters. Catalysts, 12(12), 1660. https://doi.org/10.3390/catal12121660