Microbiota of Urine, Glans and Prostate Biopsies in Patients with Prostate Cancer Reveals a Dysbiosis in the Genitourinary System
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Population and Clinical Data
2.2. Specimen Collection
2.3. DNA Extraction
2.4. Illumina High-Throughput Sequencing
2.5. Bioinformatic and Statistical Analysis
3. Results
3.1. Clinical Characteristics of Participants
3.2. Sequencing Characteristics
3.3. Bacterial Community Structure and Composition
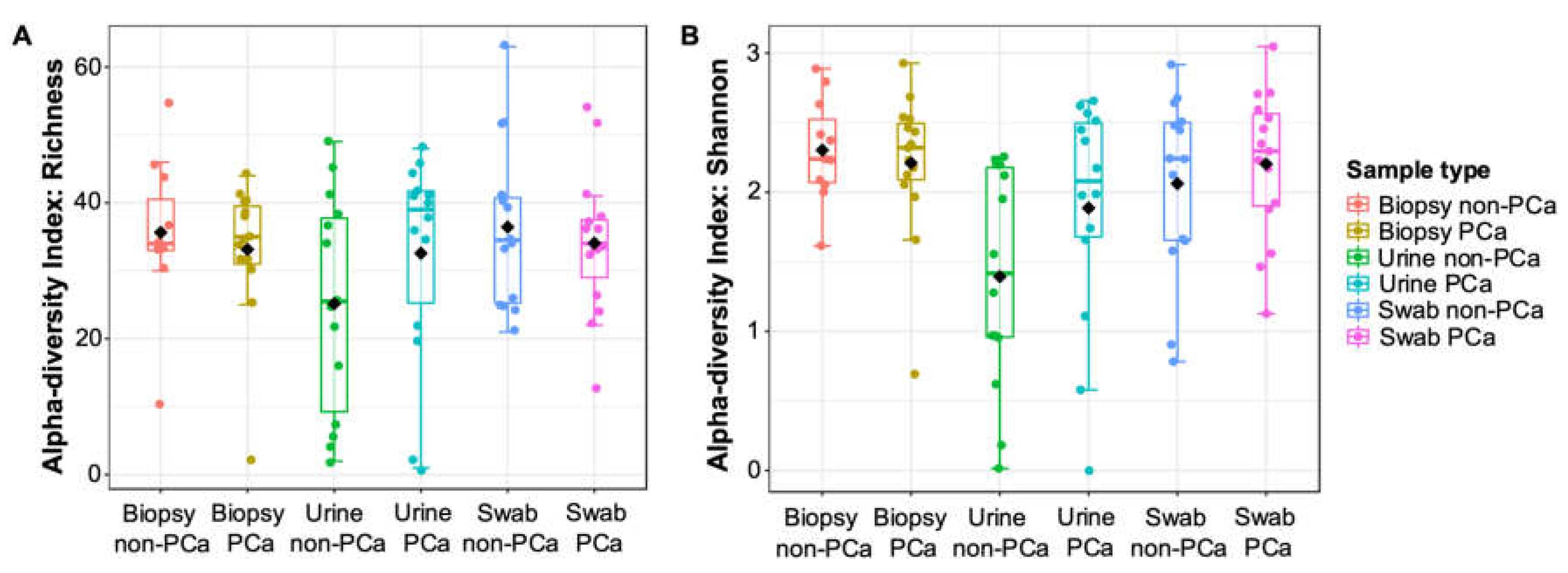
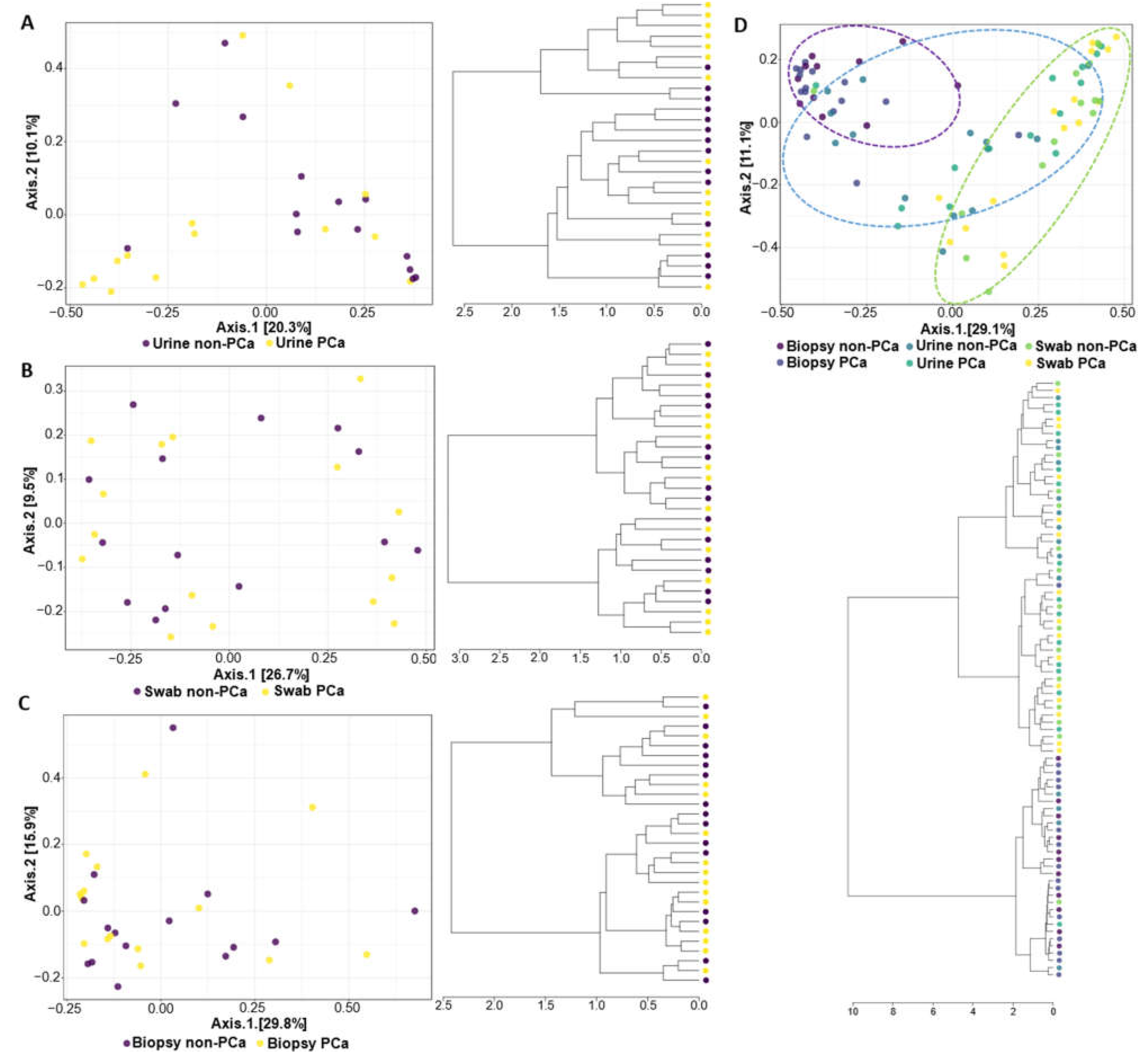
3.3.1. α- and β-Diversity
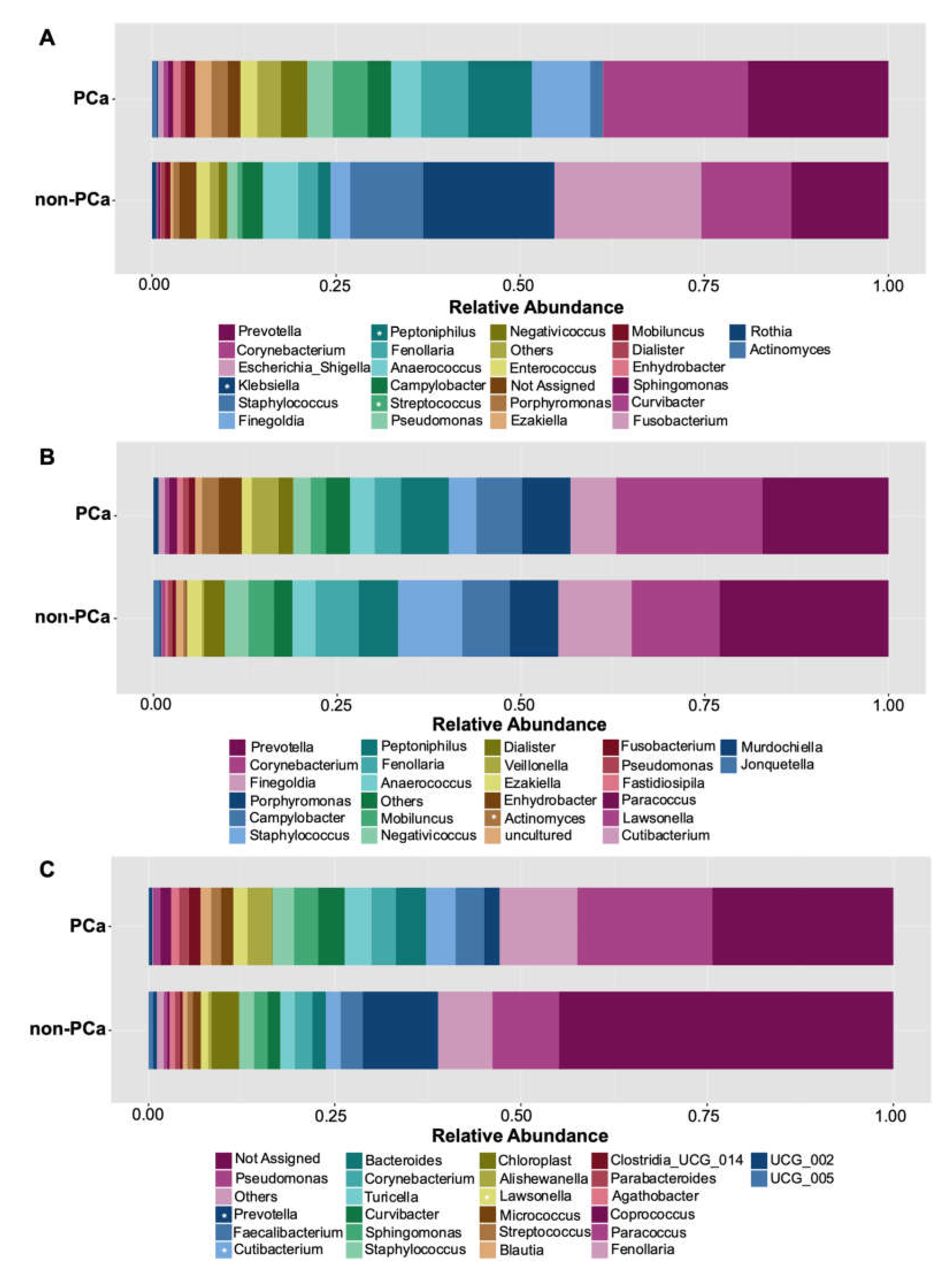
3.3.2. Taxonomic Composition of Bacterial Communities
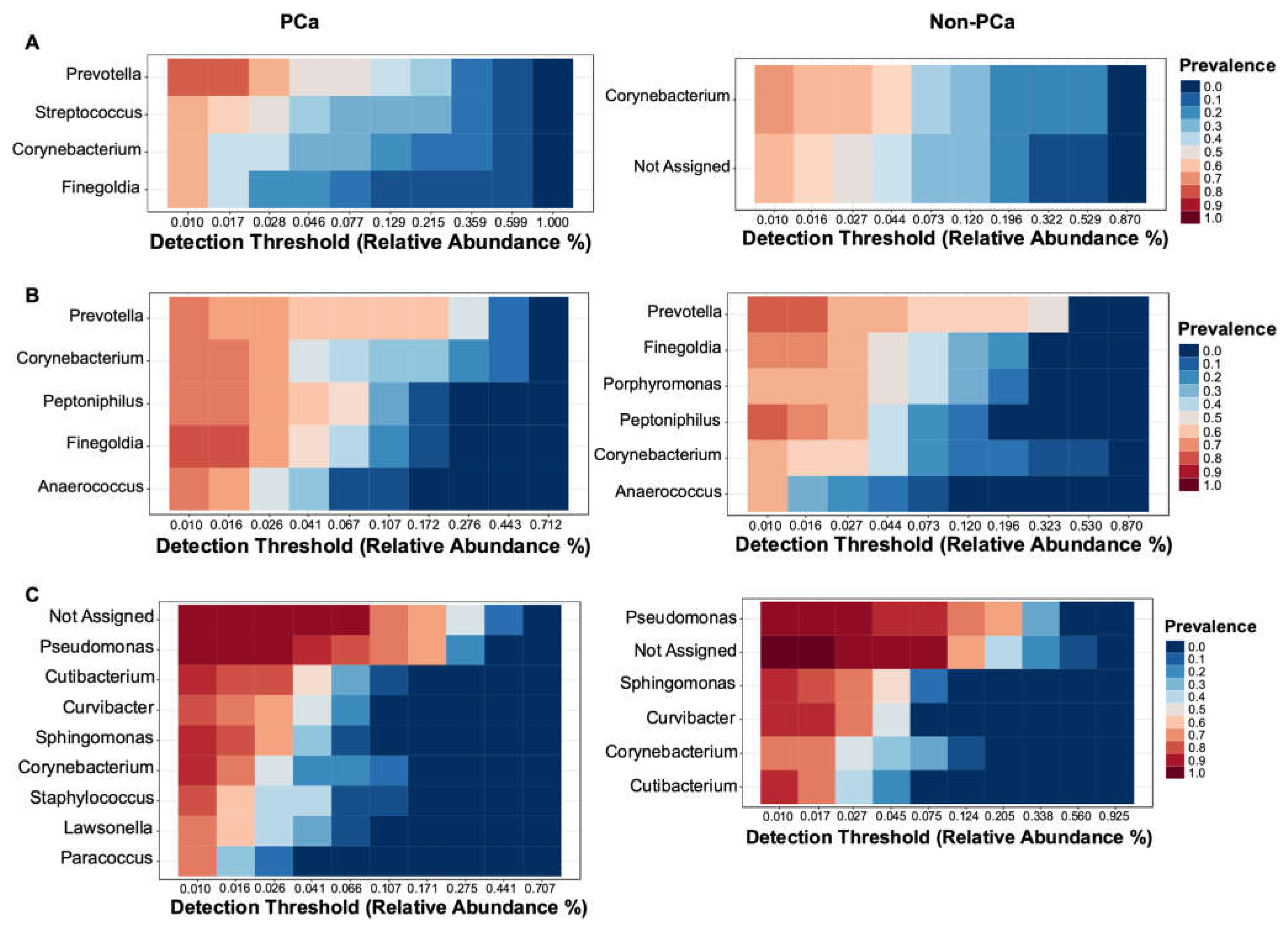
3.3.3. Bacterial Core Community
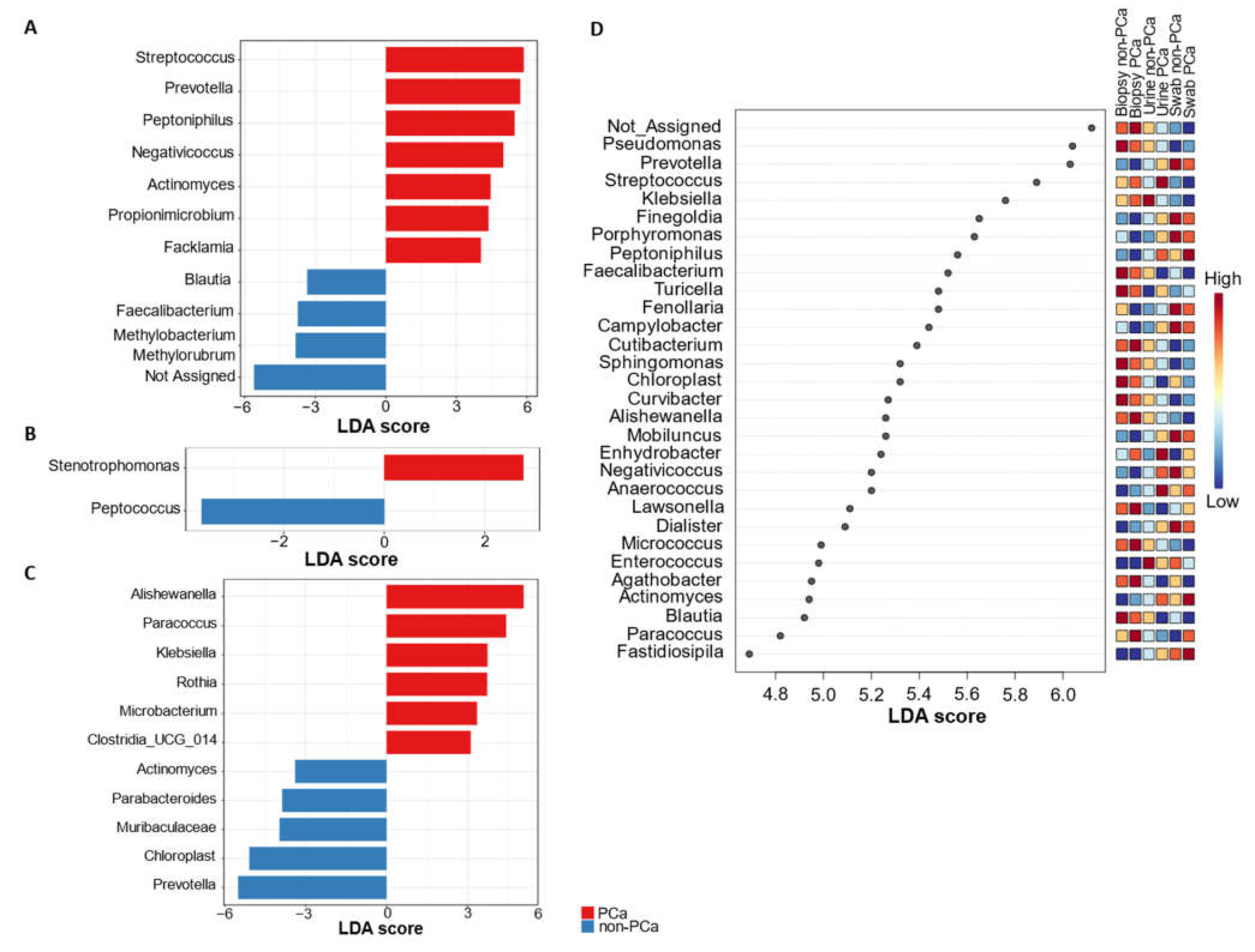
3.3.4. Differential Abundance of Bacterial Taxa
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- World Health Organization. International Agency for Research on Cancer; World Health Organization: Geneva, Switzerland, 2019. [Google Scholar]
- Ohadian Moghadam, S.; Momeni, S.A. Human microbiome and prostate cancer development: Current insights into the prevention and treatment. Front. Med. 2021, 15, 11–32. [Google Scholar] [CrossRef] [PubMed]
- Garbas, K.; Zapala, P.; Zapala, L.; Radziszewski, P. The role of microbial factors in prostate cancer development—An up-to-date review. J. Clin. Med. 2021, 10, 4772. [Google Scholar] [CrossRef] [PubMed]
- Peisch, S.F.; Van Blarigan, E.L.; Chan, J.M.; Stampfer, M.J.; Kenfield, S.A. Prostate cancer progression and mortality: A review of diet and lifestyle factors. World J. Urol. 2017, 35, 867–874. [Google Scholar] [CrossRef]
- Sfanos, K.S.; Isaacs, W.B.; De Marzo, A.M. Infections and inflammation in prostate cancer. Am. J. Clin. Exp. Urol. 2013, 1, 3–11. [Google Scholar] [PubMed]
- Sfanos, K.S.; Yegnasubramanian, S.; Nelson, W.G.; De Marzo, A.M. The inflammatory microenvironment and microbiome in prostate cancer development. Nat. Rev. Urol. 2018, 15, 11–24. [Google Scholar] [CrossRef] [PubMed]
- Chipman, J.J.; Sanda, M.G.; Dunn, R.L.; Wei, J.T.; Litwin, M.S.; Crociani, C.M.; Regan, M.M.; Chang, P.; Consortium, P.-Q. Measuring and predicting prostate cancer related quality of life changes using EPIC for clinical practice. J. Urol. 2014, 191, 638–645. [Google Scholar] [CrossRef] [PubMed]
- Costello, E.K.; Stagaman, K.; Dethlefsen, L.; Bohannan, B.J.; Relman, D.A. The application of ecological theory toward an understanding of the human microbiome. Science 2012, 336, 1255–1262. [Google Scholar] [CrossRef]
- Chu, H.; Khosravi, A.; Kusumawardhani, I.P.; Kwon, A.H.; Vasconcelos, A.C.; Cunha, L.D.; Mayer, A.E.; Shen, Y.; Wu, W.L.; Kambal, A.; et al. Gene-microbiota interactions contribute to the pathogenesis of inflammatory bowel disease. Science 2016, 352, 1116–1120. [Google Scholar] [CrossRef]
- Garrett, W.S. The gut microbiota and colon cancer. Science 2019, 364, 1133–1135. [Google Scholar] [CrossRef]
- Irrazabal, T.; Belcheva, A.; Girardin, S.E.; Martin, A.; Philpott, D.J. The multifaceted role of the intestinal microbiota in colon cancer. Mol. Cell 2014, 54, 309–320. [Google Scholar] [CrossRef]
- Salachan, P.V.; Rasmussen, M.; Fredsoe, J.; Ulhoi, B.; Borre, M.; Sorensen, K.D. Microbiota of the prostate tumor environment investigated by whole-transcriptome profiling. Genome Med. 2022, 14, 9. [Google Scholar] [CrossRef] [PubMed]
- Poore, G.D.; Kopylova, E.; Zhu, Q.; Carpenter, C.; Fraraccio, S.; Wandro, S.; Kosciolek, T.; Janssen, S.; Metcalf, J.; Song, S.J.; et al. Microbiome analyses of blood and tissues suggest cancer diagnostic approach. Nature 2020, 579, 567–574. [Google Scholar] [CrossRef] [PubMed]
- Zhu, G.; Su, H.; Johnson, C.H.; Khan, S.A.; Kluger, H.; Lu, L. Intratumour microbiome associated with the infiltration of cytotoxic CD8+ T cells and patient survival in cutaneous melanoma. Eur. J. Cancer 2021, 151, 25–34. [Google Scholar] [CrossRef] [PubMed]
- Hernandez, B.Y.; Zhu, X.; Risch, H.A.; Lu, L.; Ma, X.; Irwin, M.L.; Lim, J.K.; Taddei, T.H.; Pawlish, K.S.; Stroup, A.M.; et al. Oral cyanobacteria and hepatocellular carcinoma. Cancer Epidemiol. Biomark. Prev. 2022, 31, 221–229. [Google Scholar] [CrossRef]
- Urbaniak, C.; Gloor, G.B.; Brackstone, M.; Scott, L.; Tangney, M.; Reid, G. The microbiota of breast tissue and its association with breast cancer. Appl. Environ. Microbiol. 2016, 82, 5039–5048. [Google Scholar] [CrossRef]
- Katongole, P.; Sande, O.J.; Joloba, M.; Reynolds, S.J.; Niyonzima, N. The human microbiome and its link in prostate cancer risk and pathogenesis. Infect. Agents Cancer 2020, 15, 53. [Google Scholar] [CrossRef]
- Cavarretta, I.; Ferrarese, R.; Cazzaniga, W.; Saita, D.; Luciano, R.; Ceresola, E.R.; Locatelli, I.; Visconti, L.; Lavorgna, G.; Briganti, A.; et al. The microbiome of the prostate tumor microenvironment. Eur. Urol. 2017, 72, 625–631. [Google Scholar] [CrossRef]
- Wang, F.; Meng, W.; Wang, B.; Qiao, L. Helicobacter pylori-induced gastric inflammation and gastric cancer. Cancer Lett. 2014, 345, 196–202. [Google Scholar] [CrossRef]
- Davidsson, S.; Molling, P.; Rider, J.R.; Unemo, M.; Karlsson, M.G.; Carlsson, J.; Andersson, S.O.; Elgh, F.; Soderquis, B.; Andren, O. Frequency and typing of Propionibacterium acnes in prostate tissue obtained from men with and without prostate cancer. Infect. Agents Cancer 2016, 11, 26. [Google Scholar] [CrossRef]
- Twu, O.; Dessi, D.; Vu, A.; Mercer, F.; Stevens, G.C.; de Miguel, N.; Rappelli, P.; Cocco, A.R.; Clubb, R.T.; Fiori, P.L.; et al. Trichomonas vaginalis homolog of macrophage migration inhibitory factor induces prostate cell growth, invasiveness, and inflammatory responses. Proc. Natl. Acad. Sci. USA 2014, 111, 8179–8184. [Google Scholar] [CrossRef]
- Vazquez-Salas, R.A.; Torres-Sanchez, L.; Lopez-Carrillo, L.; Romero-Martinez, M.; Manzanilla-Garcia, H.A.; Cruz-Ortiz, C.H.; Mendoza-Pena, F.; Jimenez-Rios, M.A.; Rodriguez-Covarrubias, F.; Hernandez-Toriz, N.; et al. History of gonorrhea and prostate cancer in a population-based case-control study in Mexico. Cancer Epidemiol. 2016, 40, 95–101. [Google Scholar] [CrossRef] [PubMed]
- Han, I.H.; Song, H.O.; Ryu, J.S. IL-6 produced by prostate epithelial cells stimulated with Trichomonas vaginalis promotes proliferation of prostate cancer cells by inducing M2 polarization of THP-1-derived macrophages. PLoS Negl. Trop. Dis. 2020, 14, e0008126. [Google Scholar] [CrossRef]
- Miyake, M.; Ohnishi, K.; Hori, S.; Nakano, A.; Nakano, R.; Yano, H.; Ohnishi, S.; Owari, T.; Morizawa, Y.; Itami, Y.; et al. Mycoplasma genitalium infection and chronic inflammation in human prostate cancer: Detection using prostatectomy and needle biopsy specimens. Cells 2019, 8, 212. [Google Scholar] [CrossRef] [PubMed]
- Alanee, S.; El-Zawahry, A.; Dynda, D.; Dabaja, A.; McVary, K.; Karr, M.; Braundmeier-Fleming, A. A prospective study to examine the association of the urinary and fecal microbiota with prostate cancer diagnosis after transrectal biopsy of the prostate using 16sRNA gene analysis. Prostate 2019, 79, 81–87. [Google Scholar] [CrossRef] [PubMed]
- Shrestha, E.; White, J.R.; Yu, S.H.; Kulac, I.; Ertunc, O.; De Marzo, A.M.; Yegnasubramanian, S.; Mangold, L.A.; Partin, A.W.; Sfanos, K.S. Profiling the urinary microbiome in men with positive versus negative biopsies for prostate cancer. J. Urol. 2018, 199, 161–171. [Google Scholar] [CrossRef]
- Golombos, D.M.; Ayangbesan, A.; O’Malley, P.; Lewicki, P.; Barlow, L.; Barbieri, C.E.; Chan, C.; DuLong, C.; Abu-Ali, G.; Huttenhower, C.; et al. The role of gut microbiome in the pathogenesis of prostate cancer: A prospective, pilot study. Urology 2018, 111, 122–128. [Google Scholar] [CrossRef]
- Sfanos, K.S.; Markowski, M.C.; Peiffer, L.B.; Ernst, S.E.; White, J.R.; Pienta, K.J.; Antonarakis, E.S.; Ross, A.E. Compositional differences in gastrointestinal microbiota in prostate cancer patients treated with androgen axis-targeted therapies. Prostate Cancer Prostatic Dis. 2018, 21, 539–548. [Google Scholar] [CrossRef]
- Liss, M.A.; White, J.R.; Goros, M.; Gelfond, J.; Leach, R.; Johnson-Pais, T.; Lai, Z.; Rourke, E.; Basler, J.; Ankerst, D.; et al. Metabolic biosynthesis pathways identified from fecal microbiome associated with prostate cancer. Eur. Urol. 2018, 74, 575–582. [Google Scholar] [CrossRef]
- Banerjee, S.; Alwine, J.C.; Wei, Z.; Tian, T.; Shih, N.; Sperling, C.; Guzzo, T.; Feldman, M.D.; Robertson, E.S. Microbiome signatures in prostate cancer. Carcinogenesis 2019, 40, 749–764. [Google Scholar] [CrossRef]
- Feng, Y.; Jaratlerdsiri, W.; Patrick, S.M.; Lyons, R.J.; Haynes, A.M.; Collins, C.C.; Stricker, P.D.; Bornman, M.S.R.; Hayes, V.M. Metagenomic analysis reveals a rich bacterial content in high-risk prostate tumors from African men. Prostate 2019, 79, 1731–1738. [Google Scholar] [CrossRef]
- Feng, Y.; Ramnarine, V.R.; Bell, R.; Volik, S.; Davicioni, E.; Hayes, V.M.; Ren, S.; Collins, C.C. Metagenomic and metatranscriptomic analysis of human prostate microbiota from patients with prostate cancer. BMC Genom. 2019, 20, 146. [Google Scholar] [CrossRef] [PubMed]
- Ma, X.; Chi, C.; Fan, L.; Dong, B.; Shao, X.; Xie, S.; Li, M.; Xue, W. The microbiome of prostate fluid is associated with prostate cancer. Front. Microbiol. 2019, 10, 1664. [Google Scholar] [CrossRef] [PubMed]
- Caporaso, J.G.; Kuczynski, J.; Stombaugh, J.; Bittinger, K.; Bushman, F.D.; Costello, E.K.; Fierer, N.; Pena, A.G.; Goodrich, J.K.; Gordon, J.I.; et al. QIIME allows analysis of high-throughput community sequencing data. Nat. Methods 2010, 7, 335–336. [Google Scholar] [CrossRef] [PubMed]
- Callahan, B.J.; McMurdie, P.J.; Rosen, M.J.; Han, A.W.; Johnson, A.J.; Holmes, S.P. DADA2: High-resolution sample inference from Illumina amplicon data. Nat. Methods 2016, 13, 581–583. [Google Scholar] [CrossRef]
- Glockner, F.O.; Yilmaz, P.; Quast, C.; Gerken, J.; Beccati, A.; Ciuprina, A.; Bruns, G.; Yarza, P.; Peplies, J.; Westram, R.; et al. 25 years of serving the community with ribosomal RNA gene reference databases and tools. J. Biotechnol. 2017, 261, 169–176. [Google Scholar] [CrossRef]
- Davis, N.M.; Proctor, D.M.; Holmes, S.P.; Relman, D.A.; Callahan, B.J. Simple statistical identification and removal of contaminant sequences in marker-gene and metagenomics data. Microbiome 2018, 6, 226. [Google Scholar] [CrossRef]
- Chong, J.; Liu, P.; Zhou, G.; Xia, J. Using MicrobiomeAnalyst for comprehensive statistical, functional, and meta-analysis of microbiome data. Nat. Protoc. 2020, 15, 799–821. [Google Scholar] [CrossRef]
- Segata, N.; Izard, J.; Waldron, L.; Gevers, D.; Miropolsky, L.; Garrett, W.S.; Huttenhower, C. Metagenomic biomarker discovery and explanation. Genome Biol. 2011, 12, R60. [Google Scholar] [CrossRef]
- Sanders, D.J.; Inniss, S.; Sebepos-Rogers, G.; Rahman, F.Z.; Smith, A.M. The role of the microbiome in gastrointestinal inflammation. Biosci. Rep. 2021, 41, BSR20203850. [Google Scholar] [CrossRef]
- Ahn, H.K.; Kim, K.; Park, J.; Kim, K.H. Urinary microbiome profile in men with genitourinary malignancies. Investig. Clin. Urol. 2022, 63, 569–576. [Google Scholar] [CrossRef]
- Alexeyev, O.; Bergh, J.; Marklund, I.; Thellenberg-Karlsson, C.; Wiklund, F.; Gronberg, H.; Bergh, A.; Elgh, F. Association between the presence of bacterial 16S RNA in prostate specimens taken during transurethral resection of prostate and subsequent risk of prostate cancer (Sweden). Cancer Causes Control 2006, 17, 1127–1133. [Google Scholar] [CrossRef] [PubMed]
- Kruslin, B.; Tomas, D.; Dzombeta, T.; Milkovic-Perisa, M.; Ulamec, M. Inflammation in prostatic hyperplasia and carcinoma-basic scientific approach. Front. Oncol. 2017, 7, 77. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.Y.; Wang, J.W.; Juan, Y.S.; Li, C.C.; Liu, C.J.; Cho, S.Y.; Yeh, H.C.; Chueh, K.S.; Wu, W.J.; Wu, D.C. The impact of urine microbiota in patients with lower urinary tract symptoms. Ann. Clin. Microbiol. Antimicrob. 2021, 20, 23. [Google Scholar] [CrossRef] [PubMed]
- Dong, Q.; Nelson, D.E.; Toh, E.; Diao, L.; Gao, X.; Fortenberry, J.D.; Van der Pol, B. The microbial communities in male first catch urine are highly similar to those in paired urethral swab specimens. PLoS ONE 2011, 6, e19709. [Google Scholar] [CrossRef] [PubMed]
- Gonçalves, M.F.M.; Fernandes, Â.R.; Rodrigues, A.G.; Lisboa, C. Microbiome in male genital mucosa (prepuce, glans, and coronal sulcus): A systematic review. Microorganisms 2022, 10, 2312. [Google Scholar] [CrossRef] [PubMed]
- Riemersma, W.A.; van der Schee, C.J.; van der Meijden, W.I.; Verbrugh, H.A.; van Belkum, A. Microbial population diversity in the urethras of healthy males and males suffering from nonchlamydial, nongonococcal urethritis. J. Clin. Microbiol. 2003, 41, 1977–1986. [Google Scholar] [CrossRef] [PubMed]
- Rosenthal, M.E.; Rojtman, A.D.; Frank, E. Finegoldia magna (formerly Peptostreptococcus magnus): An overlooked etiology for toxic shock syndrome? Med. Hypotheses 2012, 79, 138–140. [Google Scholar] [CrossRef] [PubMed]
- So, K.A.; Yang, E.J.; Kim, N.R.; Hong, S.R.; Lee, J.H.; Hwang, C.S.; Shim, S.H.; Lee, S.J.; Kim, T.J. Changes of vaginal microbiota during cervical carcinogenesis in women with human papillomavirus infection. PLoS ONE 2020, 15, e0238705. [Google Scholar] [CrossRef] [PubMed]
- Ikeda, M.; Kobayashi, T.; Suzuki, T.; Wakabayashi, Y.; Ohama, Y.; Maekawa, S.; Takahashi, S.; Homma, Y.; Tatsuno, K.; Sato, T.; et al. Propionimicrobium lymphophilum and Actinotignum schaalii bacteraemia: A case report. New Microbes New Infect. 2017, 18, 18–21. [Google Scholar] [CrossRef]
- Williams, G.D. Two cases of urinary tract infection caused by Propionimicrobium lymphophilum. J. Clin. Microbiol. 2015, 53, 3077–3080. [Google Scholar] [CrossRef]
- Boyanova, L.; Marteva-Proevska, Y.; Markovska, R.; Yordanov, D.; Gergova, R. Urinary tract infections: Should we think about the anaerobic cocci? Anaerobe 2022, 77, 102509. [Google Scholar] [CrossRef] [PubMed]
- Bschleipfer, T.; Karl, I. Bladder Microbiome in the context of urological disorders—Is there a biomarker potential for interstitial cystitis? Diagnostics 2022, 12, 281. [Google Scholar] [CrossRef] [PubMed]
- Hurst, R.; Meader, E.; Gihawi, A.; Rallapalli, G.; Clark, J.; Kay, G.L.; Webb, M.; Manley, K.; Curley, H.; Walker, H.; et al. Microbiomes of urine and the prostate are linked to human prostate cancer risk groups. Eur. Urol. Oncol. 2022, 5, 412–419. [Google Scholar] [CrossRef]
- Ma, J.; Gnanasekar, A.; Lee, A.; Li, W.T.; Haas, M.; Wang-Rodriguez, J.; Chang, E.Y.; Rajasekaran, M.; Ongkeko, W.M. Influence of intratumor microbiome on clinical outcome and immune processes in prostate cancer. Cancers 2020, 12, 2524. [Google Scholar] [CrossRef]
- Shinohara, D.B.; Vaghasia, A.M.; Yu, S.H.; Mak, T.N.; Bruggemann, H.; Nelson, W.G.; De Marzo, A.M.; Yegnasubramanian, S.; Sfanos, K.S. A mouse model of chronic prostatic inflammation using a human prostate cancer-derived isolate of Propionibacterium acnes. Prostate 2013, 73, 1007–1015. [Google Scholar] [CrossRef] [PubMed]
- Davidsson, S.; Carlsson, J.; Greenberg, L.; Wijkander, J.; Soderquist, B.; Erlandsson, A. Cutibacterium acnes induces the expression of immunosuppressive genes in macrophages and is associated with an increase of regulatory T-cells in prostate cancer. Microbiol. Spectr. 2021, 9, e0149721. [Google Scholar] [CrossRef] [PubMed]
- Ugge, H.; Carlsson, J.; Soderquist, B.; Fall, K.; Anden, O.; Davidsson, S. The influence of prostatic Cutibacterium acnes infection on serum levels of IL6 and CXCL8 in prostate cancer patients. Infect. Agent Cancer 2018, 13, 34. [Google Scholar] [CrossRef] [PubMed]
- Yu, H.; Meng, H.; Zhou, F.; Ni, X.; Shen, S.; Das, U.N. Urinary microbiota in patients with prostate cancer and benign prostatic hyperplasia. Arch. Med. Sci. 2015, 11, 385–394. [Google Scholar] [CrossRef]
- Yow, M.A.; Tabrizi, S.N.; Severi, G.; Bolton, D.M.; Pedersen, J.; Giles, G.G.; Southey, M.C.; Australian Prostate Cancer, Bioresource. Characterisation of microbial communities within aggressive prostate cancer tissues. Infect. Agent Cancer 2017, 12, 4. [Google Scholar] [CrossRef] [PubMed]
- Sarkar, P.; Malik, S.; Banerjee, A.; Datta, C.; Pal, D.K.; Ghosh, A.; Saha, A. Differential microbial signature associated with benign prostatic hyperplasia and prostate cancer. Front. Cell Infect. Microbiol. 2022, 12, 894777. [Google Scholar] [CrossRef]
- Ahmed, W.; Dewar, S.; Williams, R.; Stansby, G.; Harris, K.; Weiand, D. Lawsonella clevelandensis is a rare cause of infected chronic contained rupture of abdominal aortic aneurysm. Access Microbiol. 2021, 3, acmi000183. [Google Scholar] [CrossRef] [PubMed]
- Favila Menezes, M.; Sousa, M.J.; Paixão, P.; Atouguia, J.; Negreiros, I.; Simões, M.J. Lawsonella clevelandensis as the causative agent of a breast abscess. IDCases 2018, 12, 95–96. [Google Scholar] [CrossRef] [PubMed]
- Ramesh, R.; Assi, M.; Esquer Garrigos, Z.; Sohail, M.R. Lawsonella clevelandensis: An emerging cause of vascular graft infection. BMJ Case Rep. 2021, 14, e237350. [Google Scholar] [CrossRef] [PubMed]
- Gajdacs, M.; Urban, E. The pathogenic role of Actinomyces spp. and related organisms in genitourinary infections: Discoveries in the new, modern diagnostic era. Antibiotics 2020, 9, 524. [Google Scholar] [CrossRef]
- Kononen, E.; Wade, W.G. Actinomyces and related organisms in human infections. Clin. Microbiol. Rev. 2015, 28, 419–442. [Google Scholar] [CrossRef]
- Sabbe, L.J.; Van De Merwe, D.; Schouls, L.; Bergmans, A.; Vaneechoutte, M.; Vandamme, P. Clinical spectrum of infections due to the newly described Actinomyces species A. turicensis, A. radingae, and A. europaeus. J. Clin. Microbiol. 1999, 37, 8–13. [Google Scholar] [CrossRef]
- Groeneveld, G.H.; Veldkamp, K.E.; van Dissel, J.T. Repetitive urinary tract infections and two prostatic masses: Prostatic soft tissue infection with Actinomyces neuii. Int. J. Infect. Dis. 2019, 86, 55–56. [Google Scholar] [CrossRef]
- Shukla, S.; Srivastava, J.K.; Shankar, E.; Kanwal, R.; Nawab, A.; Sharma, H.; Bhaskaran, N.; Ponsky, L.E.; Fu, P.; MacLennan, G.T.; et al. Oxidative stress and antioxidant status in high-risk prostate cancer subjects. Diagnostics 2020, 10, 126. [Google Scholar] [CrossRef]
- Di Minno, A.; Aveta, A.; Gelzo, M.; Tripodi, L.; Pandolfo, S.D.; Crocetto, F.; Imbimbo, C.; Castaldo, G. 8-Hydroxy-2-Deoxyguanosine and 8-Iso-Prostaglandin F2α: Putative biomarkers to assess oxidative stress damage following robot-assisted radical prostatectomy (RARP). J. Clin. Med. 2022, 11, 6102. [Google Scholar] [CrossRef]
- Hilt, E.E.; McKinley, K.; Pearce, M.M.; Rosenfeld, A.B.; Zilliox, M.J.; Mueller, E.R.; Brubaker, L.; Gai, X.; Wolfe, A.J.; Schreckenberger, P.C. Urine is not sterile: Use of enhanced urine culture techniques to detect resident bacterial flora in the adult female bladder. J. Clin. Microbiol. 2014, 52, 871–876. [Google Scholar] [CrossRef]

| Characteristics | PCa Group | Non-PCa Group | p-Value | |
|---|---|---|---|---|
| Subjects | 15 | 15 | nd | |
| Age, years: mean (SD) | 68 (9) | 69 (8) | 0.621 | |
| PSA, ng/mL: median (range) | 9 (3–28) | 9 (3–32) | 0.640 | |
| Prostate volume, g: mean (range) | 37 (56) | 79 (230) | 0.015 | |
| Charlson index: n (%) | 1 | 2 (13%) | 2 (13%) | 0.799 |
| 2 | 4 (27%) | 2 (13%) | ||
| 3 | 4 (27%) | 5 (33%) | ||
| 4 | 1 (7%) | 3 (20%) | ||
| 5 | 2 (13%) | 2 (13%) | ||
| 6 | 1 (7%) | 1 (7%) | ||
| 7 | 1 (7%) | 0 | ||
| ISUP grade: n (%) | 1 | 3 (20%) | nd | nd |
| 2 | 6 (40%) | nd | ||
| 3 | 2 (13%) | nd | ||
| 4 | 2 (13%) | nd | ||
| 5 | 2 (13%) | nd | ||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gonçalves, M.F.M.; Pina-Vaz, T.; Fernandes, Â.R.; Miranda, I.M.; Silva, C.M.; Rodrigues, A.G.; Lisboa, C. Microbiota of Urine, Glans and Prostate Biopsies in Patients with Prostate Cancer Reveals a Dysbiosis in the Genitourinary System. Cancers 2023, 15, 1423. https://doi.org/10.3390/cancers15051423
Gonçalves MFM, Pina-Vaz T, Fernandes ÂR, Miranda IM, Silva CM, Rodrigues AG, Lisboa C. Microbiota of Urine, Glans and Prostate Biopsies in Patients with Prostate Cancer Reveals a Dysbiosis in the Genitourinary System. Cancers. 2023; 15(5):1423. https://doi.org/10.3390/cancers15051423
Chicago/Turabian StyleGonçalves, Micael F. M., Teresa Pina-Vaz, Ângela Rita Fernandes, Isabel M. Miranda, Carlos Martins Silva, Acácio Gonçalves Rodrigues, and Carmen Lisboa. 2023. "Microbiota of Urine, Glans and Prostate Biopsies in Patients with Prostate Cancer Reveals a Dysbiosis in the Genitourinary System" Cancers 15, no. 5: 1423. https://doi.org/10.3390/cancers15051423
APA StyleGonçalves, M. F. M., Pina-Vaz, T., Fernandes, Â. R., Miranda, I. M., Silva, C. M., Rodrigues, A. G., & Lisboa, C. (2023). Microbiota of Urine, Glans and Prostate Biopsies in Patients with Prostate Cancer Reveals a Dysbiosis in the Genitourinary System. Cancers, 15(5), 1423. https://doi.org/10.3390/cancers15051423






