Radiobiological Meta-Analysis of the Response of Prostate Cancer to Different Fractionations: Evaluation of the Linear–Quadratic Response at Large Doses and the Effect of Risk and ADT
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Clinical Dataset
2.2. Radiobiological Modelling: Dose–Response
2.3. Statistical Methods
2.4. Radiobiological Modelling: and Number of Clonogens
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Brenner, D.J.; Hall, E.J. Fractionation and protraction for radiotherapy of prostate carcinoma. Int. J. Radiat. Oncol. Biol. Phys. 1999, 43, 1095–1101. [Google Scholar] [CrossRef] [PubMed]
- Bentzen, S.M.; Ritter, M.A. The alpha/beta ratio for prostate cancer: What is it, really? Radiother. Oncol. 2005, 76, 1–3. [Google Scholar] [CrossRef] [PubMed]
- Fowler, J.F. The radiobiology of prostate cancer including new aspects of fractionated radiotherapy. Acta Oncol. 2005, 44, 265–276. [Google Scholar] [CrossRef] [PubMed]
- Miralbell, R.; Roberts, S.A.; Zubizarreta, E.; Hendry, J.H. Dose-fractionation sensitivity of prostate cancer deduced from radiotherapy outcomes of 5969 patients in seven international institutional datasets: α/β = 1.4 (0.9–2.2) Gy. Int. J. Radiat. Oncol. Biol. Phys. 2012, 82, e17–e24. [Google Scholar] [CrossRef] [PubMed]
- Shaffer, R.; Pickles, T.; Lee, R.; Moiseenko, V. Deriving prostate alpha-beta ratio using carefully matched groups, long follow-up and the phoenix definition of biochemical failure. Int. J. Radiat. Oncol. Biol. Phys. 2011, 79, 1029–1036. [Google Scholar] [CrossRef]
- Oliveira, S.M.; Teixeira, N.J.; Fernandes, L. What do we know about the alpha/beta for prostate cancer? Med. Phys. 2012, 39, 3189–3201. [Google Scholar] [CrossRef]
- Dasu, A. Is the alpha/beta value for prostate tumours low enough to be safely used in clinical trials? Clin. Oncol. 2007, 19, 289–301. [Google Scholar] [CrossRef]
- Nahum, A.E.; Movsas, B.; Horwitz, E.M.; Stobbe, C.C.; Chapman, J.D. Incorporating clinical measurements of hypoxia into tumour local control modelling of prostate cancer: Implications for the alpha/beta ratio. Int. J. Radiat. Oncol. Biol. Phys. 2003, 57, 391–401. [Google Scholar] [CrossRef]
- Pedicini, P.; Strigari, L.; Benassi, M. Estimation of a self-consistent set of radiobiological parameters from hypofractionated versus standard radiation therapy of prostate cancer. Int. J. Radiat. Oncol. Biol. Phys. 2013, 85, e231–e237. [Google Scholar] [CrossRef]
- Datta, N.R.; Stutz, E.; Rogers, S.; Bodis, S. Clinical estimation of α/β values for prostate cancer from isoeffective phase III randomized trials with moderately hypofractionated radiotherapy. Acta Oncol. 2018, 57, 883–894. [Google Scholar] [CrossRef]
- Vogelius, I.R.; Bentzen, S.M. Diminishing Returns From Ultrahypofractionated Radiation Therapy for Prostate Cancer. Int. J. Radiat. Oncol. Biol. Phys. 2020, 107, 299–304. [Google Scholar] [CrossRef]
- Lo, S.S.; Fakiris, A.J.; Chang, E.L.; Mayr, N.A.; Wang, J.Z.; Papiez, L.; Teh, B.S.; McGarry, R.C.; Cardenes, H.R.; Timmerman, R.D. Stereotactic body radiation therapy: A novel treatment modality. Nat. Rev. Clin. Oncol. 2010, 7, 44–54. [Google Scholar] [CrossRef]
- Zietman, A.L.; Bae, K.; Slater, J.D.; Shipley, W.U.; Efstathiou, J.A.; Coen, J.J.; Bush, D.A.; Lunt, M.; Spiegel, D.Y.; Skowronski, R.; et al. Randomized trial comparing conventional-dose with high-dose conformal radiation therapy in early-stage adenocarcinoma of the prostate: Long-term results from proton radiation oncology group/american college of radiology 95-09. J. Clin. Oncol. 2010, 28, 1106–1111. [Google Scholar] [CrossRef]
- Arcangeli, G.; Saracino, B.; Arcangeli, S.; Gomellini, S.; Petrongari, M.G.; Sanguineti, G.; Strigari, L. Moderate hypofractionation in high-risk, organ-confined prostate cancer: Final results of a phase III randomized trial. J. Clin. Oncol. 2017, 35, 1891–1897. [Google Scholar] [CrossRef]
- Dearnaley, D.P.; Jovic, G.; Syndikus, I.; Khoo, V.; Cowan, R.A.; Graham, J.D.; Aird, E.G.; Bottomley, D.; Huddart, R.A.; Jose, C.C.; et al. Escalated-dose versus control-dose conformal radiotherapy for prostate cancer: Long-term results from the MRC RT01 randomised controlled trial. Lancet Oncol. 2014, 15, 464–473. [Google Scholar] [CrossRef]
- Kirkpatrick, J.P.; Meyer, J.J.; Marks, L.B. The linear–quadratic model is inappropriate to model high dose per fraction effects in radiosurgery. Semin. Radiat. Oncol. 2008, 18, 240–243. [Google Scholar] [CrossRef]
- Brenner, D.J. The linear–quadratic model is an appropriate methodology for determining isoeffective doses at large doses per fraction. Semin. Radiat. Oncol. 2008, 18, 234–239. [Google Scholar] [CrossRef]
- Sperduto, P.W.; Song, C.W.; Kirkpatrick, J.P.; Glatstein, E. A hypothesis: Indirect cell death in the radiosurgery era. Int. J. Radiat. Oncol. Biol. Phys. 2015, 91, 11–13. [Google Scholar] [CrossRef]
- Guerrero, M.; Li, X.A. Extending the linear–quadratic model for large fraction doses pertinent to stereotactic radiotherapy. Phys. Med. Biol. 2004, 49, 4825–4835. [Google Scholar] [CrossRef]
- Wang, J.Z.; Huang, Z.; Lo, S.S.; Yuh, W.T.C.; Mayr, N.A. A generalized linear–quadratic model for radiosurgery, stereotactic body radiation therapy, and high-dose rate brachytherapy. Sci. Transl. Med. 2010, 2, 39ra48. [Google Scholar] [CrossRef]
- Song, C.W.; Lee, Y.J.; Griffin, R.J.; Park, I.; Koonce, N.A.; Hui, S.; Kim, M.-S.; Dusenbery, K.E.; Sperduto, P.W.; Cho, L.C. Indirect Tumour Cell Death after High-Dose Hypofractionated Irradiation: Implications for Stereotactic Body Radiation Therapy and Stereotactic Radiation Surgery. Int. J. Radiat. Oncol. Biol. Phys. 2015, 93, 166–172. [Google Scholar] [CrossRef] [PubMed]
- Song, C.W.; Glatstein, E.; Marks, L.B.; Emami, B.; Grimm, J.; Sperduto, P.W.; Kim, M.-S.; Hui, S.; Dusenbery, K.E.; Cho, L.C. Biological Principles of Stereotactic Body Radiation Therapy (SBRT) and Stereotactic Radiation Surgery (SRS): Indirect Cell Death. Int. J. Radiat. Oncol. Biol. Phys. 2021, 110, 21–34. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Barbeito, P.; Díaz-Botana, P.; Gago-Arias, A.; Feijoo, M.; Neira, S.; Guiu-Souto, J.; López-Pouso, Ó.; Gómez-Caamaño, A.; Pardo-Montero, J. A model of indirect cell death caused by tumour vascular damage after high-dose radiotherapy. Cancer Res. 2019, 79, 6044–6053. [Google Scholar] [CrossRef] [PubMed]
- Serre, R.; Benzekry, S.; Padovani, L.; Meille, C.; André, N.; Ciccolini, J.; Barlesi, F.; Muracciole, X.; Barbolosi, D. Mathematical Modelling of Cancer Immunotherapy and Its Synergy with Radiotherapy. Cancer Res. 2016, 76, 4931–4940. [Google Scholar] [CrossRef] [PubMed]
- Poleszczuk, J.; Enderling, H. The optimal radiation dose to induce robust systemic anti-tumour immunity. Int. J. Mol. Sci. 2018, 19, 3377. [Google Scholar] [CrossRef]
- Gago-Arias, A.; Neira, S.; Pombar, M.; Gómez-Caamaño, A.; Pardo-Montero, J. Evaluation of indirect damage and damage saturation effects in dose–response curves of hypofractionated radiotherapy of early-stage NSCLC and brain metastases. Radiother. Oncol. 2021, 2021 161, 1–8. [Google Scholar] [CrossRef]
- González-Crespo, I.; Gómez-Caamaño, A.; Pouso, O.L.; Fenwick, J.D.; Pardo-Montero, J. A biomathematical model of tumour response to radioimmunotherapy with αPDL1 and αCTLA4. IEEE/ACM Trans. Comput. Biol. Bioinform. 2023, 20, 808–821. [Google Scholar] [CrossRef]
- Royce, T.J.; Mavroidis, P.; Wang, K.; Falchook, A.D.; Sheets, N.C.; Fuller, D.B. Tumour Control Probability Modelling and Systematic Review of the Literature of Stereotactic Body Radiation Therapy for Prostate Cancer. Int. J. Radiat. Oncol. Biol. Phys. 2021, 110, 227–236. [Google Scholar] [CrossRef]
- Alayed, Y.; Cheung, P.; Pang, G.; Mamedov, A.; D’Alimonte, L.; Deabreu, A.; Commisso, K.; Commisso, A.; Zhang, L.; Quon, H.C.; et al. Dose escalation for prostate stereotactic ablative radiotherapy (SABR): Late outcomes from two prospective clinical trials. Radiother. Oncol. 2018, 127, 213–218. [Google Scholar] [CrossRef]
- Arcangeli, S.; Strigari, L.; Gomellini, S.; Saracino, B.; Petrongari, M.G.; Pinnarò, P.; Pinzi, V.; Arcangeli, G. Updated Results and Patterns of Failure in a Randomized Hypofractionation Trial for High-risk Prostate Cancer. Int. J. Radiat. Oncol. Biol. Phys. 2012, 84, 1172–1184. [Google Scholar] [CrossRef]
- Bernetich, M.; Oliai, C.; Lanciano, R.; Hanlon, A.; Lamond, J.; Arrigo, S.; Yang, J.; Good, M.; Feng, J.; Brown, R.; et al. SBRT for the primary treatment of localized prostate cancer: The effect of Gleason score, dose and heterogeneity of intermediate risk on outcome utilizing 2.2014 NCCN risk stratification guidelines. Front. Oncol. 2014, 4, 312. [Google Scholar] [CrossRef]
- Catton, C.N.; Lukka, H.; Gu, C.; Martin, J.M.; Supiot, S.; Chung, P.W.M.; Bauman, G.S.; Bahary, J.; Ahmed, S.; Cheung, P.; et al. Randomized Trial of a Hypofractionated Radiation Regimen for the Treatment of Localized Prostate Cancer. J. Clin. Oncol. 2017, 35, 1884–1890. [Google Scholar] [CrossRef]
- Davis, J.; Sharma, S.; Shumway, R.; Perry, D.; Bydder, S.; Simpson, C.K.; D’Ambrosio, D. Stereotactic Body Radiotherapy for Clinically Localized Prostate Cancer: Toxicity and Biochemical Disease-Free Outcomes from a Multi-Institutional Patient Registry. Cureus 2015, 7, e395. [Google Scholar] [CrossRef]
- Dearnaley, D.; Syndikus, I.; Mossop, H.; Khoo, V.; Birtle, A.; Bloomfield, D.; Graham, J.; Kirkbride, P.; Logue, J.; Malik, Z.; et al. Conventional versus hypofractionated high-dose intensity-modulated radiotherapy for prostate cancer: 5-year outcomes of the randomised, non-inferiority, phase 3 CHHiP trial. Lancet Oncol. 2016, 17, 1047–1060. [Google Scholar] [CrossRef]
- Fuller, D.F.; Naitoh, J.; Mardirossian, G. Virtual HDR CyberKnife SBRT for localized prostatic carcinoma: 5-year disease-free survival and toxicity observations. Front. Oncol. 2014, 4, 321. [Google Scholar] [CrossRef]
- Hannah, R.; Tumati, V.; Xie, X.; Cho, C.; Kavanagh, B.D.; Brindle, J.; Raben, D.; Nanda, A.; Cooley, S.; Kim, D.W.N.; et al. Stereotactic body radiation therapy for low and intermediate risk prostate cancer—Results from a multi-institutional clinical trial. Eur. J. Cancer 2016, 59, 142–151. [Google Scholar] [CrossRef]
- Incrocci, L.; Wortel, R.C.; Alemayehu, W.G.; Aluwini, S.; Schimmel, E.; Krol, S.; van der Toorn, P.; de Jager, H.; Heemsbergen, W.; Heijmen, B.; et al. Hypofractionated versus conventionally fractionated radiotherapy for patients with localised prostate cancer (HYPRO): Final efficacy results from a randomised, multicentre, open-label, phase 3 trial. Lancet Oncol. 2016, 17, 1061–1069. [Google Scholar] [CrossRef]
- Kang, J.; Cho, C.K.; Choi, C.W.; Yoo, S.; Kim, M.; Yang, K.; Yoo, H.; Kim, J.H.; Seo, Y.S.; Lee, D.H.; et al. Image-guided stereotactic body radiation therapy for localized prostate cancer. Tumori 2011, 97, 43–48. [Google Scholar] [CrossRef]
- Katz, A.; Formenti, S.C.; Kang, J. Predicting Biochemical Disease-Free survival after Prostate stereotactic Body radiotherapy: Risk-stratification and Patterns of Failure. Front. Oncol. 2016, 6, 168. [Google Scholar] [CrossRef]
- King, C.R.; Brooks, J.D.; Gill, H.; Presti, J.C. Long-term outcomes from a prospective trial of stereotactic body radiotherapy for low-risk prostate cancer. Int. J. Radiat. Oncol. Biol. Phys. 2012, 82, 877–882. [Google Scholar] [CrossRef]
- Kuban, D.A.; Nogueras-Gonzalez, G.M.; Hamblin, L.; Lee, A.K.; Choi, S.; Frank, S.J.; Nguyen, Q.N.; Hoffman, K.E.; McGuire, S.E.; Munsell, M.F. Preliminary Report of a Randomized Dose Escalation Trial for Prostate Cancer using Hypofractionation. Int. J. Radiat. Oncol. Biol. Phys. 2010, 78, S58–S59. [Google Scholar] [CrossRef]
- Kupelian, P.A.; Willoughby, T.R.; Reddy, C.A.; Klein, E.A.; Mahadevan, A. Hypofractionated intensity-modulated radiotherapy (70 Gy at 2.5 Gy per fraction) for localized prostate cancer: Cleveland Clinic experience. Int. J. Radiat. Oncol. Biol. Phys. 2007, 68, 1424–1430. [Google Scholar] [CrossRef] [PubMed]
- Lee, W.R.; Dignam, J.J.; Amin, M.B.; Bruner, D.W.; Low, D.; Swanson, G.P.; Shah, A.B.; D’Souza, D.P.; Michalski, J.M.; Dayes, I.S.; et al. Randomized Phase III Noninferiority Study Comparing Two Radiotherapy Fractionation Schedules in Patients With Low-Risk Prostate Cancer. J. Clin. Oncol. 2016, 34, 2325–2332. [Google Scholar] [CrossRef] [PubMed]
- Levin-Epstein, R.G.; Jiang, N.Y.; Wang, X.; Upadhyaya, S.K.; Collins, S.P.; Guy, S.; Aghdam, N.; Mantz, C.; Katz, A.J.; Miszczyk, L.; et al. Dose–response with stereotactic body radiotherapy for prostate cancer: A multi-institutional analysis of prostate-specific antigen kinetics and biochemical control. Radiother. Oncol. 2021, 154, 207–213. [Google Scholar] [CrossRef]
- Lukka, H.; Hayter, C.; Julian, J.A.; Warde, P.; Morris, W.J.; Gospodarowicz, M.; Levine, M.; Sathya, J.; Choo, R.; Prichard, H.; et al. Randomized Trial Comparing Two Fractionation Schedules for Patients With Localized Prostate Cancer. J. Clin. Oncol. 2005, 23, 6132–6138. [Google Scholar] [CrossRef]
- Madsen, B.L.; Hsi, R.A.; Pham, H.T.; Fowler, J.F.; Esagui, L.; Corman, J. Stereotactic hypofractionated accurate radiotherapy of the prostate (SHARP), 33.5 Gy in five fractions for localized disease: First clinical trial results. Int. J. Radiat. Oncol. Biol. Phys. 2007, 67, 1099–1105. [Google Scholar] [CrossRef]
- Mantz, C. A phase II trial of stereotactic ablative body radiotherapy for low-risk prostate cancer using a non-robotic linear accelerator and real-time target tracking: Report of toxicity, quality of life, and disease control outcomes with 5-year minimum follow-up. Front. Oncol. 2014, 4, 279. [Google Scholar] [CrossRef]
- McBride, S.M.; Wong, D.S.; Dombrowski, J.J.; Harkins, B.; Tapella, P.; Hanscom, H.N.; Collins, S.P.; Kaplan, I.D. Hypofractionated Stereotactic Body Radiotherapy in Low-Risk Prostate Adenocarcinoma. Cancer 2012, 118, 3681–3690. [Google Scholar] [CrossRef]
- Yeoh, E.E.; Holloway, R.H.; Fraser, R.J.; Botten, R.J.; Di Matteo, A.C.; Butters, J.; Weerasinghe, S.; Abeysinghe, P. Hypofractionated versus conventional fractionated radiation therapy for prostate carcinoma: Updated results of a phase III randomized trial. Int. J. Radiat. Oncol. Biol. Phys. 2006, 66, 1072–1083. [Google Scholar] [CrossRef]
- Zelefsky, M.J.; Fuks, Z.; Hunt, M.; Yamada, Y.; Marion, C.; Ling, C.C.; Amols, H.; Venkatraman, E.S.; Leibel, S.A. High-dose intensity modulated radiation therapy for prostate cancer: Early toxicity and biochemical outcome in 772 patients. Int. J. Radiat. Oncol. Biol. Phys. 2002, 53, 1111–1116. [Google Scholar] [CrossRef]
- Bentzen, S.M.; Tucker, S.L. Quantifying the position and steepness of radiation dose–response curves. Int. J. Radiat. Biol. 1997, 71, 531–542. [Google Scholar]
- Royston, P. Profile likelihood for estimation and confidence intervals. Stata J. 2007, 7, 376–387. [Google Scholar] [CrossRef]
- Rao, S.S. The Profile Likelihood. Available online: https://web.stat.tamu.edu/suhasini/teaching613/chapter3.pdf (accessed on 1 January 2023).
- Akaike, H. A new look at the statistical model identification. IEEE Trans. Autom. Control 1974, 19, 716–723. [Google Scholar] [CrossRef]
- Webb, S.; Nahum, A.E. A model for calculating tumour control probability in radiotherapy including the effects of inhomogeneous distributions of dose and clonogenic cell density. Phys. Med. Biol. 1993, 38, 653–666. [Google Scholar] [CrossRef]
- Fowler, J.F. 21 years of biologically effective dose. Br. J. Radiol. 2010, 83, 554–568. [Google Scholar] [CrossRef]
- Shuryak, I.; Carlson, D.J.; Brown, M.; Brenner, D.J. High-dose and fractionation effects in stereotactic radiotherapy: Analysis of tumour control data from 2965 patients. Radiother. Oncol. 2015, 115, 327–334. [Google Scholar] [CrossRef]
- Symonds, M.R.; Moussalli, A. A brief guide to model selection, multimodel inference and model averaging in behavioural ecology using Akaike’s information criterion. Behav. Ecol. Sociobiol. 2011, 65, 13–21. [Google Scholar] [CrossRef]
- Kuperman, V.Y.; Lubich, L.M. Effect of reoxygenation on hypofractionated radiotherapy of prostate cancer. Med. Phys. 2020, 47, 5383–5391. [Google Scholar] [CrossRef]
- Kerns, S.L.; Fachal, L.; Dorling, L.; Barnett, G.C.; Baran, A.; Peterson, D.R.; Hollenberg, M.; Hao, K.; Narzo, A.D.; Ahsen, M.E.; et al. Radiogenomics Consortium Genome-Wide Association Study Meta-Analysis of Late Toxicity After Prostate Cancer Radiotherapy. JNCI J. Natl. Cancer Inst. 2020, 112, 179–190. [Google Scholar] [CrossRef]
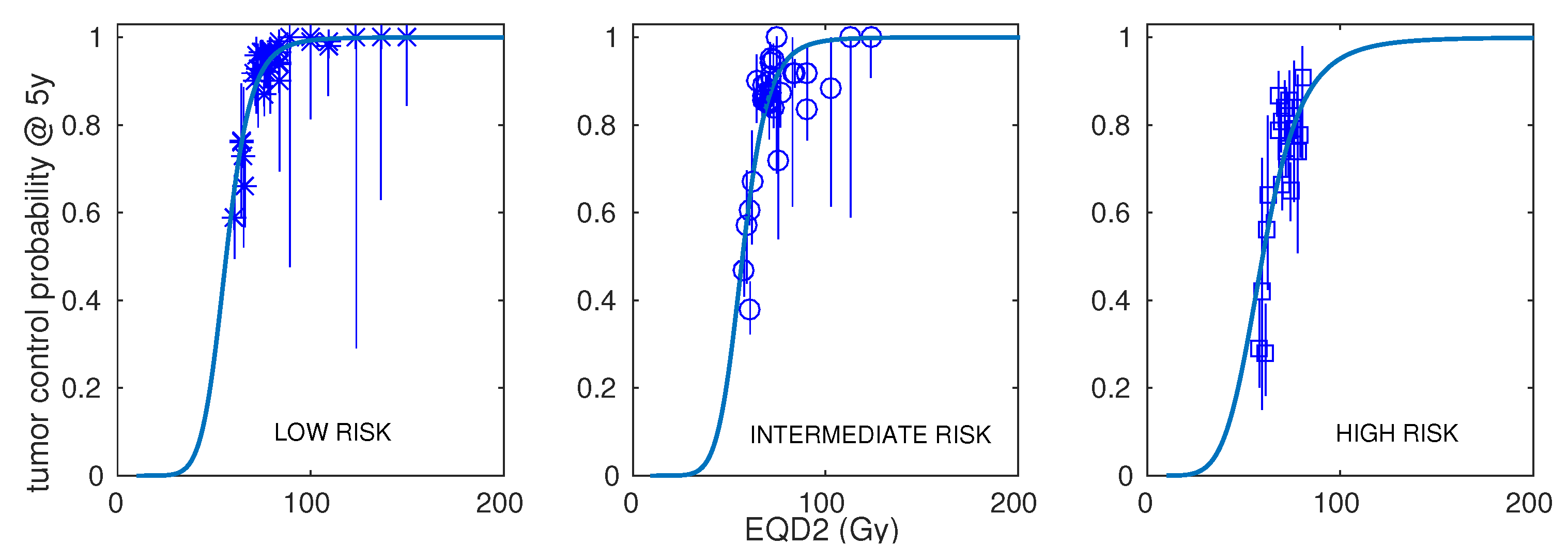
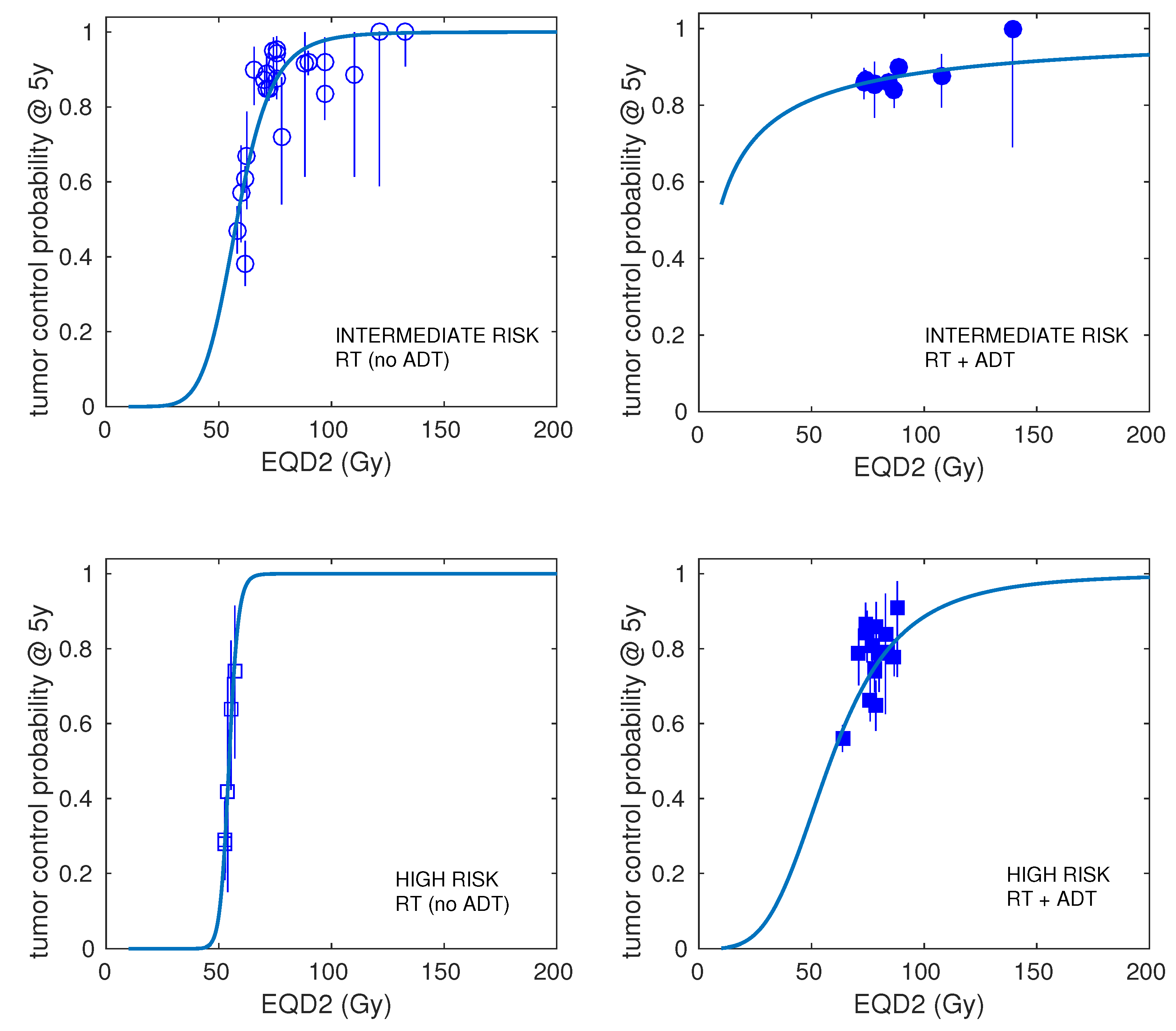
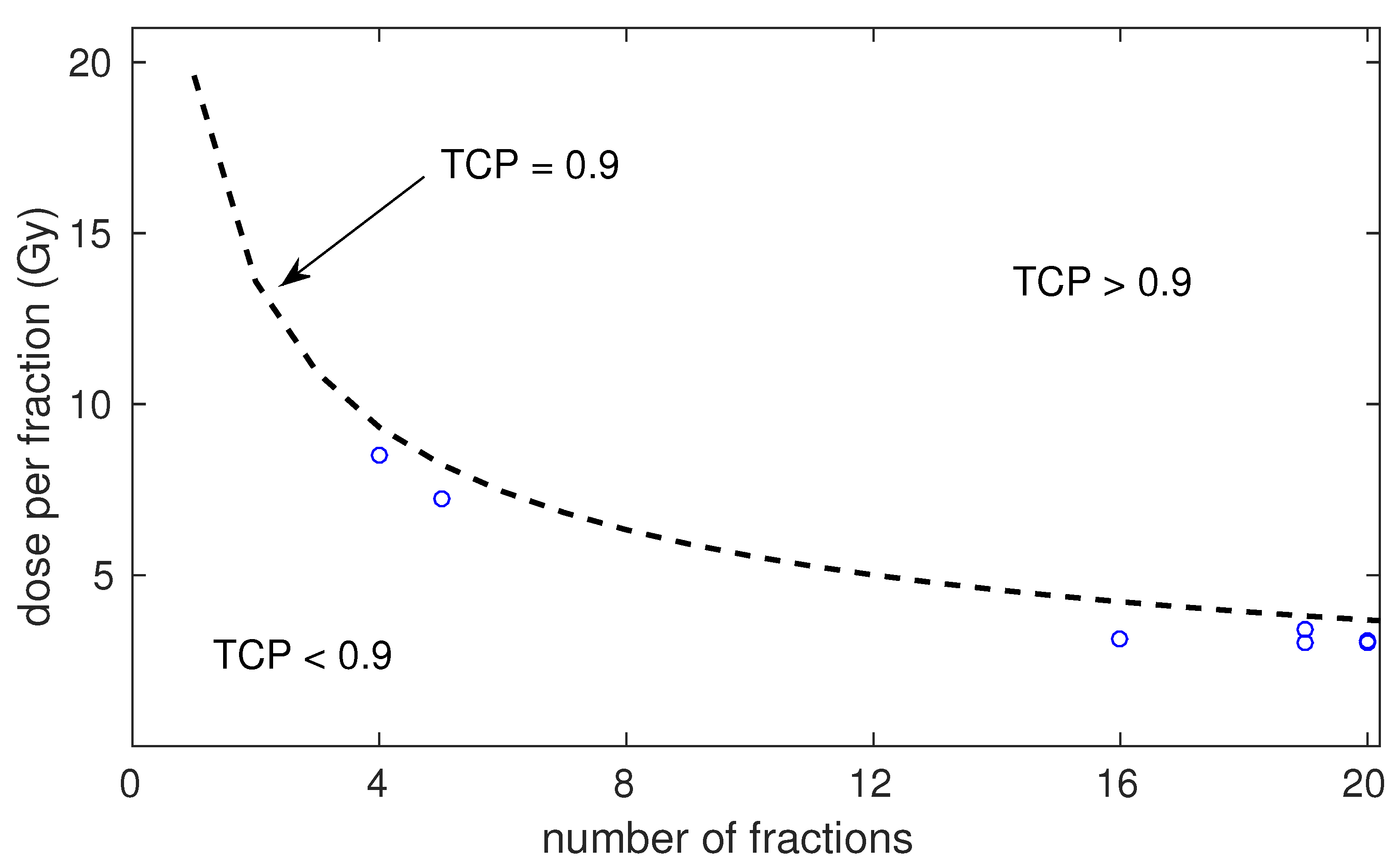
| Risk | Number of Schedules | Number of Patients (Range) | Dose per Fraction (Range) | Total Dose (Range) | Treatment Time (Range) | ADT (Fraction of Schedules) | Control at 5 Years (Range) |
|---|---|---|---|---|---|---|---|
| LR | 35 | 3–550 | 1.8–10 Gy | 33.5–81 Gy | 3–62 days | 3/35 | 0.59–1.00 |
| IR | 32 | 7–839 | 1.8–10 Gy | 34–81 Gy | 3–62 days | 9/32 | 0.38–1.00 |
| HR | 20 | 12–812 | 1.8–8.5 Gy | 34–81 Gy | 3–62 days | 15/20 | 0.28–0.908 |
| Risk | Model | Parameters | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| (Gy) | (Gy day) | (day) | (Gy) | (Gy) | (Gy) | ||||||
| LR | LQ | 2.0 | 0.00 | - | - | - | 56.2 | 2.17 | 89.4 | 190.8 | - |
| LQL | 2.0 | 0.00 | - | 0.00 | - | 56.2 | 2.17 | 89.4 | 193.7 | −2.9 | |
| LQ | 2.6 | 0.00 | - | - | 0.07 | 55.8 | 2.11 | 89.2 | 193.4 | −2.6 | |
| IR | LQ | 3.4 | 0.41 | 24.0 | - | - | 56.9 | 2.14 | 220.9 | 454.2 | - |
| LQL | 0.4 | 0.00 | - | 0.28 | - | 62.8 | 2.18 | 201.3 | 418.0 | 36.2 | |
| LQ | 3.5 | 0.41 | 23.9 | - | 0.00 | 56.8 | 2.15 | 220.9 | 457.2 | −3.0 | |
| HR | LQ | 2.8 | 0.35 | 21.0 * | - | - | 59.8 | 1.45 | 105.0 | 224.4 | - |
| LQL | 2.8 | 0.35 | 21.0 * | 0.00 | - | 59.8 | 1.45 | 105.0 | 228.5 | −4.2 | |
| LQ | 11.2 | 0.34 | 21.0 * | - | 0.75 | 58.7 | 1.47 | 103.9 | 226.2 | −1.8 | |
| IR | LQ | 2.8 | 0.32 | 21.0 * | - | - | 58.1 | 1.85 | 157.6 | 328.7 | - |
| (no ADT) | LQL | 0.5 | 0.00 | - | 0.24 | - | 63.6 | 2.01 | 138.7 | 294.6 | 34.1 |
| LQ | 2.8 | 0.32 | 21.0 * | - | 0.00 | 58.1 | 1.85 | 157.6 | 332.4 | −3.7 | |
| HR | LQ | 2.1 | 0.00 | - | - | - | 58.5 | 0.95 | 72.6 | 161.8 | - |
| (ADT) | LQL | 2.1 | 0.00 | - | 0.00 | - | 58.5 | 0.95 | 72.5 | 167.6 | -5.8 |
| LQ | 18.7 | 0.00 | - | - | 1.99 | 56.8 | 0.88 | 71.2 | 164.9 | −3.1 | |
| (Gy) | (Gy day) | (day) | (Gy) | ||
|---|---|---|---|---|---|
| LR | 2.0 | 0 | - | 56.2 | 2.17 |
| (1.7, 2.3) | (0, 0.13) | (54.4, 58.0) | (1.90, 2.47) | ||
| IR | 3.4 | 0.41 | 24.0 | 56.9 | 2.14 |
| (3.0, 4.0) | (0.31, 0.49) | (21.0, 25.5) | (55.5, 57.9) | (1.92, 2.40) | |
| HR | 2.8 | 0.35 | 21.0 | 59.8 | 1.45 |
| (1.4, 4.2) | (0, ∞) | (21.0, ∞) | (57.1, 63.9) | (1.07, 1.83) | |
| IR | 2.8 | 0.32 | 21.0 | 58.1 | 1.85 |
| (no ADT) | (2.1, 3.5) | (0.09, 0.46) | (21.0, 27.3) | (56.5, 60.0) | (1.55, 2.14) |
| HR | 2.1 | 0 | - | 58.5 | 0.95 |
| (ADT) | (1.5, 3.5) | (0, 0.31) | (54.3, 61.5) | (0.75, 1.25) | |
| IR | 0.1 | 0 | - | 8.1 | 0.20 |
| (ADT) | (0, ∞) | (0, ∞) | (0.4, 40.5) | (0.11, 0.80) | |
| HR | 100.0 | 3.31 | 39.9 | 54.6 | 6.68 |
| (no ADT) | (7.1, ∞) | (1.09, ∞) | (21.0, 40.8) | (49.8, 60.5) | (2.70, 10.67) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pardo-Montero, J.; González-Crespo, I.; Gómez-Caamaño, A.; Gago-Arias, A. Radiobiological Meta-Analysis of the Response of Prostate Cancer to Different Fractionations: Evaluation of the Linear–Quadratic Response at Large Doses and the Effect of Risk and ADT. Cancers 2023, 15, 3659. https://doi.org/10.3390/cancers15143659
Pardo-Montero J, González-Crespo I, Gómez-Caamaño A, Gago-Arias A. Radiobiological Meta-Analysis of the Response of Prostate Cancer to Different Fractionations: Evaluation of the Linear–Quadratic Response at Large Doses and the Effect of Risk and ADT. Cancers. 2023; 15(14):3659. https://doi.org/10.3390/cancers15143659
Chicago/Turabian StylePardo-Montero, Juan, Isabel González-Crespo, Antonio Gómez-Caamaño, and Araceli Gago-Arias. 2023. "Radiobiological Meta-Analysis of the Response of Prostate Cancer to Different Fractionations: Evaluation of the Linear–Quadratic Response at Large Doses and the Effect of Risk and ADT" Cancers 15, no. 14: 3659. https://doi.org/10.3390/cancers15143659
APA StylePardo-Montero, J., González-Crespo, I., Gómez-Caamaño, A., & Gago-Arias, A. (2023). Radiobiological Meta-Analysis of the Response of Prostate Cancer to Different Fractionations: Evaluation of the Linear–Quadratic Response at Large Doses and the Effect of Risk and ADT. Cancers, 15(14), 3659. https://doi.org/10.3390/cancers15143659






