Efficient Heat Shock Response Affects Hyperthermia-Induced Radiosensitization in a Tumor Spheroid Control Probability Assay
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Cell Lines and Culture Conditions
2.2. Spheroid Routine Culturing and Monitoring
2.3. Hyperthermia and Irradiation Treatment
2.4. Exposure to Chemical Chaperones
2.5. Spheroid Volume Growth Delay and Spheroid Control Probability (SCP) Assays
2.6. Reverse Transcription (RT-) and Real-Time Quantitative-(q-)PCR Analyses
2.7. Western Blotting
2.8. Transcriptomics
3. Results
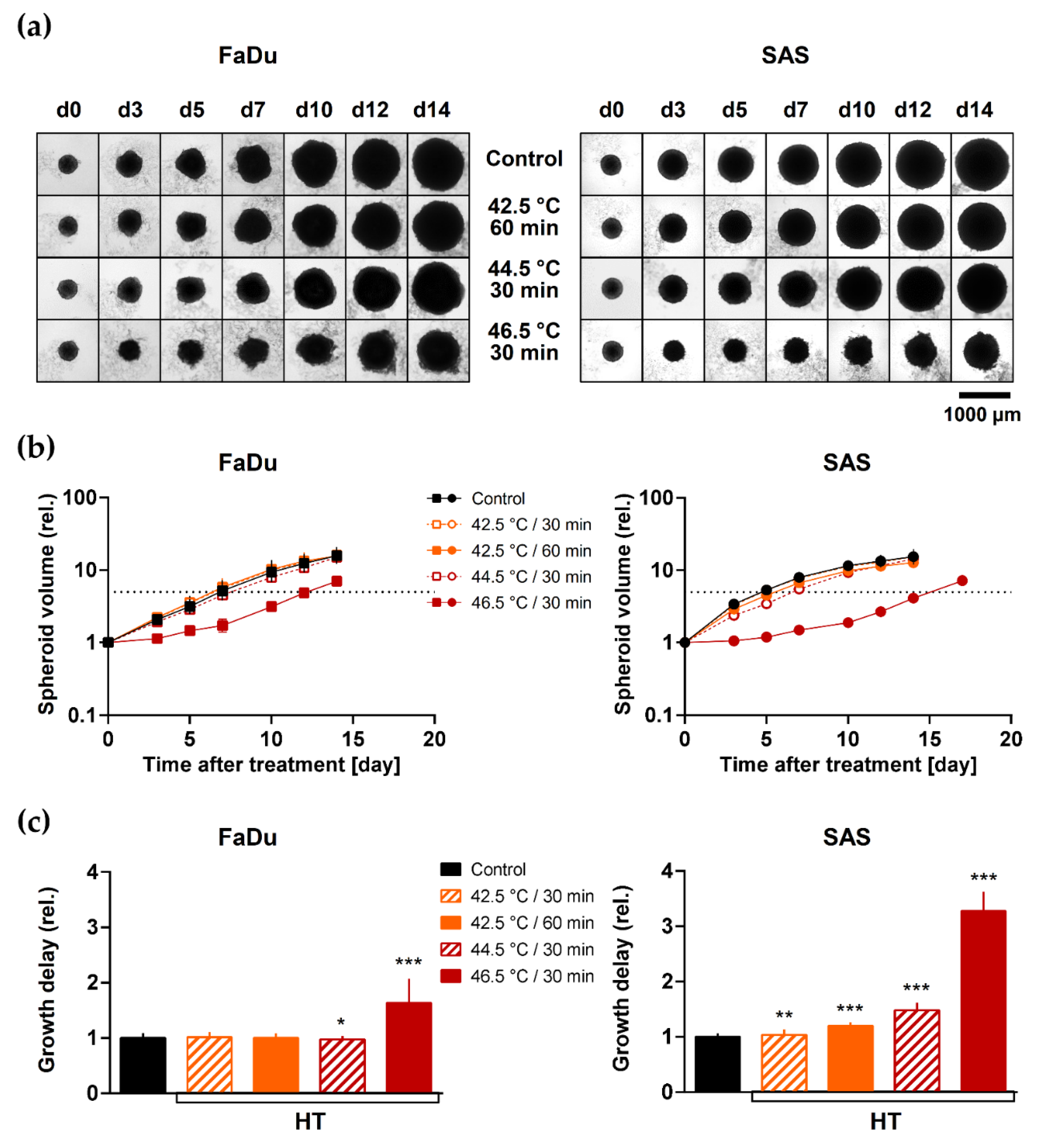
3.1. HT Induces Minimal Spheroid Growth Delay at Clinically Relevant Temperatures
3.2. HT Induces Sensitization of HNSCC Spheroids to Single Dose Irradiation
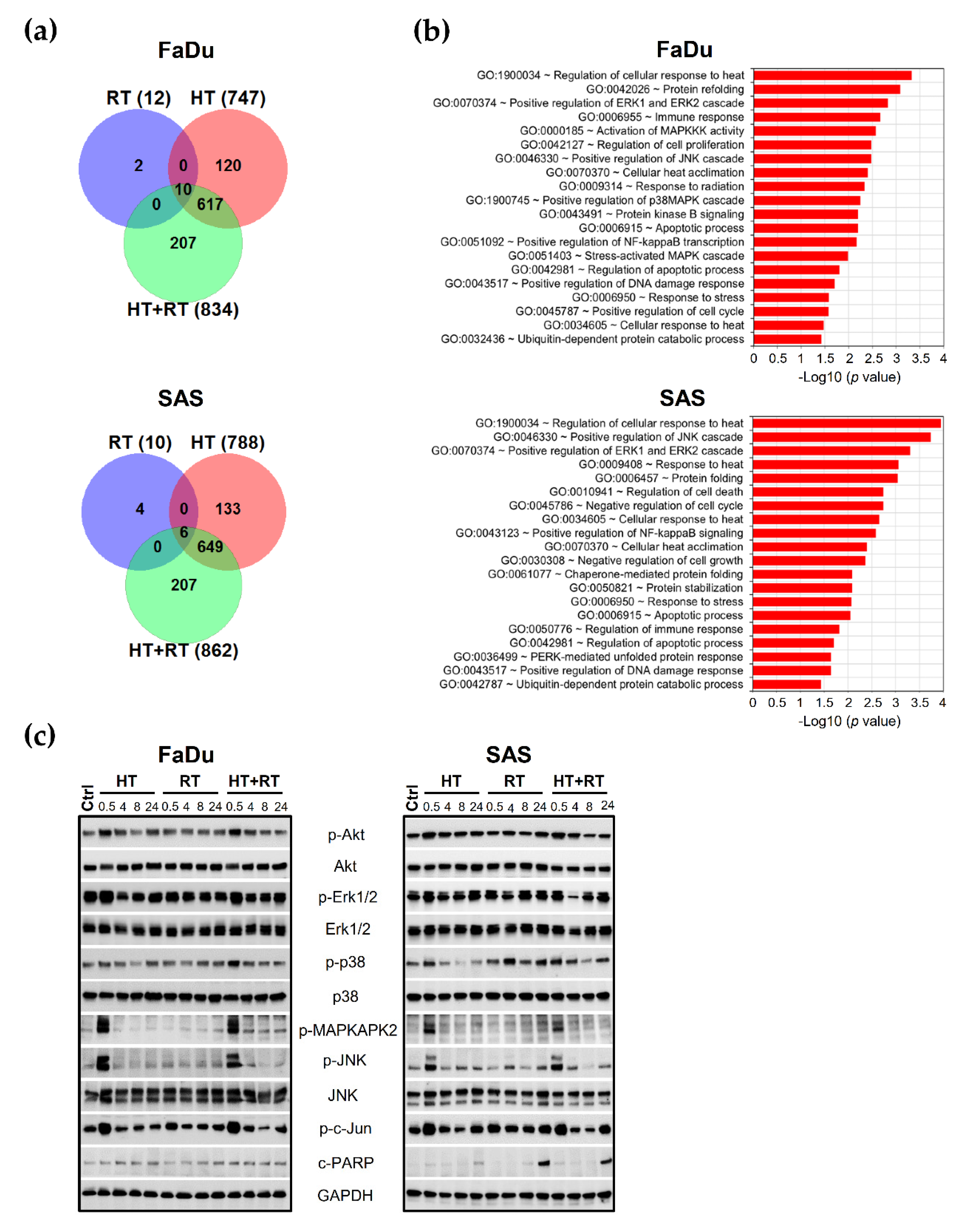
3.3. Global Changes in Gene Expression Profiles of HNSCC Spheroids upon HT + RT
3.4. HT Affects Main Survival and Stress Response Signaling in HNSCC Spheroids
3.5. HT Triggers Heat Shock Response (HSR) and Proteotoxic Stress in HNSCC Spheroids
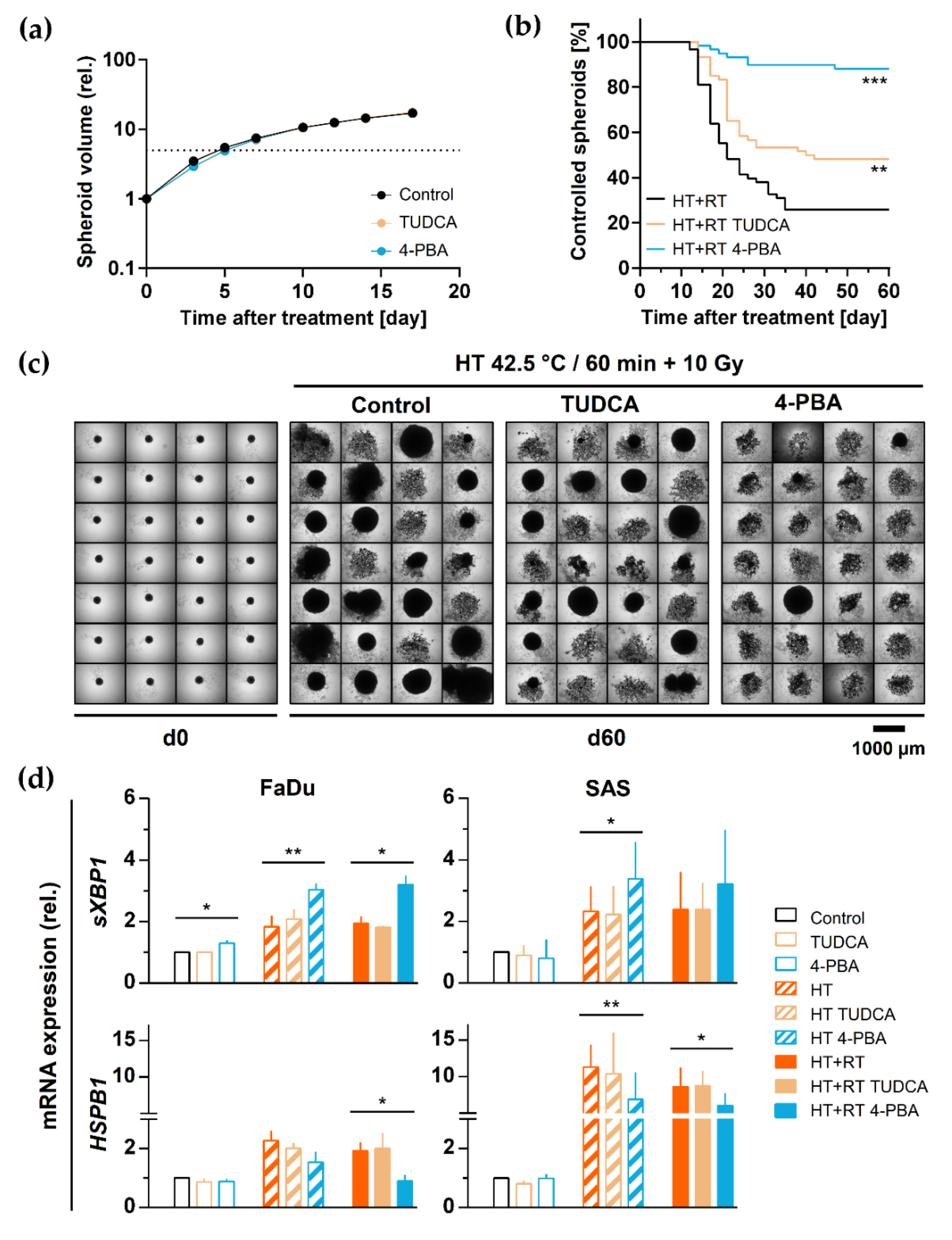
3.6. HT-Induced Radiosensitization Is Adversely Affected by the Proteotoxic Stress Response
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Datta, N.; Ordóñez, S.G.; Gaipl, U.; Paulides, M.; Crezee, H.; Gellermann, J.; Marder, D.; Puric, E.; Bodis, S. Local hyperthermia combined with radiotherapy and-/or chemotherapy: Recent advances and promises for the future. Cancer Treat. Rev. 2015, 41, 742–753. [Google Scholar] [CrossRef]
- Crezee, J.; Van Leeuwen, C.M.; Oei, A.L.; Stalpers, L.J.A.; Bel, A.; Franken, N.; Kok, H. Thermoradiotherapy planning: Integration in routine clinical practice. Int. J. Hyperth. 2015, 32, 41–49. [Google Scholar] [CrossRef] [Green Version]
- Peeken, J.C.; Vaupel, P.; Combs, S.E. Integrating Hyperthermia into Modern Radiation Oncology: What Evidence Is Necessary? Front. Oncol. 2017, 7, 7. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Datta, N.R.; Kok, H.P.; Crezee, H.; Gaipl, U.S.; Bodis, S. Integrating Loco-Regional Hyperthermia Into the Current Oncology Practice: SWOT and TOWS Analyses. Front. Oncol. 2020, 10, 819. [Google Scholar] [CrossRef] [PubMed]
- Kang, J.K.; Kim, J.C.; Shin, Y.; Han, S.M.; Won, W.R.; Her, J.; Park, J.Y.; Oh, K.T. Principles and applications of nanomaterial-based hyperthermia in cancer therapy. Arch. Pharmacal Res. 2020, 43, 46–57. [Google Scholar] [CrossRef] [PubMed]
- Kok, H.P.; Cressman, E.N.K.; Ceelen, W.; Brace, C.L.; Ivkov, R.; Grüll, H.; Ter Haar, G.; Wust, P.; Crezee, J. Heating technology for malignant tumors: A review. Int. J. Hyperth. 2020, 37, 711–741. [Google Scholar] [CrossRef] [PubMed]
- Paulides, M.M.; Verduijn, G.M.; Van Holthe, N. Status quo and directions in deep head and neck hyperthermia. Radiat. Oncol. 2016, 11, 21. [Google Scholar] [CrossRef] [Green Version]
- Gao, S.; Zheng, M.; Ren, X.; Tang, Y.; Liang, X. Local hyperthermia in head and neck cancer: Mechanism, application and advance. Oncotarget 2016, 7, 57367–57378. [Google Scholar] [CrossRef] [Green Version]
- Baumann, M.; Krause, M.; Zips, D.; Eicheler, W.; Dörfler, A.; Ahrens, J.; Petersen, C.; Brüchner, K.; Hilberg, F. Selective inhibition of the epidermal growth factor receptor tyrosine kinase by BIBX1382BS and the improvement of growth delay, but not local control, after fractionated irradiation in human FaDu squamous cell carcinoma in the nude mouse. Int. J. Radiat. Biol. 2003, 79, 547–559. [Google Scholar] [CrossRef] [PubMed]
- Krause, M.; Prager, J.; Zhou, X.; Yaromina, A.; Dörfler, A.; Eicheler, W.; Baumann, M. EGFR-TK inhibition before radiotherapy reduces tumour volume but does not improve local control: Differential response of cancer stem cells and nontumourigenic cells? Radiother. Oncol. 2007, 83, 316–325. [Google Scholar] [CrossRef]
- Yaromina, A.; Krause, M.; Thames, H.; Rosner, A.; Krause, M.; Hessel, F.; Grenman, R.; Zips, D.; Baumann, M. Pre-treatment number of clonogenic cells and their radiosensitivity are major determinants of local tumour control after fractionated irradiation. Radiother. Oncol. 2007, 83, 304–310. [Google Scholar] [CrossRef]
- Yaromina, A.; Thames, H.; Zhou, X.; Hering, S.; Eicheler, W.; Dörfler, A.; Leichtner, T.; Zips, D.; Baumann, M. Radiobiological hypoxia, histological parameters of tumour microenvironment and local tumour control after fractionated irradiation. Radiother. Oncol. 2010, 96, 116–122. [Google Scholar] [CrossRef]
- Kunz-Schughart, L.; Freyer, J.P.; Hofstaedter, F.; Ebner, R. The Use of 3-D Cultures for High-Throughput Screening: The Multicellular Spheroid Model. J. Biomol. Screen. 2004, 9, 273–285. [Google Scholar] [CrossRef] [Green Version]
- Friedrich, J.; Ebner, R.; Kunz-Schughart, L. Experimental anti-tumor therapy in 3-D: Spheroids—old hat or new challenge? Int. J. Radiat. Biol. 2007, 83, 849–871. [Google Scholar] [CrossRef] [PubMed]
- Costa, E.C.; Moreira, A.F.; De Melo-Diogo, D.; Gaspar, V.; Carvalho, M.P.; Correia, I.J. 3D tumor spheroids: An overview on the tools and techniques used for their analysis. Biotechnol. Adv. 2016, 34, 1427–1441. [Google Scholar] [CrossRef] [PubMed]
- Hirschhaeuser, F.; Menne, H.; Dittfeld, C.; West, J.; Mueller-Klieser, W.; Kunz-Schughart, L.A. Multicellular tumor spheroids: An underestimated tool is catching up again. J. Biotechnol. 2010, 148, 3–15. [Google Scholar] [CrossRef] [PubMed]
- Weiswald, L.-B.; Bellet, D.; Dangles-Marie, V. Spherical Cancer Models in Tumor Biology. Neoplasia 2015, 17, 1–15. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Durand, R.E. Effects of Hyperthermia on the Cycling, Noncycling, and Hypoxic Cells of Irradiated and Unirradiated Mul-ticell Spheroids. Radiat. Res. 1978, 75, 373–384. [Google Scholar] [CrossRef]
- Lücke-Huhle, C.; Dertinger, H. “Tumour-Model” (V 79 Spheroids) to 42 °C Hyperthermia. Eur. J. Cancer 1977, 13, 23–28. [Google Scholar] [CrossRef]
- Dertinger, H.; Seiter, A. Recovery and cycle progression in multicell spheroids after fractionated gamma-irradiation and combined hyperthermic treatment (author’s transl). Strahlentherapie 1979, 155, 39–43. [Google Scholar]
- Neshasteh-Riz, A.; Rahdani, R.; Mostaar, A. Evaluation of The Combined Effects of Hyperthermia, Cobalt-60 Gamma Rays and IUdR on Cultured Glioblastoma Spheroid Cells and Dosimetry Using TLD-100. Cell J. 2014, 16, 335–342. [Google Scholar]
- Asayesh, T.; Changizi, V.; Eyvazzadeh, N. Assessment of cytotoxic damage induced by irradiation combined with hyper-thermia and Gemcitabine on cultured glioblastoma spheroid cells. Radiat. Phys. Chem. 2016, 120, 44–48. [Google Scholar] [CrossRef]
- Mahdavi, S.R.; Yahyapour, R.; Nikoofar, A. Cytotoxic effects of hyperthermia, chemotherapy (Navelbine) and radiation on glioma spheroids. Radiat. Phys. Chem. 2016, 123, 20–24. [Google Scholar] [CrossRef]
- Esfahani, A.J.; Mahdavi, S.R.; Shiran, M.B.; Khoei, S. The Role of Radiofrequency Hyperthermia in The Radiosensitization of A Human Prostate Cancer Cell Line. Cell J. 2017, 19, 86–95. [Google Scholar] [CrossRef]
- Mahdavi, S.R.; Esfahani, A.J.; Shiran, M.B.; Khoei, S.; Estiri, N. Enhanced DNA Damages of Human Prostate Cancer Cells Induced by Radiofrequency Capacitive Hyperthermia Pre- and Post X-rays: 6 MV versus 15 MV. Cell J. 2017, 19, 79–85. [Google Scholar] [CrossRef] [PubMed]
- Rajaee, Z.; Khoei, S.; Mahdavi, S.R.; Ebrahimi, M.; Shirvalilou, S.; Mahdavian, A. Evaluation of the effect of hyperthermia and electron radiation on prostate cancer stem cells. Radiat. Environ. Biophys. 2018, 57, 133–142. [Google Scholar] [CrossRef]
- Brüningk, S.C.; Ziegenhein, P.; Rivens, I.; Oelfke, U.; Ter Haar, G. A cellular automaton model for spheroid response to radiation and hyperthermia treatments. Sci. Rep. 2019, 9, 17674. [Google Scholar] [CrossRef]
- Brüningk, S.C.; Rivens, I.; Box, C.; Oelfke, U.; Ter Haar, G. 3D tumour spheroids for the prediction of the effects of radiation and hyperthermia treatments. Sci. Rep. 2020, 10, 1653. [Google Scholar] [CrossRef]
- Friedrich, J.; Seidel, C.; Ebner, R.; Kunz-Schughart, L. Spheroid-based drug screen: Considerations and practical approach. Nat. Protoc. 2009, 4, 309–324. [Google Scholar] [CrossRef]
- Ingargiola, M.; Runge, R.; Heldt, J.-M.; Freudenberg, R.; Steinbach, J.; Cordes, N.; Baumann, M.; Kotzerke, J.; Brockhoff, G.; Kunz-Schughart, L. Potential of a Cetuximab-based radioimmunotherapy combined with external irradiation manifests in a 3-D cell assay. Int. J. Cancer 2014, 135, 968–980. [Google Scholar] [CrossRef] [Green Version]
- Hinrichs, C.N.; Ingargiola, M.; Käubler, T.; Löck, S.; Temme, A.; Köhn-Luque, A.; Deutsch, A.; Vovk, O.; Stasyk, O.; Kunz-Schughart, L.A. Arginine Deprivation Therapy: Putative Strategy to Eradicate Glioblastoma Cells by Radiosensitization. Mol. Cancer Ther. 2017, 17, 393–406. [Google Scholar] [CrossRef] [Green Version]
- Kaur, P.; Hurwitz, M.D.; Krishnan, S.; Asea, A. Combined Hyperthermia and Radiotherapy for the Treatment of Cancer. Cancers 2011, 3, 3799–3823. [Google Scholar] [CrossRef]
- Oei, A.L.; Vriend, L.E.M.; Crezee, J.; Franken, N.A.P.; Krawczyk, P.M. Effects of hyperthermia on DNA repair pathways: One treatment to inhibit them all. Radiat. Oncol. 2015, 10, 165. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Richter, K.; Haslbeck, M.; Buchner, J. The Heat Shock Response: Life on the Verge of Death. Mol. Cell 2010, 40, 253–266. [Google Scholar] [CrossRef]
- Schröder, M.; Kaufman, R.J. The mammalian unfolded protein response. Annu. Rev. Biochem. 2005, 74, 739–789. [Google Scholar] [CrossRef] [PubMed]
- Vahid, S.; Thaper, D.; Zoubeidi, A. Chaperoning the Cancer: The Proteostatic Functions of the Heat Shock Proteins in Cancer. Recent Pat. Anticancer. Drug Discov. 2017, 12, 35–47. [Google Scholar] [CrossRef] [PubMed]
- Eicheler, W.; Zips, D.; Dörfler, A.; Grénman, R.; Baumann, M. Splicing Mutations in TP53 in Human Squamous Cell Carcinoma Lines Influence Immunohistochemical Detection. J. Histochem. Cytochem. 2002, 50, 197–204. [Google Scholar] [CrossRef] [Green Version]
- Chen, O.; Manig, F.; Lehmann, L.; Sorour, N.; Löck, S.; Yu, Z.; Dubrovska, A.; Baumann, M.; Kessler, B.M.; Stasyk, O.; et al. Dual role of ER stress in response to metabolic co-targeting and radiosensitivity in head and neck cancer cells. Cell. Mol. Life Sci. 2021, 78, 3021–3044. [Google Scholar] [CrossRef]
- Walker, A.M.; Suit, H.D. Assessment of local tumor control using censored tumor response data. Int. J. Radiat. Oncol. 1983, 9, 383–386. [Google Scholar] [CrossRef]
- Vynnytska-Myronovska, B.O.; Kurlishchuk, Y.; Chen, O.; Bobak, Y.; Dittfeld, C.; Hüther, M.; Kunz-Schughart, L.; Stasyk, O.V. Arginine starvation in colorectal carcinoma cells: Sensing, impact on translation control and cell cycle distribution. Exp. Cell Res. 2016, 341, 67–74. [Google Scholar] [CrossRef]
- Gurgis, F.M.S.; Ziaziaris, W.; Munoz, L. Mitogen-Activated Protein Kinase–Activated Protein Kinase 2 in Neuroinflammation, Heat Shock Protein 27 Phosphorylation, and Cell Cycle: Role and Targeting. Mol. Pharmacol. 2013, 85, 345–356. [Google Scholar] [CrossRef] [Green Version]
- Leppä, S.; Bohmann, D. Diverse functions of JNK signaling and c-Jun in stress response and apoptosis. Oncogene 1999, 18, 6158–6162. [Google Scholar] [CrossRef] [Green Version]
- Potapova, O.; Basu, S.; Mercola, D.; Holbrook, N.J. Protective Role for c-Jun in the Cellular Response to DNA Damage. J. Biol. Chem. 2001, 276, 28546–28553. [Google Scholar] [CrossRef] [Green Version]
- Yoshida, H.; Matsui, T.; Yamamoto, A.; Okada, T.; Mori, K. XBP1 mRNA Is Induced by ATF6 and Spliced by IRE1 in Response to ER Stress to Produce a Highly Active Transcription Factor. Cell 2001, 107, 881–891. [Google Scholar] [CrossRef] [Green Version]
- Arrigo, A.-P. Mammalian HspB1 (Hsp27) is a molecular sensor linked to the physiology and environment of the cell. Cell Stress Chaperon. 2017, 22, 517–529. [Google Scholar] [CrossRef] [Green Version]
- Aloy, M.-T.; Hadchity, E.; Bionda, C.; Diaz-Latoud, C.; Claude, L.; Rousson, R.; Arrigo, A.-P.; Rodriguez-Lafrasse, C. Protective Role of Hsp27 Protein Against Gamma Radiation–Induced Apoptosis and Radiosensitization Effects of Hsp27 Gene Silencing in Different Human Tumor Cells. Int. J. Radiat. Oncol. 2008, 70, 543–553. [Google Scholar] [CrossRef] [PubMed]
- Alsahafi, E.; Begg, K.; Amelio, I.; Raulf, N.; Lucarelli, P.; Sauter, T.; Tavassoli, M. Clinical update on head and neck cancer: Molecular biology and ongoing challenges. Cell Death Dis. 2019, 10, 1–17. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cramer, J.D.; Burtness, B.; Le, Q.T.; Ferris, R.L. The changing therapeutic landscape of head and neck cancer. Nat. Rev. Clin. Oncol. 2019, 16, 669–683. [Google Scholar] [CrossRef]
- Roti, J.L.R. Cellular responses to hyperthermia (40–46°C): Cell killing and molecular events. Int. J. Hyperth. 2008, 24, 3–15. [Google Scholar] [CrossRef] [PubMed]
- Krawczyk, P.M.; Eppink, B.; Essers, J.; Stap, J.; Rodermond, H.; Odijk, H.; Zelensky, A.; van Bree, C.; Stalpers, L.J.; Buist, M.R.; et al. Mild hyperthermia inhibits homologous recombination, induces BRCA2 degradation, and sensitizes cancer cells to poly (ADP-ribose) polymerase-1 inhibition. Proc. Natl. Acad. Sci. USA 2011, 108, 9851–9856. [Google Scholar] [CrossRef] [Green Version]
- Ihara, M.; Takeshita, S.; Okaichi, K.; Okumura, Y.; Ohnishi, T. Heat exposure enhances radiosensitivity by depressing DNA-PK kinase activity during double strand break repair. Int. J. Hyperth. 2014, 30, 102–109. [Google Scholar] [CrossRef]
- Hetz, C. The unfolded protein response: Controlling cell fate decisions under ER stress and beyond. Nat. Rev. Mol. Cell Biol. 2012, 13, 89–102. [Google Scholar] [CrossRef]
- Morimoto, R.I. Proteotoxic stress and inducible chaperone networks in neurodegenerative disease and aging. Genes Dev. 2008, 22, 1427–1438. [Google Scholar] [CrossRef] [Green Version]
- Lindquist, S. The Heat hock Response. Annu. Rev. Biochem. 1986, 55, 1151–1191. [Google Scholar] [CrossRef] [PubMed]
- Vabulas, R.M.; Raychaudhuri, S.; Hayer-Hartl, M.; Hartl, F.U. Protein Folding in the Cytoplasm and the Heat Shock Response. Cold Spring Harb. Perspect. Biol. 2010, 2, a004390. [Google Scholar] [CrossRef] [PubMed]
- Labbadia, J.; Morimoto, R.I. The Biology of Proteostasis in Aging and Disease. Annu. Rev. Biochem. 2015, 84, 435–464. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Morimoto, R.I. Regulation of the heat shock transcriptional response: Cross talk between a family of heat shock factors, molecular chaperones, and negative regulators. Genes Dev. 1998, 12, 3788–3796. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Calderwood, S.K.; Gong, J. Heat Shock Proteins Promote Cancer: It’s a Protection Racket. Trends Biochem. Sci. 2016, 41, 311–323. [Google Scholar] [CrossRef] [Green Version]
- Chen, O.; Bobak, Y.P.; Stasyk, O.V.; Kunz-Schughart, L. A Complex Scenario and Underestimated Challenge: The Tumor Microenvironment, ER Stress, and Cancer Treatment. Curr. Med. Chem. 2018, 25, 2465–2502. [Google Scholar] [CrossRef]
- Overgaard, J.; Nielsen, O. The importance of thermotolerance for the clinical treatment with hyperthermia. Radiother. Oncol. 1983, 1, 167–178. [Google Scholar] [CrossRef]
- Dai, C.; Dai, S.; Cao, J. Proteotoxic stress of cancer: Implication of the heat-shock response in oncogenesis. J. Cell. Physiol. 2012, 227, 2982–2987. [Google Scholar] [CrossRef] [Green Version]
- Guang, M.H.Z.; Kavanagh, E.; Dunne, L.; Dowling, P.; Zhang, L.; Lindsay, S.; Bazou, D.; Goh, C.; Hanley, C.; Bianchi, G.; et al. Targeting Proteotoxic Stress in Cancer: A Review of the Role that Protein Quality Control Pathways Play in Oncogenesis. Cancers 2019, 11, 66. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Morrison, D.K. MAP Kinase Pathways. Cold Spring Harb. Perspect. Biol. 2012, 4, a011254. [Google Scholar] [CrossRef]
- Kariya, A.; Tabuchi, Y.; Yunoki, T.; Kondo, T. Identification of common gene networks responsive to mild hyperthermia in human cancer cells. Int. J. Mol. Med. 2013, 32, 195–202. [Google Scholar] [CrossRef] [Green Version]
- Tabuchi, Y.; Takasaki, I.; Wada, S.; Zhao, Q.-L.; Hori, T.; Nomura, T.; Ohtsuka, K.; Kondo, T. Genes and genetic networks responsive to mild hyperthermia in human lymphoma U937 cells. Int. J. Hyperth. 2008, 24, 613–622. [Google Scholar] [CrossRef] [PubMed]
- Pluquet, O.; Galmiche, A. Impact and Relevance of the Unfolded Protein Response in HNSCC. Int. J. Mol. Sci. 2019, 20, 2654. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Uppala, J.K.; Gani, A.R.; Ramaiah, K.V.A. Chemical chaperone, TUDCA unlike PBA, mitigates protein aggregation efficiently and resists ER and non-ER stress induced HepG2 cell death. Sci. Rep. 2017, 7, 3831. [Google Scholar] [CrossRef]
- Kusaczuk, M.; Krętowski, R.; Bartoszewicz, M.; Cechowska-Pasko, M. Phenylbutyrate—a pan-HDAC inhibitor—suppresses proliferation of glioblastoma LN-229 cell line. Tumor Biol. 2016, 37, 931–942. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chatterjee, S.; Burns, T.F. Targeting Heat Shock Proteins in Cancer: A Promising Therapeutic Approach. Int. J. Mol. Sci. 2017, 18, 1978. [Google Scholar] [CrossRef] [Green Version]
- Clarke, H.J.; Chambers, J.; Liniker, E.; Marciniak, S.J. Endoplasmic Reticulum Stress in Malignancy. Cancer Cell 2014, 25, 563–573. [Google Scholar] [CrossRef] [Green Version]
- Yuno, A.; Lee, M.-J.; Lee, S.; Tomita, Y.; Rekhtman, D.; Moore, B.; Trepel, J.B. Clinical Evaluation and Biomarker Profiling of Hsp90 Inhibitors. Methods Mol. Biol. 2017, 1709, 423–441. [Google Scholar] [CrossRef]
- Heinrich, J.C.; Donakonda, S.; Haupt, V.J.; Lennig, P.; Zhang, Y.; Schroeder, M. New HSP27 inhibitors efficiently suppress drug resistance development in cancer cells. Oncotarget 2016, 7, 68156–68169. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Choi, S.-K.; Kam, H.; Kim, K.-Y.; Park, S.I.; Lee, Y.-S. Targeting Heat Shock Protein 27 in Cancer: A Druggable Target for Cancer Treatment? Cancers 2019, 11, 1195. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chi, K.N.; Yu, E.Y.; Jacobs, C.; Bazov, J.; Kollmannsberger, C.; Higano, C.S.; Mukherjee, S.D.; Gleave, M.; Stewart, P.S.; Hotte, S.J. A phase I dose-escalation study of apatorsen (OGX-427), an antisense inhibitor targeting heat shock protein 27 (Hsp27), in patients with castration-resistant prostate cancer and other advanced cancers. Ann. Oncol. 2016, 27, 1116–1122. [Google Scholar] [CrossRef]
- Yu, E.Y.; Ellard, S.L.; Hotte, S.J.; Gingerich, J.R.; Joshua, A.; Gleave, M.E.; Chi, K.N. A randomized phase 2 study of a HSP27 targeting antisense, apatorsen with prednisone versus prednisone alone, in patients with metastatic castration resistant prostate cancer. Investig. New Drugs 2017, 36, 278–287. [Google Scholar] [CrossRef]
- Nappi, L.; Aguda, A.H.; Al Nakouzi, N.; Lelj-Garolla, B.; Beraldi, E.; Lallous, N.; Thi, M.; Moore, S.; Fazli, L.; Battsogt, D.; et al. Ivermectin inhibits HSP27 and potentiates efficacy of oncogene targeting in tumor models. J. Clin. Investig. 2019, 130, 699–714. [Google Scholar] [CrossRef]
- Lee, S.; Son, B.; Park, G.; Kim, H.; Kang, H.; Jeon, J.; Youn, H.; Youn, B. Immunogenic Effect of Hyperthermia on Enhancing Radiotherapeutic Efficacy. Int. J. Mol. Sci. 2018, 19, 2795. [Google Scholar] [CrossRef] [Green Version]
- Toraya-Brown, S.; Fiering, S. Local tumour hyperthermia as immunotherapy for metastatic cancer. Int. J. Hyperth. 2014, 30, 531–539. [Google Scholar] [CrossRef] [PubMed]
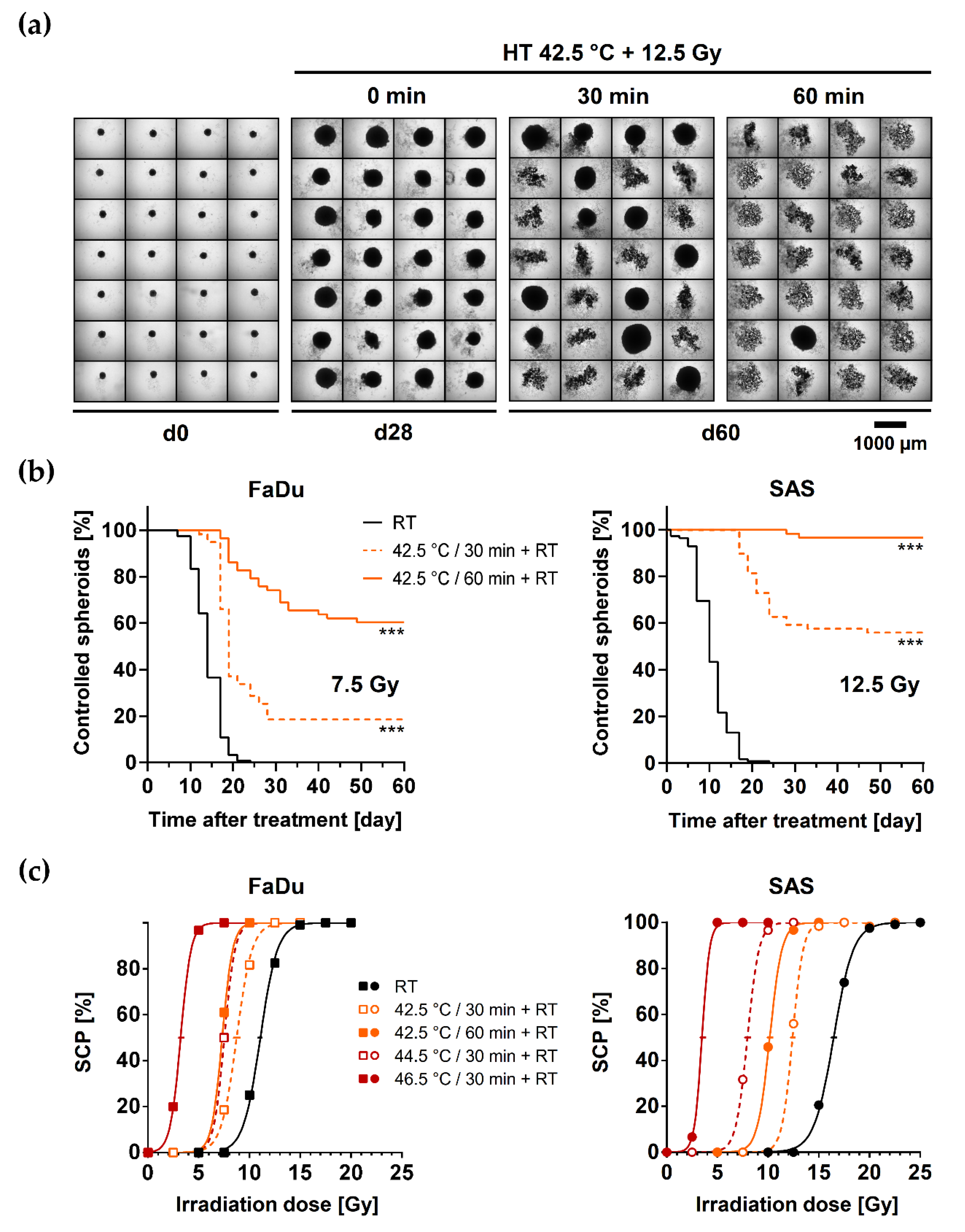

| Spheroid Type | Readout | RT Only | HT + RT | |||
|---|---|---|---|---|---|---|
| 42.5 °C 30 min | 42.5 °C 60 min | 44.5 °C 30 min | 46.5 °C 30 min | |||
| FaDu | SCD50 (Gy) | 11.1 | 8.7 | 7.3 | 7.5 | 3.2 |
| 95% CI (Gy) | 10.8–11.3 | 8.4–9.1 | 7.1–7.6 | 7.3–7.8 | 3.0–3.6 | |
| TER | 1.3 | 1.5 | 1.5 | 3.4 | ||
| 95% CI | 1.2–1.3 | 1.4–1.6 | 1.4–1.5 | 3.1–3.8 | ||
| SAS | SCD50 (Gy) | 16.5 | 12.4 | 10.2 | 8.0 | 3.5 |
| 95% CI (Gy) | 16.2–16.8 | 12.2–12.7 | 9.9–10.5 | 7.7–8.3 | 3.3–3.9 | |
| TER | 1.3 | 1.6 | 2.1 | 4.6 | ||
| 95% CI | 1.3–1.4 | 1.6–1.7 | 2.0–2.1 | 4.3–5.0 | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, O.; Michlíková, S.; Eckhardt, L.; Wondrak, M.; De Mendoza, A.M.; Krause, M.; McLeod, D.D.; Kunz-Schughart, L.A. Efficient Heat Shock Response Affects Hyperthermia-Induced Radiosensitization in a Tumor Spheroid Control Probability Assay. Cancers 2021, 13, 3168. https://doi.org/10.3390/cancers13133168
Chen O, Michlíková S, Eckhardt L, Wondrak M, De Mendoza AM, Krause M, McLeod DD, Kunz-Schughart LA. Efficient Heat Shock Response Affects Hyperthermia-Induced Radiosensitization in a Tumor Spheroid Control Probability Assay. Cancers. 2021; 13(13):3168. https://doi.org/10.3390/cancers13133168
Chicago/Turabian StyleChen, Oleg, Soňa Michlíková, Lisa Eckhardt, Marit Wondrak, Adriana M. De Mendoza, Mechthild Krause, Damian D. McLeod, and Leoni A. Kunz-Schughart. 2021. "Efficient Heat Shock Response Affects Hyperthermia-Induced Radiosensitization in a Tumor Spheroid Control Probability Assay" Cancers 13, no. 13: 3168. https://doi.org/10.3390/cancers13133168
APA StyleChen, O., Michlíková, S., Eckhardt, L., Wondrak, M., De Mendoza, A. M., Krause, M., McLeod, D. D., & Kunz-Schughart, L. A. (2021). Efficient Heat Shock Response Affects Hyperthermia-Induced Radiosensitization in a Tumor Spheroid Control Probability Assay. Cancers, 13(13), 3168. https://doi.org/10.3390/cancers13133168






