Hyperthermic Intraperitoneal Chemotherapy: A Critical Review
Abstract
:Simple Summary
Abstract
1. Introduction
2. Basic Concepts
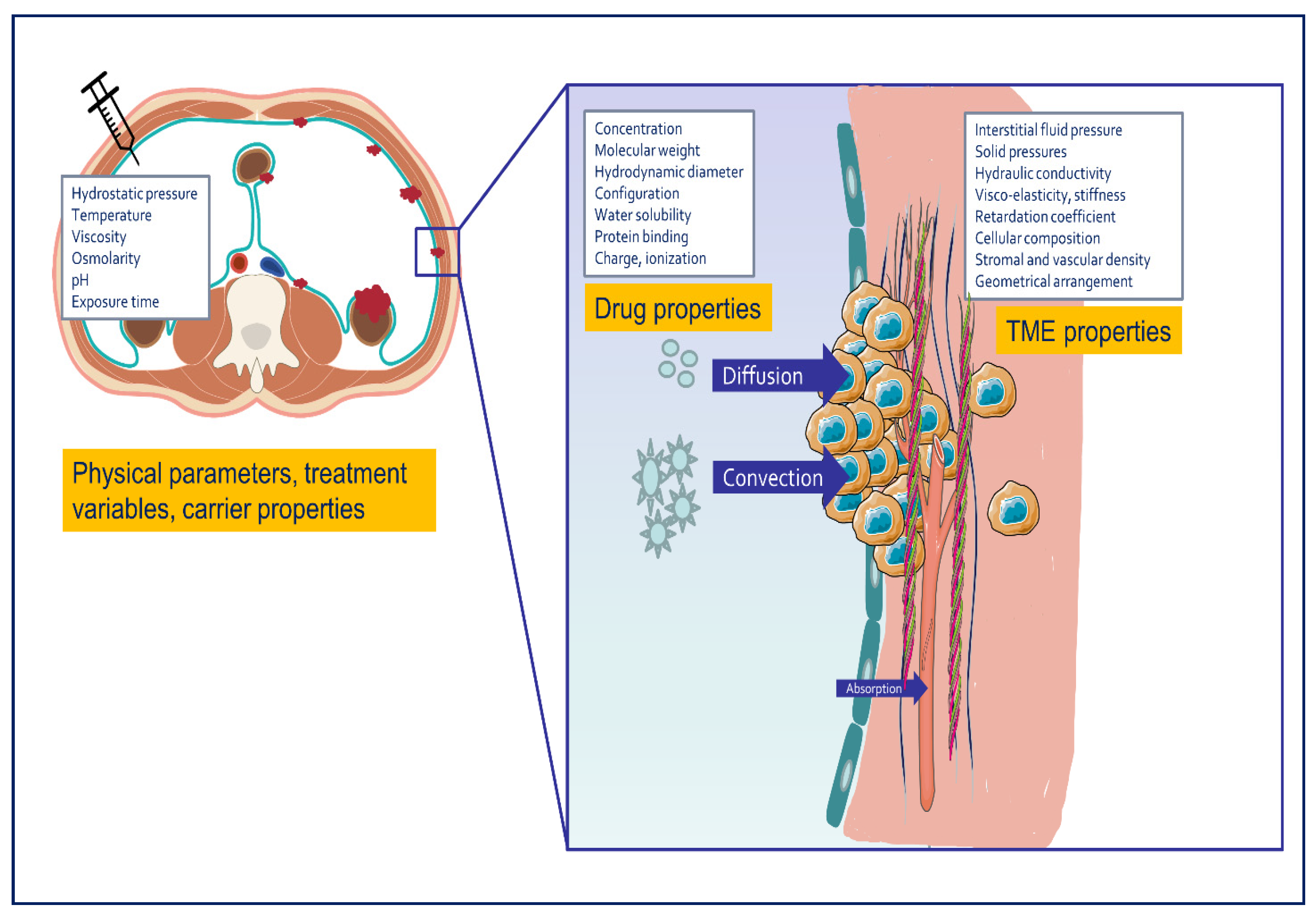
2.1. Pharmacokinetic Behavior and Drug Tissue Transport after Intraperitoneal Chemotherapy
2.1.1. Convection
2.1.2. Diffusion
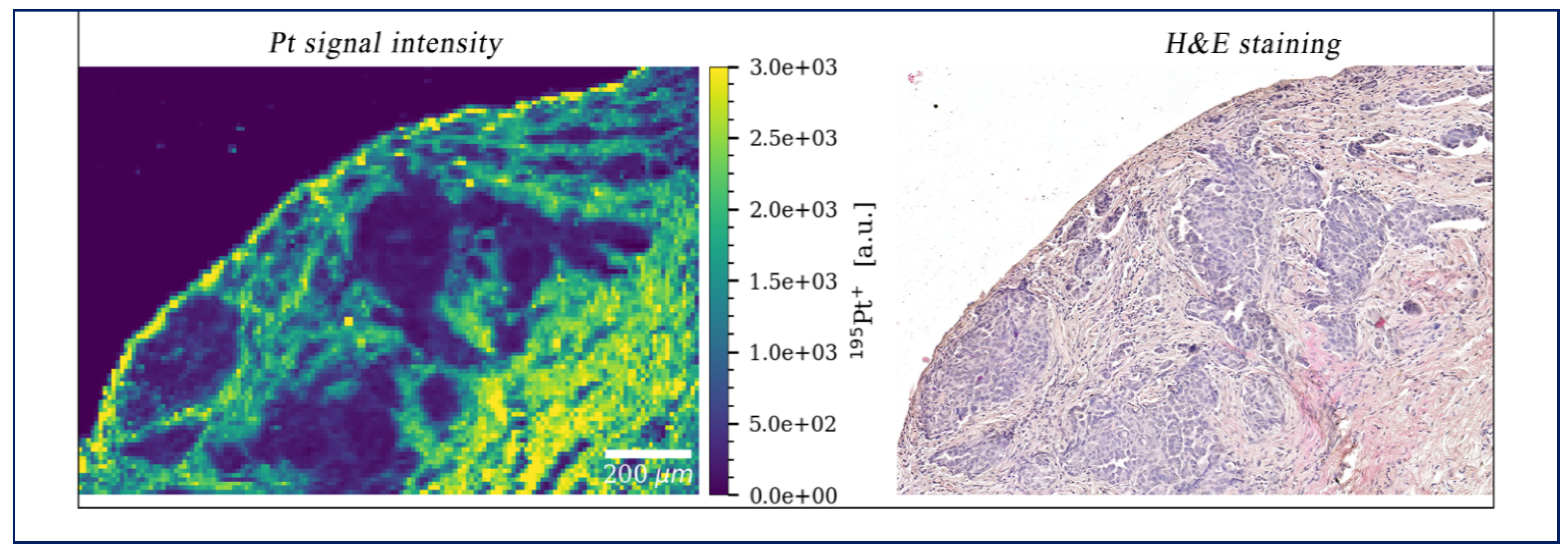
2.2. Penetration Depth after IPDD
2.3. Use of Hyperthermia
3. Clinical Implementation of Hyperthermic Intraperitoneal Drug Delivery
3.1. Choice and Combination of Chemotherapy
3.2. Open Versus Closed Abdomen Perfusion
4. Clinical Results of HIPEC
4.1. Ovarian Cancer
4.2. Colorectal Cancer
4.3. Pseudomyxoma Peritonei
4.4. Gastric Cancer
4.5. Other Intra-Abdominal Cancers
5. Addressing Current Limitations of HIPEC: The Road to Progress
5.1. Development of Novel Anticancer Compounds and Carriers
5.2. Improved Heat Delivery Methods
5.3. Clinically Relevant Preclinical Models
5.4. Elucidation of the Tumor Microenvironment and the Peritoneal Ecosystem
5.5. High Quality Clinical Trials
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Lurvink, R.J.; Bakkers, C.; Rijken, A.; van Erning, F.N.; Nienhuijs, S.W.; Burger, J.W.; Creemers, G.J.; Verhoef, C.; Lemmens, V.E.; De Hingh, I.H. Increase in the incidence of synchronous and metachronous peritoneal metastases in patients with colorectal cancer: A nationwide study. Eur. J. Surg. Oncol. 2020, 47, 1026–1033. [Google Scholar] [CrossRef]
- Koemans, W.J.; Lurvink, R.J.; Grootscholten, C.; Verhoeven, R.H.A.; de Hingh, I.H.; van Sandick, J.W. Synchronous peritoneal metastases of gastric cancer origin: Incidence, treatment and survival of a nationwide Dutch cohort. Gastric Cancer 2021. [Google Scholar] [CrossRef]
- Lengyel, E. Ovarian cancer development and metastasis. Am. J. Pathol. 2010, 177, 1053–1064. [Google Scholar] [CrossRef] [PubMed]
- Flanagan, M.; Solon, J.; Chang, K.H.; Deady, S.; Moran, B.; Cahill, R.; Shields, C.; Mulsow, J. Peritoneal metastases from extra-abdominal cancer—A population-based study. Eur. J. Surg. Oncol. 2018, 44, 1811–1817. [Google Scholar] [CrossRef]
- Lurvink, R.J.; Rijken, A.; Bakkers, C.; Aarts, M.J.; Kunst, P.W.A.; van de Borne, B.E.; van Erning, F.N.; de Hingh, I. Synchronous peritoneal metastases from lung cancer: Incidence, associated factors, treatment and survival: A Dutch population-based study. Clin. Exp. Metastasis 2021, 38, 295–303. [Google Scholar] [CrossRef]
- Franko, J.; Shi, Q.; Meyers, J.P.; Maughan, T.S.; Adams, R.A.; Seymour, M.T.; Saltz, L.; Punt, C.J.A.; Koopman, M.; Tournigand, C.; et al. Prognosis of patients with peritoneal metastatic colorectal cancer given systemic therapy: An analysis of individual patient data from prospective randomised trials from the Analysis and Research in Cancers of the Digestive System (ARCAD) database. Lancet Oncol. 2016, 17, 1709–1719. [Google Scholar] [CrossRef]
- Warrick, C. An Improvement on the practice of tapping; whereby that operation, instead of a relief for symptoms, becomes an absolute cure for an ascites, exemplified in the case of Jane Roman; And Recommended to the Consideration of the Royal Society, by Christopher Warrick, of Truro, Surgeon. Philos. Trans. 1744, 43, 12–19. [Google Scholar]
- Saxena, A.; Yan, T.D.; Morris, D.L. A critical evaluation of risk factors for complications after cytoreductive surgery and perioperative intraperitoneal chemotherapy for colorectal peritoneal carcinomatosis. World J. Surg. 2010, 34, 70–78. [Google Scholar] [CrossRef] [PubMed]
- Chua, T.C.; Yan, T.D.; Zhao, J.; Morris, D.L. Peritoneal carcinomatosis and liver metastases from colorectal cancer treated with cytoreductive surgery perioperative intraperitoneal chemotherapy and liver resection. Ejso 2009, 35, 1299–1305. [Google Scholar] [CrossRef] [PubMed]
- Dedrick, R.L.; Myers, C.E.; Bungay, P.M.; DeVita, V.T., Jr. Pharmacokinetic rationale for peritoneal drug administration in the treatment of ovarian cancer. Cancer Treat. Rep. 1978, 62, 1–11. [Google Scholar] [PubMed]
- Dedrick, R.L. Theoretical and experimental bases of intraperitoneal chemotherapy. Semin. Oncol. 1985, 12, 1–6. [Google Scholar]
- Euler, J.; Prieschi, A.; Wenzl, J.; Sauerman, G.; Klockler, K.; Kretschm, G. Hyperthermic peritoneal perfusion in ascites tumors in rats. Wien. Klin. Wochenschr. 1974, 86, 220–225. [Google Scholar] [PubMed]
- Spratt, J.S.; Adcock, R.A.; Muskovin, M.; Sherrill, W.; Mckeown, J. Clinical delivery system for intra-peritoneal hyperthermic chemotherapy. Cancer Res. 1980, 40, 256–260. [Google Scholar]
- Bushati, M.; Rovers, K.P.; Sommariva, A.; Sugarbaker, P.H.; Morris, D.L.; Yonemura, Y.; Quadros, C.A.; Somashekhar, S.P.; Ceelen, W.; Dubé, P.; et al. The current practice of cytoreductive surgery and HIPEC for colorectal peritoneal metastases: Results of a worldwide web-based survey of the Peritoneal Surface Oncology Group International (PSOGI). Eur. J. Surg. Oncol. 2018, 44, 1942–1948. [Google Scholar] [CrossRef]
- Quénet, F.; Elias, D.; Roca, L.; Goéré, D.; Ghouti, L.; Pocard, M.; Facy, O.; Arvieux, C.; Lorimier, G.; Pezet, D.; et al. Cytoreductive surgery plus hyperthermic intraperitoneal chemotherapy versus cytoreductive surgery alone for colorectal peritoneal metastases (PRODIGE 7): A multicentre, randomised, open-label, phase 3 trial. Lancet Oncol. 2021, 22, 256–266. [Google Scholar] [CrossRef]
- Hasovits, C.; Clarke, S. Pharmacokinetics and Pharmacodynamics of Intraperitoneal Cancer Chemotherapeutics. Clin. Pharmacokinet. 2012, 51, 203–224. [Google Scholar] [CrossRef]
- Leypoldt, J.K. Solute transport across the peritoneal membrane. J. Am. Soc. Nephrol. 2002, 13 (Suppl. 1), S84–S91. [Google Scholar] [CrossRef]
- Butler, T.P.; Grantham, F.H.; Gullino, P.M. Bulk transfer of fluid in the interstitial compartment of mammary tumors. Cancer Res. 1975, 35, 3084–3088. [Google Scholar] [PubMed]
- Stylianopoulos, T. The solid mechanics of cancer and strategies for improved therapy. J. Biomech. Eng. 2017, 139. [Google Scholar] [CrossRef] [PubMed]
- McGrail, D.J.; McAndrews, K.M.; Brandenburg, C.P.; Ravikumar, N.; Kieu, Q.M.; Dawson, M.R. Osmotic regulation is required for cancer cell survival under solid stress. Biophys. J. 2015, 109, 1334–1337. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Netti, P.A.; Berk, D.A.; Swartz, M.A.; Grodzinsky, A.J.; Jain, R.K. Role of extracellular matrix assembly in interstitial transport in solid tumors. Cancer Res. 2000, 60, 2497–2503. [Google Scholar] [PubMed]
- Jain, R.K. Transport of molecules in the tumor interstitium: A review. Cancer Res. 1987, 47, 3039–3051. [Google Scholar]
- Young, E.W. Cells, tissues, and organs on chips: Challenges and opportunities for the cancer tumor microenvironment. Integr. Biol. 2013, 5, 1096–1109. [Google Scholar] [CrossRef]
- Baker, A.M.; Bird, D.; Lang, G.; Cox, T.R.; Erler, J.T. Lysyl oxidase enzymatic function increases stiffness to drive colorectal cancer progression through FAK. Oncogene 2013, 32, 1863–1868. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Butcher, D.T.; Alliston, T.; Weaver, V.M. A tense situation: Forcing tumour progression. Nat. Rev. Cancer 2009, 9, 108–122. [Google Scholar] [CrossRef]
- Steuperaert, M.; Debbaut, C.; Carlier, C.; De Wever, O.; Descamps, B.; Vanhove, C.; Ceelen, W.; Segers, P. A 3D CFD model of the interstitial fluid pressure and drug distribution in heterogeneous tumor nodules during intraperitoneal chemotherapy. Drug Deliv. 2019, 26, 404–415. [Google Scholar] [CrossRef] [Green Version]
- Carlier, C.; Mathys, A.; De Jaeghere, E.; Steuperaert, M.; De Wever, O.; Ceelen, W. Tumour tissue transport after intraperitoneal anticancer drug delivery. Int. J. Hyperth. 2017, 33, 534–542. [Google Scholar] [CrossRef] [Green Version]
- Cox, T.R. The matrix in cancer. Nat. Rev. Cancer 2021, 21, 217–238. [Google Scholar] [CrossRef] [PubMed]
- Hildebrandt, B.; Wust, P.; Ahlers, O.; Dieing, A.; Sreenivasa, G.; Kerner, T.; Felix, R.; Riess, H. The cellular and molecular basis of hyperthermia. Crit. Rev. Oncol. Hematol. 2002, 43, 33–56. [Google Scholar] [CrossRef]
- Datta, N.R.; Ordóñez, S.G.; Gaipl, U.S.; Paulides, M.M.; Crezee, H.; Gellermann, J.; Marder, D.; Puric, E.; Bodis, S. Local hyperthermia combined with radiotherapy and-/or chemotherapy: Recent advances and promises for the future. Cancer Treat. Rev. 2015, 41, 742–753. [Google Scholar] [CrossRef]
- Los, G.; Sminia, P.; Wondergem, J.; Mutsaers, P.H.A.; Havemen, J.; Huinink, D.T.; Smals, O.; Gonzalezgonzalez, D.; Mcvie, J.G. Optimization of intraperitoneal cisplatin therapy with regional hyperthermia in rats. Eur. J. Cancer 1991, 27, 472–477. [Google Scholar] [CrossRef]
- Pestieau, S.R.; Belliveau, J.F.; Griffin, H.; Stuart, O.A.; Sugarbaker, P.H. Pharmacokinetics of intraperitoneal oxaliplatin: Experimental studies. J. Surg. Oncol. 2001, 76, 106–114. [Google Scholar] [CrossRef]
- Jacquet, P.; Averbach, A.; Stuart, O.A.; Chang, D.; Sugarbaker, P.H. Hyperthermic intraperitoneal doxorubicin: Pharmacokinetics, metabolism, and tissue distribution in a rat model. Cancer Chemother. Pharmacol. 1998, 41, 147–154. [Google Scholar] [CrossRef]
- Helderman, R.; Löke, D.R.; Verhoeff, J.; Rodermond, H.M.; van Bochove, G.G.W.; Boon, M.; van Kesteren, S.; Garcia Vallejo, J.J.; Kok, H.P.; Tanis, P.J.; et al. The temperature-dependent effectiveness of platinum-based drugs mitomycin-C and 5-FU during hyperthermic intraperitoneal chemotherapy (HIPEC) in colorectal cancer cell lines. Cells 2020, 9, 1775. [Google Scholar] [CrossRef]
- Boussios, S.; Moschetta, M.; Karathanasi, A.; Tsiouris, A.K.; Kanellos, F.S.; Tatsi, K.; Katsanos, K.H.; Christodoulou, D.K. Malignant peritoneal mesothelioma: Clinical aspects, and therapeutic perspectives. Ann. Gastroenterol. 2018, 31, 659–669. [Google Scholar] [CrossRef]
- Forsythe, S.D.; Sasikumar, S.; Moaven, O.; Sivakumar, H.; Shen, P.; Levine, E.A.; Soker, S.; Skardal, A.; Votanopoulos, K.I. Personalized identification of optimal HIPEC perfusion protocol in patient-derived tumor organoid platform. Ann. Surg. Oncol. 2020, 27, 4950–4960. [Google Scholar] [CrossRef]
- Kepenekian, V.; Aloy, M.T.; Magne, N.; Passot, G.; Armandy, E.; Decullier, E.; Sayag-Beaujard, A.; Gilly, F.N.; Glehen, O.; Rodriguez-Lafrasse, C. Impact of hyperthermic intraperitoneal chemotherapy on Hsp27 protein expression in serum of patients with peritoneal carcinomatosis. Cell Stress Chaperones 2013, 18, 623–630. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Grimmig, T.; Moll, E.M.; Kloos, K.; Thumm, R.; Moench, R.; Callies, S.; Kreckel, J.; Vetterlein, M.; Pelz, J.; Polat, B.; et al. Upregulated heat shock proteins after hyperthermic chemotherapy point to induced cell survival mechanisms in affected tumor cells from peritoneal carcinomatosis. Cancer Growth Metastasis 2017, 10, 1179064417730559. [Google Scholar] [CrossRef] [PubMed]
- Fujimoto, S.; Kokubun, M.; Shrestha, R.D.; Kobayashi, K.; Kiuchi, S.; Konno, C.; Takahashi, M.; Okui, K. Prevention of scald injury on the peritoneo-serosal surface in advanced gastric cancer patients treated with intraperitoneal hyperthermic perfusion. Int. J. Hyperth. 1991, 7, 543–550. [Google Scholar] [CrossRef] [PubMed]
- Yonemura, Y.; de Aretxabala, X.; Fujimura, T.; Fushida, S.; Katayama, K.; Bandou, E.; Sugiyama, K.; Kawamura, T.; Kinoshita, K.; Endou, Y.; et al. Intraoperative chemohyperthermic peritoneal perfusion as an adjuvant to gastric cancer: Final results of a randomized controlled study. Hepatogastroenterology 2001, 48, 1776–1782. [Google Scholar]
- Chang, M.; Hou, Z.; Wang, M.; Li, C.; Lin, J. Recent advances in hyperthermia therapy-based synergistic immunotherapy. Adv. Mater. 2021, 33, e2004788. [Google Scholar] [CrossRef]
- Minnaar, C.A.; Kotzen, J.A.; Ayeni, O.A.; Vangu, M.D.; Baeyens, A. Potentiation of the abscopal effect by modulated electro-hyperthermia in locally advanced cervical cancer patients. Front. Oncol. 2020, 10, 376. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bear, A.S.; Kennedy, L.C.; Young, J.K.; Perna, S.K.; Mattos Almeida, J.P.; Lin, A.Y.; Eckels, P.C.; Drezek, R.A.; Foster, A.E. Elimination of metastatic melanoma using gold nanoshell-enabled photothermal therapy and adoptive T cell transfer. PLoS ONE 2013, 8, e69073. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Seclì, L.; Fusella, F.; Avalle, L.; Brancaccio, M. The dark-side of the outside: How extracellular heat shock proteins promote cancer. Cell. Mol. Life Sci. 2021, 78, 4069–4083. [Google Scholar] [CrossRef] [PubMed]
- Ramanathan, R.; Choudry, H.; Jones, H.; Girgis, M.; Gooding, W.; Kalinski, P.; Bartlett, D.L. Phase II trial of adjuvant dendritic cell vaccine in combination with celecoxib, interferon-α, and rintatolimod in patients undergoing cytoreductive surgery and hyperthermic intraperitoneal chemotherapy for peritoneal metastases. Ann. Surg. Oncol. 2021, 1–10. [Google Scholar] [CrossRef]
- Franko, J.; Brahmbhatt, R.; Tee, M.; Raman, S.; Ferrel, B.; Gorvet, M.; Andres, M. Cellular immunoprofile of peritoneal environment during a HIPEC procedure. Ann. Surg. Oncol. 2020, 27, 5005–5013. [Google Scholar] [CrossRef]
- Van der Speeten, K.; Lemoine, L.; Sugarbaker, P. Overview of the optimal perioperative intraperitoneal chemotherapy regimens used in current clinical practice. Pleura Peritoneum 2017, 2, 63–72. [Google Scholar] [CrossRef]
- Helderman, R.; Löke, D.R.; Kok, H.P.; Oei, A.L.; Tanis, P.J.; Franken, N.; Crezee, J. Variation in clinical application of hyperthermic intraperitoneal chemotherapy: A Review. Cancers 2019, 11, 78. [Google Scholar] [CrossRef] [Green Version]
- Wisselink, D.D.; Braakhuis, L.L.F.; Gallo, G.; van Grevenstein, W.M.U.; van Dieren, S.; Kok, N.F.M.; de Reuver, P.R.; Tanis, P.J.; de Hingh, I. Systematic review of published literature on oxaliplatin and mitomycin C as chemotherapeutic agents for hyperthermic intraperitoneal chemotherapy in patients with peritoneal metastases from colorectal cancer. Crit. Rev. Oncol. Hematol. 2019, 142, 119–129. [Google Scholar] [CrossRef]
- Levine, E.A.; Votanopoulos, K.I.; Shen, P.; Russell, G.; Fenstermaker, J.; Mansfield, P.; Bartlett, D.; Stewart, J.H. A multicenter randomized trial to evaluate hematologic toxicities after hyperthermic intraperitoneal chemotherapy with oxaliplatin or mitomycin in patients with appendiceal tumors. J. Am. Coll. Surg. 2018, 226, 434–443. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Moaven, O.; Votanopoulos, K.I.; Shen, P.; Mansfield, P.; Bartlett, D.L.; Russell, G.; McQuellon, R.; Stewart, J.H.; Levine, E.A. Health-related quality of life after cytoreductive surgery/HIPEC for mucinous appendiceal cancer: Results of a multicenter randomized trial comparing oxaliplatin and mitomycin. Ann. Surg. Oncol. 2020, 27, 772–780. [Google Scholar] [CrossRef]
- Ceelen, W. HIPEC with oxaliplatin for colorectal peritoneal metastasis: The end of the road? Eur. J. Surg. Oncol. 2019, 45, 400–402. [Google Scholar] [CrossRef] [PubMed]
- Quenet, F.; Goéré, D.; Mehta, S.S.; Roca, L.; Dumont, F.; Hessissen, M.; Saint-Aubert, B.; Elias, D. Results of two bi-institutional prospective studies using intraperitoneal oxaliplatin with or without irinotecan during HIPEC after cytoreductive surgery for colorectal carcinomatosis. Ann. Surg. 2011, 254, 294–301. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez Silva, C.; Moreno Ruiz, F.J.; Bellido Estévez, I.; Carrasco Campos, J.; Titos García, A.; Ruiz López, M.; González Poveda, I.; Toval Mata, J.A.; Mera Velasco, S.; Santoyo Santoyo, J. Are there intra-operative hemodynamic differences between the Coliseum and closed HIPEC techniques in the treatment of peritoneal metastasis? A retrospective cohort study. World J. Surg. Oncol. 2017, 15, 51. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Leiting, J.L.; Cloyd, J.M.; Ahmed, A.; Fournier, K.; Lee, A.J.; Dessureault, S.; Felder, S.; Veerapong, J.; Baumgartner, J.M.; Clarke, C.; et al. Comparison of open and closed hyperthermic intraperitoneal chemotherapy: Results from the United States hyperthermic intraperitoneal chemotherapy collaborative. World J. Gastrointest. Oncol. 2020, 12, 756–767. [Google Scholar] [CrossRef]
- Cianci, S.; Abatini, C.; Fagotti, A.; Chiofalo, B.; Tropea, A.; Biondi, A.; Scambia, G.; Pacelli, F. Hyperthermic intraperitoneal chemotherapy (HIPEC) for peritoneal malignancies using new hybrid CO (2) system: Preliminary experience in referral center. Updates Surg. 2019, 71, 555–560. [Google Scholar] [CrossRef]
- Arjona-Sanchez, A.; Aziz, O.; Passot, G.; Salti, G.; Esquivel, J.; Van der Speeten, K.; Piso, P.; Nedelcut, D.S.; Sommariva, A.; Yonemura, Y.; et al. Laparoscopic cytoreductive surgery and hyperthermic intraperitoneal chemotherapy for limited peritoneal metastasis. The PSOGI international collaborative registry. Eur. J. Surg. Oncol. 2020. [Google Scholar] [CrossRef]
- Verwaal, V.J.; Bruin, S.; Boot, H.; van Slooten, G.; van Tinteren, H. 8-year follow-up of randomized trial: Cytoreduction and hyperthermic intraperitoneal chemotherapy versus systemic chemotherapy in patients with peritoneal carcinomatosis of colorectal cancer. Ann. Surg. Oncol. 2008, 15, 2426–2432. [Google Scholar] [CrossRef]
- Klaver, C.E.L.; Wisselink, D.D.; Punt, C.J.A.; Snaebjornsson, P.; Crezee, J.; Aalbers, A.G.J.; Brandt, A.; Bremers, A.J.A.; Burger, J.W.A.; Fabry, H.F.J.; et al. Adjuvant hyperthermic intraperitoneal chemotherapy in patients with locally advanced colon cancer (COLOPEC): A multicentre, open-label, randomised trial. Lancet Gastroenterol. Hepatol. 2019, 4, 761–770. [Google Scholar] [CrossRef]
- Goéré, D.; Glehen, O.; Quenet, F.; Guilloit, J.M.; Bereder, J.M.; Lorimier, G.; Thibaudeau, E.; Ghouti, L.; Pinto, A.; Tuech, J.J.; et al. Second-look surgery plus hyperthermic intraperitoneal chemotherapy versus surveillance in patients at high risk of developing colorectal peritoneal metastases (PROPHYLOCHIP-PRODIGE 15): A randomised, phase 3 study. Lancet Oncol. 2020, 21, 1147–1154. [Google Scholar] [CrossRef]
- Rovers, K.P.; Bakkers, C.; Nienhuijs, S.W.; Burger, J.W.A.; Creemers, G.M.; Thijs, A.M.J.; Brandt-Kerkhof, A.R.M.; Madsen, E.V.E.; van Meerten, E.; Tuynman, J.B.; et al. Perioperative systemic therapy vs cytoreductive surgery and hyperthermic intraperitoneal chemotherapy alone for resectable colorectal peritoneal metastases: A phase 2 randomized clinical trial. JAMA Surg. 2021. [Google Scholar] [CrossRef] [PubMed]
- Spiliotis, J.; Halkia, E.; Lianos, E.; Kalantzi, N.; Grivas, A.; Efstathiou, E.; Giassas, S. Cytoreductive surgery and HIPEC in recurrent epithelial ovarian cancer: A prospective randomized phase III study. Ann. Surg. Oncol. 2015, 22, 1570–1575. [Google Scholar] [CrossRef] [PubMed]
- van Driel, W.J.; Koole, S.N.; Sikorska, K.; Schagen van Leeuwen, J.H.; Schreuder, H.W.R.; Hermans, R.H.M.; de Hingh, I.; van der Velden, J.; Arts, H.J.; Massuger, L.; et al. Hyperthermic intraperitoneal chemotherapy in ovarian cancer. N. Engl. J. Med. 2018, 378, 230–240. [Google Scholar] [CrossRef] [PubMed]
- Zivanovic, O.; Chi, D.S.; Zhou, Q.; Iasonos, A.; Konner, J.A.; Makker, V.; Grisham, R.N.; Brown, A.K.; Nerenstone, S.; Diaz, J.P.; et al. Secondary Cytoreduction and Carboplatin Hyperthermic Intraperitoneal Chemotherapy for Platinum-Sensitive Recurrent Ovarian Cancer: An MSK Team Ovary Phase II Study. J. Clin. Oncol. 2021, Jco2100605. [Google Scholar] [CrossRef]
- Rassy, E.; Assi, T.; Boussios, S.; Kattan, J.; Smith-Gagen, J.; Pavlidis, N. Narrative review on serous primary peritoneal carcinoma of unknown primary site: Four questions to be answered. Ann. Transl. Med. 2020, 8, 1709. [Google Scholar] [CrossRef] [PubMed]
- Koole, S.N.; Kieffer, J.M.; Sikorska, K.; Schagen van Leeuwen, J.H.; Schreuder, H.W.R.; Hermans, R.H.; de Hingh, I.H.; van der Velden, J.; Arts, H.J.; van Ham, M.; et al. Health-related quality of life after interval cytoreductive surgery with or without hyperthermic intraperitoneal chemotherapy (HIPEC) in patients with stage III ovarian cancer. Eur. J. Surg. Oncol. 2021, 47, 101–107. [Google Scholar] [CrossRef] [PubMed]
- Koole, S.N.; van Lieshout, C.; van Driel, W.J.; van Schagen, E.; Sikorska, K.; Kieffer, J.M.; Schagen van Leeuwen, J.H.; Schreuder, H.W.R.; Hermans, R.H.; de Hingh, I.H.; et al. Effectiveness of Interval Cytoreductive Surgery with Hyperthermic Intraperitoneal Chemotherapy in Stage III Ovarian Cancer on the Basis of a Randomized Phase III Trial. J. Clin. Oncol. 2019, 37, 2041–2050. [Google Scholar] [CrossRef]
- Armstrong, D.K.; Alvarez, R.D.; Bakkum-Gamez, J.N.; Barroilhet, L.; Behbakht, K.; Berchuck, A.; Berek, J.S.; Chen, L.M.; Cristea, M.; DeRosa, M.; et al. NCCN Guidelines Insights: Ovarian Cancer, Version 1.2019. J. Natl. Compr. Cancer Netw. JNCCN 2019, 17, 896–909. [Google Scholar] [CrossRef] [Green Version]
- Boussios, S.; Pavlidis, N. Ovarian cancer: State of the art and perspectives of clinical research. Ann. Transl. Med. 2020, 8, 1702. [Google Scholar] [CrossRef]
- Lim, M.C.; Chang, S.-J.; Yoo, H.J.; Nam, B.-H.; Bristow, R.; Park, S.-Y. Randomized trial of hyperthermic intraperitoneal chemotherapy (HIPEC) in women with primary advanced peritoneal, ovarian, and tubal cancer. J. Clin. Oncol. 2017, 35, 5520. [Google Scholar] [CrossRef]
- Koole, S.; van Stein, R.; Sikorska, K.; Barton, D.; Perrin, L.; Brennan, D.; Zivanovic, O.; Mosgaard, B.J.; Fagotti, A.; Colombo, P.E.; et al. Primary cytoreductive surgery with or without hyperthermic intraperitoneal chemotherapy (HIPEC) for FIGO stage III epithelial ovarian cancer: OVHIPEC-2, a phase III randomized clinical trial. Int. J. Gynecol. Cancer 2020, 30, 888–892. [Google Scholar] [CrossRef]
- Farrell, R.; Burling, M. HIPEC: Turning up the heat on ovarian cancer. Aust. N. Z. J. Obstet. Gynaecol. 2021, 61, 11–15. [Google Scholar] [CrossRef] [PubMed]
- Verwaal, V.J.; van Ruth, S.; de Bree, E.; van Sloothen, G.W.; van Tinteren, H.; Boot, H.; Zoetmulder, F.A. Randomized trial of cytoreduction and hyperthermic intraperitoneal chemotherapy versus systemic chemotherapy and palliative surgery in patients with peritoneal carcinomatosis of colorectal cancer. J. Clin. Oncol. 2003, 21, 3737–3743. [Google Scholar] [CrossRef] [PubMed]
- Flood, M.; Narasimhan, V.; Waters, P.; Ramsay, R.; Michael, M.; Warrier, S.; Heriot, A. Survival after cytoreductive surgery and hyperthermic intraperitoneal chemotherapy for colorectal peritoneal metastases: A systematic review and discussion of latest controversies. Surgeon 2020. [Google Scholar] [CrossRef] [PubMed]
- Chua, T.C.; Yan, T.D.; Saxena, A.; Morris, D.L. Should the treatment of peritoneal carcinomatosis by cytoreductive surgery and hyperthermic intraperitoneal chemotherapy still be regarded as a highly morbid procedure? A systematic review of morbidity and mortality. Ann. Surg. 2009, 249, 900–907. [Google Scholar] [CrossRef] [PubMed]
- Nagourney, R.A.; Evans, S.; Tran, P.H.; Nagourney, A.J.; Sugarbaker, P.H. Colorectal cancer cells from patients treated with FOLFOX or CAPOX are resistant to oxaliplatin. Eur. J. Surg. Oncol. 2021, 47, 738–742. [Google Scholar] [CrossRef]
- Ubink, I.; Bolhaqueiro, A.C.F.; Elias, S.G.; Raats, D.A.E.; Constantinides, A.; Peters, N.A.; Wassenaar, E.C.E.; de Hingh, I.; Rovers, K.P.; van Grevenstein, W.M.U.; et al. Organoids from colorectal peritoneal metastases as a platform for improving hyperthermic intraperitoneal chemotherapy. Br. J. Surg. 2019, 106, 1404–1414. [Google Scholar] [CrossRef]
- Rovers, K.P.; Simkens, G.A.; Punt, C.J.; van Dieren, S.; Tanis, P.J.; de Hingh, I.H. Perioperative systemic therapy for resectable colorectal peritoneal metastases: Sufficient evidence for its widespread use? A critical systematic review. Crit. Rev. Oncol. Hematol. 2017, 114, 53–62. [Google Scholar] [CrossRef]
- Waite, K.; Youssef, H. The Role of Neoadjuvant and Adjuvant Systemic Chemotherapy with Cytoreductive Surgery and Heated Intraperitoneal Chemotherapy for Colorectal Peritoneal Metastases: A Systematic Review. Ann. Surg. Oncol. 2017, 24, 705–720. [Google Scholar] [CrossRef]
- Repullo, D.J.; Barbois, S.; Leonard, D.; Bohlok, A.; Van den Audenaeren, E.T.; Hendlisz, A.; Van den Eynde, M.; Donckier, V.; Kartheuser, A.; Liberale, G. The absence of benefit of perioperative chemotherapy in initially resectable peritoneal metastases of colorectal cancer origin treated with complete cytoreductive surgery and hyperthermic intraperitoneal chemotherapy: A retrospective analysis. Eur. J. Surg. Oncol. 2021. [Google Scholar] [CrossRef]
- Rovers, K.P.; Bakkers, C.; Simkens, G.; Burger, J.W.A.; Nienhuijs, S.W.; Creemers, G.M.; Thijs, A.M.J.; Brandt-Kerkhof, A.R.M.; Madsen, E.V.E.; Ayez, N.; et al. Perioperative systemic therapy and cytoreductive surgery with HIPEC versus upfront cytoreductive surgery with HIPEC alone for isolated resectable colorectal peritoneal metastases: Protocol of a multicentre, open-label, parallel-group, phase II-III, randomised, superiority study (CAIRO6). BMC Cancer 2019, 19, 390. [Google Scholar]
- Goere, D.; Glehen, O.; Quenet, F.; Ducreux, M.; Guilloit, J.-M.; Texier, M.; Benhamou, E.; Elias, D. BIG-RENAPE.; PRODIGE, Results of a randomized phase 3 study evaluating the potential benefit of a second-look surgery plus HIPEC in patients at high risk of developing colorectal peritoneal metastases (PROPHYLOCHIP- NTC01226394). J. Clin. Oncol. 2018, 36, 3531. [Google Scholar] [CrossRef]
- Klaver, C.E.L.; Wisselink, D.D.; Punt, C.J.A.; Snaebjornsson, P.; Crezee, J.; Aalbers, A.; Bemelman, W.A.; Brandt, A.; Bremers, A.; Burger, J.W.A.; et al. Adjuvant HIPEC in patients with colon cancer at high risk of peritoneal metastases: Primary outcome of the COLOPEC multicenter randomized trial. J. Clin. Oncol. 2019, 37, 482. [Google Scholar] [CrossRef]
- Ripley, R.T.; Davis, J.L.; Kemp, C.D.; Steinberg, S.M.; Toomey, M.A.; Avital, I. Prospective randomized trial evaluating mandatory second look surgery with HIPEC and CRS vs. standard of care in patients at high risk of developing colorectal peritoneal metastases. Trials 2010, 11, 1–8. [Google Scholar] [CrossRef] [Green Version]
- Pai, R.K.; Longacre, T.A. Appendiceal mucinous tumors and pseudomyxoma peritonei—Histologic features, diagnostic problems, and proposed classification. Adv. Anat. Pathol. 2005, 12, 291–311. [Google Scholar] [CrossRef]
- Panarelli, N.C.; Yantiss, R.K. Mucinous neoplasms of the appendix and peritoneum. Arch. Pathol. Lab. Med. 2011, 135, 1261–1268. [Google Scholar] [CrossRef]
- Legue, L.M.; Creemers, G.J.; de Hingh, I.; Lemmens, V.; Huysentruyt, C.J. Review: Pathology and its clinical relevance of mucinous appendiceal neoplasms and pseudomyxoma peritonei. Clin Colorectal Cancer 2019, 18, 1–7. [Google Scholar] [CrossRef]
- Smeenk, R.M.; Verwaal, V.J.; Zoetmulder, F.A.N. Pseudomyxoma peritonei. Cancer Treat. Rev. 2007, 33, 138–145. [Google Scholar] [CrossRef]
- Sugarbaker, P.H.; Chang, D. Results of treatment of 385 patients with peritoneal surface spread of appendiceal malignancy. Ann. Surg. Oncol. 1999, 6, 727–731. [Google Scholar] [CrossRef]
- Elias, D.; Honore, C.; Ciuchendea, R.; Billard, V.; Raynard, B.; Lo Dico, R.; Dromain, C.; Duvillard, P.; Goere, D. Peritoneal pseudomyxoma: Results of a systematic policy of complete cytoreductive surgery and hyperthermic intraperitoneal chemotherapy. Br. J. Surg. 2008, 95, 1164–1171. [Google Scholar] [CrossRef]
- Smeenk, R.M.; Verwaal, V.J.; Antonini, N.; Zoetmulder, F.A.N. Survival analysis of pseudomyxoma peritonei patients treated by cytoreductive surgery and hyperthermic intraperitoneal chemotherapy. Ann. Surg. 2007, 245, 104–109. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.S.; Gong, C.Y.; Yang, L.; Wu, Q.J.; Shi, S.A.; Shi, H.S.; Qian, Z.Y.; Wei, Y.Q. 5-FU-hydrogel inhibits colorectal peritoneal carcinomatosis and tumor growth in mice. BMC Cancer 2010, 10, 1–8. [Google Scholar] [CrossRef] [Green Version]
- Kusamura, S.; Barretta, F.; Yonemura, Y.; Sugarbaker, P.H.; Moran, B.J.; Levine, E.A.; Goere, D.; Baratti, D.; Nizri, E.; Morris, D.L.; et al. The role of hyperthermic intraperitoneal chemotherapy in pseudomyxoma peritonei After cytoreductive surgery. JAMA Surg. 2021, 156, e206363. [Google Scholar] [CrossRef] [PubMed]
- Sugarbaker, P.H. New standard of care for appendiceal epithelial neoplasms and pseudomyxoma peritonei syndrome? Lancet Oncology 2006, 7, 69–76. [Google Scholar] [CrossRef]
- Miner, T.J.; Shia, J.R.; Jaques, D.P.; Klimstra, D.S.; Brennan, M.F.; Coit, D.G. Long-term survival following treatment of pseudomyxoma peritonei—An analysis of surgical therapy. Ann. Surg. 2005, 241, 300–308. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, D.; Kodera, Y. Intraperitoneal chemotherapy for gastric cancer with peritoneal metastasis. Gastric Cancer 2017, 20, 111–121. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Feingold, P.L.; Kwong, M.L.; Davis, J.L.; Rudloff, U. Adjuvant intraperitoneal chemotherapy for the treatment of gastric cancer at risk for peritoneal carcinomatosis: A systematic review. J. Surg. Oncol. 2017, 115, 192–201. [Google Scholar] [CrossRef]
- Yan, T.D.; Black, D.; Sugarbaker, P.H.; Zhu, J.Q.; Yonemura, Y.; Petrou, G.; Morris, D.L. A systematic review and meta-analysis of the randomized controlled trials on adjuvant intraperitoneal chemotherapy for resectable gastric cancer. Ann. Surg. Oncol. 2007, 14, 2702–2713. [Google Scholar] [CrossRef]
- Desiderio, J.; Chao, J.; Melstrom, L.; Warner, S.; Tozzi, F.; Fong, Y.; Parisi, A.; Woo, Y. The 30-year experience-A meta-analysis of randomised and high-quality non-randomised studies of hyperthermic intraperitoneal chemotherapy in the treatment of gastric cancer. Eur. J. Cancer 2017, 79, 1–14. [Google Scholar] [CrossRef]
- van der Kaaij, R.T.; Wassenaar, E.C.E.; Koemans, W.J.; Sikorska, K.; Grootscholten, C.; Los, M.; Huitema, A.; Schellens, J.H.M.; Veenhof, A.; Hartemink, K.J.; et al. Treatment of PERItoneal disease in Stomach Cancer with cytOreductive surgery and hyperthermic intraPEritoneal chemotherapy: PERISCOPE I initial results. Br. J. Surg. 2020, 107, 1520–1528. [Google Scholar] [CrossRef]
- Yan, T.D.; Welch, L.; Black, D.; Sugarbaker, P.H. A systematic review on the efficacy of cytoreductive surgery combined with perioperative intraperitoneal chemotherapy for diffuse malignancy peritoneal mesothelioma. Ann. Oncol. 2007, 18, 827–834. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Yonemura, Y.; Levine, E.A.; Glehen, O.; Goere, D.; Elias, D.; Morris, D.L.; Sugarbaker, P.H.; Tuech, J.J.; Cashin, P.; et al. Cytoreductive surgery plus hyperthermic intraperitoneal chemotherapy for peritoneal metastases from a small bowel adenocarcinoma: Multi-institutional experience. Ann. Surg. Oncol. 2018, 25, 1184–1192. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Subbiah, V.; Lamhamedi-Cherradi, S.E.; Cuglievan, B.; Menegaz, B.A.; Camacho, P.; Huh, W.; Ramamoorthy, V.; Anderson, P.M.; Pollock, R.E.; Lev, D.C.; et al. Multimodality treatment of desmoplastic small round cell tumor: Chemotherapy and complete cytoreductive surgery improve patient survival. Clin. Cancer Res. 2018, 24, 4865–4873. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Horsman, M.R.; Overgaard, J. Hyperthermia: A potent enhancer of radiotherapy. Clin. Oncol. 2007, 19, 418–426. [Google Scholar] [CrossRef] [PubMed]
- Gardner, S.N. A mechanistic, predictive model of dose-response curves for cell cycle phase-specific and -nonspecific drugs. Cancer Res. 2000, 60, 1417–1425. [Google Scholar] [PubMed]
- Dakwar, G.R.; Shariati, M.; Willaert, W.; Ceelen, W.; De Smedt, S.C.; Remaut, K. Nanomedicine-based intraperitoneal therapy for the treatment of peritoneal carcinomatosis—Mission possible? Adv. Drug Deliv. Rev. 2017, 108, 13–24. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhu, L.; Xu, Y.; Shan, Y.; Zheng, R.; Wu, Z.; Ma, S. Intraperitoneal perfusion chemotherapy and whole abdominal hyperthermia using external radiofrequency following radical D2 resection for treatment of advanced gastric cancer. Int. J. Hyperth. 2019, 36, 403–407. [Google Scholar] [CrossRef]
- McCabe-Lankford, E.; McCarthy, B.; Berwick, M.A.; Salafian, K.; Galarza-Paez, L.; Sarkar, S.; Sloop, J.; Donati, G.; Brown, A.J.; Levi-Polyachenko, N. Binding of targeted semiconducting photothermal polymer nanoparticles for intraperitoneal detection and treatment of colorectal cancer. Nanotheranostics 2020, 4, 107–118. [Google Scholar] [CrossRef] [Green Version]
- Beck, M.; Ghadjar, P.; Weihrauch, M.; Burock, S.; Budach, V.; Nadobny, J.; Sehouli, J.; Wust, P. Regional hyperthermia of the abdomen, a pilot study towards the treatment of peritoneal carcinomatosis. Radiat. Oncol. 2015, 10, 157. [Google Scholar] [CrossRef] [Green Version]
- Kok, H.P.; Beck, M.; Löke, D.R.; Helderman, R.; van Tienhoven, G.; Ghadjar, P.; Wust, P.; Crezee, H. Locoregional peritoneal hyperthermia to enhance the effectiveness of chemotherapy in patients with peritoneal carcinomatosis: A simulation study comparing different locoregional heating systems. Int. J. Hyperth. 2020, 37, 76–88. [Google Scholar] [CrossRef]
- Löke, D.R.; Helderman, R.; Rodermond, H.M.; Tanis, P.J.; Streekstra, G.J.; Franken, N.A.P.; Oei, A.L.; Crezee, J.; Kok, H.P. Demonstration of treatment planning software for hyperthermic intraperitoneal chemotherapy in a rat model. Int. J. Hyperth. 2021, 38, 38–54. [Google Scholar] [CrossRef]
- McCabe-Lankford, E.; Peterson, M.; McCarthy, B.; Brown, A.J.; Terry, B.; Galarza-Paez, L.; Levi-Polyachenko, N. Murine models of intraperitoneal perfusion for disseminated colorectal cancer. J. Surg. Res. 2019, 233, 310–322. [Google Scholar] [CrossRef]
- Liesenfeld, L.F.; Hillebrecht, H.C.; Klose, J.; Schmidt, T.; Schneider, M. Impact of perfusate concentration on hyperthermic intraperitoneal chemotherapy efficacy and toxicity in a rodent model. J. Surg. Res. 2020, 253, 262–271. [Google Scholar] [CrossRef]
- Schuitevoerder, D.; Sherman, S.K.; Izquierdo, F.J.; Eng, O.S.; Turaga, K.K. Assessment of the surgical workforce pertaining to cytoreductive surgery and hyperthermic intraperitoneal chemotherapy in the United States. Ann. Surg. Oncol. 2020, 27, 3097–3102. [Google Scholar] [CrossRef]
- Ceelen, W.P. Clinical research in surgery: Threats and opportunities. Eur. Surg. Res. 2014, 53, 95–107. [Google Scholar] [CrossRef]
- Ceelen, W.; Ramsay, R.G.; Narasimhan, V.; Heriot, A.G.; De Wever, O. Targeting the tumor microenvironment in colorectal peritoneal metastases. Trends Cancer 2020, 6, 236–246. [Google Scholar] [CrossRef]
- Harvin, J.A.; Zarzaur, B.L.; Nirula, R.; King, B.T.; Malhotra, A.K. Alternative clinical trial designs. Trauma Surg. Acute Care Open 2020, 5, e000420. [Google Scholar] [CrossRef] [Green Version]
| Tumor | Study, Year | Inclusion | Primary Endpoint | Treatment and N Randomized | Results | 95% CI of Effect and P Value |
|---|---|---|---|---|---|---|
| Colorectal cancer | Verwaal [58] (2003, updated 2008) | Histologically proven PM, age <71 yrs, no distant metastasis | Disease specific survival | Chemotherapy alone (5-FU-LV) N = 51 | 12.6 m | P = 0.028 |
| CRS and HIPEC (MMC, 90 min) N = 54 | 22.2 m | |||||
| Prodige 7 (2021) [15] | Histologically proven PM, PCI ≤25 | Overall survival | CRS N = 132 | 41.2 m | HR 0.63–1.58, P = 0.99 | |
| CRS and HIPEC (OX, 30 min) N = 133 | 41.7 m | |||||
| COLOPEC (2019) [59] | Clinical or pathological T4N0–2M0-or perforated colon cancer | Peritoneal metastasis free survival at 18 months | Adjuvant HIPEC (OX, 30 min) and adjuvant chemotherapy N = 102 | 80.9% | P = 0.28 | |
| Adjuvant chemotherapy N = 102 | 76.2% | |||||
| PROPHYLOCHIP (2020) [60] | Synchronous and resected PM, resected ovarian metastases, perforated tumor | Disease free survival | Adjuvant chemotherapy and HIPEC (OX ± IRI, 30 min) N = 75 | 44% | HR 0.61–1.56, P = 0.82 | |
| Adjuvant chemotherapy N = 75 | 53% | |||||
| Rovers (2021) [61] | Histologically proven isolated resectable PM | % complete CRS/% Clavien-Dindo ≥ grade 3 morbidity | Perioperative chemotherapy and CRS-HIPEC (MMC, 90 min or OX, 30 min) N = 40 | 89%/22% | RR 0.88-1.23, P = 0.74/0.31–1.37, P = 0.25 | |
| CRS and HIPEC alone N = 40 | 86%/33% | |||||
| Ovarian cancer | Spiliotis (2015) [62] | Recurrent EOC | Overall survival | CRS and HIPEC (CIS or DOX with PTX or MMC, 60 min) N = 60 | 26.7 m | P = 0.006 |
| CRS alone N = 60 | 13.4 m | |||||
| OVHIPEC (2018) [63] | EOC with at least stable disease after three cycles of carboplatin–PTX | Recurrence free survival | Interval CRS and HIPEC (CIS, 90 min) N = 122 | 14.2 m | HR 0.50–0.87, P = 0.003 | |
| Interval CRS alone N = 123 | 10.7 m | |||||
| Zivanovic (2021) [64] | Recurrent EOC | Proportion free of progression at 24 months (‘pick the winner’) | CRS and HIPEC (Carboplatin, 90 min) followed by 5 cycles of Carboplatin based IV chemotherapy N = 49 | 16.3% | Not applicable (no winner determined) | |
| CRS alone followed by 6 cycles of Carboplatin based IV chemotherapy N = 49 | 24.5% |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ceelen, W.; Demuytere, J.; de Hingh, I. Hyperthermic Intraperitoneal Chemotherapy: A Critical Review. Cancers 2021, 13, 3114. https://doi.org/10.3390/cancers13133114
Ceelen W, Demuytere J, de Hingh I. Hyperthermic Intraperitoneal Chemotherapy: A Critical Review. Cancers. 2021; 13(13):3114. https://doi.org/10.3390/cancers13133114
Chicago/Turabian StyleCeelen, Wim, Jesse Demuytere, and Ignace de Hingh. 2021. "Hyperthermic Intraperitoneal Chemotherapy: A Critical Review" Cancers 13, no. 13: 3114. https://doi.org/10.3390/cancers13133114
APA StyleCeelen, W., Demuytere, J., & de Hingh, I. (2021). Hyperthermic Intraperitoneal Chemotherapy: A Critical Review. Cancers, 13(13), 3114. https://doi.org/10.3390/cancers13133114







