Adjuvant Chemotherapy for Stage III Colon Cancer
Simple Summary
Abstract
1. Introduction
2. Timing of Adjuvant Chemotherapy
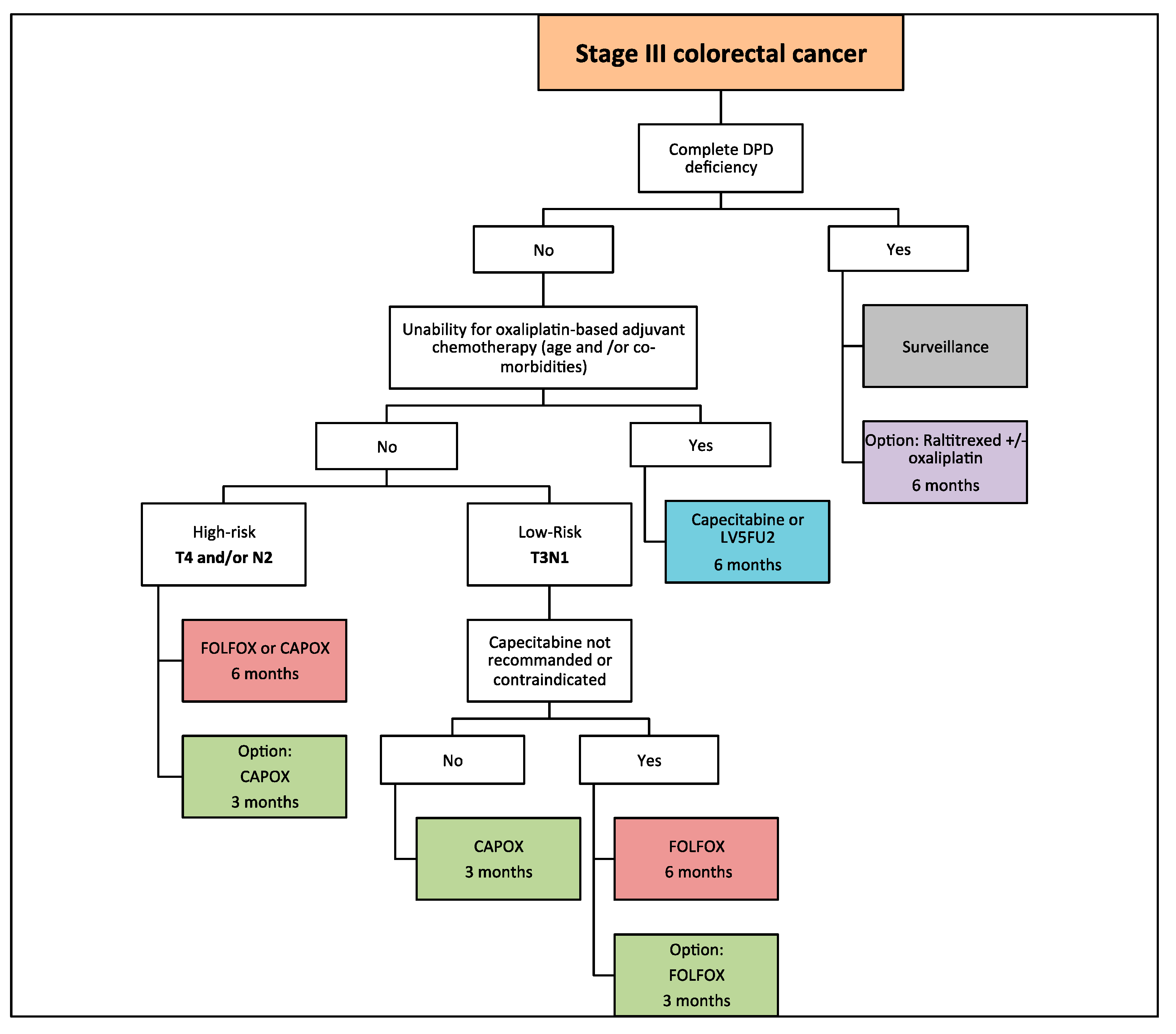
3. Chemotherapy Regimens
4. Duration of Adjuvant Chemotherapy: IDEA Collaboration
5. Adjuvant Chemotherapy in Elderly Patients
6. Neo-Adjuvant Chemotherapy for Locally Advanced Colon Cancer
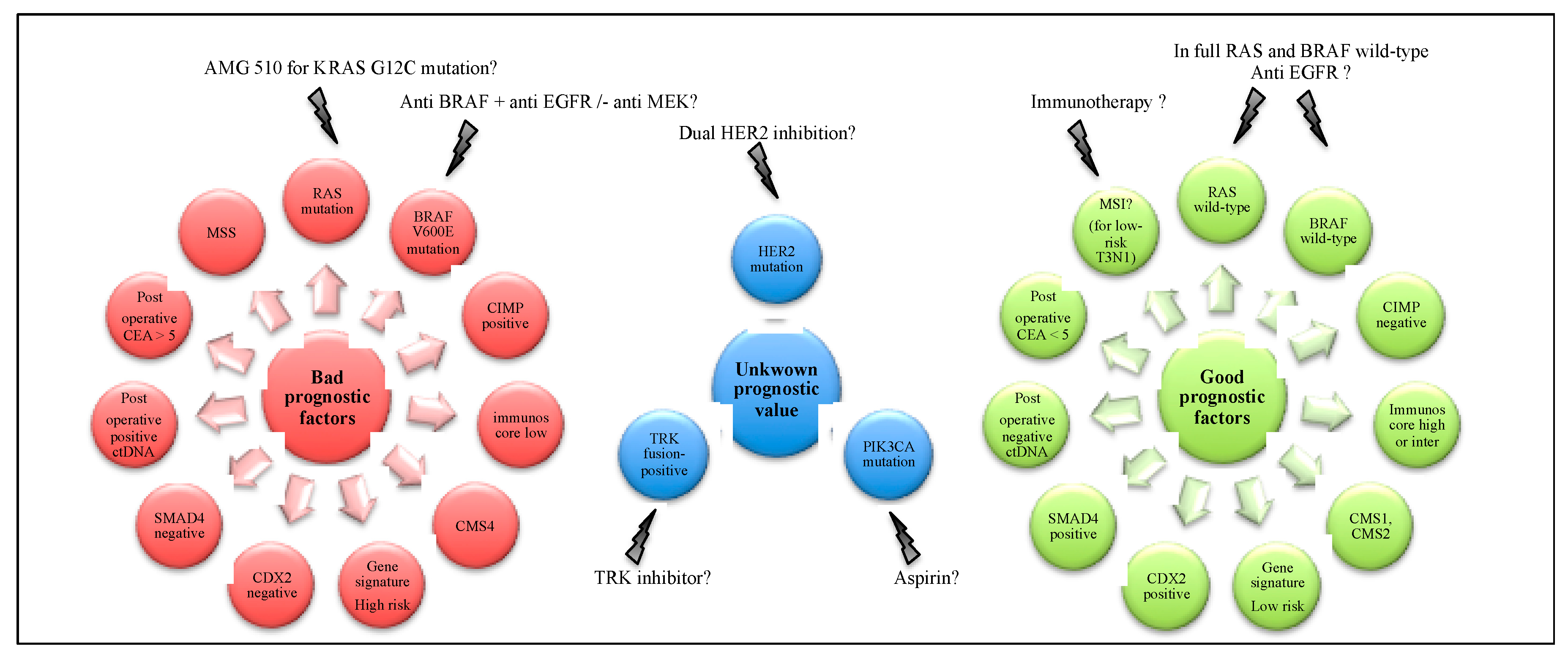
7. Biomarkers and Efficacy of Adjuvant Chemotherapy
8. Perspectives for (Neo)Adjuvant Chemotherapy in Stage III Colon Cancer
9. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2018, 68, 394–424. [Google Scholar] [CrossRef] [PubMed]
- Brierley, J.D.; Gospodarowicz, M.K.; Wittekind, C. TNM Classification of Malignant Tumours; John Wiley & Sons: Hoboken, NJ, USA, 2017; ISBN 978-1-119-26357-9. [Google Scholar]
- Moertel, C.G.; Fleming, T.R.; Macdonald, J.S.; Haller, D.G.; Laurie, J.A.; Goodman, P.J.; Ungerleider, J.S.; Emerson, W.A.; Tormey, D.C.; Glick, J.H. Levamisole and fluorouracil for adjuvant therapy of resected colon carcinoma. N. Engl. J. Med. 1990, 322, 352–358. [Google Scholar] [CrossRef] [PubMed]
- André, T.; Boni, C.; Mounedji-Boudiaf, L.; Navarro, M.; Tabernero, J.; Hickish, T.; Topham, C.; Zaninelli, M.; Clingan, P.; Bridgewater, J.; et al. Oxaliplatin, fluorouracil, and leucovorin as adjuvant treatment for colon cancer. N. Engl. J. Med. 2004, 350, 2343–2351. [Google Scholar] [CrossRef] [PubMed]
- André, T.; de Gramont, A.; Vernerey, D.; Chibaudel, B.; Bonnetain, F.; Tijeras-Raballand, A.; Scriva, A.; Hickish, T.; Tabernero, J.; Van Laethem, J.L.; et al. Adjuvant Fluorouracil, Leucovorin, and Oxaliplatin in Stage II to III Colon Cancer: Updated 10-Year Survival and Outcomes According to BRAF Mutation and Mismatch Repair Status of the MOSAIC Study. J. Clin. Oncol. 2015, 33, 4176–4187. [Google Scholar] [CrossRef]
- Taieb, J.; André, T.; Auclin, E. Refining adjuvant therapy for non-metastatic colon cancer, new standards and perspectives. Cancer Treat. Rev. 2019, 75, 1–11. [Google Scholar] [CrossRef]
- Grothey, A.; Sobrero, A.F.; Shields, A.F.; Yoshino, T.; Paul, J.; Taieb, J.; Souglakos, J.; Shi, Q.; Kerr, R.; Labianca, R.; et al. Duration of Adjuvant Chemotherapy for Stage III Colon Cancer. N. Engl. J. Med. 2018, 378, 1177–1188. [Google Scholar] [CrossRef]
- Des Guetz, G.; Nicolas, P.; Perret, G.-Y.; Morere, J.-F.; Uzzan, B. Does delaying adjuvant chemotherapy after curative surgery for colorectal cancer impair survival? A meta-analysis. Eur. J. Cancer 2010, 46, 1049–1055. [Google Scholar] [CrossRef]
- Gao, P.; Huang, X.-Z.; Song, Y.-X.; Sun, J.-X.; Chen, X.-W.; Sun, Y.; Jiang, Y.-M.; Wang, Z.-N. Impact of timing of adjuvant chemotherapy on survival in stage III colon cancer: A population-based study. BMC Cancer 2018, 18, 234. [Google Scholar] [CrossRef]
- Turner, M.C.; Farrow, N.E.; Rhodin, K.E.; Sun, Z.; Adam, M.A.; Mantyh, C.R.; Migaly, J. Delay in Adjuvant Chemotherapy and Survival Advantage in Stage III Colon Cancer. J. Am. Coll. Surg. 2018, 226, 670–678. [Google Scholar] [CrossRef]
- Labianca, R.; Marsoni, S.; Pancera, G.; Torri, V.; Zaniboni, A.; Erlichman, C.; Pater, J.; Shepherd, L.; Zee, B.; Seitz, J.F.; et al. Efficacy of adjuvant fluorouracil and folinic acid in colon cancer. Lancet 1995, 345, 939–944. [Google Scholar]
- Quasar Collaborative Group; Gray, R.; Barnwell, J.; McConkey, C.; Hills, R.K.; Williams, N.S.; Kerr, D.J. Adjuvant chemotherapy versus observation in patients with colorectal cancer: A randomised study. Lancet 2007, 370, 2020–2029. [Google Scholar] [CrossRef] [PubMed]
- Francini, G.; Petrioli, R.; Lorenzini, L.; Mancini, S.; Armenio, S.; Tanzini, G.; Marsili, S.; Aquino, A.; Marzocca, G.; Civitelli, S. Folinic acid and 5-fluorouracil as adjuvant chemotherapy in colon cancer. Gastroenterology 1994, 106, 899–906. [Google Scholar] [CrossRef]
- O’Connell, M.J.; Mailliard, J.A.; Kahn, M.J.; Macdonald, J.S.; Haller, D.G.; Mayer, R.J.; Wieand, H.S. Controlled trial of fluorouracil and low-dose leucovorin given for 6 months as postoperative adjuvant therapy for colon cancer. J. Clin. Oncol. 1997, 15, 246–250. [Google Scholar] [CrossRef]
- Andre, T.; Colin, P.; Louvet, C.; Gamelin, E.; Bouche, O.; Achille, E.; Colbert, N.; Boaziz, C.; Piedbois, P.; Tubiana-Mathieu, N.; et al. Semimonthly versus monthly regimen of fluorouracil and leucovorin administered for 24 or 36 weeks as adjuvant therapy in stage II and III colon cancer: Results of a randomized trial. J. Clin. Oncol. 2003, 21, 2896–2903. [Google Scholar] [CrossRef] [PubMed]
- Twelves, C.; Wong, A.; Nowacki, M.P.; Abt, M.; Burris, H.; Carrato, A.; Cassidy, J.; Cervantes, A.; Fagerberg, J.; Georgoulias, V.; et al. Capecitabine as adjuvant treatment for stage III colon cancer. N. Engl. J. Med. 2005, 352, 2696–2704. [Google Scholar] [CrossRef] [PubMed]
- Kuebler, J.P.; Wieand, H.S.; O’Connell, M.J.; Smith, R.E.; Colangelo, L.H.; Yothers, G.; Petrelli, N.J.; Findlay, M.P.; Seay, T.E.; Atkins, J.N.; et al. Oxaliplatin combined with weekly bolus fluorouracil and leucovorin as surgical adjuvant chemotherapy for stage II and III colon cancer: Results from NSABP C-07. J. Clin. Oncol. 2007, 25, 2198–2204. [Google Scholar] [CrossRef]
- Yothers, G.; O’Connell, M.J.; Allegra, C.J.; Kuebler, J.P.; Colangelo, L.H.; Petrelli, N.J.; Wolmark, N. Oxaliplatin as adjuvant therapy for colon cancer: Updated results of NSABP C-07 trial, including survival and subset analyses. J. Clin. Oncol. 2011, 29, 3768–3774. [Google Scholar] [CrossRef]
- Haller, D.G.; Tabernero, J.; Maroun, J.; de Braud, F.; Price, T.; Van Cutsem, E.; Hill, M.; Gilberg, F.; Rittweger, K.; Schmoll, H.-J. Capecitabine plus oxaliplatin compared with fluorouracil and folinic acid as adjuvant therapy for stage III colon cancer. J. Clin. Oncol. 2011, 29, 1465–1471. [Google Scholar] [CrossRef]
- Allegra, C.J.; Yothers, G.; O’Connell, M.J.; Sharif, S.; Petrelli, N.J.; Colangelo, L.H.; Atkins, J.N.; Seay, T.E.; Fehrenbacher, L.; Goldberg, R.M.; et al. Phase III trial assessing bevacizumab in stages II and III carcinoma of the colon: Results of NSABP protocol C-08. J. Clin. Oncol. 2011, 29, 11–16. [Google Scholar] [CrossRef]
- Alberts, S.R.; Sargent, D.J.; Nair, S.; Mahoney, M.R.; Mooney, M.; Thibodeau, S.N.; Smyrk, T.C.; Sinicrope, F.A.; Chan, E.; Gill, S.; et al. Effect of Oxaliplatin, Fluorouracil, and Leucovorin With or Without Cetuximab on Survival Among Patients With Resected Stage III Colon Cancer. JAMA 2012, 307, 1383–1393. [Google Scholar] [CrossRef]
- Saltz, L.B.; Niedzwiecki, D.; Hollis, D.; Goldberg, R.M.; Hantel, A.; Thomas, J.P.; Fields, A.L.A.; Mayer, R.J. Irinotecan fluorouracil plus leucovorin is not superior to fluorouracil plus leucovorin alone as adjuvant treatment for stage III colon cancer: Results of CALGB 89803. J. Clin. Oncol. 2007, 25, 3456–3461. [Google Scholar] [CrossRef] [PubMed]
- Ychou, M.; Raoul, J.-L.; Douillard, J.-Y.; Gourgou-Bourgade, S.; Bugat, R.; Mineur, L.; Viret, F.; Becouarn, Y.; Bouché, O.; Gamelin, E.; et al. A phase III randomised trial of LV5FU2 + irinotecan versus LV5FU2 alone in adjuvant high-risk colon cancer (FNCLCC Accord02/FFCD9802). Ann. Oncol. 2009, 20, 674–680. [Google Scholar] [CrossRef] [PubMed]
- Van Cutsem, E.; Labianca, R.; Bodoky, G.; Barone, C.; Aranda, E.; Nordlinger, B.; Topham, C.; Tabernero, J.; André, T.; Sobrero, A.F.; et al. Randomized phase III trial comparing biweekly infusional fluorouracil/leucovorin alone or with irinotecan in the adjuvant treatment of stage III colon cancer: PETACC-3. J. Clin. Oncol. 2009, 27, 3117–3125. [Google Scholar] [CrossRef] [PubMed]
- Taieb, J.; Tabernero, J.; Mini, E.; Subtil, F.; Folprecht, G.; Van Laethem, J.-L.; Thaler, J.; Bridgewater, J.; Petersen, L.N.; Blons, H.; et al. Oxaliplatin, fluorouracil, and leucovorin with or without cetuximab in patients with resected stage III colon cancer (PETACC-8): An open-label, randomised phase 3 trial. Lancet Oncol. 2014, 15, 862–873. [Google Scholar] [CrossRef]
- de Gramont, A.; Van Cutsem, E.; Schmoll, H.-J.; Tabernero, J.; Clarke, S.; Moore, M.J.; Cunningham, D.; Cartwright, T.H.; Hecht, J.R.; Rivera, F.; et al. Bevacizumab plus oxaliplatin-based chemotherapy as adjuvant treatment for colon cancer (AVANT): A phase 3 randomised controlled trial. Lancet Oncol. 2012, 13, 1225–1233. [Google Scholar] [CrossRef]
- Kerr, R.S.; Love, S.; Segelov, E.; Johnstone, E.; Falcon, B.; Hewett, P.; Weaver, A.; Church, D.; Scudder, C.; Pearson, S.; et al. Adjuvant capecitabine plus bevacizumab versus capecitabine alone in patients with colorectal cancer (QUASAR 2): An open-label, randomised phase 3 trial. Lancet Oncol. 2016, 17, 1543–1557. [Google Scholar] [CrossRef]
- Taieb, J.; Balogoun, R.; Le Malicot, K.; Tabernero, J.; Mini, E.; Folprecht, G.; Van Laethem, J.-L.; Emile, J.-F.; Mulot, C.; Fratté, S.; et al. Adjuvant FOLFOX +/− cetuximab in full RAS and BRAF wildtype stage III colon cancer patients. Ann. Oncol. 2017, 28, 824–830. [Google Scholar] [CrossRef]
- Popov, I.; Carrato, A.; Sobrero, A.; Vincent, M.; Kerr, D.; Labianca, R.; Raffaele Bianco, A.; El-Serafi, M.; Bedenne, L.; Paillot, B.; et al. Raltitrexed (Tomudex) versus standard leucovorin-modulated bolus 5-fluorouracil: Results from the randomised phase III Pan-European Trial in Adjuvant Colon Cancer 01 (PETACC-1). Eur. J. Cancer 2008, 44, 2204–2211. [Google Scholar] [CrossRef]
- Lapeyre-Prost, A.; Hug de Larauze, M.; Chibaudel, B.; Garcia, M.L.; Guering-Meyer, V.; Bouché, O.; Boucher, E.; Ychou, M.; Dauba, J.; Obled, S.; et al. Feasibility of Capecitabine and Oxaliplatin Combination Chemotherapy Without Central Venous Access Device in Patients With Stage III Colorectal Cancer. Clin. Colorectal Cancer 2016, 15, 250–256. [Google Scholar] [CrossRef]
- Chang, H.J.; Lee, K.-W.; Kim, J.H.; Bang, S.M.; Kim, Y.J.; Kim, D.W.; Kang, S.B.; Lee, J.S. Adjuvant capecitabine chemotherapy using a tailored-dose strategy in elderly patients with colon cancer. Ann. Oncol. 2012, 23, 911–918. [Google Scholar] [CrossRef]
- Diasio, R.B.; Beavers, T.L.; Carpenter, J.T. Familial deficiency of dihydropyrimidine dehydrogenase. Biochemical basis for familial pyrimidinemia and severe 5-fluorouracil-induced toxicity. J. Clin. Investig. 1988, 81, 47–51. [Google Scholar] [CrossRef] [PubMed]
- Sobrero, A.F.; Andre, T.; Meyerhardt, J.A.; Grothey, A.; Iveson, T.; Yoshino, T.; Sougklakos, I.; Meyers, J.P.; Labianca, R.; Saunders, M.P.; et al. Overall survival (OS) and long-term disease-free survival (DFS) of three versus six months of adjuvant (adj) oxaliplatin and fluoropyrimidine-based therapy for patients (pts) with stage III colon cancer (CC): Final results from the IDEA (International Duration Evaluation of Adj chemotherapy) collaboration. JCO 2020, 38, 4004. [Google Scholar] [CrossRef]
- Sargent, D.J.; Goldberg, R.M.; Jacobson, S.D.; Macdonald, J.S.; Labianca, R.; Haller, D.G.; Shepherd, L.E.; Seitz, J.F.; Francini, G. A pooled analysis of adjuvant chemotherapy for resected colon cancer in elderly patients. N. Engl. J. Med. 2001, 345, 1091–1097. [Google Scholar] [CrossRef] [PubMed]
- Mayer, B.; Sander, S.; Paschke, S.; Henne-Bruns, D.; Link, K.-H.; Kornmann, M.; Study Group Oncology of Gastrointestinal Tumors (FOGT). Stratified Survival Analysis After Adjuvant Chemotherapy of Colon Cancer Reveals a Benefit for Older Patients. Anticancer Res. 2015, 35, 5587–5593. [Google Scholar]
- Sanoff, H.K.; Carpenter, W.R.; Stürmer, T.; Goldberg, R.M.; Martin, C.F.; Fine, J.P.; McCleary, N.J.; Meyerhardt, J.A.; Niland, J.; Kahn, K.L.; et al. Effect of adjuvant chemotherapy on survival of patients with stage III colon cancer diagnosed after age 75 years. J. Clin. Oncol. 2012, 30, 2624–2634. [Google Scholar] [CrossRef] [PubMed]
- McCleary, N.J.; Meyerhardt, J.A.; Green, E.; Yothers, G.; de Gramont, A.; Van Cutsem, E.; O’Connell, M.; Twelves, C.J.; Saltz, L.B.; Haller, D.G.; et al. Impact of age on the efficacy of newer adjuvant therapies in patients with stage II/III colon cancer: Findings from the ACCENT database. J. Clin. Oncol. 2013, 31, 2600–2606. [Google Scholar] [CrossRef]
- Haller, D.G.; O’Connell, M.J.; Cartwright, T.H.; Twelves, C.J.; McKenna, E.F.; Sun, W.; Saif, M.W.; Lee, S.; Yothers, G.; Schmoll, H.-J. Impact of age and medical comorbidity on adjuvant treatment outcomes for stage III colon cancer: A pooled analysis of individual patient data from four randomized, controlled trials. Ann. Oncol. 2015, 26, 715–724. [Google Scholar] [CrossRef]
- Aparicio, T.; Francois, E.; Cristol-Dalstein, L.; Carola, E.; Maillard, E.; Paillaud, E.; Retornaz, F.; Faroux, R.; André, T.; Bedenne, L.; et al. PRODIGE 34-FFCD 1402-ADAGE: Adjuvant chemotherapy in elderly patients with resected stage III colon cancer: A randomized phase 3 trial. Dig. Liver Dis. 2016, 48, 206–207. [Google Scholar] [CrossRef]
- Karoui, M.; Rullier, A.; Piessen, G.; Legoux, J.L.; Barbier, E.; De Chaisemartin, C.; Lecaille, C.; Bouche, O.; Ammarguellat, H.; Brunetti, F.; et al. Perioperative FOLFOX 4 Versus FOLFOX 4 Plus Cetuximab Versus Immediate Surgery for High-Risk Stage II and III Colon Cancers: A Phase II Multicenter Randomized Controlled Trial (PRODIGE 22). Ann. Surg. 2020, 271, 637–645. [Google Scholar] [CrossRef]
- Seymour, M.T.; Morton, D. FOxTROT: An international randomised controlled trial in 1052 patients (pts) evaluating neoadjuvant chemotherapy (NAC) for colon cancer. JCO 2019, 37, 3504. [Google Scholar] [CrossRef]
- Gavin, P.G.; Colangelo, L.H.; Fumagalli, D.; Tanaka, N.; Remillard, M.Y.; Yothers, G.; Kim, C.; Taniyama, Y.; Kim, S.I.; Choi, H.J.; et al. Mutation profiling and microsatellite instability in stage II and III colon cancer: An assessment of their prognostic and oxaliplatin predictive value. Clin. Cancer Res. 2012, 18, 6531–6541. [Google Scholar] [CrossRef] [PubMed]
- Taieb, J.; Shi, Q.; Pederson, L.; Alberts, S.; Wolmark, N.; Van Cutsem, E.; de Gramont, A.; Kerr, R.; Grothey, A.; Lonardi, S.; et al. Prognosis of microsatellite instability and/or mismatch repair deficiency stage III colon cancer patients after disease recurrence following adjuvant treatment: Results of an ACCENT pooled analysis of seven studies. Ann. Oncol. 2019, 30, 1466–1471. [Google Scholar] [CrossRef] [PubMed]
- Ribic, C.M.; Sargent, D.J.; Moore, M.J.; Thibodeau, S.N.; French, A.J.; Goldberg, R.M.; Hamilton, S.R.; Laurent-Puig, P.; Gryfe, R.; Shepherd, L.E.; et al. Tumor microsatellite-instability status as a predictor of benefit from fluorouracil-based adjuvant chemotherapy for colon cancer. N. Engl. J. Med. 2003, 349, 247–257. [Google Scholar] [CrossRef] [PubMed]
- Sargent, D.J.; Marsoni, S.; Monges, G.; Thibodeau, S.N.; Labianca, R.; Hamilton, S.R.; French, A.J.; Kabat, B.; Foster, N.R.; Torri, V.; et al. Defective mismatch repair as a predictive marker for lack of efficacy of fluorouracil-based adjuvant therapy in colon cancer. J. Clin. Oncol. 2010, 28, 3219–3226. [Google Scholar] [CrossRef] [PubMed]
- Sinicrope, F.A.; Foster, N.R.; Thibodeau, S.N.; Marsoni, S.; Monges, G.; Labianca, R.; Kim, G.P.; Yothers, G.; Allegra, C.; Moore, M.J.; et al. DNA mismatch repair status and colon cancer recurrence and survival in clinical trials of 5-fluorouracil-based adjuvant therapy. J. Natl. Cancer Inst. 2011, 103, 863–875. [Google Scholar] [CrossRef] [PubMed]
- Taieb, J.; Le Malicot, K.; Shi, Q.; Penault Lorca, F.; Bouché, O.; Tabernero, J.; Mini, E.; Goldberg, R.M.; Folprecht, G.; Luc Van Laethem, J.; et al. Prognostic Value of BRAF and KRAS Mutations in MSI and MSS Stage III Colon Cancer. J. Natl. Cancer Inst. 2017, 109. [Google Scholar] [CrossRef]
- Hutchins, G.; Southward, K.; Handley, K.; Magill, L.; Beaumont, C.; Stahlschmidt, J.; Richman, S.; Chambers, P.; Seymour, M.; Kerr, D.; et al. Value of mismatch repair, KRAS, and BRAF mutations in predicting recurrence and benefits from chemotherapy in colorectal cancer. J. Clin. Oncol. 2011, 29, 1261–1270. [Google Scholar] [CrossRef]
- Sinicrope, F.A.; Huebner, L.J.; Laurent-Puig, P.; Smyrk, T.C.; Tabernero, J.; Mini, E.; Goldberg, R.M.; Folprecht, G.; Zaanan, A.; Le Malicot, K.; et al. Relative contribution of clinical and molecular features to outcome within low and high risk T and N groups in stage III colon cancer (CC). JCO 2019, 37, 3520. [Google Scholar] [CrossRef]
- Tougeron, D.; Mouillet, G.; Trouilloud, I.; Lecomte, T.; Coriat, R.; Aparicio, T.; Des Guetz, G.; Lécaille, C.; Artru, P.; Sickersen, G.; et al. Efficacy of Adjuvant Chemotherapy in Colon Cancer With Microsatellite Instability: A Large Multicenter AGEO Study. J. Natl. Cancer Inst. 2016, 108. [Google Scholar] [CrossRef]
- Zaanan, A.; Fléjou, J.-F.; Emile, J.-F.; Des, G.G.; Cuilliere-Dartigues, P.; Malka, D.; Lecaille, C.; Validire, P.; Louvet, C.; Rougier, P.; et al. Defective mismatch repair status as a prognostic biomarker of disease-free survival in stage III colon cancer patients treated with adjuvant FOLFOX chemotherapy. Clin. Cancer Res. 2011, 17, 7470–7478. [Google Scholar] [CrossRef]
- Mireia, G.R.; Gallach, S.; Jantus-Lewintre, E.; Safont, M.J.; Maria Luisa, G.M.; Garde-Noguera, J.; Maria Dolores, R.C.; Blasco, S.; Giner-Bosch, V.; Camps, C. Molecular subtyping of colon cancer (CC) based on mutational status of RAS, BRAF, and DNA mismatch repair (MMR) proteins. Prognostic value. JCO 2016, 34, e15094. [Google Scholar] [CrossRef]
- Mouradov, D.; Sloggett, C.; Jorissen, R.N.; Love, C.G.; Li, S.; Burgess, A.W.; Arango, D.; Strausberg, R.L.; Buchanan, D.; Wormald, S.; et al. Colorectal cancer cell lines are representative models of the main molecular subtypes of primary cancer. Cancer Res. 2014, 74, 3238–3247. [Google Scholar] [CrossRef]
- Van Rijnsoever, M.; Elsaleh, H.; Joseph, D.; McCaul, K.; Iacopetta, B. CpG island methylator phenotype is an independent predictor of survival benefit from 5-fluorouracil in stage III colorectal cancer. Clin. Cancer Res. 2003, 9, 2898–2903. [Google Scholar] [PubMed]
- Gallois, C.; Taieb, J.; Le Corre, D.; Le Malicot, K.; Tabernero, J.; Mulot, C.; Seitz, J.-F.; Aparicio, T.; Folprecht, G.; Lepage, C.; et al. Prognostic value of methylator phenotype in stage III colon cancer treated with oxaliplatin-based adjuvant chemotherapy. Clin. Cancer Res. 2018. [Google Scholar] [CrossRef]
- Shiovitz, S.; Bertagnolli, M.M.; Renfro, L.A.; Nam, E.; Foster, N.R.; Dzieciatkowski, S.; Luo, Y.; Lao, V.V.; Monnat, R.J.; Emond, M.J.; et al. CpG island methylator phenotype is associated with response to adjuvant irinotecan-based therapy for stage III colon cancer. Gastroenterology 2014, 147, 637–645. [Google Scholar] [CrossRef] [PubMed]
- Domingo, E.; Church, D.N.; Sieber, O.; Ramamoorthy, R.; Yanagisawa, Y.; Johnstone, E.; Davidson, B.; Kerr, D.J.; Tomlinson, I.P.M.; Midgley, R. Evaluation of PIK3CA Mutation As a Predictor of Benefit From Nonsteroidal Anti-Inflammatory Drug Therapy in Colorectal Cancer. JCO 2013, 31, 4297–4305. [Google Scholar] [CrossRef] [PubMed]
- Liao, X.; Lochhead, P.; Nishihara, R.; Morikawa, T.; Kuchiba, A.; Yamauchi, M.; Imamura, Y.; Qian, Z.R.; Baba, Y.; Shima, K.; et al. Aspirin use, tumor PIK3CA mutation, and colorectal-cancer survival. N. Engl. J. Med. 2012, 367, 1596–1606. [Google Scholar] [CrossRef]
- Michel, P.; Boige, V.; Andre, T.; Aparicio, T.; Bachet, J.B.; Dahan, L.; Guimbaud, R.; Lepage, C.; Manfredi, S.; Tougeron, D.; et al. Aspirin versus placebo in stage III or high-risk stage II colon cancer with PIK3CA mutation: A French randomised double-blind phase III trial (PRODIGE 50-ASPIK). Dig. Liver Dis. 2018, 50, 305–307. [Google Scholar] [CrossRef]
- Gray, R.G.; Quirke, P.; Handley, K.; Lopatin, M.; Magill, L.; Baehner, F.L.; Beaumont, C.; Clark-Langone, K.M.; Yoshizawa, C.N.; Lee, M.; et al. Validation study of a quantitative multigene reverse transcriptase-polymerase chain reaction assay for assessment of recurrence risk in patients with stage II colon cancer. J. Clin. Oncol. 2011, 29, 4611–4619. [Google Scholar] [CrossRef]
- Venook, A.P.; Niedzwiecki, D.; Lopatin, M.; Ye, X.; Lee, M.; Friedman, P.N.; Frankel, W.; Clark-Langone, K.; Millward, C.; Shak, S.; et al. Biologic Determinants of Tumor Recurrence in Stage II Colon Cancer: Validation Study of the 12-Gene Recurrence Score in Cancer and Leukemia Group B (CALGB) 9581. J. Clin. Oncol. 2013, 31, 1775–1781. [Google Scholar] [CrossRef]
- Yothers, G.; O’Connell, M.J.; Lee, M.; Lopatin, M.; Clark-Langone, K.M.; Millward, C.; Paik, S.; Sharif, S.; Shak, S.; Wolmark, N. Validation of the 12-gene colon cancer recurrence score in NSABP C-07 as a predictor of recurrence in patients with stage II and III colon cancer treated with fluorouracil and leucovorin (FU/LV) and FU/LV plus oxaliplatin. J. Clin. Oncol. 2013, 31, 4512–4519. [Google Scholar] [CrossRef]
- Di Narzo, A.F.; Tejpar, S.; Rossi, S.; Yan, P.; Popovici, V.; Wirapati, P.; Budinska, E.; Xie, T.; Estrella, H.; Pavlicek, A.; et al. Test of four colon cancer risk-scores in formalin fixed paraffin embedded microarray gene expression data. J. Natl. Cancer Inst. 2014, 106. [Google Scholar] [CrossRef] [PubMed]
- Kopetz, S.; Jiang, Z.-Q.; Overman, M.J.; Rosenberg, R.; Salazar, R.; Tabernero, J.; Stork, L.; Li, Y.; Simon, I.; Chang, G.J.; et al. Genomic classifier (ColoPrint) to predict outcome and chemotherapy benefit in stage II and III colon cancer patients. JCO 2013, 31, 3612. [Google Scholar] [CrossRef]
- Wang, Y.; Jatkoe, T.; Zhang, Y.; Mutch, M.G.; Talantov, D.; Jiang, J.; McLeod, H.L.; Atkins, D. Gene expression profiles and molecular markers to predict recurrence of Dukes’ B colon cancer. J. Clin. Oncol. 2004, 22, 1564–1571. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Y.; Casey, G.; Lavery, I.C.; Zhang, Y.; Talantov, D.; Martin-McGreevy, M.; Skacel, M.; Manilich, E.; Mazumder, A.; Atkins, D.; et al. Development of a clinically feasible molecular assay to predict recurrence of stage II colon cancer. J. Mol. Diagn. 2008, 10, 346–354. [Google Scholar] [CrossRef] [PubMed]
- Kennedy, R.D.; Bylesjo, M.; Kerr, P.; Davison, T.; Black, J.M.; Kay, E.W.; Holt, R.J.; Proutski, V.; Ahdesmaki, M.; Farztdinov, V.; et al. Development and independent validation of a prognostic assay for stage II colon cancer using formalin-fixed paraffin-embedded tissue. J. Clin. Oncol. 2011, 29, 4620–4626. [Google Scholar] [CrossRef]
- Agesen, T.H.; Sveen, A.; Merok, M.A.; Lind, G.E.; Nesbakken, A.; Skotheim, R.I.; Lothe, R.A. ColoGuideEx: A robust gene classifier specific for stage II colorectal cancer prognosis. Gut 2012, 61, 1560–1567. [Google Scholar] [CrossRef]
- Zhang, J.-X.; Song, W.; Chen, Z.-H.; Wei, J.-H.; Liao, Y.-J.; Lei, J.; Hu, M.; Chen, G.-Z.; Liao, B.; Lu, J.; et al. Prognostic and predictive value of a microRNA signature in stage II colon cancer: A microRNA expression analysis. Lancet Oncol. 2013, 14, 1295–1306. [Google Scholar] [CrossRef]
- Guinney, J.; Dienstmann, R.; Wang, X.; de Reyniès, A.; Schlicker, A.; Soneson, C.; Marisa, L.; Roepman, P.; Nyamundanda, G.; Angelino, P.; et al. The consensus molecular subtypes of colorectal cancer. Nat. Med. 2015, 21, 1350–1356. [Google Scholar] [CrossRef]
- Marisa, L.; Ayadi, M.; Balogoun, R.; Pilati, C.; Le Malicot, K.; Lepage, C.; Emile, J.-F.; Salazar, R.; Aust, D.E.; Duval, A.; et al. Clinical utility of colon cancer molecular subtypes: Validation of two main colorectal molecular classifications on the PETACC-8 phase III trial cohort. JCO 2017, 35, 3509. [Google Scholar] [CrossRef]
- Laurent-Puig, P.; Marisa, L.; Ayadi, M.; Blum, Y.; Balogoun, R.; Pilati, C.; Le Malicot, K.; Lepage, C.; Emile, J.-F.; Salazar, R.; et al. 60PDColon cancer molecular subtype intratumoral heterogeneity and its prognostic impact: An extensive molecular analysis of the PETACC-8. Ann. Oncol. 2018, 29. [Google Scholar] [CrossRef]
- Dalerba, P.; Sahoo, D.; Paik, S.; Guo, X.; Yothers, G.; Song, N.; Wilcox-Fogel, N.; Forgó, E.; Rajendran, P.S.; Miranda, S.P.; et al. CDX2 as a Prognostic Biomarker in Stage II and Stage III Colon Cancer. N. Engl. J. Med. 2016, 374, 211–222. [Google Scholar] [CrossRef] [PubMed]
- Zhang, B.; Zhang, B.; Chen, X.; Bae, S.; Singh, K.; Washington, M.K.; Datta, P.K. Loss of Smad4 in colorectal cancer induces resistance to 5-fluorouracil through activating Akt pathway. Br. J. Cancer 2014, 110, 946–957. [Google Scholar] [CrossRef] [PubMed]
- Kozak, M.M.; von Eyben, R.; Pai, J.; Vossler, S.R.; Limaye, M.; Jayachandran, P.; Anderson, E.M.; Shaffer, J.L.; Longacre, T.; Pai, R.K.; et al. Smad4 inactivation predicts for worse prognosis and response to fluorouracil-based treatment in colorectal cancer. J. Clin. Pathol. 2015, 68, 341–345. [Google Scholar] [CrossRef]
- Yan, P.; Klingbiel, D.; Saridaki, Z.; Ceppa, P.; Curto, M.; McKee, T.A.; Roth, A.; Tejpar, S.; Delorenzi, M.; Bosman, F.T.; et al. Reduced Expression of SMAD4 Is Associated with Poor Survival in Colon Cancer. Clin. Cancer Res. 2016, 22, 3037–3047. [Google Scholar] [CrossRef] [PubMed]
- Pagès, F.; Mlecnik, B.; Marliot, F.; Bindea, G.; Ou, F.-S.; Bifulco, C.; Lugli, A.; Zlobec, I.; Rau, T.T.; Berger, M.D.; et al. International validation of the consensus Immunoscore for the classification of colon cancer: A prognostic and accuracy study. Lancet 2018, 391, 2128–2139. [Google Scholar] [CrossRef]
- Pagès, F.; André, T.; Taieb, J.; Vernerey, D.; Henriques, J.; Borg, C.; Marliot, F.; Ben Jannet, R.; Louvet, C.; Mineur, L.; et al. Prognostic and predictive value of the Immunoscore in stage III colon cancer patients treated with oxaliplatin in the prospective IDEA France PRODIGE-GERCOR cohort study. Ann. Oncol. 2020, 31, 921–929. [Google Scholar] [CrossRef]
- Reichling, C.; Taieb, J.; Derangere, V.; Klopfenstein, Q.; Le Malicot, K.; Gornet, J.-M.; Becheur, H.; Fein, F.; Cojocarasu, O.; Kaminsky, M.C.; et al. Artificial intelligence-guided tissue analysis combined with immune infiltrate assessment predicts stage III colon cancer outcomes in PETACC08 study. Gut 2020, 69, 681–690. [Google Scholar] [CrossRef]
- Lecomte, T.; Berger, A.; Zinzindohoué, F.; Micard, S.; Landi, B.; Blons, H.; Beaune, P.; Cugnenc, P.-H.; Laurent-Puig, P. Detection of free-circulating tumor-associated DNA in plasma of colorectal cancer patients and its association with prognosis. Int. J. Cancer 2002, 100, 542–548. [Google Scholar] [CrossRef]
- Fan, G.; Zhang, K.; Yang, X.; Ding, J.; Wang, Z.; Li, J. Prognostic value of circulating tumor DNA in patients with colon cancer: Systematic review. PLoS ONE 2017, 12, e0171991. [Google Scholar] [CrossRef]
- Tarazona, N.; Gimeno-Valiente, F.; Gambardella, V.; Zuñiga, S.; Rentero-Garrido, P.; Huerta, M.; Roselló, S.; Martinez-Ciarpaglini, C.; Carbonell-Asins, J.A.; Carrasco, F.; et al. Targeted next-generation sequencing of circulating-tumor DNA for tracking minimal residual disease in localized colon cancer. Ann. Oncol. 2019. [Google Scholar] [CrossRef] [PubMed]
- Tie, J.; Cohen, J.D.; Wang, Y.; Christie, M.; Simons, K.; Lee, M.; Wong, R.; Kosmider, S.; Ananda, S.; McKendrick, J.; et al. Circulating Tumor DNA Analyses as Markers of Recurrence Risk and Benefit of Adjuvant Therapy for Stage III Colon Cancer. JAMA Oncol. 2019. [Google Scholar] [CrossRef] [PubMed]
- Reinert, T.; Henriksen, T.V.; Christensen, E.; Sharma, S.; Salari, R.; Sethi, H.; Knudsen, M.; Nordentoft, I.; Wu, H.-T.; Tin, A.S.; et al. Analysis of Plasma Cell-Free DNA by Ultradeep Sequencing in Patients With Stages I to III Colorectal Cancer. JAMA Oncol. 2019. [Google Scholar] [CrossRef]
- Taieb, J.; Taly, V.; Vernerey, D.; Bourreau, C.; Bennouna, J.; Faroux, R.; Desrame, J.; Bouche, O.; Borg, C.; Egreteau, J.; et al. LBA30_PR-Analysis of circulating tumour DNA (ctDNA) from patients enrolled in the IDEA-FRANCE phase III trial: Prognostic and predictive value for adjuvant treatment duration. Ann. Oncol. 2019, 30, v867. [Google Scholar] [CrossRef]
- Overman, M.J.; McDermott, R.; Leach, J.L.; Lonardi, S.; Lenz, H.-J.; Morse, M.A.; Desai, J.; Hill, A.; Axelson, M.; Moss, R.A.; et al. Nivolumab in patients with metastatic DNA mismatch repair-deficient or microsatellite instability-high colorectal cancer (CheckMate 142): An open-label, multicentre, phase 2 study. Lancet Oncol. 2017, 18, 1182–1191. [Google Scholar] [CrossRef]
- Andre, T.; Shiu, K.-K.; Kim, T.W.; Jensen, B.V.; Jensen, L.H.; Punt, C.J.A.; Smith, D.M.; Garcia-Carbonero, R.; Benavides, M.; Gibbs, P.; et al. Pembrolizumab versus chemotherapy for microsatellite instability-high/mismatch repair deficient metastatic colorectal cancer: The phase 3 KEYNOTE-177 Study. JCO 2020, 38, LBA4. [Google Scholar] [CrossRef]
- Chalabi, M.; Fanchi, L.F.; Dijkstra, K.K.; Van den Berg, J.G.; Aalbers, A.G.; Sikorska, K.; Lopez-Yurda, M.; Grootscholten, C.; Beets, G.L.; Snaebjornsson, P.; et al. Neoadjuvant immunotherapy leads to pathological responses in MMR-proficient and MMR-deficient early-stage colon cancers. Nat. Med. 2020, 26, 566–576. [Google Scholar] [CrossRef]
- Sinicrope, F.A.; Ou, F.-S.; Zemla, T.; Nixon, A.B.; Mody, K.; Levasseur, A.; Dueck, A.C.; Dhanarajan, A.R.; Lieu, C.H.; Cohen, D.J.; et al. Randomized trial of standard chemotherapy alone or combined with atezolizumab as adjuvant therapy for patients with stage III colon cancer and deficient mismatch repair (ATOMIC, Alliance A021502). JCO 2019, 37, e15169. [Google Scholar] [CrossRef]
- Lau, D.; Cunningham, D.; Gillbanks, A.; Crux, R.; Powell, R.; Kalaitzaki, E.; Annels, N.E.; Sclafani, F.; Gerlinger, M.; Chau, I.; et al. POLEM: Avelumab plus fluoropyrimidine-based chemotherapy as adjuvant treatment for stage III dMMR or POLE exonuclease domain mutant colon cancer—A phase III randomized study. JCO 2019, 37, TPS3615. [Google Scholar] [CrossRef]
- Kopetz, S.; Grothey, A.; Yaeger, R.; Van Cutsem, E.; Desai, J.; Yoshino, T.; Wasan, H.; Ciardiello, F.; Loupakis, F.; Hong, Y.S.; et al. Encorafenib, Binimetinib, and Cetuximab in BRAF V600E-Mutated Colorectal Cancer. N. Engl. J. Med. 2019. [Google Scholar] [CrossRef]
- Sartore-Bianchi, A.; Trusolino, L.; Martino, C.; Bencardino, K.; Lonardi, S.; Bergamo, F.; Zagonel, V.; Leone, F.; Depetris, I.; Martinelli, E.; et al. Dual-targeted therapy with trastuzumab and lapatinib in treatment-refractory, KRAS codon 12/13 wild-type, HER2-positive metastatic colorectal cancer (HERACLES): A proof-of-concept, multicentre, open-label, phase 2 trial. Lancet Oncol. 2016, 17, 738–746. [Google Scholar] [CrossRef]
- Drilon, A.; Laetsch, T.W.; Kummar, S.; DuBois, S.G.; Lassen, U.N.; Demetri, G.D.; Nathenson, M.; Doebele, R.C.; Farago, A.F.; Pappo, A.S.; et al. Efficacy of Larotrectinib in TRK Fusion-Positive Cancers in Adults and Children. N. Engl. J. Med. 2018, 378, 731–739. [Google Scholar] [CrossRef] [PubMed]
- Canon, J.; Rex, K.; Saiki, A.Y.; Mohr, C.; Cooke, K.; Bagal, D.; Gaida, K.; Holt, T.; Knutson, C.G.; Koppada, N.; et al. The clinical KRAS(G12C) inhibitor AMG 510 drives anti-tumour immunity. Nature 2019, 575, 217–223. [Google Scholar] [CrossRef] [PubMed]
- Bennouna, J.; André, T.; Campion, L.; Hiret, S.; Miglianico, L.; Mineur, L.; Touchefeu, Y.; Artru, P.; Asmis, T.; Bouché, O.; et al. Rationale and Design of the IROCAS Study: Multicenter, International, Randomized Phase 3 Trial Comparing Adjuvant Modified (m) FOLFIRINOX to mFOLFOX6 in Patients With High-Risk Stage III (pT4 and/or N2) Colon Cancer-A UNICANCER GI-PRODIGE Trial. Clin. Colorectal Cancer 2019, 18, e69–e73. [Google Scholar] [CrossRef] [PubMed]
- Conroy, T.; Lamfichekh, N.; Etienne, P.-L.; Rio, E.; Francois, E.; Mesgouez-Nebout, N.; Vendrely, V.; Artignan, X.; Bouché, O.; Gargot, D.; et al. Total neoadjuvant therapy with mFOLFIRINOX versus preoperative chemoradiation in patients with locally advanced rectal cancer: Final results of PRODIGE 23 phase III trial, a UNICANCER GI trial. JCO 2020, 38, 4007. [Google Scholar] [CrossRef]
- Taïeb, J.; Benhaim, L.; Laurent Puig, P.; Le Malicot, K.; Emile, J.F.; Geillon, F.; Tougeron, D.; Manfredi, S.; Chauvenet, M.; Taly, V.; et al. Decision for adjuvant treatment in stage II colon cancer based on circulating tumor DNA:The CIRCULATE-PRODIGE 70 trial. Dig. Liver Dis. 2020, 52, 730–733. [Google Scholar] [CrossRef]
| First Author | Study Name | N | Year | Stage | Chemotherapy | Duration | DFS | OS | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 3–year DFS | 5–year DFS | HR | p | 3–year OS | 5–year OS | HR | p | |||||||
| IMPACT [11] | IMPACT | 652 | 1982–1989 | III | FULV | 6 months | 62% | 0.55 | < 10−4 | 76% | 0.71 | 0.02 | ||
| Surgery alone | 44% | 64% | ||||||||||||
| Francini et al. [13] | 118 | 1985–1990 | III | FULV | 12 months | 69% | 0.0025 | |||||||
| Surgery alone | 43% | |||||||||||||
| O Connell et al. [14] | 317 | 1988–1989 | II–III | FULV | 6 months | 74% | 0.01 | |||||||
| Surgery alone | 58% | |||||||||||||
| André et al. [15] | 905 | 1996–1999 | II–III | LV5FU2 | 6/9 months | 73% | 1.04 | 0.7 | 86% | 1.26 | 0.18 | |||
| FULV | 6/9 months | 72% | 88% | |||||||||||
| Twelves et al. [16] | X-ACT | 1987 | 1998–2001 | III | Capecitabine | 6 months | 64% | 0.87 | 0.12 | 81% | 0.84 | 0.07 | ||
| FULV | 6 months | 61% | 78% | |||||||||||
| André et al. [4] | MOSAIC | 2246 | 1998–2001 | II–III | FOLFOX | 6 months | 78% | 0.76 | 0.002 | 88% | 0.90 | |||
| LV5FU2 | 6 months | 73% | 87% | |||||||||||
| Ychou et al. [23] | FNCLCC Accord02 /FFCD9802 | 400 | 1998–2002 | III | FOLFIRI | 6 months | 51% | 1.19 | 0.22 | 61% | 1.20 | 0.26 | ||
| LV5FU2 | 6 months | 60% | 67% | |||||||||||
| Popov et al. [29] | PETACC-1 | 1921 | 1999 | III | Raltitrexed | 6 months | 62% | 1.04 | ||||||
| FU/LV | 6 months | 62% | ||||||||||||
| Saltz et al. [22] | CALGB 89803 | 1264 | 1999–2001 | III | CPT-11 + FU/LV | 8 months | 66% | 59% | 0.85 | 80% | 68% | 0.74 | ||
| FU/LV | 8 months | 69% | 61% | 81% | 71% | |||||||||
| Kuebler et al. [17] Yothers et al. [18] | NSABP C-07 | 2409 | 2000–2002 | II–III | FLOX | 6 months | 69% | 0.82 | 0.002 | 80% | 0.88 | 0.08 | ||
| FULV | 6 months | 64% | 78% | 0.09 | ||||||||||
| Van Cutsem et al. [24] | PETACC-3 | 2094 | 2000–2002 | III | FOLFIRI | 6 months | 57% | 0.9 | 0.1 | 74% | ||||
| LV5FU2 | 6 months | 54% | 71% | |||||||||||
| Haller et al. [19] | XELOXA | 1886 | 2003–2004 | III | XELOX | 6 months | 66% | 0.80 | 0.004 | 78% | 0.87 | 0.15 | ||
| FU/FA | 6 months | 60% | 74% | |||||||||||
| Allegra et al. [20] | NSABP C-08 | 2672 | 2004–2006 | II–III | FOLFOX + bevacizumab | FOLFOX 6 months Bevacizumab 1 year | 77% | 0.89 | 0.15 | 82% | 0.95 | 0.56 | ||
| FOLFOX | 6 months | 75% | 81% | |||||||||||
| De Gramont et al. [26] | AVANT | 2867 | 2004–2007 | III | FOLFOX +Bevacizumab | 6 months 1 year | 73% | 1.17 | 0.07 | 81% | 1.27 | 0.02 | ||
| CAPOX + Bevacizumab | 6 months 1 year | 75% | 1.07 | 0.44 | 82% | 1.15 | 0.21 | |||||||
| FOLFOX | 76% | 85% | ||||||||||||
| Alberts et al. [21] | NCCTG NO147 | 1863 (KRAS WT) | 2004–2009 | III | FOLFOX + cetuximab | 6 months | 71% | 1.21 | 0.08 | 86% | 1.25 | 0.15 | ||
| FOLFOX | 6 months | 75% | 87% | |||||||||||
| Taieb et al. [25] | PETACC-8 | 1602 (KRAS WT) | 2005–2009 | III | FOLFOX + cetuximab | 6 months | 75% | 1.05 | 0.66 | 88% | 1.09 | 0.56 | ||
| FOLFOX | 6 months | 78% | 90% | |||||||||||
| Kerr et al. [27] | QUASAR,2 | 1941 | 2005–2010 | II–III | Capecitabine +Bevacizumab | 6 months 1 year | 75% | 1.06 | 0.54 | 87% | 1.11 | 0.33 | ||
| Capecitabine | 6 months | 78% | 89% | |||||||||||
| Grothey et al. [7] Sobrero et al. [33] | IDEA | 12834 | 2007–2015 | III | FOLFOX/CAPOX | 3 months | 75% | 1.07 | 0.11 | 82% | 1.02 | 0.64 | ||
| FOLFOX/CAPOX | 6 months | 75% | 83% | |||||||||||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Taieb, J.; Gallois, C. Adjuvant Chemotherapy for Stage III Colon Cancer. Cancers 2020, 12, 2679. https://doi.org/10.3390/cancers12092679
Taieb J, Gallois C. Adjuvant Chemotherapy for Stage III Colon Cancer. Cancers. 2020; 12(9):2679. https://doi.org/10.3390/cancers12092679
Chicago/Turabian StyleTaieb, Julien, and Claire Gallois. 2020. "Adjuvant Chemotherapy for Stage III Colon Cancer" Cancers 12, no. 9: 2679. https://doi.org/10.3390/cancers12092679
APA StyleTaieb, J., & Gallois, C. (2020). Adjuvant Chemotherapy for Stage III Colon Cancer. Cancers, 12(9), 2679. https://doi.org/10.3390/cancers12092679





