Micro Milled Microfluidic Photoionization Detector for Volatile Organic Compounds
Abstract
:1. Introduction
2. Micro PID
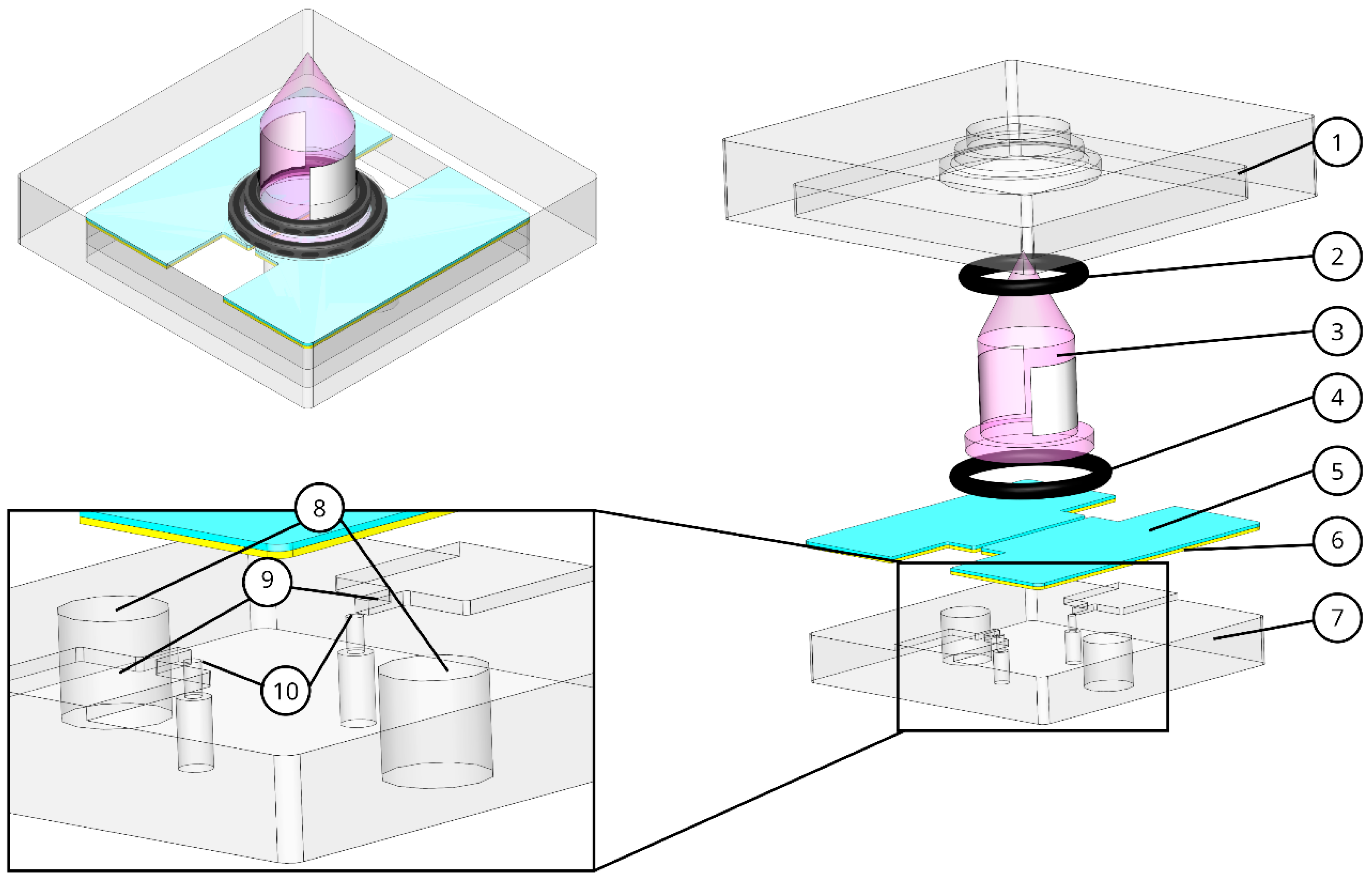
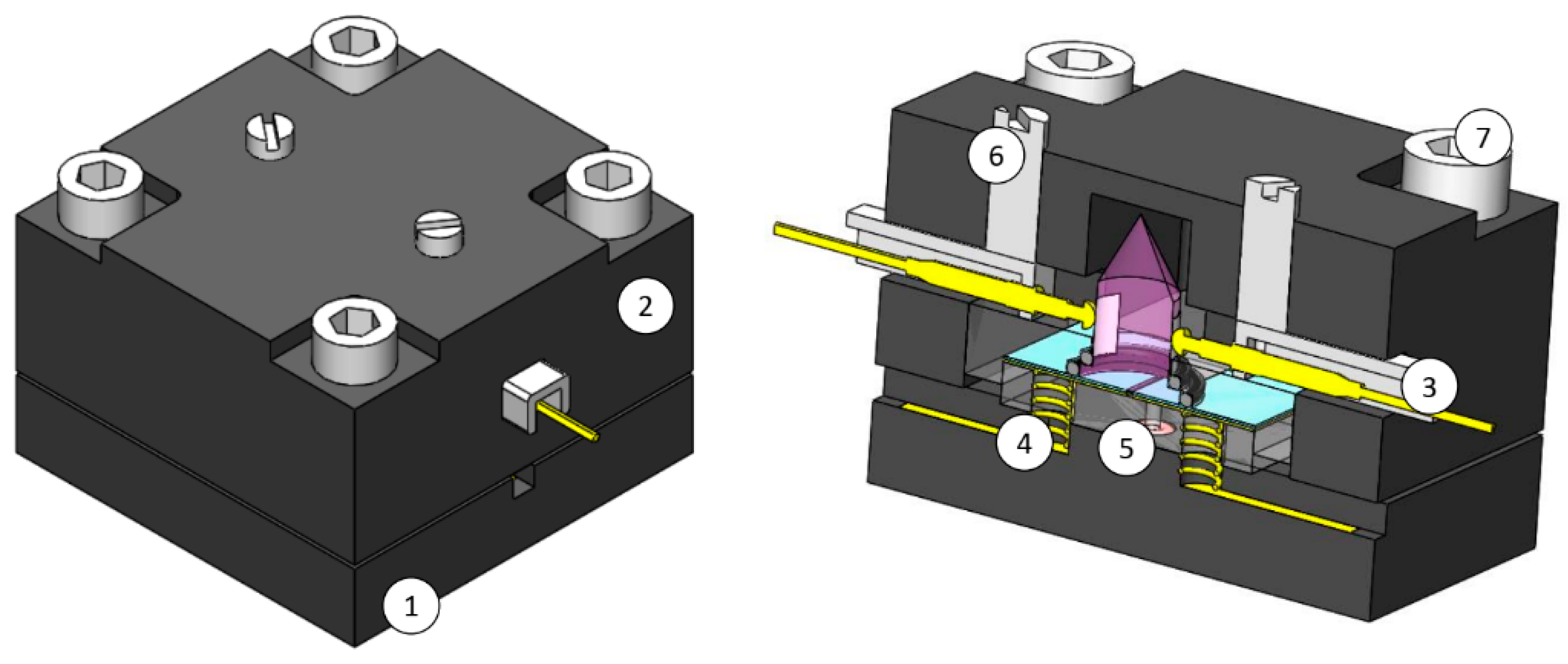
2.1. Design, Fabrication and Assembly
2.1.1. Core
2.1.2. Shell
2.2. Ionization Chamber
2.3. Coating Shield
2.3.1. DLC Coating
2.3.2. PMMA Coating
3. Results
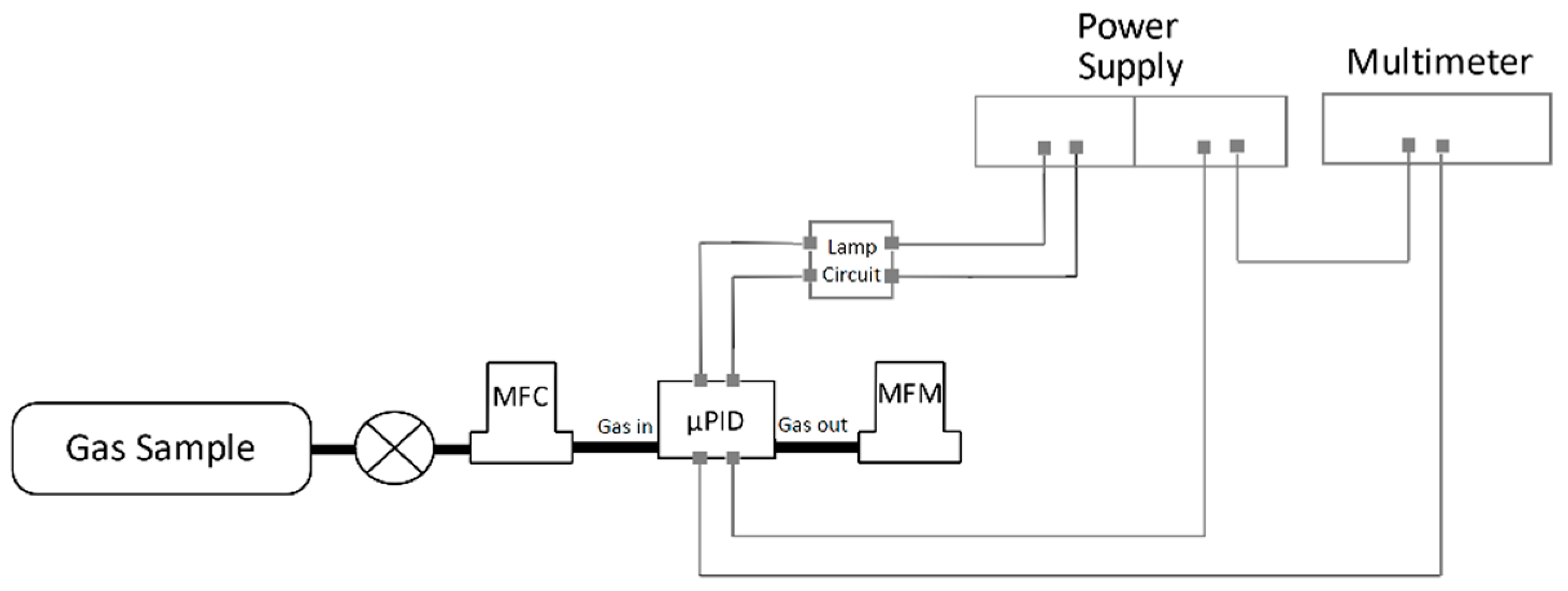
3.1. Experimental Setup and Material
3.2. Optical Microscope Images
3.3. Experimental Results and Discussion
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Wallace, L.A.; Pellizzari, E.; Leaderer, B.; Zelon, H.; Sheldon, L. Emissions of Volatile Organic Compounds from Building Materials and Consumer Products. Atmos. Environ. 1987, 21, 385–393. [Google Scholar] [CrossRef]
- WHO. Air Quality Guidelines for Europe, 2nd ed.; WHO Regional Office for Europe: Copenhagen, Denmark, 2000. [Google Scholar]
- WHO. WHO Guidelines for Indoor Air Quality: Selected Pollutants; WHO Regional Office for Europe: Copenhagen, Denmark, 2010. [Google Scholar]
- Council of the European Union; European Parliament. DIRECTIVE 2000/69/EC OF THE EUROPEAN PARLIAMENT AND OF THE COUNCIL of 16 November 2000 Relating to Limit Values for Benzene and Carbon Monoxide in Ambient Air. Off. J. Eur. Communities 2000, 313, 12–21. [Google Scholar]
- Ministère de l’Ecologie. Décret No 2011-1727 Du 2 Décembre 2011 Relatif Aux Valeurs-Guides Pour l’air Intérieur Pour Le Formaldéhyde et Le Benzène. J. Off. de la République Française 2011. [Google Scholar]
- BTEX Analyzer with PID Detector: ChromaPID. Available online: http://www.chromatotec.com/BTEX,analyzer,with,PID,detector,chromaPID-Article-138-ChromaGC-Product-14.html (accessed on 19 July 2018).
- BTEX (Model GC955-600) Benzene/Toluene/Xylene Analyser. Available online: http://www.et.co.uk/products/air-quality-monitoring/continuous-gas-analysers/synspec-gc955-601-btx/ (accessed on 19 July 2018).
- Baseline MOCON. Available online: http://www.baseline-mocon.com (accessed on 6 July 2018).
- Khan, S.; Newport, D.; Le Calvé, S. Development of a Toluene Detector Based on Deep UV Absorption Spectrophotometry Using Glass and Aluminum Capillary Tube Gas Cells with a LED Source. Micromachines 2019, 10, 193. [Google Scholar] [CrossRef] [PubMed]
- Rezende, G.C.; Le Calvé, S.; Brandner, J.J.; Newport, D. Micro photoionization detectors. Sens. Actuators B Chem. 2019, 287, 86–94. [Google Scholar] [CrossRef]
- Model 4430, IO Analytical. Available online: http://aimanalytical.com/Manuals/PIDFID manual.pdf (accessed on 20 July 2018).
- Photoionization Detector, SRI Instruments. Available online: https://www.srigc.com/home/product_detail/pid---photo-ionization-detector (accessed on 4 October 2018).
- Ion Science. Available online: https://www.ionscience.com/ (accessed on 6 July 2018).
- Sun, J.; Guan, F.; Cui, D.; Chen, X.; Zhang, L.; Chen, J. An Improved photoionization detector with a micro gas chromatography column for portable rapid gas chromatography system. Sens. Actuators B Chem. 2013, 188, 513–518. [Google Scholar] [CrossRef]
- Zhu, H.; Nidetz, R.; Zhou, M.; Lee, J.; Buggaveeti, S.; Kurabayashi, K.; Fan, X. Flow-through microfluidic photoionization detectors for rapid and highly sensitive vapor detection. Lab Chip 2015, 15, 3021–3029. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Zhou, M.; Zhu, H.; Nidetz, R.; Kurabayashi, K.; Fan, X. Fully Automated Portable Comprehensive 2-Dimensional Gas Chromatography Device. Anal. Chem. 2016, 88, 10266–10274. [Google Scholar] [CrossRef] [PubMed]
- Akbar, M.; Shakeel, H.; Agah, M. GC-on-Chip: Integrated column and photoionization detector. Lab Chip 2015, 15, 1748–1758. [Google Scholar] [CrossRef] [PubMed]
- Zhu, H.; Zhou, M.; Lee, J.; Nidetz, R.; Kurabayashi, K.; Fan, X. Low-power miniaturized helium dielectric barrier discharge photoionization detectors for highly sensitive vapor detection. Anal. Chem. 2016, 88, 8780–8786. [Google Scholar] [CrossRef] [PubMed]
- Narayanan, S.; Rice, G.; Agah, M. A Micro-discharge photoionization detector for micro-gas chromatography. Microchim. Acta 2014, 181, 493–499. [Google Scholar] [CrossRef]
- Narayanan, S.; Rice, G.; Agah, M. Characterization of a micro-helium discharge detector for gas chromatography. Sens. Actuators B Chem. 2015, 206, 190–197. [Google Scholar] [CrossRef]
- Akbar, M.; Restaino, M.; Agah, M. Chip-scale gas chromatography: from injection through detection. Microsyst. Nanoeng. 2015, 1, 15039. [Google Scholar] [CrossRef]
- Zhou, M.; Lee, J.; Zhu, H.; Nidetz, R.; Kurabayashi, K.; Fan, X. A fully automated portable gas chromatography system for sensitive and rapid quantification of volatile organic compounds in water. RSC Adv. 2016, 6, 49416–49424. [Google Scholar] [CrossRef]
- Haynes, W.M. CRC Handbook of Chemistry and Physics, 97th ed.; CRC Press: Boca Raton, FL, USA, 2016. [Google Scholar]
- Rahman, F. Nanostructures in Electronics and Photonics, 1st ed.; CRC Press: Boca Raton, FL, USA, 2008. [Google Scholar]
- Mednikarov, B.; Spasov, G.; Pirov, J.; Sahatchieva, M.; Popov, C.; Kulischa, W. Optical properties of diamond-like carbon and nanocrystalline diamond films. J. Optoelectron. Adv. Mater. 2005, 7, 1407–1413. [Google Scholar]
- Lin, C.R.; Wei, D.H.; Chang, C.K.; Liao, W.H. Optical properties of diamond-like carbon films for antireflection coating by RF magnetron sputtering method. Phys. Procedia. 2011, 18, 46–50. [Google Scholar] [CrossRef]
- Joram, C. Transmission Curves of Plexiglass (PMMA) and Optical Grease; PH-EP-Tech-Note-2009-003; CERN Document Server: Meyrin, Switzerland, 2009. [Google Scholar]



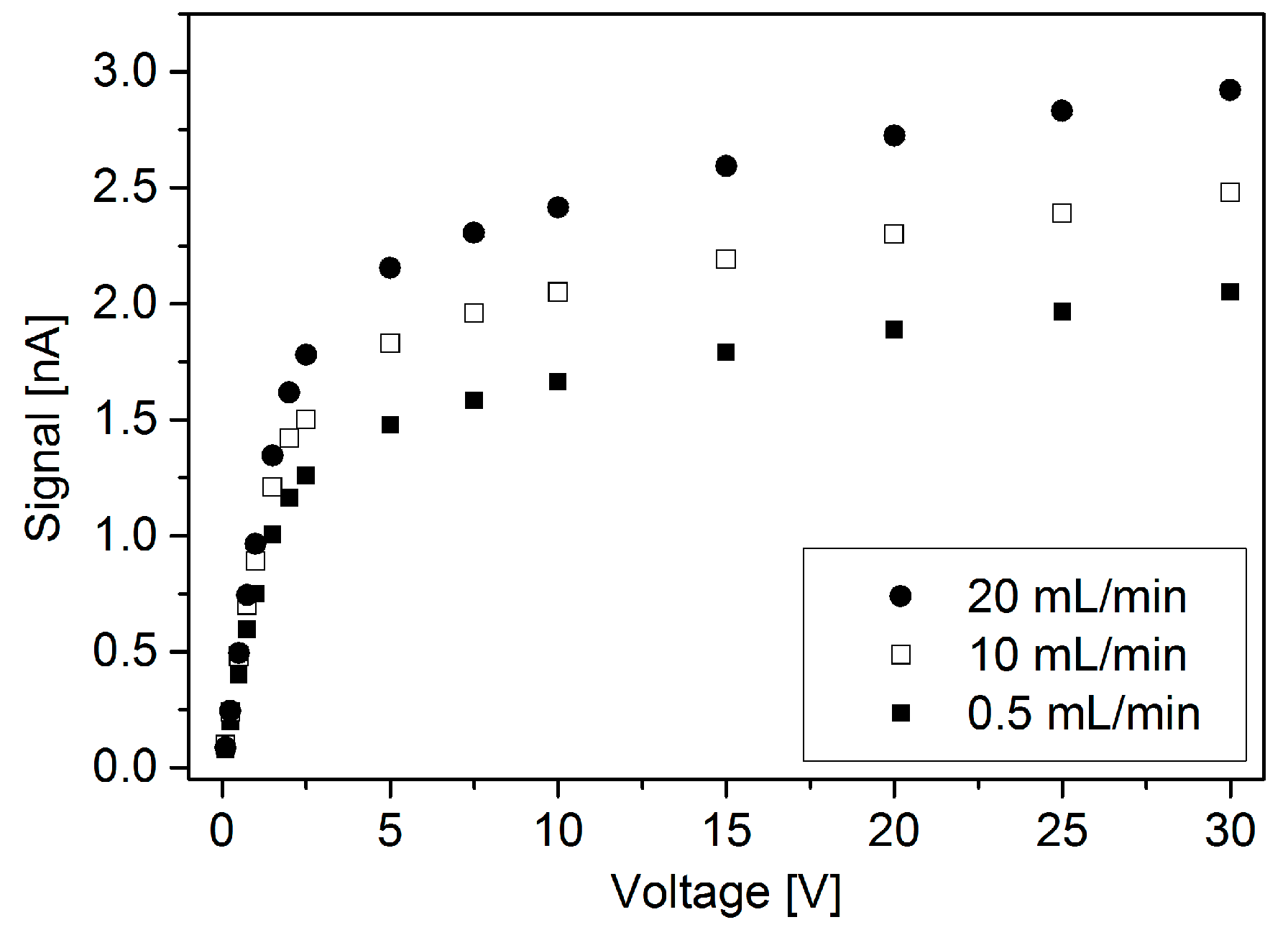
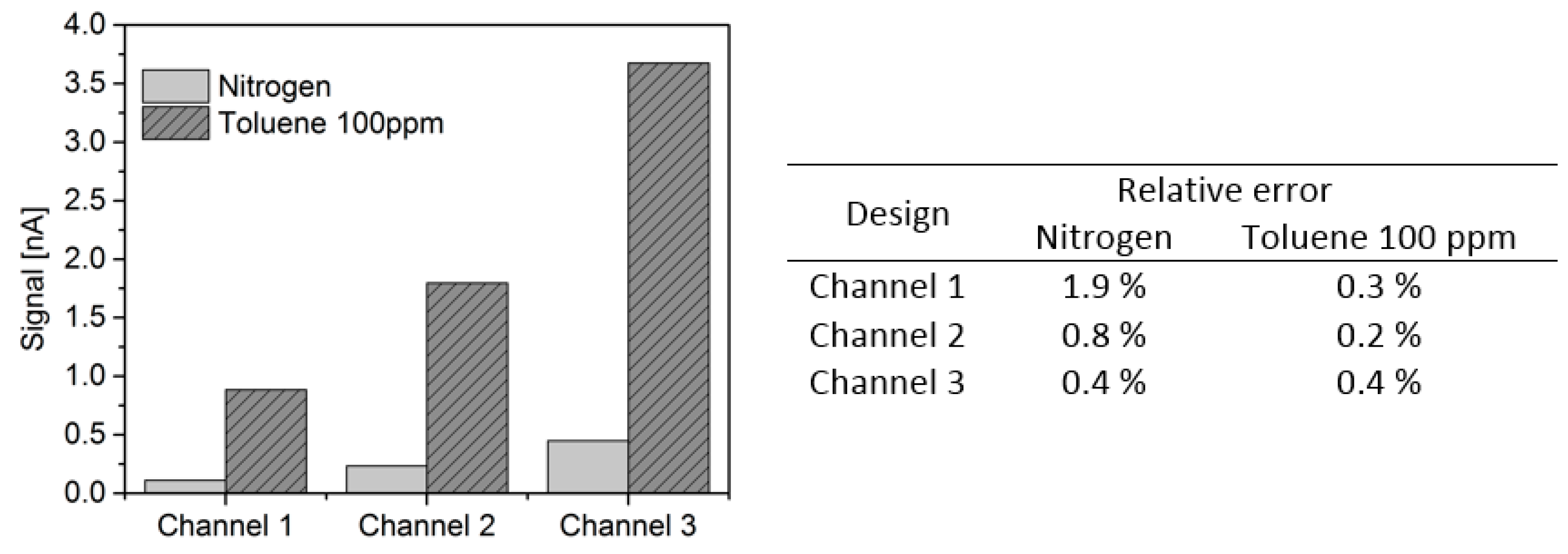
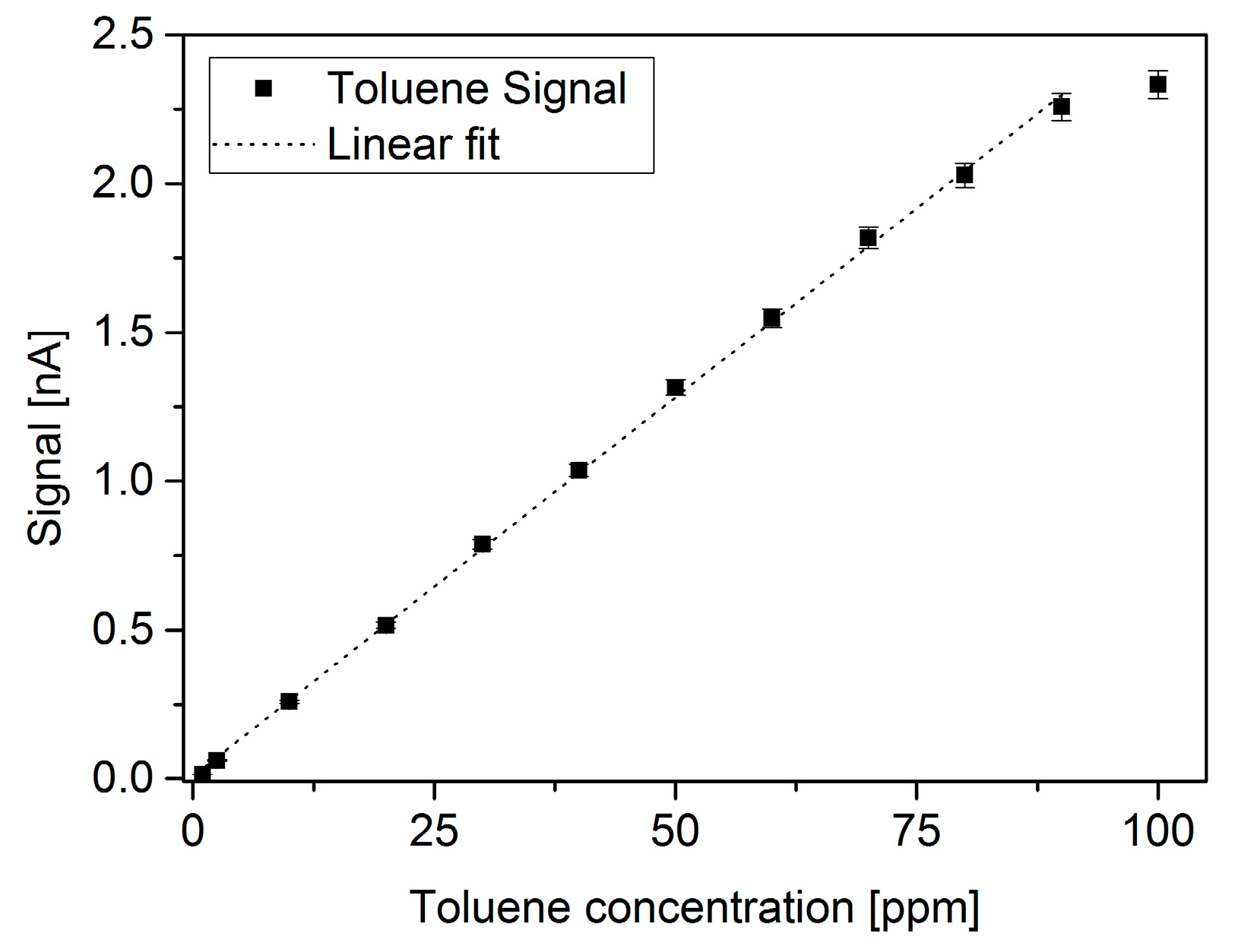
| Reference | Ionization Source | Manufacturing Main Materials | Design Main Features and Dimensions | Ionization Chamber |
|---|---|---|---|---|
| [14] | UV Lamp, 10.6 eV | - | Introduced nozzle inside a conventional ionization chamber. | 10 µL |
| [15] | UV Lamp, 10.6 eV | Highly doped p-type <100> single-sided polished conductive Si wafers with resistivity 0.001–0.005 Ω.cm and 380 µm thickness; 500 μm thick Pyrex glass wafers. | Ionization chamber is a microchannel with cross-section 150 μm (width), 380 μm (depth) and length 2.3 cm. Entire overall channel size is 15 mm × 15 mm. Microchannel area covered by lamp is 2.4 mm × 2.4 mm. | 1.3 μL |
| [16] | UV Lamp, 10.6 eV | Conductive p-type <100> silicon wafer and glass. | Channel etched 380 μm (width) × 380 μm (depth) × 2 cm (length). | 0.5 µL |
| [17] | Helium discharge | Silicon and glass architecture. | Micro separation column fabricated on the same chip. Overall size (1.5 cm × 3 cm) | Not mentioned |
| [18] | Helium discharge | 500 μm thick p-type <100> double side polished Si wafer with 500 nm thick thermal oxide layers; 100 μm thick Borofloat 33 glass wafer; 500 μm thick Borofloat 33 glass wafer. | Microchannels formed by Si and glass. Three main channels: 1) Auxiliary helium; 2) Analytes; 3) Outlet channel. Cross-section 380 μm (width) and 500 μm (depth); | 1.4 µL |
| [19] | Helium discharge | Two (bottom and top) Borosilicate glass wafers 700 μm thickness and 100 mm diameter used as substrate. | Channel etched 250 µm (depth). | Not mentioned |
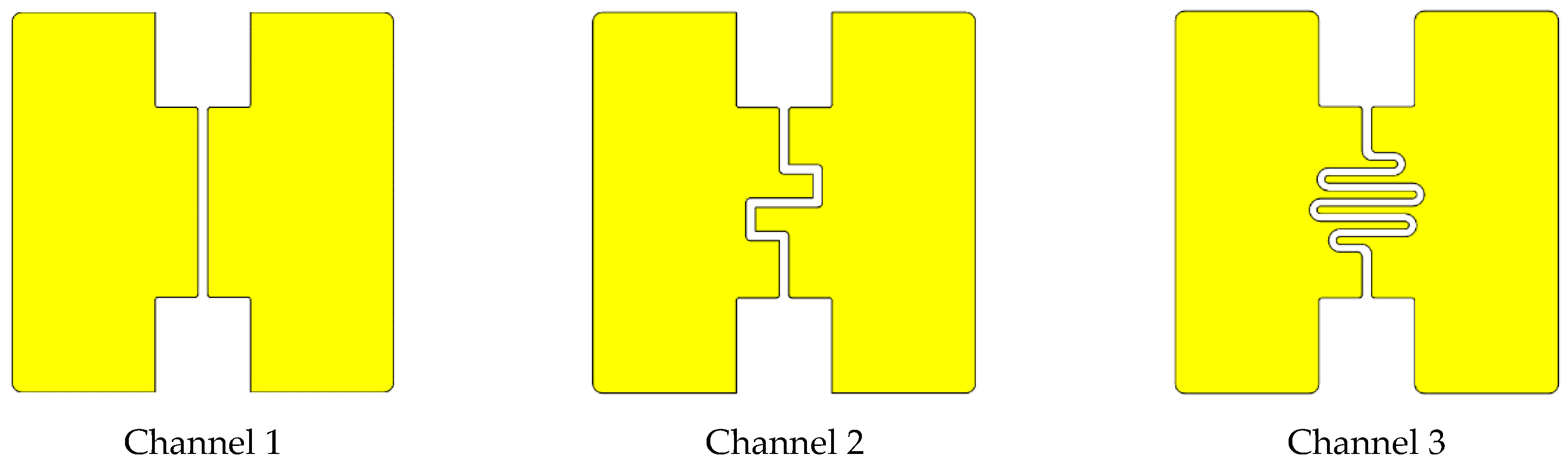
| This work | UV Lamp, 10.6 eV | Micromilled PMMA and PVC. Copper plate. | Modular assembly of components. No use of glue. Microchannels width vary between 400 and 500 µm. | 1.75–6.42 µL |
| Channel n | w [µm] | Aelectrodes [mm²] | Vinlet/outlet [µL] | A/A1 | V/V1 |
|---|---|---|---|---|---|
| 1 | 500 | 5.98 | 1.75 | 1.0 | 1.0 |
| 2 | 500 | 12.67 | 2.75 | 2.1 | 1.6 |
| 3 | 400 | 31.17 | 6.42 | 5.2 | 3.7 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rezende, G.C.; Le Calvé, S.; Brandner, J.J.; Newport, D. Micro Milled Microfluidic Photoionization Detector for Volatile Organic Compounds. Micromachines 2019, 10, 228. https://doi.org/10.3390/mi10040228
Rezende GC, Le Calvé S, Brandner JJ, Newport D. Micro Milled Microfluidic Photoionization Detector for Volatile Organic Compounds. Micromachines. 2019; 10(4):228. https://doi.org/10.3390/mi10040228
Chicago/Turabian StyleRezende, Gustavo C., Stéphane Le Calvé, Jürgen J. Brandner, and David Newport. 2019. "Micro Milled Microfluidic Photoionization Detector for Volatile Organic Compounds" Micromachines 10, no. 4: 228. https://doi.org/10.3390/mi10040228
APA StyleRezende, G. C., Le Calvé, S., Brandner, J. J., & Newport, D. (2019). Micro Milled Microfluidic Photoionization Detector for Volatile Organic Compounds. Micromachines, 10(4), 228. https://doi.org/10.3390/mi10040228







