Controlled Focused Ion Beam Milling of Composite Solid State Nanopore Arrays for Molecule Sensing
Abstract
1. Introduction
1.1. Transport in Nanoscale
- Permselectivity induces asymmetric pressure or electric field driven molecule transport through the nanochannels or nanopores.
- Size dependent transpore permeation of molecules is caused by the “Born repulsion” evolved due to the electrical interaction between the overlapping electron clouds of translocating molecules and atoms of the pore surface.
- Reconfiguration of macromolecules can also be experienced (in the case of deoxyribonucleic acid (DNA) chains) due to the entropic barrier conditions.
1.2. Applications of Nanofluidic Systems
- Development of robust and engineered fabrication techniques for reproducible nanostructure formation.
- Clarification the comprehensive theory and construction predictive modelling of nanoscale transport phenomena.
- Precise control and in-situ monitoring of physical or chemical parameters (e.g., transport properties or local surface modification) inside the nanopores.
1.3. Nanopore Structures and Their Fabrication Techniques
- Biological nanoporous layers are mainly based on pore-forming transmembrane proteins embedded in lipid bilayer membranes.
- Hybrid type nanopore membranes consist of similar biological pore-forming molecules integrated in synthetic layers.
- Artificial nanopore structures are fabricated by dedicated nanoprocessing methods in synthetic materials, polymer, glass, quartz or dielectric layers compatible with silicon technology (silicon-nitride, silicon-oxide, silicon-carbide, possibly Atomic Layer Deposited metal-oxides), or 2D materials (graphene, molybdenum-oxide, etc.).
2. Materials and Methods
2.1. Fabrication of Nanopore Arrays by Focused Ion Beam (FIB)
2.2. Characterisation of Nanopore Geometries
3. Results and Discussion—Study and Improvement of FIB Process
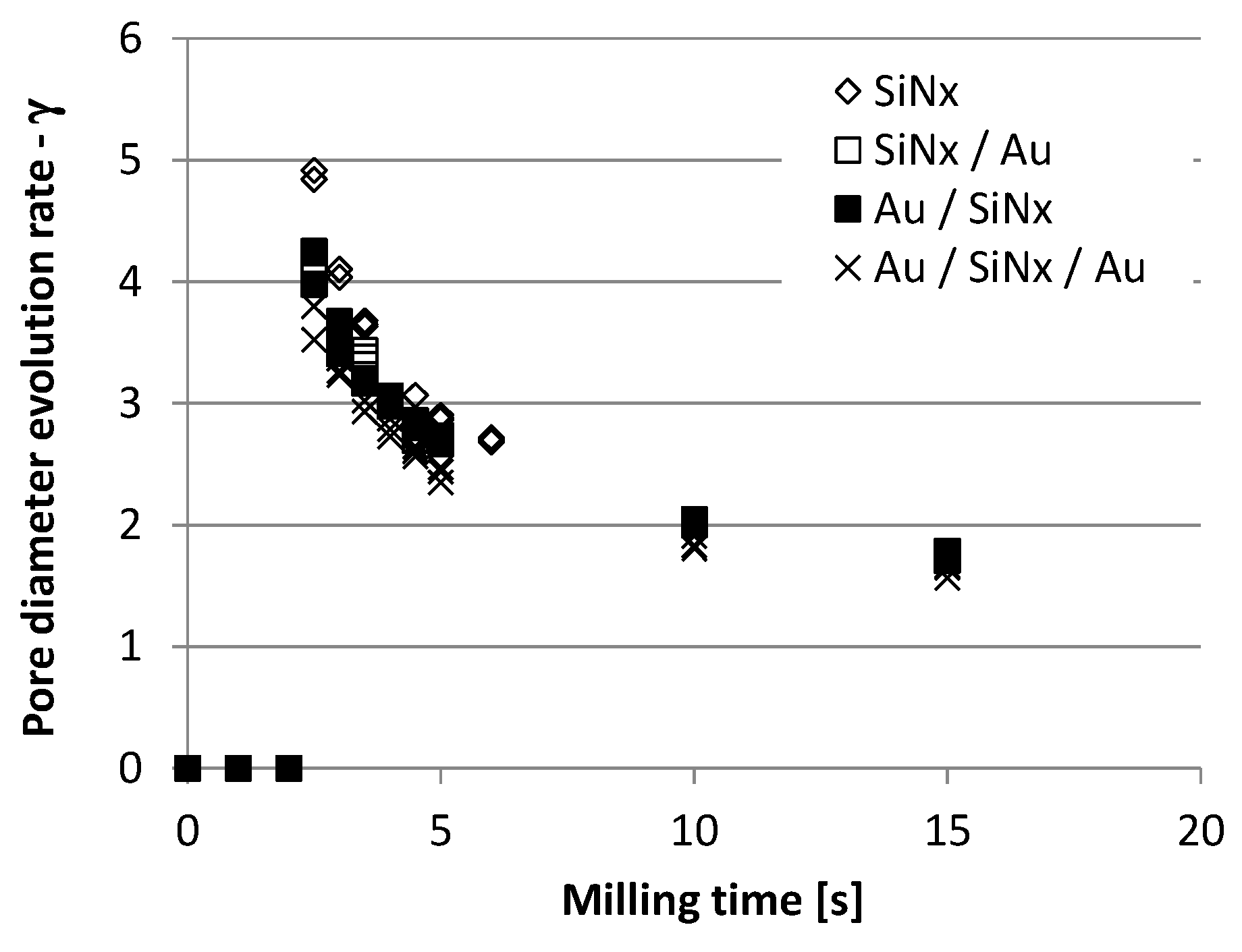
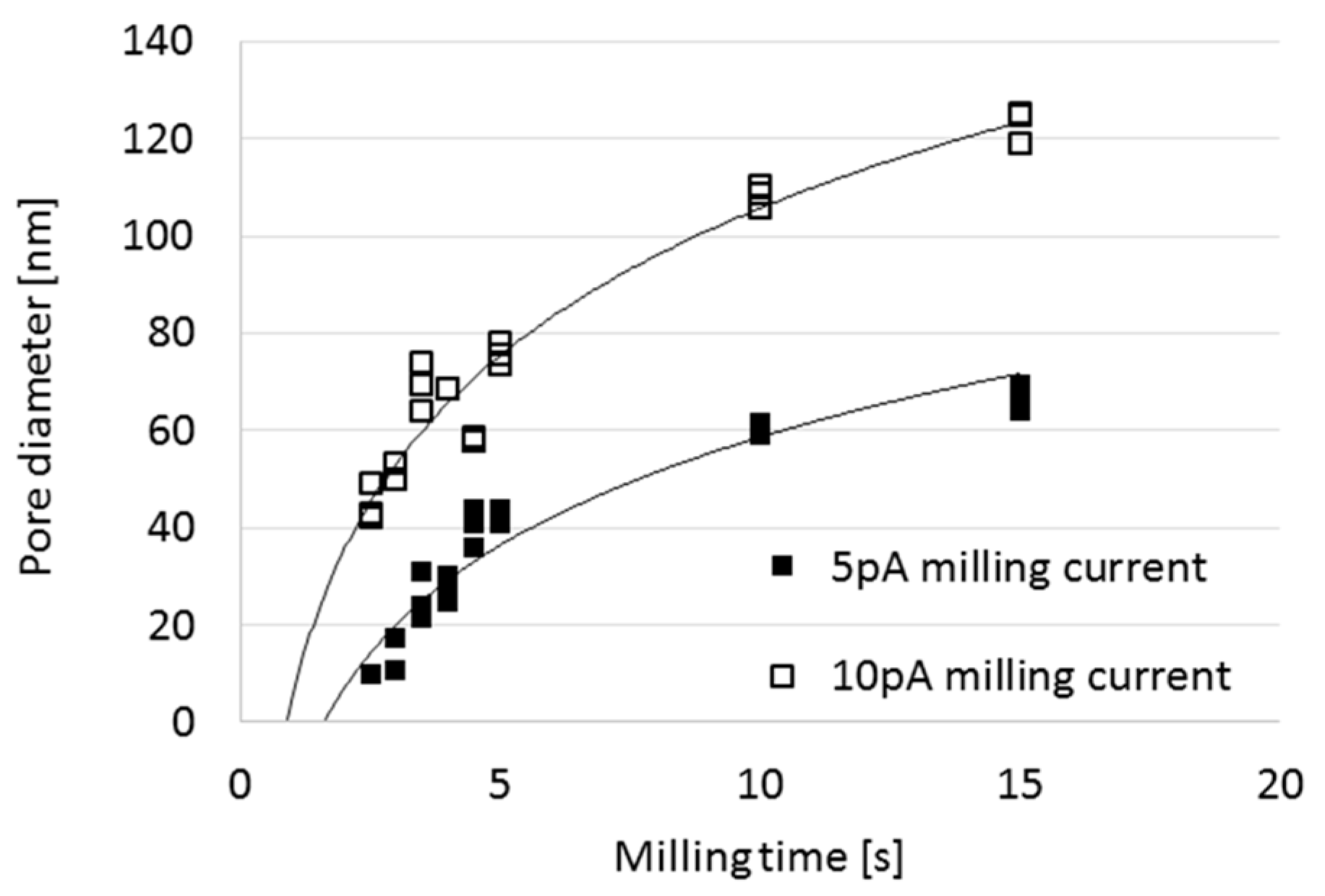
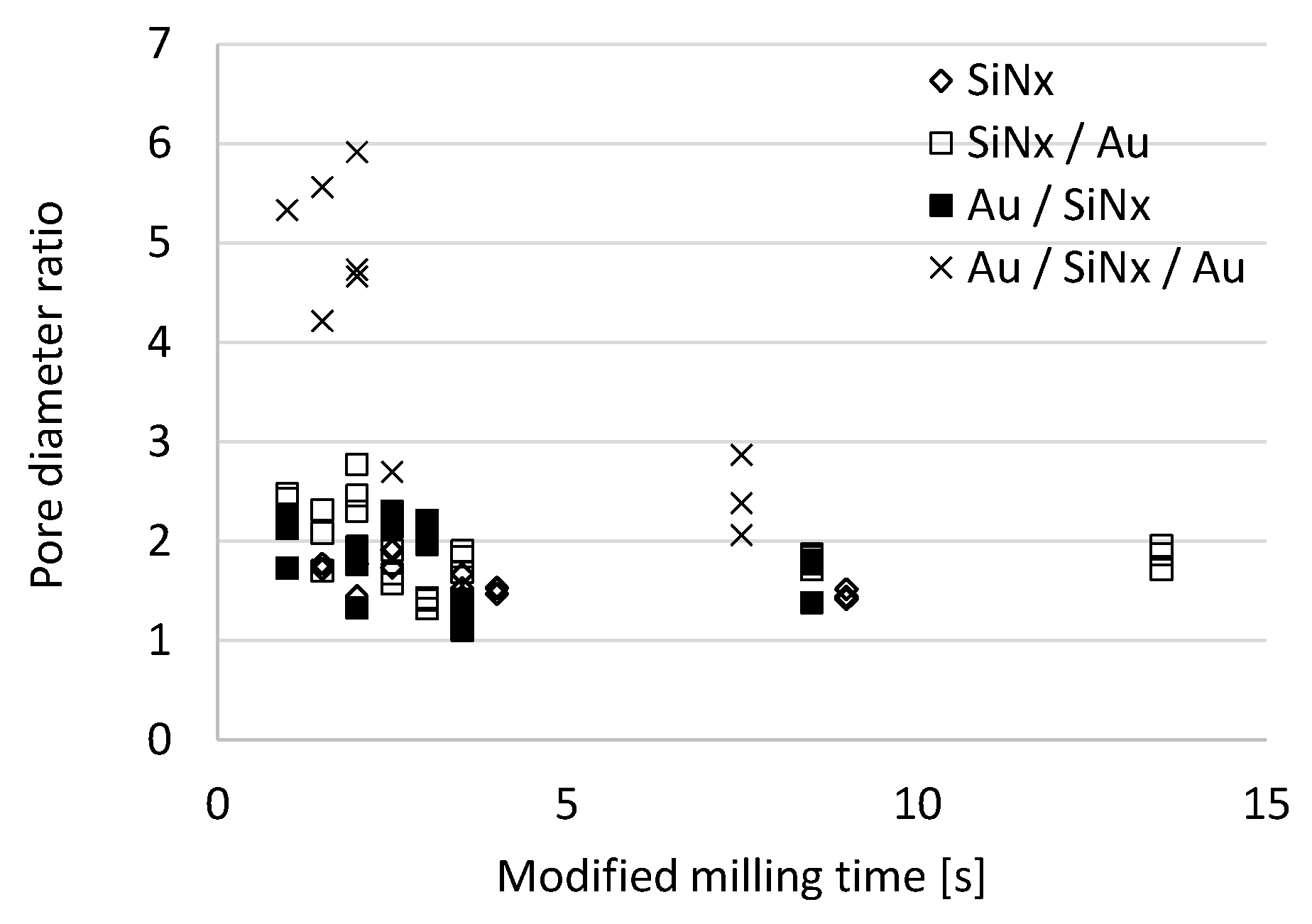
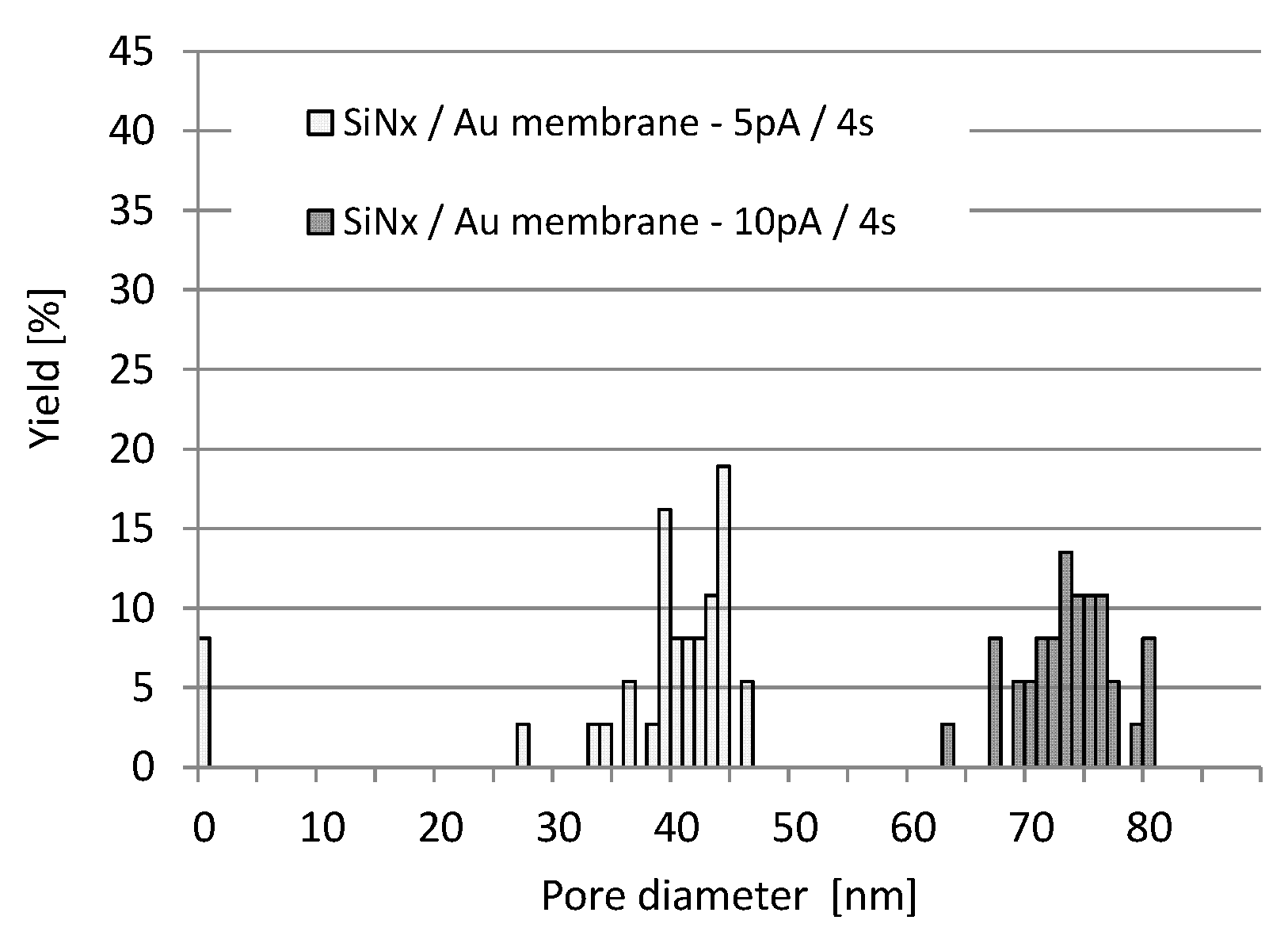
3.1. Analysis and Control of Pore Diameter Evolution
3.2. FIB Process Development
3.3. Pore size Dependent Sensitivity of Molecule Detection
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Napoli, M.; Eijkel, J.C.T.; Pennathur, S. Nanofluidic technology for biomolecule applications: A critical review. Lab Chip 2010, 20, 957–985. [Google Scholar] [CrossRef]
- Sackmann, E.K.; Fulton, A.L.; Beebe, D.J. The present and future role of microfluidics in biomedical research. Nature 2014, 507, 181–189. [Google Scholar] [CrossRef]
- Tagliazucchi, M.; Szleifer, I. Transport mechanisms in nanopores and nanochannels: Can we mimic nature? Mater. Today 2015, 18, 131–142. [Google Scholar] [CrossRef]
- Gyurcsányi, R.E. Chemically-modified nanopores for sensing. Trends Anal. Chem. 2008, 27, 627–639. [Google Scholar] [CrossRef]
- Jiang, Y.; Guo, W. Nanopore-based sensing and analysis: Beyond the resistive-pulse method. Sci. Bull. 2015, 60, 491–502. [Google Scholar] [CrossRef][Green Version]
- Lin, Y.; Ying, Y.L.; Long, Y.T. Nanopore confinement for electrochemical sensing at the single-molecule level. Curr. Opin. Electrochem. 2018, 7, 172–178. [Google Scholar] [CrossRef]
- Bayley, H.; Cremer, P.S. Stochastic sensors inspired by biology. Nature 2001, 413, 226–230. [Google Scholar] [CrossRef] [PubMed]
- Oxford Nanopore Technologies. Available online: https://www.nanoporetech.com/ (accessed on 30 September 2019).
- Schaffer, A. 10 Breakthrough Technologies–Nanopore Sequencing. MIT Technology Review. 2012. Available online: http://www2.technologyreview.com/news/427677/nanopore-sequencing/ (accessed on 30 September 2019).
- Terejánszky, P.; Makra, I.; Fürjes, P.; Gyurcsányi, R.E. Calibration-Less Sizing and Quantitation of Polymeric Nanoparticles and Viruses with Quartz Nanopipets. Anal. Chem. 2014, 86, 4688–4697. [Google Scholar] [CrossRef]
- Makra, I.; Gyurcsányi, R.E. Electrochemical sensing with nanopores: A mini review. Electrochem. Commun. 2014, 43, 55–59. [Google Scholar] [CrossRef]
- Makra, I.; Brajnovits, A.; Jágerszki, G.; Fürjes, P.; Gyurcsanyi, R.E. Potentiometric sensing of nucleic acids using chemically modified nanopores. Nanoscale 2017, 9, 739–747. [Google Scholar] [CrossRef]
- Lautner, G.; Plesz, M.; Jágerszki, G.; Fürjes, P.; Gyurcsányi, R.E. Nanoparticle displacement assay with electrochemical nanopore-based sensors. Electrochem. Commun. 2016, 71, 13–17. [Google Scholar] [CrossRef][Green Version]
- Lepoitevin, M.; Ma, T.; Bechelany, M.; Janot, J.-M.; Balme, S. Balme, Functionalization of single solid state nanopores to mimic biological ion channels: A review. Adv. Colloid Interface Sci. 2017, 250, 195–213. [Google Scholar] [CrossRef]
- Haque, F.; Li, J.; Wu, H.-C.; Liang, X.-J.; Guo, P. Solid-state and biological nanopore for real-time sensing of single chemical and sequencing of DNA. Nano Today 2013, 8, 56–74. [Google Scholar] [CrossRef] [PubMed]
- Feng, Y.; Zhang, Y.; Ying, C.; Wang, D.; Du, C. Nanopore-based Fourth-generation DNA Sequencing Technology. Genom. Proteom. Bioinform. 2015, 13, 4–16. [Google Scholar] [CrossRef] [PubMed]
- Dekker, C. Solid State Nanopores. Nat. Nanotechnol. 2007, 2, 209–215. [Google Scholar] [CrossRef]
- Wu, D.; Bi, S.; Zhang, L.; Yang, J. Single-Molecule Study of Proteins by Biological Nanopore Sensors. Sensors 2014, 14, 18211–18222. [Google Scholar] [CrossRef]
- Zhao, Q.; Wang, D.; Jayawardhana, A.; Guan, X. Stochastic sensing of biomolecules in a nanopore sensor array. Nanotechnology 2008, 19, 505504. [Google Scholar] [CrossRef]
- Wang, Y.; Zheng, D.; Tan, Q.; Wang, M.X.; Gu, L.-Q. Nanopore-based detection of circulating microRNAs in lung cancer patients. Nat. Nanotechnol. 2011, 6, 668–674. [Google Scholar] [CrossRef]
- Wanunu, M. Nanopores: A journey towards DNA sequencing. Phys. Life Rev. 2012, 9, 125–158. [Google Scholar] [CrossRef]
- Bayley, H. Nanopore Sequencing: From Imagination to Reality. Clin. Chem. 2015, 61, 25–31. [Google Scholar] [CrossRef]
- Manrao, E.A.; Derrington, I.M.; Laszlo, A.H.; Langford, K.W.; Hopper, M.K.; Gillgren, N.; Pavlenok, M.; Niederweis, M.; Gundlach, J.H. Reading DNA at single-nucleotide resolution with a mutant MspA nanopore and phi29 DNA polymerase. Nat. Biotechnol. 2012, 30, 349–353. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Stein, D.; McMullan, C.; Branton, S.; Aziz, M.J.; Golovchenko, J.A. Ion-beam sculpting at nanometer length scales. Nature 2001, 412, 166–169. [Google Scholar] [CrossRef] [PubMed]
- Miles, B.N.; Ivanov, A.P.; Wilson, K.A.; Dogan, F.; Japrung, D.; Edel, J.B. Single molecule sensing with solid-state nanopores: Novel materials, methods, and applications. Chem. Soc. Rev. 2013, 42, 15–28. [Google Scholar] [CrossRef] [PubMed]
- Venkatesan, B.M.; Bashir, R. Nanopore sensors for nucleic acid analysis. Nat. Nanotechnol. 2011, 6, 615–624. [Google Scholar] [CrossRef]
- Li, J.; Yu, D.; Zhao, Q. Solid-state nanopore-based DNA single molecule detection and sequencing. Microchim. Acta 2016, 183, 941–953. [Google Scholar] [CrossRef]
- Wei, R.; Gatterdam, V.; Wieneke, R.; Tampe, R.; Rant, U. Stochastic sensing of proteins with receptor-modified solid-state nanopores. Nat. Nanotechnol. 2012, 7, 257–263. [Google Scholar] [CrossRef]
- Albrecht, T. Progress in single-biomolecule analysis with solid-state nanopores. Curr. Opin. Electrochem. 2017, 4, 159–165. [Google Scholar] [CrossRef]
- Tsutsui, M.; Hongo, S.; He, Y.; Taniguchi, M.; Gemma, N.; Kawai, T. Single-Nanoparticle Detection Using a Low-Aspect-Ratio Pore. ACS Nano 2012, 6, 3499–3505. [Google Scholar] [CrossRef]
- Yang, J.; Ferranti, D.C.; A Stern, L.; A Sanford, C.; Huang, J.; Ren, Z.; Qin, L.-C.; Hall, A.R. Rapid and precise scanning helium ion microscope milling of solid-state nanopores for biomolecule detection. Nanotechnology 2011, 22, 285310. [Google Scholar] [CrossRef]
- Yamazaki, H.; Mizuguchi, T.; Esashika, K.; Saiki, T. Electro-osmotic trapping and compression of single DNA molecules while passing through a nanopore. Analyst 2019, 144, 5381–5388. [Google Scholar] [CrossRef]
- Coglitore, D.; Merenda, A.; Giamblanco, N.; Dumée, L.F.; Janot, J.-M.; Balme, S. Metal alloy solid-state nanopores for single nanoparticle detection. Phys. Chem. Chem. Phys. 2018, 20, 12799–12807. [Google Scholar] [CrossRef] [PubMed]
- Duan, C.; Wang, W.; Xie, Q. Review article: Fabrication of nanofluidic devices. Biomicrofluidics 2013, 7, 26501. [Google Scholar] [CrossRef] [PubMed]
- Deng, T.; Li, M.; Wang, Y.; Liu, Z. Development of solid-state nanopore fabrication technologies. Sci. Bull. 2015, 60, 304–319. [Google Scholar] [CrossRef]
- Stoloff, D.H.; Wanunu, M. Recent trends in nanopores for biotechnology. Curr. Opin. Biotechnol. 2013, 14, 699–704. [Google Scholar] [CrossRef]
- Wei, R.; Pedone, D.; Zürner, A.; Döblinger, M.; Rant, U. Fabrication of Metallized Nanopores in Silicon Nitride Membranes for Single-Molecule Sensing. Small 2010, 13, 1406–1414. [Google Scholar] [CrossRef]
- Xue, T.; Xu, Q.; Li, Y.-X.; Qi, H.-Y.; Wang, Z.-B.; Yang, Y.; Ren, T.-L. A method of controlling the hole size of nanopores array on anodic aluminum oxide. Mater. Lett. 2018, 215, 183–186. [Google Scholar] [CrossRef]
- Venkatesan, B.M.; Shah, A.B.; Zuo, J.M.; Bashir, R. DNA Sensing Using Nanocrystalline Surface-Enhanced Al2O3 Nanopore Sensors. Adv. Funct. Mater. 2010, 20, 1266–1275. [Google Scholar] [CrossRef]
- Nilsson, J.; Lee, J.R.I.; Ratto, T.V.; Létant, S.E. Localized Functionalization of Single Nanopores. Adv. Mater. 2006, 18, 427–431. [Google Scholar] [CrossRef]
- Danelon, C.; Santschi, C.; Brugger, J.; Vogel, H. Fabrication and Functionalization of Nanochannels by Electron-Beam-Induced Silicon Oxide Deposition. Langmuir 2006, 22, 10711–10715. [Google Scholar] [CrossRef]
- Kwok, H.; Briggs, K.; Tabard-Cossa, V. Nanopore Fabrication by Controlled Dielectric Breakdown. PLoS ONE 2014, 9, e92880. [Google Scholar] [CrossRef]
- Yanagi, I.; Akahori, R.; Hatano, T.; Takeda, K.-I. Fabricating nanopores with diameters of sub-1 nm to 3 nm using multilevel pulse-voltage injection. Sci. Rep. 2014, 4, 5000. [Google Scholar] [CrossRef]
- Zhang, Y.; Miyahara, Y.; Derriche, N.; Yang, W.; Yazda, K.; Capaldi, X.; Liu, Z.; Grutter, P.; Reisner, W. Nanopore Formation via Tip-Controlled Local Breakdown Using an Atomic Force Microscope. Small Methods 2019, 3, 1900147. [Google Scholar] [CrossRef]
- Ying, C.; Houghtaling, J.; Eggenberger, O.M.; Guha, A.; Nirmalraj, P.; Awasthi, S.; Tian, J.; Mayer, M. Formation of Single Nanopores with Diameters of 20-50 Nanometers in Silicon Nitride Membranes Using Laser-Assisted Controlled Breakdown. ACS Nano 2018, 12, 11458–11470. [Google Scholar] [CrossRef]
- Yamazaki, H.; Hu, R.; Zhao, Q.; Wanunu, M. Photothermally Assisted Thinning of Silicon Nitride, Membranes for Ultrathin Asymmetric Nanopores. ACS Nano 2018, 12, 12472–12481. [Google Scholar] [CrossRef]
- Gilboa, T.; Zrehen, A.; Girsault, A.; Meller, A. Optically-Monitored Nanopore Fabrication Using a Focused Laser Beam. Sci. Rep. 2018, 8, 9765. [Google Scholar] [CrossRef]
- Venkatesan, B.M.; Dorvel, B.; Yemenicioglu, S.; Watkins, N.; Petrov, I.; Bashir, R. Highly Sensitive, Mechanically Stable Nanopore Sensors for DNA Analysis. Adv. Materials 2009, 21, 2771–2776. [Google Scholar] [CrossRef]
- Fürjes, P. Precisely tailored solid state nanopores for molecule recognition. In Proceedings of the Design, Test, Integration and Packaging of MEMS/MOEMS–DTIP 2016 Conference, Budapest, Hungary, 30 May–2 June 2016. IEEE XPlore. [Google Scholar] [CrossRef]
- Tseng, A.A. Recent Developments in Nanofabrication Using Focused Ion Beams. Small 2005, 1, 924–939. [Google Scholar] [CrossRef]
- Sawafta, F.; Carlsen, A.T.; Hall, A.R. Membrane Thickness Dependence of Nanopore Formation with a Focused Helium Ion Beam. Sensors 2014, 14, 8150–8161. [Google Scholar] [CrossRef]
- Liu, S.; Yuzvinsky, T.D.; Schmidt, H. Effect of Fabrication-Dependent Shape and Composition of Solid-State Nanopores on Single Nanoparticle Detection. ACS Nano 2013, 7, 5621–5627. [Google Scholar] [CrossRef]
- Varga, M.; Berczes, Z.; Illés, L.; Safrany, G.; Barsony, I.; Fürjes, P.; Gyurcsányi, R.E.; Jágerszki, G. Fluidically and electrically integrated solid state nanopore arrays for biochemical sensing. In Proceedings of the IEEE SENSORS, Valencia, Spain, 2–5 November 2014; pp. 870–872. [Google Scholar]
- NHS-SS-Biotin. Available online: http://www.apexbt.com/nhs-ss-biotin.html (accessed on 30 September 2019).
- Gyurcsányi, R.E.; Vigassy, T.; Pretsch, E. Biorecognition-modulated ion fluxes through functionalized gold nanotubules as a novel label-free biosensing approach. Chem. Commun. 2003, 20, 2560–2561. [Google Scholar] [CrossRef]
- Gamry Instruments. Available online: https://www.gamry.com/ (accessed on 30 September 2019).
- Vitarelli, M.J., Jr.; Prakash, S.; David, S. Talaga, determining Nanocapillary Geometry from Electrochemical Impedance Spectroscopy Using a Variable Topology Network Circuit Model. Anal. Chem. 2011, 83, 533–541. [Google Scholar] [CrossRef]
- Rollings, R.; Graef, E.; Walsh, N.; Nandivada, S.; Benamara, M.; Li, J. The effects of geometry and stability of solid-state nanopores on detecting single DNA molecules. Nanotechnology 2015, 26, 044001. [Google Scholar] [CrossRef]
- Anzai, J.; Hoshi, T.; Osa, T. Avidin—Biotin Mediated Biosensors. In Biosensors and Their Applications; Yang, V.C., Ngo, T.T., Eds.; Springer: Berlin/Heidelberg, Germany, 2000; ISBN 978-1-4613-6875-5. [Google Scholar] [CrossRef]
- Johannssen, W.; Attwood, P.V.; Wallace, J.C.; Keech, D.B. Localisation of the Active Site of Pyruvate Carboxylase by Electron Microscopic Examination of Avidin-Enzyme Complexes. Eur. J. Biochem. 1983, 133, 201–206. [Google Scholar] [CrossRef] [PubMed]
- Pugliese, L.; Coda, A.; Malcovati, M.; Bolognesi, M. Three-dimensional Structure of the Tetragonal Crystal Form of Egg-white Avidin in its functional Complex with Biotin at 2·7 Å Resolution. J. Mol. Biol. 1993, 231, 698–710. [Google Scholar] [CrossRef] [PubMed]
- Rauscher, M.; Dietrich, S. Wetting Phenomena in Nanofluidics. Annu. Rev. Mater. Res. 2008, 38, 143–172. [Google Scholar] [CrossRef]
- Zhang, Y.; Liu, L.; Sha, J.; Ni, Z.; Yi, H.; Chen, Y. Nanopore detection of DNA molecules in magnesium chloride solutions. Nanoscale Res. Lett. 2013, 8, 245. [Google Scholar] [CrossRef]
- Acar, E.T.; Buchsbaum, S.F.; Combs, C.; Fornasiero, F.; Siwy, Z.S. Biomimetic potassium-selective nanopores. Sci. Adv. 2019, 5, eaav2568. [Google Scholar] [CrossRef]
- Farimani, A.B.; Heiranian, M.; Aluru, N.R. Identification of amino acids with sensitive nanoporous MoS2: Towards machine learning-based prediction. npj 2D Mater. Appl. 2018, 2, 2–14. [Google Scholar] [CrossRef]
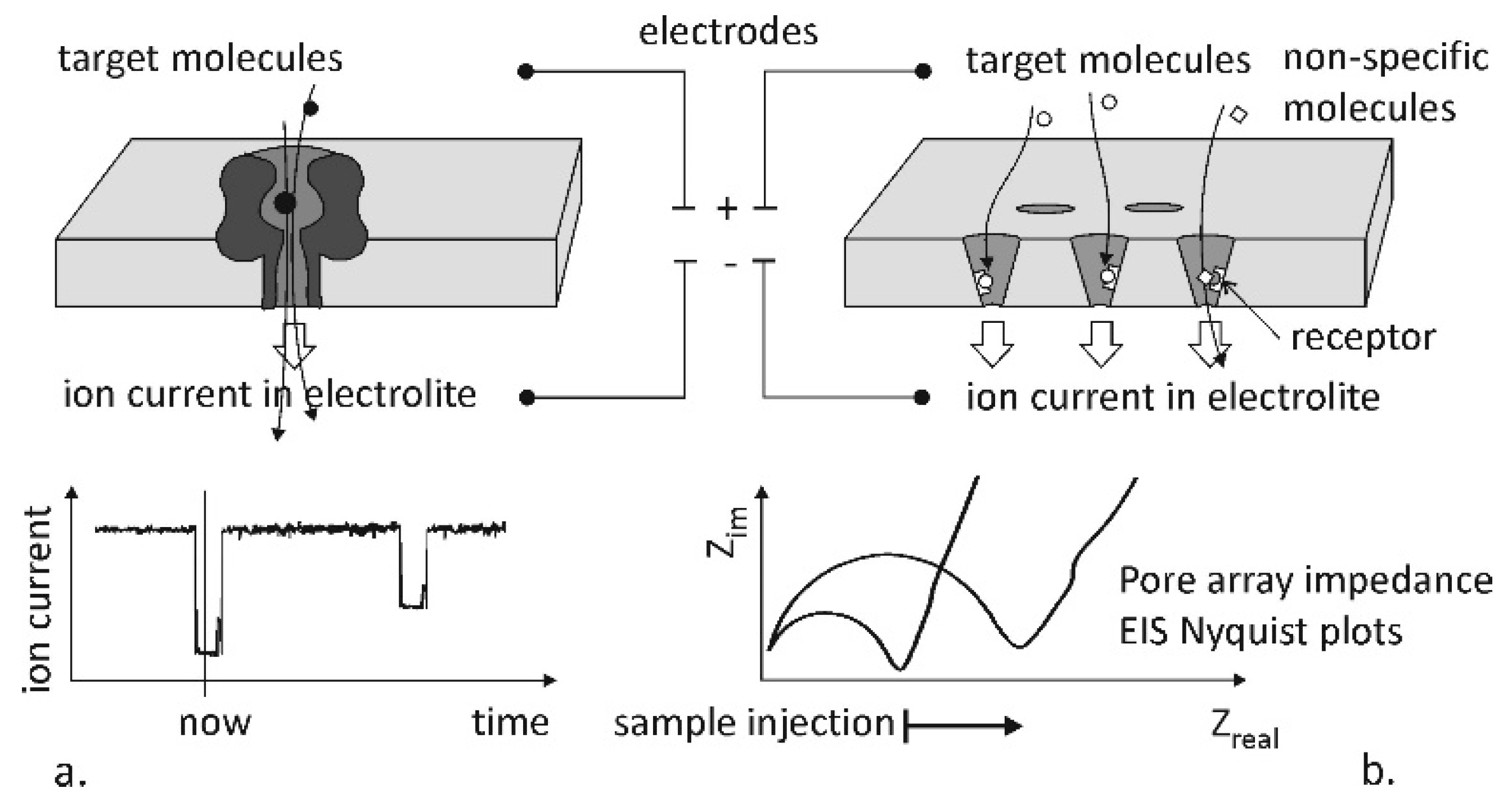
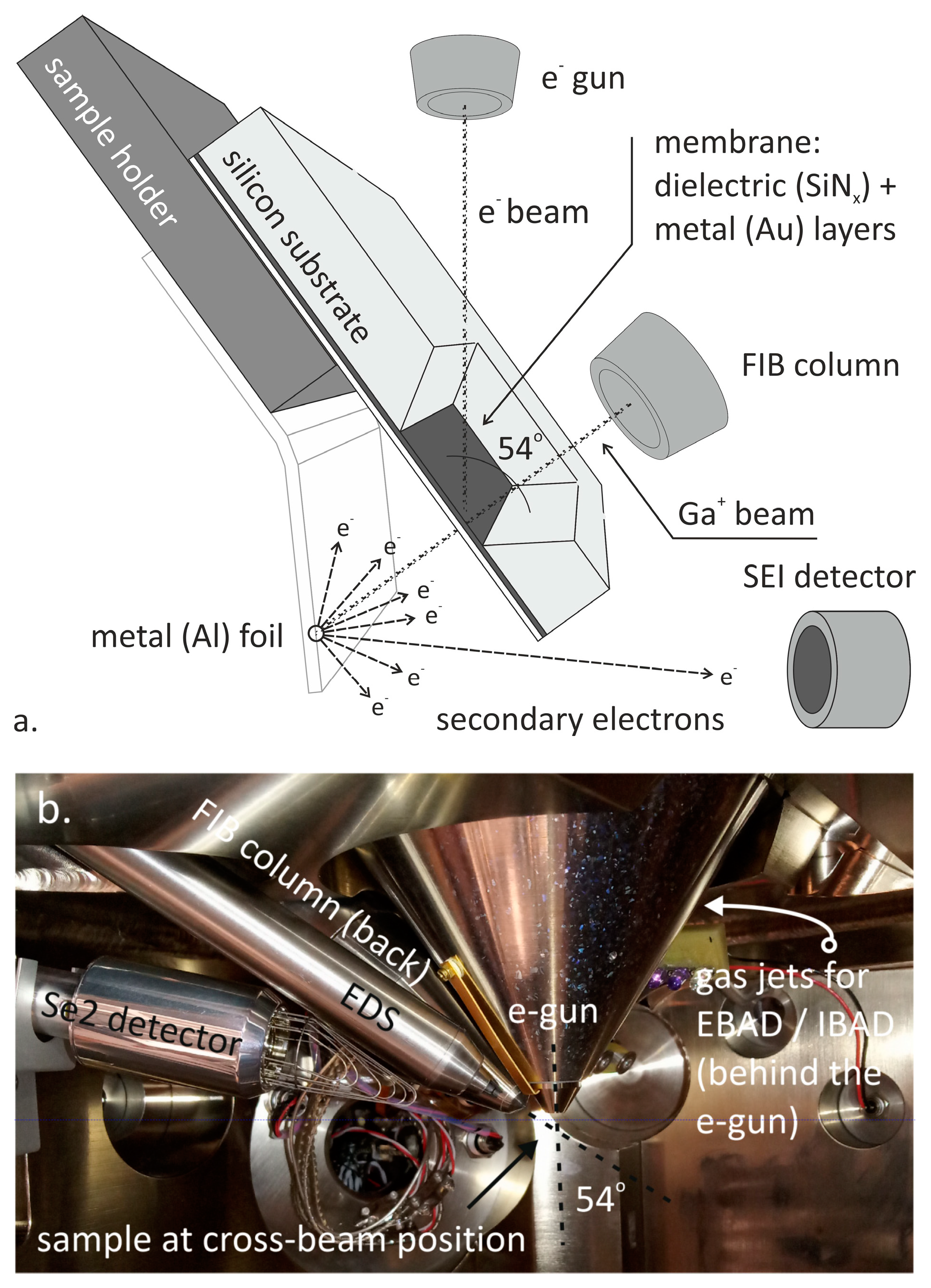

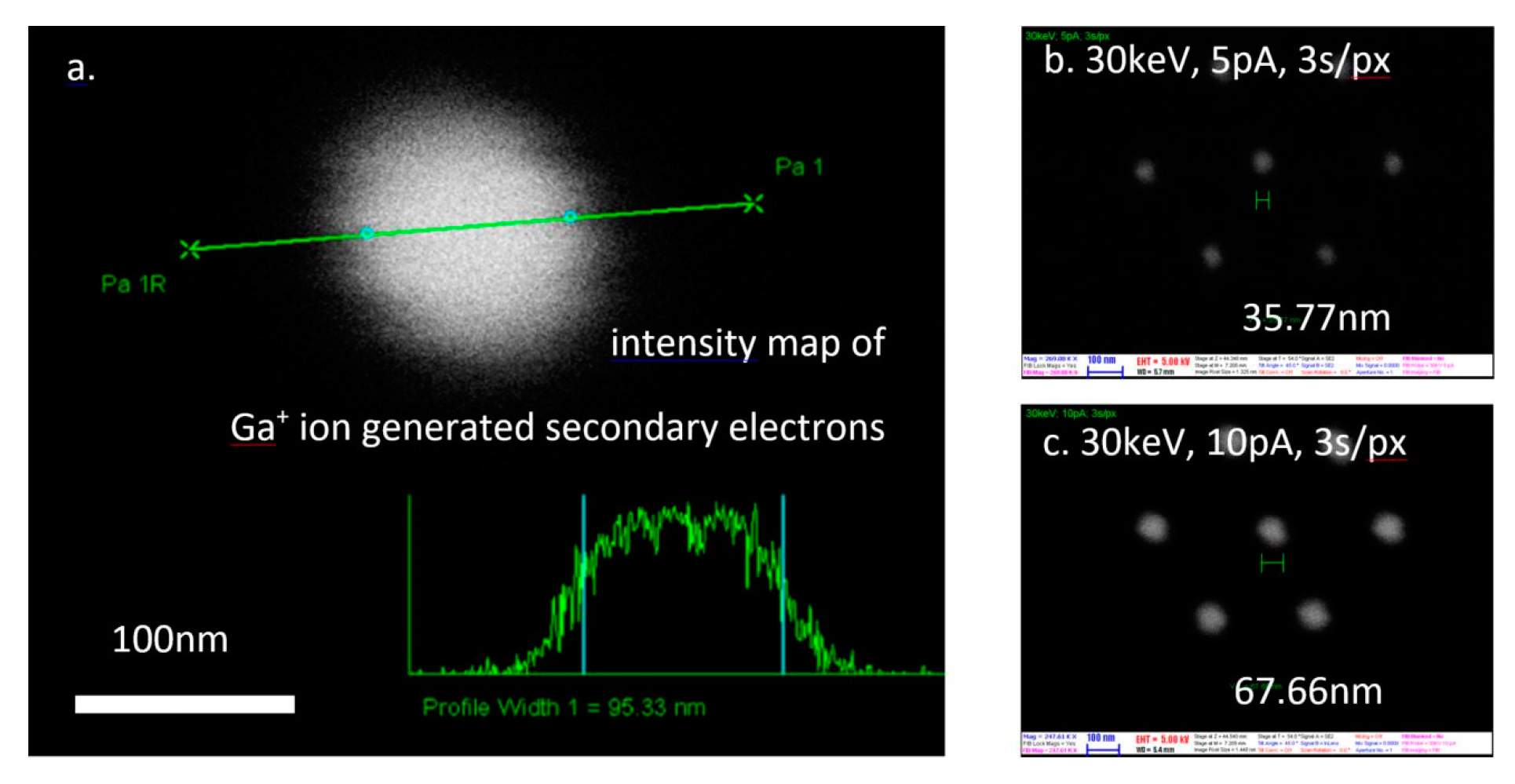
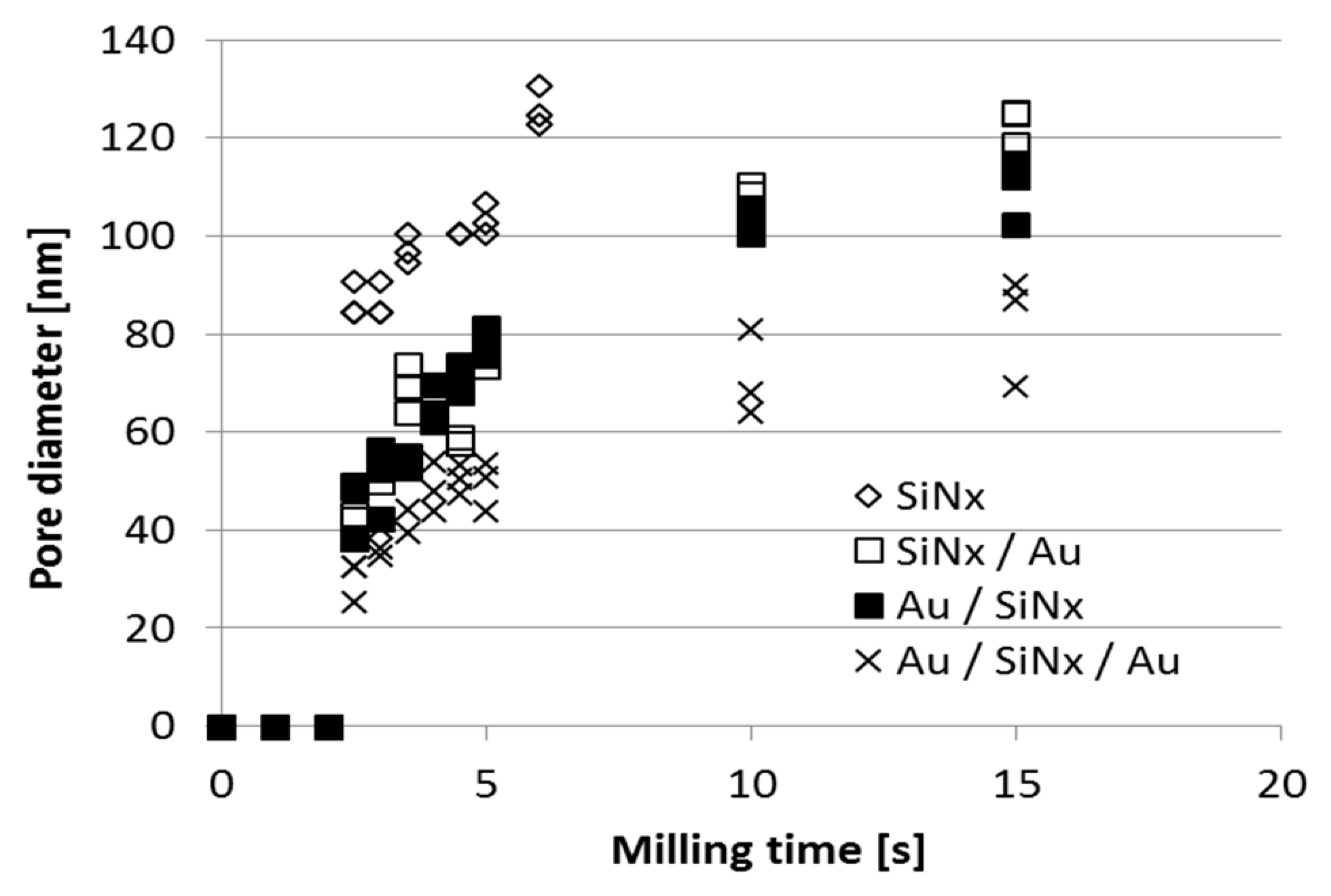


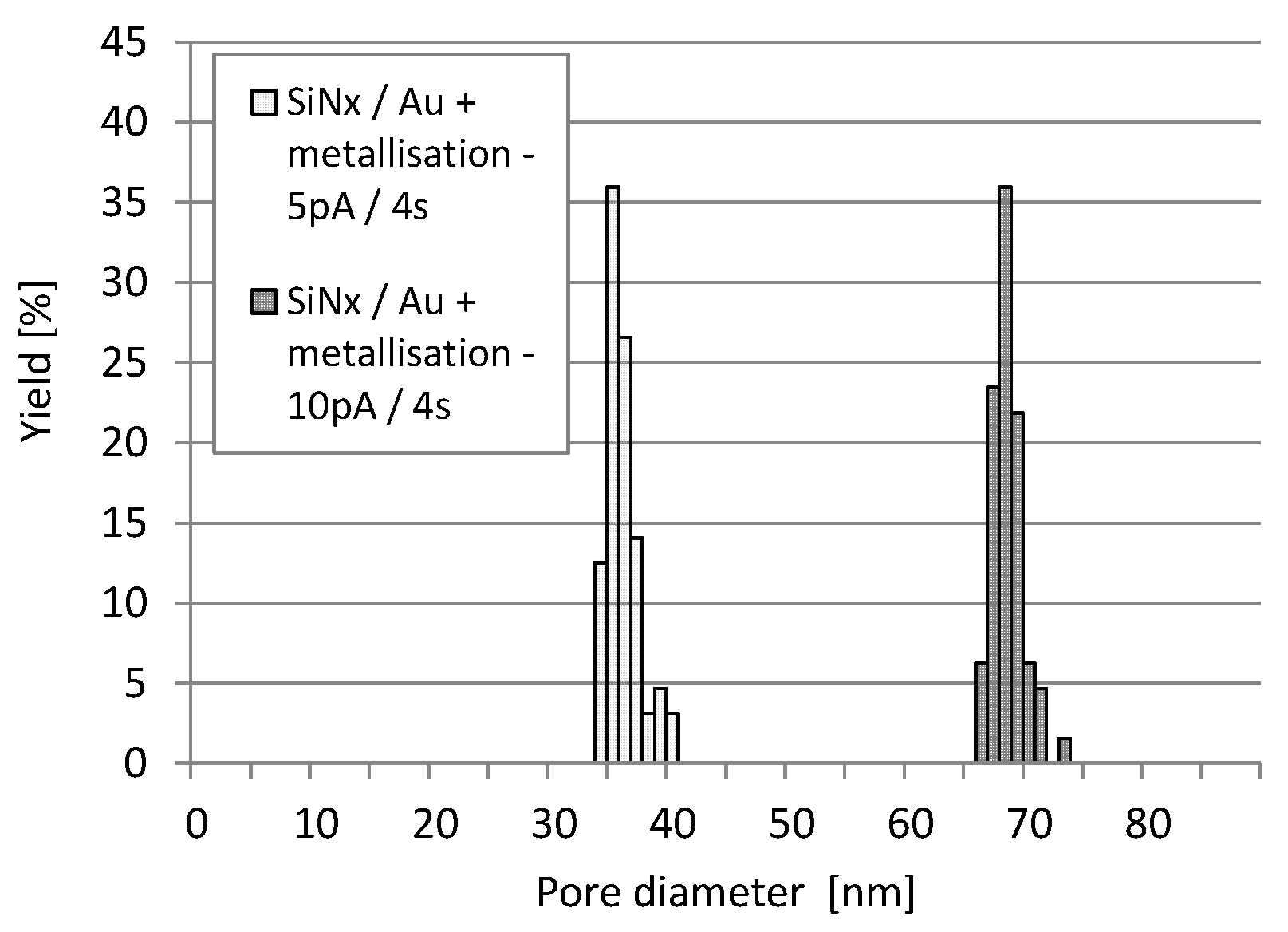
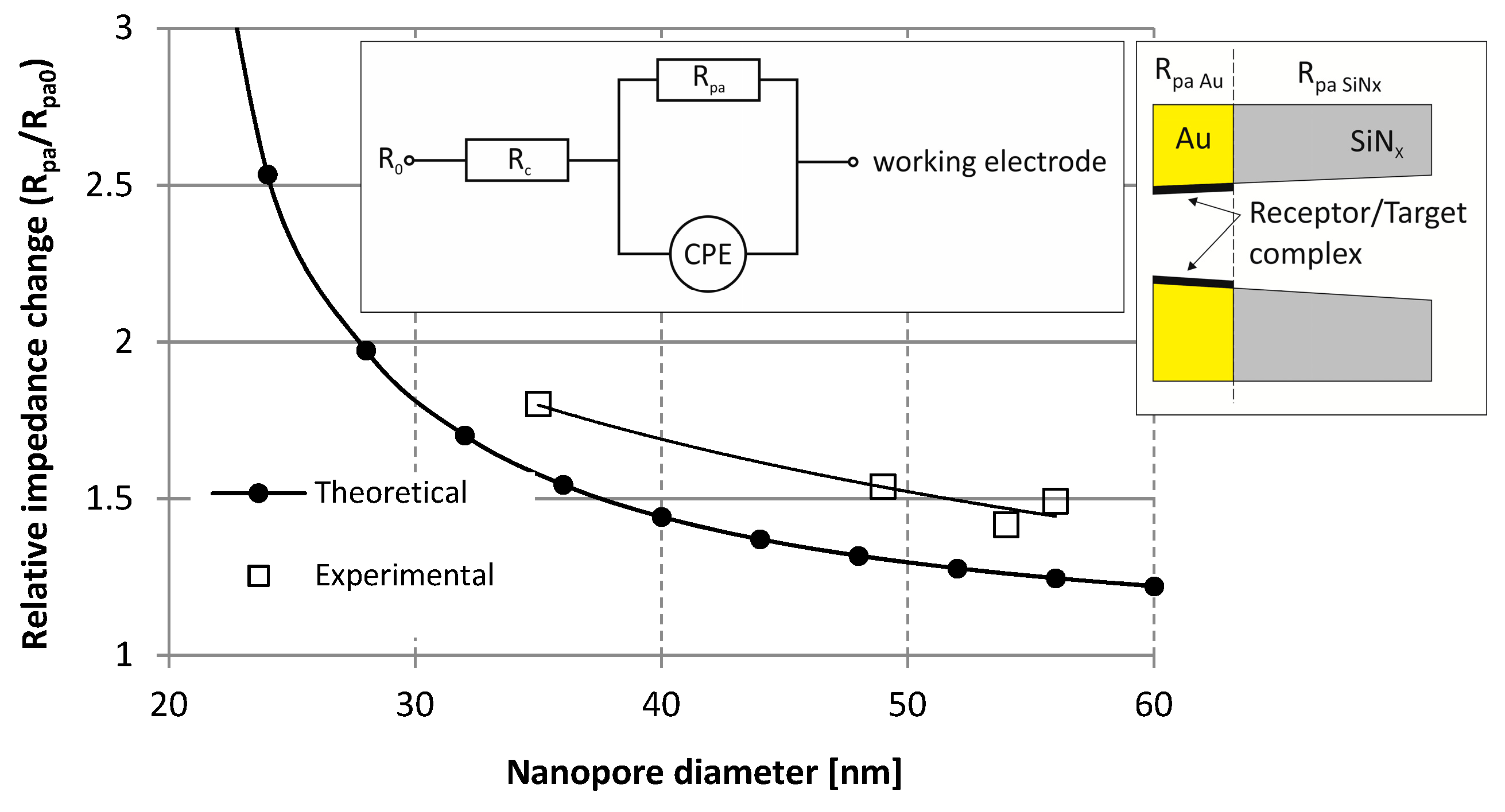
| Fabrication Technique | e-BEAM [17,34,35,37] | FIB or TEM [17,24,25,31,34,35,36] | NIL [34] | MEMS Based: Sacrificial Layer or Channel Sealing by Bonding [34] | |
|---|---|---|---|---|---|
| Geometry | Feature size | sub 10nm | >10nm or sub 10nm | >20nm | >5nm |
| Reproducibility | excellent | excellent | excellent | good | |
| Shape | channel array | pore/channel array | pore/channel array | lateral/buried channels | |
| Material | silicon or compatible | ||||
| Manufacturing | Equipment/Infrastructure | E-BEAM system + MEMS (RIE/DRIE *) | FIB/SEM or TEM system + MEMS (RIE/DRIE *) | NIL system + MEMS (RIE/DRIE *) | MEMS |
| Cost | High | High | High/Medium | Medium | |
| Process | Wafer scale | Chip/wafer scale | Wafer scale | Wafer scale | |
| Process compatibility | MEMS | ||||
| Main advantages | Advanced production rate, reproducibility | Geometrical and material flexibility | Advanced production rate | Advancedproduction rate | |
| Main drawbacks | High infrastructural demand | ||||
| Complex pre- and post-processing | Moderate production rate and reproducibility | Complex pre- and post-processing | Moderate reproducibility | ||
| Proposed Surface Functionalisation | Structural Materials |
|---|---|
| thiol chemistry | Au/SiNx/Au SiNx/Au Au/SiNx |
| silane chemistry | bare SiNx PFA/SiNx/PFA * |
| Pore outlet diameters | 20–60 nm |
| Pore length | 450 nm (300 nm in SiNx and 150 nm in Au) |
| Pore wall angle (see Figure 7) | 5° |
| Pore inlet diameters (estimated) | 68–108 nm |
| Number of nanopores/array | 64 |
| Target molecule/estimated size | Avidin/5.7 nm × 4.4 nm × 4.4 nm |
| Receptor molecule/estimated length | Biotin/2.43 nm |
| Electrolyte conductivity (0.01 M PBS) | 150–200 S/m (15–20 mS/cm, 15–20 mho/cm) |
© 2019 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fürjes, P. Controlled Focused Ion Beam Milling of Composite Solid State Nanopore Arrays for Molecule Sensing. Micromachines 2019, 10, 774. https://doi.org/10.3390/mi10110774
Fürjes P. Controlled Focused Ion Beam Milling of Composite Solid State Nanopore Arrays for Molecule Sensing. Micromachines. 2019; 10(11):774. https://doi.org/10.3390/mi10110774
Chicago/Turabian StyleFürjes, Péter. 2019. "Controlled Focused Ion Beam Milling of Composite Solid State Nanopore Arrays for Molecule Sensing" Micromachines 10, no. 11: 774. https://doi.org/10.3390/mi10110774
APA StyleFürjes, P. (2019). Controlled Focused Ion Beam Milling of Composite Solid State Nanopore Arrays for Molecule Sensing. Micromachines, 10(11), 774. https://doi.org/10.3390/mi10110774





