Nanomaterials-Based Electrochemical Immunosensors
Abstract
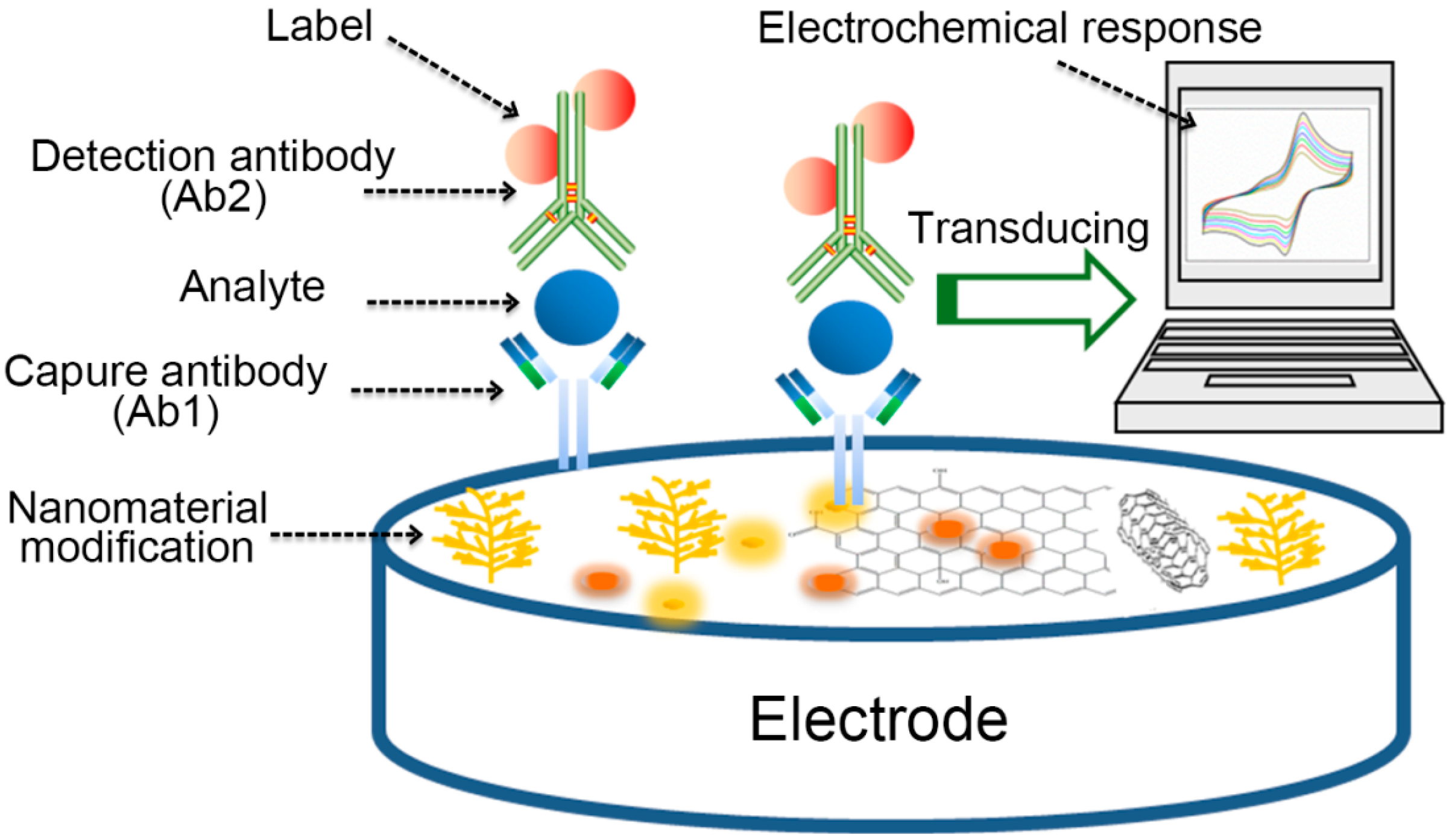
1. Introduction
2. Nanomaterials Based Electrochemical Immunosensor
2.1. Electrochemical Immunosensor Based on Carbon Nanomaterials
2.2. Electrochemical Immunosensor Based on Quantum Dots
3. Conclusions and Outlook
Author Contributions
Funding
Conflicts of Interest
References
- Keçili, R.; Büyüktiryaki, S.; Hussain, C.M. Advancement in bioanalytical science through nanotechnology: Past, present and future. TrAC Trends Anal. Chem. 2019, 110, 259–276. [Google Scholar] [CrossRef]
- Genet, C.; Errabi, K.; Gauthier, C. Which model of technology transfer for nanotechnology? A comparison with biotech and microelectronics. Technovation 2012, 32, 205–215. [Google Scholar] [CrossRef]
- Wang, L.; Gong, C.; Yuan, X.; Wei, G. Controlling the Self-Assembly of Biomolecules into Functional Nanomaterials through Internal Interactions and External Stimulations: A Review. Nanomaterial 2019, 9, 285. [Google Scholar] [CrossRef] [PubMed]
- Kaushik, N.K.; Kaushik, N.; Linh, N.N.; Ghimire, B.; Pengkit, A.; Sornsakdanuphap, J.; Lee, S.-J.; Choi, E.H. Plasma and Nanomaterials: Fabrication and Biomedical Applications. Nanomaterial 2019, 9, 98. [Google Scholar] [CrossRef] [PubMed]
- Mohajeri, M.; Behnam, B.; Sahebkar, A. Biomedical applications of carbon nanomaterials: Drug and gene delivery potentials. J. Cell. Physiol. 2019, 234, 298–319. [Google Scholar] [CrossRef]
- Vaithiyanathan, M.; Bajgiran, K.R.; Darapaneni, P.; Safa, N.; Dorman, J.A.; Melvin, A.T. Luminescent nanomaterials for droplet tracking in a microfluidic trapping array. Anal. Bioanal. Chem. 2019, 411, 157–170. [Google Scholar] [CrossRef]
- Sethi, M.; Pacardo, D.B.; Knecht, M.R. Biological Surface Effects of Metallic Nanomaterials for Applications in Assembly and Catalysis. Langmuir 2010, 26, 15121–15134. [Google Scholar] [CrossRef]
- Yao, Y.; Chen, S. Surface effect on resonant properties of nanowires predicted by an elastic theory for nanomaterials. J. Appl. Phys. 2015, 118, 044303. [Google Scholar] [CrossRef]
- Sun, J.; Liu, L.; Zhao, X.; Yang, S.; Komarneni, S.; Yang, D. Capture of radioactive cations from water using niobate nanomaterials with layered and tunnel structures. RSC Adv. 2015, 5, 75354–75359. [Google Scholar] [CrossRef]
- Sumpter, B.G.; Garashchuk, S.; Jakowski, J.; Wang, L. Quantum Trajectory-Electronic Structure Approach for Exploring Nuclear Effects in the Dynamics of Nanomaterials. J. Chem. Theory Comput. 2013, 9, 5221–5235. [Google Scholar]
- Li, M.; Wang, Z.; Zhang, R.; Soon, A.; Zhang, R. Size and dimension effect on volume plasmon energy of nanomaterials. Solid State Commun. 2012, 152, 1564–1566. [Google Scholar] [CrossRef]
- Batsanov, S.S.; Poyarkov, K.B.; Gavrilkin, S.M. The effect of the atomic structure on dielectric properties of nanomaterials. Dokl. Phys. 2009, 54, 407–409. [Google Scholar] [CrossRef]
- Aziz, A.; Ahmed, E.; Ali, I.; Athar, M.; Ehsan, M.F.; Ashiq, M.N. Effect of Gd and Cu on the Structural, Electrical, and Dielectric Properties of MnFeO3 Nanomaterials Synthesized by the Sol–Gel Method. J. Electron. Mater. 2015, 44, 4300–4307. [Google Scholar] [CrossRef]
- Lee, A.-Y.; Ra, H.-S.; Kwak, D.-H.; Jeong, M.-H.; Park, J.-H.; Kang, Y.-S.; Chae, W.-S.; Lee, J.-S.; Min-Hye, J. Hybrid Black Phosphorus/Zero-Dimensional Quantum Dot Phototransistors: Tunable Photodoping and Enhanced Photoresponsivity. ACS Appl. Mater. Interfaces 2018, 10, 16033–16040. [Google Scholar] [CrossRef] [PubMed]
- Zeng, Z.; Phan, H.; Chen, S.; Yu, Z.; Wang, R.; Nguyen, T.-Q.T.; Tan, T.T.Y.; Xiao, F. Unraveling the cooperative synergy of zero-dimensional graphene quantum dots and metal nanocrystals enabled by layer-by-layer assembly. J. Mater. Chem. A 2018, 6, 1700–1713. [Google Scholar] [CrossRef]
- Yu, L.; Ruan, S.; Xu, X.; Zou, R.; Hu, J. One-dimensional nanomaterial-assembled macroscopic membranes for water treatment. Nano Today 2017, 17, 79–95. [Google Scholar] [CrossRef]
- Wang, Z.L. Splendid One-Dimensional Nanostructures of Zinc Oxide: A New Nanomaterial Family for Nanotechnology. ACS Nano 2008, 2, 1987–1992. [Google Scholar] [CrossRef] [PubMed]
- Zhu, C.; Fang, Y.; Wen, D.; Dong, S. One-pot synthesis of functional two-dimensional graphene/SnO2 composite nanosheets as a building block for self-assembly and an enhancing nanomaterial for biosensing. J. Mater. Chem. 2011, 21, 16911. [Google Scholar] [CrossRef]
- Maurya, J.B.; François, A.; Prajapati, Y.K. Two-Dimensional Layered Nanomaterial-Based One-Dimensional Photonic Crystal Refractive Index Sensor. Sensors 2018, 18, 857. [Google Scholar] [CrossRef]
- Wang, S.; Ye, B.Y.; An, C.W.; Wang, J.Y.; Li, Q.B. Synergistic effects between Cu metal-organic framework (Cu-MOF) and carbon nanomaterials for the catalyzation of the thermal decomposition of ammonium perchlorate (AP). J. Mater. Sci. 2019, 54, 4928–4941. [Google Scholar] [CrossRef]
- Xiang, L.; Zhao, C.; Wang, J. Nanomaterials-Based Electrochemical Sensors and Biosensors for Pesticide Detection. Sens. Lett. 2011, 9, 1184–1189. [Google Scholar] [CrossRef]
- Holland-Moritz, H.; Scheeler, S.; Stanglmair, C.; Pacholski, C.; Ronning, C. Enhanced sputter yields of ion irradiated Au nano particles: Energy and size dependence. Nanotechnology 2015, 26, 325301. [Google Scholar] [CrossRef] [PubMed]
- Prakash, A.; Pathrose, B.P.; Nampoori, V.P.N.; Radhakrishnan, P.; Mujeeb, A. Thermal diffusivity of neutral red dye using dual beam thermal lens technique: A comparison on the effects using nano pulsed laser ablated silver and gold nanoparticles. Phys. E 2019, 107, 203–208. [Google Scholar] [CrossRef]
- Dhanunjaya, M.; A Khan, S.; Pathak, A.P.; Avasthi, D.K.; Rao, S.V.S.N. Ion induced crystallization and grain growth of hafnium oxide nano-particles in thin-films deposited by radio frequency magnetron sputtering. J. Phys. D Appl. Phys. 2017, 50, 505301. [Google Scholar] [CrossRef]
- Wang, H.; Hou, P.; Zhao, G.; Yu, L.; Gao, Y.-W.; He, H. Development and evaluation of serotype-specific recombinase polymerase amplification combined with lateral flow dipstick assays for the diagnosis of foot-and-mouth disease virus serotype A, O and Asia1. BMC Vet. Res. 2018, 14, 359. [Google Scholar] [CrossRef] [PubMed]
- Wei, Y.-H.; Hsieh, C.-K.; Tseng, F.-G. Highly-Sensitive Non-Enzymatic Glucose Sensor via Nano Platinum Crystals Fabricated by Phase-Controlled Electrochemical Deposition. J. Electrochem. Soc. 2018, 165, B48–B54. [Google Scholar] [CrossRef]
- Fuentes, S.; Zarate, R.A.; Chavez, E.; Munoz, P.; Diaz-Droguett, D.; Leyton, P. Preparation of SrTiO3 nanomaterial by a sol-gel-hydrothermal method. J. Mater. Sci. 2010, 45, 1448–1452. [Google Scholar] [CrossRef]
- Taylor, A.; Fekete, L.; Hubík, P.; Jäger, A.; Janicek, P.; Mortet, V.; Mistrík, J.; Vacik, J. Large area deposition of boron doped nano-crystalline diamond films at low temperatures using microwave plasma enhanced chemical vapour deposition with linear antenna delivery. Diam. Relat. Mater. 2014, 47, 27–34. [Google Scholar] [CrossRef]
- Chen, S.; Zhang, Y.; Han, W.; Wellburn, D.; Liang, J.; Liu, C. Synthesis and magnetic properties of Fe2O3–TiO2 nano-composite particles using pulsed laser gas phase evaporation–liquid phase collecting method. Appl. Surf. Sci. 2013, 283, 422–429. [Google Scholar] [CrossRef]
- Liu, Q.L.; Yan, X.H.; Yin, X.M.; Situ, B.; Zhou, H.K.; Lin, L.; Li, B.; Gan, N.; Zheng, L. Electrochemical Enzyme-Linked Immunosorbent Assay (ELISA) for alpha-Fetoprotein Based on Glucose Detection with Multienzyme-Nanoparticle Amplification. Molecules 2013, 18, 12675–12686. [Google Scholar] [CrossRef]
- Yang, G.; Guo, H.; Li, H.; Shan, S.; Zhang, X.; Rombout, J.H.; An, L. Molecular characterization of LEAP-2 cDNA in common carp (Cyprinus carpio L.) and the differential expression upon a Vibrio anguillarum stimulus; indications for a significant immune role in skin. Fish Shellfish Immunol. 2014, 37, 22–29. [Google Scholar] [CrossRef] [PubMed]
- Li, T.; Wang, L.; Zhang, Y.; Guo, X.; Chen, X.; Zhang, F.; Yang, G.; Wen, W.; Li, H. Molecular characterization of three novel perforins in common carp (Cyprinus carpio L.) and their expression patterns during larvae ontogeny and in response to immune challenges. BMC Vet. Res. 2018, 14, 299. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Yan, J.; Shi, J.; Sun, W.; Chen, Z.; Yu, J.; Qi, J.; Du, Y.; Zhang, H.; Feng, L. Zebrafish intelectin 1 (zITLN1) plays a role in the innate immune response. Fish Shellfish Immunol. 2018, 83, 96–103. [Google Scholar] [CrossRef] [PubMed]
- Zhou, F.; Lu, M.; Wang, W.; Bian, Z.-P.; Zhang, J.-R.; Zhu, J.-J. Electrochemical Immunosensor for Simultaneous Detection of Dual Cardiac Markers Based on a Poly(Dimethylsiloxane)-Gold Nanoparticles Composite Microfluidic Chip: A Proof of Principle. Clin. Chem. 2010, 56, 1701–1707. [Google Scholar] [CrossRef] [PubMed]
- Beitollahi, H.; Ivari, S.G.; Torkzadeh-Mahani, M. Application of antibody–nanogold–ionic liquid–carbon paste electrode for sensitive electrochemical immunoassay of thyroid-stimulating hormone. Biosens. Bioelectron. 2018, 110, 97–102. [Google Scholar] [CrossRef] [PubMed]
- Du, X.; Zhou, J. Application of biosensors to detection of epidemic diseases in animals. Res. Vet. Sci. 2018, 118, 444–448. [Google Scholar] [CrossRef] [PubMed]
- Karimi-Maleh, H. Electrochemical Sensors Based on Metal Nanoparticles, Carbon Based Nanomaterials or Ionic Liquids. Curr. Anal. Chem. 2017, 13, 4. [Google Scholar] [CrossRef]
- Zhang, L.; Wang, J.; Tian, Y. Electrochemical in-vivo sensors using nanomaterials made from carbon species, noble metals, or semiconductors. Microchim. Acta 2014, 181, 1471–1484. [Google Scholar] [CrossRef]
- Hashimoto, A.; Suenaga, K.; Gloter, A.; Urita, K.; Iijima, S. Direct evidence for atomic defects in graphene layers. Nature 2004, 430, 870–873. [Google Scholar] [CrossRef]
- Zhu, Z.; Su, D.; Weinberg, G.; Schlögl, R. Supermolecular Self-Assembly of Graphene Sheets: Formation of Tube-in-Tube Nanostructures. Nano Lett. 2004, 4, 2255–2259. [Google Scholar] [CrossRef]
- Khalil, I.; Julkapli, N.M.; Yehye, W.A.; Basirun, W.J.; Bhargava, S.K. Graphene-Gold Nanoparticles Hybrid-Synthesis, Functionalization, and Application in a Electrochemical and Surface-Enhanced Raman Scattering Biosensor. Materials 2016, 9, 406. [Google Scholar] [CrossRef] [PubMed]
- Tran, H.V.; Piro, B.; Reißberg, S.; Duc, H.T.; Pham, M. Antibodies Directed to RNA/DNA Hybrids: An Electrochemical Immunosensor for MicroRNAs Detection using Graphene-Composite Electrodes. Anal. Chem. 2013, 85, 8469–8474. [Google Scholar] [CrossRef] [PubMed]
- Tran, H.V.; Piro, B.; Reißberg, S.; Nguyen, L.H.; Nguyen, T.D.; Duc, H.; Pham, M. An electrochemical ELISA-like immunosensor for miRNAs detection based on screen-printed gold electrodes modified with reduced graphene oxide and carbon nanotubes. Biosens. Bioelectron. 2014, 62, 25–30. [Google Scholar] [CrossRef] [PubMed]
- Yang, Z.; Lan, Q.; Li, J.; Wu, J.; Tang, Y.; Hu, X. Efficient streptavidin-functionalized nitrogen-doped graphene for the development of highly sensitive electrochemical immunosensor. Biosens. Bioelectron. 2017, 89, 312–318. [Google Scholar] [CrossRef] [PubMed]
- Khetani, S.; Ozhukil Kollath, V.; Kundra, V.; Nguyen, M.D.; Debert, C.; Sen, A.; Karan, K.; Sanati-Nezhad, A. Polyethylenimine Modified Graphene-Oxide Electrochemical Immunosensor for the Detection of Glial Fibrillary Acidic Protein in Central Nervous System Injury. ACS Sens. 2018, 3, 844–851. [Google Scholar] [CrossRef] [PubMed]
- AfzaliTabar, M.; Alaei, M.; Khojasteh, R.R.; Motiee, F.; Rashidi, A.M. Preference of multi-walled carbon nanotube (MWCNT) to single-walled carbon nanotube (SWCNT) and activated carbon for preparing silica nanohybrid pickering emulsion for chemical enhanced oil recovery (C-EOR). J. Solid State Chem. 2017, 245, 164–173. [Google Scholar] [CrossRef]
- Wei, X.P.; Luo, Y.L.; Xu, F.; Chen, Y.S.; Yang, L.H. In-situ non-covalent dressing of multi-walled carbon nanotubes@titanium dioxides with carboxymethyl chitosan nanocomposite electrochemical sensors for detection of pesticide residues. Mater. Des. 2016, 111, 445–452. [Google Scholar] [CrossRef]
- Ruchika, M.; Vyomesh, P.; Jose, P.V.; Gutkind, J.S.; Rusling, J.F. Ultrasensitive Electrochemical Immunosensor for Oral Cancer Biomarker IL-6 Using Carbon Nanotube Forest Electrodes and Multilabel Amplification. Anal. Chem. 2010, 82, 3118–3123. [Google Scholar]
- Singh, V.; Krishnan, S. Voltammetric immunosensor assembled on carbon-pyrenyl nanostructures for clinical diagnosis of type of diabetes. Anal. Chem. 2015, 87, 2648–2654. [Google Scholar] [CrossRef]
- Zhang, H.; Han, Z.; Wang, X.; Li, F.; Cui, H.; Yang, D.; Bian, Z. Sensitive immunosensor for N-terminal pro-brain natriuretic peptide based on N-(aminobutyl)-N-(ethylisoluminol)-functionalized gold nanodots/multiwalled carbon nanotube electrochemiluminescence nanointerface. ACS Appl. Mater. Interfaces 2015, 7, 7599–7604. [Google Scholar] [CrossRef]
- Samadi Pakchin, P.; Ghanbari, H.; Saber, R.; Omidi, Y. Electrochemical immunosensor based on chitosan-gold nanoparticle/carbon nanotube as a platform and lactate oxidase as a label for detection of CA125 oncomarker. Biosens. Bioelectron. 2018, 122, 68–74. [Google Scholar] [CrossRef] [PubMed]
- Han, J.; Zhuo, Y.; Chai, Y.Q.; Xiang, Y.; Yuan, R. New type of redox nanoprobe: C60-based nanomaterial and its application in electrochemical immunoassay for doping detection. Anal. Chem. 2015, 87, 1669–1675. [Google Scholar] [CrossRef]
- Gandhi, M.; Rajagopal, D.; Parthasarathy, S.; Raja, S.; Huang, S.T.; Senthil Kumar, A. In Situ Immobilized Sesamol-Quinone/Carbon Nanoblack-Based Electrochemical Redox Platform for Efficient Bioelectrocatalytic and Immunosensor Applications. ACS Omega 2018, 3, 10823–10835. [Google Scholar] [CrossRef] [PubMed]
- Akyuz, D.; Keles, T.; Biyiklioglu, Z.; Koca, A. Electrochemical pesticide sensors based on electropolymerized metallophthalocyanines. J. Electroanal. Chem. 2017, 804, 53–63. [Google Scholar] [CrossRef]
- Ali, M.; Shah, I.; Kim, S.W.; Sajid, M.; Lim, J.H.; Choi, K.H. Quantitative detection of uric acid through ZnO quantum dots based highly sensitive electrochemical biosensor. Sens. Actuators A Phys. 2018, 283, 282–290. [Google Scholar] [CrossRef]
- Baluta, S.; Lesiak, A.; Cabaj, J. Graphene Quantum Dots-based Electrochemical Biosensor for Catecholamine Neurotransmitters Detection. Electroanalysis 2018, 30, 1773–1782. [Google Scholar] [CrossRef]
- Chen, C.; Wang, L.; Jiang, G.H.; Yu, H.J. Chemical preparation of special-shaped metal nanomaterials through encapsulation or inducement in soft solution. Rev. Adv. Mater. Sci. 2006, 11, 1–18. [Google Scholar]
- McCarthy, S.A.; Ratkic, R.; Purcell-Milton, F.; Perova, T.S.; Gun’ko, Y.K. Adaptable surfactant-mediated method for the preparation of anisotropic metal chalcogenide nanomaterials. Sci. Rep. 2018, 8, 2860. [Google Scholar] [CrossRef]
- Zhou, J.; Soontornworajit, B.; Wang, Y. DNA aptamer-based nanomaterial for targeted cell labeling. In Abstracts of Papers of the American Chemical Society; American Chemical Society: Washington, DC, USA, 2009; Volume 238. [Google Scholar]
- Zeng, H.J.; Agyapong, D.A.Y.; Li, C.X.; Zhao, R.L.; Yang, H.; Wu, C.H.; Jiang, Y.D.; Liu, Y.Y. A carcinoembryonic antigen optoelectronic immunosensor based on thiol-derivative-nanogold labeled anti-CEA antibody nanomaterial and gold modified ITO. Sens. Actuators B Chem. 2015, 221, 22–27. [Google Scholar] [CrossRef]
- Jampani, P.; Wang, Z.Q. Improving biocompatibility of nano-scale metal-organic supercontainers (MOSCs) for functional applications. In Abstracts of Papers of the American Chemical Society; American Chemical Society: Washington, DC, USA, 2018; Volume 255. [Google Scholar]
- Moyo, M.; Okonkwo, J.O.; Agyei, N.M. Recent Advances in Polymeric Materials Used as Electron Mediators and Immobilizing Matrices in Developing Enzyme Electrodes. Sensors 2012, 12, 923–953. [Google Scholar] [CrossRef]
- Wang, J.; Wang, Y.; Chen, H.; Xu, H.; Wang, W.; Bai, L. Sensitive and simultaneous detection of tumor markers assisted by novel functional polymer brush/Au nanoparticles composite. Sens. Actuators B Chem. 2018, 258, 998–1007. [Google Scholar] [CrossRef]
- Wang, Y.; Wang, Y.; Wu, D.; Ma, H.; Zhang, Y.; Fan, D.; Pang, X.; Du, B.; Wei, Q. Label-free electrochemical immunosensor based on flower-like Ag/MoS2/rGO nanocomposites for ultrasensitive detection of carcinoembryonic antigen. Sens. Actuators B Chem. 2018, 255, 125–132. [Google Scholar] [CrossRef]
- Wang, Y.; Ma, H.; Wang, X.; Pang, X.; Wu, D.; Du, B.; Wei, Q. Novel signal amplification strategy for ultrasensitive sandwich-type electrochemical immunosensor employing Pd-Fe3O4-GS as the matrix and SiO2 as the label. Biosens. Bioelectron. 2015, 74, 59–65. [Google Scholar] [CrossRef] [PubMed]
- Sheng, Y.; Li, P.L.; Chen, Y.H. The Synergistic Effect of Transition Metal Nitrate in Polypropylene/Magnesium Hydroxide Flame Retarded Composite. Adv. Polym. Technol. 2014, 33, 33. [Google Scholar] [CrossRef]
- Wang, L.; Lei, J.; Ma, R.; Ju, H. Host-guest interaction of adamantine with a beta-cyclodextrin-functionalized AuPd bimetallic nanoprobe for ultrasensitive electrochemical immunoassay of small molecules. Anal. Chem. 2013, 85, 6505–6510. [Google Scholar] [CrossRef] [PubMed]
- Sheng, Y.; Chen, Y.H.; Bai, Y.Z. Catalytically Synergistic Effects of Novel LaMnO3 Composite Metal Oxide in Intumescent Flame-Retardant Polypropylene System. Polym. Compos. 2014, 35, 2390–2400. [Google Scholar] [CrossRef]
- Li, F.; Feng, J.; Gao, Z.; Shi, L.; Wu, D.; Du, B.; Wei, Q. Facile Synthesis of Cu2O@TiO2-PtCu Nanocomposites as a Signal Amplification Strategy for the Insulin Detection. ACS Appl. Mater. Interfaces 2019, 11, 8945–8953. [Google Scholar] [CrossRef]
- Chae, J.; Oh, M.; Quy, V.H.V.; Kwon, J.; Kim, J.H.; Kang, S.H.; Kim, H.; Vijayakumar, E.; Ahn, K.S. Enhanced electrocatalytic activity and electrochemical stability of copper(I) sulfide electrode electrodeposited on a Ti interlayer-coated fluorine-doped tin oxide substrate and its application to quantum dot-sensitized solar cells. Thin Solid Films 2018, 660, 46–53. [Google Scholar] [CrossRef]
- Li, Q.; Liu, D.; Xu, L.; Xing, R.; Liu, W.; Sheng, K.; Song, H. Wire-in-Tube IrOx Architectures: Alternative Label-Free Immunosensor for Amperometric Immunoassay toward alpha-Fetoprotein. ACS Appl. Mater. Interfaces 2015, 7, 22719–22726. [Google Scholar] [CrossRef]
- Wang, Y.; Zhao, G.; Wang, H.; Cao, W.; Du, B.; Wei, Q. Sandwich-type electrochemical immunoassay based on Co3O4@MnO2-thionine and pseudo-ELISA method toward sensitive detection of alpha fetoprotein. Biosens. Bioelectron. 2018, 106, 179–185. [Google Scholar] [CrossRef]
- Yan, Q.; Cao, L.; Dong, H.; Tan, Z.; Hu, Y.; Liu, Q.; Liu, H.; Zhao, P.; Chen, L.; Liu, Y.; et al. Label-free immunosensors based on a novel multi-amplification signal strategy of TiO2-NGO/Au@Pd hetero-nanostructures. Biosens. Bioelectron. 2019, 127, 174–180. [Google Scholar] [CrossRef] [PubMed]
- Shen, W.J.; Zhuo, Y.; Chai, Y.Q.; Yang, Z.H.; Han, J.; Yuan, R. Enzyme-free electrochemical immunosensor based on host-guest nanonets catalyzing amplification for procalcitonin detection. ACS Appl. Mater. Interfaces 2015, 7, 4127–4134. [Google Scholar] [CrossRef] [PubMed]
- Cao, X.; Liu, S.; Feng, Q.; Wang, N. Silver nanowire-based electrochemical immunoassay for sensing immunoglobulin G with signal amplification using strawberry-like ZnO nanostructures as labels. Biosens. Bioelectron. 2013, 49, 256–262. [Google Scholar] [CrossRef] [PubMed]
- Wang, P.; Li, M.; Pei, F.; Li, Y.; Liu, Q.; Dong, Y.; Chu, Q.; Zhu, H. An ultrasensitive sandwich-type electrochemical immunosensor based on the signal amplification system of double-deck gold film and thionine unite with platinum nanowire inlaid globular SBA-15 microsphere. Biosens. Bioelectron. 2017, 91, 424–430. [Google Scholar] [CrossRef] [PubMed]
- Valera, A.E.; Nesbitt, N.T.; Archibald, M.M.; Naughton, M.J.; Chiles, T.C. On-Chip Electrochemical Detection of Cholera Using a Polypyrrole-Functionalized Dendritic Gold Sensor. ACS Sens. 2019, 4, 654–659. [Google Scholar] [CrossRef] [PubMed]
- Zhao, C.Z.; Qiu, J.J.; Agarwal, G.; Wang, J.S.; Ren, X.Z.; Xia, H.; Guo, B.Z.; Ma, C.L.; Wan, S.B.; Bertioli, D.J.; et al. Genome-Wide Discovery of Microsatellite Markers from Diploid Progenitor Species, Arachis duranensis and A. ipaensis, and Their Application in Cultivated Peanut (A. hypogaea). Front. Plant Sci. 2017, 8, 1209. [Google Scholar] [CrossRef] [PubMed]
- Shadjou, N.; Hasanzadeh, M.; Omari, A. Electrochemical quantification of some water soluble vitamins in commercial multi-vitamin using poly-amino acid caped by graphene quantum dots nanocomposite as dual signal amplification elements. Anal. Biochem. 2017, 539, 70–80. [Google Scholar] [CrossRef]
- Cadkova, M.; Kovarova, A.; Dvorakova, V.; Metelka, R.; Bilkova, Z.; Korecka, L. Electrochemical quantum dots-based magneto-immunoassay for detection of HE4 protein on metal film-modified screen-printed carbon electrodes. Talanta 2018, 182, 111–115. [Google Scholar] [CrossRef]
- Kokkinos, C.; Prodromidis, M.; Economou, A.; Petrou, P.; Kakabakos, S. Quantum dot-based electrochemical DNA biosensor using a screen-printed graphite surface with embedded bismuth precursor. Electrochem. Commun. 2015, 60, 47–51. [Google Scholar] [CrossRef]
- Li, C.C.; Hu, J.; Lu, M.F.; Zhang, C.Y. Quantum dot-based electrochemical biosensor for stripping voltammetric detection of telomerase at the single-cell level. Biosens. Bioelectron. 2018, 122, 51–57. [Google Scholar] [CrossRef]
- Chen, Z.P.; Mou, K.W.; Wang, X.H.; Liu, L.C. Nitrogen-Doped Graphene Quantum Dots Enhance the Activity of Bi2O3 Nanosheets for Electrochemical Reduction of CO2 in a Wide Negative Potential Region. Angew. Chem. Int. Ed. 2018, 57, 12790–12794. [Google Scholar] [CrossRef] [PubMed]
- Wu, P.; Xu, Y.X.; Zhan, J.Y.; Li, Y.; Xue, H.G.; Pang, H. The Research Development of Quantum Dots in Electrochemical Energy Storage. Small 2018, 14, 14. [Google Scholar] [CrossRef] [PubMed]
- Pinwattana, K.; Wang, J.; Lin, C.T.; Wu, H.; Du, D.; Lin, Y.; Chailapakul, O. CdSe/ZnS quantum dots based electrochemical immunoassay for the detection of phosphorylated bovine serum albumin. Biosens. Bioelectron. 2010, 26, 1109–1113. [Google Scholar] [CrossRef] [PubMed]
- Lu, D.; Wang, J.; Wang, L.; Du, D.; Timchalk, C.; Barry, R.; Lin, Y. A Novel Nanoparticle-Based Disposable Electrochemical Immunosensor for Diagnosis of Exposure to Toxic Organophosphorus Agents. Adv. Funct. Mater. 2011, 21, 4371–4378. [Google Scholar] [CrossRef]
- Yang, M.; Javadi, A.; Gong, S. Sensitive electrochemical immunosensor for the detection of cancer biomarker using quantum dot functionalized graphene sheets as labels. Sens. Actuators B Chem. 2011, 155, 357–360. [Google Scholar] [CrossRef]
- Qin, D.M.; Jiang, X.H.; Mo, G.C.; Feng, J.S.; Yu, C.H.; Deng, B.Y. A Novel Carbon Quantum Dots Signal Amplification Strategy Coupled with Sandwich Electrochemiluminescence Immunosensor for the Detection of CA15-3 in Human Serum. ACS Sens. 2019, 4, 504–512. [Google Scholar] [CrossRef] [PubMed]
- Martin-Yerga, D.; Gonzalez-Garcia, M.B.; Costa-Garcia, A. Electrochemical immunosensor for anti-tissue transglutaminase antibodies based on the in situ detection of quantum dots. Talanta 2014, 130, 598–602. [Google Scholar] [CrossRef]
- Wu, Y.; Xue, P.; Kang, Y.; Hui, K.M. Highly Specific and Ultrasensitive Graphene-Enhanced Electrochemical Detection of Low-Abundance Tumor Cells Using Silica Nanoparticles Coated with Antibody-Conjugated Quantum Dots. Anal. Chem. 2013, 85, 3166–3173. [Google Scholar] [CrossRef]
- Zhou, S.; Wang, Y.; Zhu, J.J. Simultaneous Detection of Tumor Cell Apoptosis Regulators Bcl-2 and Bax through a Dual-Signal-Marked Electrochemical Immunosensor. ACS Appl. Mater. Interfaces 2016, 8, 7674–7682. [Google Scholar] [CrossRef]
- Liu, K.; Zhang, J.; Liu, Q.; Huang, H. Electrochemical immunosensor for alpha-fetoprotein determination based on ZnSe quantum dots/Azure I/gold nanoparticles/poly (3,4-ethylenedioxythiophene) modified Pt electrode. Electrochim. Acta 2013, 114, 448–454. [Google Scholar] [CrossRef]
- Feng, D.; Li, L.; Fang, X.; Han, X.; Zhang, Y. Dual signal amplification of horseradish peroxidase functionalized nanocomposite as trace label for the electrochemical detection of carcinoembryonic antigen. Electrochim. Acta 2014, 127, 334–341. [Google Scholar] [CrossRef]


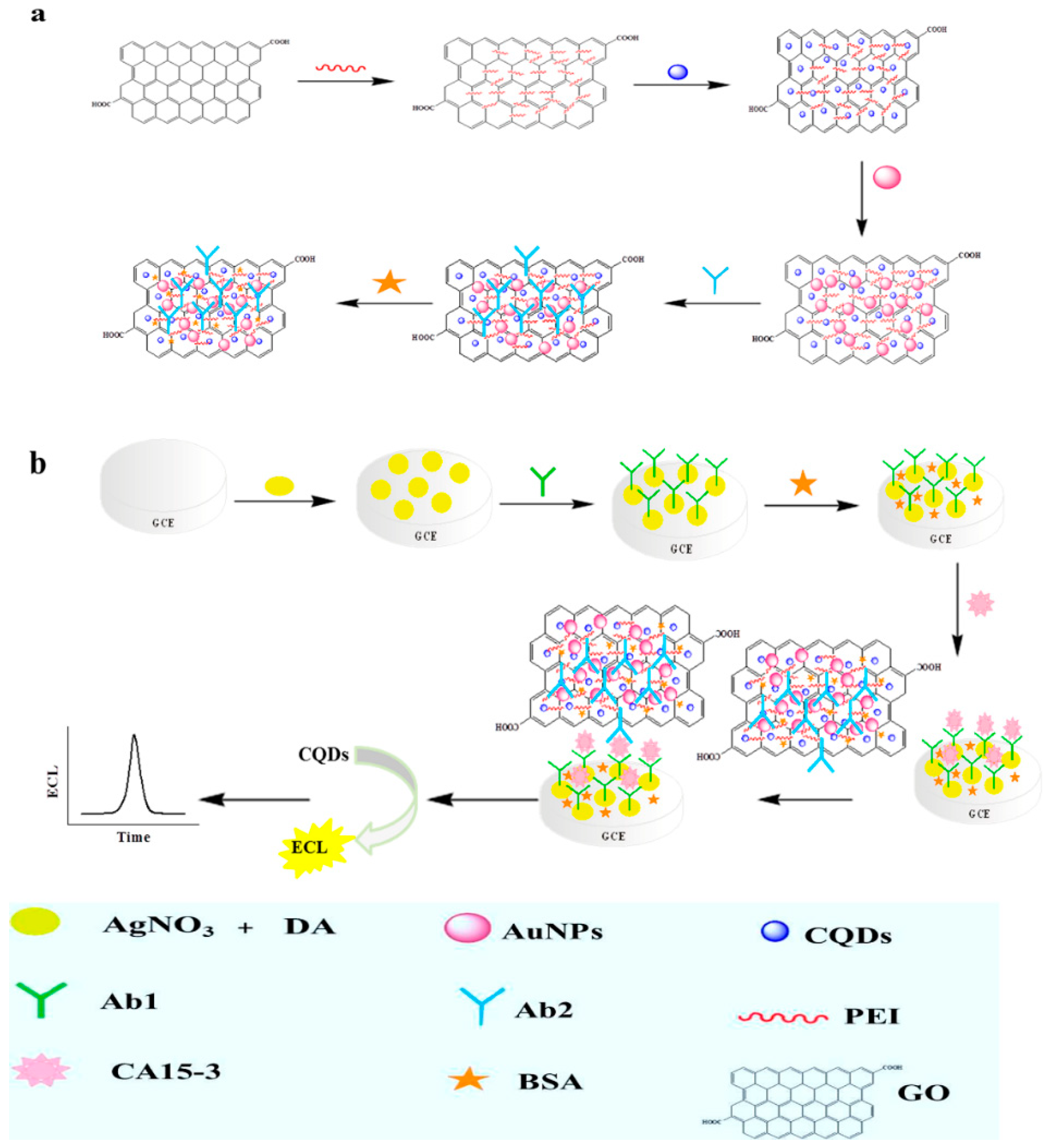
| Electrode Modification | Label | Analyte | Detection Range and LOD | Reproducibility | Stability | Reference |
|---|---|---|---|---|---|---|
| CP/RGO | Label-free | miRNA | Detection range: 1 fM–1 nM LOD: 5 fM | - | - | [42] |
| RGO-carbon nanotubes | Label-free | miRNA | LOD: 10 fM | - | - | [43] |
| streptavidin-functionalized NG | HRP | CEA | Linear range: 0.02–12 ng/mL LOD: 0.01 ng/mL | 3.6% | 95.8% (4 weeks) | [44] |
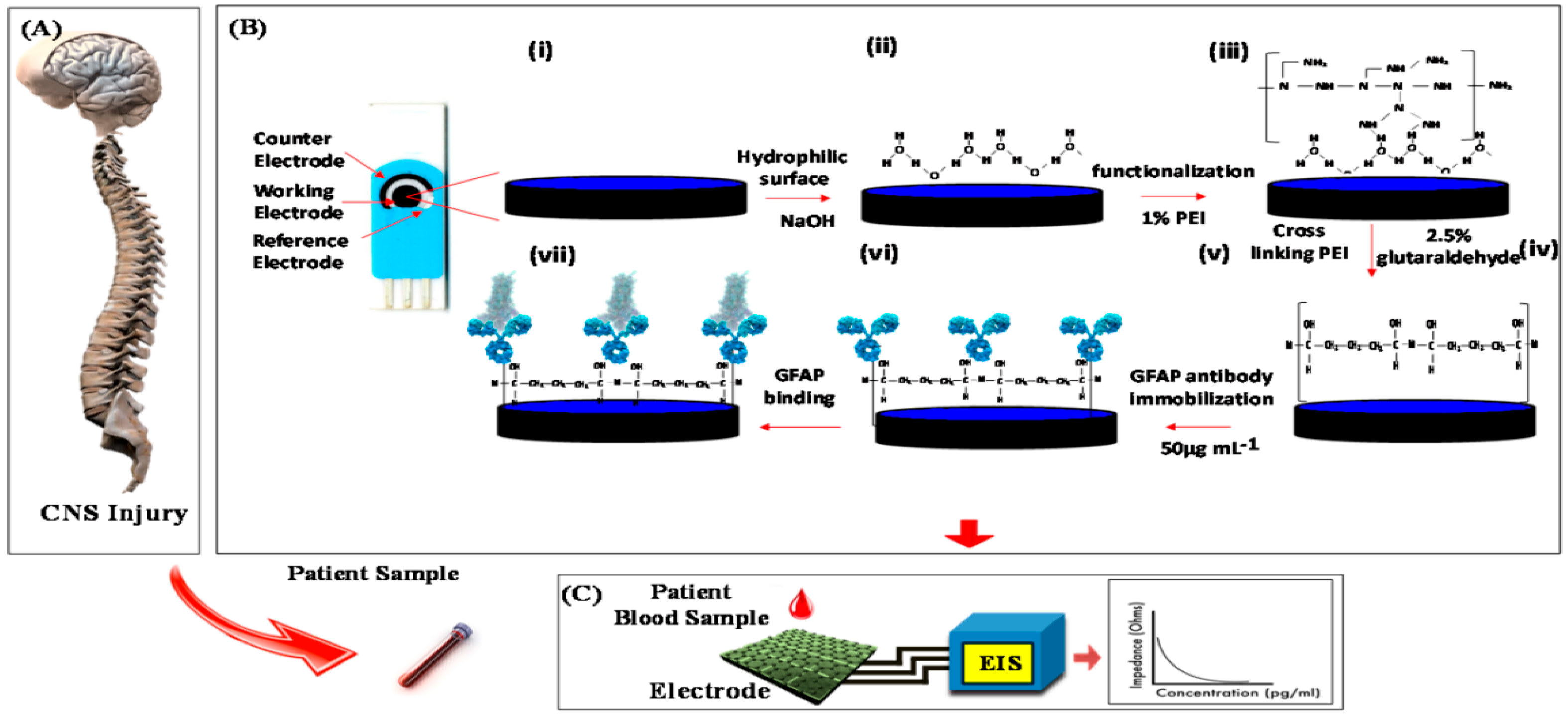
| PEI coated graphene | Label-free | GFAP | Linear range: 1 pg/mL–100 ng/mL | 4.5% | - | [45] |
| SWNT forests | Carboxylated MWCNT-HRP | IL-6 | LOD: 0.5 pg/mL | - | - | [48] |
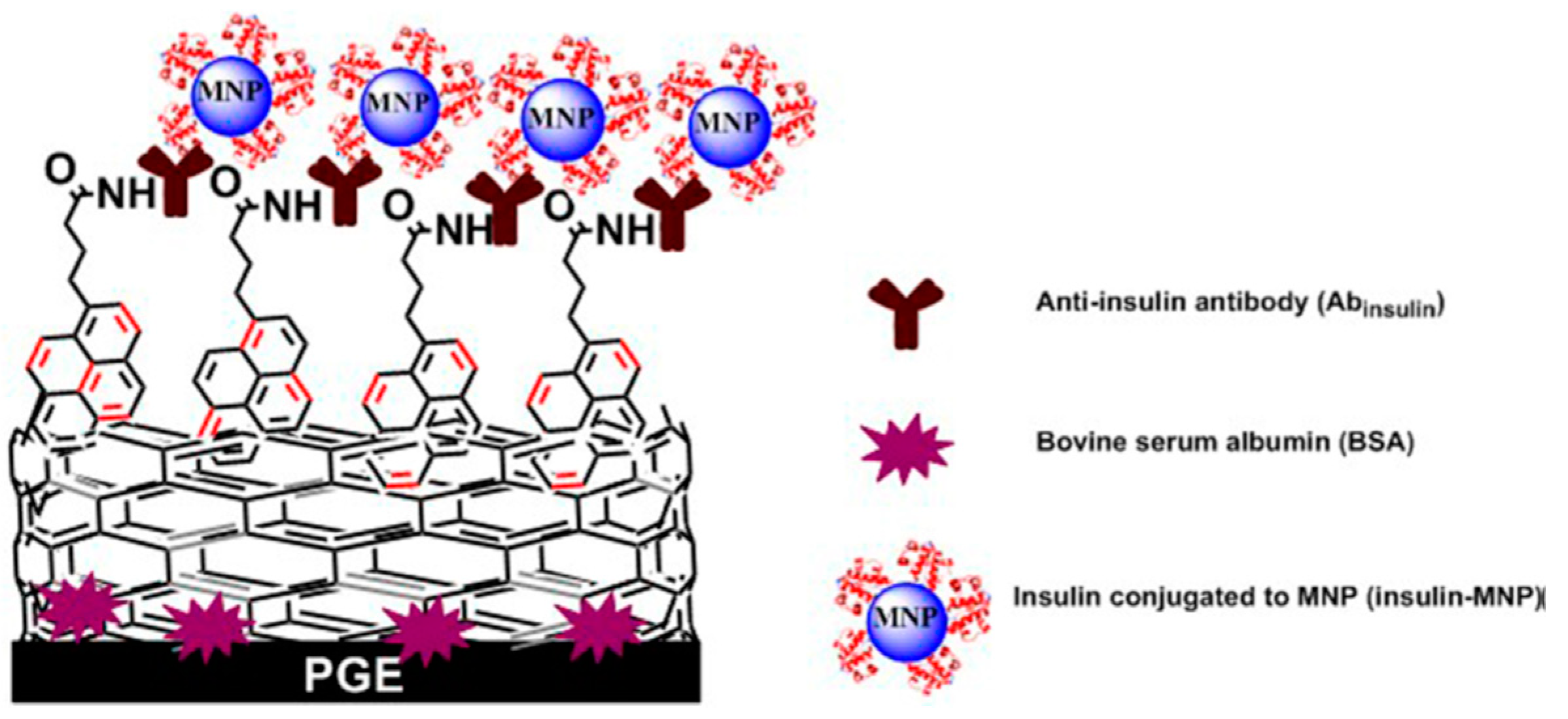
| PGE/MWNT/Py | MNP | Insulin | LOD: 5 pM | - | - | [49] |
| MWCNTs/chitosan/GNDs | Label-free | NT-proBNP | Linear range: 0.01–100 pg/mL LOD: 3.86 fg/mL | 3.3–5.9% | - | [50] |
| CS-AuNP/MWCNT/GO | Lactate oxidase | CHA | Linear range: 0.01–0.5 U/mL/0.5–100 U/mL LOD: 0.002 U/mL | 7.6% | 94.5% (2 weeks) | [51] |
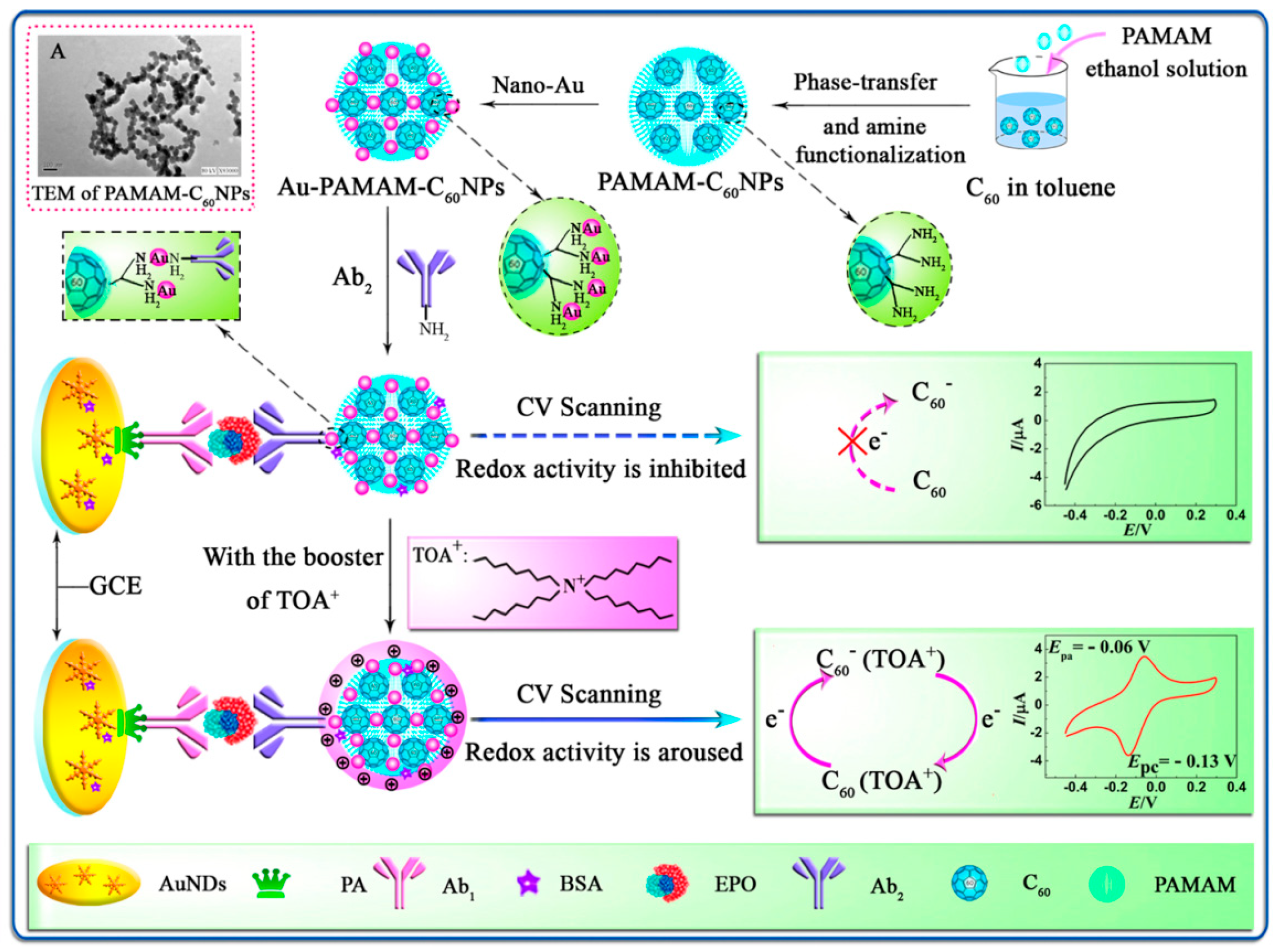
| AuNPs-protein A | C60NPs-Au-PAMAM | Erythropoietin | Linear range: 0.01–80 mIU/mL LOD: 0.0027 mIU/mL | 4% | 86.3% (2 weeks) | [52] |
| CB@Ses-Qn | HRP | White spot syndrome virus | LOD: 990 nM | 1.3% | - | [53] |
| Electrode Modification | Label | Analyte | Detection Range and LOD | Reproducibility | Stability | Reference |
|---|---|---|---|---|---|---|
| AuNPs/PGMA-g-PAN | Aq, FeC-COOH | PSA, AFP | Linear range: 10 pg/mL–100 ng/mL LOD: 2.2 pg/mL (PSA), 1.8 pg/mL (AFP) | - | - | [63] |
| Ag/MoS2/rGO | Label-free | CEA | Linear range: 0.01 pg/mL–100 ng/mL LOD: 1.6 fg/mL | <5% | 100% (4 weeks) | [64] |
| Pd–Fe3O4-GS | SiO2 | IgG | Linear range: 5 × 10−6–5 ng/mL LOD: 3.2 fg/mL | 3.2% | 100% (4 weeks) | [65] |
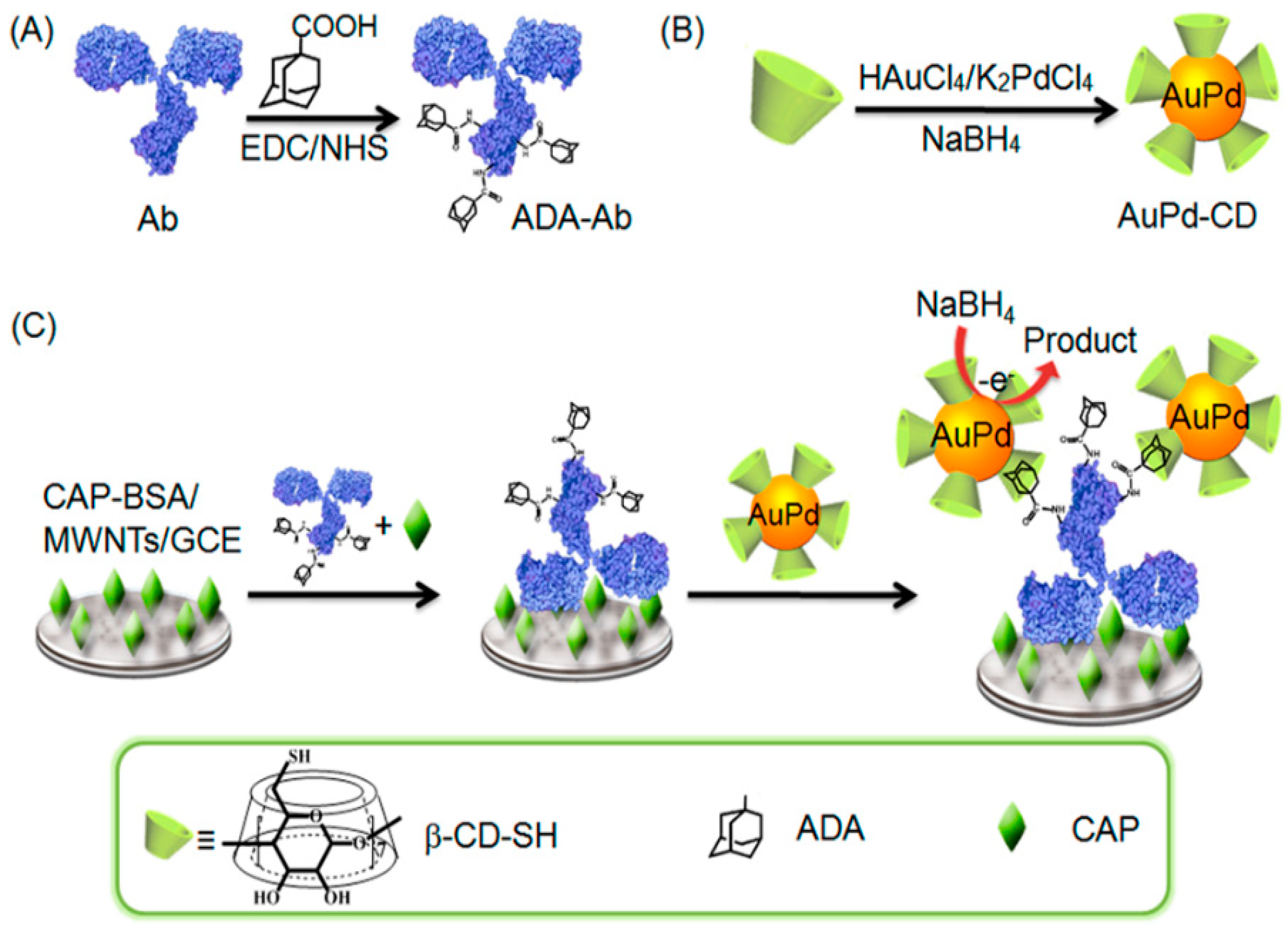
| CAP-MWCNTs | AuPd−CD | Adamantine | Linear range: 50 pg/mL–50 μg/mL LOD: 4.6 pg/mL | 2.9% | 93.5% (1 week) | [66] |
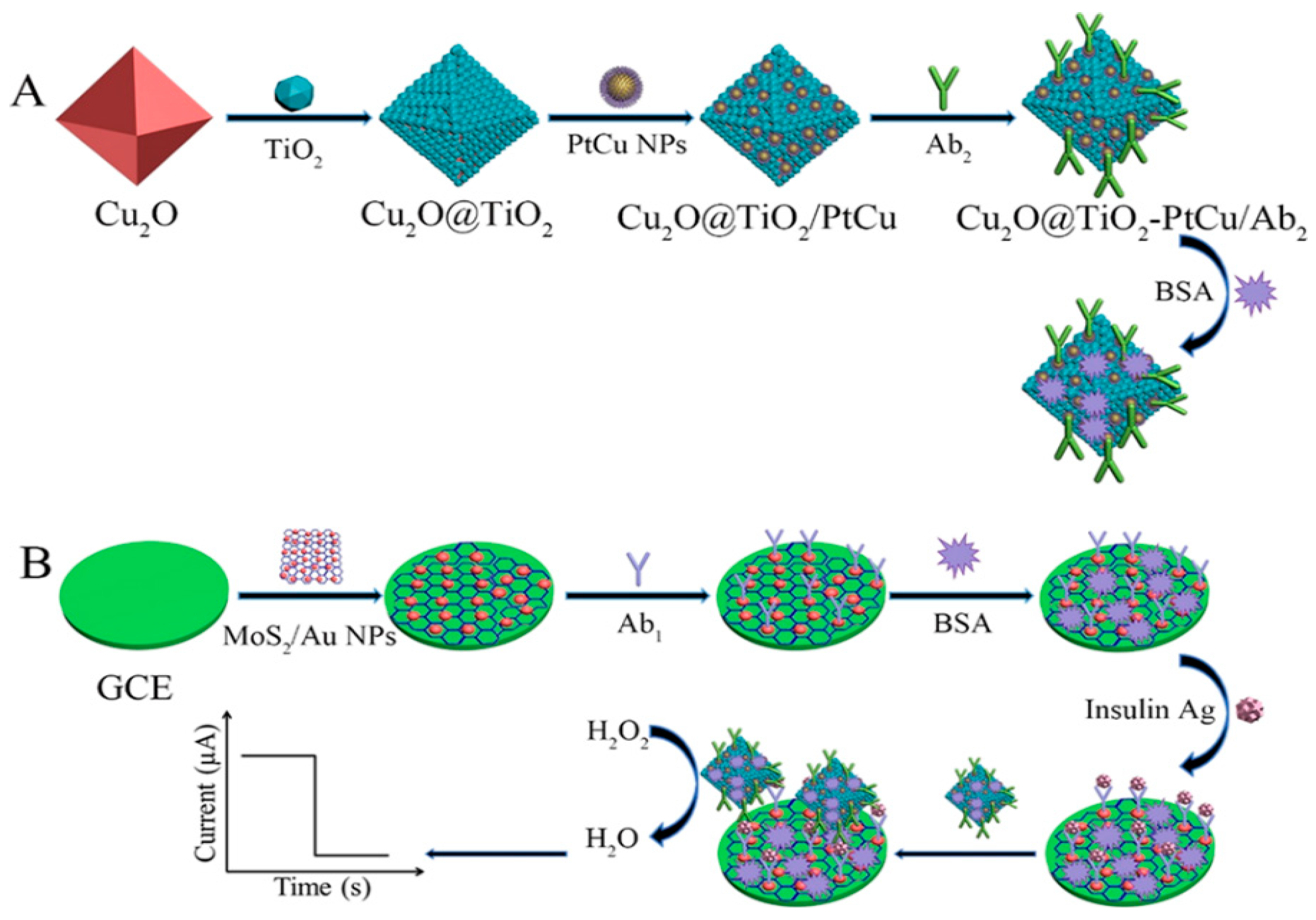
| MoS2/Au NPs | PtCu NPs/Cu2O@TiO2-NH2 | Insulin | Linear range: 0.1 pg/mL–100 ng/mL LOD: 0.024 pg/mL | 2.54–4.28% | - | [69] |
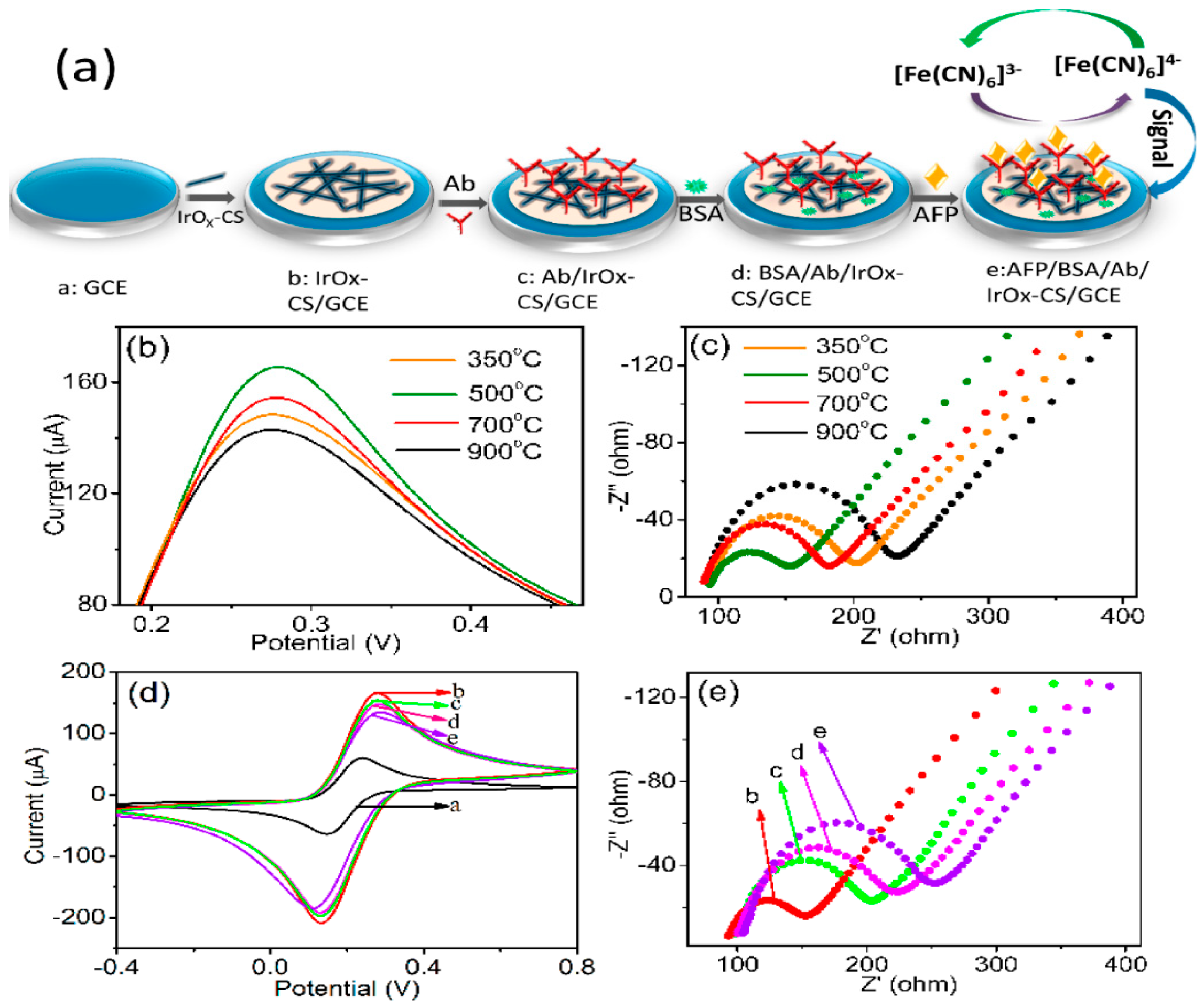
| IrOx | Label-free | AFP | Linear range: 0.05–150 ng/mL LOD: 20 pg/mL | <5.0% | 14% (2 weeks) | [71] |
| Ag NPs | Co3O4@MnO2-Th | AFP | Linear range: 0.001–100 ng/mL LOD: 0.33 pg/mL. | <5.0% | 86% (1 week) | [72] |
| TiO2-NGO/Au@Pd HSs | Label-free | HE4 Ag | Linear range: 40 fM–60 nM LOD: 13.33 fM | <2.3% | 90.2% (4 weeks) | [73] |
| DpAu | Fc-Fc/β-CD/PAMAM−Au | PCT | Linear range: 1.80 pg/mL–500 ng/mL LOD: 0.36 pg/mL | 3.7% | 83.7% (4 weeks) | [74] |
| TiO2-NGO/Au@Pd HSs | Label-free | HE4 Ag | Linear range: 40 fM–60 nM LOD: 13.33 fM | <2.3% | 90.2% (4 weeks) | [75] |
| D-Au film | Pt NWs@g-SBA-15/Thi | HBs Ag | Linear range: 10 fg/mL–100 ng/mL LOD: 3.3 fg/mL | 1.31% | 85.37% (4 weeks) | [76] |
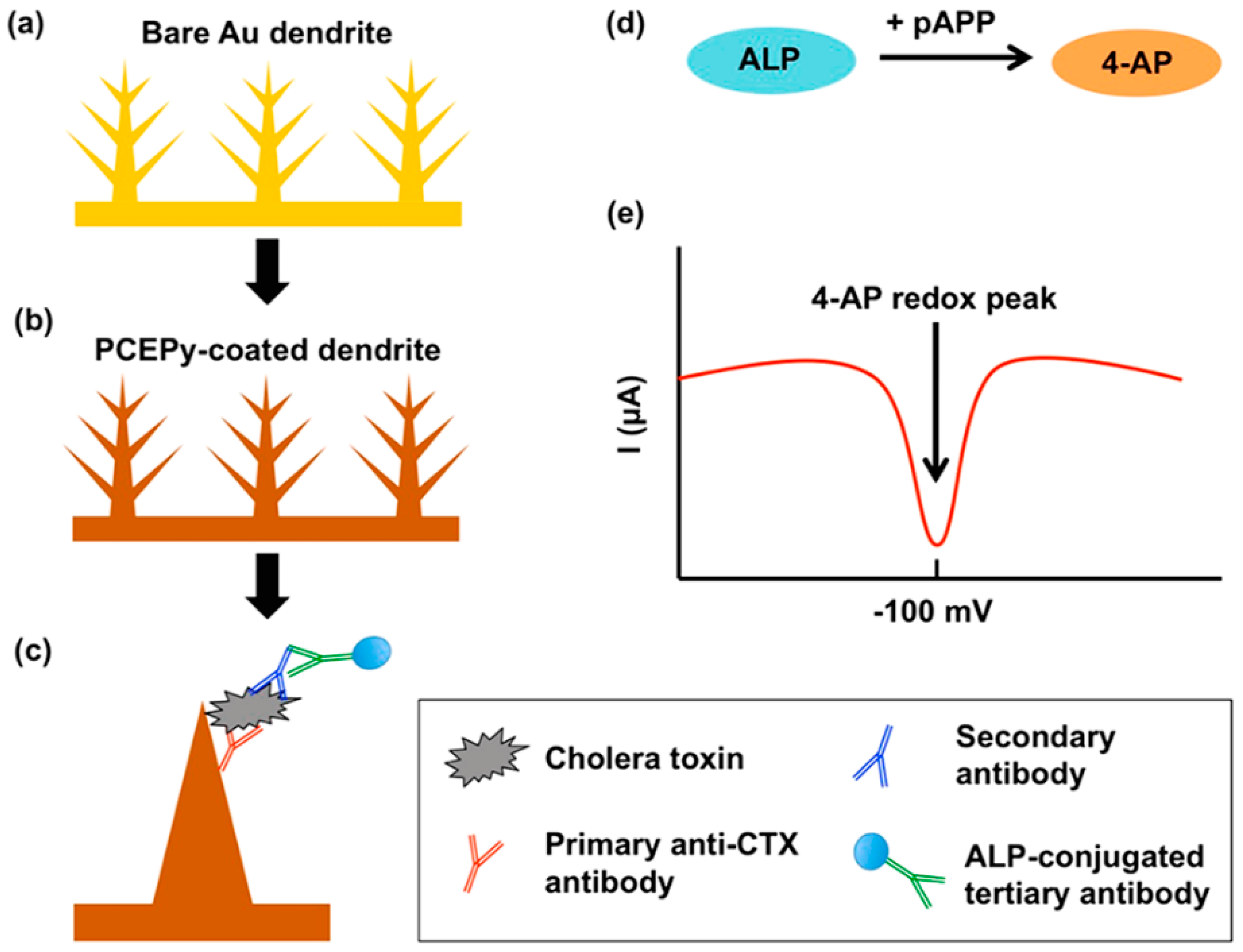
| Dendritic gold-PCEPy | ALP | CTX | LOD: 1 ng/mL | - | - | [77] |
| Electrode Modification | Label | Analyte | Detection Range and LOD | Reproducibility | Stability | Reference |
|---|---|---|---|---|---|---|
| - | CdSe/ZnS QD | BSA-OP | Linear range: 0.5–500 ng/mL LOD: 0.5 ng/mL | 8.6% | - | [85] |
| ZrO2 | QD | OP-BChE | Linear range: 0.1–30 nM LOD: 0.03 nM | 4.5% | - | [86] |
| GS–PBSE | GS–QD | PSA | Linear range: 0.005–10 ng/mL LOD: 3 pg/ML | 7.9% | 88% (3 weeks) | [87] |
| AgNPs-PDA | CQDs-PEI-GO/AuNPs | CA15-3 | Linear range: 0.005–10 ng/mL LOD: 3 pg/mL | 2.3% | 89.6% (4 weeks) | [88] |
| - | CdSe/ZnS QDs | tTG | LOD: 2.2 U/mL | 5.9% | 100% (4 weeks) | [89] |
| CS-GO | ZnSe QD-coated silica nanoparticles | EpCAM, GPC3 on the surface of Hep3B cell | Linear range: 5–1 × 106 cells/mL LOD: 5 cells/mL | 4.6% and 6.2% | >90% (2 weeks) | [90] |
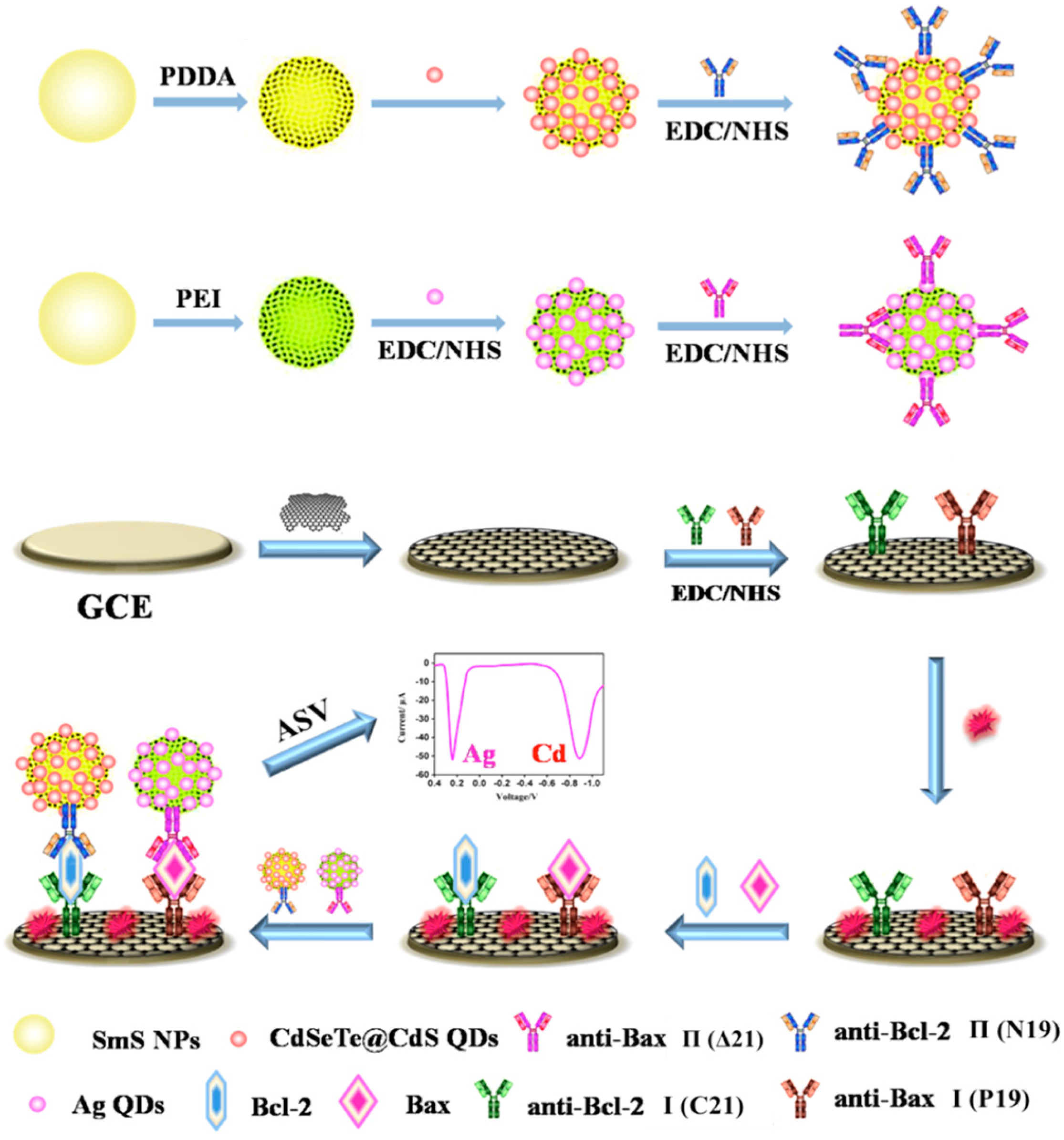
| RGO | CdSeTe@CdS QDs, Ag nanoclusters | Bcl-2, Bax | LOD: 1 × 103 cells | - | - | [91] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, Z.; Cong, Y.; Huang, Y.; Du, X. Nanomaterials-Based Electrochemical Immunosensors. Micromachines 2019, 10, 397. https://doi.org/10.3390/mi10060397
Zhang Z, Cong Y, Huang Y, Du X. Nanomaterials-Based Electrochemical Immunosensors. Micromachines. 2019; 10(6):397. https://doi.org/10.3390/mi10060397
Chicago/Turabian StyleZhang, Zhenguo, Yulin Cong, Yichun Huang, and Xin Du. 2019. "Nanomaterials-Based Electrochemical Immunosensors" Micromachines 10, no. 6: 397. https://doi.org/10.3390/mi10060397
APA StyleZhang, Z., Cong, Y., Huang, Y., & Du, X. (2019). Nanomaterials-Based Electrochemical Immunosensors. Micromachines, 10(6), 397. https://doi.org/10.3390/mi10060397





