Minimal Concentrations of Deoxynivalenol Reduce Cytokine Production in Individual Lymphocyte Populations in Pigs
Abstract
1. Introduction
2. Results
2.1. Viability of PBMC after DON Exposure
2.2. PBMC Proliferation Activity after DON Exposure
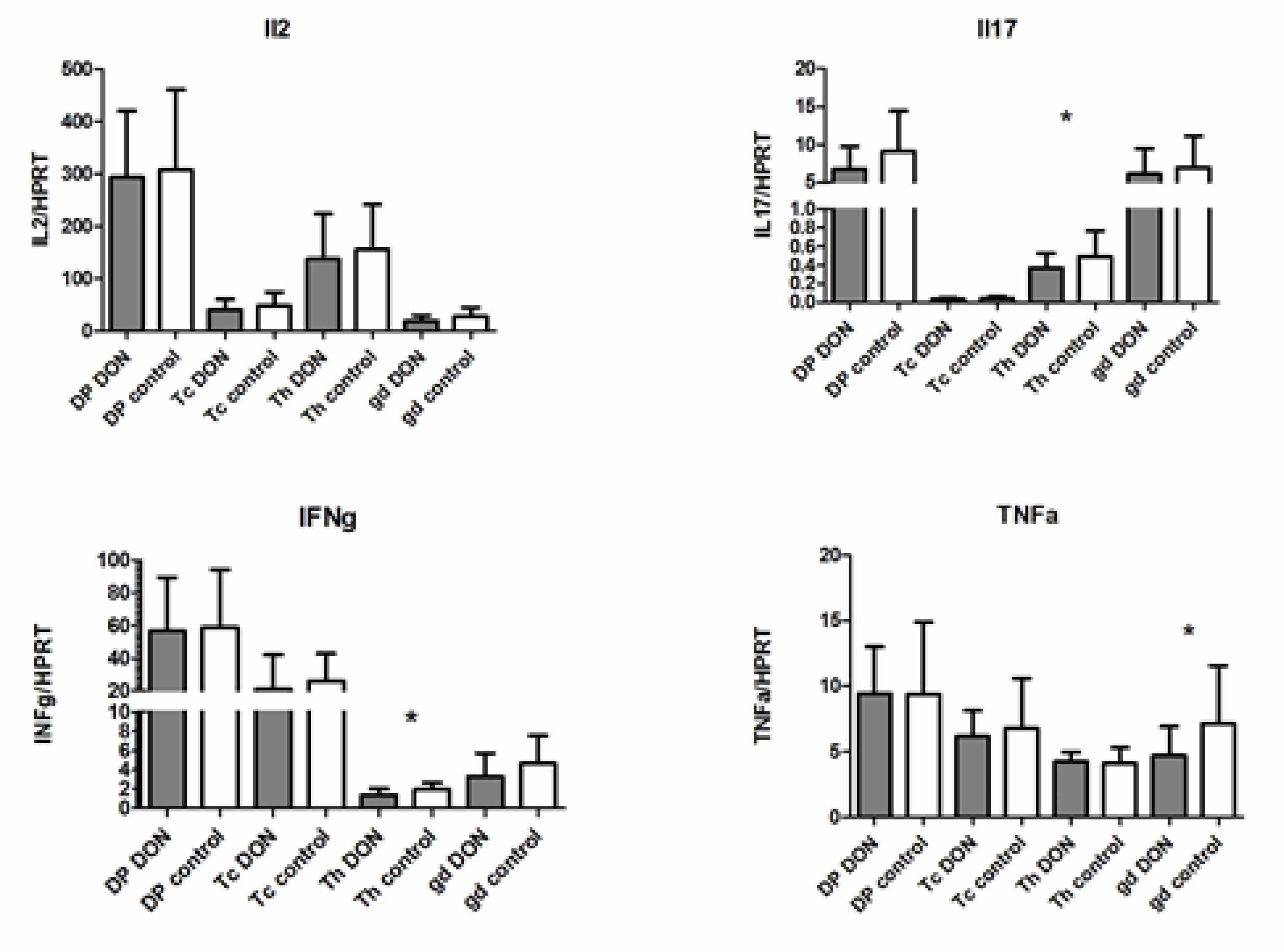
2.3. The Expression of mRNA Encoding Cytokine Genes in PBMC and Lymphocyte Subpopulations after DON Exposure
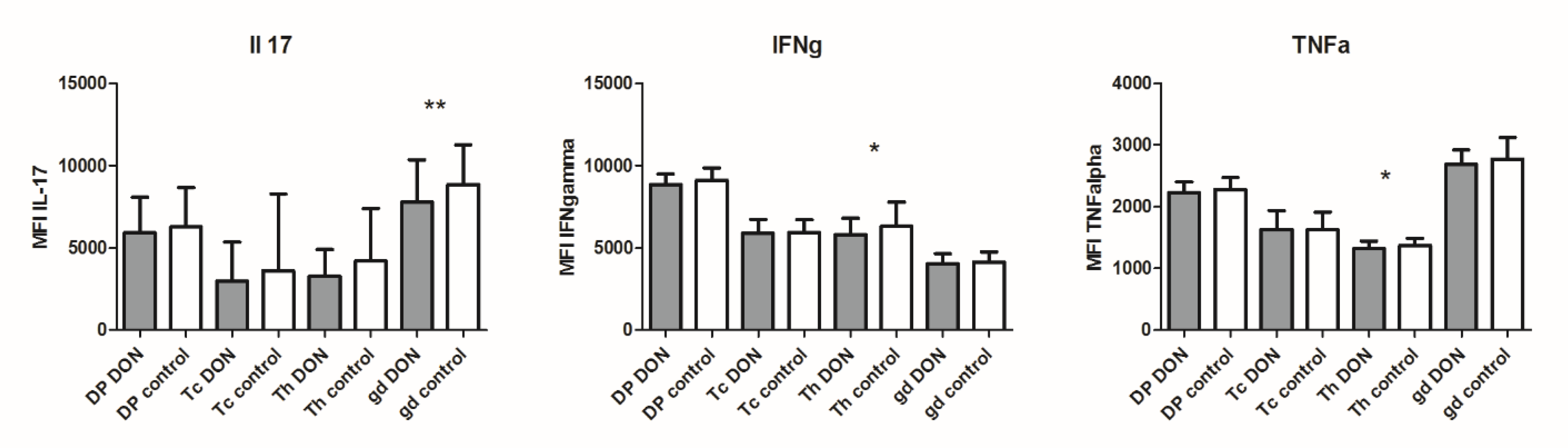
2.4. Cytokine Production at the Protein Level in Lymphocyte Subpopulations after DON Exposure
3. Discussion
4. Conclusions
5. Materials and Methods
5.1. Animals and Blood Samples
5.2. Isolation of Peripheral Blood Mononuclear Cells
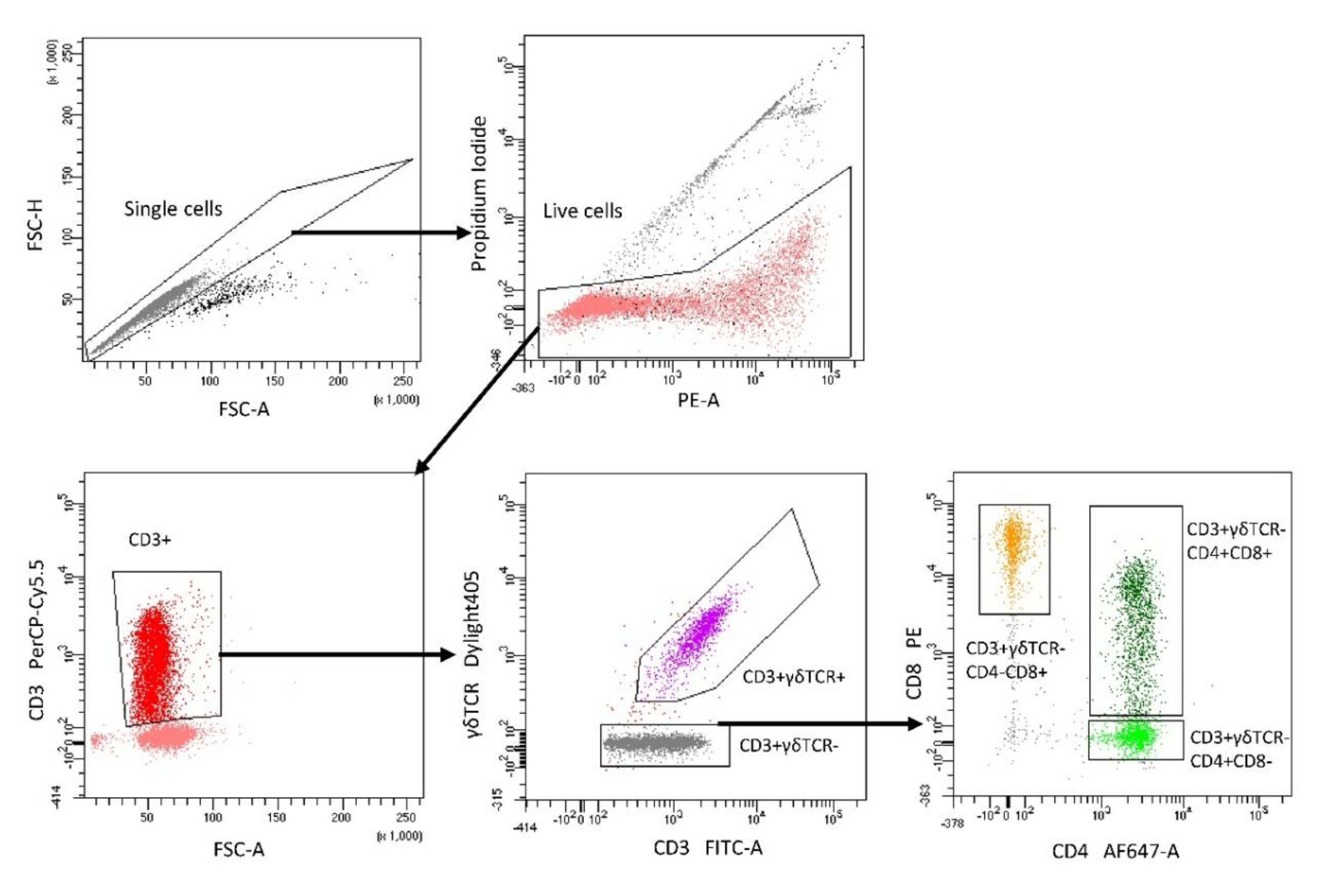
5.3. Isolation of Lymphocyte Subpopulations with FACS
5.4. Viability Assay
5.5. PBMC Proliferation Activity Assay
5.6. Cytokine Gene Expression Analysis—RNA Preparation and Quantitative RT-PCR Analysis
5.7. Flow Cytometry Analysis of Cytokine Production in Lymphocyte Subpopulations after DON Exposure
5.8. Statistical Analysis
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A

References
- Rotter, B.A. Invited Review: Toxicology of Deoxynivalenol (Vomitoxin). J. Toxicol. Environ. Health 1996, 48, 1–34. [Google Scholar] [CrossRef] [PubMed]
- BIOMIN World Mycotoxin Survey the Global Threat 2018 Q1; BIOMIN: Inzersdorf-Getzersdorf, Austria, 2018; pp. 1–5.
- Prelusky, D.B.; Gerdes, R.G.; Underhill, K.L.; Rotter, B.A.; Jui, P.Y.; Trenholm, H.L. Effects of low-level dietary deoxynivalenol on haematological and clinical parameters of the pig. Nat. Toxins 1994, 2, 97–104. [Google Scholar] [CrossRef]
- Rotter, B.A.; Prelusky, D.B.; Pestka, J.J. Toxicology of deoxynivalenol (vomitoxin). J. Toxicol. Environ. Health 1996, 48, 1–2. [Google Scholar] [CrossRef] [PubMed]
- Trenholm, H.L.; Hamilton, R.M.; Friend, D.W.; Thompson, B.K.; Hartin, K.E. Feeding trials with vomitoxin (deoxynivalenol)-contaminated wheat: Effects on swine, poultry, and dairy cattle. J. Am. Vet. Med. Assoc. 1984, 185, 527–531. [Google Scholar] [PubMed]
- King, R.R.; McQueen, R.E.; Levesque, D.; Greenhalgh, R. Transformation of deoxynivalenol (vomitoxin) by rumen microorganisms. J. Agric. Food Chem. 1984, 32, 1181–1183. [Google Scholar] [CrossRef]
- Maresca, M. From the gut to the brain: Journey and pathophysiological effects of the food-associated trichothecene mycotoxin deoxynivalenol. Toxins 2013, 5, 784–820. [Google Scholar] [CrossRef] [PubMed]
- Young, L.G.; McGirr, L.; Valli, V.E.; Lumsden, J.H.; Lun, A. Vomitoxin in corn fed to young pigs. J. Anim. Sci. 1983, 57, 655–664. [Google Scholar] [CrossRef]
- Bergsjø, B.; Langseth, W.; Nafstad, I.; Jansen, J.H.; Larsen, H.J.S. The effects of naturally deoxynivalenol-contaminated oats on the clinical condition, blood parameters, performance and carcass composition of growing pigs. Vet. Res. Commun. 1993, 17, 283–294. [Google Scholar] [CrossRef]
- Pestka, J.J. Deoxynivalenol: Toxicity, mechanisms and animal health risks. Anim. Feed Sci. Technol. 2007, 137, 283–298. [Google Scholar] [CrossRef]
- Pestka, J.J. Deoxynivalenol-induced proinflammatory gene expression: Mechanisms and pathological sequelae. Toxins 2010, 2, 1300–1317. [Google Scholar] [CrossRef]
- Pestka, J.J.; Smolinski, A.T. Deoxynivalenol: Toxicology and Potential Effects on Humans. J. Toxicol. Environ. Health Part B 2005, 8, 39–69. [Google Scholar] [CrossRef] [PubMed]
- Meky, F.A.; Hardie, L.J.; Evans, S.W.; Wild, C.P. Deoxynivalenol-induced immunomodulation of human lymphocyte proliferation and cytokine production. Food Chem. Toxicol. 2001, 39, 827–836. [Google Scholar] [CrossRef]
- Dąbrowski, M.; Jakimiuk, E.; Baranowski, M.; Gajȩcka, M.; Zielonka, Ł.; Gajȩcki, M.T. The effect of deoxynivalenol on selected populations of immunocompetent cells in porcine blood-a preliminary study. Molecules 2017, 22, 691. [Google Scholar] [CrossRef]
- Zhou, H.-R.; Islam, Z.; Pestka, J.J. Rapid, sequential activation of mitogen-activated protein kinases and transcription factors precedes proinflammatory cytokine mRNA expression in spleens of mice exposed to the trichothecene vomitoxin. Toxicol. Sci. 2003, 72, 130–142. [Google Scholar] [CrossRef] [PubMed]
- Ouyang, Y.L.; Azcona-Olivera, J.I.; Murtha, J.; Pestka, J.J. Vomitoxin-mediated IL-2, IL-4, and IL-5 superinduction in murine CD4+T cells stimulated with phorbol ester and calcium ionophore: Relation to kinetics of proliferation. Toxicol. Appl. Pharmacol. 1996, 138, 324–334. [Google Scholar] [CrossRef]
- Severino, L.; Luongo, D.; Bergamo, P.; Lucisano, A.; Rossi, M. Mycotoxins nivalenol and deoxynivalenol differentially modulate cytokine mRNA expression in Jurkat T cells. Cytokine 2006, 36, 75–82. [Google Scholar] [CrossRef] [PubMed]
- Pestka, J.; Uzarski, R.; Islam, Z. Induction of apoptosis and cytokine production in the Jurkat human T cells by deoxynivalenol: Role of mitogen-activated protein kinases and comparison to other 8-ketotrichothecenes. Toxicology 2005, 206, 207–219. [Google Scholar] [CrossRef]
- Azcona-Olivera, J.I.; Ouyang, Y.-L.; Warner, R.L.; Linz, J.E.; Pestka, J.J. Effects of vomitoxin (Deoxynivalenol) and cycloheximide on IL-2, 4, 5 and 6 secretion and mRNA levels in murine CD4+ cells. Food Chem. Toxicol. 1995, 33, 433–441. [Google Scholar] [CrossRef]
- European Commission. COMMISSION RECOMMENDATION 2006/576/EC of 17 August 2006 on the presence of deoxynivalenol, zearalenone, ochratoxin A, T-2 and HT-2 and fumonisins in products intended for animal feeding. Off. J. Eur. Union 2006, 229, 7–9. [Google Scholar]
- Dänicke, S.; Beyer, M.; Breves, G.; Valenta, H.; Humpf, H.-U. Effects of oral exposure of pigs to deoxynivalenol (DON) sulfonate (DONS) as the non-toxic derivative of DON on tissue residues of DON and de-epoxy-DON and on DONS blood levels. Food Addit. Contam. Part A 2010, 27, 1558–1565. [Google Scholar] [CrossRef]
- Van Limbergen, T.; Devreese, M.; Croubels, S.; Broekaert, N.; Michiels, A.; De Saeger, S.; Maes, D. Role of mycotoxins in herds with and without problems with tail necrosis in neonatal pigs. Vet. Rec. 2017, 181, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Kullik, K.; Brosig, B.; Kersten, S.; Valenta, H.; Diesing, A.-K.; Panther, P.; Reinhardt, N.; Kluess, J.; Rothkötter, H.-J.; Breves, G.; et al. Interactions between the Fusarium toxin deoxynivalenol and lipopolysaccharides on the in vivo protein synthesis of acute phase proteins, cytokines and metabolic activity of peripheral blood mononuclear cells in pigs. Food Chem. Toxicol. 2013, 57, 11–20. [Google Scholar] [CrossRef] [PubMed]
- Goyarts, T.; Dänicke, S.; Brüssow, K.-P.; Valenta, H.; Ueberschär, K.-H.; Tiemann, U. On the transfer of the Fusarium toxins deoxynivalenol (DON) and zearalenone (ZON) from sows to their fetuses during days 35–70 of gestation. Toxicol. Lett. 2007, 171, 38–49. [Google Scholar] [CrossRef] [PubMed]
- Stastny, K.; Stepanova, H.; Hlavova, K.; Faldyna, M. Identification and determination of deoxynivalenol (DON) and deepoxy-deoxynivalenol (DOM-1) in pig colostrum and serum using liquid chromatography in combination with high resolution mass spectrometry (LC-MS/MS (HR)). J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2019, 1126–1127, 121735. [Google Scholar] [CrossRef]
- Rohweder, D.; Kersten, S.; Valenta, H.; Sondermann, S.; Schollenberger, M.; Drochner, W.; Dänicke, S. Bioavailability of the Fusarium toxin deoxynivalenol (DON) from wheat straw and chaff in pigs. Arch. Anim. Nutr. 2013, 67, 37–47. [Google Scholar] [CrossRef]
- Dänicke, S.; Valenta, H.; Döll, S. On the toxicokinetics and the metabolism of deoxynivalenol (DON) in the pig. Arch. Anim. Nutr. 2004, 58, 169–180. [Google Scholar] [CrossRef]
- Goyarts, T.; Dänicke, S. Bioavailability of the Fusarium toxin deoxynivalenol (DON) from naturally contaminated wheat for the pig. Toxicol. Lett. 2006, 163, 171–182. [Google Scholar] [CrossRef]
- Marin, D.E.; Taranu, I.; Manda, G.; Nagoe, I.; Oswald, I.P. In vitro effect of deoxynivalenol on porcine lymphocyte immune functions. Arch. Zootech. 2006, 9, 10–18. [Google Scholar]
- Gerez, J.R.; Pinton, P.; Callu, P.; Grosjean, F.; Oswald, I.P.; Bracarense, A.P.F.L.; Ois Grosjean, F.; Oswald, I.P.; Paula, A.; Bracarense, F.L. Deoxynivalenol alone or in combination with nivalenol and zearalenone induce systemic histological changes in pigs. Exp. Toxicol. Pathol. 2015, 67, 89–98. [Google Scholar] [CrossRef]
- Atkinson, H.A.C.A.; Miller, K. Inhibitory effect of deoxynivalenol, 3-acetyldeoxynivalenol and zearalenone on induction of rat and human lymphocyte proliferation. Toxicol. Lett. 1984, 23, 215–221. [Google Scholar] [CrossRef]
- Pinton, P.; Accensi, F.; Beauchamp, E.; Cossalter, A.-M.M.; Callu, P.; Grosjean, F.; Oswald, I.P. Ingestion of deoxynivalenol (DON) contaminated feed alters the pig vaccinal immune responses. Toxicol. Lett. 2008, 177, 215–222. [Google Scholar] [CrossRef] [PubMed]
- Luongo, D.; De Luna, R.; Russo, R.; Severino, L. Effects of four Fusarium toxins (fumonisin B1, α-zearalenol, nivalenol and deoxynivalenol) on porcine whole-blood cellular proliferation. Toxicon 2008, 52, 156–162. [Google Scholar] [CrossRef] [PubMed]
- Janossy, G.; Greaves, M.F. Lymphocyte activation. II. Discriminating stimulation of lymphocyte subpopulations by phytomitogens and heterologous antilymphocyte sera. Clin. Exp. Immunol. 1972, 10, 525–536. [Google Scholar] [PubMed]
- Goyarts, T.; Dänicke, S.; Tiemann, U.; Rothkötter, H.J. Effect of the Fusarium toxin deoxynivalenol (DON) on IgA, IgM and IgG concentrations and proliferation of porcine blood lymphocytes. Toxicol. In Vitro 2006, 20, 858–867. [Google Scholar] [CrossRef] [PubMed]
- Taranu, I.; Marin, D.E.; Burlacu, R.; Pinton, P.; Damian, V.; Oswald, I.P. Comparative aspects of in vitro proliferation of human and porcine lymphocytes exposed to mycotoxins. Arch. Anim. Nutr. 2010, 64, 383–393. [Google Scholar] [CrossRef]
- Pan, X.; Whitten, D.A.; Wu, M.; Chan, C.; Wilkerson, C.G.; Pestka, J.J. Global protein phosphorylation dynamics during deoxynivalenol-induced ribotoxic stress response in the macrophage. Toxicol. Appl. Pharmacol. 2013, 268, 201–211. [Google Scholar] [CrossRef]
- Geppert, T. Phytohemagglutinin (PHA). In Encyclopedia of Immunology; Elsevier: Amsterdam, The Netherlands, 1998; pp. 1952–1953. [Google Scholar]
- Palacios, R. Concanavalin A triggers T lymphocytes by directly interacting with their receptors for activation. J. Immunol. 1982, 128, 337–342. [Google Scholar]
- Pestka, J.J.; Zhou, H.-R.; Moon, Y.; Chung, Y.J. Cellular and molecular mechanisms for immune modulation by deoxynivalenol and other trichothecenes: Unraveling a paradox. Toxicol. Lett. 2004, 153, 61–73. [Google Scholar] [CrossRef]
- Kopecký, O.; Krejsek, J. KlinickáImunologie, 1st ed.; Nucleus HK: Hradec Králové, Czech Republic, 2004; ISBN 80-86225-50-X. [Google Scholar]
- Ghareeb, K.; Awad, W.A.; Soodoi, C.; Sasgary, S.; Strasser, A.; Böhm, J.; Arditi, M.; Mengheri, E.; Oswald, I. Effects of Feed Contaminant Deoxynivalenol on Plasma Cytokines and mRNA Expression of Immune Genes in the Intestine of Broiler Chickens. PLoS ONE 2013, 8, e71492. [Google Scholar] [CrossRef]
- Becker, C.; Reiter, M.; Pfaffl, M.W.; Meyer, H.H.D.; Bauer, J.; Meyer, K.H.D. Expression of immune relevant genes in pigs under the influence of low doses of deoxynivalenol (DON). Mycotoxin Res. 2011, 27, 287–293. [Google Scholar] [CrossRef]
- Baggiolini, M.; Clark-Lewis, I. Interleukin-8, a chemotactic and inflammatory cytokine. FEBS Lett. 1992, 307, 97–101. [Google Scholar] [CrossRef]
- Dinarello, C.A. Immunological and Inflammatory Functions of the Interleukin-1 Family. Annu. Rev. Immunol. 2009, 27, 519–550. [Google Scholar] [CrossRef] [PubMed]
- Cano, P.M.; Seeboth, J.; Meurens, F.; Cognie, J.; Abrami, R.; Oswald, I.P.; Guzylack-Piriou, L. Deoxynivalenol as a New Factor in the Persistence of Intestinal Inflammatory Diseases: An Emerging Hypothesis through Possible Modulation of Th17-Mediated Response. PLoS ONE 2013, 8, e53647. [Google Scholar] [CrossRef] [PubMed]
- Vandenbroucke, V.; Pasmans, F.; Martel, A.; Verbrugghe, E.; Goossens, J.; Van Deun, K.; Boyen, F.; De Backer, P.; Haesebrouck, F.; Croubels, S. Combined Effects of Deoxynivalenol and Salmonella Typhimurium on Intestinal Inflammation in the Pig; Mycotoxin Workshop; Ghent University: Ghent, Belgium, 2011; p. 22. [Google Scholar]
- Grenier, B.; Loureiro-Bracarense, A.-P.; Lucioli, J.; Pacheco, G.D.; Cossalter, A.-M.; Moll, W.-D.; Schatzmayr, G.; Oswald, I.P. Individual and combined effects of subclinical doses of deoxynivalenol and fumonisins in piglets. Mol. Nutr. Food Res. 2011, 55, 761–771. [Google Scholar] [CrossRef]
- Schoenborn, J.R.; Wilson, C.B. Regulation of Interferon-γ during innate and adaptive immune responses. Adv. Immunol. 2007, 96, 41–101. [Google Scholar]
- Liao, W.; Lin, J.-X.; Leonard, W.J. IL-2 family cytokines: New insights into the complex roles of IL-2 as a broad regulator of T helper cell differentiation. Curr. Opin. Immunol. 2011, 23, 598–604. [Google Scholar] [CrossRef]
- Stepanova, H.; Mensikova, M.; Chlebova, K.; Faldyna, M. CD4+ and γδTCR+ T lymphocytes are sources of interleukin-17 in swine. Cytokine 2012, 58, 152–157. [Google Scholar] [CrossRef]
- Mehta, A.K.; Gracias, D.T.; Croft, M. TNF activity and T cells. Cytokine 2018, 101, 14–18. [Google Scholar] [CrossRef]
- Gerner, W.; Käser, T.; Saalmüller, A. Porcine T lymphocytes and NK cells--an update. Dev. Comp. Immunol. 2009, 33, 310–320. [Google Scholar] [CrossRef]
- Swamy, H.V.L.N.; Smith, T.K.; MacDonald, E.J.; Karrow, N.A.; Woodward, B.; Boermans, H.J. Effects of feeding a blend of grains naturally contaminated with Fusarium mycotoxins on growth and immunological measurements of starter pigs, and the efficacy of a polymeric glucomannan mycotoxin adsorbent1. J. Anim. Sci. 2003, 81, 2792–2803. [Google Scholar] [CrossRef]
- Ferrari, L.; Cantoni, A.M.; Borghetti, P.; De Angelis, E.; Corradi, A. Cellular immune response and immunotoxicity induced by DON (deoxynivalenol) in piglets. Vet. Res. Commun. 2009, 33, 133–135. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.; Slight, S.R.; Khader, S.A. Th17 cytokines and vaccine-induced immunity. Semin. Immunopathol. 2010, 32, 79–90. [Google Scholar] [CrossRef] [PubMed]
- Vandenbroeck, K.; Nauwynck, H.; Vanderpooten, A.; Van Reeth, K.; Goddeeris, B.; Billiau, A. Recombinant porcine IFN-γ potentiates the secondary IgG and IgA responses to an inactivated suid Herpesvirus-1 vaccine and reduces postchallenge weight loss and fever in pigs. J. Interferon Cytokine Res. 2009, 18, 739–744. [Google Scholar] [CrossRef] [PubMed]
- Chin, K.L.; Anis, F.Z.; Sarmiento, M.E.; Norazmi, M.N.; Acosta, A. Role of Interferons in the Development of Diagnostics, Vaccines, and Therapy for Tuberculosis. J. Immunol. Res. 2017, 2017, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Aringer, M. Vaccination under TNF blockade—Less effective, but worthwhile. Arthritis Res. Ther. 2012, 14, 117. [Google Scholar] [CrossRef][Green Version]
- Priebe, G.P.; Walsh, R.L.; Cederroth, T.A.; Kamei, A.; Coutinho-Sledge, Y.S.; Goldberg, J.B.; Pier, G.B. IL-17 Is a Critical Component of Vaccine-induced Protection against Lung Infection by LPS-heterologous Strains of Pseudomonas aeruginosa. J. Immunol. 2008, 181, 4965. [Google Scholar] [CrossRef]
- Nygard, A.-B.; Jørgensen, C.B.; Cirera, S.; Fredholm, M. Selection of reference genes for gene expression studies in pig tissues using SYBR green qPCR. BMC Mol. Biol. 2007, 8, 67. [Google Scholar] [CrossRef]
- Pavlova, B.; Volf, J.; Ondrackova, P.; Matiasovic, J.; Stepanova, H.; Crhanova, M.; Karasova, D.; Faldyna, M.; Rychlik, I. SPI-1-encoded type III secretion system of Salmonella enterica is required for the suppression of porcine alveolar macrophage cytokine expression. Vet. Res. 2011, 42, 16. [Google Scholar] [CrossRef]
- Volf, J.; Boyen, F.; Faldyna, M.; Pavlova, B.; Navratilova, J.; Rychlik, I. Cytokine response of porcine cell lines to Salmonella enterica serovar Typhimurium and its hilA and ssrA mutants. Zoonoses Public Health 2007, 54, 286–293. [Google Scholar] [CrossRef]
- Zelnickova, P.; Matiasovic, J.; Pavlova, B.; Kudlackova, H.; Kovaru, F.; Faldyna, M. Quantitative nitric oxide production by rat, bovine and porcine macrophages. Nitric Oxide 2008, 19, 36–41. [Google Scholar] [CrossRef]
| Time of Stimulation | Mitogen | Control | DON 100 ng/mL | DON 10 ng/mL | DON 1 ng/mL |
|---|---|---|---|---|---|
| 18 h | ConA | 65.55 ± 23.47 | 39.31 ± 21.19 * | 54.05 ± 37.55 | 50.63 ± 28.83 |
| PHA | 9.77 ± 5.61 | 10.71 ± 5.78 | 5.69 ± 3.21 | 9.75 ± 8.32 | |
| PWM | 86.63 ± 39.08 | 47.47 ± 23.37 ** | 69.04 ± 42.59 | 69.67 ± 37.81 | |
| 5 days | ConA | 12.62 ± 11.31 | 8.74 ± 7.26 | 8.66 ± 10.51 | 8.93 ± 6.90 |
| PHA | 7.60 ± 6.06 | 6.74 ± 6.30 | 6.34 ± 3.97 | 10.08 ± 5.85 | |
| PWM | 13.37 ± 8.83 | 8.27 ± 6.54 * | 16.73 ± 15.84 | 18.19 ± 15.51 |
| Time of Stimulation | Control | DON 100 ng/mL | DON 10 ng/mL | DON 1 ng/mL |
|---|---|---|---|---|
| 18 h | 52.33 ± 18.4 | 73.11 ± 27.93 * | 56.25 ± 16.22 | 56.75 ± 12.06 |
| 5 days | 47.44 ± 17.88 | 48.44 ± 19.15 | 51.22 ± 17.54 | 48.56 ± 18.90 |
| Time of Exposure | Cytokine | Relative Expression of Cytokine mRNA | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| DON Concentration (Mean ± SD) | ||||||||||||
| Control 0 ng/mL | 100 ng/mL | 10 ng/mL | 1 ng/mL | |||||||||
| Mean | SD | Mean | SD | p Value | Mean | SD | p Value | Mean | SD | p Value | ||
| 18 h | IL-1β | 2.28 | ±1.23 | 3.14 | ±1.608 | * | 2.77 | ±1.85 | NS | 1.83 | ±1.20 | NS |
| IL-2 | 56.52 | ±16.77 | 49.76 | ±8.89 | NS | 45.14 | ±21.23 | * | 35.94 | ±20.87 | ** | |
| IL-8 | 9.56 | ±4.51 | 13.18 | ±6.40 | ** | 7.98 | ±3.02 | NS | 7.17 | ±3.52 | NS | |
| IL-17 | 3.41 | ±1.73 | 3.11 | ±1.25 | NS | 2.87 | ±1.51 | * | 2.62 | ±1.29 | ** | |
| IFNγ | 7.97 | ±3.44 | 7.47 | ±2.43 | NS | 7.01 | ±2.58 | ** | 6.96 | ±2.35 | NS | |
| TNFα | 1.78 | ±0.78 | 1.87 | ±0.68 | NS | 1.11 | ±0.53 | ** | 1.11 | ±0.62 | ** | |
| 5 days | IL-1β | 2.53 | ±2.91 | 0.98 | ±0.71 | ** | 1.12 | ±1.1 | ** | 1.28 | ±0.95 | ** |
| IL-2 | 49.22 | ±25.73 | 27.17 | ±12.1 | *** | 35.10 | ±21.5 | ** | 37.21 | ±17.11 | NS | |
| IL-8 | 13.05 | ±12.38 | 16.37 | ±15.55 | NS | 9.68 | ±9.05 | * | 9.37 | ±6.69 | * | |
| IL-17 | 2.48 | ±1.81 | 1.30 | ±0.56 | *** | 1.54 | ±0.63 | * | 1.41 | ±0.6 | * | |
| IFNγ | 9.60 | ±3.57 | 6.10 | ±2.11 | *** | 9.08 | ±3.59 | NS | 8.98 | ±4.56 | * | |
| TNFα | 2.34 | ±1.66 | 1.82 | ±1.26 | NS | 1.47 | ±0.95 | ** | 1.69 | ±1.18 | ** | |
| Gene | Forward 5′-3′ | Reverse 5′-3′ |
|---|---|---|
| HPRT | GAGCTACTGTAATGACCAGTCAACG | CCAGTGTCAATTATATCTTCAACAATCAA |
| IFNγ | TGCAGATCCAGCGCAAAGCCATCAG | TTGATGCTCTCTGGCCTTGGAACATAGTC |
| IL-1β | GGGACTTGAAGAGAGAAGTGG | CTTTCCCTTGATCCCTAAGGT |
| IL-8 | TGAAGAGAACTGAGAAGCAACAACAACAGCAG | TCTTGGGAGCCACGGAGAATGGGT |
| IL-17 | ACATGCTGAGGGAAGTTCTTGTC | ATCCTCGTCCCTGTCACTGC |
| TNFα | CCCCCAGAAGGAAGAGTTTC | CGGGCTTATCTGAGGTTTGA |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hlavová, K.; Štěpánová, H.; Šťastný, K.; Levá, L.; Hodkovicová, N.; Vícenová, M.; Matiašovic, J.; Faldyna, M. Minimal Concentrations of Deoxynivalenol Reduce Cytokine Production in Individual Lymphocyte Populations in Pigs. Toxins 2020, 12, 190. https://doi.org/10.3390/toxins12030190
Hlavová K, Štěpánová H, Šťastný K, Levá L, Hodkovicová N, Vícenová M, Matiašovic J, Faldyna M. Minimal Concentrations of Deoxynivalenol Reduce Cytokine Production in Individual Lymphocyte Populations in Pigs. Toxins. 2020; 12(3):190. https://doi.org/10.3390/toxins12030190
Chicago/Turabian StyleHlavová, Karolina, Hana Štěpánová, Kamil Šťastný, Lenka Levá, Nikola Hodkovicová, Monika Vícenová, Ján Matiašovic, and Martin Faldyna. 2020. "Minimal Concentrations of Deoxynivalenol Reduce Cytokine Production in Individual Lymphocyte Populations in Pigs" Toxins 12, no. 3: 190. https://doi.org/10.3390/toxins12030190
APA StyleHlavová, K., Štěpánová, H., Šťastný, K., Levá, L., Hodkovicová, N., Vícenová, M., Matiašovic, J., & Faldyna, M. (2020). Minimal Concentrations of Deoxynivalenol Reduce Cytokine Production in Individual Lymphocyte Populations in Pigs. Toxins, 12(3), 190. https://doi.org/10.3390/toxins12030190





