Antibody Microarray Immunoassay for Simultaneous Quantification of Multiple Mycotoxins in Corn Samples
Abstract
1. Introduction
2. Results and Discussion
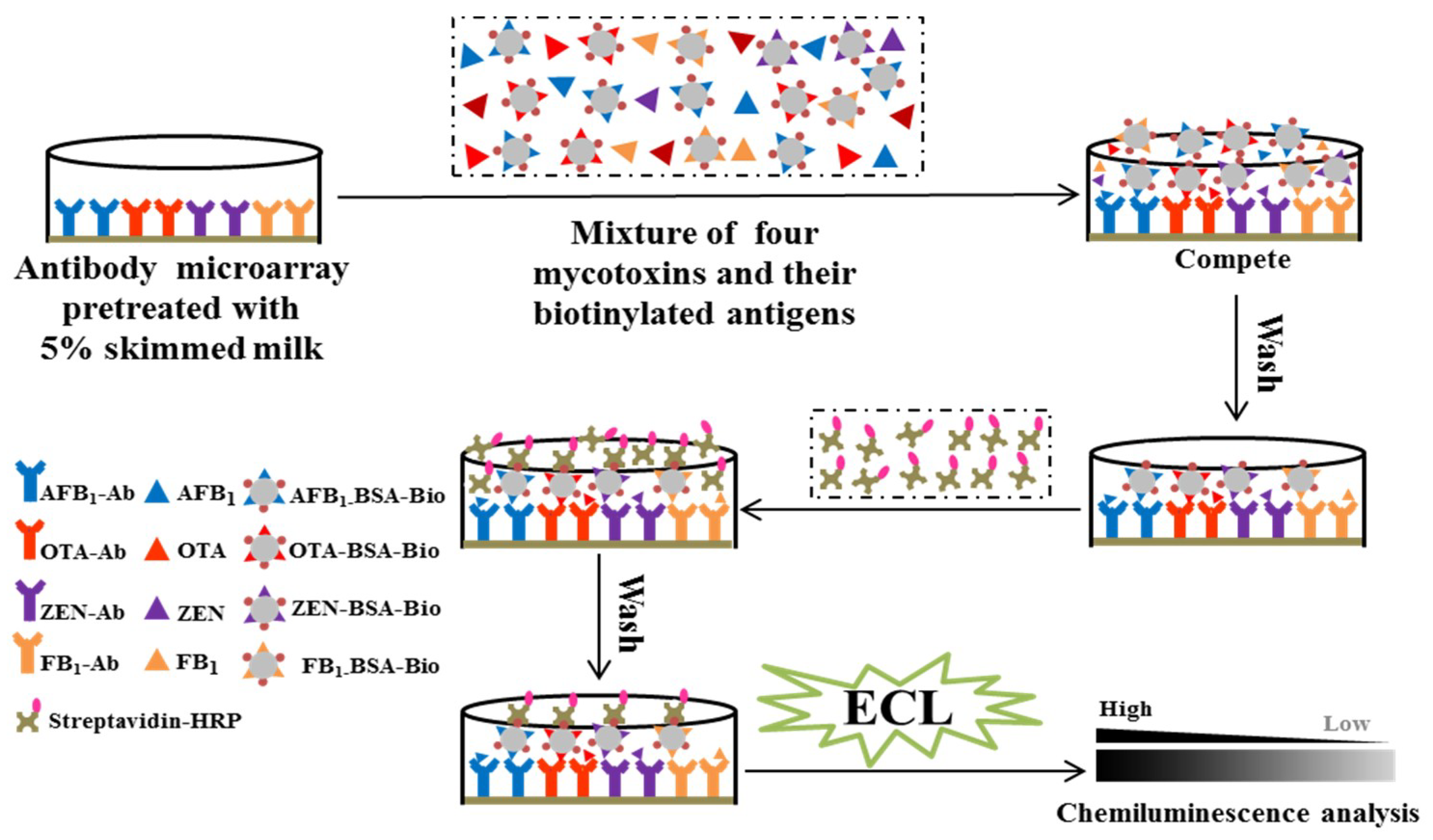
2.1. Development of Concept for Multiplex Antibody Microarray Immunoassay
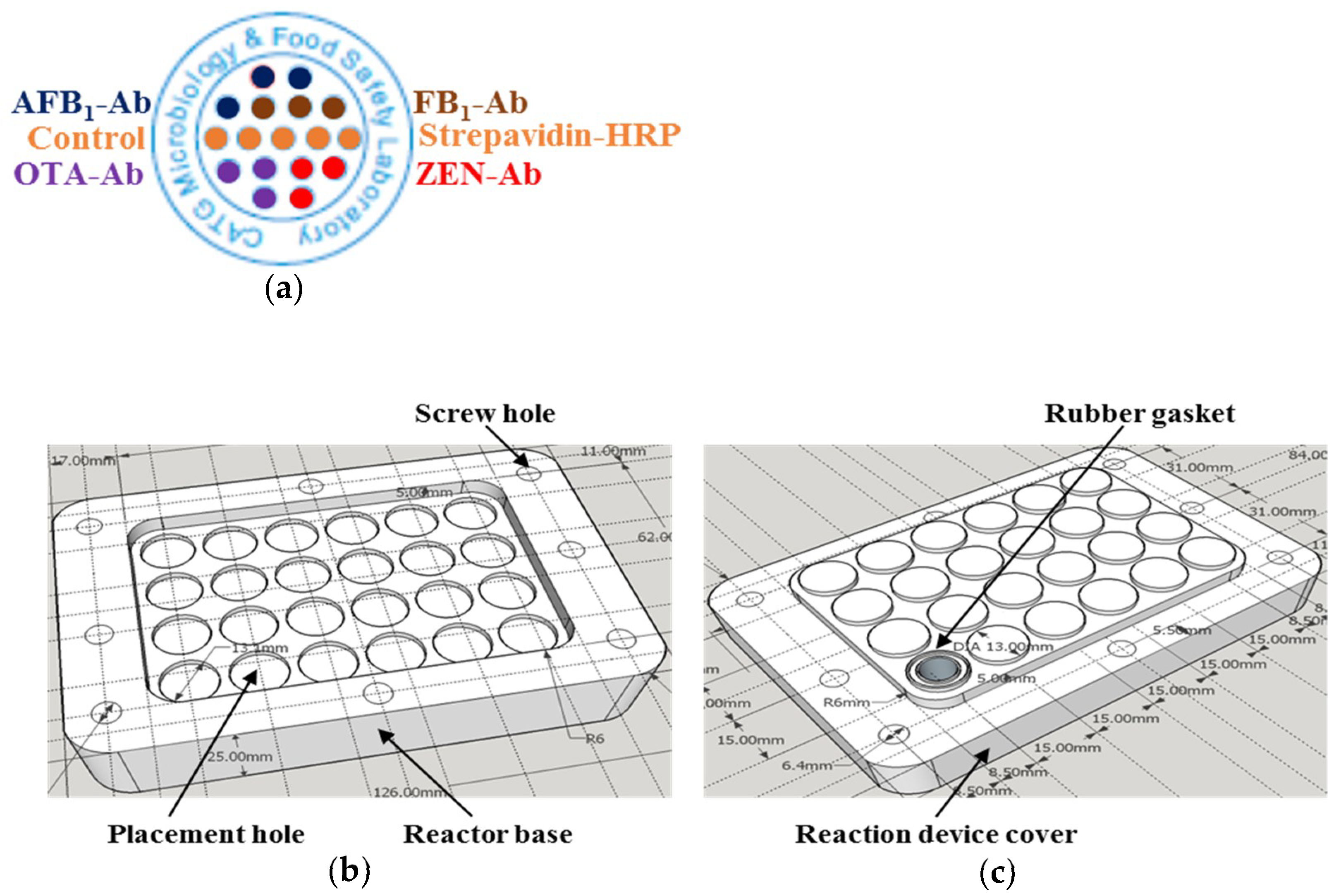
2.2. Preparation of the Hardware Prototype for Antibody Microarray Reaction
2.3. Evaluation of Specificity Among Ag-Ab Pairs for the Antibody Microarray
2.4. Calibration Curves for AFB1, OTA, ZEN, and FB1 Detection
2.5. Recovery Studies
2.6. Commercial Samples Analysis
3. Conclusions
4. Materials and Methods
4.1. Reagents
4.2. Apparatus
4.3. Biotinylation and Identification of Mycotoxin-Protein Conjugates
4.4. Antibody Microarray and Reaction Device Design
4.5. Preparation of the Antibody Microarray
4.6. Detection Procedure of the Antibody Microarray
4.7. Signal Acquisition and Data Analysis
4.8. Specificity Among the Four Antibodies and Biotinylated Conjugates
4.9. Recovery Studies
4.10. Detection of Commercial Samples by Antibody Microarray and LC-MS/MS.
Author Contributions
Funding
Conflicts of Interest
References
- Wang, G.; Lian, C.; Xi, Y.; Sun, Z.; Zheng, S. Evaluation of nonionic surfactant modified montmorillonite as mycotoxins adsorbent for aflatoxin B1 and zearalenone. J. Colloid Interface Sci. 2018, 518, 48–56. [Google Scholar] [CrossRef] [PubMed]
- Vila-Donat, P.; Marin, S.; Sanchis, V.; Ramos, A.J. A review of the mycotoxin adsorbing agents, with an emphasis on their multi-binding capacity, for animal feed decontamination. Food. Chem. Toxicol. 2018, 114, 246–259. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.D.; Abdul, N.S.; Phulukdaree, A.; Tiloke, C.; Nagiah, S.; Baijnath, S.; Chuturgoon, A.A. Toxicity assessment of mycotoxins extracted from contaminated commercial dog pelleted feed on canine blood mononuclear cells. Food. Chem. Toxicol. 2018, 114, 112–118. [Google Scholar] [CrossRef] [PubMed]
- Smith, M.C.; Madec, S.; Coton, E.; Hymery, N. Natural Co-Occurrence of Mycotoxins in Foods and Feeds and Their in vitro Combined Toxicological Effects. Toxins 2016, 8, 94. [Google Scholar] [CrossRef] [PubMed]
- Alassane-Kpembi, I.; Schatzmayr, G.; Taranu, I.; Marin, D.; Puel, O.; Oswald, I.P. Mycotoxins co-contamination: Methodological aspects and biological relevance of combined toxicity studies. Crit. Rev. Food Sci. Nutr. 2017, 57, 3489–3507. [Google Scholar] [CrossRef] [PubMed]
- Adekoya, I.; Obadina, A.; Adaku, C.C.; de Boevre, M.; Okoth, S.; de Saeger, S.; Njobeh, P. Mycobiota and co-occurrence of mycotoxins in South African maize-based opaque beer. Int. J. Food Microbiol. 2018, 270, 22–30. [Google Scholar] [CrossRef] [PubMed]
- Wei, Y.; Feng, M.; Zhu, Z.; He, J.; Shen, J.; He, Z.; Qin, X.; Zhou, L. Simultaneous determination of 11 mycotoxins in feeds by high performance liquid chromatography-tandem mass spectrometry. Se Pu 2017, 35, 891–896. [Google Scholar] [CrossRef] [PubMed]
- Abdallah, M.F.; Girgin, G.; Baydar, T.; Krska, R.; Sulyok, M. Occurrence of multiple mycotoxins and other fungal metabolites in animal feed and maize samples from Egypt using LC-MS/MS. J. Sci. Food Agric. 2017, 97, 4419–4428. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Carrasco, Y.; Molto, J.C.; Manes, J.; Berrada, H. Development of a GC-MS/MS strategy to determine 15 mycotoxins and metabolites in human urine. Talanta 2014, 128, 125–131. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Wen, K.; Mi, T.; Zhang, X.; Zhang, H.; Zhang, S.; Shen, J.; Wang, Z. A universal multi-wavelength fluorescence polarization immunoassay for multiplexed detection of mycotoxins in maize. Biosens. Bioelectron. 2016, 79, 258–265. [Google Scholar] [CrossRef] [PubMed]
- Oswald, S.; Dietrich, R.; Martlbauer, E.; Niessner, R.; Knopp, D. Microarray-Based Immunoassay for Parallel Quantification of Multiple Mycotoxins in Oat. Methods Mol. Biol. 2017, 1536, 143–156. [Google Scholar] [PubMed]
- Li, C.; Mi, T.; Conti, G.O.; Yu, Q.; Wen, K.; Shen, J.; Ferrante, M.; Wang, Z. Development of a screening fluorescence polarization immunoassay for the simultaneous detection of fumonisins B1 and B2 in maize. J. Agric. Food Chem. 2015, 63, 4940. [Google Scholar] [CrossRef] [PubMed]
- Goud, K.Y.; Kalisa, S.K.; Kumar, V.; Tsang, Y.F.; Lee, S.E.; Gobi, K.V.; Kim, K.H. Progress on nanostructured electrochemical sensors and their recognition elements for detection of mycotoxins: A review. Biosens. Bioelectron. 2018, 121, 205–222. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.K.; Yan, Y.X.; Li, S.Q.; Wang, H.A.; Ji, W.H.; Sun, J.H. Simultaneous quantitative determination of multiple mycotoxins in cereal and feedstuff samples by a suspension array immunoassay. J. Agric. Food Chem. 2013, 61, 10948–10953. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Ning, B.; Peng, Y.; Bai, J.; Liu, M.; Fan, X.; Sun, Z.; Lv, Z.; Zhou, C.; Gao, Z. Application of suspension array for simultaneous detection of four different mycotoxins in corn and peanut. Biosens. Bioelectron. 2013, 41, 391–396. [Google Scholar] [CrossRef] [PubMed]
- Peters, J.; Bienenmann-Ploum, M.; de Rijk, T.; Haasnoot, W. Development of a multiplex flow cytometric microsphere immunoassay for mycotoxins and evaluation of its application in feed. Mycotoxin Res. 2011, 27, 63–72. [Google Scholar] [CrossRef] [PubMed]
- Deng, G.; Xu, K.; Sun, Y.; Chen, Y.; Zheng, T.; Li, J. High sensitive immunoassay for multiplex mycotoxin detection with photonic crystal microsphere suspension array. Anal. Chem. 2013, 85, 2833–2840. [Google Scholar] [CrossRef] [PubMed]
- Kloth, K.; Rye-Johnsen, M.; Didier, A.; Dietrich, R.; Martlbauer, E.; Niessner, R.; Seidel, M. A regenerable immunochip for the rapid determination of 13 different antibiotics in raw milk. Analyst 2009, 134, 1433–1439. [Google Scholar] [CrossRef] [PubMed]
- Lan, M.; Guo, Y.; Zhao, Y.; Liu, Y.; Gui, W.; Zhu, G. Multi-residue detection of pesticides using a sensitive immunochip assay based on nanogold enhancement. Anal. Chim. Acta 2016, 938, 146–155. [Google Scholar]
- Chiriaco, M.S.; Primiceri, E.; de Feo, F.; Montanaro, A.; Monteduro, A.G.; Tinelli, A.; Megha, M.; Carati, D.; Maruccio, G. Simultaneous detection of multiple lower genital tract pathogens by an impedimetric immunochip. Biosens. Bioelectron. 2016, 79, 9–14. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Liu, N.; Ning, B.; Liu, M.; Lv, Z.; Sun, Z.; Peng, Y.; Chen, C.; Li, J.; Gao, Z. Simultaneous and rapid detection of six different mycotoxins using an immunochip. Biosens. Bioelectron. 2012, 34, 44–50. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Wang, X.; Sun, M.; Zhang, X.; Song, H.; Yan, Y.; Sun, J.; Li, X.; Fang, W. A Magnetic Nanoparticle Based Enzyme-Linked Immunosorbent Assay for Sensitive Quantification of Zearalenone in Cereal and Feed Samples. Toxins 2015, 7, 4216–4231. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Sun, M.; Kang, Y.; Xie, H.; Wang, X.; Song, H.; Li, X.; Fang, W. Identification of a high-affinity monoclonal antibody against ochratoxin A and its application in enzyme-linked immunosorbent assay. Toxicon 2015, 106, 89–96. [Google Scholar] [CrossRef] [PubMed]
- Mercader, J.V.; Montoya, A. Development of monoclonal ELISAs for azinphos-methyl. 2. Assay optimization and water sample analysis. J. Agric. Food Chem. 1999, 47, 1285–1293. [Google Scholar] [PubMed]
- Chun, H.S.; EChoi, H.; Chang, H.J.; Choi, S.W.; Eremin, S.A. A fluorescence polarization immunoassay for the detection of zearalenone in corn. Anal. Chim. Acta 2009, 639, 83–89. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.K.; Yan, Y.X.; Ji, W.H.; Wang, H.A.; Zou, Q.; Sun, J.H. Novel chemiluminescence immunoassay for the determination of zearalenone in food samples using gold nanoparticles labeled with streptavidin-horseradish peroxidase. J. Agric. Food Chem. 2013, 61, 4250–4256. [Google Scholar] [CrossRef] [PubMed]
- Sergeyeva, T.; Yarynka, D.; Piletska, E.; Lynnik, R.; Zaporozhets, O.; Brovko, O.; Piletsky, S.; El’skaya, A. Fluorescent sensor systems based on nanostructured polymeric membranes for selective recognition of Aflatoxin B1. Talanta 2017, 175, 101–107. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Wen, F.; Li, M.; Guo, X.; Li, S.; Zheng, N.; Wang, J. A simple aptamer-based fluorescent assay for the detection of Aflatoxin B1 in infant rice cereal. Food Chem. 2017, 215, 377–382. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Tan, R.; Chen, D. Fluorescence method for quickly detecting ochratoxin A in flour and beer using nitrogen doped carbon dots and silver nanoparticles. Talanta 2018, 182, 363–370. [Google Scholar] [CrossRef] [PubMed]
- Zhang, G.; Zhu, C.; Huang, Y.; Yan, J.; Chen, A. A Lateral Flow Strip Based Aptasensor for Detection of Ochratoxin A in Corn Samples. Molecules 2018, 23, 291. [Google Scholar] [CrossRef] [PubMed]
- Hu, S.; Ouyang, W.; Guo, L.; Lin, Z.; Jiang, X.; Qiu, B.; Chen, G. Facile synthesis of Fe3O4/g-C3N4/HKUST-1 composites as a novel biosensor platform for ochratoxin A. Biosens. Bioelectron. 2017, 92, 718–723. [Google Scholar] [CrossRef] [PubMed]
- Dong, G.; Pan, Y.; Wang, Y.; Ahmed, S.; Liu, Z.; Peng, D.; Yuan, Z. Preparation of a broad-spectrum anti-zearalenone and its primary analogues antibody and its application in an indirect competitive enzyme-linked immunosorbent assay. Food Chem. 2018, 247, 8–15. [Google Scholar] [CrossRef] [PubMed]
- Ji, J.; Gu, W.; Sun, C.; Sun, J.; Jiang, H.; Zhang, Y.; Sun, X. A novel recombinant cell fluorescence biosensor based on toxicity of pathway for rapid and simple evaluation of DON and ZEN. Sci Rep. 2016, 6, 31270. [Google Scholar] [CrossRef] [PubMed]
- Goud, K.Y.; Hayat, A.; Satyanarayana, M.; Kumar, V.S.; Catanante, G.; Gobi, K.V.; Marty, J.L. Aptamer-based zearalenone assay based on the use of a fluorescein label and a functional graphene oxide as a quencher. Microchim. Acta 2017, 184, 4401–4408. [Google Scholar] [CrossRef]
- Peltomaa, R.; Benito-Pena, E.; Barderas, R.; Sauer, U.; Andrade, M.G.; Moreno-Bondi, M.C. Microarray-Based Immunoassay with Synthetic Mimotopes for the Detection of Fumonisin B1. Anal. Chem. 2017, 89, 6216–6223. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.C.; Bao, M.; Li, F.H.; Fan, H.X.; Li, H.S.; Li, Y.; Feng, S.B.; Wu, J.J. Development of a sensitive, competitive, indirect ELISA for the detection of fumonisin B1 in corn originating from Anhui province, China. J. Environ. Sci. Health B 2016, 51, 107–112. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.; Zhang, Z.; Zhang, Q.; Zhang, W.; Yu, L.; Wang, D.; Li, H.; Li, P. An On-Site Simultaneous Semi-quantification of Aflatoxin B1, Zearalenone, and T-2 Toxin in Maize- and Cereal-based Feed via Multicolor Immunochromatographic Assay. Toxins 2018, 10, 87. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.K.; Yan, Y.X.; Ji, W.H.; Wang, H.A.; Li, S.Q.; Zou, Q.; Sun, J.H. Rapid simultaneous quantification of zearalenone and fumonisin B1 in corn and wheat by lateral flow dual immunoassay. J. Agric. Food Chem. 2013, 61, 5031–5036. [Google Scholar] [CrossRef] [PubMed]
- Goud, K.Y.; Moru, D.S.; Gobi, K.V.; Catanante, G.; Marty, J.L. An electrochemical aptasensor based on functionalized graphene oxide assisted electrocatalytic signal amplification of methylene blue for aflatoxin B1 detection. Electrochim. Acta 2017, 244, 96–103. [Google Scholar] [CrossRef]
- Taghdisi, S.M.; Danesh, N.M.; Ramezani, M.; Abnous, K. A new amplified fluorescent aptasensor based on hairpin structure of G-quadruplex oligonucleotide-Aptamer chimera and silica nanoparticles for sensitive detection of aflatoxin B1 in the grape juice. Food Chem. 2018, 268, 342–346. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Wang, Z.; Xie, H.; Sun, R.; Cao, T.; Paudyal, N.; Fang, W.; Song, H. Development of a Magnetic Nanoparticles-Based Screen-Printed Electrodes (MNPs-SPEs) Biosensor for the Quantification of Ochratoxin A in Cereal and Feed Samples. Toxins 2018, 10, 317. [Google Scholar] [CrossRef] [PubMed]
- Liu, F.; Ding, A.; Zheng, J.; Chen, J.; Wang, B. A Label-Free Aptasensor for Ochratoxin a Detection Based on the Structure Switch of Aptamer. Sensors 2018, 18, 1769. [Google Scholar] [CrossRef] [PubMed]
- Foubert, A.; Beloglazova, N.V.; Hedstrom, M.; de Saeger, S. Antibody immobilization strategy for the development of a capacitive immunosensor detecting zearalenone. Talanta 2019, 191, 202–208. [Google Scholar] [CrossRef] [PubMed]
- Jie, M.; Yu, S.; Yu, F.; Liu, L.; He, L.; Li, Y.; Zhang, H.; Qu, L.; Harrington, P.B.; Wu, Y. An ultrasensitive chemiluminescence immunoassay for fumonisin B1 detection in cereals based on gold-coated magnetic nanoparticles. J. Sci. Food Agric. 2018, 98, 3384–3390. [Google Scholar] [CrossRef] [PubMed]
- Soleimany, F.; Jinap, S.; Abas, F. Determination of mycotoxins in cereals by liquid chromatography tandem mass spectrometry. Food Chem. 2012, 130, 1055–1060. [Google Scholar] [CrossRef]



| Mycotoxins | Concentration of the Antibody (μg/mL) | Concentration of the Biotinylated Antigen (μg/mL) |
|---|---|---|
| AFB1 | 25 | 0.2 |
| OTA | 25 | 0.1 |
| ZEN | 50 | 0.4 |
| FB1 | 12.5 | 0.4 |
| Mycotoxins | LOD (ng/mL) | IC50 (ng/mL) | Detection Range (IC20-IC80, ng/mL) | Regression Equation |
|---|---|---|---|---|
| AFB1 | 0.21 | 5.12 | 0.47–55.69 | y = 0.2895x + 0.2946 (R2 = 0.9806) |
| OTA | 0.19 | 7.82 | 0.48–127.11 | y = 0.2477x + 0.2788 (R2 = 0.9887) |
| ZEN | 0.09 | 2.62 | 0.22–31.36 | y = 0.2782x + 0.3837 (R2 = 0.9810) |
| FB1 | 0.24 | 7.23 | 0.56–92.57 | y = 0.2709x + 0.2673 (R2 = 0.9936) |
| Samples | Concentrations (μg/kg) | Recovery Rate (%) (Mean ± SD a) | CV b (%) | |
|---|---|---|---|---|
| Spiked | Detected | |||
| AFB1 | 20 | 17.27 | 86.3 ± 6.4 | 7.4 |
| 40 | 31.68 | 79.2 ± 4.7 | 5.9 | |
| 60 | 54.87 | 91.5 ± 7.1 | 7.8 | |
| OTA | 20 | 16.71 | 83.6 ± 8.1 | 9.7 |
| 40 | 34.87 | 87.2 ± 7.6 | 8.7 | |
| 60 | 49.73 | 82.9 ± 6.8 | 8.2 | |
| ZEN | 50 | 45.86 | 91.7 ± 8.9 | 9.7 |
| 100 | 89.13 | 89.1 ± 5.7 | 6.4 | |
| 200 | 213.07 | 106.5 ± 7.3 | 6.9 | |
| FB1 | 200 | 189.27 | 94.6 ± 9.1 | 9.6 |
| 400 | 427.62 | 106.9 ± 7.8 | 7.3 | |
| 800 | 907.21 | 113.4 ± 8.6 | 7.6 | |
| Samples | Antibody Microarray (μg/kg, Mean ± SD a) | LC-MS/MS (μg/kg, Mean ± SD) | ||||||
|---|---|---|---|---|---|---|---|---|
| AFB1 | OTA | ZEN | FB1 | AFB1 | OTA | ZEN | FB1 | |
| Corn 1 | - b | 12.31 ± 2.13 | - | - | - | 14.87 ± 1.21 | - | - |
| Corn 2 | - | - | 95.37 ± 8.73 | 724.53 ± 57.29 | - | - | 109.14 ± 8.16 | 688.47 ± 32.07 |
| Corn 3 | - | - | 72.85 ± 5.26 | 920.76 ± 64.79 | - | - | 85.03 ± 7.39 | 846.59 ± 50.61 |
| Corn 4 | - | 27.64 ± 2.49 | - | - | - | 32.36 ± 2.14 | - | - |
| Corn 7 | - | 20.86 ± 2.43 | - | 593.71 ± 41.15 | - | 25.57 ± 2.71 | - | 542.04 ± 38.32 |
| Corn 11 | - | 22.59 ± 2.04 | - | - | - | 26.71 ± 2.13 | - | - |
| Wheat 1 | - | 16.76 ± 1.36 | - | - | - | 20.99 ± 2.71 | - | - |
| Wheat 6 | - | - | 29.78 ± 2.54 | 811.23 ± 62.15 | - | - | 34.21 ± 2.33 | 741.48 ± 40.32 |
| Wheat 7 | - | 42.33 ± 3.89 | - | - | - | 49.55 ± 3.23 | - | - |
| Wheat 11 | 8.79 ± 1.05 | - | 13.96 ± 1.19 | - | 10.11 ± 1.62 | - | 16.67 ± 1.35 | - |
| Feed 2 | 23.85 ± 2.68 | - | 15.97 ± 1.34 | 398.92 ± 35.67 | 27.52 ± 1.84 | - | 19.35 ± 2.17 | 347.54 ± 26.38 |
| Feed 3 | - | - | 14.69 ± 1.37 | 321.27 ± 28.94 | - | - | 17.52 ± 1.34 | 299.93 ± 21.15 |
| Feed 6 | - | - | 11.38 ± 1.67 | - | - | - | 14.84 ± 1.89 | - |
| Feed 7 | - | 39.41 ± 2.97 | - | 713.25 ± 61.68 | - | 45.62 ± 3.12 | - | 667.06 ± 48.81 |
| Feed 11 | - | - | 26.72 ± 2.51 | 684.82 ± 46.39 | - | - | 30.34 ± 2.19 | 631.28 ± 37.43 |
| Feed 12 | - | - | 21.85 ± 2.62 | 461.56 ± 35.73 | - | - | 27.03 ± 2.31 | 420.79 ± 22.27 |
| Method | Target Analyte | LOD (ng/mL) | Ref. |
|---|---|---|---|
| Fluorescent sensor | AFB1 | 14 | [27] |
| Aptamer-based fluorescent assay | AFB1 | 1.6 | [28] |
| FRET based fluorescence | OTA | 3.5 | [29] |
| Lateral flow strip based aptasensor | OTA | 0.40 | [30] |
| Novel biosensor platform | OTA | 2.57 | [31] |
| ic-ELISA | ZEN | 0.114 | [32] |
| Aptamer-based fluorescence assay | ZEN | 0.5 | [34] |
| Microarray-Based Immunoassay | FB1 | 11.1 | [35] |
| Ic-ELISA | FB1 | 1.15 | [36] |
| Cell fluorescence biosensor | ZEN and DON | ZEN: 3.2 | [33] |
| Lateral Flow Dual Immunoassay | ZEN and FB1 | ZEN: 0.35, FB1: 5.23 | [38] |
| Multicolor ICGA | AFB1, ZEN, and T-2 | AFB1: 0.5, ZEN: 2 | [37] |
| Suspension Array | AFB1, ZEN, DON, and FB1 | AFB1: 0.56, ZEN: 0.51, FB1: 6.0 | [14] |
| Microarray immunoassay | AFB1, OTA, ZEN, and FB1 | AFB1: 0.21, OTA: 0.19, ZEN: 0.09, FB1: 0.24 | This work |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, X.; Wang, Z.; Fang, Y.; Sun, R.; Cao, T.; Paudyal, N.; Fang, W.; Song, H. Antibody Microarray Immunoassay for Simultaneous Quantification of Multiple Mycotoxins in Corn Samples. Toxins 2018, 10, 415. https://doi.org/10.3390/toxins10100415
Zhang X, Wang Z, Fang Y, Sun R, Cao T, Paudyal N, Fang W, Song H. Antibody Microarray Immunoassay for Simultaneous Quantification of Multiple Mycotoxins in Corn Samples. Toxins. 2018; 10(10):415. https://doi.org/10.3390/toxins10100415
Chicago/Turabian StyleZhang, Xian, Zuohuan Wang, Yun Fang, Renjie Sun, Tong Cao, Narayan Paudyal, Weihuan Fang, and Houhui Song. 2018. "Antibody Microarray Immunoassay for Simultaneous Quantification of Multiple Mycotoxins in Corn Samples" Toxins 10, no. 10: 415. https://doi.org/10.3390/toxins10100415
APA StyleZhang, X., Wang, Z., Fang, Y., Sun, R., Cao, T., Paudyal, N., Fang, W., & Song, H. (2018). Antibody Microarray Immunoassay for Simultaneous Quantification of Multiple Mycotoxins in Corn Samples. Toxins, 10(10), 415. https://doi.org/10.3390/toxins10100415





