Meta-Analysis of the Association between Vitamin D and Autoimmune Thyroid Disease
Abstract
:1. Introduction
2. Methods
2.1. Bibliographic Search
2.2. Eligibility Criteria and Excluded Studies
2.3. Data Extraction
2.4. Statistical Method
3. Results

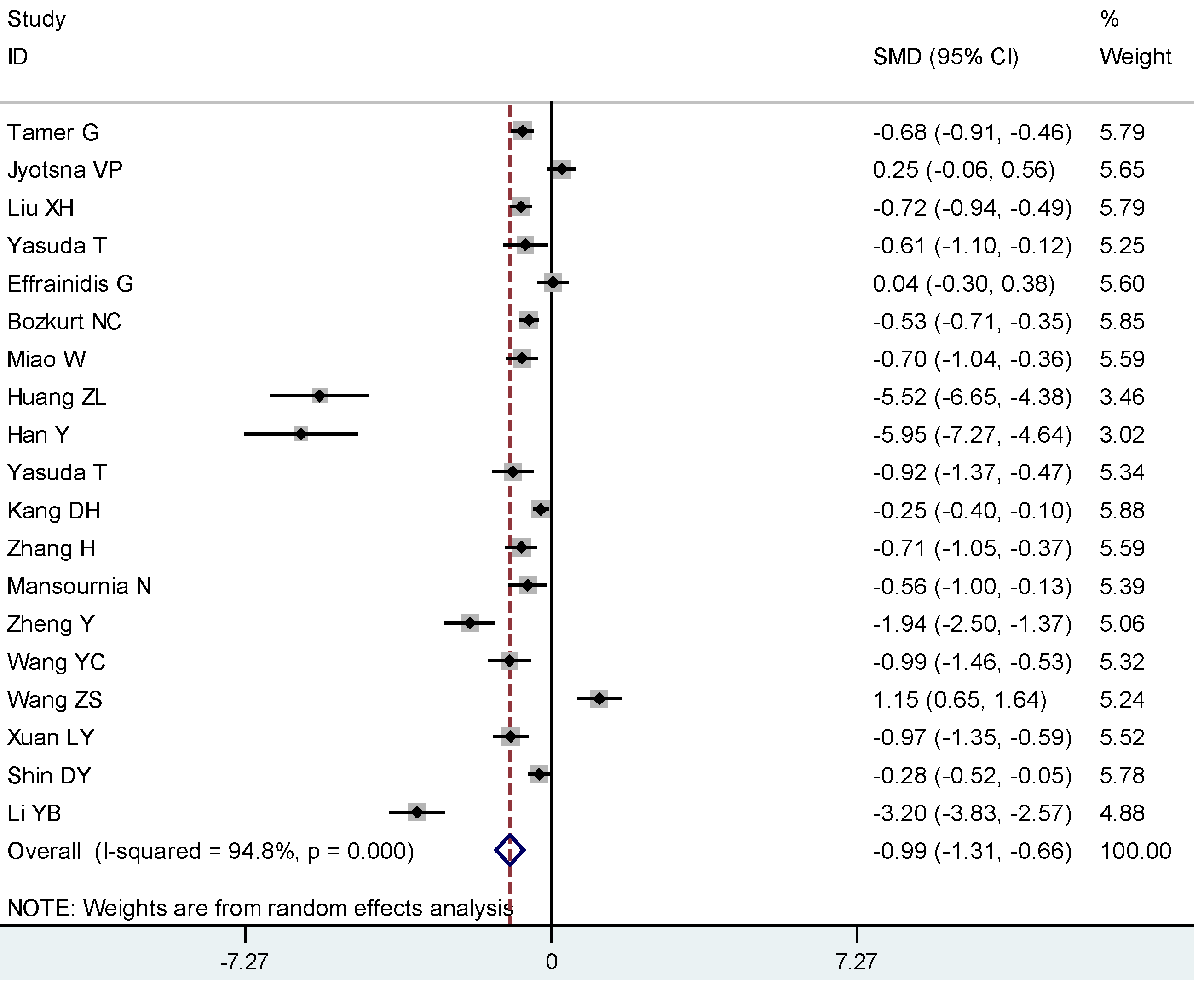
| First Author and Year | AITD (N)/Total (N) | AITD, males, % | AITD, Year, mean ± SD | Assay Method | Season of Collected Samples | 25(OH)D in AITD, ng/mL mean ± SD | 25(OH)D in Control, ng/mL mean ± SD | p-Value | Quality of Study (Score) |
|---|---|---|---|---|---|---|---|---|---|
| Yusuda T 2013 | 36/85 | 0 | 37.8 ± 8.1 | CPBA | Sum, A | 14.5 ± 2.9 | 18.6 ± 5.3 | <0.0005 | 8 |
| Tamer G 2011 | 161/323 | 6/161 | 35.4 ± 7.9 | RIA | W | 16.3 ± 10.4 | 29.6 ± 25.5 | <0.0001 | 9 |
| Yusuda T 2012 | 26/72 | 0 | 37.3 ± 13.0 | CPBA | W, S | 14.4 ± 4.9 | 17.1 ± 4.1 | <0.05 | 8 |
| Bozkurt NC 2013 | 360/540 | 114/360 | 42.55 ± 11.35 | ELISA | Sum | 12.2 ± 5.6 | 15.4 ± 6.8 | <0.001 | 8 |
| Effraimidis G 2012 | 67/134 | NG | 38.3 ± 11.5 | RIA | ALL | 21.6 ± 9.2 | 21.2 ± 9.3 | NS | 8 |
| Han Y 2013 | 30/50 | 6/30 | 35.7 ± 7.3 | HPLC | W, S | 17.51 ± 6.14 | 58.84 ± 8.01 | <0.01 | 7 |
| Miao W 2013 | 70/140 | 22/70 | 40 ± 15.2 | ECLIA | W, S | 12.7 ± 5.25 | 16.56 ± 5.8 | <0.01 | 9 |
| Huang ZL 2013 | 40/60 | 6/40 | 44.6 ± 8.5 | ECLIA | S, A | 16.26 ± 4.16 | 49.5 ± 8.68 | <0.01 | 8 |
| Liu XH 2012 | 160/325 | 25/160 | 43.25 ± 8.55 | ECLIA | W, S, Sum | 13.51 ± 5.88 | 19.48 ± 10.12 | <0.05 | 8 |
| Xuan LY 2014 | 89/134 | 32/89 | 33.92 ± 12.70 | ELISA | ALL | 19.04 ± 9.72 | 29.95 ± 13.86 | <0.01 | 7 |
| Shin DY 2014 | 111/304 | 21/111 | 48.7 ± 12.7 | RIA | ALL | 12.6 ± 5.5 | 14.5 ± 7.3 | <0.001 | 8 |
| Li YB 2014 | 40/90 | 0 | 34 ± 14 | ELISA | W, S | 13 ± 5 | 29 ± 5 | <0.05 | 8 |
| Zhang H 2014 | 70/140 | 28/70 | 31.77 ± 10.32 | ELISA | S | 21.15 ± 4.41 | 24.28 ± 4.37 | <0.05 | 8 |
| Jyotsna VP 2012 | 80/160 | 18/80 | 36.33 ± 11.15 | RIA | ALL | 12.67 ± 6.24 | 10.99 ± 7.05 | <0.05 | 7 |
| Mansournia N 2014 | 41/86 | NG | 42.3 ± 15.3 | HPLC | A | 15.9 ± 12.1 | 24.4 ± 17.3 | <0.01 | 8 |
| Zheng Y 2014 | 33/72 | 14/33 | 35.3 ± 9.23 | ELISA | ALL | 15.71 ± 6.79 | 30.84 ± 8.57 | <0.01 | 7 |
| Wang YC 2014 | 60/90 | 22/60 | 35.1 ± 7.95 | ECLIA | W, S, Sum | 12.28 ± 5.83 | 18.1 ± 5.92 | <0.01 | 7 |
| Kang DH 2013 | 280/719 | 100/280 | 42.5 ± 7.9 | ELISA | A | 21.68 ± 9.54 | 24.05 ± 9.58 | <0.01 | 7 |
| Wang ZS 2014 | 28/79 | 0 | NG | ECLIA | ALL | 26.98 ± 9.02 | 19.05 ± 5.47 | <0.01 | 7 |
| First Author and Year | AITD(N)/Total (N) | AITD, Males, % | AITD, year (Mean or Range) | Assay Method | Season of Collected Samples | 25(OH)D Deficiency in AITD (N) | 25(OH)D Deficiency in Control (N) | Criterion of 25(OH)D Deficiency | p-Value | Quality of Study (Score) |
|---|---|---|---|---|---|---|---|---|---|---|
| Yusuda T 2012 | 26/72 | 0 | 37.3 ± 13.0 | CPBA | W, S | 17 | 15 | <15 ng/mL | <0.05 | 8 |
| Bozkurt NC 2013 | 360/540 | 57/180 | 42.55 ± 11.35 | ELISA | Sum | 150 | 37 | <10 ng/mL | <0.001 | 8 |
| Effraimidis G 2012 | 67/134 | NG | 38.3 ± 11.5 | RIA | ALL | 33 | 23 | <20 ng/mL | =0.05 | 8 |
| HanY 2013 | 30/50 | 6/30 | 35.7 ± 7.3 | HPLC | W, S | 16 | 0 | <20 ng/mL | <0.01 | 7 |
| Miao W 2013 | 70/140 | 22/70 | 40 ± 15.2 | ECLIA | W, S | 65 | 54 | <20 ng/mL | <0.05 | 9 |
| Kivity S 2011 | 50/148 | 6/50 | 45 ± 16 | DCCLIA | S | 35 | 37 | <10 ng/mL | <0.001 | 8 |
| Zhang H 2014 | 70/140 | 28/70 | 31.77 ± 10.32 | ELISA | S | 30 | 10 | <20 ng/mL | <0.05 | 8 |
| Kang DH 2013 | 280/719 | 100/280 | 42.5 ± 7.9 | ELISA | A | 133 | 158 | <20 ng/mL | <0.01 | 7 |
| Mansourria N 2014 | 41/86 | NG | 42.3 ± 15.3 | HPLC | A | 34 | 24 | <20 ng/mL | 0.82 | 8 |
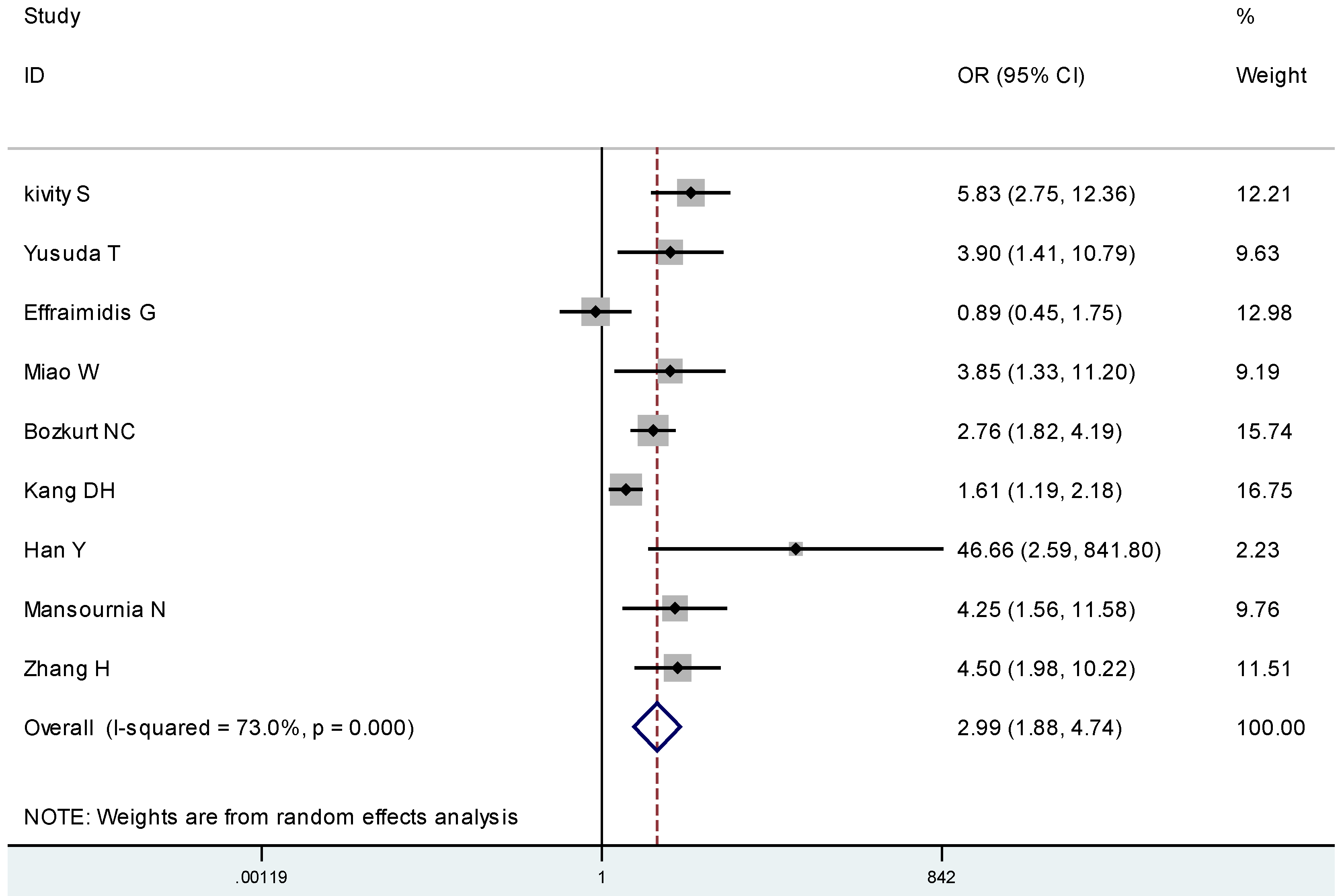
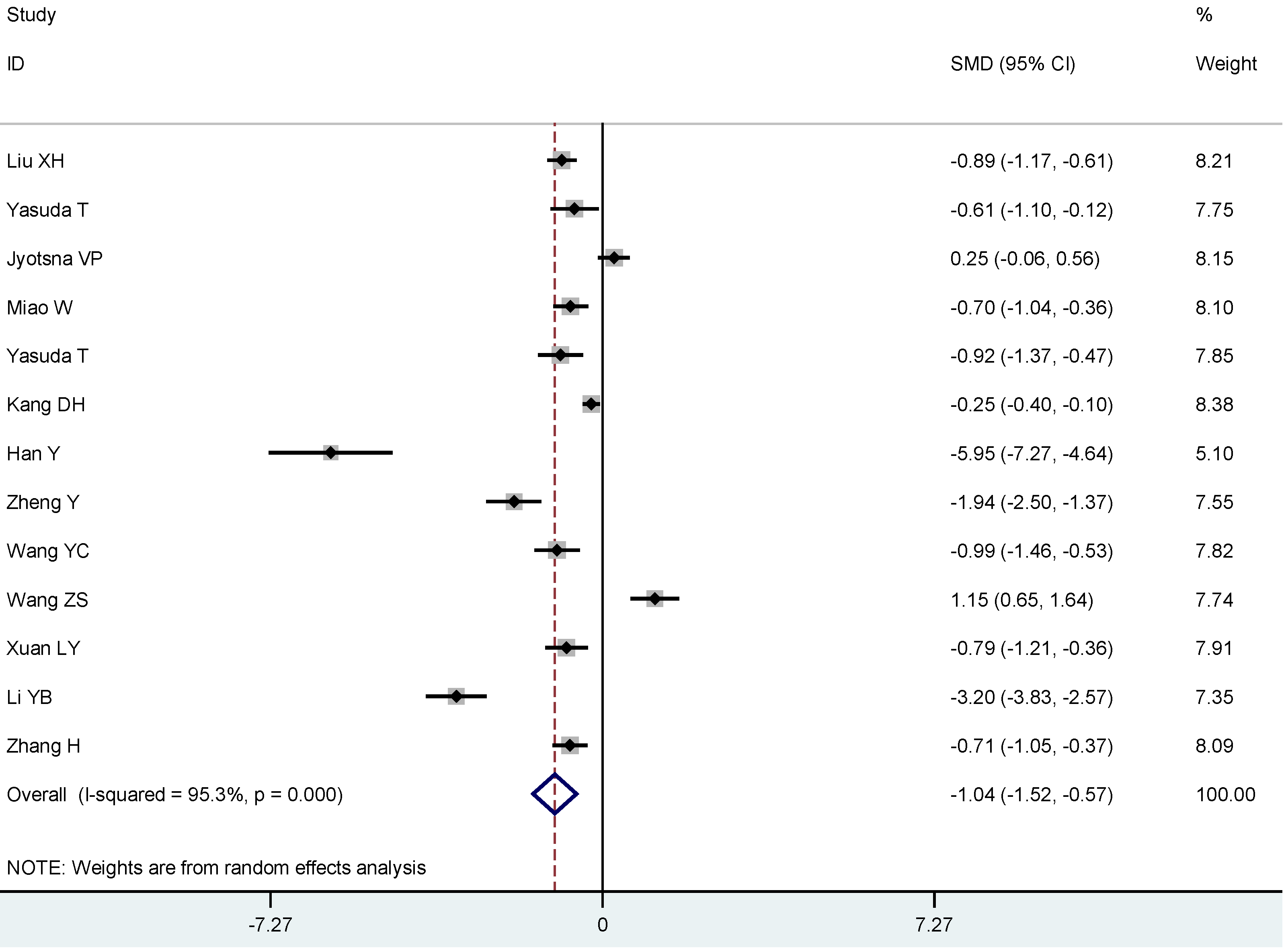
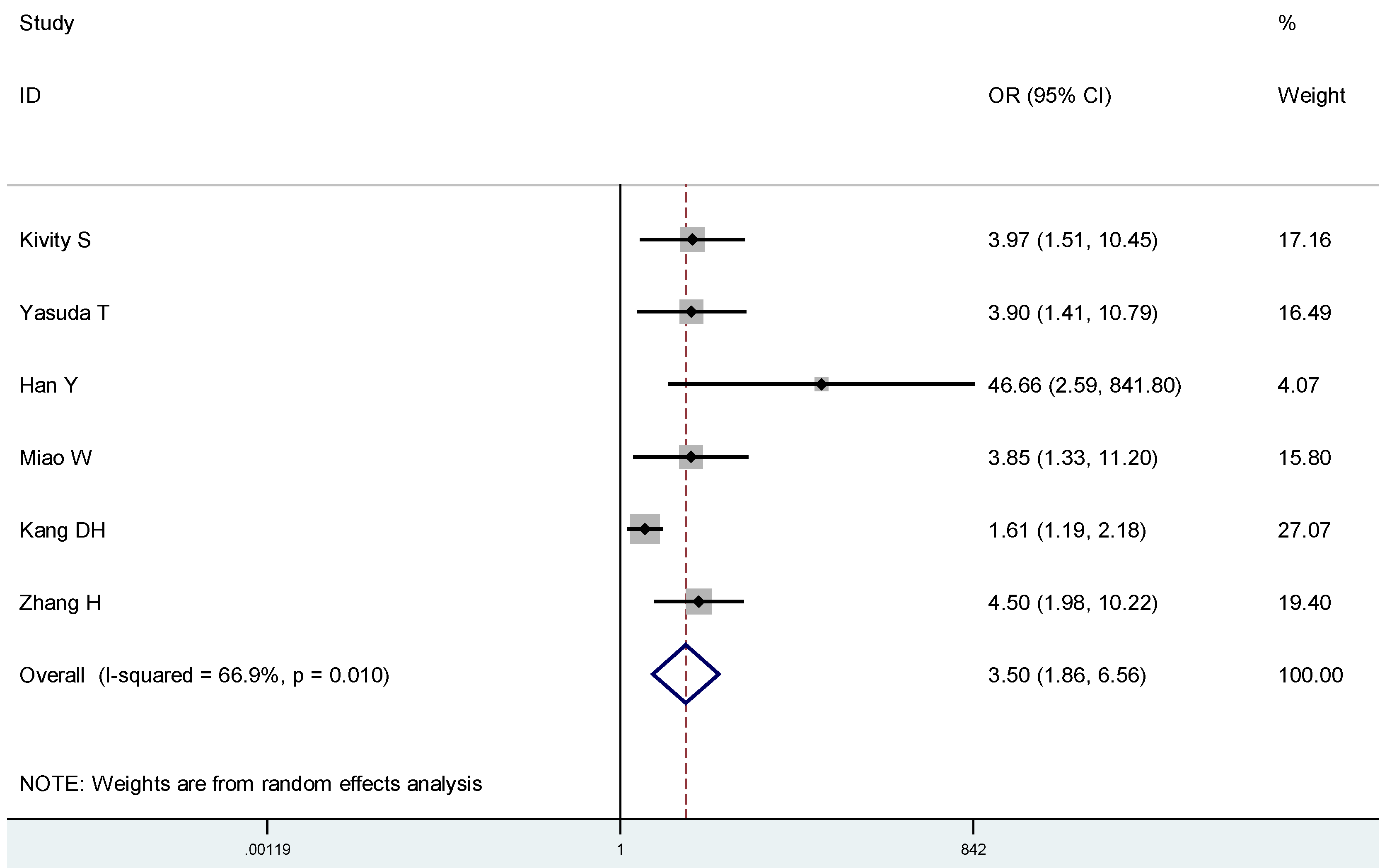
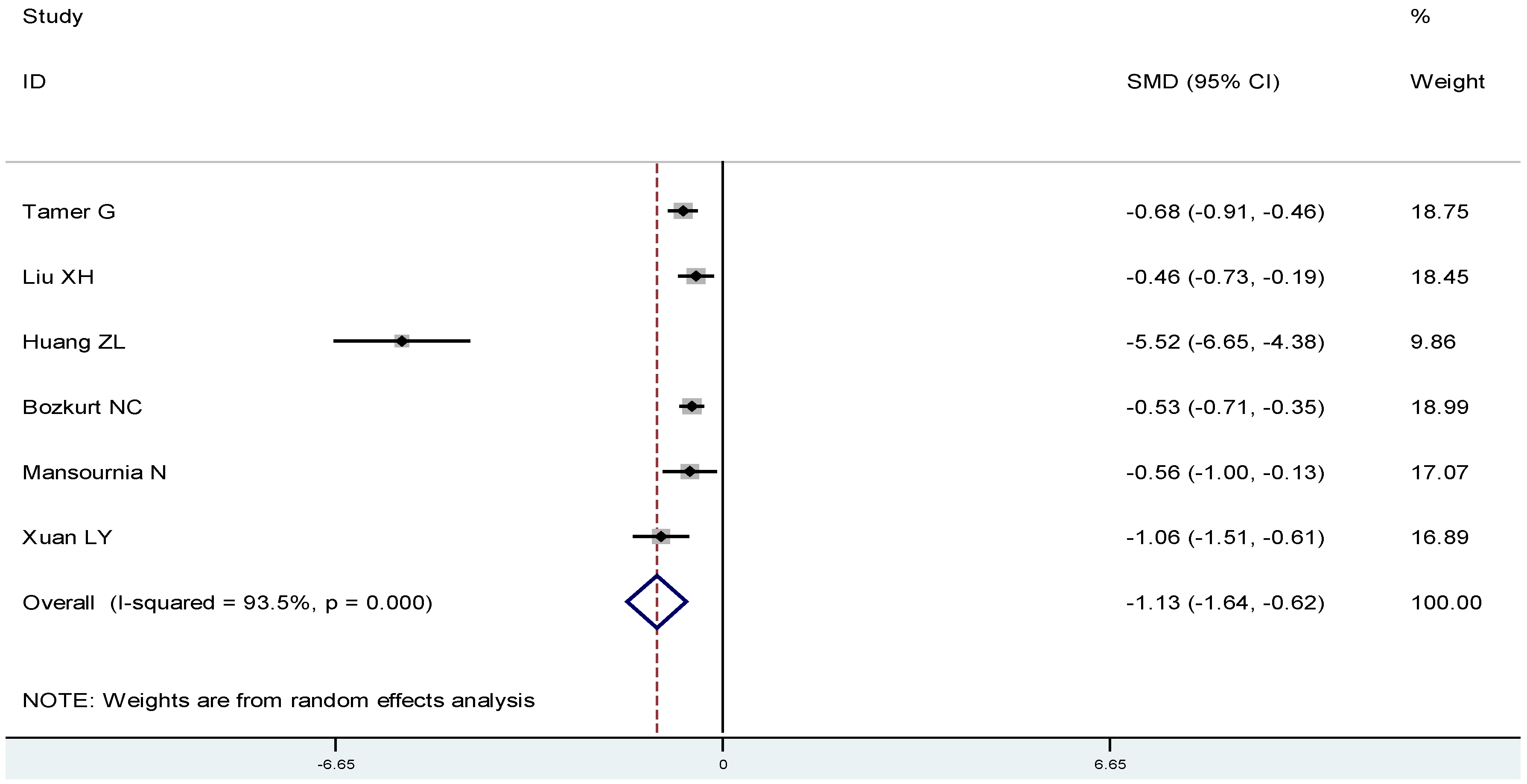
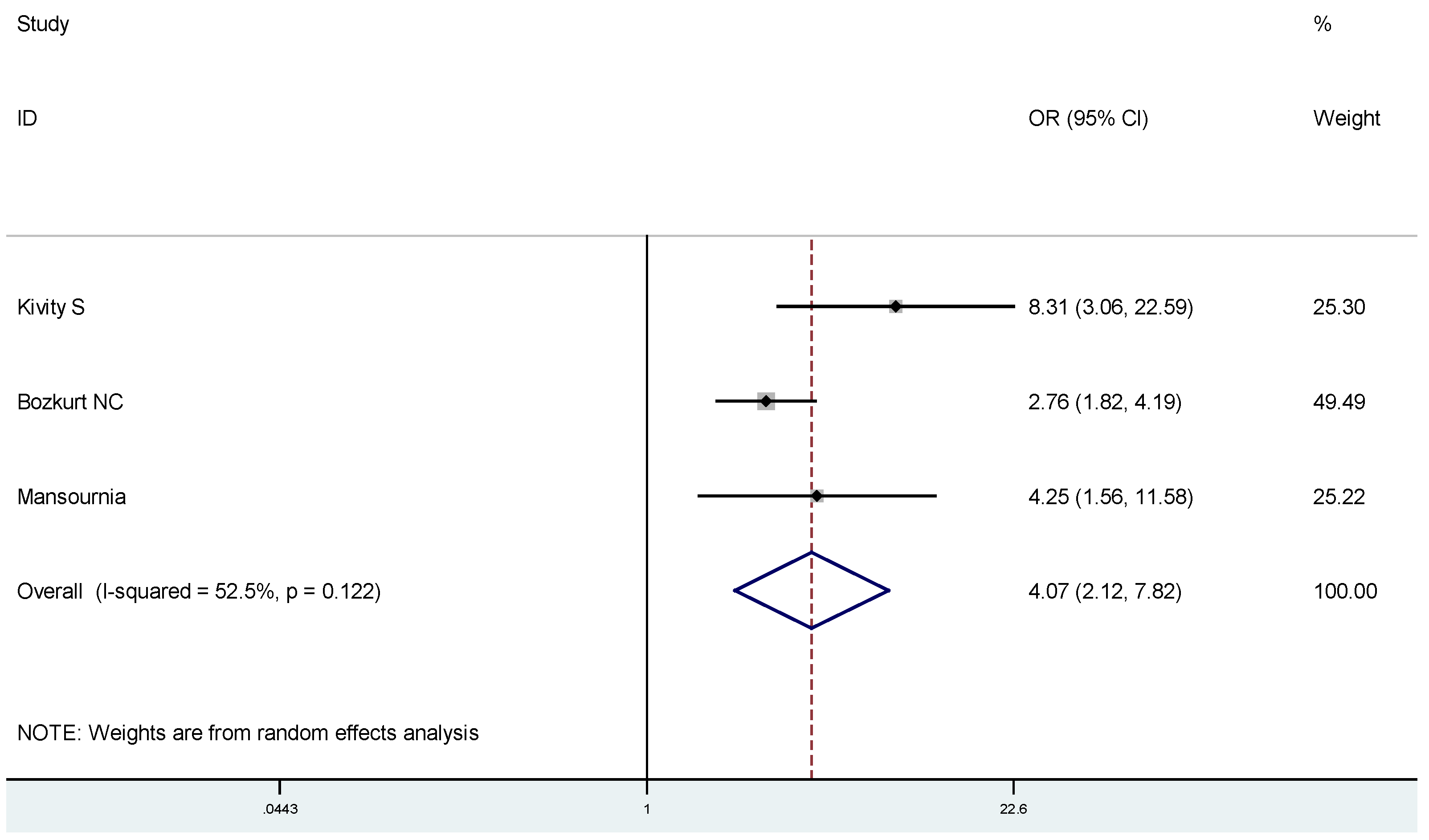
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Holick, M.F. Vitamin D deficiency. N. Engl. J. Med. 2007, 357, 266–281. [Google Scholar] [CrossRef] [PubMed]
- Lemire, J.M.; Adams, J.S.; Sakai, R.; Jordan, S.C. 1 alpha,25-dihydroxyvitamin D3 suppresses proliferation and immunoglobulin production by normal human peripheral blood mononuclear cells. J. Clin. Investig. 1984, 74, 657–661. [Google Scholar] [CrossRef] [PubMed]
- Rigby, W.F.; Stacy, T.; Fanger, M.W. Inhibition of T lymphocyte mitogenesis by 1, 25-dihydroxyvitamin D3 (Calcitriol). J. Clin. Investig. 1984, 74, 1451–1455. [Google Scholar] [CrossRef] [PubMed]
- Deluca, H.F.; Cantorna, M.T. Vitamin D: Its role and uses in immunology. FASEB J. 2001, 15, 2579–2585. [Google Scholar] [CrossRef] [PubMed]
- Abou-Raya, A.; Abou-Raya, S.; Helmii, M. The effect of vitamin D supplementation on inflammatory and hemostatic markers and disease activity in patients with systemic lupus erythematosus: A randomized placebo-controlled trial. J. Rheumatol. 2013, 40, 265–272. [Google Scholar] [CrossRef] [PubMed]
- Grishkan, I.V.; Fairchild, A.N.; Calabresi, P.A.; Gocke, A.R. 1,25-Dihydroxyvitamin D3 selectively and reversibly impairs T helper-cell CNS localization. Proc. Natl. Acad. Sci. USA 2013, 110, 21101–21106. [Google Scholar] [CrossRef] [PubMed]
- Ananthakrishnan, A.N.; Cagan, A.; Gainer, V.S.; Cheng, S.C.; Cai, T.; Szolovits, P.; Shaw, S.Y.; Churchill, S.; Karlson, E.W.; Murphy, S.N.; et al. Higher plasma vitamin D is associated with reduced risk of Clostridium difficile infection in patients with inflammatory bowel diseases. Aliment. Pharmacol. Ther. 2014, 39, 1136–1142. [Google Scholar] [CrossRef] [PubMed]
- Skaaby, T.; Husemoen, L.L.; Thuesen, B.H.; Linneberg, A. Prospective population-based study of the association between vitamin D status and incidence of autoimmune disease. Endocrine 2015. [Google Scholar] [CrossRef]
- Verstuyf, A.; Carmeliet, G.; Bouillon, R.; Mathieu, C. Vitamin D: A pleiotropic hormone. Kidney Int. 2010, 78, 140–145. [Google Scholar] [CrossRef] [PubMed]
- Huang, Z.L. The Study on Relationship between Serum 25-Hydroxyvitamin D3 Concentration and Hashimoto Thyroiditis. Master Dissertation, Jilin University, Jilin, China, 2013. [Google Scholar]
- Liu, X.H. The Study on Relation between Vitamin D3 Level and Immune Disorder in Patients with Autoimmune Thyroid Disease. Master Dissertation, Zhengzhou University, Zhengzhou, China, 2012. [Google Scholar]
- Sezgin, G.; Esref, O.M. Relationship of vitamin D deficiency and autoimmune thyroid diseases. Eur. J. Internal Med. 2011, 22, 87. [Google Scholar] [CrossRef]
- Effraimidis, G.; Badenhoop, K.; Tijssen, J.G.; Wiersinga, W.M. Vitamin D deficiency is not associated with early stages of thyroid autoimmunity. Eur. J. Endocrinol. 2012, 167, 43–48. [Google Scholar] [CrossRef] [PubMed]
- Wells, G.A.; Shea, B.; O’Connell, D.; Peterson, J.; Welch, V.; Losos, M.; Tugwell, P. The Newcastle-Ottawa Scale (NOS) for Assessing the Quality of Non-Randomized Studies in Meta-Analysis. Available online: http://www.ohri.ca/programs/clinical_epidemiology/oxford.asp (accessed on 1 December 2014).
- Eliades, M.; Spyrou, E.; Agrawal, N.; Lazo, M.; Brancati, F.L.; Potter, J.J.; Koteish, A.A.; Clark, J.M.; Guallar, E.; Hernaez, R. Meta-analysis: Vitamin D and non-alcoholic fatty liver disease. Aliment. Pharmacol. Ther. 2013, 38, 246–254. [Google Scholar] [CrossRef] [PubMed]
- Yasuda, T.; Okamoto, Y.; Hamada, N.; Miyashita, K.; Takahara, M.; Sakamoto, F.; Miyatsuka, T.; Kitamura, T.; Katakami, N.; Kawamori, D.; et al. Serum vitamin D levels are decreased in patients without remission of Graves’ disease. Endocrine 2013, 43, 230–232. [Google Scholar] [CrossRef] [PubMed]
- Tamer, G.; Arik, S.; Tamer, I.; Coksert, D. Relative vitamin D insufficiency in Hashimoto’s thyroiditis. Thyroid 2011, 21, 891–896. [Google Scholar] [CrossRef] [PubMed]
- Yasuda, T.; Okamoto, Y.; Hamada, N.; Miyashita, K.; Takahara, M.; Sakamoto, F.; Miyatsuka, T.; Kitamura, T.; Katakami, N.; Kawamori, D.; et al. Serum vitamin D levels are decreased and associated with thyroid volume in female patients with newly onset Graves’ disease. Endocrine 2012, 42, 739–741. [Google Scholar] [CrossRef] [PubMed]
- Bozkurt, N.C.; Karbek, B.; Ucan, B.; Sahin, M.; Cakal, E.; Ozbek, M.; Delibasi, T. The association between severity of vitamin D deficiency and Hashimoto’s thyroiditis. Endocr. Pract. 2013, 19, 479–484. [Google Scholar] [CrossRef] [PubMed]
- Han, Y.; Cheng, Y.K.; Chen, Y.J.; Li, Y. M.; Jiang, Y. Q.; Zhang, S. F. Abnormality of serum 25(OH)D level and its associations with hormones and auto-antibody in patients with Graves’ disease. Chin. J. Clin. Res. 2013, 26, 642–646. [Google Scholar]
- Miao, W.; Ma, J.; Guo, R.; Wang, Y. J.; Wang, G.; Guan, H. X. The correlation between serum 25(OH)D and Graves’ disease. Chin. J. Pract. Med. 2013, 33, 394–395. [Google Scholar]
- Zhang, H.; Liang, L.Y.; Xie, Z.J. Low Vitamin D status is associated with increased titers of thyroid stimulating hormone receptor antibodies in Graves’ disease. Endocr. Pract. 2014. [Google Scholar] [CrossRef]
- Shin, D.Y.; Kim, K.J.; Kim, D.; Hwang, S.; Lee, E.J. Low serum vitamin D is associated with anti-thyroid peroxidase antibody in autoimmune thyroiditis. Yonsei Med. J. 2014, 55, 476–481. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.B.; Xue, X.H.; Liu, S.W.; Xi, G.X.; Zhao, L. X.; Zhang, X.L. Serum vitamin D of early Graves’ disease patients: A clinical research. Chin. Rem. Clin. 2014, 14, 242–243. [Google Scholar]
- Kivity, S.; Agmon, L.N.; Zisappl, M.; Shapira, Y.; Nagy, E.V.; Dankó, K.; Szekanecz, Z.; Langevitz, P.; Shoenfeld, Y. Vitamin D and autoimmune thyroid diseases. Cell Mol. Immunol. 2011, 8, 243–247. [Google Scholar] [CrossRef] [PubMed]
- Jyotsna, V.P.; Sahoo, A.; Ksh, S.A.; Sreenivass, V.; Gupta, N. Bone mineral density in patients of Graves’ disease pre- & post-treatment in a predominantly vitamin D deficient population. Indian J. Med. Res. 2012, 135, 36–41. [Google Scholar] [CrossRef] [PubMed]
- Mansournia, N.; Mansournia, M.A.; Saeedi, S.; Dehghan, J. The association between serum 25(OH)D levels and hypothyroid Hashimoto’s thyroiditis. J. Endocrinol. Investig. 2014, 37, 473–476. [Google Scholar] [CrossRef]
- Xuan, L.Y.; Yang, Y.H.; Lai, X.Y. The association between the serum 25(OH)D3 of autoimmune thyroid diseases patients and the level of sFas. Shandong Med. J. 2014, 38, 61–63. [Google Scholar]
- Zheng, Y.; Zheng, F.P.; Li, H. The relationship between the blood uric acid level of Graves’ disease and bone mineral density of lumbar vertebra. Chin. J. Gerontol. 2014, 11, 3017–3019. [Google Scholar]
- Wang, Y.C. Analysis of the Relationship between 25(OH)D, IGF-1 and Bone Metabolism in Patients with Graves’ Disease. Master Dissertation, Anhui Medical University, Hefei, China, 2014. [Google Scholar]
- Kang, D.H.; Wang, Y.; Cao, W.; Wang, P.; Zhang, H.M. Higher prevalence of vitamin D deficiency in female patients with Graves’ disease. Acta Nutrimenta Sin. 2014, 35, 299–301. [Google Scholar]
- Wang, Z.S.; Wu, Y.P.; Song, Q.H. Bone mineral density and bone metabolism of Graves’ disease in premenopausal women. Guangdong Med. J. 2014, 35, 1743–1746. [Google Scholar]
- Bellastella, G.; Maiorino, M.I.; Petrizzo, M.; de Bellis, A.; Capuano, A.; Esposito, K.; Giugliano, D. Vitamin D and autoimmunity: What happens in autoimmune polyendocrine syndromes? J. Endocrinol. Investig. 2015. [Google Scholar] [CrossRef]
- Feng, M.; Li, H.; Chen, S.F.; Li, W.F.; Zhang, F.B. Polymorphism in the vitamin D receptor gene and risk of autoimmune thyroid disease: A meta-analysis. Endocrine 2013, 43, 318–326. [Google Scholar] [CrossRef] [PubMed]
- Tsatsoulis, A. The role of stress in the clinical expression of thyroid autoimmunity. Ann. N.Y. Acad. Sci. 2006, 1088, 382–395. [Google Scholar] [CrossRef] [PubMed]
- Mullins, R.J.; Cohen, S.B.; Webb, L.M.; Chernajovsky, Y.; Dayan, C.M.; Londei, M.; Feldmann, M. Identification of thyroid stimulating hormone receptor-specific T cells in Graves’ disease thyroid using autoantigen-transfected Epstein-Barrvirus-transformed B cell lines. J. Clin. Investig. 1995, 96, 30–37. [Google Scholar] [CrossRef] [PubMed]
- Kallmann, B.A.; Hüther, M.; Tubes, M.; Feldkamp, J.; Bertrams, J.; Gries, F.A.; Lampeter, E.F.; Kolb, H. Systemic bias of cytokine production toward cell-mediated immune regulation in IDDM and toward humoral immunity in Graves’ disease. Diabetes 1997, 46, 237–243. [Google Scholar] [CrossRef] [PubMed]
- Dogan, R.N.E.; Vasy, C. Absence of IL-4 and not suppression of the Th2 response, prevents development of experimental autoimmune thyroid Graves’ disease. J. Immunol. 2003, 170, 2195–2204. [Google Scholar] [CrossRef] [PubMed]
- Karanikas, G.; Schuetz, M.; Wahl, K.; Paul, M.; Kontur, S.; Pietschmann, P.; Kletter, K.; Dudczak, R.; Willheim, M. Relation of anti-TPO autoantibody titre and T-lymphocyte cytokine production patterns in Hashimoto’s thyroiditis. Clin. Endocrinol. (Oxf.) 2005, 63, 191–196. [Google Scholar] [CrossRef]
- Peng, D.; Xu, B.; Wang, Y.; Guo, H.; Jiang, Y. A high frequency of circulating Th22 and Th17 cells in patients with new onset Graves’ disease. PLoS ONE 2013, 8. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; Cai, W.; Gu, R.; Zhang, Y.; Zhang, H.; Tang, K.; Xu, P.; Katirai, F.; Shi, W.; Wang, L.; et al. Th17 cells plays a role in the pathogenesis of Hashimoto’s thyroiditis in patients. Clin. Immunol. 2013, 149, 411–420. [Google Scholar] [CrossRef]
- Qin, Q.; Liu, P.; Liu, L.; Wang, R.; Yan, N.; Yang, J.; Wang, X.; Pandey, M.; Zhang, J.A. The increased but non-predominant expression of Th17- and Th1-specific cytokines in Hashimoto’s thyroiditis but not in Graves’ disease. Braz. J. Med. Biol. Res. 2012, 45, 1202–1208. [Google Scholar] [CrossRef] [PubMed]
- Ito, C.; Watanabe, M.; Okuda, N.; Watanabe, C.; Iwatani, Y. Association between the severity of Hashimoto’s disease and the functional +874A/T polymorphism in the interferon-gamma gene. Endocr. J. 2006, 53, 473–478. [Google Scholar] [CrossRef] [PubMed]
- Staeva-Vieira, T.P.; Freedman, L.P. 1,25-Dihydroxyvitamin D3 inhabit IFN-γ and IL-4 levels during in vitro polarization of primary murine CD4+ T cells. J. Immunol. 2002, 168, 1181–1189. [Google Scholar] [CrossRef] [PubMed]
- Palmer, M.T.; Lee, Y.K.; Maynard, C.L.; Oliver, J.R.; Bikle, D.D.; Jetten, A.M.; Weaver, C.T. Lineage-specific Effects of 1, 25-Dihydroxyvitamin D3 on the development of effect CD4 T cells. J. Biol. Chem. 2011, 286, 997–1004. [Google Scholar] [CrossRef] [PubMed]
- Pichler, J.; Gerstmayr, M.; Szépfalusi, Z.; Urbanek, R.; Peterlik, M.; Willheim, M. 1α,25 (OH)2D3 inhabits not only Th1 but also Th2 differentiation in human blood T cells. Pediatr. Res. 2002, 52, 12–18. [Google Scholar] [PubMed]
- Joshi, S.; Pantalena, L.C.; Liu, X.K.; Gaffen, S.L.; Liu, H.; Rohowsky-Kochan, C.; Ichiyama, K.; Yoshimura, A.; Steinman, L.; Christakos, S.; et al. 1,25-Dihydroxyvitamin D3 ameliorates Th17 autoimmunity via transcriptional modulation of interleukin-17A. Mol. Cell Biol. 2011, 31, 3653–3669. [Google Scholar] [CrossRef] [PubMed]
- Muscogiuri, G.; Mitri, J.; Mathieu, C.; Badenhoop, K.; Tamer, G.; Orio, F.; Mezza, T.; Vieth, R.; Colao, A.; Pittas, A. Mchanisms in endocrinology: Vitamin D as a potential contributor in endocrine health and disease. Eur. J. Endocrinol. 2014, 171, 101–110. [Google Scholar] [CrossRef]
- Zhang, Q.Q.; Sun, M.; Wang, Z.X.; Fu, Q.; Shi, Y.; Yang, F.; Zheng, S.; Xu, J.J.; Huang, X.P.; Liu, X.Y.; Cui, D.; Yang, T. Relationship between serum 25-hydroxy vitamin D and thyroid autoimmunity among middle-aged and elderly individuals. Acta Univ. Med. Nanjing (Nat. Sci.) 2014, 34, 486–489. [Google Scholar]
- Shin, D.; Hwang, S. Baseline vitamin D level could be a short-term prognostic marker in patients with Graves’ disease. Thyroid 2011, 21, A48. [Google Scholar]
- Kawakami-Tani, T.; Fukawa, E.; Tanaka, H.; Abe, Y.; Makino, I. Effect of 1 alpha-hydroxyvitamin D3 on serum levels of thyroid hormones in hyperthyroid patients with untreated Graves’ disease. Metabolism 1997, 46, 1184–1188. [Google Scholar] [CrossRef] [PubMed]
- De Remigis, P.; Vianale, L.; De Remingis, A.; Napolitano, G. Vitamin D and autoimmune thyroid disease (at): Preliminary results. Thyroid 2013, 23, A81–A82. [Google Scholar]
- Amrein, K.; Zajic, P.; Schnedl, C.; Waltensdorfer, A.; Fruhwald, S.; Holl, A.; Purkart, T.; Wünsch, G.; Valentin, T.; Grisold, A.; et al. Vitamin D status and its association with season, hospital and sepsis mortality in critical illness. Crit. Care 2014, 18. [Google Scholar] [CrossRef]
- Lai, J.K.; Lucas, R.M.; Clements, M.S.; Harrison, S.L.; Banks, E. Assessing vitamin D status: Pitfalls for the unwary. Mol. Nutr. Food Res. 2010, 54, 1062–1071. [Google Scholar] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, J.; Lv, S.; Chen, G.; Gao, C.; He, J.; Zhong, H.; Xu, Y. Meta-Analysis of the Association between Vitamin D and Autoimmune Thyroid Disease. Nutrients 2015, 7, 2485-2498. https://doi.org/10.3390/nu7042485
Wang J, Lv S, Chen G, Gao C, He J, Zhong H, Xu Y. Meta-Analysis of the Association between Vitamin D and Autoimmune Thyroid Disease. Nutrients. 2015; 7(4):2485-2498. https://doi.org/10.3390/nu7042485
Chicago/Turabian StyleWang, Jiying, Shishi Lv, Guo Chen, Chenlin Gao, Jianhua He, Haihua Zhong, and Yong Xu. 2015. "Meta-Analysis of the Association between Vitamin D and Autoimmune Thyroid Disease" Nutrients 7, no. 4: 2485-2498. https://doi.org/10.3390/nu7042485
APA StyleWang, J., Lv, S., Chen, G., Gao, C., He, J., Zhong, H., & Xu, Y. (2015). Meta-Analysis of the Association between Vitamin D and Autoimmune Thyroid Disease. Nutrients, 7(4), 2485-2498. https://doi.org/10.3390/nu7042485




