Effects of Dietary Fat and Saturated Fat Content on Liver Fat and Markers of Oxidative Stress in Overweight/Obese Men and Women under Weight-Stable Conditions
Abstract
:1. Introduction
2. Subjects and Methods
2.1. Study Design
2.2. Subjects
2.3. Dietary Intervention
| Title | Baseline a | CONT LFD | LFD | CONT HFD | HFD |
|---|---|---|---|---|---|
| Daily energy (kcal) | 2326 ± 334 | 3284 ± 125 | 3321 ± 150 | 3140 ± 120 | 3208 ± 92 |
| Fat (% of total energy) | 38.6 ± 2.1 | 35.8 ± 0.6 | 20.2 ± 0.003 1 | 35.2 ± 0.02 | 54.8 ± 0.05 2,3 |
| Saturated fat (% of total energy) | 13.2 ± 0.9 | 11.9 ± 0.4 | 7.7 ± 0.01 1 | 11.6 ± 0.02 | 23.7 ± 0.05 2,3 |
| MUFA (% of total energy) | 13.7 ± 1.0 | 16.7 ± 0.1 | 7.7 ± 0.01 1 | 16.6 ± 0.04 | 22.2 ± 0.06 2,3 |
| PUFA (% of total energy) | 8.2 ± 0.8 | 4.7 ± 0.01 | 3.0 ± 0.01 1 | 4.7 ± 0.03 | 5.2 ± 0.04 2,3 |
| Carbohydrate (% of total energy) | 42.5 ± 2.7 | 46.4 ± 0.6 | 61.7 ± 0.02 1 | 46.9 ± 0.01 | 27.4 ± 0.05 2,3 |
| Protein (% of total energy) | 18.9 ± 1.3 | 17.9 ± 0.01 | 18.1 ± 0.02 1 | 17.8 ± 0.01 | 17.8 ± 0.03 3 |
| Cholesterol (mg/day) | 479 ± 93 | 378 ± 10 | 492 ± 21 1 | 352 ± 11 | 506 ± 17 2 |
| Total fiber (g/day) | 16.2 ± 2.1 | 47.2 ± 2.0 | 46.1 ± 2.1 | 45.8 ± 1.8 | 39.8 ± 1.3 2,3 |
| Fructose (g/day) | 26.7 ± 7.6 | 34.1 ± 1.8 | 46.1 ± 2.1 1 | 33.1 ± 1.4 | 10.0 ± 0.3 2,3 |
| Vitamin C (mg/day) | 68.8 ± 20.0 | 260.8 ± 13.0 | 306.6 ± 16.2 1 | 230.4 ± 20.8 | 121.1 ± 4.8 2,3 |
| Vitamin E (mg/day) | 9.0 ± 1.7 | 27.4 ± 1.1 | 14.4 ± 0.6 1 | 24.8 ± 1.6 | 28.5 ± 0.8 2,3 |
2.4. Study Procedures
2.5. Quantification of Liver Fat
2.6. Body Fat Distribution
2.7. Assays
2.8. Statistical Analysis
3. Results
3.1. Baseline Subject Characteristics and Diet Composition
3.2. Response to the LFD

3.3. Response to the HFD
| Title | Low Fat Diet | High Fat Diet | ||
|---|---|---|---|---|
| CONT | LFD | CONT | HFD | |
| Weight (kg) | 100.7 ± 4.1 | 100.5 ± 4.4 | 104.0 ± 5.8 | 104.1 ± 5.9 |
| Liver fat (%) | 9.4 ± 7.5 | 7.2 ± 7.7 1 | 8.3 ± 7.9 | 7.0 ± 7.2 |
| IAF (cm3) | 1479 ± 331 | 1447 ± 321 | 1179 ± 235 | 1202 ± 238 |
| SQF (cm3) | 2440 ± 316 | 2413 ± 301 | 2704 ± 478 | 2861 ± 483 1,2 |
| ALT (Units/L) | 26.2 ± 2.8 | 23.8 ± 2.9 | 25.2 ± 3.0 | 25.4 ± 2.1 |
| AST (Units/L) | 21 ± 1.6 | 19.0 ± 1.5 | 20.6 ± 1.9 | 22.2±2.7 |
| GGT (Units/L) | 30.8 ± 6.0 | 33.7 ± 10.2 | 29.0 ± 7.2 | 26.8 ± 8.0 2 |
| Total cholesterol (mg/dL) | 179.1 ± 6.0 | 179.4 ± 7.3 | 159.3 ± 6.2 | 166.5 ± 8.8 |
| Triglycerides (mg/dL) | 120.1 ± 10.7 | 148.7 ± 25.4 | 84.2 ± 9.8 | 88.6 ± 14.1 |
| HDL cholesterol (mg/dL) | 41.5 ± 4.4 | 40.0 ± 4.6 | 48.1 ± 4.8 | 47.4 ± 5.3 |
| LDL cholesterol (mg/dL) | 113.6 ± 6.8 | 109.7 ± 6.9 | 94.4 ± 6.8 | 101.3 ± 8.6 |
| Fasting glucose (mg/dL) | 95.5 ± 3.3 | 97.1 ± 4.0 | 95.3 ± 3.3 | 94.7 ± 2.9 |
| Fasting insulin (µU/mL) | 13.9 ± 2.1 | 12.8 ± 2.0 | 16.6 ± 3.7 | 15.0 ± 3.3 |
| Fasting NEFA (mE/L) | 0.39 ± 0.04 | 0.39 ± 0.06 | 0.34 ± 0.04 | 0.35 ± 0.04 |
| Adiponectin (µg/mL) | 3.4 ± 0.3 | 4.1 ± 1.2 | 4.2 ± 0.9 | 4.6 ± 1.2 |
| Leptin (ng/mL) | 13.9 ± 3.3 | 15.1 ± 3.3 | 17.3 ± 3.5 | 16.8 ± 4.1 |
| hsCRP (mg/L) | 3.3 ± 0.9 | 2.8 ± 0.8 | 2.3 ± 0.6 | 2.2 ± 0.4 |
| IL-6 (pg/mL) | 1.08 (1.09) | 1.01 (1.14) | 0.91 (1.4) | 0.83 (2.4) |
| IL-10 (pg/mL) | 3.7 ± 0.3 | 3.4 ± 0.2 | 3.2 ± 0.1 | 3.4 ± 0.2 |
| IL-12 (pg/mL) | 1.24 (3.02) | 1.14 (4.57) | 1.12 (6.35) | 1.04 (11.01) |
| γ-interferon (pg/mL) | 11.1 (19.0) | 14.2 (15.2) | 13.7 (21.4) | 9.9 (19.8) |
| Urinary F2α isoprostanes (ng/mg Cr) | 1.04 ± 0.12 | 1.00 ± 0.11 | 1.3 ± 0.2 | 1.3 ± 0.2 |
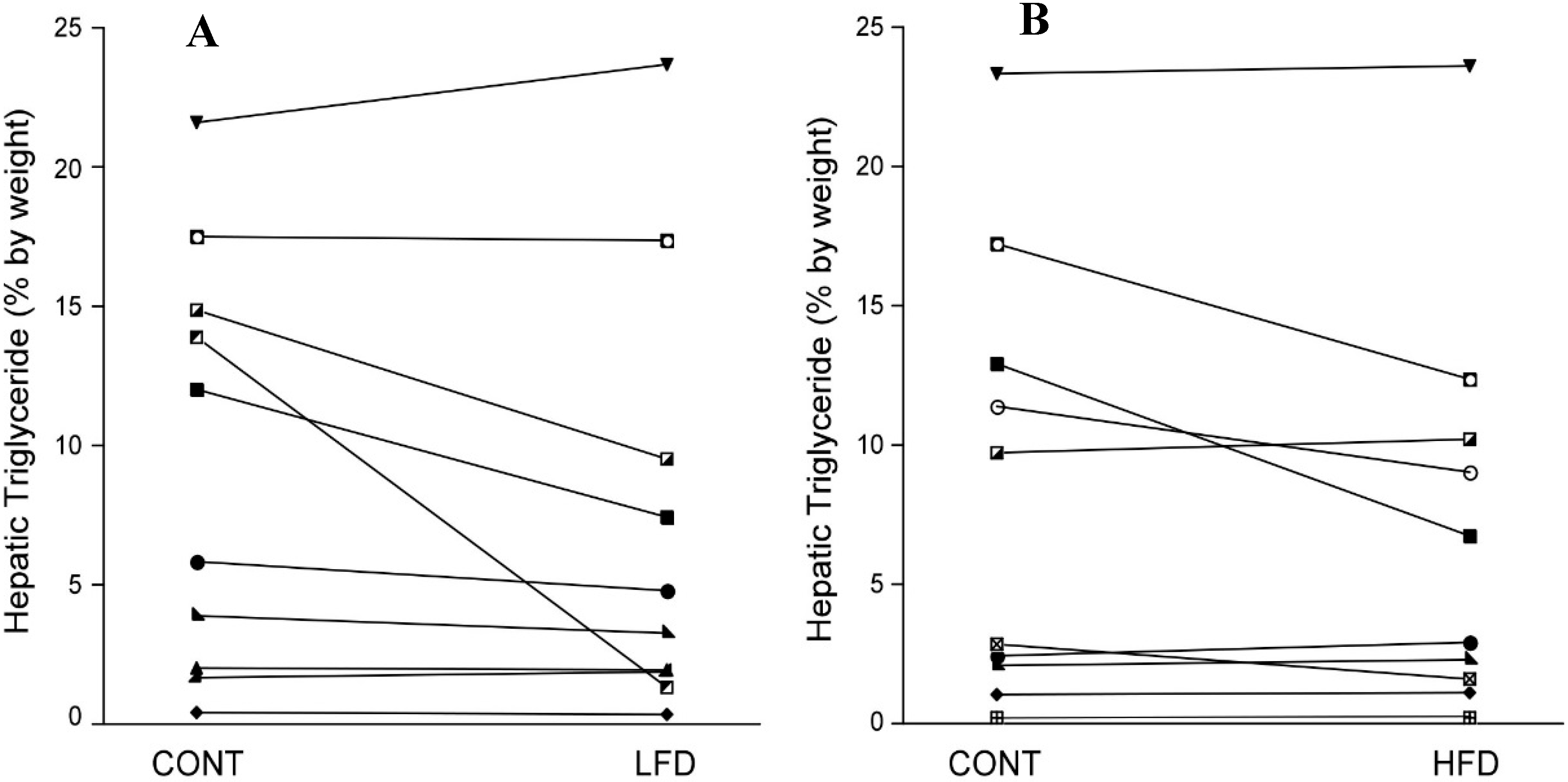
3.4. Comparison of the LFD and HFD
4. Discussion
5. Conclusions
Supplementary Files
Supplementary File 1Acknowledgments
Author Contributions
Conflicts of Interest
References
- Matteoni, C.A.; Younossi, Z.M.; Gramlich, T.; Boparai, N.; Liu, Y.C.; McCullough, A.J. Nonalcoholic fatty liver disease: A spectrum of clinical and pathological severity. Gastroenterology 1999, 116, 1413–1419. [Google Scholar] [CrossRef] [PubMed]
- Singal, A.K.; Guturu, P.; Hmoud, B.; Kuo, Y.F.; Salameh, H.; Wiesner, R.H. Evolving frequency and outcomes of liver transplantation based on etiology of liver disease. Transplantation 2013, 95, 755–760. [Google Scholar] [CrossRef] [PubMed]
- Day, C.P.; James, O.F. Steatohepatitis: A tale of two “hits”? Gastroenterology 1998, 114, 842–845. [Google Scholar] [CrossRef]
- Wang, D.; Wei, Y.; Pagliassotti, M.J. Saturated fatty acids promote endoplasmic reticulum stress and liver injury in rats with hepatic steatosis. Endocrinology 2006, 147, 943–951. [Google Scholar] [CrossRef] [PubMed]
- Lieber, C.S.; Leo, M.A.; Mak, K.M.; Xu, Y.; Cao, Q.; Ren, C.; Ponomarenko, A.; DeCarli, L.M. Model of nonalcoholic steatohepatitis. Am. J. Clin. Nutr. 2004, 79, 502–509. [Google Scholar] [PubMed]
- Van Herpen, N.A.; Schrauwen-Hinderling, V.; Schaart, G.; Mensinck, R.P.; Schrauwen, P. Three weeks on a high-fat diet increases intrahepatic lipid accumulation and decreases metabolic flexibility in healthy overweight men. JCEM 2011, 96, E691–E695. [Google Scholar]
- Westerbacka, J.; Lammi, K.; Hakkinen, A.M.; Rissanen, A.; Salminen, I.; Aro, A.; Yki-Jarvinen, H. Dietary fat content modifies liver fat in overweight nondiabetic subjects. J. Clin. Endocrinol. Metab. 2005, 90, 2804–2809. [Google Scholar] [CrossRef] [PubMed]
- Utzschneider, K.M.; Bayer-Carter, J.L.; Arbuckle, M.D.; Tidwell, J.M.; Richards, T.L.; Craft, S. Beneficial effect of a weight-stable, low-fat/low-saturated fat/low-glycaemic index diet to reduce liver fat in older subjects. Br. J. Nutr. 2013, 109, 1096–1104. [Google Scholar] [CrossRef] [PubMed]
- USDA Database for the Added Sugar Content of Selected Foods, Release 1. Available online: http://www.megaheart.com/pdf/addedsugars.pdf (accessed on 22 October 2014).
- Agricultural Research Services USDA. Available online: http://www.ars.usda.gov/SP2UserFiles/Place/80400530/pdf/fndds2_doc.pdf (accessed on 22 October 2014).
- Mifflin, M.D.; St Jeor, S.T.; Hill, L.A.; Scott, B.J.; Daugherty, S.A.; Koh, Y.O. A new predictive equation for resting energy expenditure in healthy individuals. Am. J. Clin. Nutr. 1990, 51, 241–247. [Google Scholar] [PubMed]
- Harris, J.A.; Benedict, F.G. A Biometric Study of Basal Metabolism in Man; Carnegie Institution of Washington: Washington, DC, USA, 1919. [Google Scholar]
- Craig, C.L.; Marshall, A.L.; Sjostrom, M.; Bauman, A.E.; Booth, M.L.; Ainsworth, B.E.; Pratt, M.; Ekelund, U.; Yngve, A.; Sallis, J.F.; et al. International physical activity questionnaire: 12-Country reliability and validity. Med. Sci. Sports Exerc. 2003, 35, 1381–1395. [Google Scholar] [CrossRef] [PubMed]
- Jenkinson, M.; Beckmann, C.F.; Behrens, T.E.; Woolrich, M.W.; Smith, S.M. FSL. NeuroImage 2012, 62, 782–790. [Google Scholar] [CrossRef] [PubMed]
- Milatovic, D.; vanRollins, M.; Li, K.; Montine, K.S.; Montine, T.J. Suppression of murine cerebral F2-isoprostanes and F4-neuroprostanes from excitotoxicity and innate immune response in vivo by alpha- or gamma-tocopherol. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2005, 827, 88–93. [Google Scholar] [CrossRef]
- Willett, W. Nutritional Epidemiology; Oxford University Press: New York, NY, USA, 2012. [Google Scholar]
- Bozzetto, L.; Prinster, A.; Annuzzi, G.; Costagliola, L.; Mangione, A.; Vitelli, A.; Mazzarella, R.; Longobardo, M.; Mancini, M.; Vigorito, C.; et al. Liver fat is reduced by an isoenergetic MUFA diet in a controlled randomized study in type 2 diabetic patients. Diabetes Care 2012, 35, 1429–1435. [Google Scholar] [CrossRef] [PubMed]
- Faeh, D.; Minehira, K.; Schwarz, J.M.; Periasamy, R.; Park, S.; Tappy, L. Effect of fructose overfeeding and fish oil administration on hepatic de novo lipogenesis and insulin sensitivity in healthy men. Diabetes 2005, 54, 1907–1913. [Google Scholar] [CrossRef] [PubMed]
- Le, K.A.; Ith, M.; Kreis, R.; Faeh, D.; Bortolotti, M.; Tran, C.; Boesch, C.; Tappy, L. Fructose overconsumption causes dyslipidemia and ectopic lipid deposition in healthy subjects with and without a family history of type 2 diabetes. Am. J. Clin. Nutr. 2009, 89, 1760–1765. [Google Scholar] [CrossRef] [PubMed]
- Le, K.A.; Faeh, D.; Stettler, R.; Ith, M.; Kreis, R.; Vermathen, P.; Boesch, C.; Ravussin, E.; Tappy, L. A 4-wk high-fructose diet alters lipid metabolism without affecting insulin sensitivity or ectopic lipids in healthy humans. Am. J. Clin. Nutr. 2006, 84, 1374–1379. [Google Scholar] [PubMed]
- Silbernagel, G.; Machann, J.; Unmuth, S.; Schick, F.; Stefan, N.; Haring, H.U.; Fritsche, A. Effects of 4-week very-high-fructose/glucose diets on insulin sensitivity, visceral fat and intrahepatic lipids: An exploratory trial. Br. J. Nutr. 2011, 106, 79–86. [Google Scholar] [CrossRef] [PubMed]
- Musso, G.; Gambino, R.; de Michieli, F.; Biroli, G.; Premoli, A.; Pagano, G.; Bo, S.; Durazzo, M.; Cassader, M. Nitrosative stress predicts the presence and severity of nonalcoholic fatty liver at different stages of the development of insulin resistance and metabolic syndrome: Possible role of vitamin A intake. Am. J. Clin. Nutr. 2007, 86, 661–671. [Google Scholar] [PubMed]
- Chalasani, N.; Deeg, M.A.; Crabb, D.W. Systemic levels of lipid peroxidation and its metabolic and dietary correlates in patients with nonalcoholic steatohepatitis. Am. J. Gastroenterol. 2004, 99, 1497–1502. [Google Scholar] [CrossRef] [PubMed]
- Madan, K.; Bhardwaj, P.; Thareja, S.; Gupta, S.D.; Saraya, A. Oxidant stress and antioxidant status among patients with nonalcoholic fatty liver disease (NAFLD). J. Clin. Gastroenterol. 2006, 40, 930–935. [Google Scholar] [CrossRef] [PubMed]
- Yesilova, Z.; Yaman, H.; Oktenli, C.; Ozcan, A.; Uygun, A.; Cakir, E.; Sanisoglu, S.Y.; Erdil, A.; Ates, Y.; Aslan, M.; et al. Systemic markers of lipid peroxidation and antioxidants in patients with nonalcoholic fatty liver disease. Am. J. Gastroenterol. 2005, 100, 850–855. [Google Scholar] [CrossRef] [PubMed]
- Gawrieh, S.; Opara, E.C.; Koch, T.R. Oxidative stress in nonalcoholic fatty liver disease: Pathogenesis and antioxidant therapies. J. Investig. Med. 2004, 52, 506–514. [Google Scholar] [PubMed]
- Sanyal, A.J.; Campbell-Sargent, C.; Mirshahi, F.; Rizzo, W.B.; Contos, M.J.; Sterling, R.K.; Luketic, V.A.; Shiffman, M.L.; Clore, J.N. Nonalcoholic steatohepatitis: Association of insulin resistance and mitochondrial abnormalities. Gastroenterology 2001, 120, 1183–1192. [Google Scholar] [CrossRef] [PubMed]
- Sanyal, A.J.; Chalasani, N.; Kowdley, K.V.; McCullough, A.; Diehl, A.M.; Bass, N.M.; Neuschwander-Tetri, B.A.; Lavine, J.E.; Tonascia, J.; Unalp, A.; et al. Pioglitazone, vitamin E, or placebo for nonalcoholic steatohepatitis. N. Engl. J. Med. 2010, 362, 1675–1685. [Google Scholar] [CrossRef] [PubMed]
- Roberts, C.K.; Won, D.; Pruthi, S.; Lin, S.S.; Barnard, R.J. Effect of a diet and exercise intervention on oxidative stress, inflammation and monocyte adhesion in diabetic men. Diabetes Res. Clin. Pract. 2006, 73, 249–259. [Google Scholar] [CrossRef] [PubMed]
- Roberts, C.K.; Chen, A.K.; Barnard, R.J. Effect of a short-term diet and exercise intervention in youth on atherosclerotic risk factors. Atherosclerosis 2007, 191, 98–106. [Google Scholar] [CrossRef] [PubMed]
- Fito, M.; Guxens, M.; Corella, D.; Saez, G.; Estruch, R.; de la Torre, R.; Frances, F.; Cabezas, C.; Lopez-Sabater Mdel, C.; Marrugat, J.; et al. Effect of a traditional Mediterranean diet on lipoprotein oxidation: A randomized controlled trial. Arch. Int. Med. 2007, 167, 1195–1203. [Google Scholar] [CrossRef]
- Rankin, J.W.; Turpyn, A.D. Low carbohydrate, high fat diet increases C-reactive protein during weight loss. J. Am. Coll. Nutr. 2007, 26, 163–169. [Google Scholar] [CrossRef] [PubMed]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Marina, A.; Von Frankenberg, A.D.; Suvag, S.; Callahan, H.S.; Kratz, M.; Richards, T.L.; Utzschneider, K.M. Effects of Dietary Fat and Saturated Fat Content on Liver Fat and Markers of Oxidative Stress in Overweight/Obese Men and Women under Weight-Stable Conditions. Nutrients 2014, 6, 4678-4690. https://doi.org/10.3390/nu6114678
Marina A, Von Frankenberg AD, Suvag S, Callahan HS, Kratz M, Richards TL, Utzschneider KM. Effects of Dietary Fat and Saturated Fat Content on Liver Fat and Markers of Oxidative Stress in Overweight/Obese Men and Women under Weight-Stable Conditions. Nutrients. 2014; 6(11):4678-4690. https://doi.org/10.3390/nu6114678
Chicago/Turabian StyleMarina, Anna, Anize Delfino Von Frankenberg, Seda Suvag, Holly S. Callahan, Mario Kratz, Todd L. Richards, and Kristina M. Utzschneider. 2014. "Effects of Dietary Fat and Saturated Fat Content on Liver Fat and Markers of Oxidative Stress in Overweight/Obese Men and Women under Weight-Stable Conditions" Nutrients 6, no. 11: 4678-4690. https://doi.org/10.3390/nu6114678
APA StyleMarina, A., Von Frankenberg, A. D., Suvag, S., Callahan, H. S., Kratz, M., Richards, T. L., & Utzschneider, K. M. (2014). Effects of Dietary Fat and Saturated Fat Content on Liver Fat and Markers of Oxidative Stress in Overweight/Obese Men and Women under Weight-Stable Conditions. Nutrients, 6(11), 4678-4690. https://doi.org/10.3390/nu6114678




