Dose and Latency Effects of Leucine Supplementation in Modulating Glucose Homeostasis: Opposite Effects in Healthy and Glucocorticoid-Induced Insulin-Resistance States
Abstract
:1. Introduction
2. Materials and Methods
2.1. Animals
2.2. Study 1
Groups
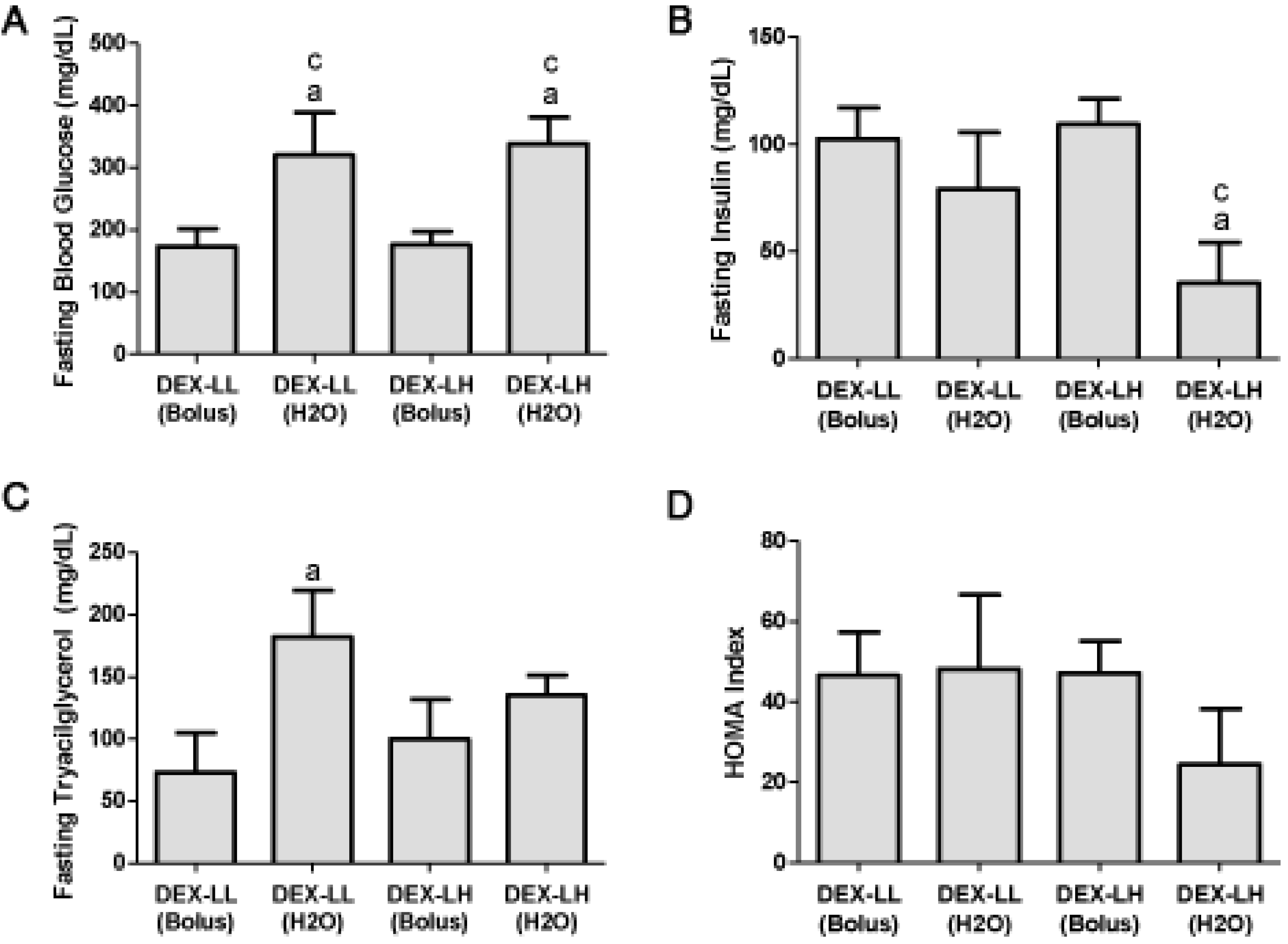
2.3. Study 2
2.3.1. Groups
2.3.2. Basal Fasting Glucose, Insulin and Tryacilglycerol (TAG) Levels
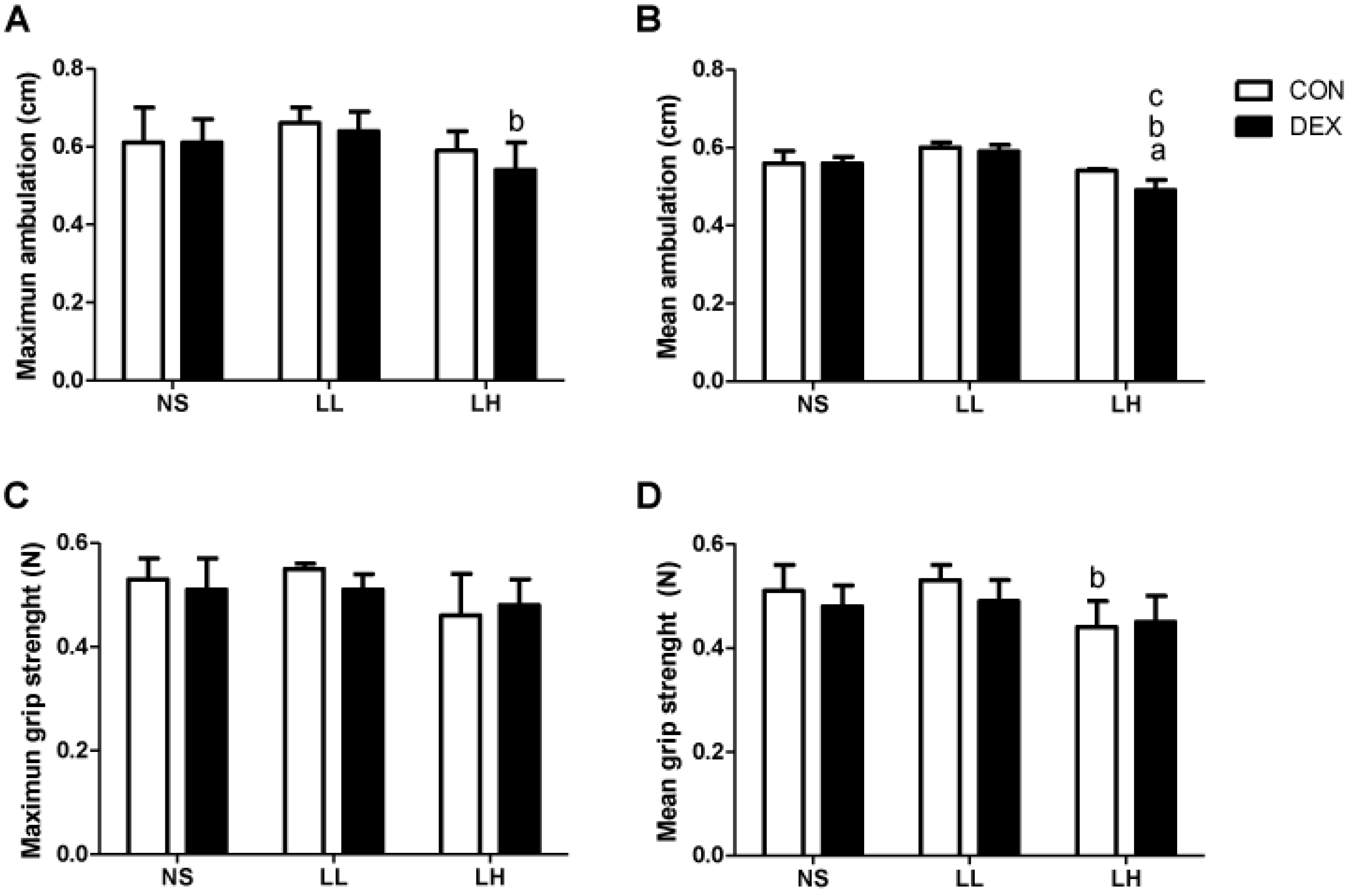
2.3.3. Motor Performance Tests
2.3.4. RNA Isolation and Realt-Time PCR
2.3.5. Statistical Analysis
3. Results
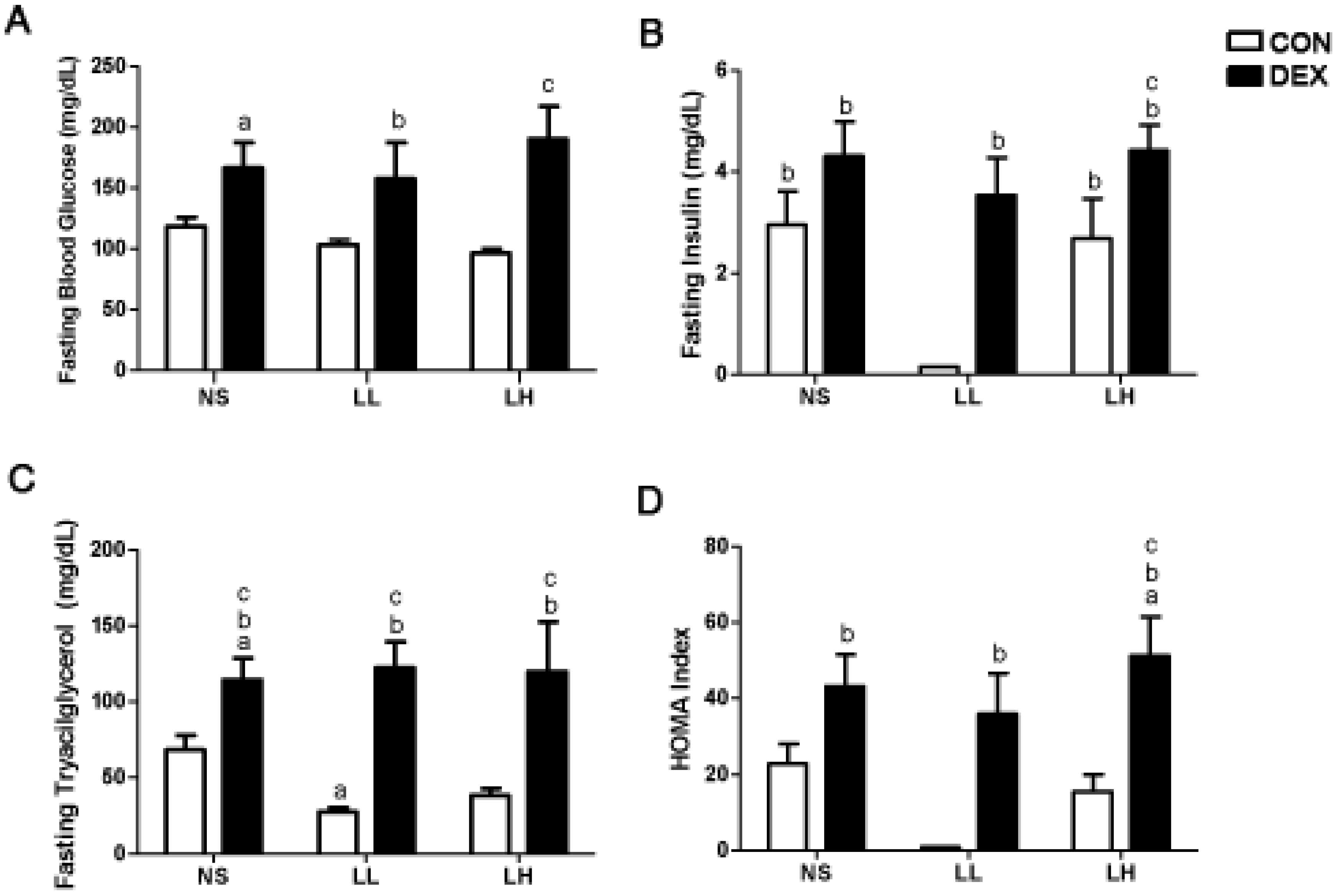
3.1. Study 1
| Variable | Group | |||||
|---|---|---|---|---|---|---|
| CON-NS | CON-LL | CON-LH | DEX-NS | DEX-LL | DEX-LH | |
| Initial BW (g) | 442.7 ± 5.90 | 442.2 ± 4.91 | 441.0 ± 4.35 | 443.8 ± 6.19 | 443.3 ± 4.19 | 444.8 ± 6.16 |
| Final BW (g) | 381.9 ± 22.1 b | 386.3 ± 20.32 b | 375.0 ± 10.21 b | 343.4 ± 11.53 a,b | 345.79 ± 15.89 a,b | 339.9 ± 11.73 a,b |
| Delta BW (g) | −60.8 ± 4.64 | −55.9 ± 5.36 | −65.17 ± 4.98 | −100.8 ± 2.40 a | −103.6 ± 2.08 a | −104.5 ± 3.35 a |
| Soleus (mg) | 214.8 ± 5.99 | 223.3 ± 5.45 | 221.0 ± 6.13 | 214.4 ± 4.29 | 210.8 ± 2.25 | 212.7 ± 5.45 |
| EDL (mg) | 189.4 ± 2.31 | 197.3 ± 2.08 | 200.1 ± 1.49 a | 179.6 ± 3.37 a | 174.8 ± 2.80 a | 174.6 ± 4.12 a |
| Day | Group | |||||
|---|---|---|---|---|---|---|
| CON-NS | CON-LL | CON-LH | DEX-NS | DEX-LL | DEX-LH | |
| 1 | 110.5 ± 5.73 | 107.3 ± 0.76 | 115.5 ± 1.47 | 120.1 ± 5.06 | 104.8 ± 4.07 | 114.7 ± 4.78 |
| 3 | 109.8 ± 5.26 | 103.5 ± 1.16 | 129.6 ± 1.28 | 215.6 ± 29.08 a,b | 196.5 ± 17.27 a,b | 178.1 ± 15.18 a,b |
| 6 | 109.8 ± 5.26 | 110.0 ± 1.64 | 130.7 ± 2.25 | 267.3 ± 31.51 a,b | 255.2 ± 23.44 a,b | 215.6 ± 22.33 a,b |
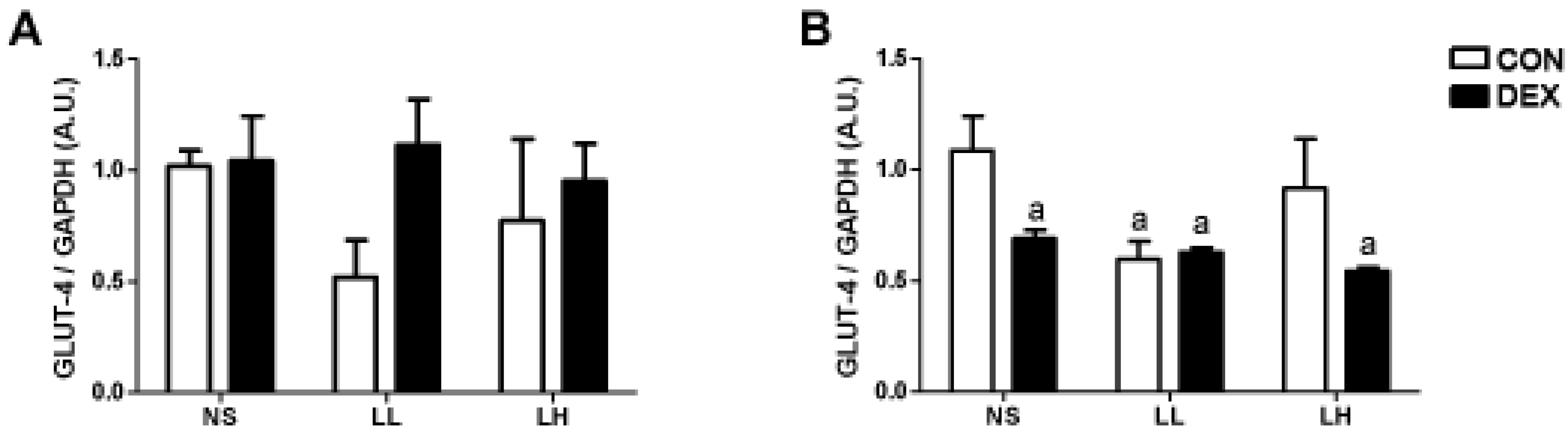
3.2. Study 2
| Variable | Group | |||
|---|---|---|---|---|
| DEX-LL | DEX-LL (H2O) | DEX-LH | DEX-LH (H2O) | |
| Initial BW (g) | 442.3 ± 1.40 | 441.8 ± 1.58 | 444.9 ± 1.49 | 446.7 ± 1.27 |
| Final BW (g) | 338.9 ± 1.83 | 346.5 ± 3.58 | 340.0 ± 2.93 | 361.0 ± 4.13 a,b,c |
| Delta BW (g) | −103.6 ± 2.07 | −95.27 ± 3.14 | −104.5 ± 3.35 | −85.67 ± 6.77 a,b |
| Soleus (mg) | 222.5 ± 5.28 | 205.0 ± 2.39 a,b | 222.9 ± 5.54 | 207.4 ± 3.22 |
| EDL (mg) | 174.8 ± 2.79 | 170.5 ± 1.99 | 174.6 ± 4.12 | 169.4 ± 1.27 |
| Day | Group | |||
|---|---|---|---|---|
| DEX-LL | DEX-LL (H2O) | DEX-LH | DEX-LH (H2O) | |
| 1 | 104.8 ± 4.07 | 100.2 ± 4.14 | 114.7 ± 4.78 | 100.3 ± 4.95 |
| 3 | 196.5 ± 17.27 b | 175.4 ± 9.49 b | 178.1 ± 15.18 | 177.4 ± 10.16 b |
| 6 | 255.2 ± 23.44 b | 286.1 ± 37.34 a,b | 215.6 ± 22.33 b | 314.8 ± 39.34 a,b |
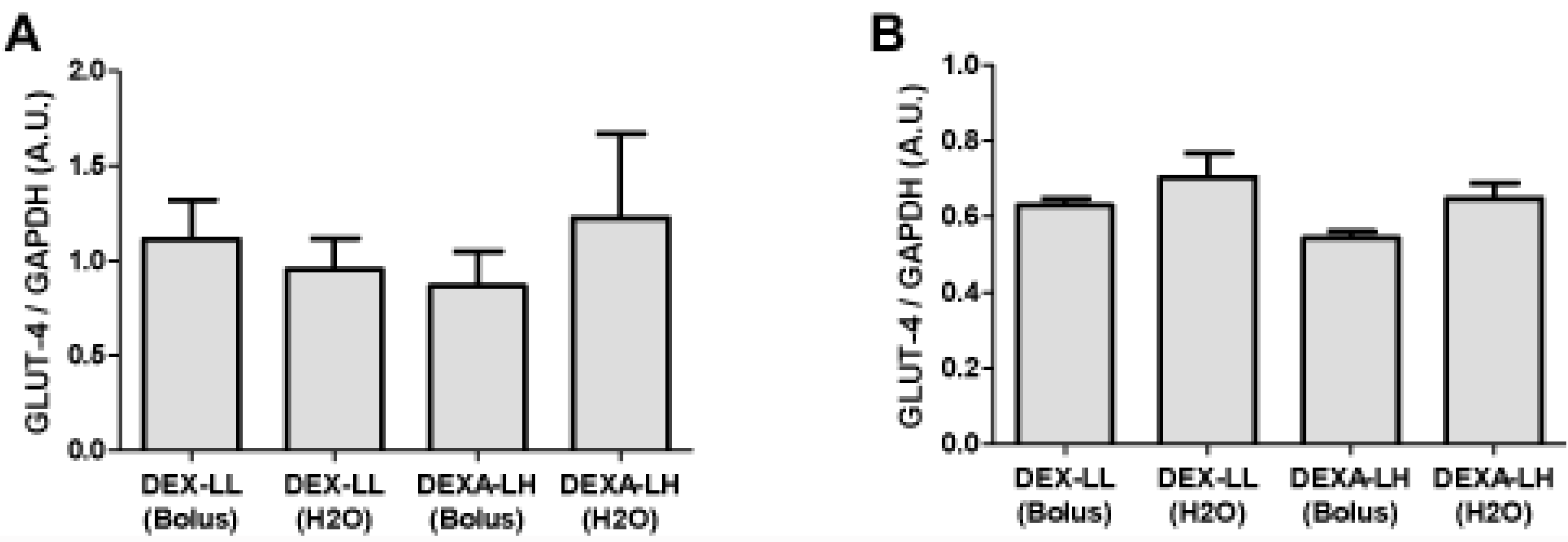
4. Discussion
5. Conclusions
Acknowledgments
Conflict of Interest
References
- Nicastro, H.; Zanchi, N.E.; da Luz, C.R.; de Moraes, W.M.A.M.; Ramona, P.; de Siqueira Filho, M.A.; Chaves, D.F.S.; Medeiros, A.; Brum, P.C.; Dardevet, D.; Lancha, A.H., Jr. Effects of leucine supplementation and resistance exercise on dexamethasone-induced muscle atrophy and insulin resistance in rats. Nutrition 2012, 28, 465–471. [Google Scholar] [CrossRef]
- Yamamoto, D.; Maki, T.; Herningtyas, E.H.; Ikeshita, N.; Shibahara, H.; Sugiyama, Y.; Nakanishi, S.; Iida, K.; Iguchi, G.; Takahashi, Y.; Kaji, H.; Chihara, K.; Okimura, Y. Branched-chain amino acids protect against dexamethasone-induced soleus muscle atrophy in rats. Muscle Nerve 2010, 41, 819–827. [Google Scholar] [CrossRef]
- Nair, K.S.; Short, K.R. Hormonal and signaling role of branched-chain amino acids. J. Nutr. 2005, 135, 1547S–1552S. [Google Scholar]
- Deshmukh, A.; Salehzadeh, F.; Metayer-Coustard, S.; Fahlman, R.; Nair, K.S.; Al-Khalili, L. Post-transcriptional gene silencing of ribosomal protein S6 kinase 1 restores insulin action in leucine-treated skeletal muscle. Cell. Mol. Life Sci. 2009, 66, 1457–1466. [Google Scholar]
- Zhang, Y.; Guo, K.; LeBlanc, R.E.; Loh, D.; Schwartz, G.J.; Yu, Y.-H. Increasing dietary leucine intake reduces diet-induced obesity and improves glucose and cholesterol metabolism in mice via multimechanisms. Diabetes 2007, 56, 1647–1654. [Google Scholar] [CrossRef]
- Lynch, C.J.; Hutson, S.M.; Patson, B.J.; Vaval, A.; Vary, T.C. Tissue-specific effects of chronic dietary leucine and norleucine supplementation on protein synthesis in rats. Am. J. Physiol. Endocrinol. Metab. 2002, 283, E824–E835. [Google Scholar]
- Nagasawa, T.; Kido, T.; Yoshizawa, F.; Ito, Y.; Nishizawa, N. Rapid suppression of protein degradation in skeletal muscle after oral feeding of leucine in rats. J. Nutr. Biochem. 2002, 13, 121–127. [Google Scholar] [CrossRef]
- Zanchi, N.E.; Nicastro, H.; Lancha, A.H., Jr. Potential antiproteolytic effects of L-leucine: Observations of in vitro and in vivo studies. Nutr. Metab. (Lond.) 2008, 5, 20. [Google Scholar] [CrossRef]
- Crozier, S.J.; Kimball, S.R.; Emmert, S.W.; Anthony, J.C.; Jefferson, L.S. Oral leucine administration stimulates protein synthesis in rat skeletal muscle. J. Nutr. 2005, 135, 376–382. [Google Scholar]
- Zanchi, N.E.; Filho, M.A.; Felitti, V.; Nicastro, H.; Lorenzeti, F.M.; Lancha, A.H., Jr. Glucocorticoids: Extensive physiological actions modulated through multiple mechanisms of gene regulation. J. Cell. Physiol. 2010, 224, 311–315. [Google Scholar] [CrossRef]
- Stahn, C.; Buttgereit, F. Genomic and nongenomic effects of glucocorticoids. Nat. Clin. Pract. Rheumatol. 2008, 4, 525–533. [Google Scholar]
- Hu, Z.; Wang, H.; Lee, I.H.; Du, J.; Mitch, W.E. Endogenous glucocorticoids and impaired insulin signaling are both required to stimulate muscle wasting under pathophysiological conditions in mice. J. Clin. Invest. 2009, 119, 3059–3069. [Google Scholar]
- Saad, M.J.; Folli, F.; Kahn, J.A.; Kahn, C.R. Modulation of insulin receptor, insulin receptor substrate-1, and phosphatidylinositol 3-kinase in liver and muscle of dexamethasone-treated rats. J. Clin. Invest. 1993, 92, 2065–2072. [Google Scholar] [CrossRef]
- Anthony, J.C.; Lang, C.H.; Crozier, S.J.; Anthony, T.G.; MacLean, D.A.; Kimball, S.R.; Jefferson, L.S. Contribution of insulin to the translational control of protein synthesis in skeletal muscle by leucine. Am. J. Physiol. Endocrinol. Metab. 2002, 282, E1092–E1101. [Google Scholar]
- Nairizi, A.; She, P.; Vary, T.C.; Lynch, C.J. Leucine supplementation of drinking water does not alter susceptibility to diet-induced obesity in mice. J. Nutr. 2009, 139, 715–719. [Google Scholar] [CrossRef]
- Adami, G.F.; Cordera, R.; Andraghetti, G.; Camerini, G.B.; Marinari, G.M.; Scopinaro, N. Changes in serum ghrelin concentration following biliopancreatic diversion for obesity. Obes. Res. 2004, 12, 684–687. [Google Scholar]
- Anderson, K.D.; Abdul, M.; Steward, O. Quantitative assessment of deficits and recovery of forelimb motor function after cervical spinal cord injury in mice. Exp. Neurol. 2004, 190, 184–191. [Google Scholar]
- Kennel, P.F.; Fonteneau, P.; Martin, E.; Schmidt, J.M.; Azzouz, M.; Borg, J.; Guenet, J.L.; Schmalbruch, H.; Warter, J.M.; Poindron, P. Electromyographical and motor performance studies in the pmn mouse model of neurodegenerative disease. Neurobiol. Dis. 1996, 3, 137–147. [Google Scholar]
- Vieira, N.M.; Bueno, C.R., Jr.; Brandalise, V.; Moraes, L.V.; Zucconi, E.; Secco, M.; Suzuki, M.F.; Camargo, M.M.; Bartolini, P.; Brum, P.C.; Vainzof, M.; Zatz, M. SJL dystrophic mice express a significant amount of human muscle proteins following systemic delivery of human adipose-derived stromal cells without immunosuppression. Stem Cells 2008, 26, 2391–2398. [Google Scholar]
- Nicastro, H.; Artioli, G.G.; Costa Ados, S.; Solis, M.Y.; da Luz, C.R.; Blachier, F.; Lancha, A.H., Jr. An overview of the therapeutic effects of leucine supplementation on skeletal muscle under atrophic conditions. Amino Acids 2011, 40, 287–300. [Google Scholar]
- Layman, D.K. The role of leucine in weight loss diets and glucose homeostasis. J. Nutr. 2003, 133, 261S–267S. [Google Scholar]
- Sun, X.; Zemel, M.B. Leucine modulation of mitochondrial mass and oxygen consumption in skeletal muscle cells and adipocytes. Nutr. Metab. (Lond.) 2009, 6, 26. [Google Scholar] [CrossRef]
- Guo, K.; Yu, Y.-H.; Hou, J.; Zhang, Y. Chronic leucine supplementation improves glycemic control in etiologically distinct mouse models of obesity and diabetes mellitus. Nutr. Metab. (Lond.) 2010, 7, 57. [Google Scholar] [CrossRef]
- Huang, S.; Czech, M.P. The GLUT4 glucose transporter. Cell Metab. 2007, 5, 237–252. [Google Scholar] [CrossRef]
- Almon, R.R.; Dubois, D.C. Fiber-type discrimination in disuse and glucocorticoid-induced atrophy. Med. Sci. Sports Exerc. 1990, 22, 304–311. [Google Scholar]
- Doi, M.; Yamaoka, I.; Fukunaga, T.; Nakayama, M. Isoleucine, a potent plasma glucose-lowering amino acid, stimulates glucose uptake in C2C12 myotubes. Biochem. Biophys. Res. Commun. 2003, 312, 1111–1117. [Google Scholar] [CrossRef]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Zanchi, N.E.; Guimarães-Ferreira, L.; De Siqueira-Filho, M.A.; Felitti, V.; Nicastro, H.; Bueno, C., Jr.; Lira, F.S.; Naimo, M.A.; Campos-Ferraz, P.; Nunes, M.T.; et al. Dose and Latency Effects of Leucine Supplementation in Modulating Glucose Homeostasis: Opposite Effects in Healthy and Glucocorticoid-Induced Insulin-Resistance States. Nutrients 2012, 4, 1851-1867. https://doi.org/10.3390/nu4121851
Zanchi NE, Guimarães-Ferreira L, De Siqueira-Filho MA, Felitti V, Nicastro H, Bueno C Jr., Lira FS, Naimo MA, Campos-Ferraz P, Nunes MT, et al. Dose and Latency Effects of Leucine Supplementation in Modulating Glucose Homeostasis: Opposite Effects in Healthy and Glucocorticoid-Induced Insulin-Resistance States. Nutrients. 2012; 4(12):1851-1867. https://doi.org/10.3390/nu4121851
Chicago/Turabian StyleZanchi, Nelo Eidy, Lucas Guimarães-Ferreira, Mário Alves De Siqueira-Filho, Vitor Felitti, Humberto Nicastro, Carlos Bueno, Jr., Fábio Santos Lira, Marshall Alan Naimo, Patrícia Campos-Ferraz, Maria Tereza Nunes, and et al. 2012. "Dose and Latency Effects of Leucine Supplementation in Modulating Glucose Homeostasis: Opposite Effects in Healthy and Glucocorticoid-Induced Insulin-Resistance States" Nutrients 4, no. 12: 1851-1867. https://doi.org/10.3390/nu4121851
APA StyleZanchi, N. E., Guimarães-Ferreira, L., De Siqueira-Filho, M. A., Felitti, V., Nicastro, H., Bueno, C., Jr., Lira, F. S., Naimo, M. A., Campos-Ferraz, P., Nunes, M. T., Seelaender, M., De Oliveira Carvalho, C. R., Blachier, F., & Lancha, A. H., Jr. (2012). Dose and Latency Effects of Leucine Supplementation in Modulating Glucose Homeostasis: Opposite Effects in Healthy and Glucocorticoid-Induced Insulin-Resistance States. Nutrients, 4(12), 1851-1867. https://doi.org/10.3390/nu4121851




