Impact of Synbiotic Intake on Liver Metabolism in Metabolically Healthy Participants and Its Potential Preventive Effect on Metabolic-Dysfunction-Associated Fatty Liver Disease (MAFLD): A Randomized, Placebo-Controlled, Double-Blinded Clinical Trial
Abstract
1. Introduction
2. Material and Methods
2.1. Ethical and Open Science Consideration
2.2. Study Design and Intervention
2.3. Participants
2.4. Anthropometrics and Categorization
- Age 20–39: physiological body fat mass: ≤20%, elevated body fat mass: >20%.
- Age 40–59: physiological body fat mass: ≤22%, elevated body fat mass: >22%.
- Age 60–79: physiological body fat mass: ≤25%, elevated body fat mass: >25%.
2.5. Dietary Intake
2.6. Blood Sample Processing and Analysis
2.7. Gut Microbiome Sample Processing and Analysis
2.8. Statistical Analysis
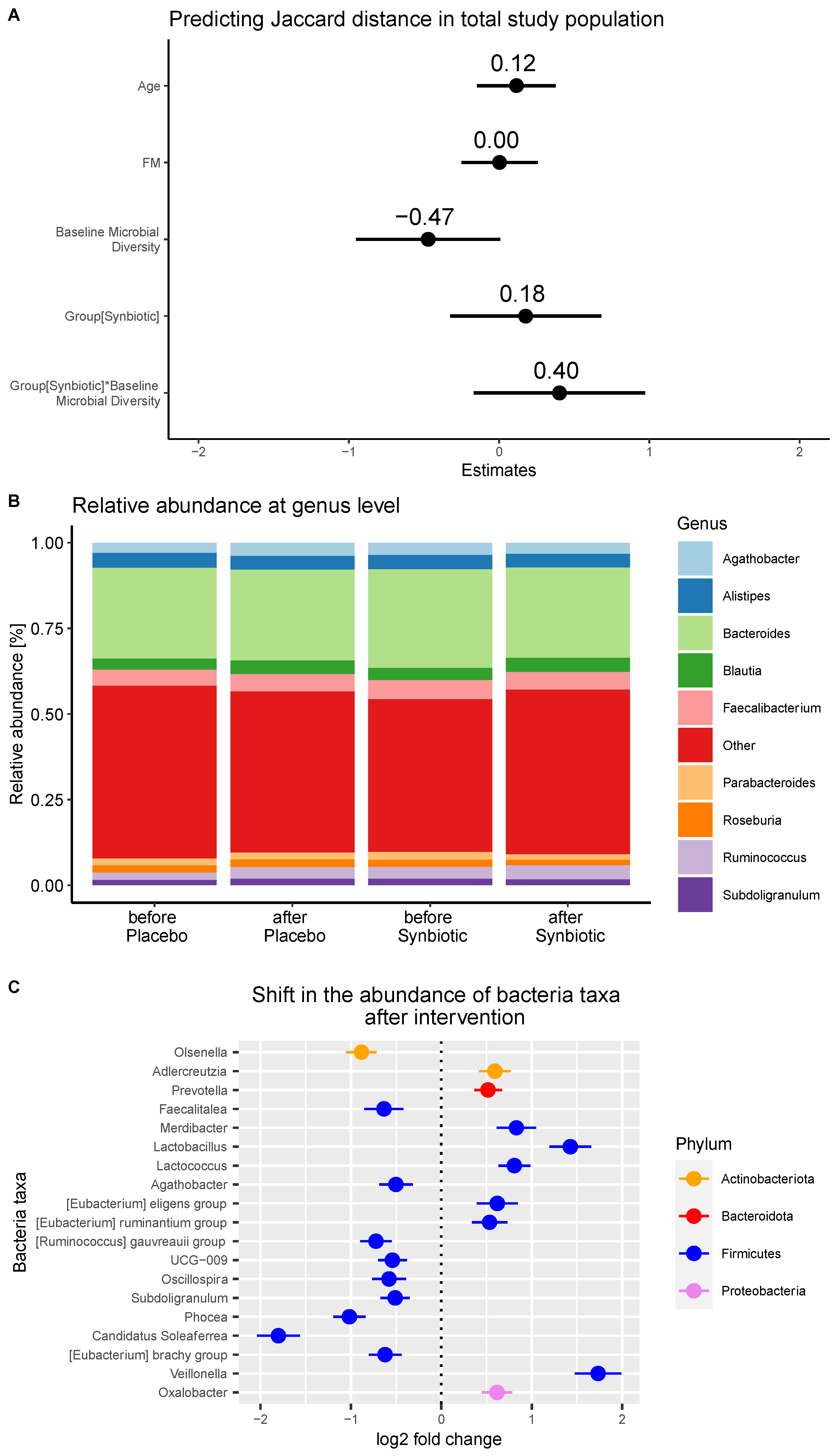
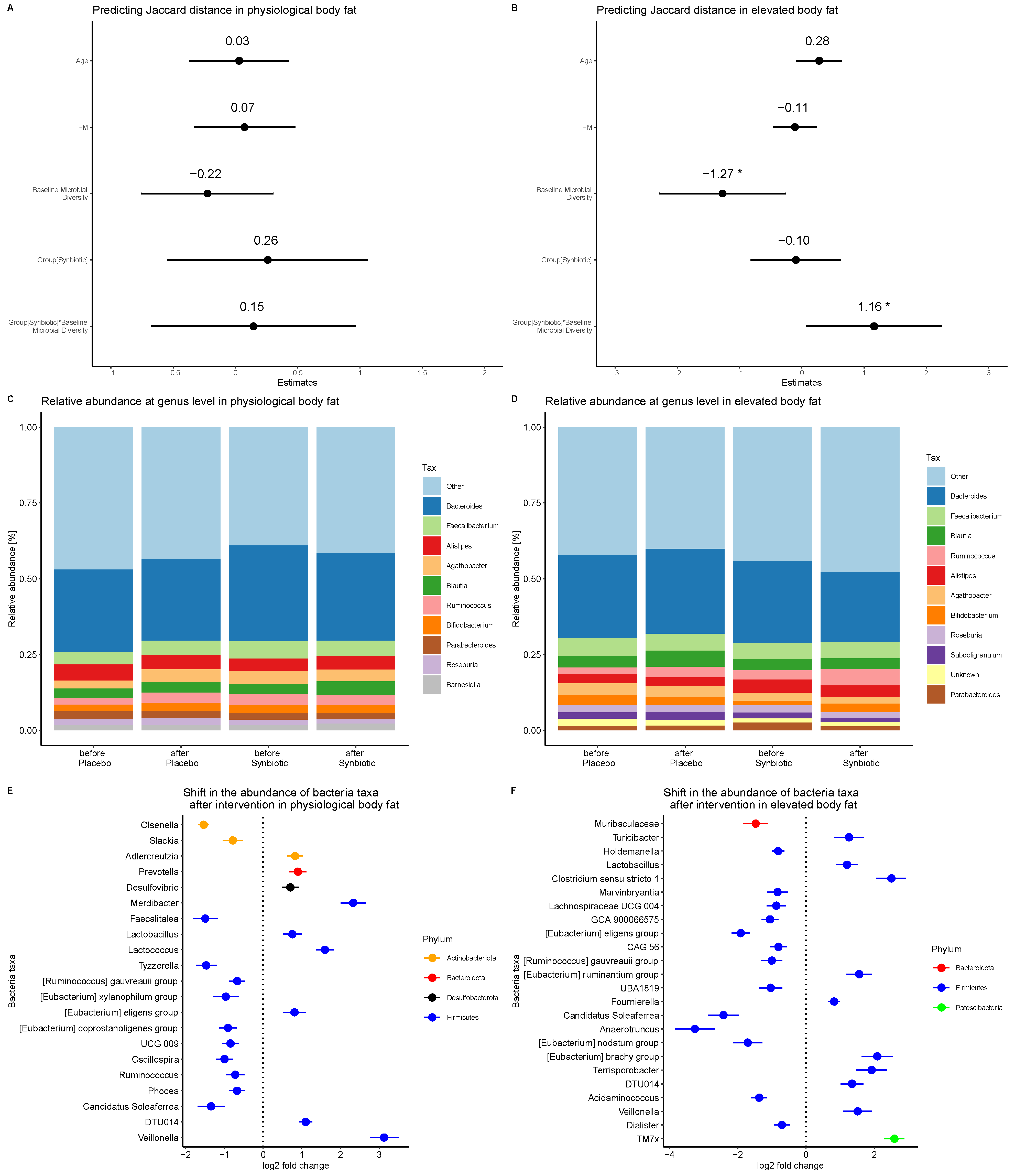
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Raman, M.; Ahmed, I.; Gillevet, P.M.; Probert, C.S.; Ratcliffe, N.M.; Smith, S.; Greenwood, R.; Sikaroodi, M.; Lam, V.; Crotty, P.; et al. Fecal Microbiome and Volatile Organic Compound Metabolome in Obese Humans with Nonalcoholic Fatty Liver Disease. Clin. Gastroenterol. Hepatol. 2013, 11, 868–875.e3. [Google Scholar] [CrossRef] [PubMed]
- Safari, Z.; Gérard, P. The Links between the Gut Microbiome and Non-Alcoholic Fatty Liver Disease (NAFLD). Cell. Mol. Life Sci. 2019, 76, 1541–1558. [Google Scholar] [CrossRef]
- European Association for the Study of the Liver (EASL); European Association for the Study of Diabetes (EASD); European Association for the Study of Obesity (EASO). EASL-EASD-EASO Clinical Practice Guidelines for the Management of Non-Alcoholic Fatty Liver Disease. J. Hepatol. 2016, 64, 1388–1402. [Google Scholar] [CrossRef] [PubMed]
- Younossi, Z.M.; Golabi, P.; de Avila, L.; Paik, J.M.; Srishord, M.; Fukui, N.; Qiu, Y.; Burns, L.; Afendy, A.; Nader, F. The Global Epidemiology of NAFLD and NASH in Patients with Type 2 Diabetes: A Systematic Review and Meta-Analysis. J. Hepatol. 2019, 71, 793–801. [Google Scholar] [CrossRef] [PubMed]
- Drobne, D.; Kurent, T.; Golob, S.; Svegl, P.; Rajar, P.; Terzic, S.; Kozelj, M.; Novak, G.; Smrekar, N.; Plut, S.; et al. Success and Safety of High Infliximab Trough Levels in Inflammatory Bowel Disease. Scand. J. Gastroenterol. 2018, 53, 940–946. [Google Scholar] [CrossRef] [PubMed]
- Crommen, S.; Rheinwalt, K.P.; Plamper, A.; Simon, M.; Rösler, D.; Fimmers, R.; Egert, S.; Metzner, C. A Specifically Tailored Multistrain Probiotic and Micronutrient Mixture Affects Nonalcoholic Fatty Liver Disease—Related Markers in Patients with Obesity after Mini Gastric Bypass Surgery. J. Nutr. 2022, 152, 408–418. [Google Scholar] [CrossRef] [PubMed]
- Mardinoglu, A.; Wu, H.; Bjornson, E.; Zhang, C.; Hakkarainen, A.; Räsänen, S.M.; Lee, S.; Mancina, R.M.; Bergentall, M.; Pietiläinen, K.H.; et al. An Integrated Understanding of the Rapid Metabolic Benefits of a Carbohydrate-Restricted Diet on Hepatic Steatosis in Humans. Cell Metab. 2018, 27, 559–571.e5. [Google Scholar] [CrossRef] [PubMed]
- Chuaypen, N.; Asumpinawong, A.; Sawangsri, P.; Khamjerm, J.; Iadsee, N.; Jinato, T.; Sutheeworapong, S.; Udomsawaengsup, S.; Tangkijvanich, P. Gut Microbiota in Patients with Non-Alcoholic Fatty Liver Disease without Type 2 Diabetes: Stratified by Body Mass Index. Int. J. Mol. Sci. 2024, 25, 1807. [Google Scholar] [CrossRef] [PubMed]
- Ley, R.E.; Turnbaugh, P.J.; Klein, S.; Gordon, J.I. Human Gut Microbes Associated with Obesity. Nature 2006, 444, 1022–1023. [Google Scholar] [CrossRef]
- Wu, C.; Yuan, Y.; Liu, H.; Li, S.; Zhang, B.; Chen, W.; An, Z.; Chen, S.; Wu, Y.; Han, B.; et al. Epidemiologic Relationship between Periodontitis and Type 2 Diabetes Mellitus. BMC Oral Health 2020, 20, 204. [Google Scholar] [CrossRef]
- Le Chatelier, E.; Nielsen, T.; Qin, J.; Prifti, E.; Hildebrand, F.; Falony, G.; Almeida, M.; Arumugam, M.; Batto, J.M.; Kennedy, S.; et al. Richness of Human Gut Microbiome Correlates with Metabolic Markers. Nature 2013, 500, 541–546. [Google Scholar] [CrossRef] [PubMed]
- Bäckhed, F.; Ley, R.E.; Sonnenburg, J.L.; Peterson, D.A.; Gordont, J.I.; Backhed, F. Host-Bacterial Mutualism in the Human Intestine. Science 2005, 307, 1915–1920. [Google Scholar] [CrossRef] [PubMed]
- Cani, P.D.; Possemiers, S.; Van de Wiele, T.; Guiot, Y.; Everard, A.; Rottier, O.; Geurts, L.; Naslain, D.; Neyrinck, A.; Lambert, D.M.; et al. Changes in Gut Microbiota Control Inflammation in Obese Mice through a Mechanism Involving GLP-2-Driven Improvement of Gut Permeability. Gut 2009, 58, 1091–1103. [Google Scholar] [CrossRef] [PubMed]
- Cani, P.D.; Bibiloni, R.; Knauf, C.; Waget, A.; Neyrinck, A.M.; Delzenne, N.M.; Burcelin, R. Changes in Gut Microbiota Control Metabolic Endotoxemia-Induced Inflammation in High-Fat Diet-Induced Obesity and Diabetes in Mice. Diabetes 2008, 57, 1470–1481. [Google Scholar] [CrossRef] [PubMed]
- Genser, L.; Aguanno, D.; Soula, H.A.; Dong, L.; Trystram, L.; Assmann, K.; Salem, J.-E.; Vaillant, J.-C.; Oppert, J.-M.; Laugerette, F.; et al. Increased Jejunal Permeability in Human Obesity Is Revealed by a Lipid Challenge and Is Linked to Inflammation and Type 2 Diabetes. J. Pathol. 2018, 246, 217–230. [Google Scholar] [CrossRef] [PubMed]
- Stolfi, C.; Maresca, C.; Monteleone, G.; Laudisi, F. Implication of Intestinal Barrier Dysfunction in Gut Dysbiosis and Diseases. Biomedicines 2022, 10, 289. [Google Scholar] [CrossRef] [PubMed]
- Kolodziejczyk, A.A.; Zheng, D.; Shibolet, O.; Elinav, E. The Role of the Microbiome in NAFLD and NASH. EMBO Mol. Med. 2019, 11, e9302. [Google Scholar] [CrossRef] [PubMed]
- Phavichitr, N.; Wang, S.; Chomto, S.; Tantibhaedhyangkul, R.; Kakourou, A.; Intarakhao, S.; Jongpiputvanich, S.; Wongteerasut, A.; Ben-Amor, K.; Martin, R.; et al. Impact of Synbiotics on Gut Microbiota during Early Life: A Randomized, Double-Blind Study. Sci. Rep. 2021, 11, 3534. [Google Scholar] [CrossRef] [PubMed]
- Lagkouvardos, I.; Intze, E.; Schaubeck, M.; Rooney, J.P.; Hecht, C.; Piloquet, H.; Clavel, T. Early Life Gut Microbiota Profiles Linked to Synbiotic Formula Effects: A Randomized Clinical Trial in European Infants. Am. J. Clin. Nutr. 2023, 117, 326–339. [Google Scholar] [CrossRef]
- Ayob, N.; Muhammad Nawawi, K.N.; Mohamad Nor, M.H.; Raja Ali, R.A.; Ahmad, H.F.; Oon, S.F.; Mohd Mokhtar, N. The Effects of Probiotics on Small Intestinal Microbiota Composition, Inflammatory Cytokines and Intestinal Permeability in Patients with Non-Alcoholic Fatty Liver Disease. Biomedicines 2023, 11, 640. [Google Scholar] [CrossRef]
- Zhang, T.; Zhang, W.; Feng, C.; Kwok, L.-Y.; He, Q.; Sun, Z. Stronger Gut Microbiome Modulatory Effects by Postbiotics than Probiotics in a Mouse Colitis Model. NPJ Sci. Food 2022, 6, 53. [Google Scholar] [CrossRef] [PubMed]
- Swanson, K.S.; Gibson, G.R.; Hutkins, R.; Reimer, R.A.; Reid, G.; Verbeke, K.; Scott, K.P.; Holscher, H.D.; Azad, M.B.; Delzenne, N.M.; et al. The International Scientific Association for Probiotics and Prebiotics (ISAPP) Consensus Statement on the Definition and Scope of Synbiotics. Nat. Rev. Gastroenterol. Hepatol. 2020, 17, 687–701. [Google Scholar] [CrossRef] [PubMed]
- Ipar, N.; Aydogdu, S.D.; Yildirim, G.K.; Inal, M.; Gies, I.; Vandenplas, Y.; Dinleyici, E.C. Effects of Synbiotic on Anthropometry, Lipid Profile and Oxidative Stress in Obese Children. Benef. Microbes 2015, 6, 775–781. [Google Scholar] [CrossRef] [PubMed]
- Safavi, M.; Farajian, S.; Kelishadi, R.; Mirlohi, M.; Hashemipour, M. The Effects of Synbiotic Supplementation on Some Cardio-Metabolic Risk Factors in Overweight and Obese Children: A Randomized Triple-Masked Controlled Trial. Int. J. Food Sci. Nutr. 2013, 64, 687–693. [Google Scholar] [CrossRef] [PubMed]
- Sanchez, M.; Darimont, C.; Drapeau, V.; Emady-Azar, S.; Lepage, M.; Rezzonico, E.; Ngom-Bru, C.; Berger, B.; Philippe, L.; Ammon-Zuffrey, C.; et al. Effect of Lactobacillus rhamnosus CGMCC1.3724 Supplementation on Weight Loss and Maintenance in Obese Men and Women. Br. J. Nutr. 2014, 111, 1507–1519. [Google Scholar] [CrossRef] [PubMed]
- Eslamparast, T.; Zamani, F.; Hekmatdoost, A.; Sharafkhah, M.; Eghtesad, S.; Malekzadeh, R.; Poustchi, H. Effects of Synbiotic Supplementation on Insulin Resistance in Subjects with the Metabolic Syndrome: A Randomised, Double-Blind, Placebo-Controlled Pilot Study. Br. J. Nutr. 2014, 112, 438–445. [Google Scholar] [CrossRef] [PubMed]
- Shakeri, H.; Hadaegh, H.; Abedi, F.; Tajabadi-Ebrahimi, M.; Mazroii, N.; Ghandi, Y.; Asemi, Z. Consumption of Synbiotic Bread Decreases Triacylglycerol and VLDL Levels While Increasing HDL Levels in Serum from Patients with Type-2 Diabetes. Lipids 2014, 49, 695–701. [Google Scholar] [CrossRef] [PubMed]
- Fujimori, S.; Gudis, K.; Mitsui, K.; Seo, T.; Yonezawa, M.; Tanaka, S.; Tatsuguchi, A.; Sakamoto, C. A Randomized Controlled Trial on the Efficacy of Synbiotic versus Probiotic or Prebiotic Treatment to Improve the Quality of Life in Patients with Ulcerative Colitis. Nutrition 2009, 25, 520–525. [Google Scholar] [CrossRef] [PubMed]
- Ustundag, G.H.; Altuntas, H.; Soysal, Y.D.; Kokturk, F. The Effects of Synbiotic “Bifidobacterium lactis B94 plus Inulin” Addition on Standard Triple Therapy of Helicobacter Pylori Eradication in Children. Can. J. Gastroenterol. Hepatol. 2017, 2017, 8130596. [Google Scholar] [CrossRef]
- Romero-Gómez, M.; Zelber-Sagi, S.; Trenell, M. Treatment of NAFLD with Diet, Physical Activity and Exercise. J. Hepatol. 2017, 67, 829–846. [Google Scholar] [CrossRef]
- Sayari, S.; Neishaboori, H.; Jameshorani, M. Combined Effects of Synbiotic and Sitagliptin versus Sitagliptin Alone in Patients with Nonalcoholic Fatty Liver Disease. Clin. Mol. Hepatol. 2018, 24, 331–338. [Google Scholar] [CrossRef] [PubMed]
- Eslamparast, T.; Poustchi, H.; Zamani, F.; Sharafkhah, M.; Malekzadeh, R.; Hekmatdoost, A. Synbiotic Supplementation in Nonalcoholic Fatty Liver Disease: A Randomized, Double-Blind, Placebo-Controlled Pilot Study. Am. J. Clin. Nutr. 2014, 99, 535–542. [Google Scholar] [CrossRef] [PubMed]
- Olas, B. Probiotics, Prebiotics and Synbiotics-A Promising Strategy in Prevention and Treatment of Cardiovascular Diseases? Int. J. Mol. Sci. 2020, 21, 9737. [Google Scholar] [CrossRef] [PubMed]
- Sáez-Lara, M.J.; Robles-Sanchez, C.; Ruiz-Ojeda, F.J.; Plaza-Diaz, J.; Gil, A. Effects of Probiotics and Synbiotics on Obesity, Insulin Resistance Syndrome, Type 2 Diabetes and Non-Alcoholic Fatty Liver Disease: A Review of Human Clinical Trials. Int. J. Mol. Sci. 2016, 17, 928. [Google Scholar] [CrossRef] [PubMed]
- Gallagher, D.; Heymsfield, S.B.; Heo, M.; Jebb, S.A.; Murgatroyd, P.R.; Sakamoto, Y. Healthy percentage body fat ranges: An approach for developing guidelines based on body mass index. Am. J. Clin. Nutr. 2000, 72, 694–701. [Google Scholar] [CrossRef]
- Bolyen, E.; Rideout, J.R.; Dillon, M.R.; Bokulich, N.A.; Abnet, C.C.; Al-Ghalith, G.A.; Alexander, H.; Alm, E.J.; Arumugam, M.; Asnicar, F.; et al. Reproducible, Interactive, Scalable and Extensible Microbiome Data Science Using QIIME 2. Nat. Biotechnol. 2019, 37, 852–857. [Google Scholar] [CrossRef] [PubMed]
- Callahan, B.J.; McMurdie, P.J.; Rosen, M.J.; Han, A.W.; Johnson, A.J.A.; Holmes, S.P. DADA2: High-Resolution Sample Inference from Illumina Amplicon Data. Nat. Methods 2016, 13, 581–583. [Google Scholar] [CrossRef] [PubMed]
- Robeson, M.S., II; O’Rourke, D.R.; Kaehler, B.D.; Ziemski, M.; Dillon, M.R.; Foster, J.T.; Bokulich, N.A. RESCRIPt: Reproducible Sequence Taxonomy Reference Database Management for the Masses. bioRxiv 2020. [Google Scholar] [CrossRef]
- Bokulich, N.A.; Kaehler, B.D.; Rideout, J.R.; Dillon, M.; Bolyen, E.; Knight, R.; Huttley, G.A.; Caporaso, J.G. Optimizing Taxonomic Classification of Marker-Gene Amplicon Sequences with QIIME 2’s Q2-Feature-Classifier Plugin. Microbiome 2018, 6, 90. [Google Scholar] [CrossRef]
- Wu, G.D.; Chen, J.; Hoffmann, C.; Bittinger, K.; Chen, Y.-Y.; Keilbaugh, S.A.; Bewtra, M.; Knights, D.; Walters, W.A.; Knight, R.; et al. Linking Long-Term Dietary Patterns with Gut Microbial Enterotypes. Science 2011, 334, 105–108. [Google Scholar] [CrossRef]
- Zhang, X.; Yi, N. NBZIMM: Negative Binomial and Zero-Inflated Mixed Models, with Application to Microbiome/Metagenomics Data Analysis. BMC Bioinform. 2020, 21, 488. [Google Scholar] [CrossRef] [PubMed]
- Mofidi, F.; Poustchi, H.; Yari, Z.; Nourinayyer, B.; Merat, S.; Sharafkhah, M.; Malekzadeh, R.; Hekmatdoost, A. Synbiotic Supplementation in Lean Patients with Non-Alcoholic Fatty Liver Disease: A Pilot, Randomised, Double-Blind, Placebo-Controlled, Clinical Trial. Br. J. Nutr. 2017, 117, 662–668. [Google Scholar] [CrossRef] [PubMed]
- Chalasani, N.; Younossi, Z.; Lavine, J.E.; Diehl, A.M.; Brunt, E.M.; Cusi, K.; Charlton, M.; Sanyal, A.J. The Diagnosis and Management of Non-Alcoholic Fatty Liver Disease: Practice Guideline by the American Association for the Study of Liver Diseases, American College of Gastroenterology, and the American Gastroenterological Association. Hepatology 2012, 55, 2005–2023. [Google Scholar] [CrossRef] [PubMed]
- Mundi, M.S.; Velapati, S.; Patel, J.; Kellogg, T.A.; Abu Dayyeh, B.K.; Hurt, R.T. Evolution of NAFLD and Its Management. Nutr. Clin. Pract. 2020, 35, 72–84. [Google Scholar] [CrossRef] [PubMed]
- Harrison, S.A.; Bedossa, P.; Guy, C.D.; Schattenberg, J.M.; Loomba, R.; Taub, R.; Labriola, D.; Moussa, S.E.; Neff, G.W.; Rinella, M.E.; et al. A Phase 3, Randomized, Controlled Trial of Resmetirom in NASH with Liver Fibrosis. N. Engl. J. Med. 2024, 390, 497–509. [Google Scholar] [CrossRef] [PubMed]
- Lee, N.Y.; Shin, M.J.; Youn, G.S.; Yoon, S.J.; Choi, Y.R.; Kim, H.S.; Gupta, H.; Han, S.H.; Kim, B.K.; Lee, D.Y.; et al. Lactobacillus Attenuates Progression of Nonalcoholic Fatty Liver Disease by Lowering Cholesterol and Steatosis. Clin. Mol. Hepatol. 2021, 27, 110–124. [Google Scholar] [CrossRef] [PubMed]
- Monserrat-Mesquida, M.; Bouzas, C.; Mascaró, C.M.; Tejada, S.; Sureda, A. Probiotics as Potential Therapy in the Management of Non-Alcoholic Fatty Liver Disease (NAFLD). Fermentation 2023, 9, 395. [Google Scholar] [CrossRef]
- Cheng, D.; Xie, M.Z. A Review of a Potential and Promising Probiotic Candidate-Akkermansia Muciniphila. J. Appl. Microbiol. 2021, 130, 1813–1822. [Google Scholar] [CrossRef] [PubMed]
- Juárez-Fernández, M.; Porras, D.; Petrov, P.; Román-Sagüillo, S.; García-Mediavilla, M.V.; Soluyanova, P.; Martínez-Flórez, S.; González-Gallego, J.; Nistal, E.; Jover, R.; et al. The Synbiotic Combination of Akkermansia Muciniphila and Quercetin Ameliorates Early Obesity and NAFLD through Gut Microbiota Reshaping and Bile Acid Metabolism Modulation. Antioxidants 2021, 10, 2001. [Google Scholar] [CrossRef]
- Mauvais-Jarvis, F. Sex Differences in Energy Metabolism: Natural Selection, Mechanisms and Consequences. Nat. Rev. Nephrol. 2023, 20, 56–69. [Google Scholar] [CrossRef]
- Link, J.C.; Reue, K. Genetic Basis for Sex Differences in Obesity and Lipid Metabolism. Annu. Rev. Nutr. 2017, 37, 225–245. [Google Scholar] [CrossRef] [PubMed]
- Ahn, S.B.; Jun, D.W.; Kang, B.K.; Lim, J.H.; Lim, S.; Chung, M.J. Randomized, Double-Blind, Placebo-Controlled Study of a Multispecies Probiotic Mixture in Nonalcoholic Fatty Liver Disease. Sci. Rep. 2019, 9, 5688. [Google Scholar] [CrossRef] [PubMed]
- Alah, M.A.; Abdeen, S.; Kehyayan, V.; Bougmiza, I. Impact of Staying at Home Measures during COVID-19 Pandemic on the Lifestyle of Qatar’s Population: Perceived Changes in Diet, Physical Activity, and Body Weight. Prev. Med. Rep. 2021, 24, 101545. [Google Scholar] [CrossRef] [PubMed]
- Stockwell, S.; Trott, M.; Tully, M.; Shin, J.; Barnett, Y.; Butler, L.; McDermott, D.; Schuch, F.; Smith, L. Changes in Physical Activity and Sedentary Behaviours from before to during the COVID-19 Pandemic Lockdown: A Systematic Review. BMJ Open Sport. Exerc. Med. 2021, 7, e000960. [Google Scholar] [CrossRef]
- González-Monroy, C.; Gómez-Gómez, I.; Olarte-Sánchez, C.M.; Motrico, E. Eating Behaviour Changes during the COVID-19 Pandemic: A Systematic Review of Longitudinal Studies. Int. J. Environ. Res. Public Health 2021, 18, 1130. [Google Scholar] [CrossRef]

| Total n = 86 | SYN n = 45 (52.3%) | PLA n = 41 (47.7%) | p Value | |
|---|---|---|---|---|
| Age (years) | 32 (20, 60) | 33 (20, 58) | 32 (20.4, 60.1) | 0.953 |
| Height (cm) | 181.38 (166.5, 198.4) | 180.44 (166.5, 192.0) | 182.41 (167.1, 198.4) | 0.162 |
| Weight (kg) | 84.67 (64.3, 124.3) | 84.18 (68.7, 124.3) | 85.22 (64.3, 123.1) | 0.607 |
| BMI (kg/m2) | 25.74 (20.5, 33.7) | 25.84 (20.6, 33.7) | 25.62 (20.5, 33.6) | 0.747 |
| Fat mass (%) | 19.49 (11.6, 33.2) | 19.41 (11.6, 33.2) | 19.57 (12.5, 32.1) | 0.888 |
| Physiological body fat | 49 (57.0%) | 25 (51.0%) | 24 (49.0%) | 0.853 |
| Elevated body fat | 32 (37.2%) | 17 (53.1%) | 15 (46.9%) | |
| TG (mg/dL) | 99.57 (36.0, 292.0) | 95.04 (36.0, 292.0) | 104.54 (40.0, 283.0) | 0.149 |
| Total chol (mg/dL) | 172.51 (107.0, 300.0) | 166.80 (107.0, 300.0) | 178.78 (108.0, 300.0) | 0.051 |
| LDL chol (mg/dL) | 108.06 (47.0, 237.0) | 103.69 (47.0, 237.0) | 112.85 (56.0, 237.0) | 0.119 |
| HDL chol (mg/dL) | 49.51 (28.0, 85.0) | 49.69 (28.0, 74.0) | 49.32 (28.0, 85.0) | 0.872 |
| AST (U/L) | 24.59 (12.0, 72.0) | 25.49 (12.0, 72.0) | 23.61 (13.0, 47.0) | 0.983 |
| ALT (U/L) | 29.76 (12.0, 72.0) | 31.07 (13.0, 144.0) | 28.33 (12.0, 119.0) | 0.411 |
| GGT (U/L) | 21.21 (8.0, 72.0) | 22.71 (8.0, 72.0) | 19.56 (10.0, 54.0) | 0.232 |
| PAL (U/L) | 64.13 (35.0, 110.0) | 65.49 (37.0, 110.0) | 62.63 (35.0, 95.0) | 0.369 |
| Glucose (mg/dL) | 91.67 (75.0, 136.0) | 92.00 (76.0, 136.0) | 91.32 (75.0, 105.0) | 0.832 |
| HbA1c (%) | 5.05 (4.3, 6.7) | 5.08 (4.3, 6.7) | 5.02 (4.6, 5.5) | 0.856 |
| Insulin (mU/L) | 8.98 (2.8, 19.9) | 8.80 (2.9, 19.9) | 9.19 (3.1, 18.3) | 0.557 |
| HOMA-IR | 2.08 (0.6, 5.7) | 2.05 (0.5, 5.7) | 2.11 (0.6, 4.2) | 0.565 |
| GLP-1 (pM) | 17.14 (7.0, 48.0) | 17.42 (7.0, 48.0) | 16.83 (8.0, 28.0) | 0.979 |
| hs-CRP (mg/L) | 0.90 (0.3, 5.1) | 0.81 (0.3, 3.7) | 1.00 (0.3, 5.1) | 0.258 |
| IL-6 (pg/mL) | 1.90 (1.5, 4.4) | 1.93 (1.5, 3.8) | 1.87 (1.5, 4.4) | 0.882 |
| SYN n = 45 | PLA n = 41 | p Value | |
|---|---|---|---|
| Weight (%) | −0.02 ± 1.92 | 0.03 ± 1.84 | 0.894 |
| BMI (%) | −0.02 ± 1.92 | 0.03 ± 1.84 | 0.894 |
| Fat mass (%) | −2.34 ± 8.15 | −1.51 ± 8.54 | 0.597 |
| Energy intake (%) | −5.50 ± 27.16 | −6.54 ± 20.98 | 0.845 |
| Carbohydrate intake (%) | −1.43 ± 33.45 | −8.55 ± 22.42 | 0.250 |
| Protein intake (%) | −4.45 ± 33.23 | −7.38 ± 26.43 | 0.979 |
| Fat intake (%) | −8.26 ± 32.08 | −3.81 ± 28.68 | 0.413 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mantri, A.; Köhlmoos, A.; Schelski, D.S.; Seel, W.; Stoffel-Wagner, B.; Krawitz, P.; Stehle, P.; Holst, J.J.; Weber, B.; Koban, L.; et al. Impact of Synbiotic Intake on Liver Metabolism in Metabolically Healthy Participants and Its Potential Preventive Effect on Metabolic-Dysfunction-Associated Fatty Liver Disease (MAFLD): A Randomized, Placebo-Controlled, Double-Blinded Clinical Trial. Nutrients 2024, 16, 1300. https://doi.org/10.3390/nu16091300
Mantri A, Köhlmoos A, Schelski DS, Seel W, Stoffel-Wagner B, Krawitz P, Stehle P, Holst JJ, Weber B, Koban L, et al. Impact of Synbiotic Intake on Liver Metabolism in Metabolically Healthy Participants and Its Potential Preventive Effect on Metabolic-Dysfunction-Associated Fatty Liver Disease (MAFLD): A Randomized, Placebo-Controlled, Double-Blinded Clinical Trial. Nutrients. 2024; 16(9):1300. https://doi.org/10.3390/nu16091300
Chicago/Turabian StyleMantri, Aakash, Anika Köhlmoos, Daniela Stephanie Schelski, Waldemar Seel, Birgit Stoffel-Wagner, Peter Krawitz, Peter Stehle, Jens Juul Holst, Bernd Weber, Leonie Koban, and et al. 2024. "Impact of Synbiotic Intake on Liver Metabolism in Metabolically Healthy Participants and Its Potential Preventive Effect on Metabolic-Dysfunction-Associated Fatty Liver Disease (MAFLD): A Randomized, Placebo-Controlled, Double-Blinded Clinical Trial" Nutrients 16, no. 9: 1300. https://doi.org/10.3390/nu16091300
APA StyleMantri, A., Köhlmoos, A., Schelski, D. S., Seel, W., Stoffel-Wagner, B., Krawitz, P., Stehle, P., Holst, J. J., Weber, B., Koban, L., Plassmann, H., & Simon, M.-C. (2024). Impact of Synbiotic Intake on Liver Metabolism in Metabolically Healthy Participants and Its Potential Preventive Effect on Metabolic-Dysfunction-Associated Fatty Liver Disease (MAFLD): A Randomized, Placebo-Controlled, Double-Blinded Clinical Trial. Nutrients, 16(9), 1300. https://doi.org/10.3390/nu16091300







