Protective Effects of Vitamin D on Proteoglycans of Human Articular Chondrocytes through TGF-β1 Signaling
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Cell Culture
2.3. Cell Viability Assay
2.4. ELISA Analysis
2.5. RNA Extraction and Real-Time Quantitative PCR
2.6. Immunohistochemistry and Immunofluorescence Analysis
2.7. Statistical Analysis
3. Results
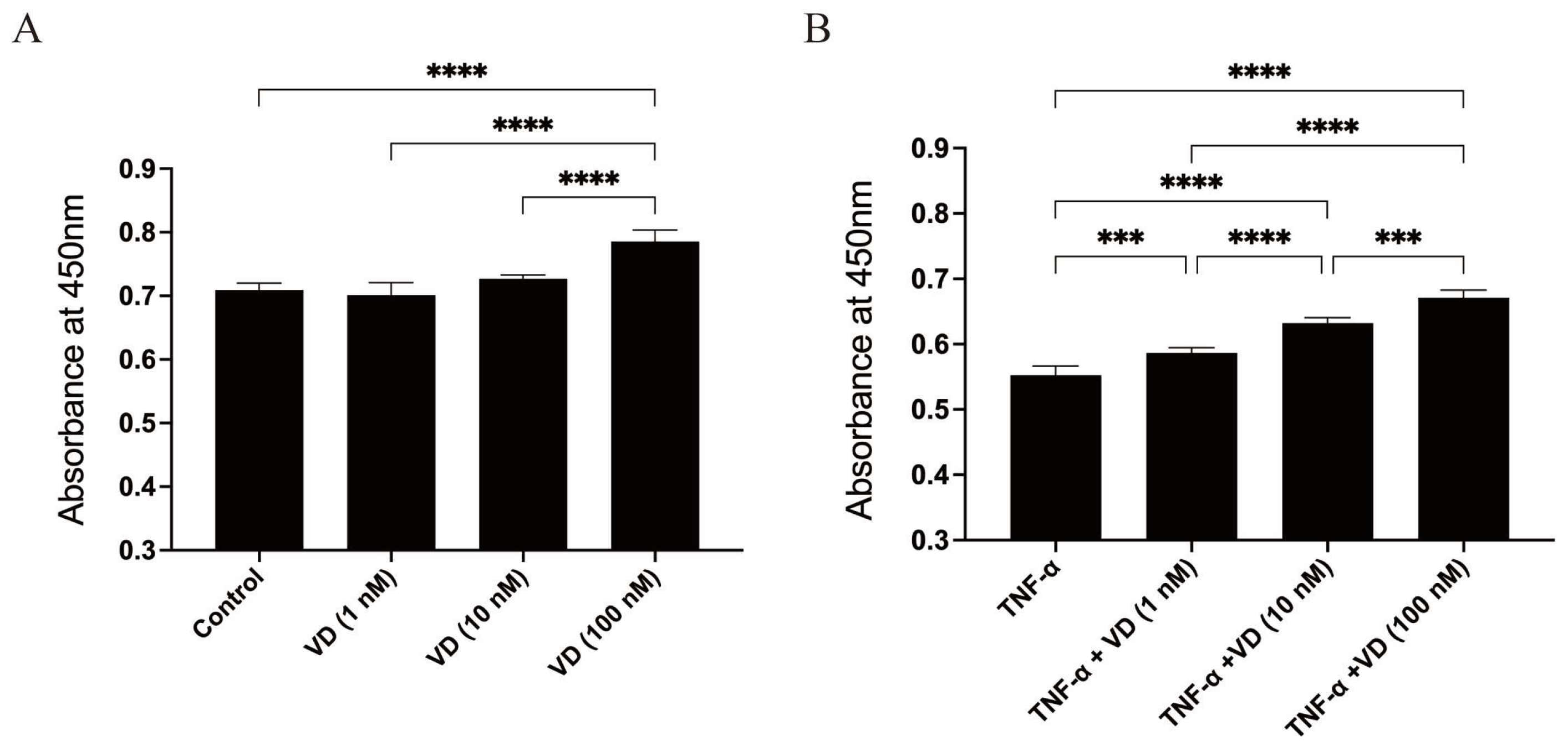
3.1. Effects of VD and TNF-α on the Viability of Human Chondrocytes
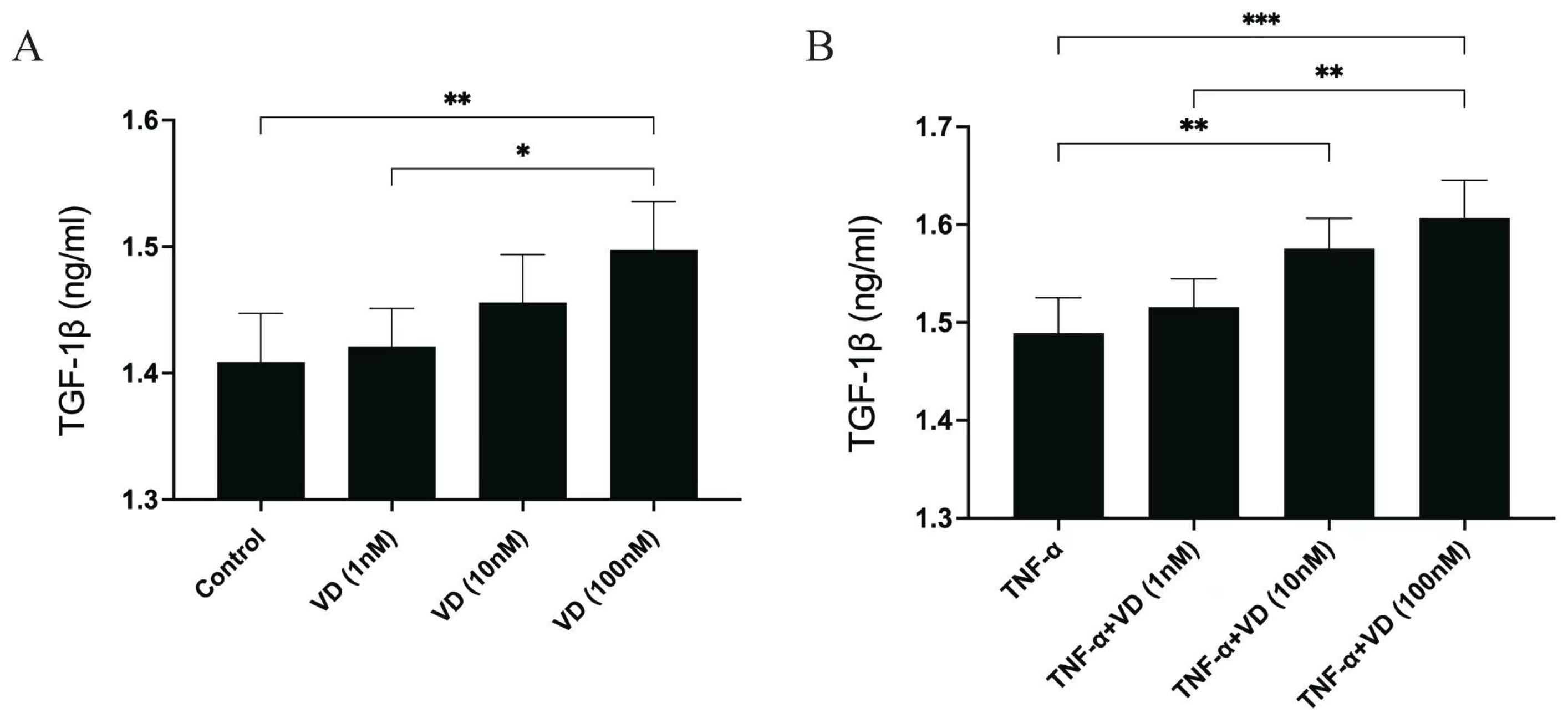
3.2. Effects of VD and TNF-α on the Release of TGF-β1
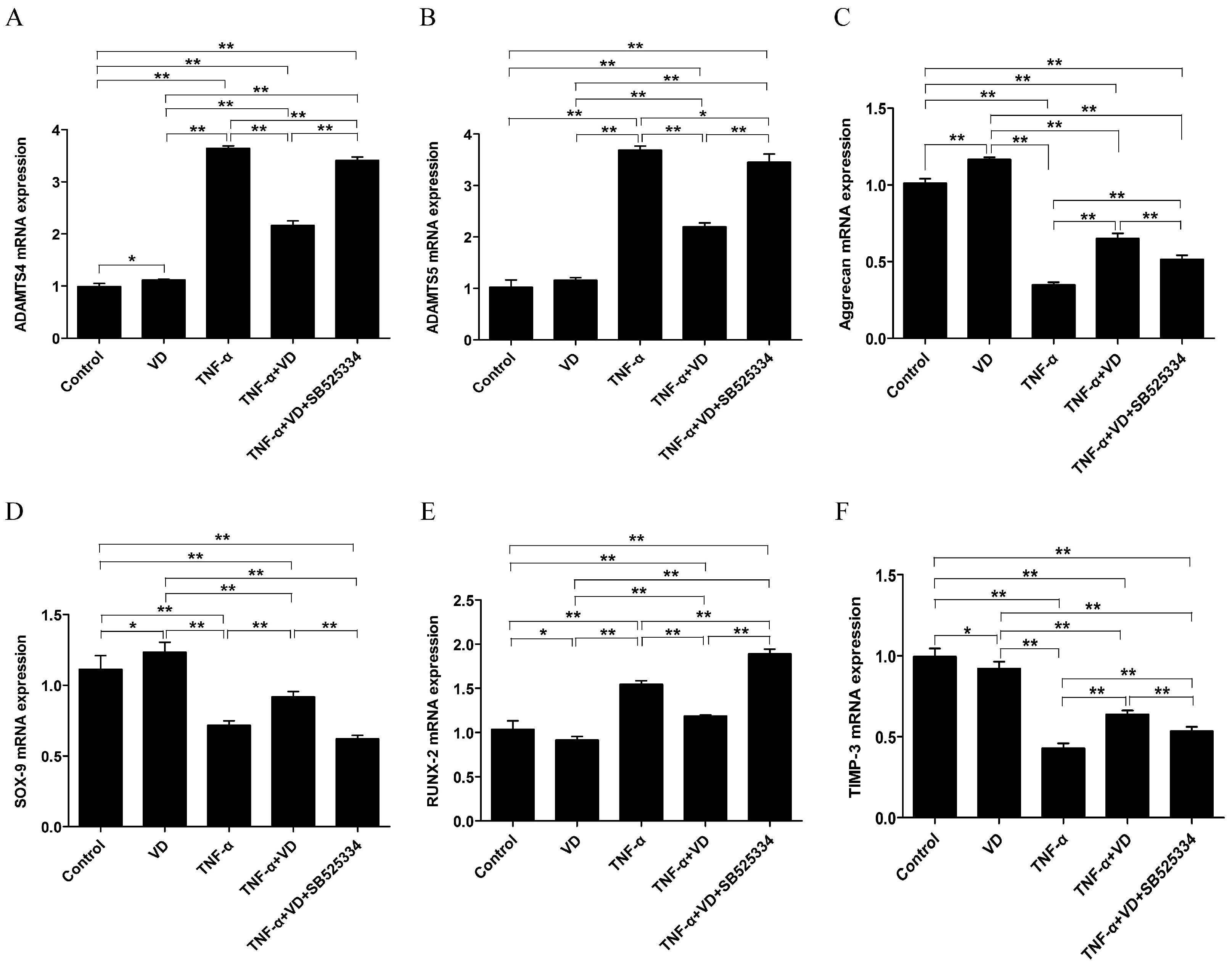
3.3. Effects of VD on the mRNA Levels of ADAMTS and Aggrecan in TNF-α Treated Human Chondrocytes
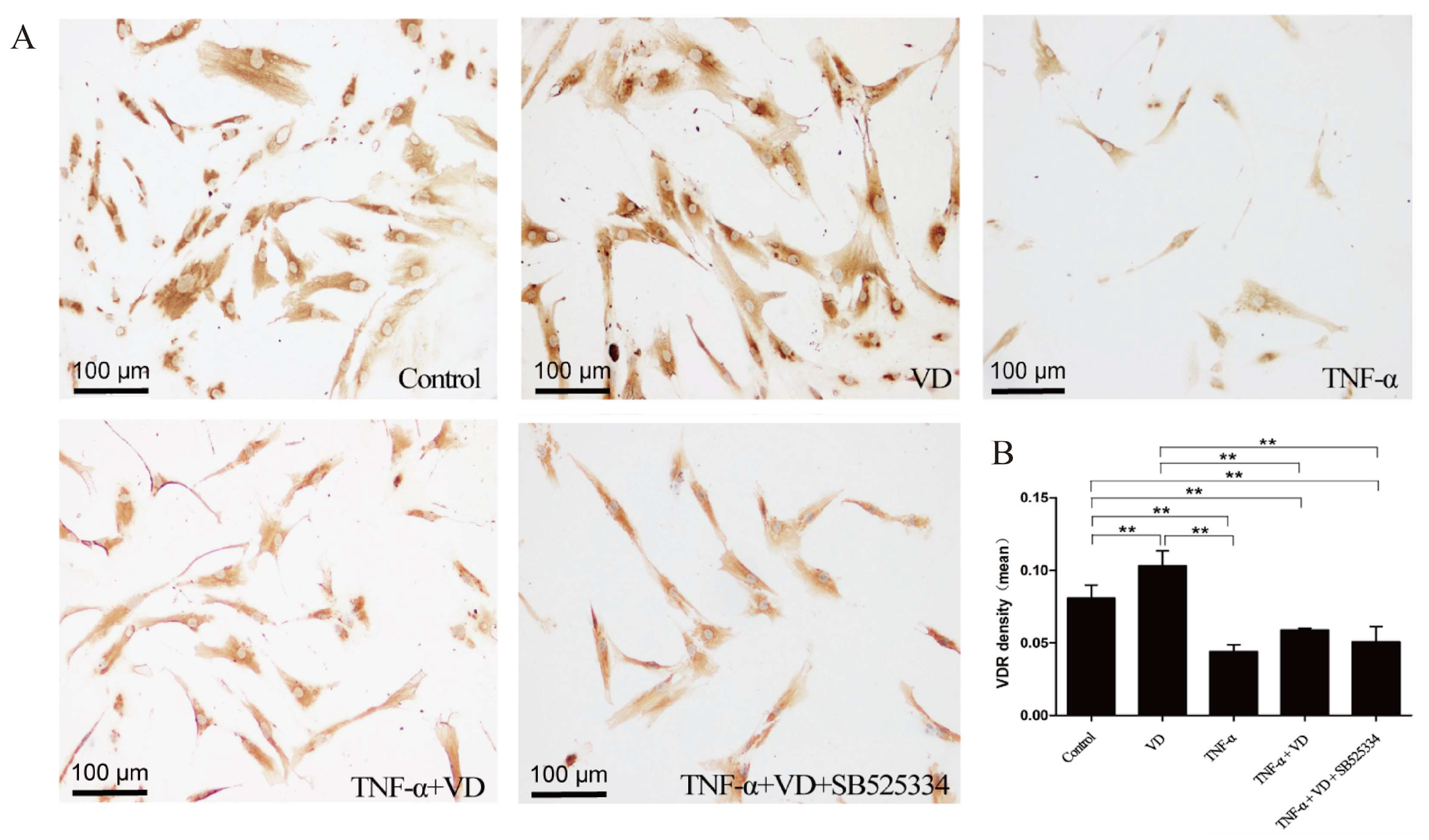
3.4. The Role of TGF-β1
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Appendix A
| Names | Primers | Sequence |
|---|---|---|
| GAPDH | Forward | 5′-TCAAGAAGGTGGTGAAGCAGG-3′ |
| Reverse | 5′-TCAAAGGTGGAGGAGTGGGT-3′ | |
| Aggrecan | Forward | 5′-TGAGCGGCAGCACTTTGAC-3′ |
| Reverse | 5′-TGAGTACAGGAGGCTTGAGG-3′ | |
| ADAMTS-4 | Forward | 5′- CAATCCTGTCAGCTTGGTGG-3′ |
| Reverse | 5′-GCTGTGTCAAAGTGGTCAGG-3′ | |
| ADAMTS-5 | Forward | 5′-CTGCCACCACACTCAAGAAC-3′ |
| Reverse | 5′-TGGAGGCCATCGTCTTCAAT-3′ | |
| SOX-9 | Forward | 5′-GAACGCACATCAAGACGGAG-3′ |
| Reverse | 5′-AGTTCTGGTGGTCGGTGTAG-3′ | |
| RUNX-2 | Forward | 5′-ATTCTGCTGAGCTCCGGAAT-3′ |
| Reverse | 5′-AGCTTCTGTCTGTGCCTTCT-3′ | |
| TIMP-3 | Forward | 5′-GTCGCGTCTATGATGGCAAG-3′ |
| Reverse | 5′-AAGCAAGGCAGGTAGTAGCA-3′ |
References
- Katz, J.N.; Arant, K.R.; Loeser, R.F. Diagnosis and Treatment of Hip and Knee Osteoarthritis: A Review. JAMA 2021, 325, 568–578. [Google Scholar] [CrossRef] [PubMed]
- Zhou, R.; Fu, W.; Vasylyev, D.; Waxman, S.G.; Liu, C.J. Ion channels in osteoarthritis: Emerging roles and potential targets. Nat. Rev. Rheumatol. 2024, 20, 545–564. [Google Scholar] [CrossRef] [PubMed]
- Yao, Q.; Wu, X.; Tao, C.; Gong, W.; Chen, M.; Qu, M.; Zhong, Y.; He, T.; Chen, S.; Xiao, G. Osteoarthritis: Pathogenic signaling pathways and therapeutic targets. Signal Transduct. Target. Ther. 2023, 8, 56. [Google Scholar] [CrossRef]
- Sao, K.; Risbud, M.V. Proteoglycan Dysfunction: A Common Link Between Intervertebral Disc Degeneration and Skeletal Dysplasia. Neurospine 2024, 21, 162–178. [Google Scholar] [CrossRef] [PubMed]
- Krawetz, R.J.; Wu, Y.E.; Bertram, K.L.; Shonak, A.; Masson, A.O.; Ren, G.; Leonard, C.; Kapoor, M.; Matyas, J.R.; Salo, P.T. Synovial mesenchymal progenitor derived aggrecan regulates cartilage homeostasis and endogenous repair capacity. Cell Death Dis. 2022, 13, 470. [Google Scholar] [CrossRef] [PubMed]
- Wilkinson, D.J.; Wang, H.; Habgood, A.; Lamb, H.K.; Thompson, P.; Hawkins, A.R.; Désilets, A.; Leduc, R.; Steinmetzer, T.; Hammami, M.; et al. Matriptase Induction of Metalloproteinase-Dependent Aggrecanolysis In Vitro and In Vivo: Promotion of Osteoarthritic Cartilage Damage by Multiple Mechanisms. Arthritis Rheumatol. 2017, 69, 1601–1611. [Google Scholar] [CrossRef]
- Li, C.; Cao, Z.; Li, W.; Liu, R.; Chen, Y.; Song, Y.; Liu, G.; Song, Z.; Liu, Z.; Lu, C.; et al. A review on the wide range applications of hyaluronic acid as a promising rejuvenating biomacromolecule in the treatments of bone related diseases. Int. J. Biol. Macromol. 2020, 165, 1264–1275. [Google Scholar] [CrossRef]
- Bouillon, R.; Marcocci, C.; Carmeliet, G.; Bikle, D.; White, J.H.; Dawson-Hughes, B.; Lips, P.; Munns, C.F.; Lazaretti-Castro, M.; Giustina, A.; et al. Skeletal and Extraskeletal Actions of Vitamin D: Current Evidence and Outstanding Questions. Endocr. Rev. 2019, 40, 1109–1151. [Google Scholar] [CrossRef]
- Chen, J.; Zhang, J.; Li, J.; Qin, R.; Lu, N.; Goltzman, D.; Miao, D.; Yang, R. 1,25-Dihydroxyvitamin D Deficiency Accelerates Aging-related Osteoarthritis via Downregulation of Sirt1 in Mice. Int. J. Biol. Sci. 2023, 19, 610–624. [Google Scholar]
- Li, S.; Niu, G.; Wu, Y.; Du, G.; Huang, C.; Yin, X.; Liu, Z.; Song, C.; Leng, H. Vitamin D prevents articular cartilage erosion by regulating collagen II turnover through TGF-β1 in ovariectomized rats. Osteoarthr. Cartil. 2016, 24, 345–353. [Google Scholar]
- Apostu, D.; Lucaciu, O.; Mester, A.; Oltean-Dan, D.; Baciut, M.; Baciut, G.; Bran, S.; Onisor, F.; Piciu, A.; Pasca, R.D.; et al. Systemic drugs with impact on osteoarthritis. Drug Metab. Rev. 2019, 51, 498–523. [Google Scholar] [CrossRef] [PubMed]
- Boyan, B.D.; Hyzy, S.L.; Pan, Q.; Scott, K.M.; Coutts, R.D.; Healey, R.; Schwartz, Z. 24R,25-Dihydroxyvitamin D3 Protects against Articular Cartilage Damage following Anterior Cruciate Ligament Transection in Male Rats. PLoS ONE 2016, 11, e0161782. [Google Scholar] [CrossRef] [PubMed]
- van Loo, G.; Bertrand, M.J.M. Death by TNF: A road to inflammation. Nat. Rev. Immunol. 2023, 23, 289–303. [Google Scholar] [CrossRef] [PubMed]
- Xiang, X.; Zhou, Y.; Sun, H.; Tan, S.; Lu, Z.; Huang, L.; Wang, W. Ivabradine abrogates TNF-α-induced degradation of articular cartilage matrix. Int. Immunopharmacol. 2019, 66, 347–353. [Google Scholar] [CrossRef]
- Hu, S.L.; Mamun, A.A.; Shaw, J.; Li, S.L.; Shi, Y.F.; Jin, X.M.; Yu, Y.X.; Pang, C.Z.; Li, Z.Y.; Lu, J.J.; et al. TBK1-medicated DRP1 phosphorylation orchestrates mitochondrial dynamics and autophagy activation in osteoarthritis. Acta Pharmacol. Sin. 2023, 44, 610–621. [Google Scholar] [CrossRef]
- Tang, H.; Gong, X.; Dai, J.; Gu, J.; Dong, Z.; Xu, Y.; Hu, Z.; Zhao, C.; Deng, J.; Dong, S. The IRF1/GBP5 axis promotes osteoarthritis progression by activating chondrocyte pyroptosis. J. Orthop. Transl. 2023, 44, 47–59. [Google Scholar] [CrossRef]
- Guo, P.; Gao, X.; Nelson, A.L.; Huard, M.; Lu, A.; Hambright, W.S.; Huard, J. TIPE2 gene transfer ameliorates aging-associated osteoarthritis in a progeria mouse model by reducing inflammation and cellular senescence. Mol. Ther. J. Am. Soc. Gene Ther. 2024. [Google Scholar]
- Liu, S.C.; Tsai, C.H.; Wang, Y.H.; Su, C.M.; Wu, H.C.; Fong, Y.C.; Yang, S.F.; Tang, C.H. Melatonin abolished proinflammatory factor expression and antagonized osteoarthritis progression in vivo. Cell Death Dis. 2022, 13, 215. [Google Scholar] [CrossRef]
- Liao, C.R.; Wang, S.N.; Zhu, S.Y.; Wang, Y.Q.; Li, Z.Z.; Liu, Z.Y.; Jiang, W.S.; Chen, J.T.; Wu, Q. Advanced oxidation protein products increase TNF-α and IL-1β expression in chondrocytes via NADPH oxidase 4 and accelerate cartilage degeneration in osteoarthritis progression. Redox Biol. 2020, 28, 101306. [Google Scholar] [CrossRef]
- Kong, C.; Wang, C.; Shi, Y.; Yan, L.; Xu, J.; Qi, W. Active vitamin D activates chondrocyte autophagy to reduce osteoarthritis via mediating the AMPK-mTOR signaling pathway. Biochem. Cell Biol. 2020, 98, 434–442. [Google Scholar] [CrossRef]
- Szychlinska, M.A.; Imbesi, R.; Castrogiovanni, P.; Guglielmino, C.; Ravalli, S.; Di Rosa, M.; Musumeci, G. Assessment of Vitamin D Supplementation on Articular Cartilage Morphology in a Young Healthy Sedentary Rat Model. Nutrients 2019, 11, 1260. [Google Scholar] [CrossRef]
- Zhang, S.; Xing, J.; Gong, Y.; Li, P.; Wang, B.; Xu, L. Downregulation of VDR in benign paroxysmal positional vertigo patients inhibits otolith associated protein expression levels. Mol. Med. Rep. 2021, 24, 591. [Google Scholar] [CrossRef] [PubMed]
- Peng, Z.; Sun, H.; Bunpetch, V.; Koh, Y.; Wen, Y.; Wu, D.; Ouyang, H. The regulation of cartilage extracellular matrix homeostasis in joint cartilage degeneration and regeneration. Biomaterials 2021, 268, 120555. [Google Scholar] [CrossRef]
- Verma, P.; Dalal, K.; Chopra, M. Pharmacophore development and screening for discovery of potential inhibitors of ADAMTS-4 for osteoarthritis therapy. J. Mol. Model. 2016, 22, 178. [Google Scholar] [CrossRef]
- Meng, P.; Zhang, F.; Zhang, Y.; Wei, H.; Tan, S.; Guo, X.; Wang, S.; Yu, Y. ADAMTS4 and ADAMTS5 may be considered as new molecular therapeutic targets for cartilage damages with Kashin-Beck Disease. Med. Hypotheses 2020, 135, 109440. [Google Scholar] [CrossRef] [PubMed]
- Cuffaro, D.; Ciccone, L.; Rossello, A.; Nuti, E.; Santamaria, S. Targeting Aggrecanases for Osteoarthritis Therapy: From Zinc Chelation to Exosite Inhibition. J. Med. Chem. 2022, 65, 13505–13532. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Du, C.; Wang, H.; Zhang, C. Increased serum ADAMTS-4 in knee osteoarthritis: A potential indicator for the diagnosis of osteoarthritis in early stages. Genet. Mol. Res. 2014, 13, 9642–9649. [Google Scholar] [CrossRef]
- van den Bosch, M.H.J.; Blom, A.B.; van der Kraan, P.M. Inflammation in osteoarthritis: Our view on its presence and involvement in disease development over the years. Osteoarthr. Cartil. 2024, 32, 355–364. [Google Scholar] [CrossRef]
- Xu, X.; Zheng, L.; Yuan, Q.; Zhen, G.; Crane, J.L.; Zhou, X.; Cao, X. Transforming growth factor-β in stem cells and tissue homeostasis. Bone Res. 2018, 6, 2. [Google Scholar] [CrossRef]
- Wang, T.Y.; Chen, D. Differential roles of TGF-beta signalling in joint tissues during osteoarthritis development. Ann. Rheum. Dis. 2016, 75, e72. [Google Scholar] [CrossRef]
- Xu, X.; Yang, C.; Yu, X.; Wang, J. Fibulin-3 regulates the inhibitory effect of TNF-α on chondrocyte differentiation partially via the TGF-β/Smad3 signaling pathway. Biochimica et biophysica acta. Mol. Cell Res. 2022, 1869, 119285. [Google Scholar]
- Lin, S.; Li, H.; Wu, B.; Shang, J.; Jiang, N.; Peng, R.; Xing, B.; Xu, X.; Lu, H. TGF-β1 regulates chondrocyte proliferation and extracellular matrix synthesis via circPhf21a-Vegfa axis in osteoarthritis. Cell Commun. Signal. 2022, 20, 75. [Google Scholar] [CrossRef]


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Guan, J.; Li, Z.; Niu, G.; Li, S.; Li, W.; Song, C.; Leng, H. Protective Effects of Vitamin D on Proteoglycans of Human Articular Chondrocytes through TGF-β1 Signaling. Nutrients 2024, 16, 2991. https://doi.org/10.3390/nu16172991
Guan J, Li Z, Niu G, Li S, Li W, Song C, Leng H. Protective Effects of Vitamin D on Proteoglycans of Human Articular Chondrocytes through TGF-β1 Signaling. Nutrients. 2024; 16(17):2991. https://doi.org/10.3390/nu16172991
Chicago/Turabian StyleGuan, Jian, Zhuoxin Li, Guodong Niu, Siwei Li, Weishi Li, Chunli Song, and Huijie Leng. 2024. "Protective Effects of Vitamin D on Proteoglycans of Human Articular Chondrocytes through TGF-β1 Signaling" Nutrients 16, no. 17: 2991. https://doi.org/10.3390/nu16172991
APA StyleGuan, J., Li, Z., Niu, G., Li, S., Li, W., Song, C., & Leng, H. (2024). Protective Effects of Vitamin D on Proteoglycans of Human Articular Chondrocytes through TGF-β1 Signaling. Nutrients, 16(17), 2991. https://doi.org/10.3390/nu16172991







