A Review on Berry Seeds—A Special Emphasis on Their Chemical Content and Health-Promoting Properties
Abstract
1. Introduction
2. Characterization of Berries
3. Chemical Content of Berries and Berry Seeds
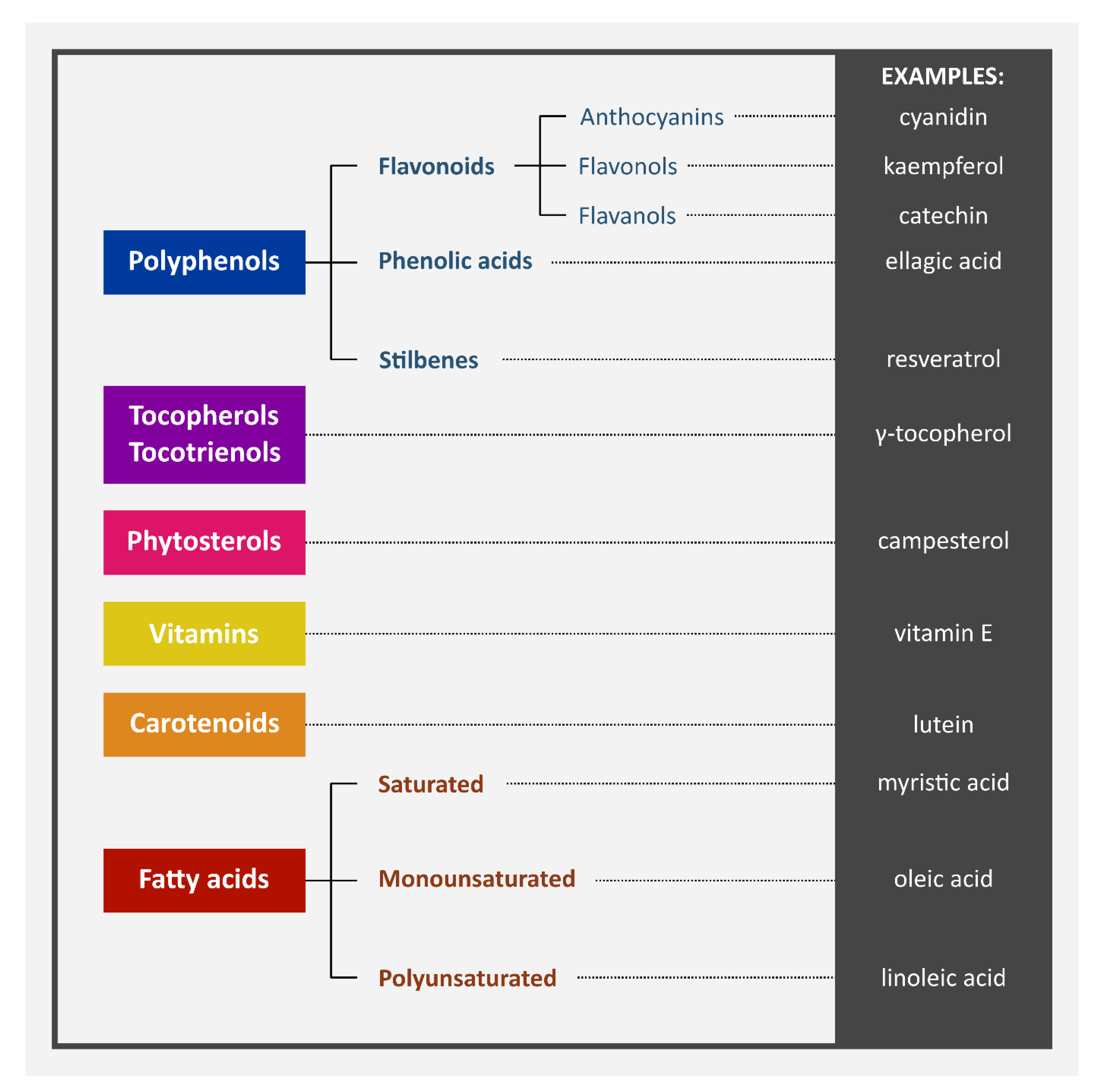
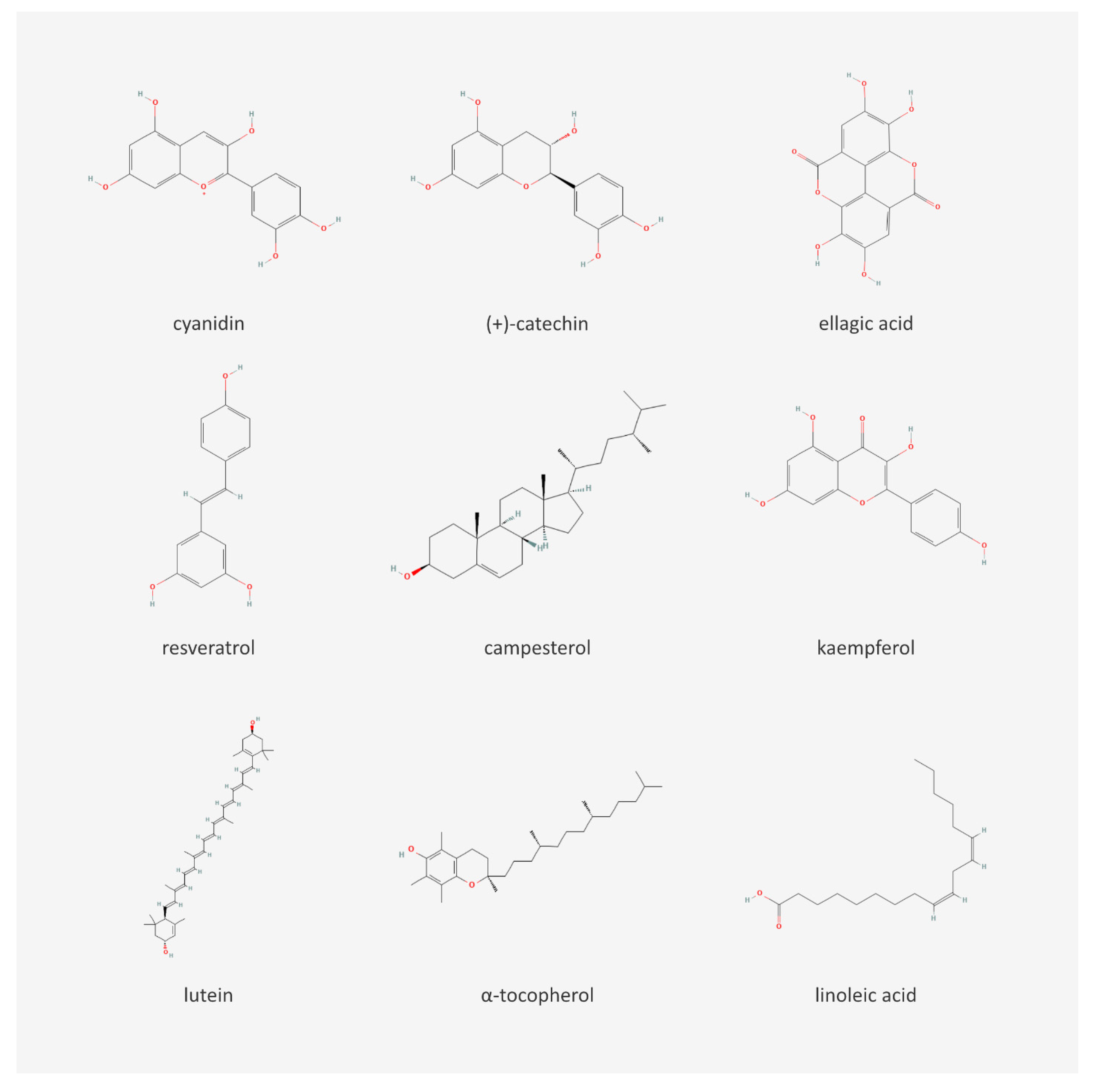
| Chemical Content of Berry Seeds | |||
|---|---|---|---|
| Type of Compound | Compound | Berries | Citation |
| Phytosterols | campesterol | raspberry | [7,23] |
| strawberry | |||
| sea buckthorn | |||
| cranberry | |||
| stigmasterol | raspberry | [7] | |
| strawberry | |||
| cranberry | |||
| β-sitosterol | raspberry | [7,14] | |
| strawberry | |||
| sea buckthorn | |||
| cranberry | |||
| Δ5-avenasterol | raspberry | [7,23] | |
| strawberry | |||
| sea buckthorn | |||
| Δ7-avenasterol | raspberry | [7] | |
| strawberry | |||
| cranberry | |||
| Tocopherols and tocotrienols | α-tocopherol | raspberry | [7,14,24] |
| strawberry | |||
| cranberry | |||
| sea buckthorn | |||
| α-tocotrienol | cranberry | [24] | |
| β-tocopherol | raspberry | [24,25] | |
| cranberry | |||
| γ-tocopherol | raspberry | [7,24,25] | |
| strawberry | |||
| cranberry | |||
| γ-tocotrienol | raspberry | [7,24] | |
| cranberry | |||
| δ-tocopherol | raspberry | [7,24] | |
| strawberry | |||
| β-carotene | raspberry | [23,26] | |
| sea buckthorn | |||
| zeaxanthin | sea buckthorn | [27] | |
| Anthocyanins | cyanidin | raspberry | [14] |
| grape | |||
| peonidin | raspberry | [14] | |
| Flavanols | epicatechin | raspberry | [28,29] |
| grape | |||
| catechin | raspberry | [14] | |
| grape | |||
| Flavonols | kaempferol | grape | [14] |
| sea buckthorn | |||
| Vitamins | vitamin C | sea buckthorn | [23,24] |
| vitamin A | sea buckthorn | [23] | |
| Phenolic acids | chlorogenic acid | grape | [14] |
| ellagic acid | raspberry | [30] | |
| strawberry | |||
| gallic acid | raspberry | [14,31] | |
| sea buckthorn | |||
| Stilbenes | resveratrol | raspberry | [14] |
| grape | |||
| Chemical Content | Oil from Berry Seeds | ||||
|---|---|---|---|---|---|
| Cranberry | Raspberry | Strawberry | Sea Buckthorn | Grape | |
| Fatty acids (g/100 g) | |||||
| Lauric acid | 0.08 | - | - | - | 0.01 |
| Myristic acid | 0.08 | 0.07 | 0.05 | 0.14 | 0.05 |
| Palmitic acid | 5.38 | 2.43 | 4.32 | 9.27 | 6.6 |
| Stearic acid | 1.25 | 0.90 | 1.68 | 2.47 | 3.5 |
| Oleic acid | 25.30 | 10.87 | 14.55 | 25.02 | 14.3 |
| Linoleic acid | 37.68 | 53.67 | 42.22 | 37.33 | 74.7 |
| Linolenic acid | 30.09 | 31.68 | 36.48 | 23.13 | 0.15 |
| Arachidonic acid | 0.07 | 0.37 | 0.71 | 0.44 | 0.40 |
| Saturated fatty acids | 6.88 | 3.82 | 6.85 | 13.39 | 10.4 |
| Monounsaturated fatty acids | 25.14 | 11.02 | 14.71 | 26.15 | 14.8 |
| Polyunsaturated fatty acids | 67.98 | 85.16 | 78.44 | 60.46 | 74.9 |
| Sterols (%) | |||||
| Campesterol | 4.2 | 4.5 | 5.4 | 4.0 | 9.3 |
| Stigmasterol | 1.3 | 1.2 | 2.3 | 17.0 | 10.8 |
| β-sitosterol | 60.4 | 79.6 | 71.1 | 66.5 | 67.4 |
4. Health-Promoting Properties of Berry Seeds—In Vitro and In Vivo Studies
4.1. Raspberry (R. idaeus L. and R. coreanus Miq.) Seeds
4.1.1. In Vitro Studies
4.1.2. In Vivo Studies
4.2. Strawberry (F. x ananassa) Seeds
4.2.1. In Vitro Studies
4.2.2. In Vivo Studies
4.3. Grape (V. vinifera L.) Seeds
4.3.1. In Vitro Studies
4.3.2. In Vivo Studies
4.4. Sea Buckthorn (H. rhamnoides L.) Seeds
4.4.1. In Vitro Studies
4.4.2. In Vivo Studies
4.5. Cranberry (V. macrocarpon Ait.) Seeds
4.5.1. In Vitro Studies
4.5.2. In Vivo Studies
| Berry | Preparation | Dose | Length of Study/ Incubation Time | Experimental Model | Biological Properties | References |
|---|---|---|---|---|---|---|
| In vitro studies | ||||||
| Strawberry | Extract | 0.1 and 0.3 µg/mL | 4. 5. and 7 days | Human stratum corneum cells | Increased expression of PPARα | [52] |
| Cranberry | Flour | 110.5 ± 22.0 μmol TE)/g flour | - | ORAC assay | Antioxidant | [84] |
| 3 and 6 mg/mL | 4 days | HT-29 (colon cancer) cells | Antitumor | |||
| Oil | 22.5 ± 1.22 μmol TE)/g | - | ABTS assay | Antioxidant | [83] | |
| 11.3 mg oil equivalent/mL | - | DPPH assay | ||||
| TBARS reduced by 2.8 mg/g oil | 1 h | LDL oxidation assay | ||||
| Raspberry | Oil | 30 µg/mL | 48 h | A-549 (human lung cancer) cells | Inhibition of cell growth | [39] |
| Extracts | IC50 = 27.1–49% of the extract (100 mg/mL) | 72 h | LS174 (human colon carcinoma) | Antiproliferative | [36] | |
| Ellagitannins | 5–30 μg/mL | 24 and 48 h | HT-29 cells | Antitumor | [38] | |
| Fresh seed extract and wine seed extract | 50–1000 μg/mL | 1 h | HepG2 cell line | Antioxidant | [42] | |
| 2.5–50 μg/mL | - | ORAC assay | ||||
| Extract | 10–1000 μg/mL | 1–24 h | RAW 264.7 (mouse leukemic monocyte macrophage) and CRFK (Crandell Reese feline kidney) cells | Antiviral activity against FCV-F9 and MNV-1 | [44] | |
| Extract | 0.5–50 μg/mL | 1 h | MDCK (Madin-Darby canine kidney) cells | Antiviral activity against influenza A and B | [43] | |
| Oil | 0.5–2 mg/mL | 1 h | HepG2 cells | Antioxidant | [40] | |
| Oil | 0.5–10% | 24–48 h | NHDF, LoVo, LoVo/DX, MCF7, MCF7/DX, A549 cells | Antioxidant, prooxidant, antitumor | [41] | |
| Grape | Extract (GSE) | 50 and 100 µg/mL | 48 h | LoVo and HT29 (human colorectal cancer) cells | Inihibition of cell growth | [56] |
| Extract (GSE) | 2000 µg/mL | 64 h | P. gingivalis ATCC 33277 and F. nucleatum ATCC 10953 | Antibacterial | [9] | |
| Proanthocyanidins | 10–80 μg/mL | 1 h pre-incubation | TM3 (Mouse testicular stromal) and HIEC (Human small intestinal crypt epithelial) cells irradiated with 5 or 8 Gy | Antioxidant, radioprotective | [86] | |
| Proanthocyanidins | 25–200 μg/mL | 24 and 48 h | HeLa cells | Antitumor | [62] | |
| Proanthocyanidins | 0.05–0.2 μM | 12–48 h | MCF-7 cells | Antitumor (EGFR inhibition, antiproliferative) | [58] | |
| Proanthocyanidins | 10–40 μg/mL | 12–48 h | SCC13 cells | Antitumor (EGFR inhibition, invasion inhibition) | [59] | |
| In vivo studies | ||||||
| Sea buckthorn | Proanthocyanidins | 50 and 100 mg/kg/day | 14 days | Rabbits | Retinoprotective activity | [81] |
| Oil | 1 mL/day | 18 days or 30 days | Healthy rabbits and rabbits with hypercholesterolemia | Anti-atherogenic activity | [78] | |
| Oil | 50 and 100 g/day | 42 days | Hamsters with hypercholesterolemia | Deceased total plasma cholesterol | [79] | |
| Oil | 0.75 mL/day | 30 days | Healthy and hypertensive/hypercholesterolemic humans | Decreased blood pressure, serum triglycerides, and cholesterol | [80] | |
| Cranberry | Extract | 15 mL/day | 28 days | Humans with cholesterol level over 200 mg/dL | Decreased blood cholesterol | [85] |
| Strawberry | Oil | 7% of diet | 8 weeks | Rats fed with obesogenic diet | Anti-hyperlipidemic, negative changes to microbial metabolism in the distal intestine | [54] |
| Grape | Extract | 200 mg/kg/day | 56 days | Athymic mice with human colon carcinoma tumor xenograft | Anticancer activity | [56] |
| Procyanidin extract (GSPE) | 50 mg/kg/day | 14 days | Mice with ischemia-reperfusion brain injury | Neuroprotective activity | [60] | |
| Procyanidin | 1–40 mg/kg, administered 1 h before cisplatin treatment | 30 days | Mice treated with cisplatin | Attenuation of chemotherapy-induced cognitive impairment | [63] | |
| Seeds | 50 g/kg diet | 98 days | Rabbits treated with lindane | Antioxidant | [64] | |
| Proanthocyanidins | 400 mg/kg 1 h before radiation exposure, then fed continuously by drinking for 4 weeks (2 mg/mL) | 56 days | Mice exposed to radiation (5, 8, or 27 Gy) | Radioprotective, antioxidant | [86] | |
| Raspberry | Oil | 7% of diet | 56 days | Rats fed with HF/LF diet | Improved lipid metabolism | [49] |
| Oil | 0.4 and 0.8 mL/3 times a week | 56 days | Rats fed with HFD | Potential anti-NAFLD effect | [47] | |
| Defatted seeds | 6% of diet | 4 weeks | Rats | Hypoglycemic, hypolipidemic, lower atherogenic index | [30] | |
| Flour | Equivalent of 0.03% ellagic acid | 12 weeks | Mice fed with HFD or HFD with sucrose | Anti-hyperlipidemic, hypoglycemic, antioxidant, anti-inflammatory, normalization of adipocyte size | [45] | |
| Ground seeds | 7 g/100 g of diet | 6 weeks | Hypertensive and normotensive rats | Improved vascular function | [46] | |
5. Herb-Drug Interaction (HDI)
6. Medicines and Commercial Products from Berry Seeds
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
References
- Lavefve, L.; Howard, L.R.; Carbonero, F. Berry Polyphenols Metabolism and Impact on Human Gut Microbiota and Health. Food Funct. 2020, 11, 45–65. [Google Scholar] [CrossRef]
- Hawkins, J.; Hires, C.; Baker, C.; Keenan, L.; Bush, M. Daily Supplementation with Aronia Melanocarpa (Chokeberry) Reduces Blood Pressure and Cholesterol: A Meta Analysis of Controlled Clinical Trials. J. Diet. Suppl. 2021, 18, 517–530. [Google Scholar] [CrossRef] [PubMed]
- Yang, B.; Kortesniemi, M. Clinical Evidence on Potential Health Benefits of Berries. Curr. Opin. Food Sci. 2015, 2, 36–42. [Google Scholar] [CrossRef]
- Luís, Â.; Domingues, F.; Pereira, L. Association between Berries Intake and Cardiovascular Diseases Risk Factors: A Systematic Review with Meta-Analysis and Trial Sequential Analysis of Randomized Controlled Trials. Food Funct. 2018, 9, 740–757. [Google Scholar] [CrossRef] [PubMed]
- Manganaris, G.A.; Goulas, V.; Vicente, A.R.; Terry, L.A. Berry Antioxidants: Small Fruits Providing Large Benefits. J. Sci. Food Agric. 2014, 94, 825–833. [Google Scholar] [CrossRef]
- Micić, D.M.; Ostojić, S.B.; Simonović, M.B.; Pezo, L.L.; Simonović, B.R. Thermal Behavior of Raspberry and Blackberry Seed Flours and Oils. Thermochim. Acta 2015, 617, 21–27. [Google Scholar] [CrossRef]
- Van Hoed, V.; De Clercq, N.; Echim, C.; Andjelkovic, M.; Leber, E.; Dewettinck, K.; Verhé, R. Berry seeds: A source of specialty oils with high content of bioactives and nutritional value. J. Food Lipids 2009, 16, 33–49. [Google Scholar] [CrossRef]
- Gustinelli, G.; Eliasson, L.; Svelander, C.; Andlid, T.; Lundin, L.; Ahrné, L.; Alminger, M. Supercritical Fluid Extraction of Berry Seeds: Chemical Composition and Antioxidant Activity. J. Food Qual. 2018, 2018, 6046074. [Google Scholar] [CrossRef]
- Furiga, A.; Lonvaud-Funel, A.; Badet, C. In Vitro Study of Antioxidant Capacity and Antibacterial Activity on Oral Anaerobes of a Grape Seed Extract. Food Chem. 2009, 113, 1037–1040. [Google Scholar] [CrossRef]
- Raczyk, M.; Bryś, J.; Brzezińska, R.; Ostrowska-Ligęza, E.; Wirkowska-Wojdyła, M.; Górska, A. Quality Assessment of Cold-Pressed Strawberry, Raspberry and Blackberry Seed Oils Intended for Cosmetic Purposes. Acta Sci. Pol. Technol. Aliment. 2021, 20, 127–133. [Google Scholar] [CrossRef]
- Sorrentino, C.; Di Gisi, M.; Gentile, G.; Licitra, F.; D’Angiolo, R.; Giovannelli, P.; Migliaccio, A.; Castoria, G.; Di Donato, M. Agri-Food By-Products in Cancer: New Targets and Strategies. Cancers 2022, 14, 5517. [Google Scholar] [CrossRef] [PubMed]
- León-González, A.J.; Auger, C.; Schini-Kerth, V.B. Pro-Oxidant Activity of Polyphenols and Its Implication on Cancer Chemoprevention and Chemotherapy. Biochem. Pharmacol. 2015, 98, 371–380. [Google Scholar] [CrossRef] [PubMed]
- Brewer, M.S. Natural Antioxidants: Sources, Compounds, Mechanisms of Action, and Potential Applications. Compr. Rev. Food Sci. Food Saf. 2011, 10, 221–247. [Google Scholar] [CrossRef]
- Sławińska, N.; Olas, B. Selected Seeds as Sources of Bioactive Compounds with Diverse Biological Activities. Nutrients 2022, 15, 187. [Google Scholar] [CrossRef]
- Salo, H.M.; Nguyen, N.; Alakärppä, E.; Klavins, L.; Hykkerud, A.L.; Karppinen, K.; Jaakola, L.; Klavins, M.; Häggman, H. Authentication of Berries and Berry-Based Food Products. Compr. Rev. Food Sci. Food Saf. 2021, 20, 5197–5225. [Google Scholar] [CrossRef] [PubMed]
- Ferlemi, A.-V.; Lamari, F. Berry Leaves: An Alternative Source of Bioactive Natural Products of Nutritional and Medicinal Value. Antioxidants 2016, 5, 17. [Google Scholar] [CrossRef]
- Skrovankova, S.; Sumczynski, D.; Mlcek, J.; Jurikova, T.; Sochor, J. Bioactive Compounds and Antioxidant Activity in Different Types of Berries. Int. J. Mol. Sci. 2015, 16, 24673–24706. [Google Scholar] [CrossRef]
- Olas, B. Berry Phenolic Antioxidants—Implications for Human Health? Front. Pharmacol. 2018, 9, 78. [Google Scholar] [CrossRef]
- Miller, K.; Feucht, W.; Schmid, M. Bioactive Compounds of Strawberry and Blueberry and Their Potential Health Effects Based on Human Intervention Studies: A Brief Overview. Nutrients 2019, 11, 1510. [Google Scholar] [CrossRef]
- Duba, K.S.; Fiori, L. Supercritical CO2 Extraction of Grape Seed Oil: Effect of Process Parameters on the Extraction Kinetics. J. Supercrit. Fluids 2015, 98, 33–43. [Google Scholar] [CrossRef]
- Garavaglia, J.; Markoski, M.M.; Oliveira, A.; Marcadenti, A. Grape Seed Oil Compounds: Biological and Chemical Actions for Health. Nutr. Metab. Insights 2016, 9, 59. [Google Scholar] [CrossRef]
- Wu, X.; Wang, T.T.Y.; Prior, R.L.; Pehrsson, P.R. Prevention of Atherosclerosis by Berries: The Case of Blueberries. J. Agric. Food Chem. 2018, 66, 9172–9188. [Google Scholar] [CrossRef] [PubMed]
- Olas, B. The Beneficial Health Aspects of Sea Buckthorn (Elaeagnus rhamnoides (L.) A.Nelson) Oil. J. Ethnopharmacol. 2018, 213, 183–190. [Google Scholar] [CrossRef] [PubMed]
- Helbig, D.; Böhm, V.; Wagner, A.; Schubert, R.; Jahreis, G. Berry Seed Press Residues and Their Valuable Ingredients with Special Regard to Black Currant Seed Press Residues. Food Chem. 2008, 111, 1043–1049. [Google Scholar] [CrossRef]
- Radočaj, O.; Vujasinović, V.; Dimić, E.; Basić, Z. Blackberry (Rubus fruticosus L.) and Raspberry (Rubus idaeus L.) Seed Oils Extracted from Dried Press Pomace after Longterm Frozen Storage of Berries Can Be Used as Functional Food Ingredients. Eur. J. Lipid Sci. Technol. 2014, 116, 1015–1024. [Google Scholar] [CrossRef]
- Parry, J.; Su, L.; Luther, M.; Zhou, K.; Peter Yurawecz, M.; Whittaker, P.; Yu, L. Fatty Acid Composition and Antioxidant Properties of Cold-Pressed Marionberry, Boysenberry, Red Raspberry, and Blueberry Seed Oils. J. Agric. Food Chem. 2005, 53, 566–573. [Google Scholar] [CrossRef]
- Zielińska, A.; Nowak, I. Abundance of Active Ingredients in Sea-Buckthorn Oil. Lipids Health Dis. 2017, 16, 95. [Google Scholar] [CrossRef]
- Yao, J.; Chen, J.; Yang, J.; Hao, Y.; Fan, Y.; Wang, C.; Li, N. Free, Soluble-Bound and Insoluble-Bound Phenolics and Their Bioactivity in Raspberry Pomace. LWT 2021, 135, 109995. [Google Scholar] [CrossRef]
- Valls, J.; Agnolet, S.; Haas, F.; Struffi, I.; Ciesa, F.; Robatscher, P.; Oberhuber, M. Valorization of Lagrein Grape Pomace as a Source of Phenolic Compounds: Analysis of the Contents of Anthocyanins, Flavanols and Antioxidant Activity. Eur. Food Res. Technol. 2017, 243, 2211–2224. [Google Scholar] [CrossRef]
- Kosmala, M.; Zduńczyk, Z.; Juśkiewicz, J.; Jurgoński, A.; Karlińska, E.; Macierzyński, J.; Jańczak, R.; Rój, E. Chemical Composition of Defatted Strawberry and Raspberry Seeds and the Effect of These Dietary Ingredients on Polyphenol Metabolites, Intestinal Function, and Selected Serum Parameters in Rats. J. Agric. Food Chem. 2015, 63, 2989–2996. [Google Scholar] [CrossRef]
- Arimboor, R.; Arumughan, C. HPLC-DAD-MS/MS Profiling of Antioxidant Flavonoid Glycosides in Sea Buckthorn (Hippophae rhamnoides L.) Seeds. Int. J. Food Sci. Nutr. 2012, 63, 730–738. [Google Scholar] [CrossRef]
- Zadernowski, R.; Nowak-Polakowska, H.; Lossow, B.; Nesterowicz, J. Sea-Buckthorn Lipids. J. Food Lipids 1997, 4, 165–172. [Google Scholar] [CrossRef]
- Yang, B.; Karlsson, R.M.; Oksman, P.H.; Kallio, H.P. Phytosterols in Sea Buckthorn (Hippophaë rhamnoides L.) Berries: Identification and Effects of Different Origins and Harvesting Times. J. Agric. Food Chem. 2001, 49, 5620–5629. [Google Scholar] [CrossRef] [PubMed]
- Kitrytė, V.; Povilaitis, D.; Kraujalienė, V.; Šulniūtė, V.; Pukalskas, A.; Venskutonis, P.R. Fractionation of Sea Buckthorn Pomace and Seeds into Valuable Components by Using High Pressure and Enzyme-Assisted Extraction Methods. LWT-Food Sci. Technol. 2017, 85, 534–538. [Google Scholar] [CrossRef]
- Available online: https://pubchem.ncbi.nlm.nih.gov/compound/128861 (accessed on 8 March 2023).
- Juranic, Z.; Zizak, Z.; Tasic, S.; Petrovic, S.; Nidzovic, S.; Leposavic, A.; Stanojkovic, T. Antiproliferative Action of Water Extracts of Seeds or Pulp of Five Different Raspberry Cultivars. Food Chem. 2005, 93, 39–45. [Google Scholar] [CrossRef]
- Williner, M.R.; Pirovani, M.E.; Güemes, D.R. Ellagic Acid Content in Strawberries of Different Cultivars and Ripening Stages. J. Sci. Food Agric. 2003, 83, 842–845. [Google Scholar] [CrossRef]
- Cho, H.; Jung, H.; Lee, H.; Yi, H.C.; Kwak, H.K.; Hwang, K.T. Chemopreventive Activity of Ellagitannins and Their Derivatives from Black Raspberry Seeds on HT-29 Colon Cancer Cells. Food Funct. 2015, 6, 1675–1683. [Google Scholar] [CrossRef]
- Simonovic, M.; Kojic, V.; Jakimov, D.; Glumac, M.; Pejin, B. Raspberry Seeds Extract Selectively Inhibits the Growth of Human Lung Cancer Cells in Vitro. Nat. Prod. Res. 2021, 35, 2253–2256. [Google Scholar] [CrossRef]
- Teng, H.; Lin, Q.; Li, K.; Yuan, B.; Song, H.; Peng, H.; Yi, L.; Wei, M.-C.; Yang, Y.-C.; Battino, M.; et al. Hepatoprotective Effects of Raspberry (Rubus coreanus Miq.) Seed Oil and Its Major Constituents. Food Chem. Toxicol. 2017, 110, 418–424. [Google Scholar] [CrossRef]
- Grajzer, M.; Wiatrak, B.; Gębarowski, T.; Boba, A.; Rój, E.; Gorczyca, D.; Prescha, A. Bioactive Compounds of Raspberry Oil Emulsions Induced Oxidative Stress via Stimulating the Accumulation of Reactive Oxygen Species and NO in Cancer Cells. Oxid. Med. Cell. Longev. 2021, 2021, 5561672. [Google Scholar] [CrossRef]
- Choi, M.H.; Shim, S.M.; Kim, G.H. Protective Effect of Black Raspberry Seed Containing Anthocyanins against Oxidative Damage to DNA, Protein, and Lipid. J. Food Sci. Technol. 2016, 53, 1214–1221. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.-H.; Oh, M.; Seok, J.; Kim, S.; Lee, D.; Bae, G.; Bae, H.-I.; Bae, S.; Hong, Y.-M.; Kwon, S.-O.; et al. Antiviral Effects of Black Raspberry (Rubus coreanus) Seed and Its Gallic Acid against Influenza Virus Infection. Viruses 2016, 8, 157. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.H.; Bae, S.Y.; Oh, M.; Seok, J.H.; Kim, S.; bin Chung, Y.; Gowda, K.G.; Mun, J.Y.; Chung, M.S.; Kim, K.H. Antiviral Effects of Black Raspberry (Rubus coreanus) Seed Extract and Its Polyphenolic Compounds on Norovirus Surrogates. Biosci. Biotechnol. Biochem. 2016, 80, 1196–1204. [Google Scholar] [CrossRef] [PubMed]
- Kang, I.; Espín, J.C.; Carr, T.P.; Tomás-Barberán, F.A.; Chung, S. Raspberry Seed Flour Attenuates High-Sucrose Diet-Mediated Hepatic Stress and Adipose Tissue Inflammation. J. Nutr. Biochem. 2016, 32, 64–72. [Google Scholar] [CrossRef]
- Majewski, M.; Kucharczyk, E.; Kaliszan, R.; Markuszewski, M.; Fotschki, B.; Juśkiewicz, J.; Borkowska-Sztachańska, M.; Ognik, K. The Characterization of Ground Raspberry Seeds and the Physiological Response to Supplementation in Hypertensive and Normotensive Rats. Nutrients 2020, 12, 1630. [Google Scholar] [CrossRef]
- Hendawy, O.; Gomaa, H.A.M.; Hussein, S.; Alzarea, S.I.; Qasim, S.; Abdel Rahman, F.E.Z.S.; Ali, A.T.; Ahmed, S.R. Cold-Pressed Raspberry Seeds Oil Ameliorates High-Fat Diet Triggered Non-Alcoholic Fatty Liver Disease. Saudi Pharm. J. 2021, 29, 1303–1313. [Google Scholar] [CrossRef]
- Mapfumo, M.; Lembede, B.W.; Nkomozepi, P.; Ndhlala, A.R.; Chivandi, E. Crude Moringa Oleifera Lam. Seed Extract Attenuates Non-Alcoholic Fatty Liver Disease in Growing Sprague–Dawley Rats. S. Afr. J. Bot. 2020, 129, 191–197. [Google Scholar] [CrossRef]
- Fotschki, B.; Jurgó, A.; Ju’skiewicz, J.J.; Zdú, Z. Dietary Supplementation with Raspberry Seed Oil Modulates Liver Functions, Inflammatory State, and Lipid Metabolism in Rats. J. Nutr. 2015, 145, 1793–1799. [Google Scholar] [CrossRef]
- Lee, H.J.; Jung, H.; Cho, H.; Lee, K.; Hwang, K.T. Black Raspberry Seed Oil Improves Lipid Metabolism by Inhibiting Lipogenesis and Promoting Fatty-Acid Oxidation in High-Fat Diet-Induced Obese Mice and Db/Db Mice. Lipids 2018, 53, 491–504. [Google Scholar] [CrossRef]
- Grzelak-Błaszczyk, K.; Karlińska, E.; Grzęda, K.; Rój, E.; Kołodziejczyk, K. Defatted Strawberry Seeds as a Source of Phenolics, Dietary Fiber and Minerals. LWT 2017, 84, 18–22. [Google Scholar] [CrossRef]
- Takeda, S.; Shimoda, H.; Takarada, T.; Imokawa, G. Strawberry Seed Extract and Its Major Component, Tiliroside, Promote Ceramide Synthesis in the Stratum Corneum of Human Epidermal Equivalents. PLoS ONE 2018, 13, e0205061. [Google Scholar] [CrossRef] [PubMed]
- Pieszka, M.; Tombarkiewicz, B.; Roman, A.; Migdał, W.; Niedziółka, J. Effect of Bioactive Substances Found in Rapeseed, Raspberry and Strawberry Seed Oils on Blood Lipid Profile and Selected Parameters of Oxidative Status in Rats. Environ. Toxicol. Pharmacol. 2013, 36, 1055–1062. [Google Scholar] [CrossRef] [PubMed]
- Jurgoński, A.; Fotschki, B.; Juśkiewicz, J. Dietary Strawberry Seed Oil Affects Metabolite Formation in the Distal Intestine and Ameliorates Lipid Metabolism in Rats Fed an Obesogenic Diet. Food Nutr. Res. 2015, 59, 26104. [Google Scholar] [CrossRef] [PubMed]
- Costa, G.N.S.; Tonon, R.V.; Mellinger-Silva, C.; Galdeano, M.C.; Iacomini, M.; Santiago, M.C.P.A.; Almeida, E.L.; Freitas, S.P. Grape Seed Pomace as a Valuable Source of Antioxidant Fibers. J. Sci. Food Agric. 2019, 99, 4593–4601. [Google Scholar] [CrossRef]
- Kaur, M.; Singh, R.P.; Gu, M.; Agarwal, R.; Agarwal, C. Grape Seed Extract Inhibits In Vitro and In Vivo Growth of Human Colorectal Carcinoma Cells. Clin. Cancer Res. 2006, 12, 6194–6202. [Google Scholar] [CrossRef]
- Ci, X.; Chen, L.; Ou, X. Grape Seed Proanthocyanidin Extracts Inhibit Lipopolysaccharide of Porphyromonas Gingivalis. Shanghai Kou Qiang Yi Xue 2015, 24, 433–436. [Google Scholar]
- Kong, F.T.; He, C.X.; Kong, F.L.; Han, S.F.; Kong, X.S.; Han, W.Q.; Yang, L.X. Grape Seed Procyanidins Inhibit the Growth of Breast Cancer MCF-7 Cells by Down-Regulating the EGFR/VEGF/MMP9 Pathway. Nat. Prod. Commun. 2021, 16, 1934578X21991691. [Google Scholar] [CrossRef]
- Sun, Q.; Prasad, R.; Rosenthal, E.; Katiyar, S.K. Grape Seed Proanthocyanidins Inhibit the Invasive Potential of Head and Neck Cutaneous Squamous Cell Carcinoma Cells by Targeting EGFR Expression and Epithelial-to-Mesenchymal Transition. BMC Complement. Altern. Med. 2011, 11, 134. [Google Scholar] [CrossRef]
- Kong, X.; Guan, J.; Gong, S.; Wang, R. Neuroprotective Effects of Grape Seed Procyanidin Extract on Ischemia-Reperfusion Brain Injury. Chin. Med. Sci. J. 2017, 32, 92–99. [Google Scholar] [CrossRef]
- Delimont, N.M.; Carlson, B.N. Prevention of Dental Caries by Grape Seed Extract Supplementation: A Systematic Review. Nutr. Health 2020, 26, 43–52. [Google Scholar] [CrossRef]
- Li, C.; Zhang, L.; Liu, C.; He, X.; Chen, M.; Chen, J. Lipophilic Grape Seed Proanthocyanidin Exerts Anti-Cervical Cancer Effects in HeLa Cells and a HeLa-Derived Xenograft Zebrafish Model. Antioxidants 2022, 11, 422. [Google Scholar] [CrossRef] [PubMed]
- Song, C.; Gao, C.; Wang, Z. Grape-Seed-Derived Procyanidin Attenuates Chemotherapy-Induced Cognitive Impairment by Suppressing MMP-9 Activity and Related Blood–Brain-Barrier Damage. Brain Sci. 2022, 12, 571. [Google Scholar] [CrossRef] [PubMed]
- Abu Hafsa, S.H.; Hassan, A.A. Grape Seed Alleviates Lindane-induced Oxidative Stress and Improves Growth Performance, Caecal Fermentation and Antioxidant Capacity in Growing Rabbits. J. Anim. Physiol. Anim. Nutr. 2022, 106, 899–909. [Google Scholar] [CrossRef]
- Anjom-Shoae, J.; Milajerdi, A.; Larijani, B.; Esmaillzadeh, A. Effects of Grape Seed Extract on Dyslipidaemia: A Systematic Review and Dose-Response Meta-Analysis of Randomised Controlled Trials. Br. J. Nutr. 2020, 124, 121–134. [Google Scholar] [CrossRef] [PubMed]
- Grohmann, T.; Litts, C.; Horgan, G.; Zhang, X.; Hoggard, N.; Russell, W.; de Roos, B. Efficacy of Bilberry and Grape Seed Extract Supplement Interventions to Improve Glucose and Cholesterol Metabolism and Blood Pressure in Different Populations—A Systematic Review of the Literature. Nutrients 2021, 13, 1692. [Google Scholar] [CrossRef]
- Gupta, M.; Dey, S.; Marbaniang, D.; Pal, P.; Ray, S.; Mazumder, B. Grape Seed Extract: Having a Potential Health Benefits. J. Food Sci. Technol. 2020, 57, 1205–1215. [Google Scholar] [CrossRef]
- Liu, M.; Yun, P.; Hu, Y.; Yang, J.; Khadka, R.B.; Peng, X. Effects of Grape Seed Proanthocyanidin Extract on Obesity. Obes. Facts 2020, 13, 279–291. [Google Scholar] [CrossRef]
- Foshati, S.; Rouhani, M.H.; Amani, R. The Effect of Grape Seed Extract Supplementation on Oxidative Stress and Inflammation: A Systematic Review and Meta-analysis of Controlled Trials. Int. J. Clin. Pract. 2021, 75, e14469. [Google Scholar] [CrossRef]
- Coelho, O.G.L.; Ribeiro, P.V.M.; de Alfenas, R.C.G. Can Grape Polyphenols Affect Glycation Markers? A Systematic Review. Crit. Rev. Food Sci. Nutr. 2021, 1–11. [Google Scholar] [CrossRef]
- Asbaghi, O.; Nazarian, B.; Reiner, Ž.; Amirani, E.; Kolahdooz, F.; Chamani, M.; Asemi, Z. The Effects of Grape Seed Extract on Glycemic Control, Serum Lipoproteins, Inflammation, and Body Weight: A Systematic Review and Meta-analysis of Randomized Controlled Trials. Phytother. Res. 2020, 34, 239–253. [Google Scholar] [CrossRef]
- Mahdipour, R.; Ebrahimzadeh-Bideskan, A.; Hosseini, M.; Shahba, S.; Lombardi, G.; Malvandi, A.M.; Mohammadipour, A. The Benefits of Grape Seed Extract in Neurological Disorders and Brain Aging. Nutr. Neurosci. 2022, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Bell, L.; Whyte, A.R.; Lamport, D.J.; Spencer, J.P.E.; Butler, L.T.; Williams, C.M. Grape Seed Polyphenol Extract and Cognitive Function in Healthy Young Adults: A Randomised, Placebo-Controlled, Parallel-Groups Acute-on-Chronic Trial. Nutr. Neurosci. 2022, 25, 54–63. [Google Scholar] [CrossRef] [PubMed]
- Sochorova, L.; Prusova, B.; Cebova, M.; Jurikova, T.; Mlcek, J.; Adamkova, A.; Nedomova, S.; Baron, M.; Sochor, J. Health Effects of Grape Seed and Skin Extracts and Their Influence on Biochemical Markers. Molecules 2020, 25, 5311. [Google Scholar] [CrossRef] [PubMed]
- Isopencu, G.; Stroescu, M.; Brosteanu, A.; Chira, N.; Pârvulescu, O.C.; Busuioc, C.; Stoica-Guzun, A. Optimization of Ultrasound and Microwave Assisted Oil Extraction from Sea Buckthorn Seeds by Response Surface Methodology. J. Food Process. Eng. 2019, 42, e12947. [Google Scholar] [CrossRef]
- Gęgotek, A.; Jastrząb, A.; Jarocka-Karpowicz, I.; Muszyńska, M.; Skrzydlewska, E. The Effect of Sea Buckthorn (Hippophae rhamnoides L.) Seed Oil on UV-Induced Changes in Lipid Metabolism of Human Skin Cells. Antioxidants 2018, 7, 110. [Google Scholar] [CrossRef]
- Dudau, M.; Vilceanu, A.C.; Codrici, E.; Mihai, S.; Popescu, I.D.; Albulescu, L.; Tarcomnicu, I.; Moise, G.; Ceafalan, L.C.; Hinescu, M.E.; et al. Sea-Buckthorn Seed Oil Induces Proliferation of Both Normal and Dysplastic Keratinocytes in Basal Conditions and under UVA Irradiation. J. Pers. Med. 2021, 11, 278. [Google Scholar] [CrossRef]
- Basu, M.; Prasad, R.; Jayamurthy, P.; Pal, K.; Arumughan, C.; Sawhney, R.C. Anti-Atherogenic Effects of Seabuckthorn (Hippophaea rhamnoides) Seed Oil. Phytomedicine 2007, 14, 770–777. [Google Scholar] [CrossRef] [PubMed]
- Hao, W.; He, Z.; Zhu, H.; Liu, J.; Kwek, E.; Zhao, Y.; Ma, K.Y.; He, W.S.; Chen, Z.Y. Sea Buckthorn Seed Oil Reduces Blood Cholesterol and Modulates Gut Microbiota. Food Funct. 2019, 10, 5669–5681. [Google Scholar] [CrossRef]
- Vashishtha, V.; Barhwal, K.; Kumar, A.; Hota, S.K.; Chaurasia, O.P.; Kumar, B. Effect of Seabuckthorn Seed Oil in Reducing Cardiovascular Risk Factors: A Longitudinal Controlled Trial on Hypertensive Subjects. Clin. Nutr. 2017, 36, 1231–1238. [Google Scholar] [CrossRef]
- Wang, Y.; Zhao, L.; Huo, Y.; Zhou, F.; Wu, W.; Lu, F.; Yang, X.; Guo, X.; Chen, P.; Deng, Q.; et al. Protective Effect of Proanthocyanidins from Sea Buckthorn (Hippophae rhamnoides L.) Seed against Visible Light-Induced Retinal Degeneration in Vivo. Nutrients 2016, 8, 245. [Google Scholar] [CrossRef]
- Ahmad, N.; Anwar, F.; Abbas, A. Cranberry Seed Oil. In Fruit Oils: Chemistry and Functionality; Springer International Publishing: Cham, Switzerland, 2019; pp. 663–674. [Google Scholar]
- Yu, L.L.; Zhou, K.K.; Parry, J. Antioxidant Properties of Cold-Pressed Black Caraway, Carrot, Cranberry, and Hemp Seed Oils. Food Chem. 2005, 91, 723–729. [Google Scholar] [CrossRef]
- Parry, J.; Su, L.; Moore, J.; Cheng, Z.; Luther, M.; Rao, J.N.; Wang, J.Y.; Yu, L.L. Chemical Compositions, Antioxidant Capacities, and Antiproliferative Activities of Selected Fruit Seed Flours. J. Agric. Food Chem. 2006, 54, 3773–3778. [Google Scholar] [CrossRef]
- Eno, M. The Effect on Lipid Profiles after Supplementation of Cranberry Seed Oil. 2008. Available online: http://digital.library.wisc.edu/1793/52973 (accessed on 13 December 2022).
- Shen, H.; Han, J.; Liu, C.; Cao, F.; Huang, Y. Grape Seed Proanthocyanidins Exert a Radioprotective Effect on the Testes and Intestines Through Antioxidant Effects and Inhibition of MAPK Signal Pathways. Front. Med. 2022, 8, 3102. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Thilakarathna, W.P.D.W.; Astatkie, T.; Rupasinghe, H.P.V. Optimization of Catechin and Proanthocyanidin Recovery from Grape Seeds Using Microwave-Assisted Extraction. Biomolecules 2020, 10, 243. [Google Scholar] [CrossRef]
- Chen, Y.; Wen, J.; Deng, Z.; Pan, X.; Xie, X.; Peng, C. Effective Utilization of Food Wastes: Bioactivity of Grape Seed Extraction and Its Application in Food Industry. J. Funct. Foods 2020, 73, 104113. [Google Scholar] [CrossRef]
- Elkatry, H.O.; Ahmed, A.R.; El-Beltagi, H.S.; Mohamed, H.I.; Eshak, N.S. Biological Activities of Grape Seed By-Products and Their Potential Use as Natural Sources of Food Additives in the Production of Balady Bread. Foods 2022, 11, 1948. [Google Scholar] [CrossRef]
- Seo, K.H.; Kim, D.H.; Jeong, D.; Yokoyama, W.; Kim, H. Chardonnay Grape Seed Flour Supplemented Diets Alter Intestinal Microbiota in Diet-Induced Obese Mice. J. Food Biochem. 2017, 41, e12396. [Google Scholar] [CrossRef]
- Choi, Y.H.; Chin, Y.-W.; Kim, Y.G. Herb-Drug Interactions: Focus on Metabolic Enzymes and Transporters. Arch. Pharm. Res. 2011, 34, 1843–1863. [Google Scholar] [CrossRef]
- Srinivas, N.R. Cranberry Juice Ingestion and Clinical Drug-Drug Interaction Potentials; Review of Case Studies and Perspectives. J. Pharm. Pharm. Sci. 2013, 16, 289. [Google Scholar] [CrossRef]
- Ortiz-Flores, M.; Portilla-Martínez, A.; Cabrera-Pérez, F.; Nájera, N.; Meaney, E.; Villarreal, F.; Pérez-Durán, J.; Ceballos, G. PXR Is a Target of (-)-Epicatechin in Skeletal Muscle. Heliyon 2020, 6, e05357. [Google Scholar] [CrossRef]
- Goey, A.K.L.; Meijerman, I.; Beijnen, J.H.; Schellens, J.H.M. The Effect of Grape Seed Extract on the Pharmacokinetics of Dextromethorphan in Healthy Volunteers. Eur. J. Clin. Pharmacol. 2013, 69, 1883–1890. [Google Scholar] [CrossRef] [PubMed]
- Ispiryan, A.; Viškelis, J.; Viškelis, P. Red Raspberry (Rubus idaeus L.) Seed Oil: A Review. Plants 2021, 10, 944. [Google Scholar] [CrossRef] [PubMed]
- Romanini, E.; McRae, J.M.; Bilogrevic, E.; Colangelo, D.; Gabrielli, M.; Lambri, M. Use of Grape Seeds to Reduce Haze Formation in White Wines. Food Chem. 2021, 341, 128250. [Google Scholar] [CrossRef]
- Olas, B.; Skalski, B. Preparations from Various Organs of Sea Buckthorn (Elaeagnus rhamnoides (L.) A. Nelson) as Important Regulators of Hemostasis and Their Role in the Treatment and Prevention of Cardiovascular Diseases. Nutrients 2022, 14, 991. [Google Scholar] [CrossRef]
- Zeb, A.; Ullah, S. Sea Buckthorn Seed Oil Protects against the Oxidative Stress Produced by Thermally Oxidized Lipids. Food Chem. 2015, 186, 6–12. [Google Scholar] [CrossRef] [PubMed]
- Koskovac, M.; Cupara, S.; Kipic, M.; Barjaktarevic, A.; Milovanovic, O.; Kojicic, K.; Markovic, M. Sea Buckthorn Oil—A Valuable Source for Cosmeceuticals. Cosmetics 2017, 4, 40. [Google Scholar] [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sławińska, N.; Prochoń, K.; Olas, B. A Review on Berry Seeds—A Special Emphasis on Their Chemical Content and Health-Promoting Properties. Nutrients 2023, 15, 1422. https://doi.org/10.3390/nu15061422
Sławińska N, Prochoń K, Olas B. A Review on Berry Seeds—A Special Emphasis on Their Chemical Content and Health-Promoting Properties. Nutrients. 2023; 15(6):1422. https://doi.org/10.3390/nu15061422
Chicago/Turabian StyleSławińska, Natalia, Katarzyna Prochoń, and Beata Olas. 2023. "A Review on Berry Seeds—A Special Emphasis on Their Chemical Content and Health-Promoting Properties" Nutrients 15, no. 6: 1422. https://doi.org/10.3390/nu15061422
APA StyleSławińska, N., Prochoń, K., & Olas, B. (2023). A Review on Berry Seeds—A Special Emphasis on Their Chemical Content and Health-Promoting Properties. Nutrients, 15(6), 1422. https://doi.org/10.3390/nu15061422






