Exercise and Nutrition Interventions for Prehabilitation in Hepato-Pancreato-Biliary Cancers: A Narrative Review
Abstract
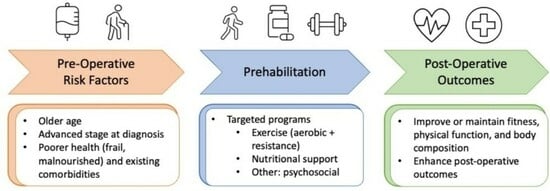
:1. Introduction
2. Methods
3. Design of Prehabilitation Programs in HPB Cancer Populations
4. Impact of Prehabilitation on Frailty and Malnutrition-Related Outcomes in HPB Cancer Populations
4.1. Fitness, Strength, and Physical Function
4.2. Biomarkers and Nutritional Status
4.3. Body Composition
4.4. Post-Operative Outcomes
5. Why Timing Matters
5.1. Alignment with Recent Guidelines
5.2. Impact of Prehabilitation on High-Risk Patients
5.3. Challenges in Assessing Prehabilitation Outcomes
6. Biologic Mechanisms of Prehabilitation Exercise and Nutrition Interventions
6.1. Exercise-Related Biological Mechanisms
6.2. Nutritional Status and Surgical Stress
7. Additional Considerations of Prehabilitation in HPB Cancer Populations
8. Ongoing Studies
9. Future Directions
10. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Arnold, M.; Abnet, C.C.; Neale, R.E.; Vignat, J.; Giovannucci, E.L.; McGlynn, K.A.; Bray, F. Global burden of 5 major types of gastrointestinal cancer. Gastroenterology 2020, 159, 335–349.e15. [Google Scholar] [CrossRef] [PubMed]
- Lu, L.; Mullins, C.S.; Schafmayer, C.; Zeißig, S.; Linnebacher, M. A global assessment of recent trends in gastrointestinal cancer and lifestyle-associated risk factors. Cancer Commun. 2021, 41, 1137–1151. [Google Scholar] [CrossRef] [PubMed]
- Ferlay, J.; Ervik, M.; Lam, F.; Colombet, M.; Mery, L.; Piñeros, M.; Znaor, A.; Soerjomataram, I.; Bray, F. Global cancer observatory: Cancer today. Lyon Fr. Int. Agency Res. Cancer 2018, 3, 2019. [Google Scholar]
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef]
- Siegel, R.L.; Miller, K.D.; Wagle, N.S.; Jemal, A. Cancer statistics, 2023. CA Cancer J. Clin. 2023, 73, 17–48. [Google Scholar] [CrossRef] [PubMed]
- Lafaro, K.; Buettner, S.; Maqsood, H.; Wagner, D.; Bagante, F.; Spolverato, G.; Xu, L.; Kamel, I.; Pawlik, T.M. Defining Post Hepatectomy Liver Insufficiency: Where do We stand? J. Gastrointest. Surg. 2015, 19, 2079–2092. [Google Scholar] [CrossRef] [PubMed]
- Vallance, A.E.; Young, A.L.; Macutkiewicz, C.; Roberts, K.J.; Smith, A.M. Calculating the risk of a pancreatic fistula after a pancreaticoduodenectomy: A systematic review. HPB 2015, 17, 1040–1048. [Google Scholar] [CrossRef]
- Hurria, A.; Jones, L.; Muss, H.B. Cancer Treatment as an Accelerated Aging Process: Assessment, Biomarkers, and Interventions. Am. Soc. Clin. Oncol. Educ. Book 2016, 36, e516–e522. [Google Scholar] [CrossRef]
- Kneuertz, P.J.; Pitt, H.A.; Bilimoria, K.Y.; Smiley, J.P.; Cohen, M.E.; Ko, C.Y.; Pawlik, T.M. Risk of Morbidity and Mortality Following Hepato-Pancreato-Biliary Surgery. J. Gastrointest. Surg. 2012, 16, 1727–1735. [Google Scholar] [CrossRef]
- Ann, S.M.; Bonney, G.K.; Balakrishnan, A. Prehabilitation for Hepatobiliary-Pancreatic Cancer Surgery. In Prehabilitation for Cancer Surgery; Springer: Singapore, 2022; pp. 251–261. [Google Scholar]
- American Cancer Society. Treating Liver Cancer: Surgery for Liver Cancer. 2022. Updated 19 April 2019. Available online: https://www.cancer.org/cancer/liver-cancer/treating/surgery.html (accessed on 1 August 2023).
- American Cancer Society. Treating Pancreatic Cancer: Treating Pancreatic Cancer, Based on Extend of the Cancer. 2022. Updated 2 January 2020. Available online: https://www.cancer.org/cancer/pancreatic-cancer/treating/by-stage.html (accessed on 1 August 2023).
- American Cancer Society. Treating Bile Duct Cancer: Treatment Options Based on the Extent of Bile Duct Cancer. 2022. Updated 30 September 2022. Available online: https://www.cancer.org/cancer/bile-duct-cancer/treating/based-on-situation.html (accessed on 1 August 2023).
- Gunasekaran, G.; Bekki, Y.; Lourdusamy, V.; Schwartz, M. Surgical Treatments of Hepatobiliary Cancers. Hepatology 2021, 73, 128–136. [Google Scholar] [CrossRef]
- Colavita, P.D.; Tsirline, V.B.; Belyansky, I.; Swan, R.Z.; Walters, A.L.; Lincourt, A.E.; Iannitti, D.A.; Heniford, B.T. Regionalization and Outcomes of Hepato-pancreato-biliary Cancer Surgery in USA. J. Gastrointest. Surg. 2014, 18, 532–541. [Google Scholar] [CrossRef]
- Ellington, T.D.; Momin, B.; Wilson, R.J.; Henley, S.J.; Wu, M.; Ryerson, A.B. Incidence and Mortality of Cancers of the Biliary Tract, Gallbladder, and Liver by Sex, Age, Race/Ethnicity, and Stage at Diagnosis: United States, 2013 to 2017 Incidence and Mortality of Cancers of the Biliary Tract. Cancer Epidemiol. Biomark. Prev. 2021, 30, 1607–1614. [Google Scholar] [CrossRef]
- Mylius, C.F.; Krijnen, W.P.; Takken, T.; Lips, D.J.; Eker, H.; Van Der Schans, C.P.; Klaase, J.M. Objectively measured preoperative physical activity is associated with time to functional recovery after hepato-pancreato-biliary cancer surgery: A pilot study. Perioper. Med. 2021, 10, 33. [Google Scholar] [CrossRef]
- Herold, Z.; Szasz, A.M.; Dank, M. Evidence based tools to improve efficiency of currently administered oncotherapies for tumors of the hepatopancreatobiliary system. World J. Gastrointest. Oncol. 2021, 13, 1109–1120. [Google Scholar] [CrossRef] [PubMed]
- Llovet, J.M.; Kelley, R.K.; Villanueva, A.; Singal, A.G.; Pikarsky, E.; Roayaie, S.; Lencioni, R.; Koike, K.; Zucman-Rossi, J.; Finn, R.S. Hepatocellular carcinoma. Nat. Rev. Dis. Primers 2021, 7, 6. [Google Scholar] [CrossRef] [PubMed]
- Buchard, B.; Boirie, Y.; Cassagnes, L.; Lamblin, G.; Coilly, A.; Abergel, A. Assessment of Malnutrition, Sarcopenia and Frailty in Patients with Cirrhosis: Which Tools Should We Use in Clinical Practice? Nutrients 2020, 12, 186. [Google Scholar] [CrossRef]
- Cornet, M.; Lim, C.; Salloum, C.; Lazzati, A.; Compagnon, P.; Pascal, G.; Azoulay, D. Prognostic value of sarcopenia in liver surgery. J. Visc. Surg. 2015, 152, 297–304. [Google Scholar] [CrossRef] [PubMed]
- Hall, A.; Smith, R.; Older, P.; French, C. Preoperative cardiopulmonary risk assessment by cardiopulmonary exercise testing. Crit. Care Resusc. 2000, 2, 198–208. [Google Scholar]
- Robinson, T.N.; Wu, D.S.; Pointer, L.; Dunn, C.L.; Cleveland, J.C., Jr.; Moss, M. Simple frailty score predicts postoperative complications across surgical specialties. Am. J. Surg. 2013, 206, 544–550. [Google Scholar] [CrossRef]
- Snowden, C.P.; Prentis, J.M.; Anderson, H.L.; Roberts, D.R.; Randles, D.; Renton, M.; Manas, D.M. Submaximal cardiopulmonary exercise testing predicts complications and hospital length of stay in patients undergoing major elective surgery. Ann. Surg. 2010, 251, 535–541. [Google Scholar] [CrossRef]
- Yokoyama, Y.; Nagino, M.; Ebata, T. Importance of “muscle” and “intestine” training before major HPB surgery: A review. J. Hepato-Biliary-Pancreat. Sci. 2021, 28, 545–555. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.; Zhou, J.; Norris, M.K.; Chow, C.; Dieli-Conwright, C.M. Prehabilitative Exercise for the Enhancement of Physical, Psychosocial, and Biological Outcomes Among Patients Diagnosed with Cancer. Curr. Oncol. Rep. 2020, 22, 71. [Google Scholar] [CrossRef] [PubMed]
- Xue, Q.-L. The Frailty Syndrome: Definition and Natural History. Clin. Geriatr. Med. 2011, 27, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.K.; Fielding, R.A. Exercise as an Intervention for Frailty. Clin. Geriatr. Med. 2011, 27, 101–110. [Google Scholar] [CrossRef] [PubMed]
- Lee, L.; Heckman, G.; Molnar, F.J. Frailty: Identifying elderly patients at high risk of poor outcomes. Can. Fam. Physician 2015, 61, 227–231. [Google Scholar] [PubMed]
- Kabashneh, S.; Alkassis, S.; Shanah, L.; Ali, H. A Complete Guide to Identify and Manage Malnutrition in Hospitalized Patients. Cureus 2020, 12, e8486. [Google Scholar] [CrossRef] [PubMed]
- Saunders, J.; Smith, T. Malnutrition: Causes and consequences. Clin. Med. 2010, 10, 624–627. [Google Scholar] [CrossRef] [PubMed]
- Argiles, J. Cancer-associated malnutrition. Eur. J. Oncol. Nurs. 2005, 9, S39–S50. [Google Scholar] [CrossRef] [PubMed]
- Lambert, J.E.; Hayes, L.D.; Keegan, T.J.; Subar, D.A.; Gaffney, C.J. The Impact of Prehabilitation on Patient Outcomes in Hepatobiliary, Colorectal, and Upper Gastrointestinal Cancer Surgery. Ann. Surg. 2021, 274, 70–77. [Google Scholar] [CrossRef]
- Dewulf, M.; Verrips, M.; Coolsen, M.M.E.; Olde Damink, S.W.M.; Den Dulk, M.; Bongers, B.C.; Dejong, K.; Bouwense, S.A.W. The effect of prehabilitation on postoperative complications and postoperative hospital stay in hepatopancreatobiliary surgery a systematic review. HPB 2021, 23, 1299–1310. [Google Scholar] [CrossRef]
- Joliat, G.R.; Kobayashi, K.; Hasegawa, K.; Thomson, J.E.; Padbury, R.; Scott, M.; Brustia, R.; Scatton, O.; Tran Cao, H.S.; Vauthey, J.N.; et al. Guidelines for Perioperative Care for Liver Surgery: Enhanced Recovery After Surgery (ERAS) Society Recommendations 2022. World J. Surg. 2023, 47, 11–34. [Google Scholar] [CrossRef] [PubMed]
- Durrand, J.; Singh, S.J.; Danjoux, G. Prehabilitation. Clin. Med. 2019, 19, 458–464. [Google Scholar] [CrossRef] [PubMed]
- Minnella, E.M.; Carli, F. Prehabilitation and functional recovery for colorectal cancer patients. Eur. J. Surg. Oncol. 2018, 44, 919–926. [Google Scholar] [CrossRef] [PubMed]
- Gurlit, S.; Gogol, M. Prehabilitation is better than cure. Curr. Opin. Anesthesiol. 2019, 32, 108–115. [Google Scholar] [CrossRef]
- Mohan, R.; Huey, C.W.T.; Junnarkar, S.; Low, J.K.; Shelat, V.G. Prehabilitation in elderly patients scheduled for liver resection and protocol for Recovery Of Surgery in Elderly. Hepatoma Res. 2020, 6, 13. [Google Scholar] [CrossRef]
- Boereboom, C.; Doleman, B.; Lund, J.N.; Williams, J.P. Systematic review of pre-operative exercise in colorectal cancer patients. Tech. Coloproctol. 2016, 20, 81–89. [Google Scholar] [CrossRef]
- Michael, C.M.; Lehrer, E.J.; Schmitz, K.H.; Zaorsky, N.G. Prehabilitation exercise therapy for cancer: A systematic review and meta-analysis. Cancer Med. 2021, 10, 4195–4205. [Google Scholar] [CrossRef]
- Bongers, B.C.; Dejong, C.H.C.; den Dulk, M. Enhanced recovery after surgery programmes in older patients undergoing hepatopancreatobiliary surgery: What benefits might prehabilitation have? Eur. J. Surg. Oncol. 2021, 47, 551–559. [Google Scholar] [CrossRef]
- Deprato, A.; Verhoeff, K.; Purich, K.; Kung, J.Y.; Bigam, D.L.; Dajani, K.Z. Surgical outcomes and quality of life following exercise-based prehabilitation for hepato-pancreatico-biliary surgery: A systematic review and meta-analysis. Hepatobiliary Pancreat. Dis. Int. 2022, 21, 207–217. [Google Scholar] [CrossRef]
- Dagorno, C.; Sommacale, D.; Laurent, A.; Attias, A.; Mongardon, N.; Levesque, E.; Langeron, O.; Rhaiem, R.; Leroy, V.; Amaddeo, G. Prehabilitation in hepato-pancreato-biliary surgery: A systematic review and meta-analysis. A necessary step forward evidence-based sample size calculation for future trials. J. Visc. Surg. 2022, 159, 362–372. [Google Scholar] [CrossRef]
- Kitahata, Y.; Hirono, S.; Kawai, M.; Okada, K.I.; Miyazawa, M.; Shimizu, A.; Kobayashi, R.; Ueno, M.; Hayami, S.; Shimokawa, T.; et al. Intensive perioperative rehabilitation improves surgical outcomes after pancreaticoduodenectomy. Langenbecks Arch. Surg. 2018, 403, 711–718. [Google Scholar] [CrossRef]
- Mikami, Y.; Kouda, K.; Kawasaki, S.; Okada, K.I.; Kawai, M.; Kitahata, Y.; Miyazawa, M.; Hirono, S.; Unno, M.; Tajima, F.; et al. Preoperative In-Hospital Rehabilitation Improves Physical Function in Patients with Pancreatic Cancer Scheduled for Surgery. Tohoku J. Exp. Med. 2020, 251, 279–285. [Google Scholar] [CrossRef] [PubMed]
- Ngo-Huang, A.; Parker, N.H.; Bruera, E.; Lee, R.E.; Simpson, R.; O’Connor, D.P.; Petzel, M.Q.B.; Fontillas, R.C.; Schadler, K.; Xiao, L.; et al. Home-Based Exercise Prehabilitation During Preoperative Treatment for Pancreatic Cancer Is Associated with Improvement in Physical Function and Quality of Life. Integr. Cancer Ther. 2019, 18. [Google Scholar] [CrossRef] [PubMed]
- Ngo-Huang, A.; Parker, N.H.; Wang, X.; Petzel, M.Q.B.; Fogelman, D.; Schadler, K.L.; Bruera, E.; Fleming, J.B.; Lee, J.E.; Katz, M.H.G. Home-based exercise during preoperative therapy for pancreatic cancer. Langenbecks Arch. Surg. 2017, 402, 1175–1185. [Google Scholar] [CrossRef] [PubMed]
- Parker, N.H.; Gorzelitz, J.; Ngo-Huang, A.; Caan, B.J.; Prakash, L.; Garg, N.; Petzel, M.Q.B.; Schadler, K.; Basen-Engquist, K.; Katz, M.H.G. The Role of Home-Based Exercise in Maintaining Skeletal Muscle During Preoperative Pancreatic Cancer Treatment. Integr. Cancer Ther. 2021, 20, 1534735420986615. [Google Scholar] [CrossRef] [PubMed]
- Parker, N.H.; Ngo-Huang, A.; Lee, R.E.; O’Connor, D.P.; Basen-Engquist, K.M.; Petzel, M.Q.B.; Wang, X.; Xiao, L.; Fogelman, D.R.; Schadler, K.L.; et al. Physical activity and exercise during preoperative pancreatic cancer treatment. Support Care Cancer 2019, 27, 2275–2284. [Google Scholar] [CrossRef] [PubMed]
- Ausania, F.; Senra, P.; Meléndez, R.; Caballeiro, R.; Ouviña, R.; Casal-Núñez, E. Prehabilitation in patients undergoing pancreaticoduodenectomy: A randomized controlled trial. Rev. Esp. Enferm. Dig. 2019, 111, 603–608. [Google Scholar] [CrossRef]
- van Wijk, L.; Bongers, B.C.; Berkel, A.E.M.; Buis, C.I.; Reudink, M.; Liem, M.S.L.; Slooter, G.D.; van Meeteren, N.L.U.; Klaase, J.M. Improved preoperative aerobic fitness following a home-based bimodal prehabilitation programme in high-risk patients scheduled for liver or pancreatic resection. Br. J. Surg. 2022, 109, 1036–1039. [Google Scholar] [CrossRef]
- Kaibori, M.; Ishizaki, M.; Matsui, K.; Nakatake, R.; Yoshiuchi, S.; Kimura, Y.; Kwon, A.H. Perioperative exercise for chronic liver injury patients with hepatocellular carcinoma undergoing hepatectomy. Am. J. Surg. 2013, 206, 202–209. [Google Scholar] [CrossRef]
- Dunne, D.F.; Jack, S.; Jones, R.P.; Jones, L.; Lythgoe, D.T.; Malik, H.Z.; Poston, G.J.; Palmer, D.H.; Fenwick, S.W. Randomized clinical trial of prehabilitation before planned liver resection. Br. J. Surg. 2016, 103, 504–512. [Google Scholar] [CrossRef]
- Baimas-George, M.; Watson, M.; Thompson, K.; Shastry, V.; Iannitti, D.; Martinie, J.B.; Baker, E.; Parala-Metz, A.; Vrochides, D. Prehabilitation for Hepatopancreatobiliary Surgical Patients: Interim Analysis Demonstrates a Protective Effect From Neoadjuvant Chemotherapy and Improvement in the Frailty Phenotype. Am. Surg. 2021, 87, 714–724. [Google Scholar] [CrossRef] [PubMed]
- Nakajima, H.; Yokoyama, Y.; Inoue, T.; Nagaya, M.; Mizuno, Y.; Kadono, I.; Nishiwaki, K.; Nishida, Y.; Nagino, M. Clinical Benefit of Preoperative Exercise and Nutritional Therapy for Patients Undergoing Hepato-Pancreato-Biliary Surgeries for Malignancy. Ann. Surg. Oncol. 2019, 26, 264–272. [Google Scholar] [CrossRef]
- Wang, B.; Shelat, V.G.; Chow, J.J.L.; Huey, T.C.W.; Low, J.K.; Woon, W.W.L.; Junnarkar, S.P. Prehabilitation Program Improves Outcomes of Patients Undergoing Elective Liver Resection. J. Surg. Res. 2020, 251, 119–125. [Google Scholar] [CrossRef] [PubMed]
- Bui Ngoc, T. Prehabilitation in Patients Undergoing Surgery for Hepatobiliary or Pancreatic Cancer. Ph.D. Thesis, Concordia University, Montreal, QC, Canada, 2019. [Google Scholar]
- Arends, J.; Bachmann, P.; Baracos, V.; Barthelemy, N.; Bertz, H.; Bozzetti, F.; Fearon, K.; Hütterer, E.; Isenring, E.; Kaasa, S.; et al. ESPEN guidelines on nutrition in cancer patients. Clin. Nutr. 2017, 36, 11–48. [Google Scholar] [CrossRef] [PubMed]
- Carli, F.; Gillis, C.; Scheede-Bergdahl, C. Promoting a culture of prehabilitation for the surgical cancer patient. Acta Oncol. 2017, 56, 128–133. [Google Scholar] [CrossRef] [PubMed]
- Trestini, I.; Cintoni, M.; Rinninella, E.; Grassi, F.; Paiella, S.; Salvia, R.; Bria, E.; Pozzo, C.; Alfieri, S.; Gasbarrini, A. Neoadjuvant treatment: A window of opportunity for nutritional prehabilitation in patients with pancreatic ductal adenocarcinoma. World J. Gastrointest. Surg. 2021, 13, 885–903. [Google Scholar] [CrossRef] [PubMed]
- Piraux, E.; Caty, G.; Reychler, G. Effects of preoperative combined aerobic and resistance exercise training in cancer patients undergoing tumour resection surgery: A systematic review of randomised trials. Surg. Oncol. 2018, 27, 584–594. [Google Scholar] [CrossRef] [PubMed]
- Steffens, D.; Beckenkamp, P.R.; Young, J.; Solomon, M.; da Silva, T.M.; Hancock, M.J. Is preoperative physical activity level of patients undergoing cancer surgery associated with postoperative outcomes? A systematic review and meta-analysis. Eur. J. Surg. Oncol. 2019, 45, 510–518. [Google Scholar] [CrossRef]
- De Luca, R.; Gianotti, L.; Pedrazzoli, P.; Brunetti, O.; Rizzo, A.; Sandini, M.; Paiella, S.; Pecorelli, N.; Pugliese, L.; Pietrabissa, A.; et al. Immunonutrition and prehabilitation in pancreatic cancer surgery: A new concept in the era of ERAS® and neoadjuvant treatment. Eur. J. Surg. Oncol. 2023, 49, 542–549. [Google Scholar] [CrossRef]
- Silver, J.K.; Baima, J.; Mayer, R.S. Impairment-driven cancer rehabilitation: An essential component of quality care and survivorship. CA Cancer J. Clin. 2013, 63, 295–317. [Google Scholar] [CrossRef]
- Brown, J.C.; Winters-Stone, K.; Lee, A.; Schmitz, K.H. Cancer, Physical Activity, and Exercise. Compr. Physiol. 2012, 2, 2775–2809. [Google Scholar] [CrossRef]
- Baracos, V.; Kazemi-Bajestani, S.M. Clinical outcomes related to muscle mass in humans with cancer and catabolic illnesses. Int. J. Biochem. Cell Biol. 2013, 45, 2302–2308. [Google Scholar] [CrossRef] [PubMed]
- Martin, L.; Birdsell, L.; Macdonald, N.; Reiman, T.; Clandinin, M.T.; McCargar, L.J.; Murphy, R.; Ghosh, S.; Sawyer, M.B.; Baracos, V.E. Cancer cachexia in the age of obesity: Skeletal muscle depletion is a powerful prognostic factor, independent of body mass index. J. Clin. Oncol. 2013, 31, 1539–1547. [Google Scholar] [CrossRef] [PubMed]
- Fulghum, K.; Hill, B.G. Metabolic mechanisms of exercise-induced cardiac remodeling. Front. Cardiovasc. Med. 2018, 5, 127. [Google Scholar] [CrossRef] [PubMed]
- Ma, C.; Lin, M.; Gao, J.; Xu, S.; Huang, L.; Zhu, J.; Huang, J.; Tao, J.; Chen, L. The impact of physical activity on blood inflammatory cytokines and neuroprotective factors in individuals with mild cognitive impairment: A systematic review and meta-analysis of randomized-controlled trials. Aging Clin. Exp. Res. 2022, 34, 1471–1484. [Google Scholar] [CrossRef]
- Proschinger, S.; Winker, M.; Joisten, N.; Bloch, W.; Palmowski, J.; Zimmer, P. The effect of exercise on regulatory T cells: A systematic review of human and animal studies with future perspectives and methodological recommendations. Exerc. Immunol. Rev. 2021, 27, 142–166. [Google Scholar]
- Gillis, C.; Wischmeyer, P.E. Pre-operative nutrition and the elective surgical patient: Why, how and what? Anaesthesia 2019, 74 (Suppl. S1), 27–35. [Google Scholar] [CrossRef] [PubMed]
- Pillinger, N.L.; Robson, J.L.; Kam, P. Nutritional prehabilitation: Physiological basis and clinical evidence. Anaesth. Intensive Care 2018, 46, 453–462. [Google Scholar] [CrossRef]
- Gillis, C.; Carli, F. Promoting Perioperative Metabolic and Nutritional Care. Anesthesiology 2015, 123, 1455–1472. [Google Scholar] [CrossRef]
- Prado, C.M.; Ford, K.L.; Gonzalez, M.C.; Murnane, L.C.; Gillis, C.; Wischmeyer, P.E.; Morrison, C.A.; Lobo, D.N. Nascent to novel methods to evaluate malnutrition and frailty in the surgical patient. J. Parenter. Enter. Nutr. 2023, 47, S54–S68. [Google Scholar] [CrossRef]
- Weimann, A. Is there a rationale for perioperative nutrition therapy in the times of ERAS? Innov. Surg. Sci. 2019, 4, 152–157. [Google Scholar] [CrossRef]
- Janevska, D.; Chaloska-Ivanova, V.; Janevski, V. Hepatocellular Carcinoma: Risk Factors, Diagnosis and Treatment. Open Access Maced. J. Med. Sci. 2015, 3, 732–736. [Google Scholar] [CrossRef]
- Mima, K.; Nakagawa, S.; Miyata, T.; Yamashita, Y.I.; Baba, H. Frailty and surgical outcomes in gastrointestinal cancer: Integration of geriatric assessment and prehabilitation into surgical practice for vulnerable patients. Ann. Gastroenterol. Surg. 2023, 7, 27–41. [Google Scholar] [CrossRef] [PubMed]
- Giallauria, F.; Cittadini, A.; Smart, N.A.; Vigorito, C. Resistance training and sarcopenia. Monaldi Arch. Chest Dis. 2015, 84. [Google Scholar] [CrossRef] [PubMed]
- Cao, A.; Ferrucci, L.M.; Caan, B.J.; Irwin, M.L. Effect of exercise on sarcopenia among cancer survivors: A systematic review. Cancers 2022, 14, 786. [Google Scholar] [CrossRef] [PubMed]
- Williams, G.R.; Dunne, R.F.; Giri, S.; Shachar, S.S.; Caan, B.J. Sarcopenia in the older adult with cancer. J. Clin. Oncol. 2021, 39, 2068. [Google Scholar] [CrossRef] [PubMed]
- Scott, J.M.; Nilsen, T.S.; Gupta, D.; Jones, L.W. Exercise Therapy and Cardiovascular Toxicity in Cancer. Circulation 2018, 137, 1176–1191. [Google Scholar] [CrossRef] [PubMed]
- Campbell, K.L.; Winters-Stone, K.M.; Wiskemann, J.; May, A.M.; Schwartz, A.L.; Courneya, K.S.; Zucker, D.S.; Matthews, C.E.; Ligibel, J.A.; Gerber, L.H.; et al. Exercise Guidelines for Cancer Survivors: Consensus Statement from International Multidisciplinary Roundtable. Med. Sci. Sports Exerc. 2019, 51, 2375–2390. [Google Scholar] [CrossRef]
- Debette-Gratien, M.; Tabouret, T.; Antonini, M.T.; Dalmay, F.; Carrier, P.; Legros, R.; Jacques, J.; Vincent, F.; Sautereau, D.; Samuel, D.; et al. Personalized adapted physical activity before liver transplantation: Acceptability and results. Transplantation 2015, 99, 145–150. [Google Scholar] [CrossRef]
- Barberan-Garcia, A.; Ubré, M.; Roca, J.; Lacy, A.M.; Burgos, F.; Risco, R.; Momblán, D.; Balust, J.; Blanco, I.; Martínez-Pallí, G. Personalised Prehabilitation in High-risk Patients Undergoing Elective Major Abdominal Surgery: A Randomized Blinded Controlled Trial. Ann. Surg. 2018, 267, 50–56. [Google Scholar] [CrossRef]
- Chakraborty, A.; Balakrishnan, A. Prehabilitation for Cancer Surgery; Springer: Singapore, 2022. [Google Scholar]
- Barnes, O.; Wilson, R.L.; Gonzalo-Encabo, P.; Kang, D.W.; Christopher, C.N.; Bentley, T.; Dieli-Conwright, C.M. The Effect of Exercise and Nutritional Interventions on Body Composition in Patients with Advanced or Metastatic Cancer: A Systematic Review. Nutrients 2022, 14, 2110. [Google Scholar] [CrossRef] [PubMed]
- Bibby, N.; Griffin, O. Nutritional considerations for the management of the older person with hepato-pancreatico-biliary malignancy. Eur. J. Surg. Oncol. 2021, 47, 533–538. [Google Scholar] [CrossRef] [PubMed]
- Al-Jumayli, M. Senior Adult Hepatobiliary Prehab Study. ClinicalTrials.gov Identifier: NCT05921552. Updated 27 June 2023. Available online: https://www.clinicaltrials.gov/study/NCT05921552?term=NCT05921552&rank=1 (accessed on 24 September 2023).
- Berardi, G. Nutrition and Exercise Prehabilitation to Reduce Morbidity Following Major Liver Surgery in Sarcopenic Patients (NEXPREM). ClinicalTrials.gov Identifier: NCT05281211. Updated 17 May 2022. Available online: https://www.clinicaltrials.gov/study/NCT05281211?term=NCT05281211&rank=1 (accessed on 24 September 2023).
- Case Comprehensive Cancer Center. Multidimensional Prehabilitation in Pancreatic Surgery for Pancreatic and Periampullary Neoplasms. ClinicalTrials.gov Identifier: NCT05225038. Updated 10 May 2023. Available online: https://www.clinicaltrials.gov/study/NCT05225038?term=NCT05225038&rank=1 (accessed on 24 September 2023).
- dels Angels Cebria i Iranzo, M. Prehabilitation and Posttransplant Training Program in Liver Transplantation (PreLiveR-T). ClinicalTrials.gov Identifier: NCT04246970. Updated 1 March 2023. Available online: https://clinicaltrials.gov/study/NCT04246970 (accessed on 24 September 2023).
- Dieli-Conwright, C. Resistance Exercise Combined with Protein Supplementation in People with Pancreatic Cancer: The RE-BUILD Trial. ClinicalTrials.gov Identifier: NCT05356117. Updated 21 February 2023. Available online: https://clinicaltrials.gov/study/NCT05356117 (accessed on 24 September 2023).
- Ejaz, A. The RIOT Trial: Re-Defining Frailty and Improving Outcomes with Prehabilitation for Pancreatic, Liver, or Gastric Cancer (RIOT). ClinicalTrials.gov Identifier: NCT04602026. Updated 29 November 2022. Available online: https://www.clinicaltrials.gov/study/NCT04602026?term=NCT04602026&rank=1 (accessed on 24 September 2023).
- Elamrani, M. Preoperative Prehabilitation for Sarcopenic Patients Prior to Pancreatic Surgery for Cancer (PSOAS). ClinicalTrials.gov Identifier: NCT04469504. Updated 8 February 2023. Available online: https://classic.clinicaltrials.gov/ct2/show/NCT04469504 (accessed on 24 September 2023).
- González, C. Evaluation of Prehabilitation as a Strategy to Minimize Surgical Risk in Pancreatic Surgery: Efficacy and Determining Factors. ClinicalTrials.gov Identifier: NCT05489419. Updated 5 August 2022. Available online: https://clinicaltrials.gov/study/NCT05489419 (accessed on 24 September 2023).
- Hedrick, T. Exercise Preconditioning with Cancer Surgeries (ExPre 01). ClinicalTrials.gov Identifier: NCT04923672. Updated 4 August 2023. Available online: https://clinicaltrials.gov/study/NCT04923672 (accessed on 24 September 2023).
- M.D. Anderson Cancer Center. PancFit: Multimodal Exercise during Preoperative Therapy for Pancreatic Cancer. ClinicalTrials.gov Identifier: NCT03187951. Updated 18 April 2023. Available online: https://clinicaltrials.gov/study/NCT03187951 (accessed on 24 September 2023).
- Mijwel, S. Metabolic Stress-Induced Exercise to Prevent Loss of Muscle Mass in Patients with Pancreatic and Biliary Tract Cancer (PREV-Ex). ClinicalTrials.gov Identifier: NCT05044065. Updated 8 April 2022. Available online: https://clinicaltrials.gov/study/NCT05044065 (accessed on 24 September 2023).
- Ngo-Huang, A. Tele-PancFit: A Multi-Site Trial of Video-Based Strengthening Exercise Prehabilitation for Patients with Resectable Pancreatic Cancer ClinicalTrials.gov Identifier: NCT05836870 Updated 20 July 2023. Available online: https://clinicaltrials.gov/study/NCT05836870 (accessed on 24 September 2023).
- Obserstein, P. Feasibility of a Health Care Provider Guided Exercise Intervention Prior to Surgical Resection of Pancreatic Cancer. ClinicalTrials.gov Identifier: NCT05483075. Updated 29 November 2022. Available online: https://classic.clinicaltrials.gov/ct2/show/NCT05483075 (accessed on 24 September 2023).
- Sell, N. Pilot Study of a Multimodal Prehabilitation Pancreatic Cancer Program. ClinicalTrials.gov Identifier: NCT03865875. Updated 17 May 2021. Available online: https://clinicaltrials.gov/study/NCT03865875 (accessed on 24 September 2023).
- Vigano, A. Improving Outcomes in Cancer Patients with a Nutritional and Physical Conditioning Prehabilitation Program. ClinicalTrials.gov Identifier: NCT03475966. Updated 8 December 2020. Available online: https://clinicaltrials.gov/study/NCT03475966 (accessed on 24 September 2023).
| Author, Year, Country | Design and Groups | Cancer Type | Cancer Treatments | Sample (n) | Exercise Type/Setting | Intervention Details | Length (Weeks) | Frequency (Times per Week) | Intensity | Time (min per Session) |
|---|---|---|---|---|---|---|---|---|---|---|
| Dunne et al., 2016, United Kingdom [54] | RCT (EX vs. SOC) | Colorectal liver metastasis | Mixed | 38 | AE/Supervised | AE: HIIT (Cycling) | 4 | 3 | AE: <60% VO2max (low intervals) to 90% VO2max (high intervals) | 30 |
| Kitahata et al., 2018, Germany [45] | Retrospective cohort (EX vs. SOC) | Pancreatic, bile duct, duodenum, and other undergoing pancreato-duodenectomy | Mixed | 576 | COMB (AE + RE)/Mixed | AE: Stationary bike, treadmill, and stair stepping RE: NR Other: Breathing exercises | 1 | 14 (twice a day) | AE: 60%VO2max RE: NR | 70 |
| Mikami et al., 2020, Japan [46] | Single group | Pancreatic | Mixed (n = 4 on preoperative chemotherapy) | 26 | COMB (AE + RE)/Supervised | AE: Cycle ergometer, handgrip ergometry, treadmill walking RE: Body weight | Varied 11.9 days (Median) | 7 (once a day) | AE: 11–13 RPE (Borg scale) RE: NR | 70 |
| Ngo-Huang et al., 2017 and 2019 [47,48] Parker et al., 2019 and 2021, United States [49,50] | Single group | Pancreatic | Chemotherapy and/or chemoradiation | 20 | COMB (AE + RE)/ Home-based | AE: Walking RE: Full body (resistance bands, dumbbells, or machines) | Varied | 2 to 3 | AE: 12–13 RPE (Borg scale) RE: 12–13 RPE (Borg scale) | AE: ≥20 RE: ≥30 |
| Author, Year, Country | Design and Groups | Cancer Type | Cancer Treatments | Sample (n) | Exercise Type/ Setting | Intervention Details | Length (Weeks) | Frequency (Times per Week) | Intensity | Time (min per Session) |
|---|---|---|---|---|---|---|---|---|---|---|
| Ausania et al., 2019, Spain [51] | RCT (EX vs. SOC) | Pancreatic or peripancreatic malignancy | No treatment | 40 | COMB (AE + RE)/ Mixed | AE: HIIT (supervised cycling and unsupervised functional exercises) RE: Home-based, NR NUT: Nutrition support (oral supplementation), endocrine and exocrine support | Varied 12.6 days (Median) | Supervised: 5 Home-based: Daily (following supervised period) | Supervised: High intensity Home-based: NR | Supervised: 60 total (20 AE + 20 RE) Home-based: NR |
| Baimas-George et al., 2020, United States [55] | Single group | HPB malignancy | Neoadjuvant chemotherapy | 19 | AE/Home-based | AE: NR NUT: NR Other: Psychological services) | 16 | NR | NR | NR |
| Bui-Ngoc et al., 2019, Canada [58] | RCT (Prehab vs. Rehab) | HPB and pancreatic | Neoadjuvant chemotherapy and no treatment | 35 | COMB (AE + RE)/Mixed | AE: Walking RE: Full body strength with body weight and resistance bands NUT: Whey protein powder Other: Relaxation for stress, anxiety, and pain management + targeted stretching | 4 | AE: Daily RE: 3–4 | AE: 3–4 RPE (Borg scale) RE: 2 sets of 8–15 reps | AE: 30 RE: 30 |
| Kaibori et al., 2013, Japan [53] | RCT (EX + Diet vs. Diet-alone) | Hepatocellular carcinoma | NR | 51 | AE/Supervised | AE: Walking (+targeted stretching) NUT: Adapted to either chronic hepatitis/liver cirrhosis or diabetes/fatty liver | 4 (pre-operatively) + 1-week post-operative for 6 months | 3 | AE: Anaerobic threshold | 60 |
| Nakajima et al., 2019, Japan [56] | Non-randomized trial (EX vs. Historical Controls on SOC) | HPB malignancy | No treatment | 142 | COMB (AE + RE)/Home-based | AE: Walking RE: Body weight or free weights (squats, calf raises, sit-ups, bridge up, and upper limb) NUT: Leucine-rich amino acid supplement | Varied | 3 (minimum) | AE: 3–4 RPE (Borg scale) RE: NR | 30 |
| Van Wijk et al., 2022, Netherlands [52] | Single group | Liver or pancreatic | NR | 26 | AE/Mixed | AE: Personalized cycle ergometer (2 HIIT and 1 MIIT sessions) NUT: Protein and multivitamin supplementation | 4 | AE: HIIT: 2 days/week AE: MIIT: 1 days/week | AE: HIIT: 60% of peak work rate to 20% of peak work rate AE: MIIT: 40% of peak work rate to 20% of peak work rate | 30 |
| Wang et al., 2020, Singapore [57] | Non-randomized trial (EX vs. Controls on SOC) | HPB | NR | 104 | COMB (AE + RE)/Home-based | AE: Walking RE: Individualized lower limb strengthening NUT: Tailored Other: Psychosocial care | 2 to 4 | 5 | NR | 30 |
| Study | Intervention | Main Result |
|---|---|---|
| Dunne et al., 2016, United Kingdom [54] | AE | ⇑ VO2 peak (EX vs. SOC) ⇑ Peak work rate (EX vs. SOC) ↔ Heart rate reserve (EX vs. SOC) |
| Mikami et al., 2020, Japan [46] | COMB | ⇑ VO2 peak (Pre vs. Post) ⇑ Peak work rate (Pre vs. Post) ⇑ 6MWT distance (Pre vs. Post) |
| Ngo-Huang et al., 2017, United States [48] | COMB | ⇑ Grip strength (Pre vs. Post) ⇑ MET min/week of physical activity (Pre vs. Post) |
| Ausania et al., 2019, Spain [51] | COMB + NUT | ⇑ Cardiopulmonary status (Pre vs. Post) ⇓ Time to complete 10 m walk test (Pre vs. Post) ⇑ Grip strength (Pre vs. Post) |
| Kaibori et al., 2013, Japan [53] | AE + NUT | ↔ Muscle strength (Pre vs. Post) ⇓ Heart rate variability (Pre vs. Post) |
| Nakajima et al., 2019, Japan [56] | COMB + NUT | ⇑ VO2 peak (Pre vs. Post) ⇑ Peak work rate (Pre vs. Post) |
| Ngo-Huang et al., 2019, United States [47] | COMB | ⇑ 6MWT distance (Pre vs. Post) ⇓ Sit-to-stand time to completion (Pre vs. Post) ↔ Grip strength (Pre vs. Post) ⇑ Gait speed (Pre vs. Post) |
| Baimas-George et al., 2020, United States [55] | AE + NUT | ↔ Grip strength (Pre vs. Post) ⇓ Frailty (Pre vs. Post)
|
| Nakajima et al., 2019, Japan [56] | COMB + NUT | ⇑ 6MWT distance (Pre vs. Post) ↔ Gait speed (Pre vs. Post) ↔ Grip strength (Pre vs. Post) |
| Bui-Ngoc et al., 2019, Canada [58] | COMB + NUT | ↔ 6MWT distance (Prehab: Baseline vs. Post) ⇓ 6MWT distance (Prehab: Pre vs. Post) ⇓ 6MWT distance (Rehab: Baseline vs. Post) ⇓ 6MWT distance (Rehab: Pre vs. Post) ↔ Grip strength (Prehab: Pre vs. Post) ↔ Grip strength (Rehab: Pre vs. Post) ↔ Arm curl—left and right side(Prehab: Pre vs. Post) ⇓ Arm curl—left side (Rehab: Pre vs. Post) ⇑ Timed-up-and-go (Prehab: Pre vs. Post) ↔ Timed-up-and-go (Rehab: Pre vs. Post) ⇓ Knee extension (Prehab: Pre vs. Post) ↔ Knee extension (Rehab: Pre vs. Post) ↔ Knee flexion (Prehab: Pre vs. Post) ↔ Knee flexion (Rehab: Pre vs. Post) |
| Study | Intervention | Main Result |
|---|---|---|
| Baimas-George et al., 2020, United States [55] | AE + NUT | ↔ Nutritional Status (Pre vs. Post) ↔ Glucose (Pre vs. Post) ↔ TSH (Pre vs. Post) ↔ Creatinine (Pre vs. Post) |
| Kaibori et al., 2013, Japan [53] | AE + NUT | ↔ Serum albumin at 6 months post-operation (EX + Diet vs. Diet-Alone) ↔ Triglyceride levels at 6 months post-operation (EX + Diet vs. Diet-Alone) ↔ Cholesterol levels at 6 months post-operation (EX + Diet vs. Diet-Alone) ↔ Glucose levels at 6 months post-operation (EX + Diet vs. Diet-Alone) ↔ Fasting serum insulin and insulin resistance at post-operation (EX + Diet vs. Diet-Alone) ⇓ Fasting serum insulin and insulin resistance at 3 months and 6 months post-operation (EX + Diet vs. Diet-Alone) |
| Nakajima et al., 2019, Japan [56] | COMB + NUT | ⇑ Prognostic nutritional index (EX vs. SOC) ⇑ Serum albumin levels (EX: vs. SOC) |
| Wang et al., 2020, Singapore [57] | COMB + NUT | ↔ Serum albumin (EX vs. SOC) |
| Study | Intervention | Main Result |
|---|---|---|
| Parker et al., 2019 and Parker et al., 2021, United States [49,50] | COMB | ↔ BMI (EX vs. SOC) ⇑ Skeletal muscle index (EX vs. SOC) ↔ Skeletal muscle density (EX vs. SOC) ⇓ BMI (EX: Pre vs. Post) ↔ Skeletal muscle index (EX: Pre vs. Post) ⇓ Skeletal muscle index (SOC: Pre vs. Post) |
| Kaibori et al., 2013, Japan [53] | AE + NUT | ⇓ Whole body mass at 6 months post-operation (EX + Diet vs. Diet-Alone) ⇓ Body mass at waist and fat mass at waist at 6 months post-operation (EX + Diet vs. Diet-Alone) ↔ Whole body fat mass, fat-free mass, and bone mineral density at 6 months post-operation (EX + Diet vs. Diet-Alone) |
| Nakajima et al., 2019, Japan [56] | COMB + NUT | ⇓ Body weight (EX: Within-group change; SOC: Within-group change) ⇓ BMI (EX: Within-group change; SOC: Within-group change) ↔ Skeletal muscle mass (EX: Within-group change) ⇓ Fat mass (EX: Within-group change) ⇑ Muscle-to-fat ratio (EX: Within-group change) |
| Wang et al., 2020, Singapore [57] | COMB + NUT | ↔ BMI (EX vs. SOC) |
| Bui-Ngoc et al., 2019, Canada [58] | COMB + NUT | ↔ Appendicular skeletal muscle index (Prehab: Pre vs. Post) ⇓ Appendicular skeletal muscle index (Rehab: Pre vs. Post) |
| Study | Intervention | Main Result |
|---|---|---|
| Dunne et al., 2016, United Kingdom [54] | AE | ↔ Readmission (EX vs. SOC) ↔ Bile duct reconstruction (EX vs. SOC) ↔ LOS (EX vs. SOC) |
| Kitahata et al., 2018, Germany [45] | COMB | ⇓ Post-operative LOS (EX vs. SOC) ⇓ Pulmonary complications (EX vs. SOC) ↔ Severe complications (EX vs. SOC) ↔ Morbidity (EX vs. SOC) ↔ Mortality (EX vs. SOC) ↔ Bile leakage (EX vs. SOC) ↔ Delayed gastric emptying (EX vs. SOC) |
| Ausania et al., 2019, Spain [51] | COMB + NUT | ↔ Complications (EX vs. SOC) ↔ Post-operative LOS (EX vs. SOC) ↔ Readmission (EX vs. SOC) ⇓ Delayed gastric emptying (EX vs. SOC) |
| Baimas-George et al., 2020, United States [55] | AE + NUT | ↔ LOS (EX + Diet vs. Diet-Alone) ↔ Mortality (EX + Diet vs. Diet-Alone) ↔ Morbidity (EX + Diet vs. Diet-Alone) ↔ Post-operative blood loss (EX + Diet vs. Diet-Alone) |
| Van Wijk et al., 2022, Netherlands [52] | AE | ⇓ Post-operative complication rate (EX vs. SOC) ⇓ Post-operative LOS (EX vs. SOC) ↔ Re-admission rate (EX vs. SOC) |
| Bui-Ngoc et al., 2019, Canada [58] | COMB + NUT | ↔ Post-operative LOS (Prehab vs. Rehab) ↔ Rehospitalization (Prehab vs. Rehab) |
| Identifier | Study Design | Population and Treatment Status | Experimental Groups | Intervention Characteristics | Outcomes of Interest |
|---|---|---|---|---|---|
| NCT05356117 [93] | RCT Length: 16 weeks | Pancreatic cancer on neoadjuvant chemotherapy aged 18 years and older |
|
|
|
| NCT03475966 [103] | RCT Length: NR | Pancreatic cancer, liver cancer, bile duct cancer, aged 18–95 years old |
|
|
|
| NCT04602026 [94] | RCT Length: NR | Pancreatic, liver, or gastric cancer, aged 18 years and older |
|
|
|
| NCT05921552 [89] | Single group Length: Up to 4 weeks before surgery | Hepatocellular carcinoma or liver metastasis, aged 70 years and older |
|
|
|
| NCT05281211 [90] | RCT Length: 6 weeks | Liver cancer, aged 18–90 years with sarcopenia |
|
|
|
| NCT05225038 [91] | RCT Length: At least 2 weeks before surgery | Pancreatic cancer receiving upfront surgery or neoadjuvant chemotherapy, aged 18 and older |
|
|
|
| NCT04246970 [92] | RCT Length: 8 weeks before surgery | Liver cancer and other candidates for liver transplants, aged 18 years and older |
|
|
|
| NCT05489419 [96] | Single group Length: 3–4 weeks | Pancreatic cancer, any ages |
|
|
|
| NCT03865875 [102] | Single group Length: NR | Pancreatic cancer, aged 18 years and older |
|
|
|
| NCT05836870 [100] | RCT Length: NR | Pancreatic cancer, receiving neoadjuvant chemotherapy, aged 18 years and older |
|
|
|
| NCT05483075 [101] | Non-randomized trial Length: 2–4 weeks | Pancreatic cancer, aged 18–90 years |
|
|
|
| NCT04469504 [95] | RCT Length: 4 weeks | Pancreatic cancer patients with sarcopenia, aged 18 years and older |
|
|
|
| NCT05044065 [99] | RCT Length: 5–6 weeks (both pre- and post-operative) | Pancreatic and biliary cancer, aged 18 years and older |
|
|
|
| NCT04923672 [97] | RCT Length: At least 3 weeks before surgery | Hepatobiliary cancer or colorectal cancer, aged 18 years and older |
|
|
|
| NCT03187951 [98] | RCT Length: During chemotherapy and up to 7 months post-operation | Pancreatic cancer scheduled to receive neoadjuvant chemotherapy and/or radiation before surgery, aged 18 years and older |
|
|
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Christopher, C.N.; Kang, D.-W.; Wilson, R.L.; Gonzalo-Encabo, P.; Ficarra, S.; Heislein, D.; Dieli-Conwright, C.M. Exercise and Nutrition Interventions for Prehabilitation in Hepato-Pancreato-Biliary Cancers: A Narrative Review. Nutrients 2023, 15, 5044. https://doi.org/10.3390/nu15245044
Christopher CN, Kang D-W, Wilson RL, Gonzalo-Encabo P, Ficarra S, Heislein D, Dieli-Conwright CM. Exercise and Nutrition Interventions for Prehabilitation in Hepato-Pancreato-Biliary Cancers: A Narrative Review. Nutrients. 2023; 15(24):5044. https://doi.org/10.3390/nu15245044
Chicago/Turabian StyleChristopher, Cami N., Dong-Woo Kang, Rebekah L. Wilson, Paola Gonzalo-Encabo, Salvatore Ficarra, Diane Heislein, and Christina M. Dieli-Conwright. 2023. "Exercise and Nutrition Interventions for Prehabilitation in Hepato-Pancreato-Biliary Cancers: A Narrative Review" Nutrients 15, no. 24: 5044. https://doi.org/10.3390/nu15245044
APA StyleChristopher, C. N., Kang, D.-W., Wilson, R. L., Gonzalo-Encabo, P., Ficarra, S., Heislein, D., & Dieli-Conwright, C. M. (2023). Exercise and Nutrition Interventions for Prehabilitation in Hepato-Pancreato-Biliary Cancers: A Narrative Review. Nutrients, 15(24), 5044. https://doi.org/10.3390/nu15245044