Possible Mechanisms of Dark Tea in Cancer Prevention and Management: A Comprehensive Review
Abstract
:1. Introduction
2. Bioactive Components in Dark Tea
3. Anti-Cancer Effects of Dark Tea and Its Components in Different Cancer Models
4. The Possible Anti-Cancer Mechanisms of Dark Tea
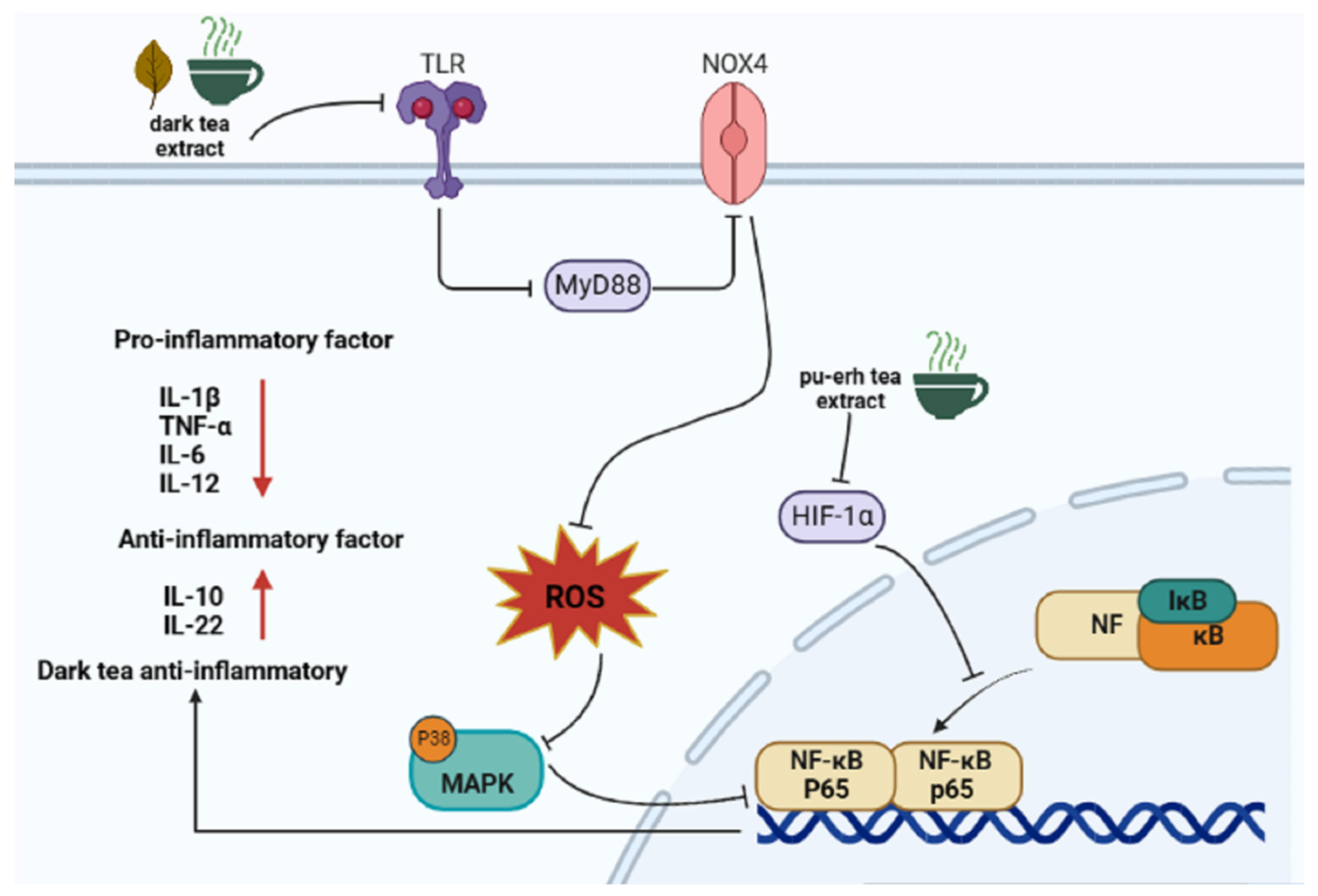
4.1. Anti-Inflammation
4.2. Antioxidation
4.3. Inhibiting the Proliferation of Cancer Cells
4.4. Promoting Cancer Cell Apoptosis
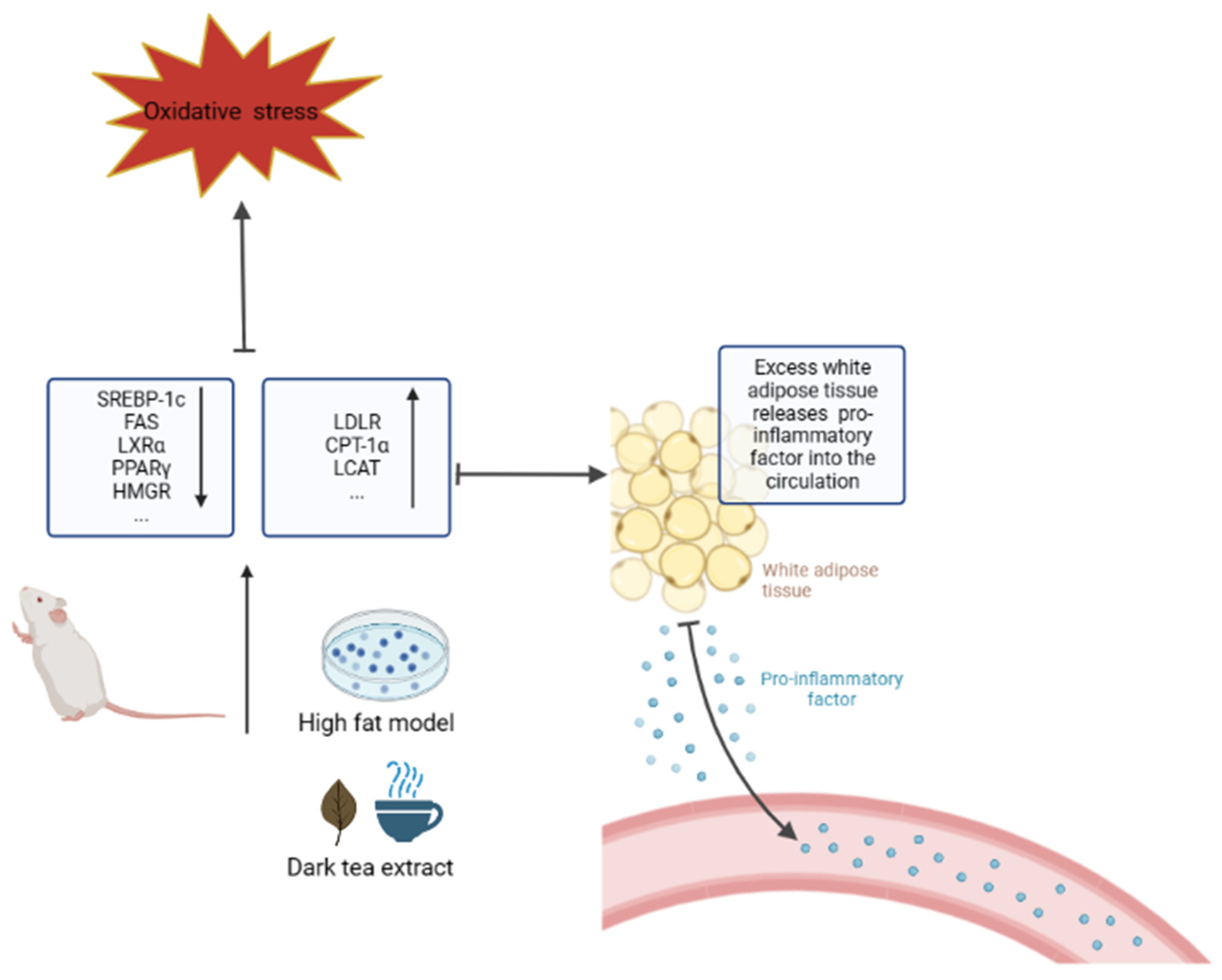
4.5. Anti-Obesity
4.6. Regulating Intestinal Flora
| Mechanisms | Related Genes/Proteins | Reference |
|---|---|---|
| Anti-inflammatory | TLR, HIF-α, NF-κB, MAPK ↓ | [41,42,43,44,45,46,51] |
| Antioxidant | MDA, ROS ↓ SOD, GSH ↑ | [78,81,82,83] |
| Inhibit proliferation | JNK, AMPK ↑ | [84,85,86,87,88] |
| Promote apoptosis | BCL-2 ↓ Caspase-9, BAX, caspase-3 ↑ | [92,93,94,95,96] |
| Inhibit obesity | SREBP, FAS, HMGR, LXRα ↓ LDLR, LCAT, CPT-1α ↑ | [106,110,111,115,117,120] |
| Regulate intestinal flora | Akkermansia muciniphila ↑ Faecalibacterium prausnitzii ↑ Lactobacillus, Actinobacteria ↑ Proteobacteria ↑ | [132,133,134,135,136] |
| Research Method/Model | Active Component | Conclusions | |
|---|---|---|---|
| In vitro study | FRAP, TEAC | Dark tea extract | 1472.27 ± 691.91 µmol Fe2+/g DW 715.99 ± 352.02 µmol Trolox/g DW 81.43 ± 40.92 mg GAE/g DW [63] |
| FRAP, DPPH ABTS, HAS, SSA | Pu’er tea extract (PRT) | As the pertinent antioxidants in PRT, EC, GC, GCG, CG, EGCG, rutin, and kaempferol contributed to the antioxidant activities [64] | |
| DPPH, SARSA, ABTS, ORAC, FRAP | Ripe and raw Pu’er tea | Raw Pu’er tea extract has better antioxidant capacity [65] | |
| ABTS, FRAP | Pu’er tea extract | Tea polysaccharides, tea polyphenols, and proteins are thought to be accountable for the biological activity of Pu’er tea [66] | |
| DPPH, ABTS | Tea polysaccharides | Compared with 12 kinds of tea, the highest total phenolic and protein content as well as the best antioxidant were found in pu’er tea polysaccharide [68] | |
| In vitro study | DPPH | Ya’an Tibetan tea | Tea polyphenols in Ya’an Tibetan tea showed higher antioxidant activity than tea polysaccharides [69] |
| DPPH, T-AOC | Teadenol A | Thealenol A isolated from fermented Pu’er tea has antioxidant properties and is an important bioactive component [70] | |
| ABTS, SOA, DPPH | Fuzhuan dark tea polysaccharides (DTPS) | The content and molecular weight of uronic acid may be the important factors affecting the oxidation resistance of DTPS [71] | |
| ROS, MDA determination | Gamma-aminobutyric acid (GABA) in Pu’er tea | Pu’er tea extract (1, 10 μ g/mL) and GABA (0.1, 1, 10 μM) decreased ROS production and lipid peroxidation in PC12 cells in a dose-dependent manner [73] | |
| ROS determination | Theabrownins (TBs) | At the concentration range of 1.25 to 6.25 mg/mL, Pu’er tea has obvious intracellular ROS clearance ability on human cancer cells (Caco2, HEp2, Hep G2 cell lines), and theabrownins are the main contributor [74] | |
| ROS determination | 2S,3R-6-methoxycarbonylgallocatechin (MCGE) | MCGE protects cells from the production of ROS in UVB-exposed keratinocytes (HACats) by activating the Nrf2 pathway [75] | |
| Cell viability | 8-C N-ethyl-2-pyrrolidinone substituted flavan-3-ols | The 8-C N-ethyl-2-pyrrolidinone substituted flavan-3-ols possessed significant antioxidant activity and could prevent HMEC damage caused by H2O2 [76] | |
| Mice exposed to 7.0 and 7.5 Gy total body irradiation | Dark tea extract | Dark tea extract reduced ROS levels in hematopoietic cells by inhibiting the expression of NOX4 [77] | |
| Obese rat model | Pu’er tea | Pu’er tea increased the activity of antioxidant enzymes such as SOD and GSH-Px, while decreased the level of lipid peroxidation product MDA in obese rats [78,79] | |
| t-BHP oxidative stress-induced rat hepatocyte model | Pu’er tea | Pu’er tea extract decreased the production of ROS marker O2− in rat hepatocytes and prevented t-BHP induced mitochondrial oxidative stress [80] | |
| SD rats Balb/c mice | Pu’er tea | Pu’er tea reduced quinocetone-induced oxidative stress [81,82] | |
| Wistar rats | Pu’er tea | Pu’er tea decreased MDA and GSH, and increased SOD and GSH-Px levels [83] | |
| Mice with acute alcoholic liver injury | Different dark tea extracts | Dark tea has greater in vivo antioxidant activity than green tea [84] |
5. Expectation and Prospect
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
References
- Bray, F.; Laversanne, M.; Weiderpass, E.; Soerjomataram, I. The Ever-Increasing Importance of Cancer as a Leading Cause of Premature Death Worldwide. Cancer 2021, 127, 3029–3030. [Google Scholar] [CrossRef] [PubMed]
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Cancer Statistics 2020: Globocan Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA A Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef] [PubMed]
- You, W.; Henneberg, M. Cancer Incidence Increasing Globally: The Role of Relaxed Natural Selection. Evol. Appl. 2018, 11, 140–152. [Google Scholar] [CrossRef] [PubMed]
- Castro-Espin, C.; Agudo, A. The Role of Diet in Prognosis among Cancer Survivors: A Systematic Review and Meta-Analysis of Dietary Patterns and Diet Interventions. Nutrients 2022, 14, 348. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.-Y.; Zhao, C.-N.; Cao, S.-Y.; Tang, G.-Y.; Gan, R.-Y.; Li, H.-B. Effects and Mechanisms of Tea for the Prevention and Management of Cancers: An Updated Review. Crit. Rev. Food Sci. Nutr. 2020, 60, 1693–1705. [Google Scholar] [CrossRef] [PubMed]
- Çakır, A.; Erbaş, O. The Effect of Coffee and Tea Consumption on Cancer. JEBMS 2022, 3, 1–5. [Google Scholar]
- Cheng, L.; Wang, Y.; Zhang, J.; Xu, L.; Zhou, H.; Wei, K.; Peng, L.; Zhang, J.; Liu, Z.; Wei, X. Integration of Non-Targeted Metabolomics and E-Tongue Evaluation Reveals the Chemical Variation and Taste Characteristics of Five Typical Dark Teas. LWT 2021, 150, 111875. [Google Scholar] [CrossRef]
- Devos, R.J.B.; de Araújo Barth, C.; Dettmer, A.; Bertolin, T.E.; Colla, L.M. Pu-Erh Tea: Fermentative Process as a Potentialized of Sensory Aspects and Bioactive Profile—A Review. Res. Soc. Dev. 2021, 10, e3510816999. [Google Scholar] [CrossRef]
- Lin, F.-J.; Wei, X.-L.; Liu, H.-Y.; Li, H.; Xia, Y.; Wu, D.-T.; Zhang, P.-Z.; Gandhi, G.R.; Li, H.-B.; Gan, R.-Y. State-of-the-Art Review of Dark Tea: From Chemistry to Health Benefits. Trends Food Sci. Technol. 2021, 109, 126–138. [Google Scholar] [CrossRef]
- Fan, X.; Xiao, X.; Mao, X.; Chen, D.; Yu, B.; Wang, J.; Yan, H. Tea Bioactive Components Prevent Carcinogenesis via Anti-Pathogen, Anti-Inflammation, and Cell Survival Pathways. IUBMB Life 2021, 73, 328–340. [Google Scholar] [CrossRef]
- Pan, H.; Le, M.; He, C.; Yang, C.S.; Ling, T. Dark Tea: A Popular Beverage with Possible Medicinal Application. Chin. Herb. Med. 2023, 15, 33–36. [Google Scholar] [CrossRef] [PubMed]
- Sanlier, N.; Atik, I.; Atik, A. A Minireview of Effects of White Tea Consumption on Diseases. Trends Food Sci. Technol. 2018, 82, 82–88. [Google Scholar] [CrossRef]
- Shang, A.; Li, J.; Zhou, D.-D.; Gan, R.-Y.; Li, H.-B. Molecular Mechanisms Underlying Health Benefits of Tea Compounds. Free Radic. Biol. Med. 2021, 172, 181–200. [Google Scholar] [CrossRef]
- Lee, L.K.; Foo, K.Y. Recent Advances on the Beneficial Use and Health Implications of Pu-Erh Tea. Food Res. Int. 2013, 53, 619–628. [Google Scholar] [CrossRef]
- Zhang, L.; Ho, C.-T.; Zhou, J.; Santos, J.S.; Armstrong, L.; Granato, D. Chemistry and Biological Activities of Processed Camellia sinensis Teas: A Comprehensive Review. Compr. Rev. Food. Sci. Food Saf. 2019, 18, 1474–1495. [Google Scholar] [CrossRef]
- Shen, N.; Wang, T.; Gan, Q.; Liu, S.; Wang, L.; Jin, B. Plant Flavonoids: Classification, Distribution, Biosynthesis, and Antioxidant Activity. Food Chem. 2022, 383, 132531. [Google Scholar] [CrossRef]
- Chen, Y.; Cheng, S.; Dai, J.; Wang, L.; Xu, Y.; Peng, X.; Xie, X.; Peng, C. Molecular Mechanisms and Applications of Tea Polyphenols: A Narrative Review. J. Food Biochem. 2021, 45, e13910. [Google Scholar] [CrossRef]
- Liu, W.; Cui, X.; Zhong, Y.; Ma, R.; Liu, B.; Xia, Y. Phenolic Metabolites as Therapeutic in Inflammation and Neoplasms: Molecular Pathways Explaining Their Efficacy. Pharmacol. Res. 2023, 193, 106812. [Google Scholar] [CrossRef]
- Turkozu, D.; Sanlier, N. L-Theanine, Unique Amino Acid of Tea, and Its Metabolism, Health Effects, and Safety. Crit. Rev. Food Sci. Nutr. 2017, 57, 1681–1687. [Google Scholar] [CrossRef] [PubMed]
- Sharma, E.; Joshi, R.; Gulati, A. L-Theanine: An Astounding Sui Generis Integrant in Tea. Food Chem. 2018, 242, 601–610. [Google Scholar] [CrossRef]
- Lin, S.; Chen, Z.; Chen, T.; Deng, W.; Wan, X.; Zhang, Z. Theanine Metabolism and Transport in Tea Plants (Camellia sinensis L.): Advances and Perspectives. Crit. Rev. Biotechnol. 2023, 43, 327–341. [Google Scholar] [CrossRef]
- Lebeau, P.F.; Byun, J.H.; Platko, K.; Saliba, P.; Sguazzin, M.; MacDonald, M.E.; Pare, G.; Steinberg, G.R.; Janssen, L.J.; Igdoura, S.A.; et al. Caffeine Blocks SREBP2-Induced Hepatic PCSK9 Expression to Enhance LDLR-Mediated Cholesterol Clearance. Nat. Commun. 2022, 13, 770. [Google Scholar] [CrossRef] [PubMed]
- Huang, F.; Zheng, X.; Ma, X.; Jiang, R.; Zhou, W.; Zhou, S.; Zhang, Y.; Lei, S.; Wang, S.; Kuang, J.; et al. Theabrownin from Pu-Erh Tea Attenuates Hypercholesterolemia via Modulation of Gut Microbiota and Bile Acid Metabolism. Nat. Commun. 2019, 10, 4971. [Google Scholar] [CrossRef]
- Hou, Y.; Zhang, Z.; Cui, Y.; Peng, C.; Fan, Y.; Tan, C.; Wang, Q.; Liu, Z.; Gong, J. Pu-Erh Tea and Theabrownin Ameliorate Metabolic Syndrome in Mice via Potential Microbiota-Gut-Liver-Brain Interactions. Food Res. Int. 2022, 162, 112176. [Google Scholar] [CrossRef] [PubMed]
- Jieyi, W.; Dan, Z.; Xiaojiao, Z.; Wei, J.I.A.; Aihua, Z. Research Progress in Biological Activities and Mechanisms of Theabrownin. J. Shanghai Jiao Tong Univ. Med. Sci. 2023, 43, 768. [Google Scholar] [CrossRef]
- WeiWei, W.; HeYuan, J.; JianYong, Z.; LuTing, Y.; LiuYan, Y. Advances in extraction and purification technology and physiological efficacy of theabrownine. J. Food Saf. Qual. 2015, 6, 1187–1192. [Google Scholar]
- Liu, M.; Gong, Z.; Liu, H.; Wang, J.; Wang, D.; Yang, Y.; Zhong, S. Structural Characterization and Anti-Tumor Activity in Vitro of a Water-Soluble Polysaccharide from Dark Brick Tea. Int. J. Biol. Macromol. 2022, 205, 615–625. [Google Scholar] [CrossRef] [PubMed]
- Zhou, L.; Wu, F.; Jin, W.; Yan, B.; Chen, X.; He, Y.; Yang, W.; Du, W.; Zhang, Q.; Guo, Y.; et al. Theabrownin Inhibits Cell Cycle Progression and Tumor Growth of Lung Carcinoma through C-Myc-Related Mechanism. Front. Pharmacol. 2017, 8, 75. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Zhong, T.; Yi, P.; Fan, C.; Zhang, Z.; Liang, G.; Xu, Y.; Fan, Y. A New Epigallocatechin Gallate Derivative Isolated from Anhua Dark Tea Sensitizes the Chemosensitivity of Gefitinib via the Suppression of PI3K/MTOR and Epithelial-Mesenchymal Transition. Fitoterapia 2020, 143, 104590. [Google Scholar] [CrossRef] [PubMed]
- He, L.; Yang, Y.; Deng, S.; Yu, G.; Gong, Y.; Yuan, P.; Cao, S.; Zhu, H.; Hao, J.; Wang, D. Ya’an Tibetan Tea Exerts Anti-Tumor and Chemotherapy Sensitizing Effect against Human HepG2 Hepatocellular Carcinoma Cells. Res. Sq. 2022; preprint. [Google Scholar] [CrossRef]
- Li, T.; Yan, B.; Xiao, X.; Zhou, L.; Zhang, J.; Yuan, Q.; Shan, L.; Wu, H.; Efferth, T. Onset of P53/NF-ΚB Signaling Crosstalk in Human Melanoma Cells in Response to Anti-Cancer Theabrownin. FASEB J. 2022, 36, e22426. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Yan, B.; Zhang, L.; Zhou, L.; Zhang, J.; Yu, W.; Dong, X.; Yao, L.; Shan, L. Theabrownin Induces Apoptosis and Tumor Inhibition of Hepatocellular Carcinoma Huh7 Cells Through ASK1-JNK-c-Jun Pathway. OncoTargets Ther. 2020, 13, 8977–8987. [Google Scholar] [CrossRef] [PubMed]
- Chang, Y.-L.; Hsu, Y.-J.; Chen, Y.; Wang, Y.-W.; Huang, S.-M. Theophylline Exhibits Anti-Cancer Activity via Suppressing SRSF3 in Cervical and Breast Cancer Cell Lines. Oncotarget 2017, 8, 101461–101474. [Google Scholar] [CrossRef]
- Zhao, X.; Qian, Y.; Zhou, Y.-L.; Wang, R.; Wang, Q.; Li, G.-J. Pu-Erh Tea Has In Vitro Anticancer Activity in TCA8113 Cells and Preventive Effects on Buccal Mucosa Cancer in U14 Cells Injected Mice In Vivo. Nutr. Cancer 2014, 66, 1059–1069. [Google Scholar] [CrossRef]
- Zheng, K.; Zhao, Q.; Chen, Q.; Xiao, W.; Jiang, Y.; Jiang, Y. The Synergic Inhibitory Effects of Dark Tea (Camellia sinensis) Extract and P38 Inhibition on the Growth of Pancreatic Cancer Cells. J. Cancer 2019, 10, 6557–6569. [Google Scholar] [CrossRef]
- Kundu, J.K.; Surh, Y.-J. Inflammation: Gearing the Journey to Cancer. Mutat. Res.-Rev. Mutat. Res. 2008, 659, 15–30. [Google Scholar] [CrossRef] [PubMed]
- Dharshini, L.C.P.; Rasmi, R.R.; Kathirvelan, C.; Kumar, K.M.; Saradhadevi, K.M.; Sakthivel, K.M. Regulatory Components of Oxidative Stress and Inflammation and Their Complex Interplay in Carcinogenesis. Appl. Biochem. Biotechnol. 2023, 195, 2893–2916. [Google Scholar] [CrossRef] [PubMed]
- Tan, Y.-F.; Wang, M.; Chen, Z.-Y.; Wang, L.; Hu, X.-H. Inhibition of BRD4 Prevents Proliferation and Epithelial-Mesenchymal Transition in Renal Cell Carcinoma via NLRP3 Inflammasome-Induced Pyroptosis. Cell Death Dis. 2020, 11, 239. [Google Scholar] [CrossRef] [PubMed]
- Mussbacher, M.; Derler, M.; Basílio, J.; Schmid, J.A. NF-ΚB in Monocytes and Macrophages—An Inflammatory Master Regulator in Multitalented Immune Cells. Front. Immunol. 2023, 14, 1134661. [Google Scholar] [CrossRef] [PubMed]
- Wang, K.; Sun, J.-Z.; Wu, Q.-X.; Li, Z.-Y.; Li, D.-X.; Xiong, Y.-F.; Zhong, G.-C.; Shi, Y.; Li, Q.; Zheng, J.; et al. Long-Term Anti-Inflammatory Diet in Relation to Improved Breast Cancer Prognosis: A Prospective Cohort Study. NPJ Breast Cancer 2020, 6, 36. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Xing, K.; Qiu, L.; Wu, Q.; Wei, H. Therapeutic Implications of Functional Tea Ingredients for Ameliorating Inflammatory Bowel Disease: A Focused Review. Crit. Rev. Food Sci. Nutr. 2022, 62, 5307–5321. [Google Scholar] [CrossRef] [PubMed]
- Crusz, S.M.; Balkwill, F.R. Inflammation and Cancer: Advances and New Agents. Nat. Rev. Clin. Oncol. 2015, 12, 584–596. [Google Scholar] [CrossRef] [PubMed]
- Wu, Z.; Sun, L.; Chen, R.; Wen, S.; Li, Q.; Lai, X.; Zhang, Z.; Cao, F.; Sun, S. Chinese Tea Alleviates CCl4-Induced Liver Injury through the NF-ΚBorNrf2Signaling Pathway in C57BL-6J Mice. Nutrients 2022, 14, 972. [Google Scholar] [CrossRef]
- Zhao, L.; Miao, Y.; Shan, B.; Zhao, C.; Peng, C.; Gong, J. Theabrownin Isolated from Pu-Erh Tea Enhances the Innate Immune and Anti-Inflammatory Effects of RAW264.7 Macrophages via the TLR2/4-Mediated Signaling Pathway. Foods 2023, 12, 1468. [Google Scholar] [CrossRef]
- Liu, H.; Chen, R.; Wen, S.; Li, Q.; Lai, X.; Zhang, Z.; Sun, L.; Sun, S.; Cao, F. Tea (Camellia sinensis) Ameliorates DSS-Induced Colitis and Liver Injury by Inhibiting TLR4/NF-ΚB/NLRP3 Inflammasome in Mice. Biomed. Pharmacother. 2023, 158, 114136. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Qiu, L.; Mi, X.; Zhang, Z.; Xu, D.; Tao, X.; Xing, K.; Wu, Q.; Wei, H. Hot-Water Extract of Ripened Pu-Erh Tea Attenuates DSS-Induced Colitis through Modulation of the NF-ΚB and HIF-1α Signaling Pathways in Mice. Food Funct. 2020, 11, 3459–3470. [Google Scholar] [CrossRef] [PubMed]
- Deok, J.Y.; Kim, S.-J. Ameliorative Effect of Pu-Erh Tea on DSS-Induced Colitis through Regulation of NF-ΚB Activation in Mice. J. Biol. Sci. Korea. 2021, 27, 248–254. [Google Scholar] [CrossRef]
- Hu, S.; Li, S.; Liu, Y.; Sun, K.; Luo, L.; Zeng, L. Aged Ripe Pu-Erh Tea Reduced Oxidative Stress-Mediated Inflammation in Dextran Sulfate Sodium-Induced Colitis Mice by Regulating Intestinal Microbes. J. Agric. Food Chem. 2021, 69, 10592–10605. [Google Scholar] [CrossRef] [PubMed]
- Liu, B.; Yang, T.; Zeng, L.; Shi, L.; Li, Y.; Xia, Z.; Xia, X.; Lin, Q.; Luo, F. Crude Extract of Fuzhuan Brick Tea Ameliorates DSS-Induced Colitis in Mice. Int. J. Food Sci. Technol. 2016, 51, 2574–2582. [Google Scholar] [CrossRef]
- Zeng, Z.; Xie, Z.; Chen, G.; Sun, Y.; Zeng, X.; Liu, Z. Anti-Inflammatory and Gut Microbiota Modulatory Effects of Polysaccharides from Fuzhuan Brick Tea on Colitis in Mice Induced by Dextran Sulfate Sodium. Food Funct. 2022, 13, 649–663. [Google Scholar] [CrossRef] [PubMed]
- Dai, X.; Ge, B.; Zhu, M.; Wang, H.; Zeng, T.; Liu, Z.; Fu, D.; Dai, X.; Ge, B.; Zhu, M.; et al. Anti-Inflammatory Properties of Fu Brick Tea Water Extract Contribute to the Improvement of Diarrhea in Mice. Beverage Plant Res. 2022, 2, 3. [Google Scholar] [CrossRef]
- Hu, S.; Lin, Z.; Zhao, S.; Zhang, B.; Luo, L.; Zeng, L. Pu-Erh Tea Alleviated Colitis-Mediated Brain Dysfunction by Promoting Butyric Acid Production. Food Chem. Toxicol. 2023, 172, 113594. [Google Scholar] [CrossRef]
- Liu, C.; Guo, Y.; Sun, L.; Lai, X.; Li, Q.; Zhang, W.; Xiang, L.; Sun, S.; Cao, F. Six Types of Tea Reduce High-Fat-Diet-Induced Fat Accumulation in Mice by Increasing Lipid Metabolism and Suppressing Inflammation. Food Funct. 2019, 10, 2061–2074. [Google Scholar] [CrossRef]
- Xiao, Y.; He, M.; Liang, X.; She, J.; He, L.; Liu, Y.; Zou, J.; Yuan, Z. Pu-Erh Tea Ameliorates Atherosclerosis Associated with Promoting Macrophage Apoptosis by Reducing NF-ΚB Activation in ApoE Knockout Mice. Oxid. Med. Cell Longev. 2018, 2018, 3197829. [Google Scholar] [CrossRef]
- Zhu, J.; Yu, C.; Zhou, H.; Wei, X.; Wang, Y. Comparative Evaluation for Phytochemical Composition and Regulation of Blood Glucose, Hepatic Oxidative Stress and Insulin Resistance in Mice and HepG2 Models of Four Typical Chinese Dark Teas. J. Sci. Food Agric. 2021, 101, 6563–6577. [Google Scholar] [CrossRef] [PubMed]
- Cai, X.; Fang, C.; Hayashi, S.; Hao, S.; Zhao, M.; Tsutsui, H.; Nishiguchi, S.; Sheng, J. Pu-Erh Tea Extract Ameliorates High-Fat Diet-Induced Nonalcoholic Steatohepatitis and Insulin Resistance by Modulating Hepatic IL-6/STAT3 Signaling in Mice. J. Gastroenterol. 2016, 51, 819–829. [Google Scholar] [CrossRef] [PubMed]
- Rahman, S.U.; Huang, Y.; Zhu, L.; Chu, X.; Junejo, S.A.; Zhang, Y.; Khan, I.M.; Li, Y.; Feng, S.; Wu, J.; et al. Tea Polyphenols Attenuate Liver Inflammation by Modulating Obesity-Related Genes and down-Regulating COX-2 and INOS Expression in High Fat-Fed Dogs. BMC Vet. Res. 2020, 16, 234. [Google Scholar] [CrossRef] [PubMed]
- Jumat, N.R.; Chai, P.S.; Loh, C.Y.; Vidyadaran, S.; Seman, Z. Immunomodulatory Effect of Papaya, Mata Kucing, Dang Shen and Pu-Erh Tea on Cytokines Profile. Malays. J. Med. Health Sci. 2019, 15, 21. [Google Scholar]
- Hayes, J.D.; Dinkova-Kostova, A.T.; Tew, K.D. Oxidative Stress in Cancer. Cancer Cell 2020, 38, 167–197. [Google Scholar] [CrossRef]
- Yang, C.-Y.; Hung, K.-C.; Yen, Y.-Y.; Liao, H.-E.; Lan, S.-J.; Lin, H.-C. Anti-Oxidative Effect of Pu-Erh Tea in Animals Trails: A Systematic Review and Meta-Analysis. Foods 2022, 11, 1333. [Google Scholar] [CrossRef]
- Yang, C.S.; Wang, X.; Lu, G.; Picinich, S.C. Cancer Prevention by Tea: Animal Studies, Molecular Mechanisms and Human Relevance. Nat. Rev. Cancer 2009, 9, 429–439. [Google Scholar] [CrossRef]
- Rha, C.-S.; Jung, Y.S.; Lee, J.-D.; Jang, D.; Kim, M.-S.; Lee, M.-S.; Hong, Y.D.; Kim, D.-O. Chemometric Analysis of Extracts and Fractions from Green, Oxidized, and Microbial Fermented Teas and Their Correlation to Potential Antioxidant and Anticancer Effects. Antioxidants 2020, 9, 1015. [Google Scholar] [CrossRef]
- Zhao, C.-N.; Tang, G.-Y.; Cao, S.-Y.; Xu, X.-Y.; Gan, R.-Y.; Liu, Q.; Mao, Q.-Q.; Shang, A.; Li, H.-B. Phenolic Profiles and Antioxidant Activities of 30 Tea Infusions from Green, Black, Oolong, White, Yellow and Dark Teas. Antioxidants 2019, 8, 215. [Google Scholar] [CrossRef]
- Zhou, B.; Ma, B.; Ma, C.; Xu, C.; Wang, J.; Wang, Z.; Yin, D.; Xia, T. Classification of Pu-Erh Ripened Teas and Their Differences in Chemical Constituents and Antioxidant Capacity. LWT-Food Sci. Technol. 2022, 153, 112370. [Google Scholar] [CrossRef]
- Roda, G.; Marinello, C.; Grassi, A.; Picozzi, C.; Aldini, G.; Carini, M.; Regazzoni, L. Ripe and Raw Pu-Erh Tea: LC-MS Profiling, Antioxidant Capacity and Enzyme Inhibition Activities of Aqueous and Hydro-Alcoholic Extracts. Molecules 2019, 24, 473. [Google Scholar] [CrossRef]
- Huang, Q.; Chen, S.; Chen, H.; Wang, Y.; Wang, Y.; Hochstetter, D.; Xu, P. Studies on the Bioactivity of Aqueous Extract of Pu-Erh Tea and Its Fractions: In Vitro Antioxidant Activity and α-Glycosidase Inhibitory Property, and Their Effect on Postprandial Hyperglycemia in Diabetic Mice. Food Chem. Toxicol. 2013, 53, 75–83. [Google Scholar] [CrossRef]
- Zhang, H.M.; Wang, C.F.; Shen, S.M.; Wang, G.L.; Liu, P.; Liu, Z.M.; Wang, Y.Y.; Du, S.S.; Liu, Z.L.; Deng, Z.W. Antioxidant Phenolic Compounds from Pu-Erh Tea. Molecules 2012, 17, 14037–14045. [Google Scholar] [CrossRef]
- Guo, H.; Fu, M.-X.; Wu, D.-T.; Zhao, Y.-X.; Li, H.; Li, H.-B.; Gan, R.-Y. Structural Characteristics of Crude Polysaccharides from 12 Selected Chinese Teas, and Their Antioxidant and Anti-Diabetic Activities. Antioxidants 2021, 10, 1562. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Q.; Li, W.; Zhang, H.; Gao, X.; Tan, S. Optimizing Synchronous Extraction and Antioxidant Activity Evaluation of Polyphenols and Polysaccharides from Ya’an Tibetan Tea (Camellia sinensis). Food Sci. Nutr. 2020, 8, 489–499. [Google Scholar] [CrossRef]
- Su, X.; Zhang, G.; Ma, Y.; Chen, M.; Chen, S.; Duan, S.; Wan, J.; Hashimoto, F.; Lv, H.; Li, J.; et al. Isolation, Identification, and Biotransformation of Teadenol A from Solid State Fermentation of Pu-Erh Tea and In Vitro Antioxidant Activity. Appl. Sci.-Basel 2016, 6, 161. [Google Scholar] [CrossRef]
- Zhu, J.; Zhou, H.; Zhang, J.; Li, F.; Wei, K.; Wei, X.; Wang, Y. Valorization of Polysaccharides Obtained from Dark Tea: Preparation, Physicochemical, Antioxidant, and Hypoglycemic Properties. Foods 2021, 10, 2276. [Google Scholar] [CrossRef]
- Yang, X.; Huang, M.; Qin, C.; Lv, B.; Mao, Q.; Liu, Z. Structural Characterization and Evaluation of the Antioxidant Activities of Polysaccharides Extracted from Qingzhuan Brick Tea. Int. J. Biol. Macromol. 2017, 101, 768–775. [Google Scholar] [CrossRef] [PubMed]
- Hou, C.-W. Pu-Erh Tea and GABA Attenuates Oxidative Stress in Kainic Acid-Induced Status Epilepticus. J. Biomed. Sci. 2011, 18, 75. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Belscak-Cvitanovic, A.; Durgo, K.; Chisti, Y.; Gong, J.; Sirisansaneeyakul, S.; Komes, D. Physicochemical Properties and Biological Activities of a High-Theabrownins Instant Pu-Erh Tea Produced Using Aspergillus Tubingensis. LWT-Food Sci. Technol. 2018, 90, 598–605. [Google Scholar] [CrossRef]
- Kim, D.; Hu, R.; Fan, Y.; Xu, Y.-N.; Park, H.J.; Lee, S.K. Photoprotective Effects of 2S,3R-6-Methoxycarbonylgallocatechin Isolated from Anhua Dark Tea on UVB-Induced Inflammatory Responses in Human Keratinocytes. J. Photochem. Photobiol. B Biol. 2020, 202, 111704. [Google Scholar] [CrossRef]
- Wang, W.; Zhang, L.; Wang, S.; Shi, S.; Jiang, Y.; Li, N.; Tu, P. 8-C N-Ethyl-2-Pyrrolidinone Substituted Flavan-3-Ols as the Marker Compounds of Chinese Dark Teas Formed in the Post-Fermentation Process Provide Significant Antioxidative Activity. Food Chem. 2014, 152, 539–545. [Google Scholar] [CrossRef]
- Long, W.; Zhang, G.; Dong, Y.; Li, D. Dark Tea Extract Mitigates Hematopoietic Radiation Injury with Antioxidative Activity. J. Radiat. Res. 2018, 59, 387–394. [Google Scholar] [CrossRef]
- Xiong, C.; Peng, Y.; Liu, B.; Cui, W.; Liu, X. Anti-Obesity, Anti-Atherosclerotic and Anti-Oxidant Effects of Pu-Erh Tea on a High Fat Diet-Induced Obese Rat Model. J. Biosci. Med. 2019, 7, 120. [Google Scholar] [CrossRef]
- Su, J.; Wang, X.; Song, W.; Bai, X.; Li, C. Reducing Oxidative Stress and Hepatoprotective Effect of Water Extracts from Pu-Erh Tea on Rats with High-Fat Diet. Food Sci. Hum. Wellness 2016, 5, 199–206. [Google Scholar] [CrossRef]
- Braud, L.; Peyre, L.; de Sousa, G.; Armand, M.; Rahmani, R.; Maixent, J.-M. Effect of Brewing Duration on the Antioxidant and Hepatoprotective Abilities of Tea Phenolic and Alkaloid Compounds in a T-BHP Oxidative Stress-Induced Rat Hepatocyte Model. Molecules 2015, 20, 14985–15002. [Google Scholar] [CrossRef]
- Yu, M.; Wang, D.; Yang, W.; Xu, M.; Liu, Y.; Xu, S.; Yao, P.; Yan, H.; Liu, L. Mechanisms of Nrf2/HO-1 Pathway up-Regulation Induced by Pu-Erh Black Tea Extract Supplementation for Quinocetone-Treated Sprague-Dawley Rats. J. Funct. Food. 2015, 14, 767–778. [Google Scholar] [CrossRef]
- Wang, D.; Zhong, Y.; Luo, X.; Wu, S.; Xiao, R.; Bao, W.; Yang, W.; Yan, H.; Yao, P.; Liu, L. Pu-Erh Black Tea Supplementation Decreases Quinocetone-Induced ROS Generation and Oxidative DNA Damage in Balb/c Mice. Food Chem. Toxicol. 2011, 49, 477–484. [Google Scholar] [CrossRef]
- Zheng, X.-N.; Wang, X.-W.; Li, L.-Y.; Xu, Z.-W.; Huang, H.-Y.; Zhao, J.-S.; Zhang, D.; Yin, X.; Sheng, J.; Tang, J.-T. Pu-Erh Tea Powder Preventive Effects on Cisplatin-Induced Liver Oxidative Damage in Wistar Rats. Asian Pac. J. Cancer Prev. 2014, 15, 7389–7394. [Google Scholar] [CrossRef]
- Cao, S.-Y.; Li, B.-Y.; Gan, R.-Y.; Mao, Q.-Q.; Wang, Y.-F.; Shang, A.; Meng, J.-M.; Xu, X.-Y.; Wei, X.-L.; Li, H.-B. The In Vivo Antioxidant and Hepatoprotective Actions of Selected Chinese Teas. Foods 2020, 9, 262. [Google Scholar] [CrossRef] [PubMed]
- Xie, J.; Yu, H.; Song, S.; Fang, C.; Wang, X.; Bai, Z.; Ma, X.; Hao, S.; Zhao, H.-Y.; Sheng, J. Pu-Erh Tea Water Extract Mediates Cell Cycle Arrest and Apoptosis in MDA-MB-231 Human Breast Cancer Cells. Front. Pharmacol. 2017, 8, 190. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Jia, S.; Tang, W.; Sheng, J.; Luo, Y. Pu-Erh Tea Inhibits Tumor Cell Growth by Down-Regulating Mutant P53. Int. J. Mol. Sci. 2011, 12, 7581–7593. [Google Scholar] [CrossRef] [PubMed]
- Way, T.-D.; Lin, H.-Y.; Kuo, D.-H.; Tsai, S.-J.; Shieh, J.-C.; Wu, J.-C.; Lee, M.-R.; Lin, J.-K. Pu-Erh Tea Attenuates Hyperlipogenesis and Induces Hepatoma Cells Growth Arrest through Activating AMP-Activated Protein Kinase (AMPK) in Human HepG2 Cells. J. Agric. Food Chem. 2009, 57, 5257–5264. [Google Scholar] [CrossRef] [PubMed]
- Yoshimura, H.; Yoshida, H.; Matsuda, S.; Ryoke, T.; Ohta, K.; Ohmori, M.; Yamamoto, S.; Kiyoshima, T.; Kobayashi, M.; Sano, K. The Therapeutic Potential of Epigallocatechin-3-gallate against Human Oral Squamous Cell Carcinoma through Inhibition of Cell Proliferation and Induction of Apoptosis: In Vitro and in Vivo Murine Xenograft Study. Mol. Med. Rep. 2019, 20, 1139–1148. [Google Scholar] [CrossRef] [PubMed]
- Zhao, H.; Zhang, M.; Zhao, L.; Ge, Y.; Sheng, J.; Shi, W. Changes of Constituents and Activity to Apoptosis and Cell Cycle During Fermentation of Tea. Int. J. Mol. Sci. 2011, 12, 1862–1875. [Google Scholar] [CrossRef]
- Chen, X.; Hu, Y.; Wang, B.; Chen, Y.; Yuan, Y.; Zhou, W.; Song, W.; Wu, Z.; Li, X. Characterization of Theabrownins Prepared From Tea Polyphenols by Enzymatic and Chemical Oxidation and Their Inhibitory Effect on Colon Cancer Cells. Front. Nutr. 2022, 9, 849728. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.-C.; Chen, M.-Y.; Tu, S.-H.; Pan, M.-H.; Lo, C.-Y.; Ho, C.-T.; Wu, C.-H.; Ho, Y.-S. Pu-Erh Tea Attenuates Smoking-Induced Foam Cell Formation through Inhibition of the A9-Nicotinic-Acetylcholine Receptor Expression in Monocytes: An Ex Vivo Study. J. Funct. Foods 2016, 22, 132–144. [Google Scholar] [CrossRef]
- Georgiev, K.; Iliev, I.; Jelev, I. Evaluation of antitumor effect of methylxanthine fraction isolated from pu-erh tea. World J. Pharm. Res. 2015, 4, 2236–2242. [Google Scholar]
- Zhao, X.; Song, J.-L.; Kim, J.-D.; Lee, J.-S.; Park, K.Y. Fermented Pu-Erh Tea Increases In Vitro Anticancer Activities in HT-29 Cells and Has Antiangiogenetic Effects on HUVECs. J. Environ. Pathol. Toxicol. Oncol. 2013, 32, 275–288. [Google Scholar] [CrossRef]
- Yuan, Y.; Bai, Y.; Zhang, Y.; Wan, H.; Hu, Y.; Wu, Z.; Li, X.; Song, W.; Chen, X. Physicochemical and Colon Cancer Cell Inhibitory Properties of Theabrownins Prepared by Weak Alkali Oxidation of Tea Polyphenols. Plant Food Hum. Nutr. 2022, 77, 405–411. [Google Scholar] [CrossRef] [PubMed]
- Leung, H.K.M.; Lo, E.K.K.; El-Nezami, H. Theabrownin Alleviates Colorectal Tumorigenesis in Murine AOM/DSS Model via PI3K/Akt/MTOR Pathway Suppression and Gut Microbiota Modulation. Antioxidants 2022, 11, 1716. [Google Scholar] [CrossRef] [PubMed]
- Xiao, X.; Guo, L.; Dai, W.; Yan, B.; Zhang, J.; Yuan, Q.; Zhou, L.; Shan, L.; Efferth, T. Green Tea-Derived Theabrownin Suppresses Human Non-Small Cell Lung Carcinoma in Xenograft Model through Activation of Not Only P53 Signaling but Also MAPK/JNK Signaling Pathway. J. Ethnopharmacol. 2022, 291, 115167. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Sang, S.; Xu, H.; Piao, L.; Liu, X. Lovastatin Suppresses Bacterial Therapy-Induced Neutrophil Recruitment to the Tumor by Promoting Neutrophil Apoptosis. J. Funct. Food. 2021, 86, 104693. [Google Scholar] [CrossRef]
- Kopelman, P.G. Obesity as a Medical Problem. Nature 2000, 404, 635–643. [Google Scholar] [CrossRef]
- Malik, V.S.; Willet, W.C.; Hu, F.B. Nearly a Decade on—Trends, Risk Factors and Policy Implications in Global Obesity. Nat. Rev. Endocrinol. 2020, 16, 615–616. [Google Scholar] [CrossRef] [PubMed]
- Bian, X.; Liu, R.; Meng, Y.; Xing, D.; Xu, D.; Lu, Z. Cancer Focus Lipid Metabolism and Cancer. J. Exp. Med. 2021, 218, e20201606. [Google Scholar] [CrossRef] [PubMed]
- Lathigara, D.; Kaushal, D.; Wilson, R.B. Molecular Mechanisms of Western Diet-Induced Obesity and Obesity-Related Carcinogenesis-A Narrative Review. Metabolites 2023, 13, 675. [Google Scholar] [CrossRef] [PubMed]
- Schauer, D.P.; Feigelson, H.S.; Koebnick, C.; Caan, B.; Weinmann, S.; Leonard, A.C.; Powers, J.D.; Yenumula, P.R.; Arterburn, D.E. Bariatric Surgery and the Risk of Cancer in a Large Multisite Cohort. Ann. Surg. 2019, 269, 95–101. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.-Y.; Zhao, C.-N.; Li, B.-Y.; Tang, G.-Y.; Shang, A.; Gan, R.-Y.; Feng, Y.-B.; Li, H.-B. Effects and Mechanisms of Tea on Obesity. Crit. Rev. Food Sci. Nutr. 2021, 63, 3716–3733. [Google Scholar] [CrossRef] [PubMed]
- Currie, E.; Schulze, A.; Zechner, R.; Walther, T.C.; Farese, R.V. Cellular Fatty Acid Metabolism and Cancer. Cell Metab. 2013, 18, 153–161. [Google Scholar] [CrossRef]
- Hahn, A.W.; Menk, A.V.; Rivadeneira, D.B.; Augustin, R.C.; Xu, M.; Li, J.; Wu, X.; Mishra, A.K.; Gide, T.N.; Quek, C.; et al. Obesity Is Associated with Altered Tumor Metabolism in Metastatic Melanoma. Clin. Cancer Res. 2023, 29, 154–164. [Google Scholar] [CrossRef]
- Li, B.; Mao, Q.; Zhou, D.; Luo, M.; Gan, R.; Li, H.; Huang, S.; Saimaiti, A.; Shang, A.; Li, H. Effects of Tea against Alcoholic Fatty Liver Disease by Modulating Gut Microbiota in Chronic Alcohol-Exposed Mice. Foods 2021, 10, 1232. [Google Scholar] [CrossRef] [PubMed]
- XiangTing, X.; Peng, W.; ShaoZhong, L.; Yan, H.; JiHong, Y.; WanFang, S. Effects of Pu-erh tea on hyperlipidemia, antioxidation and vascular endothelium protection in SD rats. J. Tea Sci. 2010, 30, 470–474. [Google Scholar]
- Xiao, Y.; Li, M.; Wu, Y.; Zhong, K.; Gao, H. Structural Characteristics and Hypolipidemic Activity of Theabrownins from Dark Tea Fermented by Single Species Eurotium Cristatum PW-1. Biomolecules 2020, 10, 204. [Google Scholar] [CrossRef]
- Ma, W.; Shi, Y.; Yang, G.; Shi, J.; Ji, J.; Zhang, Y.; Wang, J.; Peng, Q.; Lin, Z.; Lv, H. Hypolipidaemic and Antioxidant Effects of Various Chinese Dark Tea Extracts Obtained from the Same Raw Material and Their Main Chemical Components. Food Chem. 2022, 375, 131877. [Google Scholar] [CrossRef] [PubMed]
- Sun, L.; Cai, J.; Gonzalez, F.J. The Role of Farnesoid X Receptor in Metabolic Diseases, and Gastrointestinal and Liver Cancer. Nat. Rev. Gastroenterol. Hepatol. 2021, 18, 335–347. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Sun, H.; Yi, R.; Tan, F.; Zhao, X. Anti-Obesity Effect of Liupao Tea Extract by Modulating Lipid Metabolism and Oxidative Stress in High-Fat-Diet-Induced Obese Mice. J. Food Sci. 2021, 86, 215–227. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Wang, W.; Liang, X.; Li, P.; Zou, Y.; Du, X. Inhibitory Effect of the Theabrownin and Tea Polysaccharide Extracts of Dark Tea on Lipase. J. Phys. Conf. Ser. 2020, 1549, 032048. [Google Scholar] [CrossRef]
- Wang, Y.; Zhao, A.; Du, H.; Liu, Y.; Qi, B.; Yang, X. Theabrownin from Fu Brick Tea Exhibits the Thermogenic Function of Adipocytes in High-Fat-Diet-Induced Obesity. J. Agric. Food Chem. 2021, 69, 11900–11911. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.; Ge, B.; Zhang, X.; Wang, K.; Zhou, C.; Fu, D. Metabolomics Analysis Reveals the Effects of Compound Fuzhuan Brick Tea (CFBT) on Regulating Dyslipidemia and Metabolic Disorders in Mice Induced by High-Fat Diet. Nutrients 2022, 14, 1128. [Google Scholar] [CrossRef] [PubMed]
- Yuan, Y.; He, J.; Tang, M.; Chen, H.; Wei, T.; Zhang, B.; Liang, D.; Nie, X. Preventive Effect of Ya’an Tibetan Tea on Obesity in Rats Fed with a Hypercaloric High-Fat Diet Revealed by Gut Microbiology and Metabolomics Studies. Food Res. Int. 2023, 165, 112520. [Google Scholar] [CrossRef] [PubMed]
- Lv, H.-P.; Zhu, Y.; Tan, J.-F.; Guo, L.; Dai, W.-D.; Lin, Z. Bioactive Compounds from Pu-Erh Tea with Therapy for Hyperlipidaemia. J. Funct. Foods 2015, 19, 194–203. [Google Scholar] [CrossRef]
- Lu, C.-H.; Hwang, L.S. Polyphenol Contents of Pu-Erh Teas and Their Abilities to Inhibit Cholesterol Biosynthesis in Hep G2 Cell Line. Food Chem. 2008, 111, 67–71. [Google Scholar] [CrossRef]
- Sun, Y.; Wang, Y.; Song, P.; Wang, H.; Xu, N.; Wang, Y.; Zhang, Z.; Yue, P.; Gao, X. Anti-Obesity Effects of Instant Fermented Teas in Vitro and in Mice with High-Fat-Diet-Induced Obesity. Food Funct. 2019, 10, 3502–3513. [Google Scholar] [CrossRef]
- Ye, J.; Zhao, Y.; Chen, X.; Zhou, H.; Yang, Y.; Zhang, X.; Huang, Y.; Zhang, N.; Lui, E.M.K.; Xiao, M. Pu-Erh Tea Ameliorates Obesity and Modulates Gut Microbiota in High Fat Diet Fed Mice. Food Res. Int. 2021, 144, 110360. [Google Scholar] [CrossRef]
- Zeng, L.; Yan, J.; Luo, L.; Zhang, D. Effects of Pu-Erh Tea Aqueous Extract (PTAE) on Blood Lipid Metabolism Enzymes. Food Funct. 2015, 6, 2008–2016. [Google Scholar] [CrossRef]
- Huang, H.-C.; Lin, J.-K. Pu-Erh Tea, Green Tea, and Black Tea Suppresses Hyperlipidemia, Hyperleptinemia and Fatty Acid Synthase through Activating AMPK in Rats Fed a High- Fructose Diet. Food Funct. 2012, 3, 170–177. [Google Scholar] [CrossRef] [PubMed]
- Zhou, S.; Chen, J.; Yang, S.; Pan, Y.; Feng, X.; Ma, S.; Fan, F.; Gong, S.; Wang, Y.; Chen, P.; et al. Pu’er Raw Tea Extract Alleviates Lipid Deposition in Both LO2 Cells and Caenorhabditis Elegans. Food Biosci. 2022, 50, 102172. [Google Scholar] [CrossRef]
- Cao, Z.-H.; Yang, H.; He, Z.-L.; Luo, C.; Xu, Z.-Q.; Gu, D.-H.; Jia, J.-J.; Ge, C.-R.; Lin, Q.-Y. Effects of Aqueous Extracts of Raw Pu-Erh Tea and Ripened Pu-Erh Tea on Proliferation and Differentiation of 3T3-L1 Preadipocytes. Phytother. Res. 2013, 27, 1193–1199. [Google Scholar] [CrossRef] [PubMed]
- Kuang, J.; Zheng, X.; Huang, F.; Wang, S.; Li, M.; Zhao, M.; Sang, C.; Ge, K.; Li, Y.; Li, J.; et al. Anti-Adipogenic Effect of Theabrownin Is Mediated by Bile Acid Alternative Synthesis via Gut Microbiota Remodeling. Metabolites 2020, 10, 475. [Google Scholar] [CrossRef]
- Jensen, G.S.; Beaman, J.L.; He, Y.; Guo, Z.; Sun, H. Reduction of Body Fat and Improved Lipid Profile Associated with Daily Consumption of a Puer Tea Extract in a Hyperlipidemic Population: A Randomized Placebo-Controlled Trial. Clin. Interv. Aging 2016, 11, 367–376. [Google Scholar] [CrossRef]
- Takeda, R.; Furuno, Y.; Imai, S.; Ide, Y.; Wu, D.; Yan, K. Effect of Powdered Beverages Containing Pu-Erh Tea Extract on Postprandial Blood Glucose Levels. Funct. Foods Health Dis. 2019, 9, 532. [Google Scholar] [CrossRef]
- Chen, Y.; Li, W.; Qiu, S.; Vladmir, C.; Xu, X.; Wang, X.; Nian, X.; Chen, Q.; Wang, Q.; Tu, P.; et al. Tea Consumption and Risk of Diabetes in the Chinese Population: A Multi-Centre, Cross-Sectional Study. Br. J. Nutr. 2020, 123, 428–436. [Google Scholar] [CrossRef]
- Zitvogel, L.; Galluzzi, L.; Viaud, S.; Vétizou, M.; Daillère, R.; Merad, M.; Kroemer, G. Cancer and the Gut Microbiota: An Unexpected Link. Sci. Transl. Med. 2015, 7, 271ps1. [Google Scholar] [CrossRef]
- Vivarelli, S.; Salemi, R.; Candido, S.; Falzone, L.; Santagati, M.; Stefani, S.; Torino, F.; Banna, G.L.; Tonini, G.; Libra, M. Gut Microbiota and Cancer: From Pathogenesis to Therapy. Cancers 2019, 11, 38. [Google Scholar] [CrossRef]
- Ma, H.; Hu, Y.; Zhang, B.; Shao, Z.; Roura, E.; Wang, S. Tea Polyphenol—Gut Microbiota Interactions: Hints on Improving the Metabolic Syndrome in a Multi-Element and Multi-Target Manner. Food Sci. Hum. Wellness 2022, 11, 11–21. [Google Scholar] [CrossRef]
- Bond, T.; Derbyshire, E. Tea Compounds and the Gut Microbiome: Findings from Trials and Mechanistic Studies. Nutrients 2019, 11, 2364. [Google Scholar] [CrossRef]
- Khairudin, M.A.S.; Mhd Jalil, A.M.; Hussin, N. Effects of Polyphenols in Tea (Camellia sinensis Sp.) on the Modulation of Gut Microbiota in Human Trials and Animal Studies. Gastroenterol. Insights 2021, 12, 18. [Google Scholar] [CrossRef]
- Gao, X.; Xie, Q.; Kong, P.; Liu, L.; Sun, S.; Xiong, B.; Huang, B.; Yan, L.; Sheng, J.; Xiang, H. Polyphenol- and Caffeine-Rich Postfermented Pu-Erh Tea Improves Diet-Induced Metabolic Syndrome by Remodeling Intestinal Homeostasis in Mice. Infect. Immun. 2017, 86, e00601-17. [Google Scholar] [CrossRef] [PubMed]
- Deng, X.; Zhang, N.; Wang, Q.; Huang, Y.; Huang, Y.; Lin, Y.; Huang, M.; Zheng, F.; Xiao, M.; Ye, J. Theabrownin of Raw and Ripened Pu-Erh Tea Varies in the Alleviation of HFD-Induced Obesity via the Regulation of Gut Microbiota. Eur. J. Nutr. 2023, 62, 2177–2194. [Google Scholar] [CrossRef] [PubMed]
- Yue, S.; Shan, B.; Peng, C.; Tan, C.; Wang, Q.; Gong, J. Theabrownin-Targeted Regulation of Intestinal Microorganisms to Improve Glucose and Lipid Metabolism in Goto-Kakizaki Rats. Food Funct. 2022, 13, 1921–1940. [Google Scholar] [CrossRef]
- Yue, S.; Peng, C.; Zhao, D.; Xia, X.; Tan, C.; Wang, Q.; Gong, J. Theabrownin Isolated from Pu-Erh Tea Regulates Bacteroidetes to Improve Metabolic Syndrome of Rats Induced by High-Fat, High-Sugar and High-Salt Diet. J. Sci. Food Agric. 2022, 102, 4250–4265. [Google Scholar] [CrossRef]
- Zhou, S.; Yang, J.; Pan, Y.; Feng, X.; Hu, H.; Ma, S.; Ou, C.; Fan, F.; Gong, S.; Wang, Y.; et al. Pu’ Er Raw Tea Extract Alleviates DSS-Induced Colitis in Mice by Restoring Intestinal Barrier Function and Maintaining Gut Microbiota Homeostasis. Food Biosci. 2023, 53, 102750. [Google Scholar] [CrossRef]
- Huang, Y.; Yang, Q.; Mi, X.; Qiu, L.; Tao, X.; Zhang, Z.; Xia, J.; Wu, Q.; Wei, H. Ripened Pu-Erh Tea Extract Promotes Gut Microbiota Resilience against Dextran Sulfate Sodium Induced Colitis. J. Agric. Food Chem. 2021, 69, 2190–2203. [Google Scholar] [CrossRef]
- Al-Zalabani, A.H.; Wesselius, A.; Yi-Wen Yu, E.; van den Brandt, P.; Grant, E.J.; White, E.; Skeie, G.; Liedberg, F.; Weiderpass, E.; Zeegers, M.P. Tea Consumption and Risk of Bladder Cancer in the Bladder Cancer Epidemiology and Nutritional Determinants (BLEND) Study: Pooled Analysis of 12 International Cohort Studies. Clin. Nutr. 2022, 41, 1122–1130. [Google Scholar] [CrossRef]
- Bamia, C.; Lagiou, P.; Jenab, M.; Trichopoulou, A.; Fedirko, V.; Aleksandrova, K.; Pischon, T.; Overvad, K.; Olsen, A.; Tjønneland, A.; et al. Coffee, Tea and Decaffeinated Coffee in Relation to Hepatocellular Carcinoma in a European Population: Multicentre, Prospective Cohort Study. Int. J. Cancer 2015, 136, 1899–1908. [Google Scholar] [CrossRef]
- He, M.; Lyu, X. Application of BRAFO-Tiered Approach for Health Benefit-Risk Assessment of Dark Tea Consumption in China. Food Chem. Toxicol. 2021, 158, 112615. [Google Scholar] [CrossRef] [PubMed]
- O’Neill, E.J.; Termini, D.; Albano, A.; Tsiani, E. Anti-Cancer Properties of Theaflavins. Molecules 2021, 26, 987. [Google Scholar] [CrossRef] [PubMed]
- Zheng, F.; Chen, K.; Zhong, J.; Tang, S.; Xu, S.; Lu, W.; Wu, Y.; Xia, D. Association between Different Types of Tea Consumption and Risk of Gynecologic Cancer: A Meta-Analysis of Cohort Studies. Nutrients 2023, 15, 403. [Google Scholar] [CrossRef] [PubMed]
- Seow, W.J.; Koh, W.-P.; Jin, A.; Wang, R.; Yuan, J.-M. Associations between Tea and Coffee Beverage Consumption and the Risk of Lung Cancer in the Singaporean Chinese Population. Eur. J. Nutr. 2020, 59, 3083–3091. [Google Scholar] [CrossRef]
- Selvakumar, P.; Badgeley, A.; Murphy, P.; Anwar, H.; Sharma, U.; Lawrence, K.; Lakshmikuttyamma, A. Flavonoids and Other Polyphenols Act as Epigenetic Modifiers in Breast Cancer. Nutrients 2020, 12, 761. [Google Scholar] [CrossRef]
- Siegel, R.L.; Miller, K.D.; Wagle, N.S.; Jemal, A. Cancer Statistics, 2023. CA A Cancer J. Clin. 2023, 73, 17–48. [Google Scholar] [CrossRef] [PubMed]
- Lazzari, G.; Solazzo, A.P.; Benevento, I.; Montagna, A.; Rago, L.; Castaldo, G.; Silvano, G. Current Trends and Challenges in Real-World Breast Cancer Adjuvant Radiotherapy: A Practical Review: New Trends in Adjuvant Radiotherapy in BC. Arch. Breast Cancer 2023, 10, 6–14. [Google Scholar] [CrossRef]
- Freitas, S.C.; Sanderson, D.; Caspani, S.; Magalhães, R.; Cortés-Llanos, B.; Granja, A.; Reis, S.; Belo, J.H.; Azevedo, J.; Gómez-Gaviro, M.V.; et al. New Frontiers in Colorectal Cancer Treatment Combining Nanotechnology with Photo- and Radiotherapy. Cancers 2023, 15, 383. [Google Scholar] [CrossRef]
- Shah, R.; Shah, H.; Thakkar, K.; Parikh, N. Conventional Therapies for Oral Cancers: Highlights on Chemotherapeutic Agents and Radiotherapy; Their Adverse Effects and Cost Burden. Crit. Rev. Trade Oncog. 2023, 28, 1–10. [Google Scholar] [CrossRef]
- Zhang, S.; Mao, B.; Cui, S.; Zhang, Q.; Zhao, J.; Tang, X.; Chen, W. Absorption, Metabolism, Bioactivity, and Biotransformation of Epigallocatechin Gallate. Crit. Rev. Food Sci. Nutr. 2023; ahead of print. [Google Scholar] [CrossRef]
- Qi, C.; Liu, G.; Ping, Y.; Yang, K.; Tan, Q.; Zhang, Y.; Chen, G.; Huang, X.; Xu, D. A Comprehensive Review of Nano-Delivery System for Tea Polyphenols: Construction, Applications, and Challenges. Food Chem. X 2023, 17, 100571. [Google Scholar] [CrossRef]


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Deng, H.; Liu, J.; Xiao, Y.; Wu, J.-L.; Jiao, R. Possible Mechanisms of Dark Tea in Cancer Prevention and Management: A Comprehensive Review. Nutrients 2023, 15, 3903. https://doi.org/10.3390/nu15183903
Deng H, Liu J, Xiao Y, Wu J-L, Jiao R. Possible Mechanisms of Dark Tea in Cancer Prevention and Management: A Comprehensive Review. Nutrients. 2023; 15(18):3903. https://doi.org/10.3390/nu15183903
Chicago/Turabian StyleDeng, Huilin, Jia Liu, Ying Xiao, Jian-Lin Wu, and Rui Jiao. 2023. "Possible Mechanisms of Dark Tea in Cancer Prevention and Management: A Comprehensive Review" Nutrients 15, no. 18: 3903. https://doi.org/10.3390/nu15183903
APA StyleDeng, H., Liu, J., Xiao, Y., Wu, J.-L., & Jiao, R. (2023). Possible Mechanisms of Dark Tea in Cancer Prevention and Management: A Comprehensive Review. Nutrients, 15(18), 3903. https://doi.org/10.3390/nu15183903







