Effects of Caffeine Consumption on Attention Deficit Hyperactivity Disorder (ADHD) Treatment: A Systematic Review of Animal Studies
Abstract
:1. Introduction
2. Materials and Methods
2.1. Search Strategy
2.2. Study Selection Criteria
2.3. Study Selection
2.4. Data Extraction and Analyses
3. Results
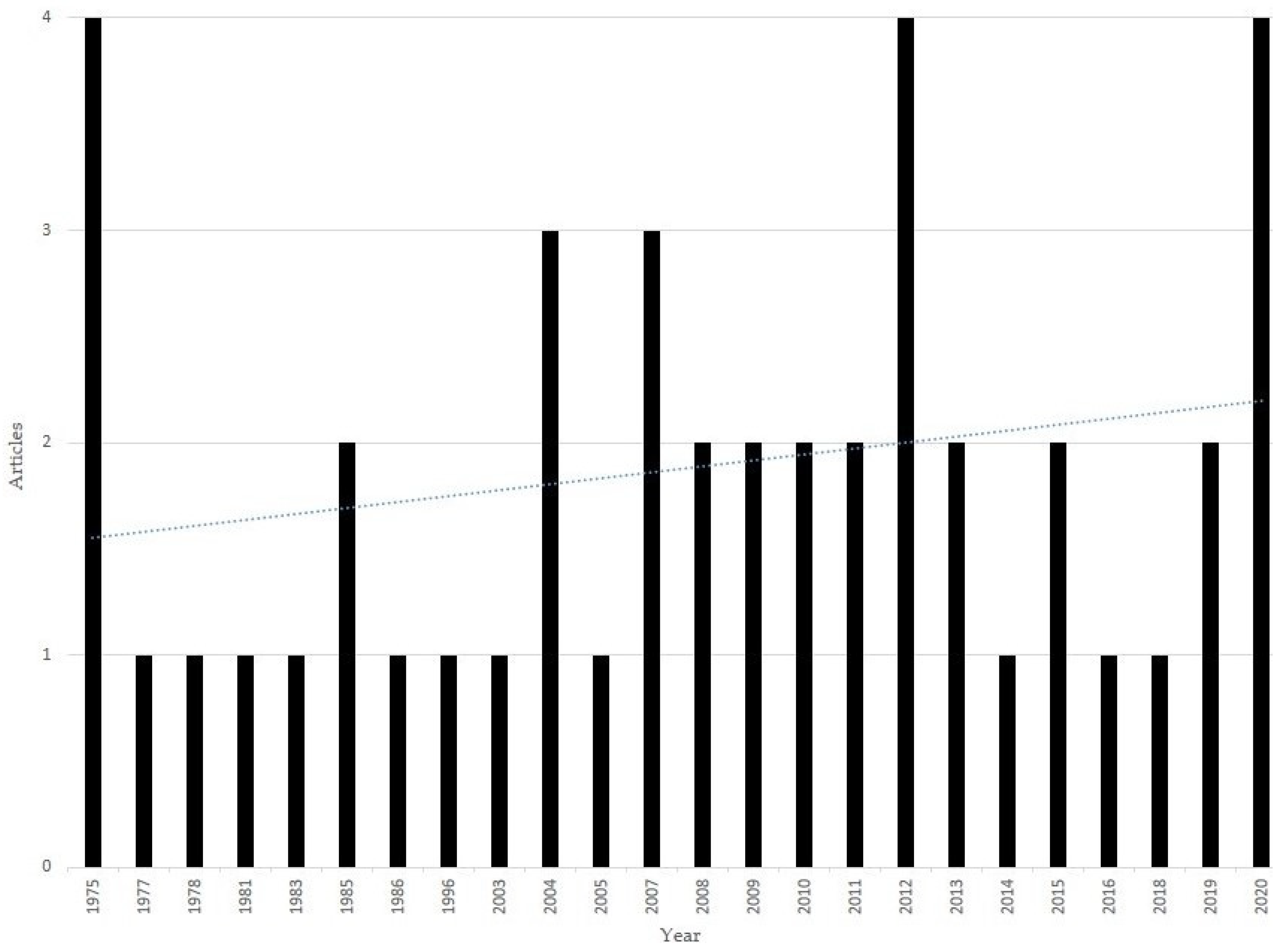
3.1. Study Selection
3.1.1. Species, Animal Model, Sex, and Treatment
3.1.2. Animal Models of ADHD
3.1.3. Behavioral Tests
3.2. Study Outcomes
3.2.1. Attention
Attention and Behavioral Flexibility
Spatial Attention
Discrimination
Selective Attention
3.2.2. Hyperactivity and Impulsivity
Locomotor Activity
Impulsive Behavior
3.2.3. Learning and Memory
Non-Associative Learning
Working Memory
Spatial Learning
Spatial Short-Term Memory
3.2.4. Olfactory Discrimination
3.2.5. Blood Pressure
3.2.6. Body Weight
3.2.7. Neurobiology
Brain Levels of Synaptosomal-Associated Protein-25
Brain Levels of Syntaxin
Brain Levels of Serotonin
Brain Levels of Dopamine
Dopamine Transporter Density
Dopamine Uptake
AdenosineA2A Receptor Density
Colocalization of Dopamine Transporter and Adenosine A2A Receptors
Brain-Derived Neurotrophic Factor
Neuronal Development In Vitro
4. Discussion
5. Conclusions
Author Contributions
Funding
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ferguson, J.H. National Institutes of Health Consensus Development Conference Statement: Diagnosis and treatment of attention-deficit/hyperactivity disorder (ADHD). J. Am. Acad. Child Adolesc. Psychiatry 2000, 39, 182–193. [Google Scholar] [CrossRef] [PubMed]
- Dopheide, J.A.; Pliszka, S.R. Attention-deficit-hyperactivity disorder: An update. Pharmacotherapy 2009, 29, 656–679. [Google Scholar] [CrossRef] [PubMed]
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders: DSM-5; American Psychiatric Association: Arlington, VA, USA, 2013. [Google Scholar]
- ICD-11: 6A05 Attention Deficit Hyperactivity Disorder. Available online: https://icd.who.int/browse11/l-m/en#/http%3a%2f%2fid.who.int%2ficd%2fentity%2f821852937 (accessed on 22 December 2021).
- Franke, B.; Michelini, G.; Asherson, P.; Banaschewski, T.; Bilbow, A.; Buitelaar, J.K.; Cormand, B.; Faraone, S.V.; Ginsberg, Y.; Haavik, J.; et al. Live fast, die young? A review on the developmental trajectories of ADHD across the lifespan. Eur. Neuropsychopharmacol. 2018, 28, 1059–1088. [Google Scholar] [CrossRef] [PubMed]
- Tsuang, M.T.; Tohen, M.; Jones, P. Textbook of Psychiatric Epidemiology, 3rd ed.; John Wiley & Sons: Chichester, UK, 2011. [Google Scholar]
- Global Health Metrics: Attention-Deficit/Hyperactivity Disorder—Level 3 Cause. Available online: https://www.thelancet.com/pb-assets/Lancet/gbd/summaries/diseases/adhd.pdf (accessed on 22 December 2021).
- NICE Guideline [NG87]—Attention Deficit Hyperactivity Disorder: Diagnosis and Management. Available online: https://www.nice.org.uk/guidance/ng87/ (accessed on 22 December 2021).
- Canadian ADHD Practice Guidelines. Available online: https://www.caddra.ca/cms4/pdfs/caddraGuidelines2011Introduction.pdf (accessed on 22 December 2021).
- Centers for Disease Control and Prevention: ADHD Treatment Recommendations. Available online: https://www.cdc.gov/ncbddd/adhd/guidelines.html (accessed on 22 December 2021).
- Wolraich, M.L.; Hagan, J.F.; Allan, C.; Chan, E.; Davison, D.; Earls, M.; Evans, S.W.; Flinn, S.K.; Froehlich, T.; Frost, J.; et al. Clinical practice guideline for the diagnosis, evaluation, and treatment of attention-deficit/hyperactivity disorder in children and adolescents. Pediatrics 2019, 144, e20192528. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schonwald, A.; Lechner, E. Attention deficit/hyperactivity disorder: Complexities and controversies. Curr. Opin. Pediatr. 2006, 18, 189–195. [Google Scholar] [CrossRef]
- Wigal, S.B. Efficacy and safety limitations of attention-deficit hyperactivity disorder pharmacotherapy in children and adults. CNS Drugs 2009, 23, 21–31. [Google Scholar] [CrossRef]
- Parker, J.; Wales, G.; Chalhoub, N.; Harpin, V. The long-term outcomes of interventions for the management of attention-deficit hyperactivity disorder in children and adolescents: A systematic review of randomized controlled trials. Psychol. Res. Behav. Manag. 2013, 6, 87. [Google Scholar] [CrossRef] [Green Version]
- Arnold, L.E.; Hodgkins, P.; Caci, H.; Kahle, J.; Young, S. Effect of treatment modality on long-term outcomes in attention-deficit/hyperactivity disorder: A systematic review. PLoS ONE 2015, 10, e0116407. [Google Scholar] [CrossRef] [Green Version]
- Castellanos, F.X.; Proal, E. Large-scale brain systems in ADHD: Beyond the prefrontal–striatal model. Trends Cogn. Sci. 2012, 16, 17–26. [Google Scholar] [CrossRef] [Green Version]
- Cortese, S.; Kelly, C.; Chabernaud, C.; Proal, E.; Di Martino, A.; Milham, M.P.; Castellanos, F.X. Toward systems neuroscience of ADHD: A meta-analysis of 55 fMRI studies. Am. J. Psychiatry 2012, 169, 1038–1055. [Google Scholar] [CrossRef]
- Stahl, S.M. Stahl’s Essential Psychopharmacology: Neuroscientific Basis and Practical Applications, 4th ed.; Cambridge University Press: Cambridge, UK, 2013. [Google Scholar]
- Haenlein, M.; Caul, W.F. Attention deficit disorder with hyperactivity: A specific hypothesis of reward dysfunction. J. Am. Acad. Child Adolesc. Psychiatry 1987, 26, 356–362. [Google Scholar] [CrossRef]
- Johansen, E.B.; Killeen, P.R.; Russell, V.A.; Tripp, G.; Wickens, J.R.; Tannock, R. Origins of altered reinforcement effects in ADHD. Behav. Brain Funct. 2009, 5, 7. [Google Scholar] [CrossRef] [Green Version]
- Kollins, S.H.; Lane, S.D.; Shapiro, S.K. The experimental analysis of childhood psychopathology: A matching analysis of the behavior of children diagnosed with Attention Deficit Hyperactivity Disorder. Psychol. Record. 1997, 47, 25–44. [Google Scholar] [CrossRef] [Green Version]
- Sonuga-Barke, E.J. The dual pathway model of AD/HD: An elaboration of neuro-developmental characteristics. Neurosci. Biobehav. Rev. 2003, 27, 593–604. [Google Scholar] [CrossRef] [PubMed]
- Tripp, G.; Wickens, J.R. Research review: Dopamine transfer deficit: A neurobiological theory of altered reinforcement mechanisms in ADHD. J. Child Psychol. Psychiatry 2008, 49, 691–704. [Google Scholar] [CrossRef]
- Nestler, E.J.; Hyman, S.E.; Malenka, R.C.; Holtzman, D.M. Molecular Neuropharmacology: A Foundation for Clinical Neuroscience, 2nd ed.; McGraw-Hill Medical: New York, NY, USA, 2009. [Google Scholar]
- Biederman, J.; Faraone, S.V. Current concepts on the neurobiology of Attention-Deficit/Hyperactivity Disorder. J. Atten. Disord. 2002, 6, 7–16. [Google Scholar] [CrossRef]
- Bidwell, L.C.; McClernon, F.J.; Kollins, S.H. Cognitive enhancers for the treatment of ADHD. Pharmacol. Biochem. Behav. 2011, 99, 262–274. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cortese, S. The neurobiology and genetics of attention-deficit/hyperactivity disorder (ADHD): What every clinician should know. Eur. J. Paediatr. Neurol. 2012, 16, 422–433. [Google Scholar] [CrossRef] [PubMed]
- Lesch, K.P.; Merker, S.; Reif, A.; Novak, M. Dances with black widow spiders: Dysregulation of glutamate signalling enters centre stage in ADHD. Eur. Neuropsychopharmacol. 2013, 23, 479–491. [Google Scholar] [CrossRef]
- Gizer, I.R.; Ficks, C.; Waldman, I.D. Candidate gene studies of ADHD: A meta-analytic review. Hum. Genet. 2009, 126, 51–90. [Google Scholar] [CrossRef]
- Kebir, O.; Joober, R. Neuropsychological endophenotypes in attention-deficit/hyperactivity disorder: A review of genetic association studies. Eur. Arch. Psychiatry Clin. Neurosci. 2011, 261, 583–594. [Google Scholar] [CrossRef] [PubMed]
- Berry, M.D. The potential of trace amines and their receptors for treating neurological and psychiatric diseases. Rev. Recent Clin. Trials. 2007, 2, 3–19. [Google Scholar] [CrossRef] [Green Version]
- Sotnikova, T.D.; Caron, M.G.; Gainetdinov, R.R. Trace amine-associated receptors as emerging therapeutic targets. Mol. Pharmacol. 2009, 76, 229–235. [Google Scholar] [PubMed] [Green Version]
- Chandler, D.J.; Waterhouse, B.D.; Gao, W.J. New perspectives on catecholaminergic regulation of executive circuits: Evidence for independent modulation of prefrontal functions by midbrain dopaminergic and noradrenergic neurons. Front. Neural Circuits 2014, 8, 53. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rahi, V.; Kumar, P. Animal models of attention-deficit hyperactivity disorder (ADHD). Int. J. Dev. Neurosci. 2021, 81, 107–124. [Google Scholar] [CrossRef]
- Russell, V.A. Hypodopaminergic and hypernoradrenergic activity in prefrontal cortex slices of an animal model for attention-deficit hyperactivity disorder—The spontaneously hypertensive rat. Behav. Brain Res. 2002, 130, 191–196. [Google Scholar] [CrossRef]
- Matias, A.; Zimmer, F.J.; Lorenzen, A.; Keil, R.; Schwabe, U. Affinity of central adenosine A1 receptors is decreased in spontaneously hypertensive rats. Eur. J. Pharmacol. 1993, 244, 223–230. [Google Scholar] [CrossRef]
- Fredholm, B.B.; Bättig, K.; Holmén, J.; Nehlig, A.; Zvartau, E.E. Actions of Caffeine in the Brain with Special Reference to Factors That Contribute to Its Widespread Use. Pharmacol. Rev. 1999, 51, 83–133. [Google Scholar]
- Latini, S.; Pedata, F. Adenosine in the central nervous system: Release mechanisms and extracellular concentrations. J. Neurochem. 2001, 79, 463–484. [Google Scholar] [CrossRef] [Green Version]
- Ferré, S. An update on the mechanisms of the psychostimulant effects of caffeine. J. Neurochem. 2008, 105, 1067–1079. [Google Scholar] [CrossRef]
- Chen, Y.; Parrish, T.B. Caffeine dose effect on activation-induced BOLD and CBF responses. Neuroimage 2009, 46, 577–583. [Google Scholar] [CrossRef] [Green Version]
- Diukova, A.; Ware, J.; Smith, J.E.; Evans, C.J.; Murphy, K.; Rogers, P.J.; Wise, R.G. Separating neural and vascular effects of caffeine using simultaneous EEG-FMRI: Differential effects of caffeine on cognitive and sensorimotor brain responses. Neuroimage 2012, 62, 239–249. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Garfinkel, B.D.; Webster, C.D.; Sloman, L. Responses to methylphenidate and varied doses of caffeine in children with attention deficit disorder. Can. J. Psychiatry 1981, 26, 395–401. [Google Scholar]
- Firestone, P.; Davey, J.; Goodman, J.T.; Peters, S. The effects of caffeine and methylphenidate on hyperactive children. J. Am. Acad. Child Psychiatry 1978, 17, 445–456. [Google Scholar] [CrossRef]
- Cunha, R.A.; Ferré, S.; Vaugeois, J.M.; Chen, J.F. Potential therapeutic interest of adenosine A2A receptors in psychiatric disorders. Curr. Pharm. Des. 2008, 14, 1512–1524. [Google Scholar] [CrossRef] [Green Version]
- Cunha, R.A. How does adenosine control neuronal dysfunction and neurodegeneration? J. Neurochem. 2016, 139, 1019–1055. [Google Scholar] [CrossRef] [PubMed]
- Harvey, D.H.; Marsh, R.W. The effects of de-caffeinated coffee versus whole coffee on hyperactive children. Dev. Med. Child Neurol. 1978, 20, 81–86. [Google Scholar] [CrossRef]
- Huestis, R.; Arnold, L.; Smeltzer, D. Caffeine versus methylphenidate and d-amphetamine in minimal brain dysfunction: A double-blind comparison. Am. J. Psychiatry 1975, 132, 868–870. [Google Scholar] [PubMed] [Green Version]
- Garfinkel, B.D.; Webster, C.D.; Sloman, L. Individual responses to methylphenidate and caffeine in children with minimal brain dysfunction. Can. Med. Assoc. J. 1975, 113, 729–732. [Google Scholar]
- Marmorstein, N.R. Energy drink and coffee consumption and psychopathology symptoms among early adolescents: Crosssectional and longitudinal associations. J. Caffeine Res. 2016, 6, 64–72. [Google Scholar] [CrossRef] [Green Version]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G.; The PRISMA Group. Preferred Reporting Items for Systematic Reviews and Meta-Analyses: The PRISMA Statement. PLoS Med. 2009, 6, e1000097. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pandolfo, P.; Machado, N.J.; Köfalvi, A.; Takahashi, R.N.; Cunha, R.A. Caffeine regulates frontocorticostriatal dopamine transporter density and improves attention and cognitive deficits in an animal model of attention deficit hyperactivity disorder. Eur. Neuropsychopharmacol. 2013, 23, 317–328. [Google Scholar] [CrossRef]
- Ouchi, H.; Ono, K.; Murakami, Y.; Matsumoto, K. Social isolation induces deficit of latent learning performance in mice: A putative animal model of attention deficit/hyperactivity disorder. Behav. Brain. Res. 2013, 238, 146–153. [Google Scholar] [CrossRef]
- Caballero, M.; Núñez, F.; Ahern, S.; Cuffí, M.L.; Carbonell, L.; Sánchez, S.; Fernández-Dueñas, V.; Ciruela, F. Caffeine improves attention deficit in neonatal 6-OHDA lesioned rats, an animal model of attention deficit hyperactivity disorder (ADHD). Neurosci. Lett. 2011, 494, 44–48. [Google Scholar] [CrossRef]
- Ruiz-Oliveira, J.; Silva, P.F.; Luchiari, A.C. Coffee time: Low caffeine dose promotes attention and focus in zebrafish. Learn. Behav. 2019, 47, 227–233. [Google Scholar] [CrossRef] [Green Version]
- Higgins, G.A.; Grzelak, M.E.; Pond, A.J.; Cohen-Williams, M.E.; Hodgson, R.A.; Varty, G.B. The effect of caffeine to increase reaction time in the rat during a test of attention is mediated through antagonism of adenosine A2A receptors. Behav. Brain. Res. 2007, 185, 32–42. [Google Scholar] [CrossRef] [PubMed]
- França, A.P.; Schamne, M.G.; de Souza, B.S.; da Luz Scheffer, D.; Bernardelli, A.K.; Corrêa, T.; de Souza Izídio, G.; Latini, A.; da Silva-Santos, J.E.; Canas, P.M.; et al. Caffeine consumption plus physical exercise improves behavioral impairments and stimulates neuroplasticity in Spontaneously Hypertensive Rats (SHR): An animal model of attention deficit hyperactivity disorder. Mol. Neurobiol. 2020, 57, 3902–3919. [Google Scholar] [CrossRef]
- Nunes, F.; Pochmann, D.; Almeida, A.S.; Marques, D.M.; Porciúncula, L.O. Differential behavioral and biochemical responses to caffeine in male and female rats from a validated model of attention deficit and hyperactivity disorder. Mol. Neurobiol. 2018, 55, 8486–8498. [Google Scholar] [CrossRef] [PubMed]
- Szczepanik, J.C.; de Oliveira, P.A.; de Oliveira, J.; Mack, J.M.; Engel, D.F.; Rial, D.; Moreira, E.L.; de Bem, A.F.; Prediger, R.D. Caffeine Mitigates the Locomotor Hyperactivity in Middle-aged Low-density Lipoprotein Receptor (LDLr)-Knockout Mice. CNS Neurosci. Ther. 2016, 22, 420. [Google Scholar] [CrossRef]
- Pires, V.A.; Pamplona, F.A.; Pandolfo, P.; Prediger, R.D.S.; Takahashi, R.N. Chronic caffeine treatment during prepubertal period confers long-term cognitive benefits in adult spontaneously hypertensive rats (SHR), an animal model of attention deficit hyperactivity disorder (ADHD). Behav. Brain. Res. 2010, 215, 39–44. [Google Scholar] [CrossRef]
- Prediger, R.D.S.; Pamplona, F.A.; Fernandes, D.; Takahashi, R.N. Caffeine improves spatial learning deficits in an animal model of attention deficit hyperactivity disorder (ADHD)-the spontaneously hypertensive rat (SHR). Int. J. Neuropsychopharmacol. 2005, 8, 583–594. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Leffa, D.T.; Ferreira, S.G.; Machado, N.J.; Souza, C.M.; Rosa, F.D.; de Carvalho, C.; Kincheski, G.C.; Takahashi, R.N.; Porciúncula, L.O.; Souza, D.O. Caffeine and cannabinoid receptors modulate impulsive behavior in an animal model of attentional deficit and hyperactivity disorder. Eur. J. Neurosci. 2019, 49, 1673–1683. [Google Scholar] [CrossRef] [PubMed]
- Pires, V.A.; Pamplona, F.A.; Pandolfo, P.; Fernandes, D.; Prediger, R.D.; Takahashi, R.N. Adenosine receptor antagonists improve short-term object-recognition ability of spontaneously hypertensive rats: A rodent model of attention-deficit hyperactivity disorder. Behav. Pharmacol. 2009, 20, 134–145. [Google Scholar] [CrossRef]
- Alves, C.B.; Almeida, A.S.; Marques, D.M.; Faé, A.H.L.; Machado, A.C.L.; Oliveira, D.L.; Porciúncula, L.O. Caffeine and adenosine A2A receptors rescue neuronal development in vitro of frontal cortical neurons in a rat model of attention deficit and hyperactivity disorder. Neuropharmacology 2020, 166, 107782. [Google Scholar] [CrossRef] [PubMed]
- Leon, M.R. Effects of caffeine on cognitive, psychomotor, and affective performance of children with attention-deficit/hyperactivity disorder. J. Atten. Disord. 2000, 4, 27–47. [Google Scholar] [CrossRef]
- Ioannidis, K.; Chamberlain, S.R.; Müller, U. Ostracising caffeine from the pharmacological arsenal for attention-deficit hyperactivity disorder—Was this a correct decision? A literature review. J. Psychopharmacol. 2014, 28, 830–836. [Google Scholar] [CrossRef]
- Fredholm, B.B. Adenosine and neuroprotection. Int. Rev. Neurobiol. 1996, 40, 259–280. [Google Scholar]
- Claghorn, G.C.; Thompson, Z.; Wi, K.; Van, L.; Garland, T., Jr. Caffeine stimulates voluntary wheel running in mice without increasing aerobic capacity. Physiol. Behav. 2017, 170, 133–140. [Google Scholar] [CrossRef]
- Cunha, R.A.; Agostinho, P.M. Chronic caffeine consumption prevents memory disturbance in different animal models of memory decline. J. Alzheimers Dis. 2010, 20, 95–116. [Google Scholar] [CrossRef] [Green Version]


| Author/s & Year | Species, Strain, Sex & Sample (n) | Animal Model | Age | Independent Variables | Caffeine Treatment | Behavioral Tests/ Type of Stress | Dependent Variables | Main Results |
|---|---|---|---|---|---|---|---|---|
| Szczepanik et al., 2016 | Mice C57Bl/6 wild-type (8) LDLr (8) Female | Genetic (LDLr) | 3 months 8 months | Treatment (caffeine or vehicle) Strain (C57Bl/6 wild type or LDLr) | 10 mg/kg oral route Chronic treatment (21 days) | Open-field arena | Spontaneous locomotor activity (total distance travelled) Anxiety (time in the center) Exploratory behavior (visual inspection of the occupation plot) | - LDLr mice travelled greater distances than the C57BI/6 wild type mice during the 5 min period of analysis. - Caffeine treatment induced a renormalization effect in 8 month-old mouse locomotion. - Caffeine treatment was unable to modify the hyperlocomotion observed in 3 month-old LDLr mice. - All animal groups spent a similar amount of time in the center of an open field. - Similar exploratory behavior between groups. |
| Higgins et al., 2007 | Rats LE (15–16) CD (12–16) Male | Not used | Not specified | Treatment (caffeine, SCH412348, KW-6002, DPCPX, CGS-21680, amphetamine) Strain (LE or CD) | 1 mL/kg i.p. route One dose, prior testing | Five-choice serial reaction time task Locomotor activity test | Selective attention (Correct/incorrect trials, omissions, premature and perseverative responses, choice accuracy, correct/incorrect, and magazine latency) Hypolocomotion (distance travelled) | - Caffeine, SCH 412348 and KW-6002 augmented time reaction in LE and CD, without effect on accuracy. - Effects of SCH 412348 were at doses that were not overtly psychostimulatory. - CGS-21680 reduced speed reaction and augmented omissions. A CGS-21680 lower dose reduced the increased premature response caused by amphetamine. - Caffeine’s attentional-enhancing effects were facilitated through A2A receptor blockade. Selective A2A receptor antagonists could be included as a potential therapy for ADHD. |
| Ruiz-Oliveira et al., 2019 | Zebrafish wild-type (40) Male Female | Not used | 4 months | Treatment (caffeine or vehicle) | 10 mg/L 50 mg/L drinking water Chronic treatment (14 days) | Discrimination task | Conditioned learning ability (average swimming speed, intergroup freezing, maximum speed, time spent in each area, latency to enter each area) | - 0 and 10 mg/L caffeine groups spent most of the time close to the target. - 10 mg caffeine group had the shortest latency to reach the target. - 0 and 10 mg/L caffeine groups increased the average speed and distance travelled. - Caffeine exposure at low doses seems to enhance visual cue discrimination and zebrafish performance. |
| Prediger et al., 2005 | Rats WKY (7–8) SHR (7–8) Female | Genetic (SHR) | 3 months | Treatment (caffeine or vehicle) Strain (WKY or SHR) | 1.3 mg/kg 10 mg/kg i.p. route One dose, prior testing | Water maze task | Spatial learning (escape latency, distance travelled, swimming speed) Mean arterial pressure | - SHR needed a larger amount of trials during the training session to learn the spatial information, although a similar profile to that of WKY rats during the test session, showing a selective spatial learning deficit. - Caffeine’s pre-training administration enhanced SHRs’ spatial learning deficit. - Caffeine’s post-training administration did not enhance SHRs’ test performance, although it improved WKY rats’ memory retention. - Mean blood pressure was not altered by caffeine. |
| Pires et al., 2009 | Rats WKY (15) SHR (18) Male Female | Genetic (SHR) | 3 months | Treatment (MPD, DPCPX, caffeine, ZM241385 or vehicle) Strain (WKY or SHR) | 1 mg/kg 3 mg/kg 10 mg/kg i.p. route One dose, prior testing | Object recognition task | Object recognition (investigation time, discrimination time) Mean arterial pressure | - SHR only discriminated between the most structurally distinct pairs of objects. - Pre-training administration of MPD, caffeine, the selective adenosine receptor antagonists DPCPX and ZM241385, or the association of ineffective doses of DPCPX and ZM241385, improved the performance of SHR in the object-recognition task. - The administration of the same doses of MPD and caffeine did not significantly alter the mean arterial pressure of either WKYs or SHRs. |
| Pires et al., 2010 | Rats WKY (37) SHR (38) Female | Genetic (SHR) | 25/38 days | Treatment (caffeine, MPD or vehicle) Strain (WKY or SHR) | 3 mg/kg i.p. Route. Chronic treatment (14 days) | Object recognition task | Object recognition (investigation time, discrimination time) Spontaneous locomotor activity Mean arterial pressure Body weight | - WKY rats distinguished all the items. SHRs were unable to distinguish pairs of items with slight structural alterations. - Caffeine or MPD chronic treatment enhanced SHR item-recognition deficits. The same treatments impaired the adult WKY rats’ short-term object recognition ability. - Effects were independent of variations in locomotion, arterial blood pressure, and body weight. |
| Caballero et al., 2011 | Rats 6-OHDA lesioned (9) Saline- lesioned (9) Male Female | Physical trauma (6-OHDA lesioned) | 25 days | Treatment (caffeine or vehicle) | 1 mg/mL drinking water Chronic treatment (14 days) | Olton maze behavioral assay | Motor behavior (number of arms crossed) Attention behavior (total number of arms walked, and total number of arms walked until one was repeated) | - Caffeine treatment significantly improved 6-OHDA lesioned rats’ attention deficit. - After caffeine consumption, no changes were found in motor activity. |
| Pandolfo et al., 2013 | Rats WKY (16) SHR (16) Male | Genetic (SHR) | 24 days | Treatment (caffeine or vehicle (saline)) Strain (WKY or SHR) | 2 mg/kg i.p. route Chronic treatment (twice daily for 21 days) | Attentional-set shifting; anxiety-related behavior; Y maze; locomotion -related behavior | Attention (regressive and never- reinforced errors, perseverative errors, total number of trials required before reaching 10 correct consecutive choices) Locomotion and anxiety (number of peripheral squares crossed, number of central squares crossed, percentage of central locomotion) Spatial recognition (number of entries; time spent per arm; random exploration) | - SHRs were hyperactive and showed poorer performance in the attentional set-shifting and Y-maze paradigms, displayed increased dopamine transporter density, and increased dopamine uptake in frontocortical and striatal terminals. - Chronic caffeine treatment improved memory and attention deficits, and normalized dopaminergic function in SHR. - First indication of adenosine A2A receptors (A2AR) in nerve terminals in frontal cortex. - First evidence that A2AR density is improved in SHR. |
| Ouichi et al., 2013 | Mice ICR (9) Male Female | Physical trauma (SI) | 4 weeks | Treatment (MPD and caffeine) SI | 0.5–1 mg/kg i.p. route One dose, prior testing | Water-finding test; aggression; modified Y-maze test; novel object recognition test; fear-conditioning test | Spatial attention (entering & drinking latency) Aggression (duration of wrestling) Spatial recognition (time spent in the new arm; total time exploring objects) Fear conditioning (freezing behavior) | - SI rats showed deficits in spatial attention on the water-finding test. Re-socialized did not reduce deficit in spatial attention. SI effect on spatial attention revealed no difference in gender or correlation with aggressive behaviour. - SI impaired conditional and contextual fear memory. - MPD and caffeine enhanced deficits in SI-induced latent learning in a manner that was reversible with cholinergic but not dopaminergic antagonists. |
| Nunes et al., 2018 | Rats WKY (5–15) SHR (5–15) Male Female | Genetic (SHR) | 15 days 28 days 50 days | Treatment (caffeine/water, caffeine/caffeine or water) Strain (WKY or SHR) | 0.3 g/L drinking water Until PND 28 | Open-field test; Novel object recognition; Y maze task | Open field test (travel distance periphery) Habituation (total travelled distance in the open field) Spatial recognition- Y maze and object recognition (exploration, discrimination ratio, number of entries, time spent in novel arm, total number of entries in three arms) | - Adolescent SHR from both sexes displayed hyperlocomotion, recognition, and spatial memory disturbances. Females displayed a lack of habituation and deteriorated spatial memory. - Caffeine was effective at improving recognition memory damage in both sexes. - Spatial memory was improved only in female SHRs. - Female SHRs displayed impaired hyperlocomotion following caffeine treatment. - SHRs of both sexes presented increases in BDNF, truncated and phospho-TrkB receptors, and phospho-CREB levels in the hippocampus. - Caffeine normalized BDNF in males and truncated TrkB receptor in both sexes. |
| Leffa et al., 2019 | Rats WKY (7–9) SHR (7–9) Male | Genetic (SHR) | 60/65 days 24 days | Treatment (WIN, AM251, caffeine or vehicle) Strain (WKY or SHR) | 2 or 5 mg/kg i.p. route Acute pretreatment, one dose Chronic treatment (21 days) | Tolerance to delay of reward; T maze | Impulsive behavior (tolerance to delay of reward) | - WIN treatment decreased large reward choices and AM251 treatment increased large reward choices in SHR. - Acute caffeine pretreatment blocked WIN effects. - Chronic caffeine treatment increased the impulsive phenotype and potentiated the WIN effects. - Cannabinoid and adenosine receptors modulate impulsive behavior in SHR. |
| Alves et al., 2020 | Rats-pregnant SHR (40–70) WKY (40–70) Female | Genetic (SHR) | In vitro | Treatment (caffeine, DMSO, LY294002, adenosine selective agonist and antagonists) Strain (WKY or SHR) | Caffeine incubation (30 µM) One dose | No behavioral task | Morphological alterations (singling, neurite branching) | - SHR neurons displayed less neurite branching, shorter maximal neurite length and decreased axonal outgrowth. - Caffeine recovered neurite branching and elongation from SHR neurons via PKA and PI3K signaling, - A2AR agonist (CGS 21680) promoted more neurite branching via PKA signaling. - The selective A2AR antagonist (SCH 58261) was efficient at recovering axonal outgrowth from SHR neurons through PI3K and not PKA signaling. |
| França et al., 2020 | Rats WKY (9) SHR (11) Male | Genetic (SHR) | 30 days 4–5 months | Treatment (caffeine or water) Physical exercise Strain (WKY or SHR) | 0.3 mg/mL, drinking water One dose | Olfactory discrimination; Open field; Object recognition; Water maze | Olfactory discrimination (time spent in compartments, numbers of crossings) Locomotor activity (total distance, time spent in the central zone) Short-term memory (total time spent exploring the objects, discrimination index) Working and procedural memories (escape latency) | - SHR showed olfactory and short-term recognition memory deficiencies from adolescence to adulthood, accompanied by lower prefrontal cortex and hippocampus SNAP-25 levels. - Caffeine and physical exercise during adolescence or adulthood repaired the olfactory discrimination ability and enhanced short-term recognition memory in SHRs. - Caffeine consumption and physical exercise during adolescence augmented hippocampus and prefrontal cortex SNAP-25, syntaxin, and serotonin levels, as well as SHRs’ striatal dopamine levels. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vázquez, J.C.; Martin de la Torre, O.; López Palomé, J.; Redolar-Ripoll, D. Effects of Caffeine Consumption on Attention Deficit Hyperactivity Disorder (ADHD) Treatment: A Systematic Review of Animal Studies. Nutrients 2022, 14, 739. https://doi.org/10.3390/nu14040739
Vázquez JC, Martin de la Torre O, López Palomé J, Redolar-Ripoll D. Effects of Caffeine Consumption on Attention Deficit Hyperactivity Disorder (ADHD) Treatment: A Systematic Review of Animal Studies. Nutrients. 2022; 14(4):739. https://doi.org/10.3390/nu14040739
Chicago/Turabian StyleVázquez, Javier C., Ona Martin de la Torre, Júdit López Palomé, and Diego Redolar-Ripoll. 2022. "Effects of Caffeine Consumption on Attention Deficit Hyperactivity Disorder (ADHD) Treatment: A Systematic Review of Animal Studies" Nutrients 14, no. 4: 739. https://doi.org/10.3390/nu14040739
APA StyleVázquez, J. C., Martin de la Torre, O., López Palomé, J., & Redolar-Ripoll, D. (2022). Effects of Caffeine Consumption on Attention Deficit Hyperactivity Disorder (ADHD) Treatment: A Systematic Review of Animal Studies. Nutrients, 14(4), 739. https://doi.org/10.3390/nu14040739






