Probiotic Lactiplantibacillus plantarum Tana Isolated from an International Weightlifter Enhances Exercise Performance and Promotes Antifatigue Effects in Mice
Abstract
1. Introduction
2. Materials and Methods
2.1. Sample Preparation
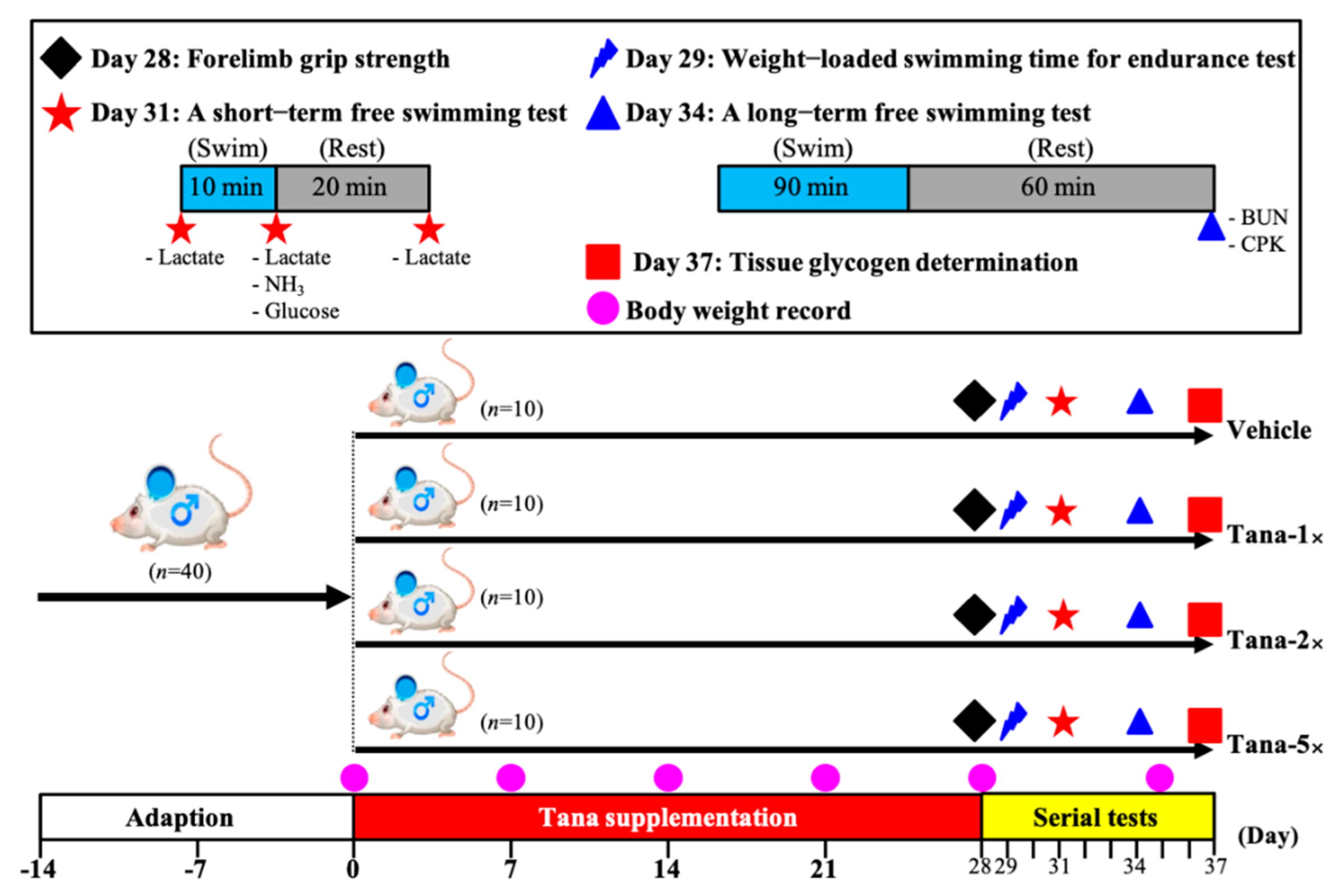
2.2. Experimental Design
2.3. Forelimb Grip Strength
2.4. Swimming Exercise Performance Test
2.5. Determination of Fatigue-Associated Serum Biomarkers
2.6. Clinical Biochemical Profiles
2.7. Visceral Tissue Weight, Histology Staining, and Glycogen Determination
2.8. Bacterial DNA Extraction and 16S rRNA Sequencing
2.9. Statistical Analysis
3. Results
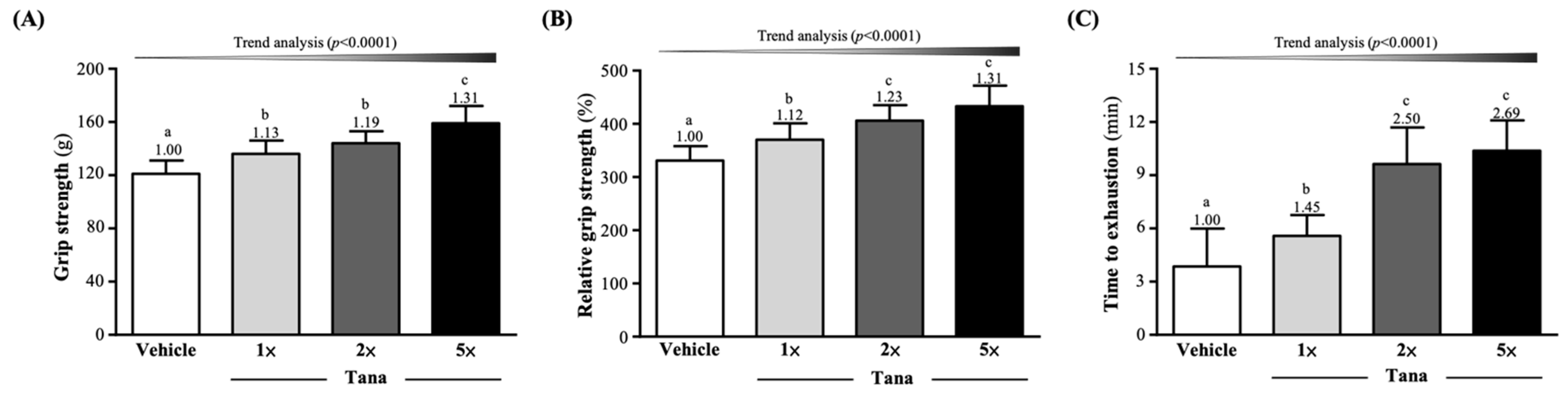
3.1. Effect of Tana Supplementation on Grip Strength and Endurance Exercise Performance
3.2. Effect of Tana Supplementation on Serum Lactate Levels after the 10 min Swim Test
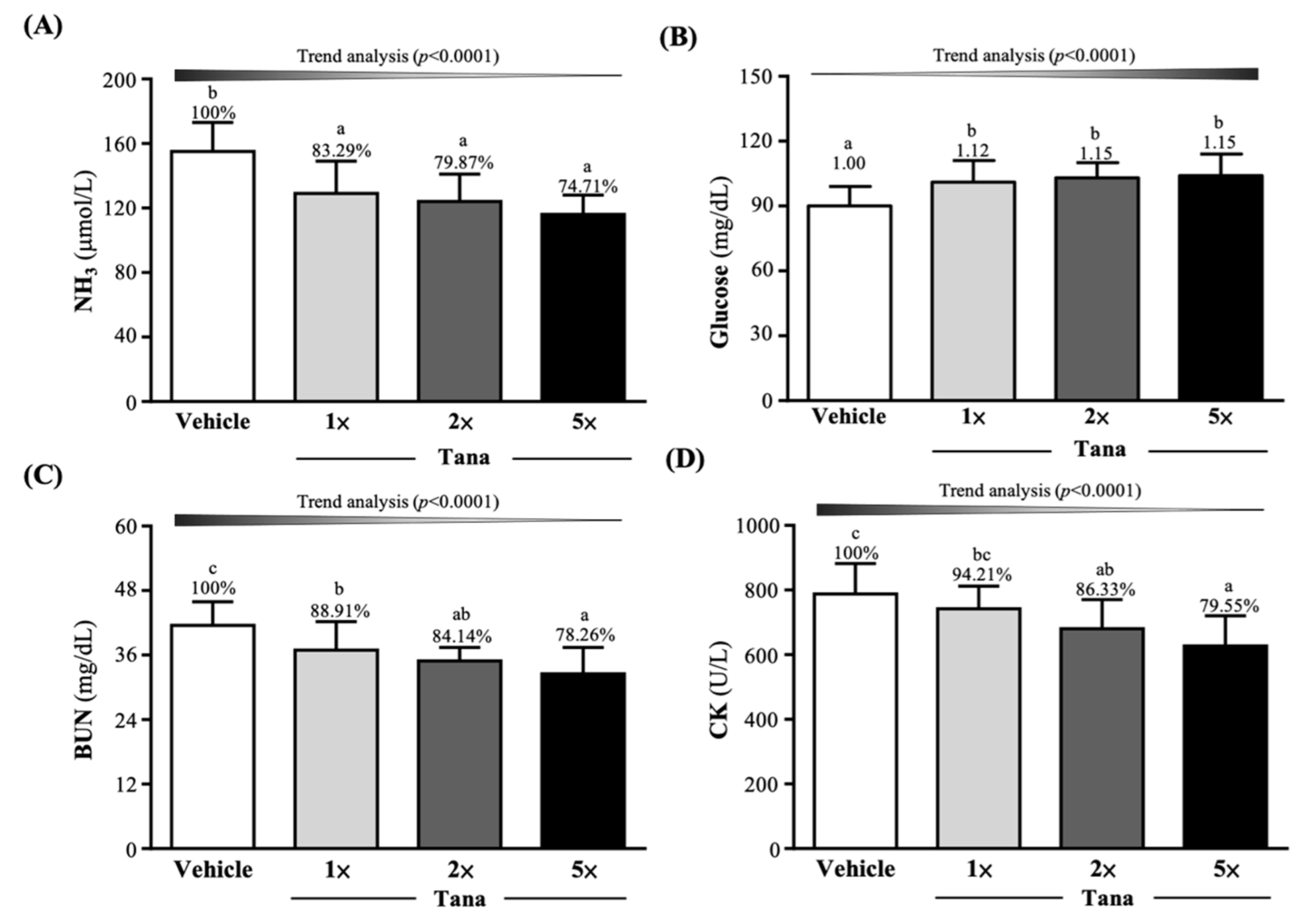
3.3. Effect of Tana Supplementation on Fatigue-Related Indexes after the 10 min Swim test or a 90 min Swim Test and 60 min Rest
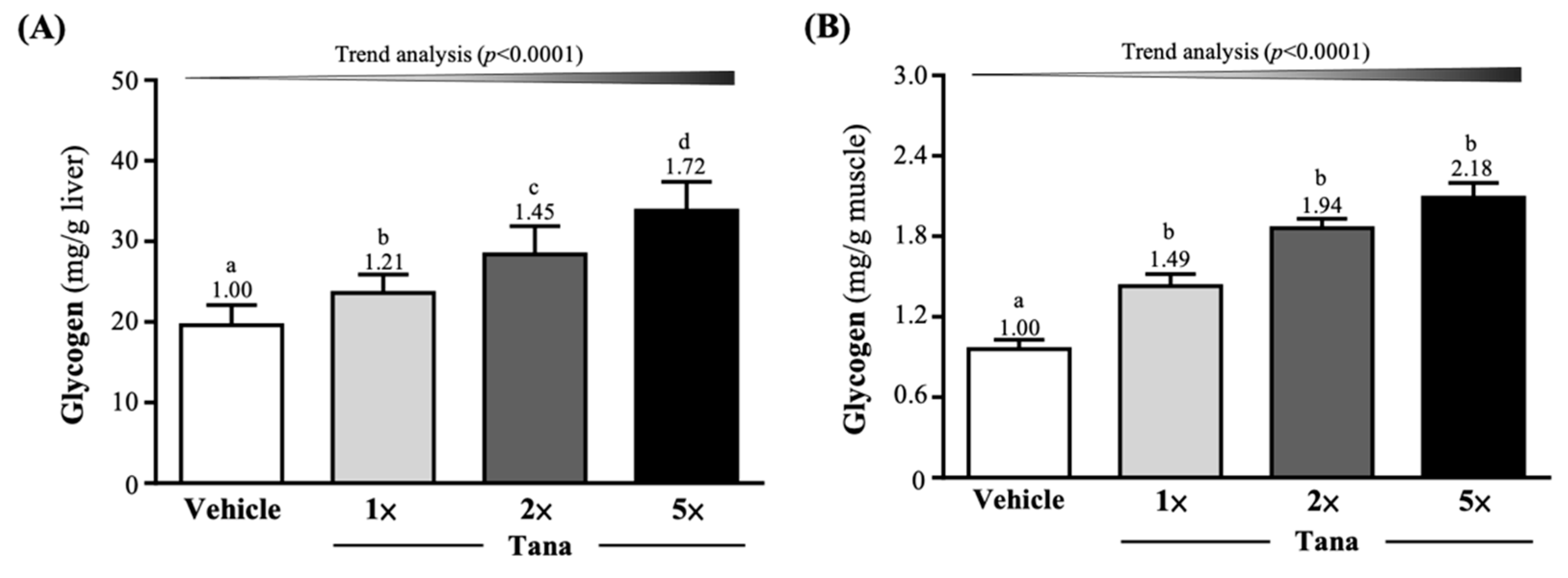
3.4. Effect of Tana Supplementation on Liver and Muscle Glycogen
3.5. General Characteristics of Mice with Tana Supplementation for Four Weeks
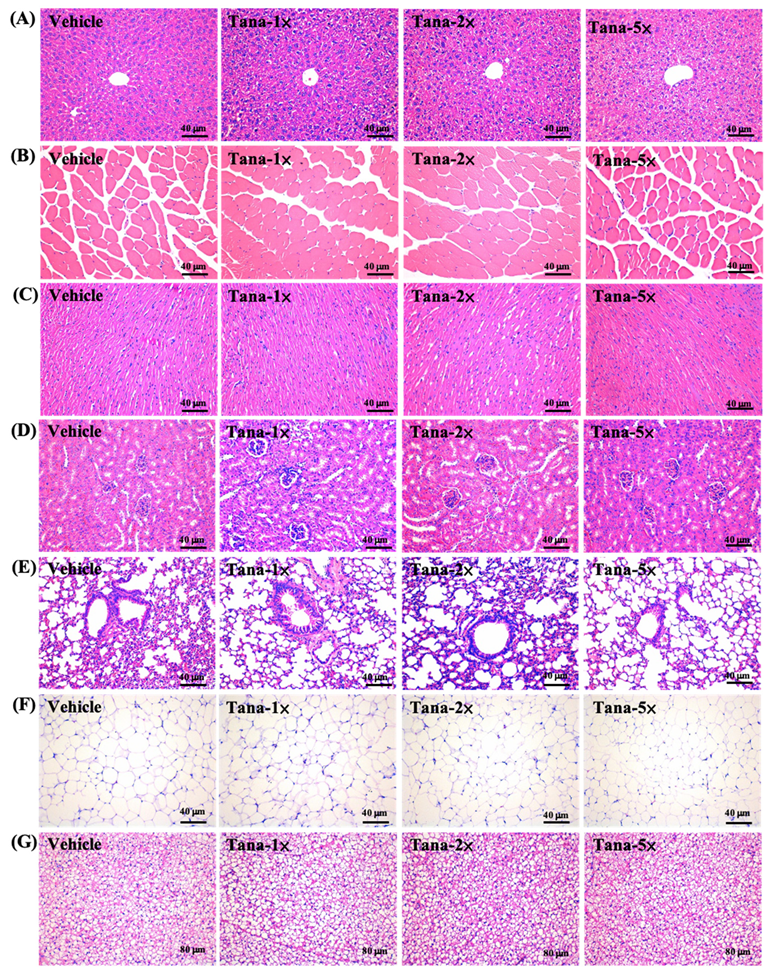
3.6. Effect of Tana Supplementation on Histopathology of Tissues and Biochemical Profiles at the End of the Study
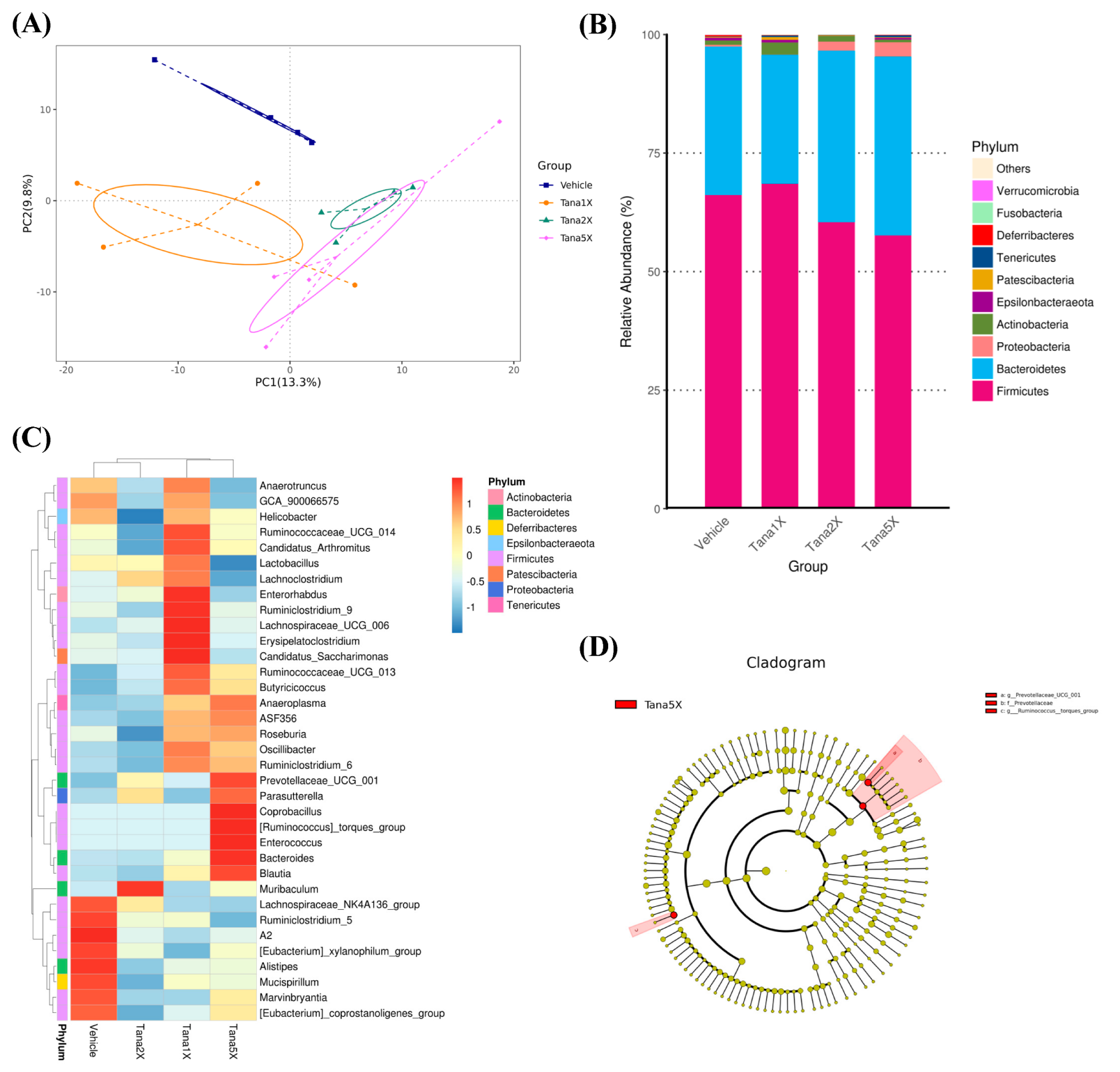
3.7. Effect of Tana Supplementation on Gut Microbiota
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Mohr, A.E.; Jäger, R.; Carpenter, K.C.; Kerksick, C.M.; Purpura, M.; Townsend, J.R.; West, N.P.; Black, K.; Gleeson, M.; Pyne, D.B.; et al. The athletic gut microbiota. J. Int. Soc. Sports Nutr. 2020, 17, 24. [Google Scholar] [CrossRef]
- Jandhyala, S.M.; Talukdar, R.; Subramanyam, C.; Vuyyuru, H.; Sasikala, M.; Nageshwar Reddy, D. Role of the normal gut microbiota. World J. Gastroenterol. 2015, 21, 8787–8803. [Google Scholar] [CrossRef] [PubMed]
- Milani, C.; Duranti, S.; Bottacini, F.; Casey, E.; Turroni, F.; Mahony, J.; Belzer, C.; Delgado Palacio, S.; Arboleya Montes, S.; Mancabelli, L.; et al. The First Microbial Colonizers of the Human Gut: Composition, Activities, and Health Implications of the Infant Gut Microbiota. Microbiol. Mol. Biol. Rev. 2017, 81. [Google Scholar] [CrossRef] [PubMed]
- Duffy, L.C.; Raiten, D.J.; Hubbard, V.S.; Starke-Reed, P. Progress and challenges in developing metabolic footprints from diet in human gut microbial cometabolism. J. Nutr. 2015, 145, 1123S–1130S. [Google Scholar] [CrossRef] [PubMed]
- Sakkas, H.; Bozidis, P.; Touzios, C.; Kolios, D.; Athanasiou, G.; Athanasopoulou, E.; Gerou, I.; Gartzonika, C. Nutritional Status and the Influence of the Vegan Diet on the Gut Microbiota and Human Health. Medicina 2020, 56, 88. [Google Scholar] [CrossRef]
- Allen, J.M.; Mailing, L.J.; Niemiro, G.M.; Moore, R.; Cook, M.D.; White, B.A.; Holscher, H.D.; Woods, J.A. Exercise Alters Gut Microbiota Composition and Function in Lean and Obese Humans. Med. Sci. Sports Exerc. 2018, 50, 747–757. [Google Scholar] [CrossRef]
- World Health Organization. Global Recommendations on Physical Activity for Health; World Health Organization: Geneva, Switzerland, 2010. [Google Scholar]
- Bressa, C.; Bailén-Andrino, M.; Pérez-Santiago, J.; González-Soltero, R.; Pérez, M.; Montalvo-Lominchar, M.G.; Maté-Muñoz, J.L.; Domínguez, R.; Moreno, D.; Larrosa, M. Differences in gut microbiota profile between women with active lifestyle and sedentary women. PLoS ONE 2017, 12, e0171352. [Google Scholar] [CrossRef]
- Clarke, S.F.; Murphy, E.F.; O’Sullivan, O.; Lucey, A.J.; Humphreys, M.; Hogan, A.; Hayes, P.; O’Reilly, M.; Jeffery, I.B.; Wood-Martin, R.; et al. Exercise and associated dietary extremes impact on gut microbial diversity. Gut 2014, 63, 1913–1920. [Google Scholar] [CrossRef]
- Barton, W.; Penney, N.C.; Cronin, O.; Garcia-Perez, I.; Molloy, M.G.; Holmes, E.; Shanahan, F.; Cotter, P.D.; O’Sullivan, O. The microbiome of professional athletes differs from that of more sedentary subjects in composition and particularly at the functional metabolic level. Gut 2018, 67, 625–633. [Google Scholar] [CrossRef] [PubMed]
- Pascale, A.; Marchesi, N.; Marelli, C.; Coppola, A.; Luzi, L.; Govoni, S.; Giustina, A.; Gazzaruso, C. Microbiota and metabolic diseases. Endocrine 2018, 61, 357–371. [Google Scholar] [CrossRef] [PubMed]
- Okamoto, T.; Morino, K.; Ugi, S.; Nakagawa, F.; Lemecha, M.; Ida, S.; Ohashi, N.; Sato, D.; Fujita, Y.; Maegawa, H. Microbiome potentiates endurance exercise through intestinal acetate production. Am. J. Physiol. Endocrinol. Metab. 2019, 316, E956–E966. [Google Scholar] [CrossRef] [PubMed]
- Nicholson, J.K.; Holmes, E.; Kinross, J.; Burcelin, R.; Gibson, G.; Jia, W.; Pettersson, S. Host-gut microbiota metabolic interactions. Science 2012, 336, 1262–1267. [Google Scholar] [CrossRef] [PubMed]
- Donohoe, D.R.; Garge, N.; Zhang, X.; Sun, W.; O’Connell, T.M.; Bunger, M.K.; Bultman, S.J. The microbiome and butyrate regulate energy metabolism and autophagy in the mammalian colon. Cell Metab. 2011, 13, 517–526. [Google Scholar] [CrossRef] [PubMed]
- Tung, Y.T.; Chen, Y.J.; Chuang, H.L.; Huang, W.C.; Lo, C.T.; Liao, C.C.; Huang, C.C. Characterization of the serum and liver proteomes in gut-microbiota-lacking mice. Int. J. Med. Sci. 2017, 14, 257–267. [Google Scholar] [CrossRef]
- Kerksick, C.M.; Wilborn, C.D.; Roberts, M.D.; Smith-Ryan, A.; Kleiner, S.M.; Jäger, R.; Collins, R.; Cooke, M.; Davis, J.N.; Galvan, E.; et al. ISSN exercise & sports nutrition review update: Research & recommendations. J. Int. Soc. Sports Nutr. 2018, 15, 38. [Google Scholar]
- Lamprecht, M.; Bogner, S.; Schippinger, G.; Steinbauer, K.; Fankhauser, F.; Hallstroem, S.; Schuetz, B.; Greilberger, J.F. Probiotic supplementation affects markers of intestinal barrier, oxidation, and inflammation in trained men; a randomized, double-blinded, placebo-controlled trial. J. Int. Soc. Sports Nutr. 2012, 9, 45. [Google Scholar] [CrossRef]
- Williams, N.T. Probiotics. Am. J. Health Syst. Pharm. 2010, 67, 449–458. [Google Scholar] [CrossRef]
- Lee, M.C.; Hsu, Y.J.; Chuang, H.L.; Hsieh, P.S.; Ho, H.H.; Chen, W.L.; Chiu, Y.S.; Huang, C.C. In Vivo Ergogenic Properties of the Bifidobacterium longum OLP-01 Isolated from a Weightlifting Gold Medalist. Nutrients 2019, 11, 2003. [Google Scholar] [CrossRef]
- Huang, W.C.; Lee, M.C.; Lee, C.C.; Ng, K.S.; Hsu, Y.J.; Tsai, T.Y.; Young, S.L.; Lin, J.S.; Huang, C.C. Effect of Lactobacillus plantarum TWK10 on Exercise Physiological Adaptation, Performance, and Body Composition in Healthy Humans. Nutrients 2019, 11, 2836. [Google Scholar] [CrossRef]
- Pyne, D.B.; West, N.P.; Cox, A.J.; Cripps, A.W. Probiotics supplementation for athletes—Clinical and physiological effects. Eur. J. Sport Sci. 2015, 15, 63–72. [Google Scholar] [CrossRef]
- George Kerry, R.; Patra, J.K.; Gouda, S.; Park, Y.; Shin, H.S.; Das, G. Benefaction of probiotics for human health: A review. J. Food Drug Anal. 2018, 26, 927–939. [Google Scholar] [CrossRef] [PubMed]
- Versalovic, J. The human microbiome and probiotics: Implications for pediatrics. Ann. Nutr. Metab. 2013, 63 (Suppl. S2), 42–52. [Google Scholar] [CrossRef] [PubMed]
- Vemuri, R.; Shinde, T.; Shastri, M.D.; Perera, A.P.; Tristram, S.; Martoni, C.J.; Gundamaraju, R.; Ahuja, K.D.K.; Ball, M.; Eri, R. A human origin strain Lactobacillus acidophilus DDS-1 exhibits superior in vitro probiotic efficacy in comparison to plant or dairy origin probiotics. Int. J. Med. Sci. 2018, 15, 840–848. [Google Scholar] [CrossRef] [PubMed]
- Nagpal, R.; Wang, S.; Ahmadi, S.; Hayes, J.; Gagliano, J.; Subashchandrabose, S.; Kitzman, D.W.; Becton, T.; Read, R.; Yadav, H. Human-origin probiotic cocktail increases short-chain fatty acid production via modulation of mice and human gut microbiome. Sci. Rep. 2018, 8, 12649. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.M.; Wei, L.; Chiu, Y.S.; Hsu, Y.J.; Tsai, T.Y.; Wang, M.F.; Huang, C.C. Lactobacillus plantarum TWK10 Supplementation Improves Exercise Performance and Increases Muscle Mass in Mice. Nutrients 2016, 8, 205. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, K.; Fujimoto, J.; Sasamoto, M.; Dugersuren, J.; Tumursuh, T.; Demberel, S. Diversity of lactic acid bacteria and yeasts in Airag and Tarag, traditional fermented milk products of Mongolia. World J. Microbio. Biotechnol. 2008, 24, 1313–1325. [Google Scholar] [CrossRef]
- Naser, S.M.; Thompson, F.L.; Hoste, B.; Gevers, D.; Dawyndt, P.; Vancanneyt, M.; Swings, J. Application of multilocus sequence analysis (MLSA) for rapid identification of Enterococcus species based on rpoA and pheS genes. Microbiology 2005, 151, 2141–2150. [Google Scholar] [CrossRef] [PubMed]
- Miyake, T.; Watanabe, K.; Watanabe, T.; Oyaizu, H. Phylogenetic analysis of the genus Bifidobacterium and related genera based on 16S rDNA sequences. Microbiol. Immunol. 1998, 42, 661–667. [Google Scholar] [CrossRef] [PubMed]
- Saitou, N.; Nei, M. The neighbor-joining method: A new method for reconstructing phylogenetic trees. Mol. Biol. Evol. 1987, 4, 406–425. [Google Scholar]
- Thompson, J.D.; Gibson, T.J.; Plewniak, F.; Jeanmougin, F.; Higgins, D.G. The CLUSTAL_X windows interface: Flexible strategies for multiple sequence alignment aided by quality analysis tools. Nucleic Acids Res. 1997, 25, 4876–4882. [Google Scholar] [CrossRef]
- Felsenstein, J. Confidence limits on phylogenies: An approach using the bootstrap. Evolution 1985, 39, 783–791. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Stecher, G.; Tamura, K. MEGA7: Molecular evolutionary genetics analysis version 7.0 for bigger datasets. Mol. Biol. Evol. 2016, 33, 1870–1874. [Google Scholar] [CrossRef] [PubMed]
- Kimura, M. A simple method for estimating evolutionary rates of base substitutions through comparative studies of nucleotide sequences. J. Mol. Evol. 1980, 16, 111–120. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.C.; Hsu, Y.J.; Lin, Y.Q.; Chen, L.N.; Chen, M.T.; Huang, C.C. Effects of Perch Essence Supplementation on Improving Exercise Performance and Anti-Fatigue in Mice. Int. J. Environ. Res. Public Health 2022, 19, 1155. [Google Scholar] [CrossRef]
- Ho, C.S.; Tung, Y.T.; Kung, W.M.; Huang, W.C.; Leung, W.K.; Huang, C.C.; Wu, J.H. Effect of Coriolus versicolor Mycelia Extract on Exercise Performance and Physical Fatigue in Mice. Int. J. Med. Sci. 2017, 14, 1110–1117. [Google Scholar] [CrossRef] [PubMed]
- Huang, S.W.; Hsu, Y.J.; Lee, M.C.; Li, H.S.; Yeo, P.C.W.; Lim, A.L.; Huang, C.C. In Vitro and In Vivo Functional Characterization of Essence of Chicken as An Ergogenic Aid. Nutrients 2018, 10, 1943. [Google Scholar] [CrossRef]
- Yeh, W.L.; Hsu, Y.J.; Ho, C.S.; Ho, H.H.; Kuo, Y.W.; Tsai, S.Y.; Huang, C.C.; Lee, M.C. Lactobacillus plantarum PL-02 Supplementation Combined With Resistance Training Improved Muscle Mass, Force, and Exercise Performance in Mice. Front. Nutr. 2022, 9, 896503. [Google Scholar] [CrossRef]
- Hsu, Y.J.; Huang, W.C.; Lin, J.S.; Chen, Y.M.; Ho, S.T.; Huang, C.C.; Tung, Y.T. Kefir Supplementation Modifies Gut Microbiota Composition, Reduces Physical Fatigue, and Improves Exercise Performance in Mice. Nutrients 2018, 10, 862. [Google Scholar] [CrossRef]
- Jäger, R.; Mohr, A.E.; Carpenter, K.C.; Kerksick, C.M.; Purpura, M.; Moussa, A.; Townsend, J.R.; Lamprecht, M.; West, N.P.; Black, K.; et al. International Society of Sports Nutrition Position Stand: Probiotics. J. Int. Soc. Sports Nutr. 2019, 16, 62. [Google Scholar] [CrossRef]
- Sánchez, B.; Delgado, S.; Blanco-Míguez, A.; Lourenço, A.; Gueimonde, M.; Margolles, A. Probiotics, gut microbiota, and their influence on host health and disease. Mol. Nutr Food Res. 2017, 61, 1600240. [Google Scholar] [CrossRef]
- Marttinen, M.; Ala-Jaakkola, R.; Laitila, A.; Lehtinen, M.J. Gut Microbiota, Probiotics and Physical Performance in Athletes and Physically Active Individuals. Nutrients 2020, 12, 2936. [Google Scholar] [CrossRef] [PubMed]
- LeBlanc, J.G.; Chain, F.; Martín, R.; Bermúdez-Humarán, L.G.; Courau, S.; Langella, P. Beneficial effects on host energy metabolism of short-chain fatty acids and vitamins produced by commensal and probiotic bacteria. Microb. Cell Fact. 2017, 16, 79. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.Q.; Zhao, T.T.; Gui, D.K.; Gao, C.L.; Gu, J.L.; Gan, W.J.; Huang, W.; Xu, Y.; Zhou, H.; Chen, W.N.; et al. Sodium Butyrate Improves Liver Glycogen Metabolism in Type 2 Diabetes Mellitus. J. Agric. Food Chem. 2019, 67, 7694–7705. [Google Scholar] [CrossRef] [PubMed]
- Samuel, B.S.; Shaito, A.; Motoike, T.; Rey, F.E.; Backhed, F.; Manchester, J.K.; Hammer, R.E.; Williams, S.C.; Crowley, J.; Yanagisawa, M.; et al. Effects of the gut microbiota on host adiposity are modulated by the short-chain fatty-acid binding G protein-coupled receptor, Gpr41. Proc. Natl. Acad. Sci. USA 2008, 105, 16767–16772. [Google Scholar] [CrossRef] [PubMed]
- Shirouchi, B.; Nagao, K.; Umegatani, M.; Shiraishi, A.; Morita, Y.; Kai, S.; Yanagita, T.; Ogawa, A.; Kadooka, Y.; Sato, M. Probiotic Lactobacillus gasseri SBT2055 improves glucose tolerance and reduces body weight gain in rats by stimulating energy expenditure. Br. J. Nutr. 2016, 116, 451–458. [Google Scholar] [CrossRef]
- Niibo, M.; Shirouchi, B.; Umegatani, M.; Morita, Y.; Ogawa, A.; Sakai, F.; Kadooka, Y.; Sato, M. Probiotic Lactobacillus gasseri SBT2055 improves insulin secretion in a diabetic rat model. J. Dairy Sci. 2019, 102, 997–1006. [Google Scholar] [CrossRef]
- Tsintzas, K.; Williams, C. Human muscle glycogen metabolism during exercise. Effect of carbohydrate supplementation. Sports Med. 1998, 25, 7–23. [Google Scholar] [CrossRef]
- Bergström, J.; Hultman, E. A study of the glycogen metabolism during exercise in man. Scand. J. Clin. Lab. Investig. 1967, 19, 218–228. [Google Scholar] [CrossRef]
- Shing, C.M.; Peake, J.M.; Lim, C.L.; Briskey, D.; Walsh, N.P.; Fortes, M.B.; Ahuja, K.D.; Vitetta, L. Effects of probiotics supplementation on gastrointestinal permeability, inflammation and exercise performance in the heat. Eur. J. Appl. Physiol. 2014, 114, 93–103. [Google Scholar] [CrossRef]
- Grosicki, G.J.; Fielding, R.A.; Lustgarten, M.S. Gut Microbiota Contribute to Age-Related Changes in Skeletal Muscle Size, Composition, and Function: Biological Basis for a Gut-Muscle Axis. Calcif. Tissue Int. 2018, 102, 433–442. [Google Scholar] [CrossRef]
- Chen, L.; Nelson, D.R.; Zhao, Y.; Cui, Z.; Johnston, J.A. Relationship between muscle mass and muscle strength, and the impact of comorbidities: A population-based, cross-sectional study of older adults in the United States. BMC Geriatr. 2013, 13, 74. [Google Scholar] [CrossRef] [PubMed]
- Petersen, L.M.; Bautista, E.J.; Nguyen, H.; Hanson, B.M.; Chen, L.; Lek, S.H.; Sodergren, E.; Weinstock, G.M. Community characteristics of the gut microbiomes of competitive cyclists. Microbiome 2017, 5, 98. [Google Scholar] [CrossRef] [PubMed]
- Kovatcheva-Datchary, P.; Nilsson, A.; Akrami, R.; Lee, Y.S.; De Vadder, F.; Arora, T.; Hallen, A.; Martens, E.; Björck, I.; Bäckhed, F. Dietary Fiber-Induced Improvement in Glucose Metabolism Is Associated with Increased Abundance of Prevotella. Cell Metab. 2015, 22, 971–982. [Google Scholar] [CrossRef]
- Negro, M.; Giardina, S.; Marzani, B.; Marzatico, F. Branched-chain amino acid supplementation does not enhance athletic performance but affects muscle recovery and the immune system. J. Sports Med. Phys. Fitness. 2008, 48, 347–351. [Google Scholar] [PubMed]
- Halson, S.L.; Bridge, M.W.; Meeusen, R.; Busschaert, B.; Gleeson, M.; Jones, D.A.; Jeukendrup, A.E. Time course of performance changes and fatigue markers during intensified training in trained cyclists. J. Appl. Physiol. 2002, 93, 947–956. [Google Scholar] [CrossRef]
- Proia, P.; Di Liegro, C.M.; Schiera, G.; Fricano, A.; Di Liegro, I. Lactate as a Metabolite and a Regulator in the Central Nervous System. Int. J. Mol. Sci. 2016, 17, 1450. [Google Scholar] [CrossRef]
- Chen, S.; Minegishi, Y.; Hasumura, T.; Shimotoyodome, A.; Ota, N. Involvement of ammonia metabolism in the improvement of endurance performance by tea catechins in mice. Sci. Rep. 2020, 10, 6065. [Google Scholar] [CrossRef]
- Bush, J.A.; Wu, G.; Suryawan, A.; Nguyen, H.V.; Davis, T.A. Somatotropin-induced amino acid conservation in pigs involves differential regulation of liver and gut urea cycle enzyme activity. J. Nutr. 2002, 132, 59–67. [Google Scholar] [CrossRef][Green Version]
- Scheiman, J.; Luber, J.M.; Chavkin, T.A.; MacDonald, T.; Tung, A.; Pham, L.D.; Wibowo, M.C.; Wurth, R.C.; Punthambaker, S.; Tierney, B.T.; et al. Meta-omics analysis of elite athletes identifies a performance-enhancing microbe that functions via lactate metabolism. Nat. Med. 2019, 25, 1104–1109. [Google Scholar] [CrossRef]
- Liu, J.; Lkhagva, E.; Chung, H.J.; Kim, H.J.; Hong, S.T. The Pharmabiotic Approach to Treat Hyperammonemia. Nutrients 2018, 10, 140. [Google Scholar] [CrossRef]
- Stojanov, S.; Berlec, A.; Štrukelj, B. The Influence of Probiotics on the Firmicutes/Bacteroidetes Ratio in the Treatment of Obesity and Inflammatory Bowel disease. Microorganisms 2020, 8, 1715. [Google Scholar] [CrossRef] [PubMed]
- Evans, C.C.; LePard, K.J.; Kwak, J.W.; Stancukas, M.C.; Laskowski, S.; Dougherty, J.; Moulton, L.; Glawe, A.; Wang, Y.; Leone, V.; et al. Exercise prevents weight gain and alters the gut microbiota in a mouse model of high fat diet-induced obesity. PLoS ONE 2014, 9, e92193. [Google Scholar] [CrossRef] [PubMed]
| Groups | Vehicle | Tana-1× | Tana-2× | Tana-5× | Trend Analysis | |
|---|---|---|---|---|---|---|
| Time Point | Lactate (mmol/L) | |||||
| Before swimming (A) | 4.33 ± 0.66 a | 4.38 ± 0.36 a | 4.39 ± 0.67 a | 4.40 ± 0.62 a | 0.9567 | |
| After swimming (B) | 7.45 ± 0.65 c | 6.79 ± 0.55 b | 6.41 ± 0.51 ab | 5.92 ± 0.59 a | <0.0001 * | |
| After a 20 min resting (C) | 6.20 ± 0.74 c | 5.38 ± 0.49 b | 5.12 ± 0.36 ab | 4.72 ± 0.24 a | <0.0001 * | |
| Rates of lactate production and clearance | ||||||
| Production rate = B/A | 1.74 ± 0.13 b | 1.55 ± 0.07 ab | 1.48 ± 0.16 a | 1.35 ± 0.08 a | 0.0003 * | |
| Clearance rate = (B − C)/B | 0.17 ± 0.04 a | 0.21 ± 0.03 a | 0.20 ± 0.02 a | 0.20 ± 0.04 a | 0.5448 | |
| Characteristics | Vehicle | Tana-1× | Tana-2× | Tana-5× | Trend Analysis |
|---|---|---|---|---|---|
| Initial BW (g) | 34.7 ± 1.1 a | 34.8 ± 1.3 a | 34.7 ± 1.5 a | 34.7 ± 1.1 a | 0.9693 |
| Final BW (g) | 38.3 ± 1.3 | 38.5 ± 2.2 a | 38.2 ± 0.8 a | 38.4 ± 1.7 a | 0.8048 |
| Water intake (mL/mouse/day) | 9.3 ± 1.8 a | 9.4 ± 2.1 a | 9.4 ± 1.3 a | 9.3 ± 1.6 a | 0.4960 |
| Diet intake (g/mouse/day) | 7.4 ± 1.4 a | 7.3 ± 1.1 a | 7.2 ± 1.6 a | 7.2 ± 1.1 a | 0.2722 |
| Liver (g) | 2.22 ± 0.09 a | 2.24 ± 0.07 a | 2.21 ± 0.06 a | 2.23 ± 0.10 a | 0.7482 |
| Muscle (g) | 0.36 ± 0.01 a | 0.38 ± 0.06 ab | 0.38 ± 0.02 ab | 0.40 ± 0.03 b | 0.0048 * |
| Kidney (g) | 0.63 ± 0.06 a | 0.64 ± 0.05 a | 0.63 ± 0.04 a | 0.63 ± 0.04 a | 0.8507 |
| Heart (g) | 0.20 ± 0.02 a | 0.20 ± 0.02 a | 0.20 ± 0.02 a | 0.20 ± 0.02 a | 0.4716 |
| Lung (g) | 0.23 ± 0.01 a | 0.23 ± 0.02 a | 0.23 ± 0.02 a | 0.23 ± 0.01 a | 0.9368 |
| EFP (g) | 0.35 ± 0.08 b | 0.34 ± 0.07 ab | 0.34 ± 0.03 ab | 0.29 ± 0.06 a | 0.0593 |
| BAT (g) | 0.11 ± 0.03 a | 0.10 ± 0.02 a | 0.11 ± 0.02 a | 0.11 ± 0.03 a | 0.8057 |
| Cecum (g) | 1.02 ± 0.11 a | 1.03 ± 0.19 | 1.04 ± 0.11 a | 1.13 ± 0.13 a | 0.0542 |
| Relative liver weight (%) | 5.80 ± 0.07 a | 5.82 ± 0.18 a | 5.79 ± 0.05 a | 5.81 ± 0.11 a | 0.9332 |
| Relative muscle weight (%) | 0.95 ± 0.01 a | 0.99 ± 0.09 ab | 0.99 ± 0.03 ab | 1.04 ± 0.05 b | <0.0001 * |
| Relative kidney weight (%) | 1.64 ± 0.10 a | 1.65 ± 0.06 a | 1.65 ± 0.08 a | 1.65 ± 0.03 a | 0.8840 |
| Relative heart weight (%) | 0.53 ± 0.05 a | 0.52 ± 0.05 a | 0.52 ± 0.04 a | 0.52 ± 0.04 a | 0.6946 |
| Relative lung weight (%) | 0.59 ± 0.03 a | 0.59 ± 0.03 a | 0.59 ± 0.03 a | 0.59 ± 0.04 a | 0.6745 |
| Relative EFP weight (%) | 0.90 ± 0.19 b | 0.88 ± 0.15 b | 0.88 ± 0.07 b | 0.74 ± 0.13 a | 0.0182 * |
| Relative BAT weight (%) | 0.28 ± 0.06 a | 0.26 ± 0.03 a | 0.27 ± 0.05 a | 0.28 ± 0.06 a | 0.7992 |
| Relative cecum weight (%) | 2.66 ± 0.21 a | 2.65 ± 0.35 a | 2.71 ± 0.23 a | 2.95 ± 0.21 b | 0.0054 * |
| Parameter | Vehicle | Tana-1× | Tana-2× | Tana-5× | Trend Analysis |
|---|---|---|---|---|---|
| AST (U/L) | 93 ± 9 a | 92 ± 8 a | 93 ± 10 a | 93 ± 10 a | 0.6056 |
| ALT (U/L) | 55 ± 10 a | 52 ± 10 a | 52 ± 8 a | 52 ± 11 a | 0.6537 |
| CK (U/L) | 250 ± 45 a | 230 ± 43 a | 229 ± 33 a | 224 ± 33 a | 0.3291 |
| GLU (mg/dL) | 182 ± 20 a | 181 ± 24 a | 181 ± 29 a | 181 ± 33 a | 0.7611 |
| CREA (mg/dL) | 0.41 ± 0.03 a | 0.41 ± 0.02 a | 0.40 ± 0.05 a | 0.40 ± 0.03 a | 0.4376 |
| BUN (mg/dL) | 25.6 ± 2.4 a | 25.8 ± 1.4 a | 25.7 ± 1.4 a | 25.5 ± 1.8 a | 0.7100 |
| UA (mg/dL) | 2.4 ± 0.6 a | 2.6 ± 0.6 a | 2.6 ± 0.5 a | 2.5 ± 0.6 a | 0.8459 |
| TC (mg/dL) | 143 ± 14 a | 144 ± 17 a | 147 ± 11 a | 144 ± 11 a | 0.5140 |
| TG (mg/dL) | 166 ± 24 a | 164 ± 26 a | 167 ± 16 a | 162 ± 18 a | 0.5624 |
| ALB (g/dL) | 3.5 ± 0.1 a | 3.5 ± 0.1 a | 3.5 ± 0.1 a | 3.5 ± 0.2 a | 0.5982 |
| TP (g/dL) | 6.0 ± 0.2 a | 6.1 ± 0.1 a | 6.0 ± 0.2 a | 6.1 ± 0.3 a | 0.1561 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, M.-C.; Chen, M.-J.; Huang, H.-W.; Wu, W.-K.; Lee, Y.-W.; Kuo, H.-C.; Huang, C.-C. Probiotic Lactiplantibacillus plantarum Tana Isolated from an International Weightlifter Enhances Exercise Performance and Promotes Antifatigue Effects in Mice. Nutrients 2022, 14, 3308. https://doi.org/10.3390/nu14163308
Lee M-C, Chen M-J, Huang H-W, Wu W-K, Lee Y-W, Kuo H-C, Huang C-C. Probiotic Lactiplantibacillus plantarum Tana Isolated from an International Weightlifter Enhances Exercise Performance and Promotes Antifatigue Effects in Mice. Nutrients. 2022; 14(16):3308. https://doi.org/10.3390/nu14163308
Chicago/Turabian StyleLee, Mon-Chien, Ming-Ju Chen, Hsiao-Wen Huang, Wei-Kai Wu, Yi-Wei Lee, Hsing-Chun Kuo, and Chi-Chang Huang. 2022. "Probiotic Lactiplantibacillus plantarum Tana Isolated from an International Weightlifter Enhances Exercise Performance and Promotes Antifatigue Effects in Mice" Nutrients 14, no. 16: 3308. https://doi.org/10.3390/nu14163308
APA StyleLee, M.-C., Chen, M.-J., Huang, H.-W., Wu, W.-K., Lee, Y.-W., Kuo, H.-C., & Huang, C.-C. (2022). Probiotic Lactiplantibacillus plantarum Tana Isolated from an International Weightlifter Enhances Exercise Performance and Promotes Antifatigue Effects in Mice. Nutrients, 14(16), 3308. https://doi.org/10.3390/nu14163308








