Effects of Cannabidiol Supplementation on Skeletal Muscle Regeneration after Intensive Resistance Training
Abstract
:1. Introduction
2. Materials and Methods
2.1. Participants
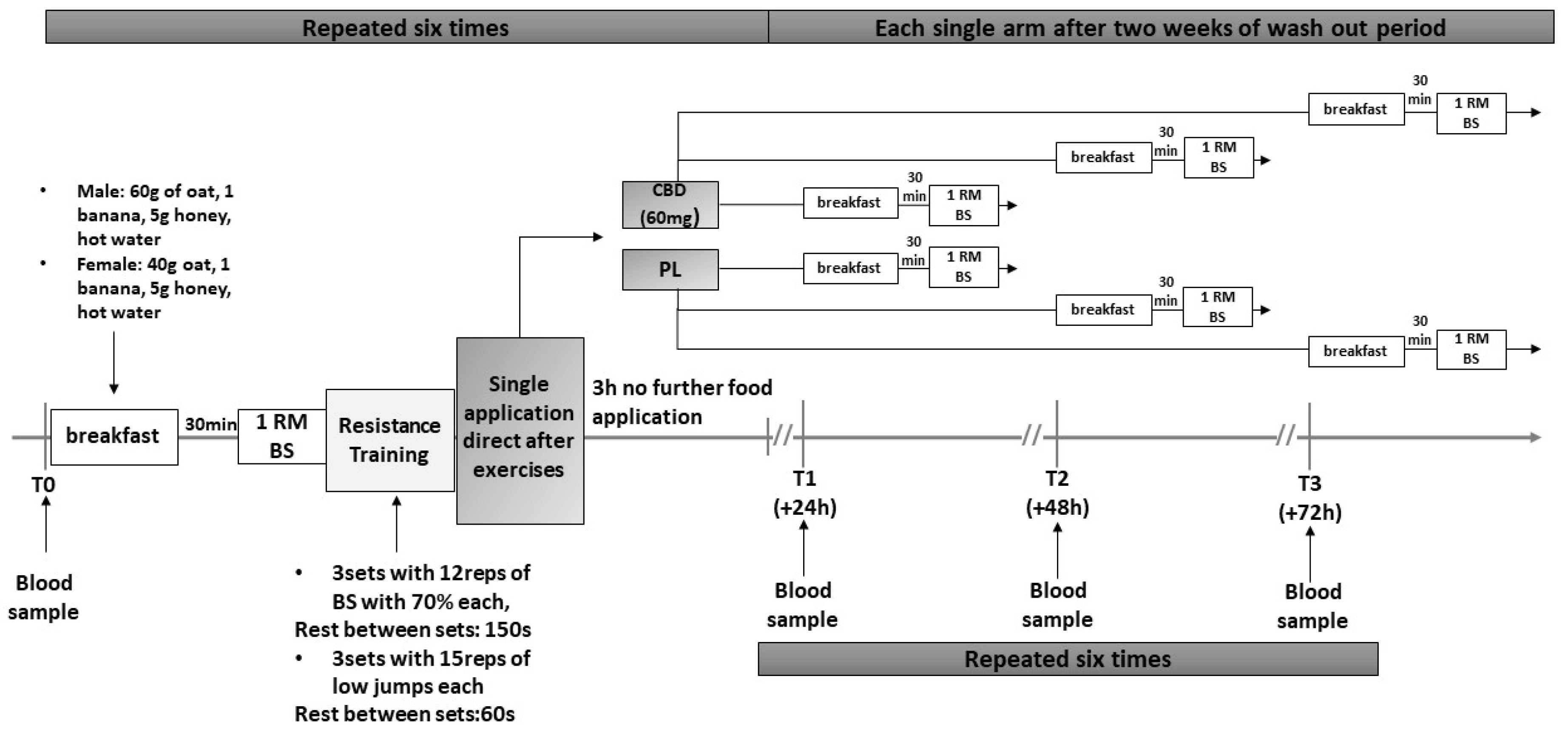
2.2. Study Design
2.3. Testing Protocol
2.4. Skeletal Muscle Serum Creatine Kinase (CK) and Myoglobin (Myo)
2.5. Strength and Power 1RM Back Squat (1RM) and Counter Movement Jump (CMJ)
2.6. Training Protocol
2.7. Supplementation
2.8. Statistical Analysis
3. Results
3.1. Physiological Adaptations over Time (Random Effects)
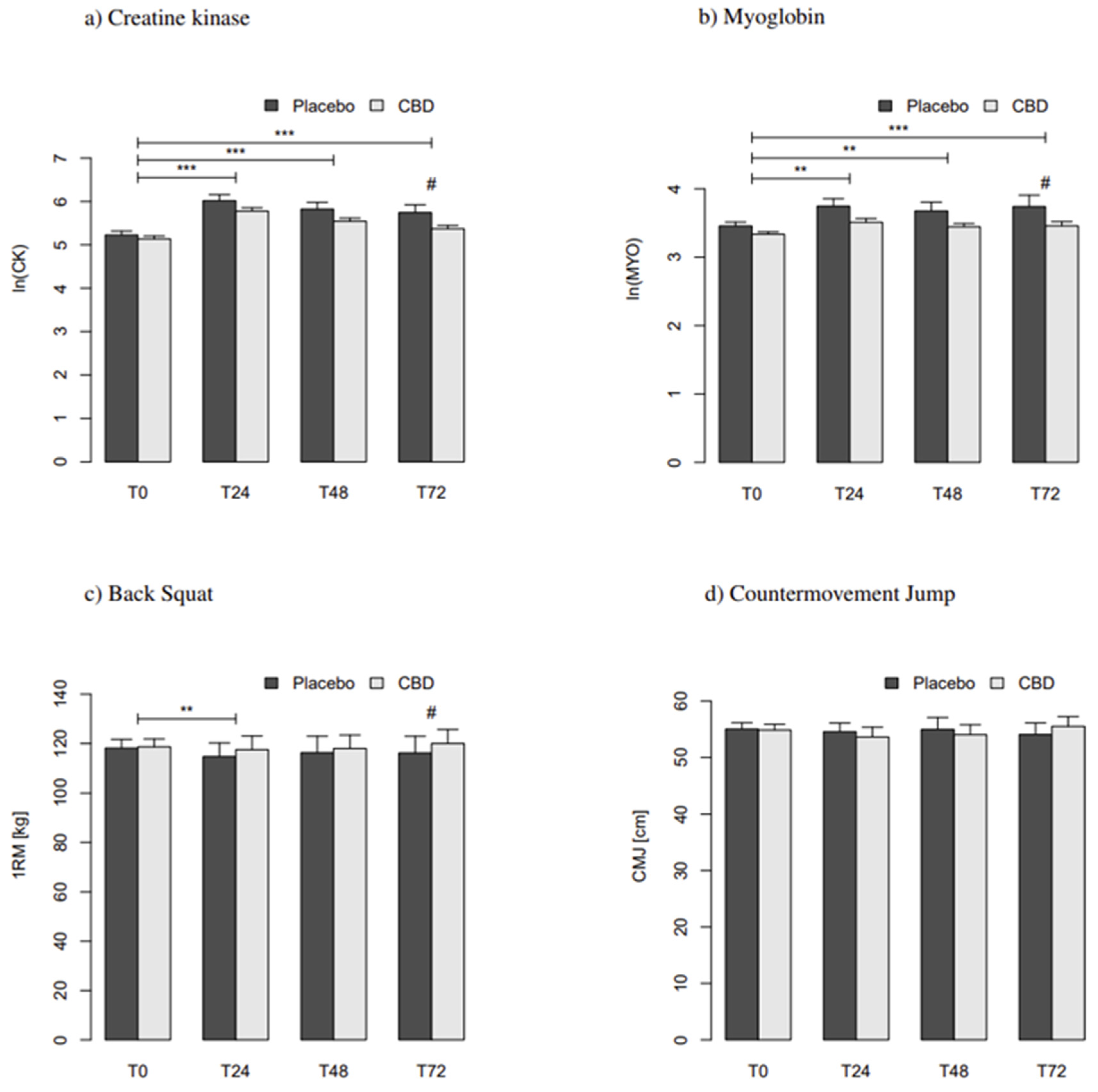
3.2. Effects on Skeletal Muscle Damage
3.3. Effects on Performance
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| 1RM | One-Repetition Maximum |
| BS | Back Squat |
| CMJ | Countermovement Jump |
| CK | Skeletal Muscle Specific Creatine Kinase |
| Myo | Myoglobin |
| ln | Logarithm |
| SEM | Standard Error of the Mean |
| ES | Effect Size |
| CBD | Cannabidiol |
| PLA | Placebo |
| ∆9-THC | ∆9-tetrahydrocannabinol |
| h | hours |
| FDA | U.S. Food and Drug Administration |
| WADA | World Anti-Doping Agency |
| NCSA | National Strength and Conditioning Association |
| AIC | Akaike Information Criterion |
| ∆ | Delta |
| t | time |
| NSAIDs | Non-Steroidal Anti-Inflammatory Drugs |
| NFL | National Footbal League |
| RI | Recovery Interval |
| LME | Linear Mixed Effect Model |
| DF | Degrees of Freedom |
| JAK | Janus Kinase |
| STAT | Signal Transducer and Activator of Transcription |
| EU | European Union |
References
- Isenmann, E.; Trittel, L.; Diel, P. The effects of alpha lipoic acid on muscle strength recovery after a single and a short-term chronic supplementation—A study in healthy well-trained individuals after intensive resistance and endurance training. J. Int. Soc. Sports Nutr. 2020, 17, 1–13. [Google Scholar] [CrossRef]
- Isenmann, E.; Blume, F.; Bizjak, D.; Hundsdörfer, V.; Pagano, S.; Schibrowski, S.; Simon, W.; Schmandra, L.; Diel, P. Comparison of pro-regenerative effects of carbohydrates and protein administrated by shake and non-macro-nutrient matched food items on the skeletal muscle after acute endurance exercise. Nutrients 2019, 11, 744. [Google Scholar] [CrossRef] [Green Version]
- Janning, M.; Isenmann, E.; Diel, P. Comparison of pro-regenerative nutritive actions given by foodstuff or shake after strength exercise. Ger. J. Sports Med. 2020, 71, 9. [Google Scholar] [CrossRef]
- Bloch, W. Muscle healing: Physiology and adverse factors. In Muscle Injuries in Sports; Müller-Wohlfahrt, H.W., Ueblacker, P., Hänsel, L., Garrett, W.E., Eds.; Thieme: Stuttgart, Germany, 2013. [Google Scholar]
- Koh, T.J. Physiology and mechanism of skeletal muscle damage. In Skeletal Muscle Damage and Repair; Tiidus, P.M., Ed.; Human Kinetics: Champaign, IL, USA, 2008. [Google Scholar]
- Sayers, S.P.; Hubal, M.J. Histological, chemical, and functional manifestation of muscle damage. In Skeletal Muscle Damage and Repair; Tiidus, P.M., Ed.; Human Kinetics: Champaign, IL, USA, 2008. [Google Scholar]
- Lieberman, H.R.; Marriott, B.P.; Williams, C.; Judelson, D.A.; Glickman, E.L.; Geiselman, P.J.; Dotson, L.; Mahoney, C.R. Patterns of dietary supplement use among college students. Clin. Nutr. 2015, 34, 976–985. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- National Study on Substance Use Habits of College Student-Athletes. 2018. Available online: https://www.wada-ama.org/sites/default/files/prohibited_list_2018_summary_of_modifications_en.pdf (accessed on 10 July 2021).
- Tscholl, P.; Alonso, J.M.; Dollé, G.; Junge, A.; Dvorak, J. The use of drugs and nutritional supplements in top-level track and field athletes. Am. J. Sports Med. 2010, 38, 133–140. [Google Scholar] [CrossRef] [PubMed]
- Zeiger, J.S.; Silvers, W.S.; Fleegler, E.M.; Zeiger, R.S. Cannabis use in active athletes: Behaviors related to subjective effects. PLoS ONE 2019, 14, e0218998. [Google Scholar] [CrossRef]
- Ware, M.A.; Jensen, D.; Barrette, A.; Vernec, A.; Derman, W. Cannabis and the health and performance of the elite athlete. Clin. J. Sport Med. 2018, 28, 480–484. [Google Scholar] [CrossRef]
- Docter, S.; Khan, M.; Gohal, C.; Ravi, B.; Bhandari, M.; Gandhi, R.; Leroux, T. Cannabis use and sport: A systematic review. Sports Health A Multidiscip. Approach 2020, 12, 189–199. [Google Scholar] [CrossRef]
- Hartsel, J.A.; Eades, J.; Hickory, B.; Makriyannis, A. Cannabis sativa and Hemp. In Nutraceuticals; Gupta, R.C., Ed.; Academic Press: Boston, CA, USA, 2016; Chapter 53; pp. 735–754. [Google Scholar] [CrossRef]
- Ligresti, A.; Petrocellis, L.D.; Marzo, V.D. From phytocannabinoids to cannabinoid receptors and endocannabinoids: Pleiotropic physiological and pathological roles through complex pharmacology. Physiol. Rev. 2016, 96, 1593–1659. [Google Scholar] [CrossRef] [Green Version]
- Laprairie, R.B.; Bagher, A.M.; Kelly, M.E.M.; Denovan-Wright, E.M. Cannabidiol is a negative allosteric modulator of the cannabinoid CB1 receptor. Br. J. Pharmacol. 2015, 172, 4790–4805. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Thomas, A.; Baillie, G.L.; Phillips, A.M.; Razdan, R.K.; Ross, R.A.; Pertwee, R.G. Cannabidiol displays unexpectedly high potency as an antagonist of CB1 and CB2 receptor agonists in vitro. Br. J. Pharmacol. 2007, 150, 613–623. [Google Scholar] [CrossRef] [Green Version]
- Maurya, N.; Velmurugan, B.K. Therapeutic applications of cannabinoids. Chem.-Biol. Interact. 2018, 293, 77–88. [Google Scholar] [CrossRef]
- Kozela, E.; Pietr, M.; Juknat, A.; Rimmerman, N.; Levy, R.; Vogel, Z. Cannabinoids ∆9-Tetrahydrocannabinol and Cannabidiol differentially inhibit the Lipopolysaccharide-activated NF-κB and Interferon-β/STAT proinflammatory pathways in BV-2 microglial cells. J. Biol. Chem. 2010, 285, 1616–1626. [Google Scholar] [CrossRef] [Green Version]
- Kozela, E.; Lev, N.; Kaushansky, N.; Eilam, R.; Rimmerman, N.; Levy, R.; Ben-Nun, A.; Juknat, A.; Vogel, Z. Cannabidiol inhibits pathogenic T cells, decreases spinal microglial activation and ameliorates multiple sclerosis-like disease in C57BL/6 mice. Br. J. Pharmacol. 2011, 163, 1507–1519. [Google Scholar] [CrossRef] [Green Version]
- Malfait, A.M.; Gallily, R.; Sumariwalla, P.F.; Malik, A.S.; Andreakos, E.; Mechoulam, R.; Feldmann, M. The nonpsychoactive cannabis constituent cannabidiol is an oral anti-arthritic therapeutic in murine collagen-induced arthritis. Proc. Natl. Acad. Sci. USA 2000, 97, 9561–9566. [Google Scholar] [CrossRef] [Green Version]
- Borrelli, F.; Aviello, G.; Romano, B.; Orlando, P.; Capasso, R.; Maiello, F.; Guadagno, F.; Petrosino, S.; Capasso, F.; Marzo, V.D.; et al. Cannabidiol, a safe and non-psychotropic ingredient of the marijuana plant Cannabis sativa, is protective in a murine model of colitis. J. Mol. Med. 2009, 87, 1111–1121. [Google Scholar] [CrossRef] [PubMed]
- Weiss, L.; Zeira, M.; Reich, S.; Har-Noy, M.; Mechoulam, R.; Slavin, S.; Gallily, R. Cannabidiol lowers incidence of diabetes in non-obese diabetic mice. Autoimmunity 2006, 39, 143–151. [Google Scholar] [CrossRef] [Green Version]
- Wilkinson, J.D.; Williamson, E.M. Cannabinoids inhibit human keratinocyte proliferation through a non-CB1/CB2 mecha- nism and have a potential therapeutic value in the treatment of psoriasis. J. Dermatol. Sci. 2007, 45, 87–92. [Google Scholar] [CrossRef] [PubMed]
- Ramirez, B.G. Prevention of Alzheimer’s disease pathology by cannabinoids: Neuroprotection mediated by blockade of microglial activation. J. Neurosci. 2005, 25, 1904–1913. [Google Scholar] [CrossRef] [Green Version]
- García-Arencibia, M.; González, S.; de Lago, E.; Ramos, J.A.; Mechoulam, R.; Fernández-Ruiz, J. Evaluation of the neuroprotective effect of cannabinoids in a rat model of Parkinson’s disease: Importance of antioxidant and cannabinoid receptor-independent properties. Brain Res. 2007, 1134, 162–170. [Google Scholar] [CrossRef] [PubMed]
- Consroe, P.; Laguna, J.; Allender, J.; Snider, S.; Stern, L.; Sandyk, R.; Kennedy, K.; Schram, K. Controlled clinical trial of cannabidiol in Huntington’s disease. Pharmacol. Biochem. Behav. 1991, 40, 701–708. [Google Scholar] [CrossRef]
- Campos, A.C.; Moreira, F.A.; Gomes, F.V.; Bel, E.A.D.; Guimarães, F.S. Multiple mechanisms involved in the large-spectrum therapeutic potential of cannabidiol in psychiatric disorders. Philos. Trans. R. Soc. B Biol. Sci. 2012, 367, 3364–3378. [Google Scholar] [CrossRef] [PubMed]
- Jones, N.A.; Glyn, S.E.; Akiyama, S.; Hill, T.D.; Hill, A.J.; Weston, S.E.; Burnett, M.D.; Yamasaki, Y.; Stephens, G.J.; Whalley, B.J.; et al. Cannabidiol exerts anti-convulsant effects in animal models of temporal lobe and partial seizures. Seizure 2012, 21, 344–352. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jones, N.A.; Hill, A.J.; Smith, I.; Bevan, S.A.; Williams, C.M.; Whalley, B.J.; Stephens, G.J. Cannabidiol displays antiepileptiform and antiseizure properties in vitro and in vivo. J. Pharmacol. Exp. Ther. 2009, 332, 569–577. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Campos, A.C.; Guimarães, F.S. Evidence for a potential role for TRPV1 receptors in the dorsolateral periaqueductal gray in the attenuation of the anxiolytic effects of cannabinoids. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2009, 33, 1517–1521. [Google Scholar] [CrossRef]
- Petrocellis, L.D.; Ligresti, A.; Moriello, A.S.; Iappelli, M.; Verde, R.; Stott, C.G.; Cristino, L.; Orlando, P.; Marzo, V.D. Non-THC cannabinoids inhibit prostate carcinoma growthin vitroandin vivo: Pro-apoptotic effects and underlying mechanisms. Br. J. Pharmacol. 2012, 168, 79–102. [Google Scholar] [CrossRef] [Green Version]
- Ligresti, A.; Moriello, A.S.; Starowicz, K.; Matias, I.; Pisanti, S.; Petrocellis, L.D.; Laezza, C.; Portella, G.; Bifulco, M.; Marzo, V.D. Antitumor activity of plant cannabinoids with emphasis on the effect of cannabidiol on human breast carcinoma. J. Pharmacol. Exp. Ther. 2006, 318, 1375–1387. [Google Scholar] [CrossRef] [Green Version]
- Campos, A.C.; Fogaça, M.V.; Sonego, A.B.; Guimarães, F.S. Cannabidiol, neuroprotection and neuropsychiatric disorders. Pharmacol. Res. 2016, 112, 119–127. [Google Scholar] [CrossRef]
- State of Colorado, U. Amendment 64 Implementation. 2012. Available online: https://www.colorado.gov/pacific/sites/default/files/13%20Amendment%2064%20LEGIS.pdf (accessed on 10 July 2021).
- Judgement of 19 November 2020. Available online: https://eur-lex.europa.eu/legal-content/DE/TXT/?uri=CELEX%3A62018CJ0663;Case C-663/18. ECLI:EU:C:2020:938 (accessed on 29 August 2021).
- World Anti Doping Agency. Summary of Major Modifications and Explanatory Notes. 2018 Prohibited List. Available online: https://www.ncaa.org/sites/default/files/2018RES_Substance_Use_Final_Report_FINAL_20180611.pdf (accessed on 10 July 2021).
- Bergamaschi, M.M.; Queiroz, R.H.C.; Zuardi, A.W.; Crippa, J.A.S. Safety and side effects of cannabidiol, a cannabis sativa constituent. Curr. Drug Saf. 2011, 6, 237–249. [Google Scholar] [CrossRef]
- Taylor, L.; Crockett, J.; Tayo, B.; Morrison, G. A phase 1, open-label, parallel-group, single-dose trial of the pharmacokinetics and safety of cannabidiol (CBD) in subjects with mild to severe hepatic impairment. J. Clin. Pharmacol. 2019, 59, 1110–1119. [Google Scholar] [CrossRef]
- Taylor, L.; Gidal, B.; Blakey, G.; Tayo, B.; Morrison, G. A phase I, randomized, double-blind, placebo-controlled, single ascending dose, multiple dose, and food effect trial of the safety, tolerability and pharmacokinetics of highly purified cannabidiol in healthy subjects. CNS Drugs 2018, 32, 1053–1067. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- McCartney, D.; Benson, M.J.; Desbrow, B.; Irwin, C.; Suraev, A.; McGregor, I.S. Cannabidiol and sports performance: A narrative review of relevant evidence and recommendations for future research. Sports Med.-Open 2020, 6. [Google Scholar] [CrossRef] [PubMed]
- Gamelin, F.X.; Cuvelier, G.; Mendes, A.; Aucouturier, J.; Berthoin, S.; Marzo, V.D.; Heyman, E. Cannabidiol in sport: Ergogenic or else? Pharmacol. Res. 2020, 156, 104764. [Google Scholar] [CrossRef]
- Crippa, J.A.; Guimarães, F.S.; Campos, A.C.; Zuardi, A.W. Translational investigation of the therapeutic potential of cannabidiol (CBD): Toward a new age. Front. Immunol. 2018, 9, 86. [Google Scholar] [CrossRef] [Green Version]
- Corroon, J.; Phillips, J.A. A cross-sectional study of cannabidiol users. Cannabis Cannabinoid Res. 2018, 3, 152–161. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Moltke, J.; Hindocha, C. Reasons for cannabidiol use: A cross-sectional study of CBD users, focusing on self-perceived stress, anxiety, and sleep problems. J. Cannabis Res. 2021, 3, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Chagas, M.H.N.; Crippa, J.A.S.; Zuardi, A.W.; Hallak, J.E.C.; de Sousa, J.P.M.; Hirotsu, C.; Maia, L.; Tufik, S.; Andersen, M.L. Effects of acute systemic administration of cannabidiol on sleep-wake cycle in rats. J. Psychopharmacol. 2013, 27, 312–316. [Google Scholar] [CrossRef]
- Murillo-Rodríguez, E.; Millán-Aldaco, D.; Cicconcelli, D.; Giorgetti, V.; Arankowsky-Sandoval, G.; Alcaraz-Silva, J.; Imperatori, C.; Machado, S.; Budde, H.; Torterolo, P. Sleep-wake cycle disturbances and NeuN-altered expression in adult rats after cannabidiol treatments during adolescence. Psychopharmacology 2021, 238, 1437–1447. [Google Scholar] [CrossRef]
- Babson, K.A.; Sottile, J.; Morabito, D. Cannabis, cannabinoids, and sleep: A review of the literature. Curr. Psychiatry Rep. 2017, 19, 1–12. [Google Scholar] [CrossRef]
- Carlini, E.A.; Cunha, J.M. Hypnotic and antiepileptic effects of cannabidiol. J. Clin. Pharmacol. 1981, 21, 417S–427S. [Google Scholar] [CrossRef]
- Malhotra, R.K. Sleep, recovery, and performance in sports. Neurol. Clin. 2017, 35, 547–557. [Google Scholar] [CrossRef]
- Hatchett, A.; Armstrong, K.; Hughes, B.; Parr, B. The influence cannabidiol on delayed onset of muscle soreness. Int. J. Phys. Educ. Sports Health 2020, 7, 89–94. [Google Scholar]
- Cochrane-Snyman, K.C.; Cruz, C.; Morales, J.; Coles, M. The effects of cannabidiol oil on noninvasive measures of muscle damage in men. Med. Sci. Sports Exerc. 2021. [Google Scholar] [CrossRef] [PubMed]
- Isenmann, E.; Veit, S.; Diel, P. Effects of cannabidiol supplementation on the skeletal muscle regeneration after intensive resistance training. Med. Sci. Sports Exerc. 2020, 52, 766. [Google Scholar] [CrossRef]
- Junior, E.R.T.S.; de Salles, B.F.; Dias, I.; Ribeiro, A.S.; Simão, R.; Willardson, J.M. Classification and determination model of resistance training status. Strength Cond. J. 2021. Online ahead of print. [Google Scholar] [CrossRef]
- Miller, T.; Haywood, K.M.; Roberton, M.A.; Getchell, N. NSCA’s Guide to Tests and Assessments; Human Kinetics: Champaign, IL, USA, 2012. [Google Scholar]
- Millar, S.A.; Stone, N.L.; Yates, A.S.; O’Sullivan, S.E. A systematic review on the pharmacokinetics of cannabidiol in humans. Front. Pharmacol. 2018, 9, 1365. [Google Scholar] [CrossRef]
- Isenmann, E.; Diel, P.; Geisler, S.; Schiffer, T. The effects of protein and carbohydrate supplementation on muscular regeneration after intense resistance training in soccer players. Curr. Dev. Nutr. 2020, 4, 1756. [Google Scholar] [CrossRef]
- Schoenfeld, B.J.; Ogborn, D.; Krieger, J.W. Dose-response relationship between weekly resistance training volume and increases in muscle mass: A systematic review and meta-analysis. J. Sports Sci. 2016, 35, 1073–1082. [Google Scholar] [CrossRef]
- Pauw, K.D.; Roelands, B.; Cheung, S.S.; de Geus, B.; Rietjens, G.; Meeusen, R. Guidelines to classify subject groups in sport-science research. Int. J. Sports Physiol. Perform. 2013, 8, 111–122. [Google Scholar] [CrossRef] [PubMed] [Green Version]
| Parameter | |
|---|---|
| Age [Years] | 24 ± 3 |
| Height [cm] | 181.4 ± 10.0 |
| Body Mass [kg] | 79.2 ± 13.7 |
| CMJ [cm] | 55.2 ± 8.5 |
| 1RM [kg] | 118.5 ± 24.7 |
| 1RM/BM [kg] | 1.4 ± 0.2 |
| PLA (n = 16) | CBD (n = 16) | |||||||
|---|---|---|---|---|---|---|---|---|
| Parameter | Units | PRE (T0) | POST | ∆ | PRE | POST | ∆ | ES |
| Creatine Kinase T 24 | [U/L] | 211.6 ± 16.9 | 399.9 ± 68.4 | 188.3 ± 58.4 *** | 197.4 ± 19.1 | 351.5 ± 61.3 | 154.1 ± 61.1 *** | ‒ |
| Creatine Kinase T 48 | [U/L] | 196.8 ± 39.1 | 530.8 ± 237.5 | 333.9 ± 238.6 *** | 188.6 ± 25.2 | 320.9 ± 65.9 | 132.4 ± 66.0 *** | ‒ |
| Creatine Kinase T 72 | [U/L] | 347.9 ± 151.3 | 3417.7 ± 2119.9 | 3069.7 ± 2106.6 *** | 181.9 ± 25.2 | 232.6 ± 35.0 | 50.7 ± 44.7 ***/# | 0.24 |
| Myoglobin T 24 | [ng/mL] | 31.8 ± 8.3 | 38.4 ± 15.1 | 6.6 ± 9.2 ** | 31.9 ± 15.0 | 31.8 ± 8.3 | −0.1 ± 12.7 ** | ‒ |
| Myoglobin T 48 | [ng/mL] | 30.5 ± 10.0 | 33.9 ± 15.4 | 3.5 ± 19.0 | 28.3 ± 6.4 | 37.5 ± 25.6 | 9.2 ± 25.0 ** | ‒ |
| Myoglobin T 72 | [ng/mL] | 30.3 ± 5.8 | 37.1 ± 15.8 | 6.8 ± 15.0 *** | 28.1 ± 5.6 | 34.4 ± 11.3 | 6.3 ± 8.6 ***/# | 0.21 |
| Back Squat T 24 | [kg] | 118.0 ± 6.0 | 114.7 ± 5.5 | −3.3 ± 1.5 ** | 118.1 ± 5.5 | 117.5 ± 5.5 | −0.6 ± 0.8 ** | ‒ |
| Back Squat T 48 | [kg] | 118.3 ± 6.0 | 116.3 ± 6.7 | −2.0 ± 1.9 | 119.1 ± 5.6 | 118.0 ± 5.4 | −1.1 ± 1.0 | ‒ |
| Back Squat T 72 | [kg] | 118.2 ± 6.5 | 116.2 ± 6.8 | −2.0 ± 1.6 | 118.8 ± 5.8 | 120.0 ± 5.6 | 1.3 ± 0.9 # | 0.37 |
| Counter Movement Jump T 24 | [cm] | 55.7 ± 2.0 | 54.6 ± 1.5 | −1.2 ± 0.7 | 54.4 ± 1.8 | 53.6 ± 1.7 | −0.8 ± 0.4 | ‒ |
| Counter Movement Jump T 48 | [cm] | 54.9 ± 2.0 | 55.0 ± 2.1 | 0.1 ± 0.6 | 54.9 ± 1.8 | 54.0 ± 1.8 | −0.8 ± 0.6 | ‒ |
| Counter Movement Jump T 72 | [cm] | 54.4 ± 2.0 | 54.1 ± 2.1 | −0.4 ± 0.8 | 55.3 ± 1.7 | 55.5 ± 1.7 | 0.2 ± 0.6 | ‒ |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Isenmann, E.; Veit, S.; Starke, L.; Flenker, U.; Diel, P. Effects of Cannabidiol Supplementation on Skeletal Muscle Regeneration after Intensive Resistance Training. Nutrients 2021, 13, 3028. https://doi.org/10.3390/nu13093028
Isenmann E, Veit S, Starke L, Flenker U, Diel P. Effects of Cannabidiol Supplementation on Skeletal Muscle Regeneration after Intensive Resistance Training. Nutrients. 2021; 13(9):3028. https://doi.org/10.3390/nu13093028
Chicago/Turabian StyleIsenmann, Eduard, Sebastian Veit, Lynn Starke, Ulrich Flenker, and Patrick Diel. 2021. "Effects of Cannabidiol Supplementation on Skeletal Muscle Regeneration after Intensive Resistance Training" Nutrients 13, no. 9: 3028. https://doi.org/10.3390/nu13093028
APA StyleIsenmann, E., Veit, S., Starke, L., Flenker, U., & Diel, P. (2021). Effects of Cannabidiol Supplementation on Skeletal Muscle Regeneration after Intensive Resistance Training. Nutrients, 13(9), 3028. https://doi.org/10.3390/nu13093028






