Health-Promoting of Polysaccharides Extracted from Ganoderma lucidum
Abstract
:1. Introduction
2. Bioactivity of Polysaccharides from Ganoderma lucidum
2.1. Antioxidative Activity
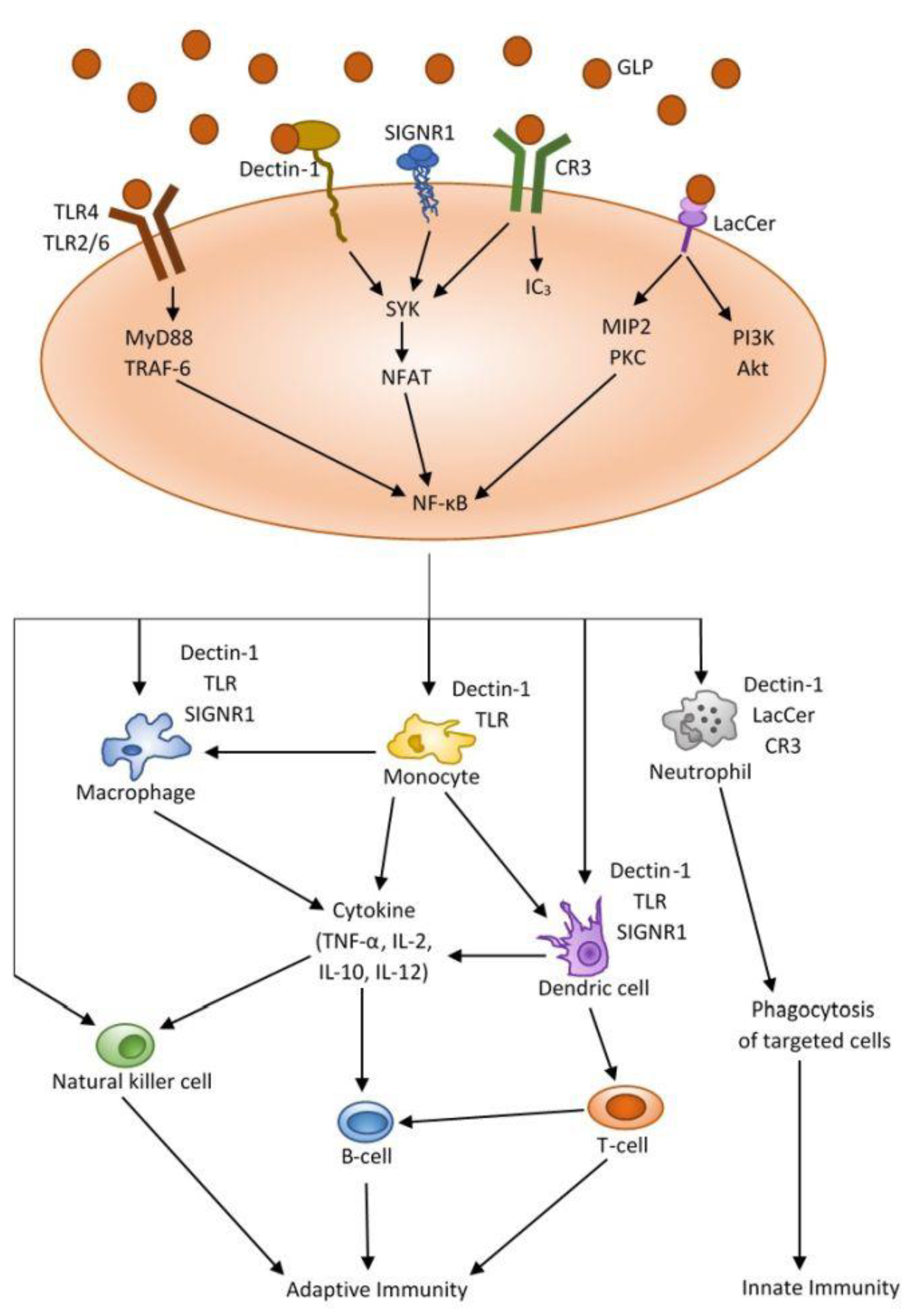
2.2. Immunomodulatory Activity
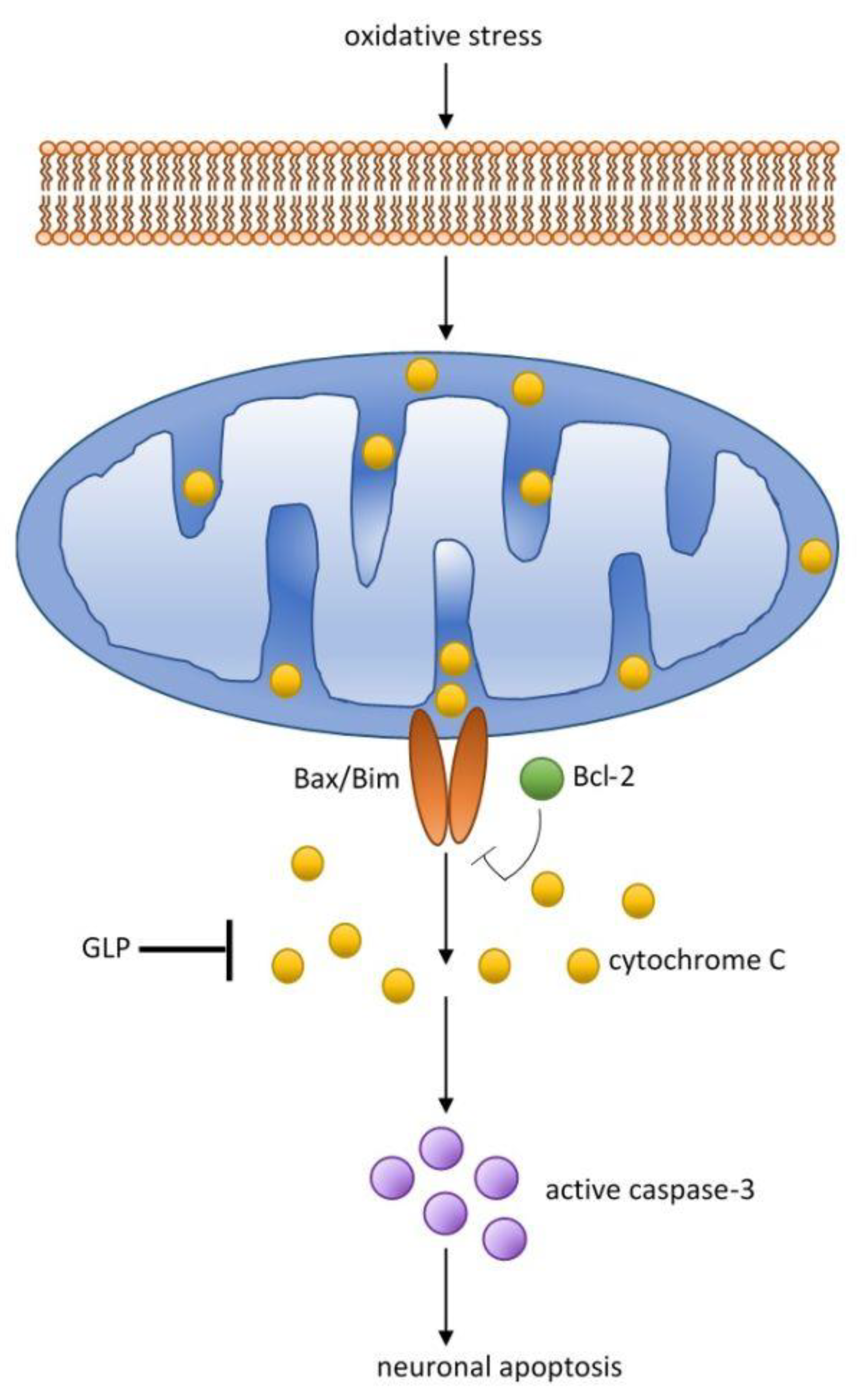
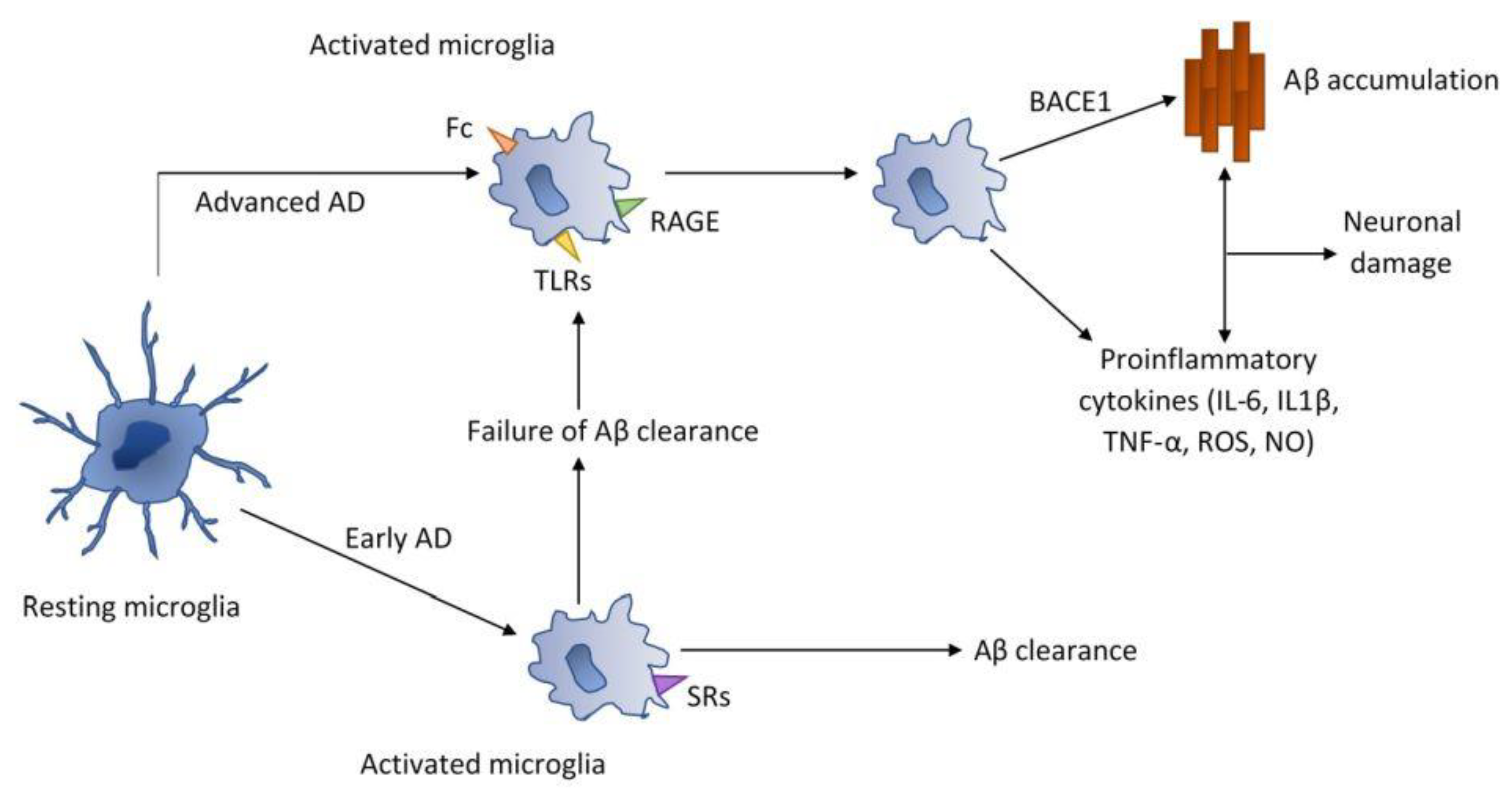
2.3. Antineurodegenerative Activity
2.4. Antidiabetic Activity
3. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Wang, X.M.; Zhang, J.; Wu, L.H.; Zhao, Y.L.; Li, T.; Li, J.Q.; Wang, Y.Z.; Liu, H.G. A mini-review of chemical composition and nutritional value of edible wild-grown mushroom from China. Food Chem. 2014, 151, 279–285. [Google Scholar] [CrossRef]
- Seweryn, E.; Ziała, A.; Gamian, A. Properties of Ganoderma lucidum triterpenes. Postepy Hig. Med. Dosw. 2019, 73, 1–8. [Google Scholar] [CrossRef]
- Wasser, S.P. Medicinal mushroom science: History, current status, future trends and unsolved problems. Int. J. Med. Mushrooms 2010, 12, 1–16. [Google Scholar] [CrossRef]
- Siwulski, M.; Sobieralski, K.; Golak-Siwulska, I.; Sokół, S.; Sękara, A. Ganoderma lucidum (Curt.: Fr. Karst.)—Health-promoting properties. A review. Herba Pol. 2015, 61, 105–118. [Google Scholar] [CrossRef] [Green Version]
- Sindhu, R.K.; Goyal, A.; Das, J.; Neha; Choden, S.; Kumar, P. Immunomodulatory potential of polysaccharides derived from plants and microbes: A narrative review. Carbohydr. Polym. Technol. Appl. 2021, 2, 100044. [Google Scholar]
- Choong, Y.K.; Ellan, K.; Chen, X.D.; Mohamad, S.A. Extraction and Fractionation of Polysaccharides from a Selected Mushroom Species, Ganoderma lucidum: A Critical Review. Intech Open 2018. [CrossRef] [Green Version]
- Synytsya, A.; Novak, M. Structural diversity of fungal glucans. Carbohydr. Polym. 2013, 92, 792–809. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Niu, D.; Zhang, Y.; Zeng, X.-A. Physicochemical properties, antioxidant and antiproliferative activities of polysaccharides from Morinda citrifolia l. (Noni) based on different extraction methods. Int. J. Biol. Macromol. 2020, 150, 114–121. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Wang, H.Y.; Pang, X.B.; Yao, W.B.; Gao, X.D. Characterization and antioxidant activity of low-molecular-weight polysaccharides purified from the fruiting bodies of Ganoderma lucidum. Int. J. Biol. Macromol. 2010, 46, 451–457. [Google Scholar] [CrossRef]
- Zeng, X.T.; Li, P.Y.; Chen, Y.; Kang, Y.; Xie, Y.; Li, X.; Xie, T.H.; Zhang, Y.K. Effects of deproteinization methods on primary structure and antioxidant activity of Ganoderma lucidum polysaccharides. Int. J. Biol. Macromol. 2019, 126, 867–876. [Google Scholar] [CrossRef]
- Xu, Y.; Wu, Y.; Sun, P.; Zhang, F.; Linhardt, R.J.; Zhang, A. Chemically modified polysaccharides: Synthesis, characterization, structure activity relationships of action. Int. J. Biol. Macromol. 2019, 13, 970–977. [Google Scholar] [CrossRef]
- Xu, J.; Liu, W.; Yao, W.B.; Pang, X.B.; Yin, D.K.; Gao, X.D. Carboxymethylation of a polysaccharide extracted from Ganoderma lucidum enhances its antioxidant activities in vitro. Carbohydr. Polym. 2009, 78, 227–234. [Google Scholar] [CrossRef]
- Zhang, J.; Liu, Y.J.; Park, H.S.; Xia, Y.M.; Kim, G.S. Antitumor activity of sulphated extracellular polysaccharides of Ganoderma lucidum from the submerged fermentation broth. Carbohydr. Polym. 2012, 87, 1539–1544. [Google Scholar] [CrossRef]
- Zhang, K.; Liu, Y.F.; Zhao, X.L.; Tang, Q.J.; Dernedde, J.; Zhang, J.S.; Fan, H. Anti-inflammatory properties of GLPss58, a sulphated polysaccharide from Ganoderma lucidum. Int. J. Biol. Macromol. 2018, 107, 486–493. [Google Scholar] [CrossRef]
- Ahmad, F. Ganoderma lucidum: Persuasive biologically active constituents and their health endorsement. Biomed. Pharmacother. 2018, 107, 507–519. [Google Scholar] [CrossRef]
- Lu, J.; He, R.; Sun, P.; Zhang, F.; Linhardt, R.J.; Zhang, A. Molecular mechanisms of bioactive polysaccharides from Ganoderma lucidum (Lingzhi), a review. Int. J. Biol. Macromol. 2020, 150, 765–774. [Google Scholar] [CrossRef]
- Yu, Y.; Shen, M.; Song, Q.; Xie, J. Biological activities and pharmaceutical applications of polysaccharide from natural resources: A review. Carbohydr. Polym. 2018, 183, 91–101. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Tang, J.; Gao, H.; Xu, Y.; Han, Y.; Shang, H.; Lu, Y.; Qin, C. Ganoderma lucidum triterpenoids and polysaccharides attenuate artherosclerotic plaque in high-fat diet rabbits. Nutr. Metab. Crdiovasc. Dis. 2021, 21, 00145-149. [Google Scholar]
- Cör, D.; Knez, Ž.; Knez-Hrnčič, M. Antitumour, antimicrobial, antioxidant and antiacetylcholinesterase effect of Ganoderma lucidum terpenoids and polysaccharides: A review. Molecules 2018, 23, 649. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, Y.; Feng, Y.; Wang, W.; Jia, L.; Zhang, J. Characterization and hepatoprotections of Ganoderma lucidum polysaccharides against multiple organ dysfunction syndrome in mice. Oxid. Med. Cell. Longev. 2021. [Google Scholar] [CrossRef]
- Li, J.; Gu, F.; Cai, C.; Hu, M.; Fan, L.; Hao, J.; Yu, G. Purification, structural characterization, and immunomodulatory activity of the polysaccharides from Ganoderma lucidum. Int. J. Biol. Macromol. 2020, 143, 806–813. [Google Scholar] [CrossRef]
- Zhang, S.L.; Pang, G.B.; Chen, C.; Qin, J.Z.; Yu, H.; Liu, Y.M.; Zhanf, X.H.; Song, Z.T.; Zhao, J.; Wang, F.J.; et al. Effective cancer immunotherapy by Ganoderma lucidum polysaccharide-gold nanocomposites through dendritic cell activation and memory T cell response. Carbohyd. Polym. 2019, 205, 192–202. [Google Scholar] [CrossRef]
- Liang, Z.; Yuan, Z.; Guo, J.; Wu, J.; Yi, J.; Deng, J.; Shan, Y. Ganoderma lucidum polysaccharides prevent palmitic acidevoked apoptosis and autophagy in intestinal porcine epithelial cell line via restoration of mitochondrial function and regulation of MAPK and AMPK/Akt/mTOR signaling pathway. Int. J. Mol. Sci. 2019, 20, 478. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lin, Y.L.; Liang, Y.C.; Lee, S.S.; Chiang, B.L. Polysaccharide purified from Ganoderma lucidum induced activation and maturation of human monocyte-derived dendric cells by the NFĸB and p38 mitogen-activated protein kinase pathways. J. Leukoc. Biol. 2005, 78, 533–543. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Habijanic, J.; Berovic, M.; Boh, B.; Planki, M.; Wraber, B. Submerged cultivation of Ganoderma lucidum and the effects of its polysaccharides on the production of human cytokines TNF-α, Il-12, IFN-ɣ, IL-2, IL-4, IL-10 and IL-17. New Biotechnol. 2015, 32, 85–95. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.Z.; Chang, Y.Z.; He, Z.M.; Chen, L.; Zhou, X.W. Immunomodulatory activity of Ganoderma lucidum immunomodulatory protein via PI3K/Akt and MAPK signalling pathways in RAW264.7 cells. J. Cell. Physiol. 2019, 234, 23337–23348. [Google Scholar] [CrossRef]
- Mallard, B.; Leach, D.N.; Wolmuth, H.; Tiralongo, J. Synergistic immunemodulatory activity in human macropages of a medicinal mushroom formulation consisting of Reishi, Shiitake and Maitake. PLoS ONE 2019, 14, e0224740. [Google Scholar] [CrossRef] [Green Version]
- Wang, J.; Cao, B.; Zhao, H.; Feng, J. Emerging roles of Ganoderma lucidum in antiaging. Aiging Dis. 2018, 8, 691–707. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sun, X.Z.; Liao, Y.; Li, W.; Guo, L. Neuroprotective effects of Ganoderma lucidum polysaccharides against oxidative stress-induced neuronal apoptosis and antimicrobial activities. Neural Regen. Res. 2017, 12, 953–958. [Google Scholar] [PubMed]
- Cai, Q.; Li, Y.; Pei, G. Polysaccharides from Ganoderma lucidum attenuate microgliamediated neuroinflammation and modulate microglial phagocytosis and behavioural response. J. Neuroinflammation 2017, 14, 63. [Google Scholar] [CrossRef] [Green Version]
- Lai, C.S.-W.; Yu, M.-S.; Yuen, W.-H.; So, K.-F.; Zee, S.-Y.; Chang, R.C.-C. Antagonizing beta-amyloid peptide neurotoxicity of the anti-aging fungus Ganoderma lucidum. Brain Res. 2008, 1190, 215–224. [Google Scholar] [CrossRef]
- Zhou, Y.; Qu, Z.; Zeng, Y.; Lin, Y.; Chung, P. Neuroprotective effect of preadministration with Ganoderma lucidum spore on rat hippocampus. Exp. Toxicol. Pathol. 2012, 64, 673–680. [Google Scholar] [CrossRef]
- Zhang, R.; Xu, S.; Cai, Y.; Zhou, M.; Zuo, X.; Chan, P. Ganoderma lucidum protects dopaminergic neuron degeneration through inhibition of microglial activation. Evid. Based Complement. Alternat. Med. 2011, 2011, 156810. [Google Scholar] [CrossRef] [Green Version]
- Lai, G.; Guo, Y.; Chen, D.; Tang, X.; Shuai, O.; Yong, T.; Wang, D.; Xiao, C.; Zhou, G.; Xie, Y.; et al. Alcohol extracts from Ganoderma lucidum delay the progress of Alzheimer‘s disease by regulating DNA methylation in rodents. Front. Pharmacol. 2019, 10, 272–298. [Google Scholar] [CrossRef] [PubMed]
- Xie, F.; Wu, M.; Lai, B.; Halim, M.; Liu, S.; Shi, D. Effects of redox interference on the pancreatic mitochondria and the abnormal blood glucose. Free Radic. Res. 2021. [Google Scholar] [CrossRef]
- Shaher, F.; Wang, S.; Qiu, H.; Hu, Y.; Zhang, Y.; Wang, W.; Al-Ward, H.; Abdulghani, M.A.M.; Baldi, S.; Zhou, S. Effect and mechanism of Ganoderma lucidum spores on alleviation of diabetic cardiomyopathy in a pilot in vivo study. Diabetes Metab. Sundr. Obes. 2020, 13, 4809–4822. [Google Scholar] [CrossRef]
- Zeng, P.; Guo, Z.; Zeng, X.; Hao, C.; Zhang, Y.; Zhang, M.; Liu, Y.; Li, H.; Li, J.; Zhang, L. Chemical, biochemical, preclinical and clinical studies of Ganoderma lucidum polysaccharide as an approved drug for treating myopathy and other diseases in China. J. Cell. Mol. Med. 2018, 22, 3278–3297. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sang, T.; Guo, C.; Guo, D.; Wu, J.; Wang, Y.; Chen, C.; Wu, K.; Na, K.; Li, K.; Fang, L.; et al. Suppression of obesity and inflammation by polysaccharide from sporoderm-broken spore of Ganoderma lucidum via gut microbiota regulation. Carbohydr. Polym. 2021, 256, 117594. [Google Scholar] [CrossRef] [PubMed]
- Xu, S.; Dou, Y.; Ye, B.; Wu, Q.; Wang, Y.; Hu, M.; Ma, F.; Rong, X.; Guo, J. Ganoderma lucidum polysaccharides improve insulin sensitivity by regulating inflammatory cytokines and gut microbiota composition in mice. J. Funct. Foods 2017, 38, 545–552. [Google Scholar] [CrossRef]
- Vitak, T.; Yurkiv, B.; Wasser, S.; Nevo, E.; Sybirna, N. Effect of medicinal mushrooms on blood cells under conditions of diabetes mellitus. World J. Diabetes 2017, 8, 187–201. [Google Scholar] [CrossRef]
- Zheng, Y.; Bai, L.; Zhou, Y.P.; Tong, R.S.; Zeng, M.H.; Li, X.F.; Shi, J.Y. Polysaccharides from Chinese herbal medicine for anti-diabetes recent advances. Int. J. Biol. Macromol. 2019, 121, 302–310. [Google Scholar] [CrossRef]
- Jia, J.; Zhang, X.; Hu, Y.S.; Wu, Y.; Wang, Q.Z.; Li, N.N.; Guo, Q.C.; Dong, X.C. Evaluation of in vivo antioxidant activities of Ganoderma lucidum polysaccharides in STZ-diabetic rats. Food Chem. 2009, 115, 32–36. [Google Scholar] [CrossRef]
- Meng, C.; Zhu, H.; Yang, S.; Wu, F.; Zheng, H.; Chen, E.; Xu, J. Attenuating effects of Ganoderma lucidum polysaccharides on myocardial collagen cross-linking relates to advanced glycation end product and antioxidant enzymes in high-fat-diet and streptozotocin- induced diabetic rats. Carbohydr. Polym. 2011, 84, 180–185. [Google Scholar] [CrossRef]
- Heriansyah, T.; Nurwidyaningtyas, W.; Sargowo, D.; Tjahjono, C.T.; Wihastuti, T.A. Polysaccharide peptide (PsP) Ganoderma lucidum: A potential inducer for vascular repair in type 2 diabetes mellitus model. Vasc. Health Risk Manag. 2019, 15, 419–427. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, M.; Xiao, D.; Liu, W.; Song, Y.; Zou, B.; Li, L.; Li, P.; Cai, Y.; Liu, D.; Liao, Q.; et al. Intake of Ganoderma lucidum polysaccharides reverse the disturbed gut microbiota and metabolism in type 2 diabetic rats. Int. J. Biol. Macromol. 2020, 155, 890–902. [Google Scholar] [CrossRef]
- Jeong, Y.U.; Park, Y.J. Ergosterol peroxide from the medicinal mushroom Ganoderma lucidum inhibits differentiation and lipid accumulation of 3T3-L1 adipocytes. Int. J. Mol. Sci. 2020, 21, 460. [Google Scholar] [CrossRef] [Green Version]
- Bishop, K.S.; Kao, C.H.J.; Xu, Y.Y.; Glucina, M.P.; Peterson, R.P.M.; Ferguson, L.R. From 2000 years of Ganoderma lucidum to recent developments in nutraceuticals. Phytochem 2015, 114, 56–65. [Google Scholar] [CrossRef] [Green Version]
- Song, L.X. Efficacy GLPSS injection combined with glucocorticoid treatment facial paralysis. Capital Med. 2010, 22, 27–32. [Google Scholar]
- Shaher, F.; Qiu, H.; Wang, S.; Hu, Y.; Wang, W.; Zhang, Y.; Wie, Y.; Al-ward, H.; Abdulghani, M.A.M.; Alenezi, S.K.; et al. Associated targets of the antioxidant cardioprotection of the Ganoderma lucidum in diabetic cardiomyopathy by using open targets platform: A systematic review. Biomed. Res. Int. 2020, 7136075. [Google Scholar] [CrossRef] [PubMed]
- Adeyi, A.O.; Awosanya, S.A.; Adeyi, O.E.; James, A.S.; Adenipekun, C.O. Ganoderma lucidum ethanol abrogates metabolic syndrome in rats: In vivo evaluation of hypoglyvemic, hypolipidemic, hypotensive and antioxidant properties. Obesity Med. 2021, 22, 100320. [Google Scholar] [CrossRef]
- Sudheesh, N.P.; Ajith, T.A.; Ramnath, V.; Janardhanan, K.J. Therapeutic potential of Ganoderma lucidum (Fr.) P.Karst. against the declined antioxidant status in the mitochondria of post-mitotic tissues of aged mice. Clin. Nutr. 2010, 29, 406–412. [Google Scholar] [CrossRef] [PubMed]
- Zhao, W.; Jiang, X.; Deng, W.; Lai, Y.; Wu, M.; Zhang, Z. Antioxidant activities of Ganoderma lucidum polysaccharides and their role on DNA damage in mice induced by cobalt-60 gamma-irradiation. Food Chem. Toxicol. 2012, 50, 303–309. [Google Scholar] [CrossRef] [PubMed]
- Cheng, X.; Chen, Y.; Li, S.; Chen, Y.; Lan, J.; Liu, L. Free radical scavenging of Ganoderma lucidum polysaccharides and its effect on antioxidant enzymes and immunity activities in cervical carcinoma rats. Carbohydr. Polym. 2009, 77, 389–393. [Google Scholar]
- Xu, Y.; Zhang, X.; Yan, X.H.; Zhang, J.L.; Wang, L.Y.; Xue, H.; Jiang, G.C.; Ma, T.X.; Liu, X.J. Characterization, hypolipidemic and antioxidant activities degraded polysaccharides from Ganoderma lucidum. Int. J. Biol. Macromol. 2019, 135, 706–716. [Google Scholar] [CrossRef]
- Liu, Z.G.; Xing, J.; Zheng, S.S.; Bo, R.N.; Luo, L.; Huang, Y.; Niu, Y.L.; Li, Z.H.; Wang, D.Y.; Hu, Y.L. Ganoderma lucidum polysaccharides encapsulated in liposome as a adjuvant to promote Th1-bias immune response. Carbohydr. Polym. 2016, 142, 141–148. [Google Scholar] [CrossRef]
- Shi, M.; Yang, Y.N.; Hu, X.S.; Zhang, Z.Y. Effect of ultrasonic extraction conditions on antioxidative and immunomodulatory activities of a Ganoderma lucidum polysaccharide originated from fermented soybean curd residue. Food Chem. 2014, 155, 50–56. [Google Scholar] [CrossRef] [PubMed]
- Sohretoglu, D.; Huang, S. Ganoderma lucidum polysaccharides as an anti-cancer agent. Anticancer Agent Med. Chem. 2018, 18, 667–674. [Google Scholar] [CrossRef]
- Rasjidi, I.; Susanto, C. Ganoderma lucidum polysaccharide peptide (GLPP) for the cancer treatment. Indones. J. Clin. Pharm. 2015, 4, 120–128. [Google Scholar] [CrossRef]
- Arranz, A.; Doxaki, C.; Vergadi, E.; Martinez de la Torre, Y.; Vaporidi, K.; Lagoudaki, E.D.; Ieronymaki, E.; Androulidaki, A.; Venihaki, M.; Margioris, A.N.; et al. Akt1 and Akt2 protein kinases differentially contribute to macrophage polarization. Proc. Natl. Acad. Sci. USA 2012, 109, 9517–9522. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vergadi, E.; Ieronymaki, E.; Lyroni, K.; Vaporidi, K.; Tsatsanis, C. Akt signaling pathway in macrophage activation and M1/M2 polarization. J. Immunol. 2017, 198, 1006–1014. [Google Scholar] [CrossRef] [Green Version]
- Schett, G.; Neurath, M.F. Resolution of chronic inflammatory disease: Universal and tissuespecific concepts. Nat. Commun. 2018, 9, 3261. [Google Scholar] [CrossRef]
- Zhao, Y.; Zhan, J.K.; Liu, Y. A perspective on roles played by immunosenescence in the pathobiology of Alzheimer’s disease. Aging Dis. 2020, 11, 16–29. [Google Scholar] [CrossRef] [PubMed]
- Hilliard, A.; Mendonca, P.; Soliman, K.F.A. Involvment of NF-ĸB and MAPK signaling pathways in the preventive effects of Ganoderma lucidum on the inflammation of BV-2 microglial cells induced by LPS. J. Neuroimmun. 2020, 345, 577269. [Google Scholar] [CrossRef]
- Phan, C.-W.; David, P.; Naidu, M.; Wong, K.-H.; Sabaratnam, V. Therapeutic potential of culinarymedicinal mushrooms for the management of neurodegenerative diseases: Diversity, metabolite and mechanism. Crit. Rev. Biotechnol. 2015, 35, 355–368. [Google Scholar] [CrossRef]
- Perry, V.H.; Nicoll, J.A.R.; Holmes, C. Microglia in neurodegenerative disease. Nat. Rev. Neurol. 2010, 6, 193–201. [Google Scholar] [CrossRef]
- Brown, G.C.; Neher, J.J. Microglial phagocytosis of live neurons. Nat. Rev. Neurosci. 2014, 15, 209–216. [Google Scholar] [CrossRef]
- Wang, W.Y.; Tan, M.S.; Yu, J.T.; Tan, L. Role of pro-inflammatory cytokines released from microglia in Alzheimer’s disease. Ann. Trans. Med. 2015, 3, 136. [Google Scholar]
- Fu, R.; Shen, Q.; Xu, P.; Luo, J.J.; Tang, Y. Phagocytosis of microglia in the central nervous system diseases. Mol. Neurobiol. 2014, 49, 1422–1434. [Google Scholar] [CrossRef] [Green Version]
- Liang, C.Y.; Liang, Y.M.; Liu, H.Z.; Zhu, D.; Hou, S.Z.; Wu, Y.Y.; Huang, S.; Lai, X.P. Effect of dendrobium officinale on D-galactose-induced aging mice. Chin. J. Integr. Med. 2017. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.-H.; Lin, W.-K.; Chang, S.-H.; Tsai, G.-J. Evaluation of the hypoglycemic and antioxidant effects of submerged Ganoderma lucidum culture in type 2 diabetic rats. Mycology 2020. [Google Scholar] [CrossRef] [Green Version]
- Bach, E.E.; Bach, E.M.; Martins, A.M.C.; Wadt, N.S.Y. Hypoglicemic and hypolipedimic effects of Ganoderma lucidum in streptozotocin-induced diabetic rats. Medicines 2018, 5, 78. [Google Scholar] [CrossRef] [Green Version]
- Nahdi, A.; John, A.; Raza, H. Elucidation of molecular mechanisms of streptozotocin-inuced oxidative stress, apoptosis and mitochondrial dysfunction in rin-5F pancreatic β-cells. Oxid. Med. Cell. Longev. 2017. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dong, K.; Ni, H.; Wu, M.; Tang, Z.; Halim, M.; Shi, D. ROS-mediated glucose metabolic reprogram induces insulin resistance in type 2 diabetes. Biochem. Biophys. Res. Commun. 2016, 476, 204–211. [Google Scholar] [CrossRef]
- Jayachandram, M.; Vinayagam, R.; Ambati, R.R.; Xu, B.; Chung, S.S.M. Guava leaf extract dimishes hyperglycemia and oxidative stress prevents β-cell death, inhibits inflammation and regulates NF-ĸB signalling pathway in STZ induced diabetic rats. Biomed. Res. Int. 2018, 2018, 4601649. [Google Scholar]
- Xiao, C.; Wu, Q.; Zhang, J.; Xie, Y.; Cai, W.; Tan, J. Antidiabetic activity of Ganoderma lucidum polysaccharides F31 downregulated hepatic glucose regulatory enzymes in diabetic mice. J. Ethnopharmacol. 2017, 196, 47–57. [Google Scholar] [CrossRef]
- Li, F.; Zhang, Y.; Zhang, Z. Antihyper glycemic effect of Ganoderma lucidum polysaccharides on streptozocin-induced diabetic mice. Int. J. Mol. Sci. 2011, 12, 6135–6145. [Google Scholar] [CrossRef] [Green Version]
- Xiao, C.; Wu, Q.P.; Cai, W.; Tan, J.B.; Yang, X.B.; Zhang, J.M. Hypoglycemic effects of Ganoderma lucidum polysaccharides in type 2 diabetic mice. Arch. Pharm. Res. 2012, 35, 1793–1801. [Google Scholar] [CrossRef]
- Wihastuti, T.A.; Heriansyah, T. The inhibitory effects of polysaccharide peptides (PsP) of Ganoderma lucidum against atherosclerosis in rats with dyslipidemia. Heart Int. 2017, 12, e1–e7. [Google Scholar] [CrossRef] [Green Version]
- Seto, S.W.; Lam, T.Y.; Tam, H.L.; Au, A.L.S.; Chan, S.W.; Wu, J.H.; Yu, P.H.F.; Leung, G.P.H.; Ngai, S.M.; Yeung, J.H.K.; et al. Novel hypoglycemic effects of Ganoderma lucidum water-extract in obese/diabetic (+db/+db) mice. Phytomedicine 2009, 16, 426–436. [Google Scholar] [CrossRef]
- Schmidt, T.S.B.; Raes, J.; Bork, P. The human gut microbiome: From association to modulation. Cell 2018, 172, 1198–1215. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wen, L.; Duffy, A. Factors influencing the gut microbiota, inflammation and type 2 diabetes. J. Nutr. 2017, 147, 14685–14755. [Google Scholar] [CrossRef] [PubMed] [Green Version]
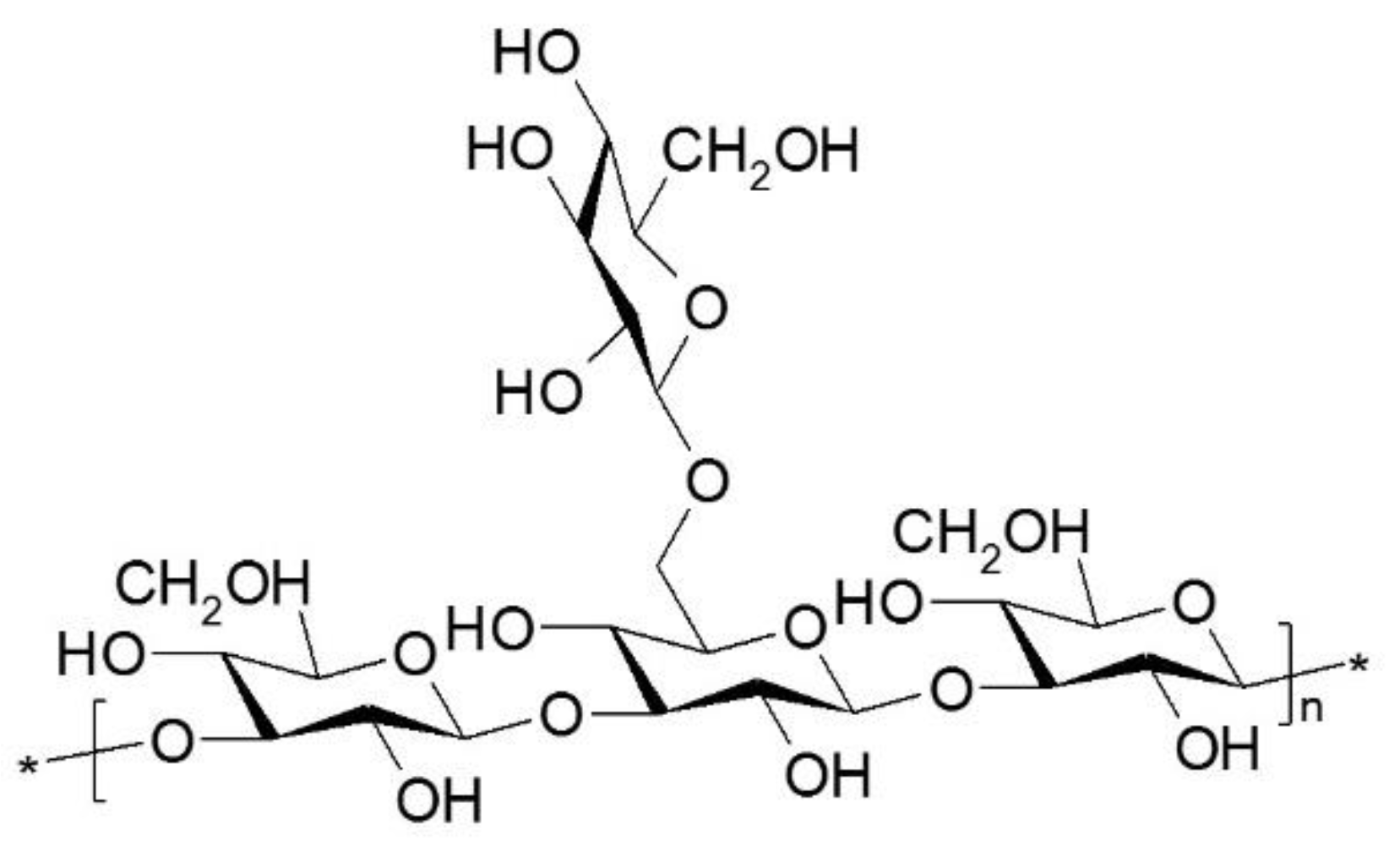
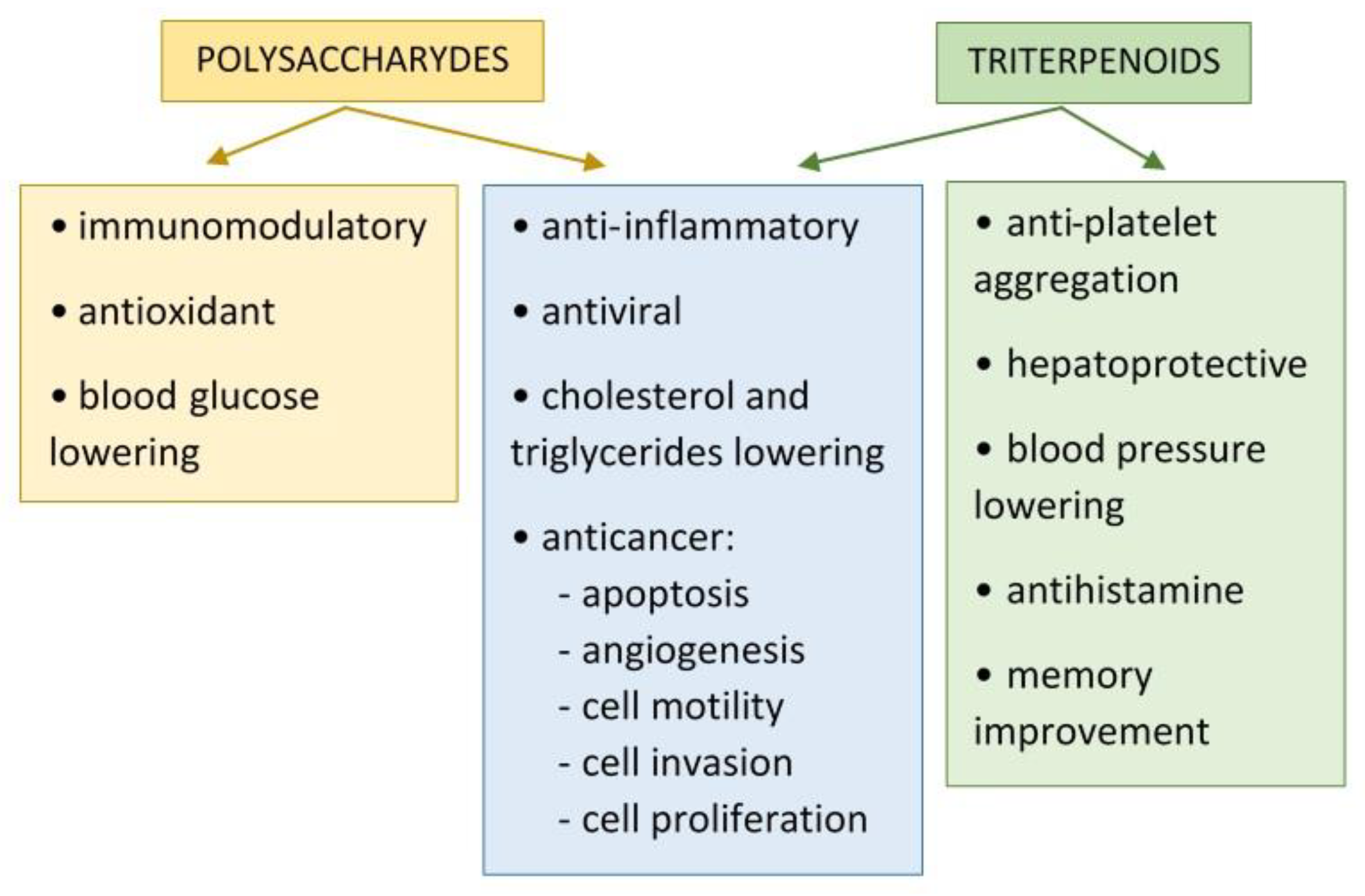
| Biological Activities | Bioactive Components/ Extracts | Origin | Mechanism of Action/ Effects | References |
|---|---|---|---|---|
| Antioxidative activity | GLPS GLPUD | Fruit body | Induce synthesis of SOD, GPx, CAT, GST, SDH, GSH in serum. Decrease in MDA. Reduce hyperlipidemia. | [17] |
| GLPS | Spore | Reduce activity of NOS, cyt. p450, xanthine oxidase, myeloperoxidase. Decrease in MDA, ROS in endothelial cells. | [18] | |
| GLPS | Fruit body | Anti-hydroxyl free radical and anti-superoxide radical ability. Decrease in lipid peroxidation. Increase activity of SOD, CAT, GPx. | [19] | |
| GLPS | Fruit body | Induce synthesis of SOD, CAT; Reduce lipid peroxidation; Decrease inflammatory cytokine: TNF-α, IL-6, IL-1β; Scavenging abilities on DPPH. | [20] | |
| Immunomodulatory activity | GLPS | Fruit body | Promote hematopoiesis. Increase in IgA levels. Regulate of IgG levels. | [21] |
| GLP-Au | Fruit body | Induce of DC maturation; Increase in T and B cells proliferation. Enhancement of NK cells, IL-2 and IFN-γ. Increase in IL-6, IL-12, IL-1β, TNF-α, IFN-ɣ. | [22] | |
| GLPS | Fruit body | Inactivation of MAPK and Akt/mTOR signaling pathways. | [23] | |
| GLPS | Fruit body | Activation of dendritic cells; Induce of IFN-ɣ, IL-10 in human DC. Activation of NF-ĸB, p38MAPK, ERK1/2 pathways during DC maturation. | [24] | |
| α-glucan β-glucan | Fruit body Mycelium | Increase in TNF-α, IFN-γ, IL-2 in human PBMC. Increase in IL-2, IFN-ɣ in Th1 cells. Induce of IL-17 in PBMC. | [25] | |
| rFIP-glu | Fruit body | Activation of RAW264,7 macrophages. Regulate expression of TNF-α, NO, IL-1, IL-6. Induce phosphorylation of Akt. | [26] | |
| β-glucan | Fruit body | Regulate expression of IL-1, IL-6, TNF-α in human macrophages. Reduce expression of IL-10. | [27] | |
| GLPS | Fruit body | Regulate expression of IL-1, IL-6, iNOS in microglial cells. Increase expression of TGFβ. Modulation of microglial morphology and phagocytosis. | [28] | |
| GLE | Fruit body | Modulation NF-ĸB and MAPK signaling pathway. Inhibition of pro-inflammatory cytokines. Inhibition of nitrate in microglial cells. | [29] | |
| Antineurodegenerative activity | GLE | Fruit body | Neuroprotective effect. Reduce amyloid toxicity. Decrease neurotoxicity. Decrease phosphorylation of JNK, c-Jun and p38MAPK. | [30,31] |
| GLE | Spore | Neuroprotective effect in hippocampus. Increase GSH, Gpx. Decrease MDA. Increase ATP and CytQx. | [32] | |
| GLPS | Fruit body | Protection dopaminergic neurons against inflammation. Inhibition of microglial activation. Decrease TNF-α, IL-1β. | [33] | |
| GLE | Fruit body | Regulation of DNA methylation. Improve morphology of hippocampal pyramidal cells. Reduce of β-amyloid. | [34] | |
| Antioxidative activity Antidiabetic activity | GLPS | Fruit body | Significant anti-hydroxyl free radical and anti-superoxide radical activity. Recover SOD activity. Restoring the redox balance. Alleviate insulin resistance. | [35] |
| GLPS | Fruit body | Reduce blood glucose level. Decrease LDH, HbA1c, total cholesterol, triglycerides. Reduce expression of TNF-α, IL-1β | [36] | |
| Anti-inflammation activity | GLPS | Spore | Effective on polymyositis, dermatomyositis and muscular dystrophy. | [37] |
| GLPS | Spore | Reduce serum levels of TNF-α, IL-1β, MCP-1, IL-6. Decrease LDL. Modulation of the intestinal dysbiosis. | [38,39] | |
| Antihyperglycemic activity | GLE | Fruit body | Decrease serum glucose and insulin levels. Increase activity of SOD, CAT, GPx, GSH. Decrease lipid peroxidation. | [40,41,42] |
| GLPS | Fruit body | Reduce blood glucose and HbA1c levels. Increase activity of SOD, CAT. Reduce serum AGE. Reduce total cholesterol and triglycerides. | [43] | |
| GLE (β-D-glucan) | Fruit body | Improve vascular endothelial dysfunction. Increase circulating endothelial cells. Decrease hydrogen peroxide; Decrease triglycerides and cholesterol. | [44] | |
| Antioxidative activity Anti-inflammation activity | GLPS | Fruit body | Decrease IL-1β, IL-6. Decrease triglycerides, total cholesterol. Increase CAT, SOD, GPx. Reduce gut microbiota dysbiosis. | [45] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Seweryn, E.; Ziała, A.; Gamian, A. Health-Promoting of Polysaccharides Extracted from Ganoderma lucidum. Nutrients 2021, 13, 2725. https://doi.org/10.3390/nu13082725
Seweryn E, Ziała A, Gamian A. Health-Promoting of Polysaccharides Extracted from Ganoderma lucidum. Nutrients. 2021; 13(8):2725. https://doi.org/10.3390/nu13082725
Chicago/Turabian StyleSeweryn, Ewa, Anna Ziała, and Andrzej Gamian. 2021. "Health-Promoting of Polysaccharides Extracted from Ganoderma lucidum" Nutrients 13, no. 8: 2725. https://doi.org/10.3390/nu13082725
APA StyleSeweryn, E., Ziała, A., & Gamian, A. (2021). Health-Promoting of Polysaccharides Extracted from Ganoderma lucidum. Nutrients, 13(8), 2725. https://doi.org/10.3390/nu13082725






