‘Old Is Gold’: How Traditional Indian Dietary Practices Can Support Pediatric Diabetes Management
Abstract
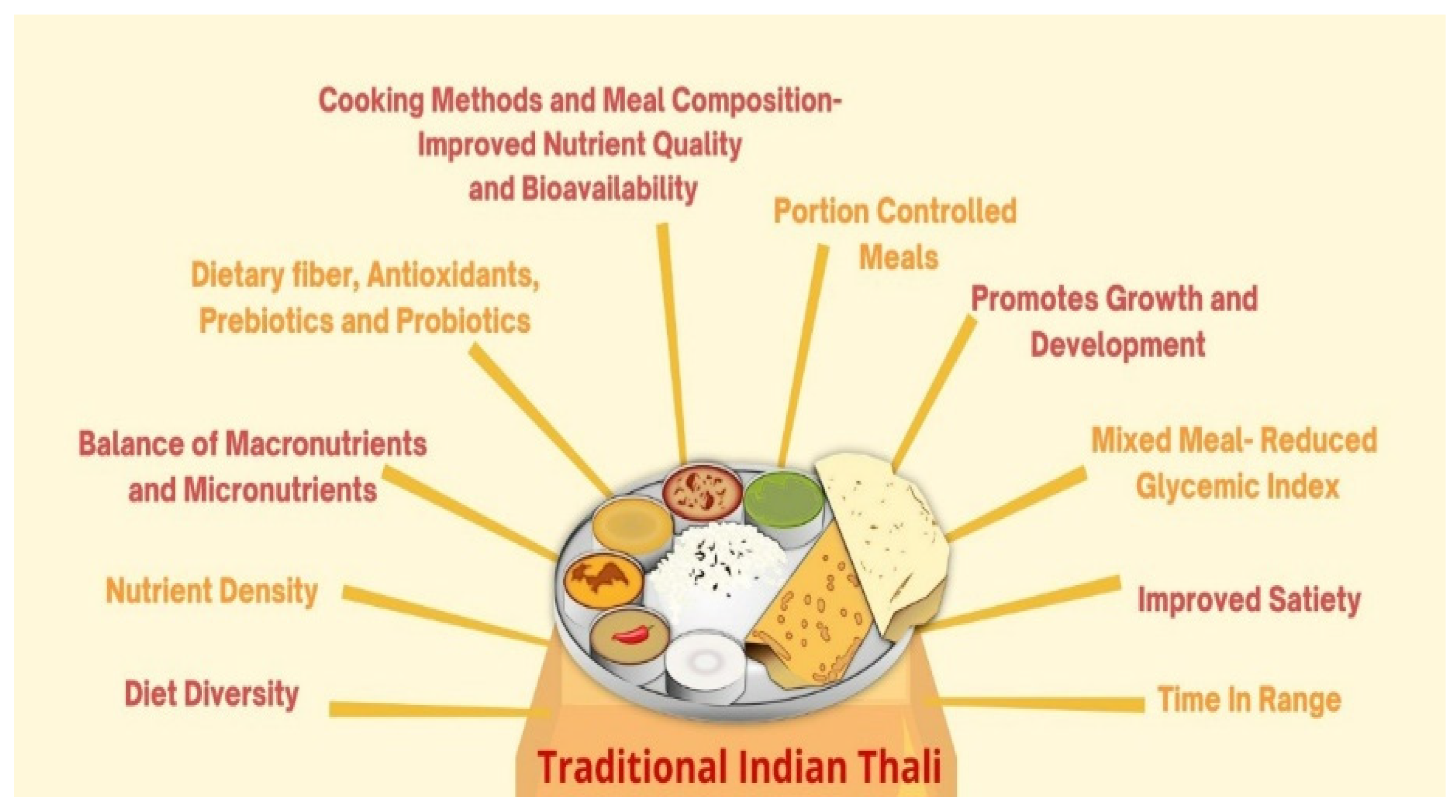
:1. Introduction
2. How Did Ancient India Manage a Healthy Diet?
3. Medical Nutrition Therapy in Type 1 Diabetes
4. Essential Components of Indian Traditional Nutrition
4.1. Carbohydrates
4.2. Dietary Fiber
4.3. Lente Carbohydrates
4.4. Resistant Starch
4.5. Sources of Carbohydrate
4.5.1. Rice
- Using hand-pounded rice: brown/red/black rice
- Using parboiled rice instead of polished white rice
- Using old (i.e., stored and aged) rice
- Using cooked rice that has been cooled overnight
- Combining rice with protein sources like pulses, yogurt, cottage cheese, egg, fish, poultry, meat
- Adding ghee or nuts and seeds in moderate amounts
- Combining rice with a variety of vegetables as a part of a mixed meal
- Squeezing lemon or adding tamarind to rice meals
4.5.2. Wheat
- Using whole wheat flour and Khapli (Emmer) wheat
- Using broken wheat and larger grit semolina
- Mixing bran and pulse flours like gram flour, soya flour, or millet flour with whole wheat flour
- Adding grated or pureed vegetables, green leafy vegetables, herbs, and spices to whole wheat flour
- Kneading whole wheat flour with yogurt, whey, milk, leftover pulse curry, or vegetable curries
- Stuffing the chapati/paratha made from whole wheat flour with protein sources such as egg, minced meat, cottage cheese, pulses, and vegetables
- Adding fat in moderate amounts
4.5.3. Barley
4.5.4. Millets
4.5.5. Maize
4.5.6. Starchy Vegetables
4.5.7. Pseudo Cereals
4.5.8. Added Sugars
4.6. Proteins
4.6.1. Pulses
- Using whole pulses or pulses with skin
- Using sprouted pulses (raw/steamed/ground/pureed)
- Adding pulses (and/or its flours) like soybean to grains like wheat, rice, and millets in meals and snacks
- Adding pulses to vegetables and meat curries
- Substituting cereals with pulses in snacks and meals e.g., in pancakes, roasted gram
- Consuming pulse-based spiced pastes (chutneys) with meals
4.6.2. Dairy Products
4.6.3. Eggs, Fish, Poultry and Meat
4.7. Fats and Oils
Nuts and Seeds
4.8. Micronutrients
4.9. Beverages
4.10. Salt
4.11. Spices and Other Superfoods
4.12. Prebiotics and Probiotics
5. Other Considerations
5.1. Meal Timings
5.2. Insulin Treatment Approaches
5.3. Celiac Disease
5.4. Festive Foods of India
6. Limitations
7. Conclusions
- Processing grains to the minimum. Using unpolished, coarse, long grain, and aged grains
- Using slow digesting (lente) carbohydrates with higher amylose and soluble fiber content such as pulses and barley
- Using meal combinations to add protein, fiber, and healthy fat to meals
- Using resistant-starch-rich foods and methods which enhance resistant starch content
- Using an acidic medium such as lemon, vinegar, or tamarind
- Using slightly unripe fruits, since the GI increases as fruit ripens
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Nambiar, V. Indian Food Anthropology and the Eat Right Movement, 1st ed.; Selective & Scientific Books: New Delhi, India, 2021. [Google Scholar]
- Sarkar, P.; DH, L.K.; Dhumal, C.; Panigrahi, S.S.; Choudhary, R. Traditional and ayurvedic foods of Indian origin. J. Ethn. Foods 2015, 2, 97–109. [Google Scholar] [CrossRef] [Green Version]
- Green, R.; Milner, J.; Joy, E.J.; Agrawal, S.; Dangour, A.D. Dietary patterns in India: A systematic review. Br. J. Nutr. 2016, 116, 142–148. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- World Health Organization. Preamble to the Constitution of the World Health Organization as adopted by the International Health Conference, New York, NY, USA, 19–22 June 1946. In Constitution of the World Health Organization; World Health Organization: Geneva, Switzerland, 1948. [Google Scholar]
- Smart, C.E.; Annan, F.; Higgins, L.A.; Jelleryd, E.; Lopez, M.; Acerini, C.L. ISPAD Clinical Practice Consensus Guidelines 2018: Nutritional management in children and adolescents with diabetes. Pediatric Diabetes 2018, 19, 136–154. [Google Scholar] [CrossRef]
- Bhushan, C.; Taneja, S.; Khurana, A. Burden of Packaged Food on Schoolchildren: Based on the CSE Survey ‘Know Your Diet’; Centre for Science and Environment: New Delhi, India, 2017. [Google Scholar]
- Holt, R.I.; DeVries, J.H.; Hess-Fischl, A.; Hirsch, I.B.; Kirkman, M.S.; Klupa, T.; Ludwig, B.; Nørgaard, K.; Pettus, J.; Renard, E. The management of type 1 diabetes in adults. A consensus report by the American Diabetes Association (ADA) and the European Association for the Study of Diabetes (EASD). Diabetologia 2021, 64, 2609–2652. [Google Scholar] [CrossRef]
- Agnihorti, R.; Farooqui, A.; Khonde, N.; Mathews, R.P.; Sharma, S.; Gahlaud, S.; Manjul, S.K.; Manjul, A.; Sawlani, R. Microscopic, biochemical and stable isotopic investigation of seven multi-nutritional food-balls from Indus archaeological site, Rajasthan (India). J. Archaeol. Sci. Rep. 2021, 37, 102917. [Google Scholar] [CrossRef]
- Pandey, M.M.; Rastogi, S.; Rawat, A.K.S. Indian Traditional Ayurvedic System of Medicine and Nutritional Supplementation. Evid. -Based Complement. Altern. Med. 2013, 2013, 376327. [Google Scholar] [CrossRef] [Green Version]
- Dhanya, S.; Ramesh, N.; Mishra, A. Traditional methods of food habits and dietary preparations in Ayurveda—The Indian system of medicine. J. Ethn. Foods 2019, 6, 1–9. [Google Scholar]
- Devarajan, A.; Mohanmarugaraja, M. A comprehensive review on Rasam: A South Indian traditional functional food. Pharmacogn. Rev. 2017, 11, 73. [Google Scholar] [CrossRef] [Green Version]
- Swaminathan, S.; Vaz, M.; Kurpad, A.V. Protein intakes in India. Br. J. Nutr. 2012, 108, S50–S58. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hemalatha, R.; Laxmaiah, A.; Sriswan, M.; Boiroju, N.; Radhakrishna, K. What India Eats. 2020. Available online: https://www.nin.res.in/nutrition2020/what_india_eats.pdf (accessed on 9 December 2021).
- Shondelmyer, K.; Knight, R.; Sanivarapu, A.; Ogino, S.; Vanamala, J.K. Focus: Nutrition and Food Science: Ancient Thali Diet: Gut Microbiota, Immunity, and Health. Yale J. Biol. Med. 2018, 91, 177. [Google Scholar] [PubMed]
- Iriti, M.; Varoni, E.M. Pulses, healthy, and sustainable food sources for feeding the planet. Int. J. Mol. Sci. 2017, 18, 255. [Google Scholar] [CrossRef] [PubMed]
- National Institute of Nutrition. ICMR-NIN Expert Group on Nutrient Requirement for Indians: Recommended Dietary Allowances (RDA) and Estimated Average Requirements (EAR)-2020. Available online: https://www.nin.res.in/RDA_Full_Report_2020.html (accessed on 9 December 2021).
- Henry, C.J.; Quek, R.Y.C.; Kaur, B.; Shyam, S.; Singh, H.K.G. A glycaemic index compendium of non-western foods. Nutr. Diabetes 2021, 11, 2. [Google Scholar] [CrossRef] [PubMed]
- Das, A.; Raychaudhuri, U.; Chakraborty, R. Cereal based functional food of Indian subcontinent: A review. J. Food Sci. Technol. 2012, 49, 665–672. [Google Scholar] [CrossRef] [PubMed]
- Satish Kumar, R.; Kanmani, P.; Yuvaraj, N.; Paari, K.; Pattukumar, V.; Arul, V. Traditional Indian fermented foods: A rich source of lactic acid bacteria. Int. J. Food Sci. Nutr. Metab. Insights 2013, 64, 415–428. [Google Scholar] [CrossRef] [PubMed]
- Nigudkar, M. Estimation of resistant starch content of selected routinely consumed Indian food preparations. Curr. Res. Nutr. Food Sci. J. 2014, 2, 73–83. [Google Scholar] [CrossRef] [Green Version]
- Sonia, S.; Witjaksono, F.; Ridwan, R. Effect of cooling of cooked white rice on resistant starch content and glycemic response. Asia Pac. J. Clin. Nutr. 2015, 24, 620–625. [Google Scholar]
- Lu, L.W.; Venn, B.; Lu, J.; Monro, J.; Rush, E. Effect of Cold Storage and Reheating of Parboiled Rice on Postprandial Glycaemic Response, Satiety, Palatability and Chewed Particle Size Distribution. Nutrients 2017, 9, 475. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Boers, H.M.; Seijen Ten Hoorn, J.; Mela, D.J. A systematic review of the influence of rice characteristics and processing methods on postprandial glycaemic and insulinaemic responses. Br. J. Nutr. 2015, 114, 1035–1045. [Google Scholar] [CrossRef] [PubMed]
- Fabbri, A.D.T.; Crosby, G.A. A review of the impact of preparation and cooking on the nutritional quality of vegetables and legumes. Int. J. Gastron. Food Sci. 2016, 3, 2–11. [Google Scholar] [CrossRef] [Green Version]
- Kone, M.L.; Samayamanthula, D.R. Chapter33—Significance of conventional Indian foods acting as immune boosters to overcome COVID-19. In Environmental Resilience and Transformation in Times of COVID-19; Elseiver: Amsterdam, The Netherlands, 2021; pp. 385–396. [Google Scholar] [CrossRef]
- Rennie, C.; Wise, A. Preferences for steaming of vegetables. J. Hum. Nutr. Diet. 2010, 23, 108–110. [Google Scholar] [CrossRef]
- Kora, A.J. Leaves as dining plates, food wraps and food packing material: Importance of renewable resources in Indian culture. Bull. Natl. Res. Cent. 2019, 43, 1–5. [Google Scholar] [CrossRef]
- Sharma, N.; Gayathri, R.; Priya, V.V. Assessment of nutritional value of overnight soaked cooked rice over un-soaked cooked rice. Int. J. Res. Pharm. Sci. 2018, 9, 616–619. [Google Scholar]
- Behera, S.S.; El Sheikha, A.F.; Hammami, R.; Kumar, A. Traditionally fermented pickles: How the microbial diversity associated with their nutritional and health benefits? J. Funct. Foods 2020, 70, 103971. [Google Scholar] [CrossRef]
- Xing, Q.; Xing, X.; Zhang, Z.; Hu, X.; Liu, F. A comparative study of the nutritional values, volatiles compounds, and sensory qualities of pea pastes cooked in iron pot and clay pot. J Food Process Preserv. 2018, 42, e13328. [Google Scholar] [CrossRef]
- Adish, A.A.; Esrey, S.A.; Gyorkos, T.W.; Jean-Baptiste, J.; Rojhani, A. Effect of consumption of food cooked in iron pots on iron status and growth of young children: A randomised trial. Lancet (Lond. Engl.) 1999, 353, 712–716. [Google Scholar] [CrossRef]
- Krishnakumar, T. Traditional foods of India. Geoscience 2019. [Google Scholar] [CrossRef]
- Evert, A.B.; Boucher, J.L.; Cypress, M.; Dunbar, S.A.; Franz, M.J.; Mayer-Davis, E.J.; Neumiller, J.J.; Nwankwo, R.; Verdi, C.L.; Urbanski, P. Nutrition therapy recommendations for the management of adults with diabetes. Diabetes Care 2014, 37, S120–S143. [Google Scholar] [CrossRef] [Green Version]
- Canadian Diabetes Association Clinical Practice Guidelines Expert Committee. Clinical practice guidelines. Nutrition Therapy. Can. J. Diabetes 2013, 37, S45–S55. [Google Scholar]
- Chiavaroli, L.; Lee, D.; Ahmed, A.; Cheung, A.; Khan, T.; Mejia, S.B.; Mirrahimi, A.; Jenkins, D.; Livesey, G.; Wolever, T.; et al. Low Glycemic Index/Load Dietary Patterns and Glycemia and Cardiometabolic Risk Factors in Diabetes: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Curr. Dev. Nutr. 2021, 5, 1018. [Google Scholar] [CrossRef]
- Ranjan, A.; Schmidt, S.; Damm-Frydenberg, C.; Steineck, I.; Clausen, T.R.; Holst, J.J.; Madsbad, S.; Nørgaard, K. Low-carbohydrate diet impairs the effect of glucagon in the treatment of insulin-induced mild hypoglycemia: A randomized crossover study. Diabetes Care 2017, 40, 132–135. [Google Scholar] [CrossRef] [Green Version]
- Seckold, R.; Fisher, E.; De Bock, M.; King, B.; Smart, C. The ups and downs of low-carbohydrate diets in the management of Type 1 diabetes: A review of clinical outcomes. Diabet. Med. 2019, 36, 326–334. [Google Scholar] [CrossRef] [PubMed]
- Smart, C.E.M.; King, B.R.; Lopez, P.E. Insulin Dosing for Fat and Protein: Is it Time? Diabetes Care 2020, 43, 13–15. [Google Scholar] [CrossRef] [Green Version]
- Bell, K.J.; Fio, C.Z.; Twigg, S.; Duke, S.A.; Fulcher, G.; Alexander, K.; McGill, M.; Wong, J.; Brand-Miller, J.; Steil, G.M. Amount and Type of Dietary Fat, Postprandial Glycemia, and Insulin Requirements in Type 1 Diabetes: A Randomized Within-Subject Trial. Diabetes Care 2020, 43, 59–66. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vujosevic, S.; Pucci, P.; Casciano, M.; Daniele, A.; Bini, S.; Berton, M.; Cavarzeran, F.; Avogaro, A.; Lapolla, A.; Midena, E. A decade-long telemedicine screening program for diabetic retinopathy in the north-east of Italy. J. Diabetes Complicat. 2017, 31, 1348–1353. [Google Scholar] [CrossRef] [PubMed]
- Kar, S.; Khandelwal, B. Fast foods and physical inactivity are risk factors for obesity and hypertension among adolescent school children in east district of Sikkim, India. J. Nat. Sci. Biol. Med. 2015, 6, 356. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Joshi, S.R.; Bhansali, A.; Bajaj, S.; Banzal, S.S.; Dharmalingam, M.; Gupta, S.; Mukhopadhyay, S.; Shah, P.R.; Sahay, R.; Sarkar, S.; et al. Results from a dietary survey in an Indian T2DM population: A STARCH study. BMJ Open 2014, 4, e005138. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Srilakshmi, B. Proteins. In Nutrition Science, 4th ed.; New age international publishers: New Delhi, India, 2012; pp. 126–145. [Google Scholar]
- Weickert, M.O.; Pfeiffer, A.F. Impact of dietary fiber consumption on insulin resistance and the prevention of type 2 diabetes. J. Nutr. 2018, 148, 7–12. [Google Scholar] [CrossRef] [Green Version]
- Li, Y.O.; Komarek, A.R. Dietary fibre basics: Health, nutrition, analysis, and applications. Food Qual. Saf. 2017, 1, 47–59. [Google Scholar] [CrossRef]
- Neelakantan, N.; Narayanan, M.; de Souza, R.J.; van Dam, R.M. Effect of fenugreek (Trigonella foenum-graecum L.) intake on glycemia: A meta-analysis of clinical trials. Nutr. J. 2014, 13. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sharma, H.; Zhang, X.; Dwivedi, C. The effect of ghee (clarified butter) on serum lipid levels and microsomal lipid peroxidation. Ayu 2010, 31, 134. [Google Scholar] [CrossRef] [PubMed]
- Jenkins, D.J.; Kendall, C.W.; Augustin, L.S.; Vuksan, V. High-complex carbohydrate or lente carbohydrate foods? Am. J. Med. 2002, 113 (Suppl. 9B), 30s–37s. [Google Scholar] [CrossRef]
- Jenkins, D.J. Lente carbohydrate: A newer approach to the dietary management of diabetes. Diabetes Care 1982, 5, 634–641. [Google Scholar] [CrossRef] [PubMed]
- Falcomer, A.L.; Riquette, R.F.R.; de Lima, B.R.; Ginani, V.C.; Zandonadi, R.P. Health Benefits of Green Banana Consumption: A Systematic Review. Nutrients 2019, 11, 1222. [Google Scholar] [CrossRef] [Green Version]
- Hoffmann Sardá, F.A.; Giuntini, E.B.; Gomez, M.L.P.A.; Lui, M.C.Y.; Negrini, J.A.E.; Tadini, C.C.; Lajolo, F.M.; Menezes, E.W. Impact of resistant starch from unripe banana flour on hunger, satiety, and glucose homeostasis in healthy volunteers. J. Funct. Foods 2016, 24, 63–74. [Google Scholar] [CrossRef]
- Phillips, K.M.; McGinty, R.C.; Couture, G.; Pehrsson, P.R.; McKillop, K.; Fukagawa, N.K. Dietary fiber, starch, and sugars in bananas at different stages of ripeness in the retail market. PLoS ONE 2021, 16, e0253366. [Google Scholar] [CrossRef]
- Kittipongpatana, O.S.; Kittipongpatana, N. Resistant starch contents of native and heat-moisture treated jackfruit seed starch. Sci. World J. 2015, 2015, 519854. [Google Scholar] [CrossRef] [Green Version]
- Gopalan, C.; Rama Sastri, B.; Balasubramanian, S. Nutritive Value of Indian Foods; National Institute of Nutrition, ICMR: Hyderabad, India, 1971. [Google Scholar]
- Shobana, S.; Lakshmipriya, N.; Bai, M.R.; Gayathri, R.; Ruchi, V.; Sudha, V.; Malleshi, N.G.; Krishnaswamy, K.; Henry, C.K.; Anjana, R.M.; et al. Even minimal polishing of an Indian parboiled brown rice variety leads to increased glycemic responses. Asia Pac. J. Clin. Nutr. 2017, 26, 829–836. [Google Scholar] [CrossRef] [PubMed]
- Bhavadharini, B.; Mohan, V.; Dehghan, M.; Rangarajan, S.; Swaminathan, S.; Rosengren, A.; Wielgosz, A.; Avezum, A.; Lopez-Jaramillo, P.; Lanas, F.; et al. White Rice Intake and Incident Diabetes: A Study of 132,373 Participants in 21 Countries. Diabetes Care 2020, 43, 2643–2650. [Google Scholar] [CrossRef]
- Prasad, V.; Hymavathi, A.; Babu, V.R.; Longvah, T. Nutritional composition in relation to glycemic potential of popular Indian rice varieties. Food Chem. 2018, 238, 29–34. [Google Scholar] [CrossRef] [PubMed]
- Malik, V.S.; Sudha, V.; Wedick, N.M.; RamyaBai, M.; Vijayalakshmi, P.; Lakshmipriya, N.; Gayathri, R.; Kokila, A.; Jones, C.; Hong, B.; et al. Substituting brown rice for white rice on diabetes risk factors in India: A randomised controlled trial. Br. J. Nutr. 2019, 121, 1389–1397. [Google Scholar] [CrossRef] [PubMed]
- Priya, T.R.; Nelson, A.R.L.E.; Ravichandran, K.; Antony, U. Nutritional and functional properties of coloured rice varieties of South India: A review. J. Ethn. Foods 2019, 6. [Google Scholar] [CrossRef] [Green Version]
- Shobana, S.; Kokila, A.; Lakshmipriya, N.; Subhashini, S.; Ramya Bai, M.; Mohan, V.; Malleshi, N.G.; Anjana, R.M.; Henry, C.J.K.; Sudha, V. Glycaemic index of three Indian rice varieties. Int. J. Food Sci. Nutr. 2011, 63, 178–183. [Google Scholar] [CrossRef]
- Hamad, S.; Zafar, T.A.; Sidhu, J. Parboiled rice metabolism differs in healthy and diabetic individuals with similar improvement in glycemic response. Nutrition 2018, 47, 43–49. [Google Scholar] [CrossRef] [PubMed]
- Larsen, H.N.; Rasmussen, O.W.; Rasmussen, P.H.; Alstrup, K.K.; Biswas, S.K.; Tetens, I.; Thilsted, S.H.; Hermansen, K. Glycaemic index of parboiled rice depends on the severity of processing: Study in type 2 diabetic subjects. Eur. J. Clin. Nutr. 2000, 54, 380–385. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wolever, T.M.S.; Jenkins, D.J.A.; Kalmusky, J.; Jenkins, A.; Giordano, C.; Giudici, S.; Josse, R.G.; Wong, G.S. Comparison of regular and parboiled rices: Explanation of discrepancies between reported glycemic responses to rice. Nutr. Res. 1986, 6, 349–357. [Google Scholar] [CrossRef]
- Beenish, P.D.; Panda, B. Effect of cooking and ageing on total glycemic index of rice, under in vitro conditions. Int. J. Chem. Stud. 2018, 6, 603–606. [Google Scholar]
- Dixit, A.A.; Azar, K.M.; Gardner, C.D.; Palaniappan, L.P. Incorporation of whole, ancient grains into a modern Asian Indian diet to reduce the burden of chronic disease. Nutr. Rev. 2011, 69, 479–488. [Google Scholar] [CrossRef]
- Wee, M.S.; Henry, C.J. Reducing the glycemic impact of carbohydrates on foods and meals: Strategies for the food industry and consumers with special focus on Asia. Compr. Rev. Food Sci. Food Saf. 2020, 19, 670–702. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Radhika, G.; Sumathi, C.; Ganesan, A.; Vasudevan, S.; Henry, J.; Mohan, V. Glycaemic index of Indian flatbreads (rotis) prepared using whole wheat flour and GÇÿatta mixGÇÖ-added whole wheat flour. Br. J. Nutr. 2010, 103, 1642–1647. [Google Scholar] [CrossRef] [Green Version]
- Vega-López, S.; Ausman, L.M.; Griffith, J.L.; Lichtenstein, A.H. Interindividual variability and intra-individual reproducibility of glycemic index values for commercial white bread. Diabetes Care 2007, 30, 1412–1417. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chavan, J.K.; Kadam, S.S. Nutritional improvement of cereals by sprouting. Crit. Rev. Food Sci. Nutr. 1989, 28, 401–437. [Google Scholar] [CrossRef] [PubMed]
- Marchini, M.; Marti, A.; Folli, C.; Prandi, B.; Ganino, T.; Conte, P.; Fadda, C.; Mattarozzi, M.; Carini, E. Sprouting of Sorghum (Sorghum bicolor [L.] Moench): Effect of Drying Treatment on Protein and Starch Features. Foods 2021, 10, 407. [Google Scholar] [CrossRef] [PubMed]
- Mundra, A.; Yenagi, N.B.; Kasturiba, B. Designing of low glycaemic chapati of dicoccum wheat for the effective management of diabetes. Karnataka J. Agric. Sci. 2010, 23, 476–479. [Google Scholar]
- Bhuvaneswari, G.; Yenagi, N.; Hanchinal, R. Carbohydrate profile of dicoccum wheat varieties. Karnataka J. Agric. Sci. 2010, 17, 781–786. [Google Scholar]
- Mansoor, R.; Mohsin Ali, T.; Arif, S.; Moin, A.; Hasnain, A. Effects of barley flour on rheological, textural, sensory and glycemic index of traditional unleavened flat bread (Roti). Cereal Chem. 2019, 96, 1170–1179. [Google Scholar] [CrossRef]
- Tharifkhan, S.A.; Perumal, A.B.; Elumalai, A.; Moses, J.A.; Anandharamakrishnan, C. Improvement of nutrient bioavailability in millets: Emphasis on the application of enzymes. J. Sci. Food Agric. 2021, 101, 4869–4878. [Google Scholar] [CrossRef] [PubMed]
- Anitha, S.; Kane-Potaka, J.; Tsusaka, T.W.; Botha, R.; Rajendran, A.; Givens, D.I.; Parasannanavar, D.J.; Subramaniam, K.; Prasad, K.D.V.; Vetriventhan, M.; et al. A Systematic Review and Meta-Analysis of the Potential of Millets for Managing and Reducing the Risk of Developing Diabetes Mellitus. Front. Nutr. 2021, 8, 687428. [Google Scholar] [CrossRef]
- Krishnan, R.; Meera, M. Pearl millet minerals: Effect of processing on bioaccessibility. J. Food Sci. Technol. 2018, 55, 3362–3372. [Google Scholar] [CrossRef]
- Thingnganing, L.; Rajendran, A.; Bhaskar, K.; Venkaiah, K. Indian Food Composition Table (IFCT); NIN-ICMR: Hyderabad, India, 2017; Available online: https://www.academia.edu/31170922/Indian_Food_Composition_Tables (accessed on 9 December 2021).
- Shobana, S.; Selvi, R.P.; Kavitha, V.; Gayathri, N.; Geetha, G.; Gayathri, R.; Vijayalakshmi, P.; Balasubramaniam, K.K.G.; Ruchi, V.; Sudha, V.; et al. Development and evaluation of nutritional, sensory and glycemic properties of finger millet (Eleusine coracana L.) based food products. Asia Pac. J. Clin. Nutr. 2018, 27, 84–91. [Google Scholar] [CrossRef] [PubMed]
- Szczepaniak, W.; Grzebisz, W.; Potarzycki, J.; Lukowiak, R.; Przygocka-Cyna, K. The magnesium and calcium mineral status of maize at physiological maturity as a tool for an evaluation of yield forming conditions. J. Elem. 2016, 21, 881–897. [Google Scholar] [CrossRef]
- Freitas, D.; Le Feunteun, S.J. Acid induced reduction of the glycaemic response to starch-rich foods: The salivary α-amylase inhibition hypothesis. Food Funct. 2018, 9, 5096–5102. [Google Scholar] [CrossRef]
- The International Glycemic Index (GI) Database; The University of Sydney: Sydney, Australia, 2012; Available online: https://researchdata.edu.au/international-glycemic-index-gi-database/11115 (accessed on 9 December 2021).
- Joseph, J.; Rao, A.G.; Nayak, S. 719-P: A Randomized, Double-Blind, Placebo-Controlled Study to Evaluate the Glucose-Lowering Effect of Green Jackfruit Flour in Type 2 Diabetes Mellitus Patients. Diabetes 2020, 69. [Google Scholar] [CrossRef]
- Ogbuji, C.A.; Odom, T.C.; Ndulaka, J.C.; Ogbodo, M.O. Effects of various processing methods of ripe and unripe plantain diets on blood glucose level. Eur. J. Biol. Med. Sci. Res. 2013, 1, 49–54. [Google Scholar]
- Rao, A.G.; Naik, K.S.; Unnikrishnan, A.G.; Joseph, J. Efficacy of green jackfruit flour as a medical nutrition therapy replacing rice or wheat in patients with type 2 diabetes mellitus: A randomized, double-blind, placebo-controlled study. Nutr. Diabetes 2021, 11, 18. [Google Scholar] [CrossRef]
- Ranasinghe, R.A.S.N.; Maduwanthi, S.D.T.; Marapana, R.A.U.J. Nutritional and Health Benefits of Jackfruit (Artocarpus heterophyllus Lam.): A Review. Int. J. Food Sci. 2019, 2019, 4327183. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kasar, C.; Thanushree, M.P.; Gupta, S.; Inamdar, A.A. Milled fractions of common buckwheat (Fagopyrum esculentum) from the Himalayan regions: Grain characteristics, functional properties and nutrient composition. J. Food Sci. Technol. 2020, 58, 3871–3881. [Google Scholar] [CrossRef]
- Chaturvedi, A.; Sarojini, G.; Nirmala, G.; Nirmalamma, N.; Satyanarayana, D. Glycemic index of grain amaranth, wheat and rice in NIDDM subjects. Plant Foods Hum. Nutr. (Dordr. Neth.) 1997, 50, 171–178. [Google Scholar] [CrossRef]
- Shanmugam, S.; Ramyabai, M.; Vasudevan, S.; Unnikrishnan, R.; Anjana, R.; Mohan, V. Nutrition and its Link with Diabetes in Asian Indians: Challenges and Solutions. Proc. Indian Natl. Sci. Acad. 2018, 84. [Google Scholar] [CrossRef]
- Misra, A.; Sharma, R.; Gulati, S.; Joshi, S.R.; Sharma, V.; Ibrahim, A.; Joshi, S.; Laxmaiah, A.; Kurpad, A.; Raj, R.K.; et al. Consensus dietary guidelines for healthy living and prevention of obesity, the metabolic syndrome, diabetes, and related disorders in Asian Indians. Diabetes Technol. Ther. 2011, 13, 683–694. [Google Scholar] [CrossRef]
- Shridhar, K.; Dhillon, P.K.; Bowen, L.; Kinra, S.; Bharathi, A.V.; Prabhakaran, D.; Reddy, K.S.; Ebrahim, S. Nutritional profile of Indian vegetarian diets–the Indian Migration Study (IMS). Nutr. J. 2014, 13, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Arens, U.; Convery, L.; Garton, L.; Hood, S.; Mason, P.; Pettinger, C.; Medici, E.; Haffner, T.; Arthur, K.; Lewis, J. One Blue Dot Eating Patterns for Health and Environmental Sustainability: A Reference Guide for Dietitians. 2018. Available online: https://www.bda.uk.com/uploads/assets/539e2268-7991-4d24-b9ee867c1b2808fc/a1283104-a0dd-476b-bda723452ae93870/one%20blue%20dot%20reference%20guide.pdf (accessed on 9 December 2021).
- Tso, R.; Forde, C.G. Unintended Consequences: Nutritional Impact and Potential Pitfalls of Switching from Animal- to Plant-Based Foods. Nutrients 2021, 13, 2527. [Google Scholar] [CrossRef]
- Mariotti, F.; Gardner, C.D. Dietary Protein and Amino Acids in Vegetarian Diets-A Review. Nutrients 2019, 11, 2661. [Google Scholar] [CrossRef] [Green Version]
- Tromba, V.; Silvestri, F. Vegetarianism and type 1 diabetes in children. Metabol. Open 2021, 11, 100099. [Google Scholar] [CrossRef] [PubMed]
- Singh, M.; Manickavasagan, A.; Shobana, S.; Mohan, V. Glycemic index of pulses and pulse-based products: A review. Crit. Rev. Food Sci. Nutr. 2021, 61, 1567–1588. [Google Scholar] [CrossRef]
- Eleazu, C.O. The concept of low glycemic index and glycemic load foods as panacea for type 2 diabetes mellitus; prospects, challenges and solutions. Afr. Health Sci. 2016, 16, 468–479. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Marton, L.T.; Pescinini-e-Salzedas, L.M.; Camargo, M.E.C.; Barbalho, S.M.; Haber, J.F.d.S.; Sinatora, R.V.; Detregiachi, C.R.P.; Girio, R.J.S.; Buchaim, D.V.; Cincotto dos Santos Bueno, P. The Effects of Curcumin on Diabetes Mellitus: A Systematic Review. Front. Endocrinol. 2021, 12, 443. [Google Scholar] [CrossRef] [PubMed]
- Facioni, M.S.; Raspini, B.; Pivari, F.; Dogliotti, E.; Cena, H. Nutritional management of lactose intolerance: The importance of diet and food labelling. J. Transl. Med. 2020, 18, 260. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Rai, D.C.; Niranjan, K.; Bhat, Z.F. Paneer-An Indian soft cheese variant: A review. J. Food Sci. Technol. 2014, 51, 821–831. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lalhriatpuii, M.; Kumar Singh, A. Goat Meat: No Less Source of Protein in Comparison to Other Meat for Human Consumption; IntechOpen: London, UK, 2021. [Google Scholar]
- Sacks, F.M.; Lichtenstein, A.H.; Wu, J.H.; Appel, L.J.; Creager, M.A.; Kris-Etherton, P.M.; Miller, M.; Rimm, E.B.; Rudel, L.L.; Robinson, J.G. Dietary fats and cardiovascular disease: A presidential advisory from the American Heart Association. Circulation 2017, 136, e1–e23. [Google Scholar] [CrossRef] [PubMed]
- Mani, I.; Kurpad, A.V. Fats & fatty acids in Indian diets: Time for serious introspection. Indian J. Med Res. 2016, 144, 507. [Google Scholar]
- Teng, M.; Zhao, Y.J.; Khoo, A.L.; Yeo, T.C.; Yong, Q.W.; Lim, B.P. Impact of coconut oil consumption on cardiovascular health: A systematic review and meta-analysis. Nutr. Rev. 2020, 78, 249–259. [Google Scholar] [CrossRef]
- Ros, E. Health benefits of nut consumption. Nutrients 2010, 2, 652–682. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Salis, S.; Joseph, M.; Agarwala, A.; Sharma, R.; Kapoor, N.; Irani, A.J. Medical nutrition therapy of pediatric type 1 diabetes mellitus in India: Unique aspects and challenges. Pediatric Diabetes 2021, 22, 93–100. [Google Scholar] [CrossRef] [PubMed]
- Vijayakumar, V.; Shankar, N.; Nanjundaiah, R.; Mooventhan, A.; Anju, S.; Krishnamurthy, M. Diet enriched with fresh coconut decreases blood glucose levels and body weight in normal adults. J. Complement. Integr. Med. 2018, 15. [Google Scholar] [CrossRef] [PubMed]
- Comprehensive National Nutrition Survey (CNNS) National Report. Ministry of Health and Family Welfare (MoHFW); Government of India, UNICEF and Population Council: New Delhi, India, 2019. [Google Scholar]
- Bharadva, K.; Mishra, S.; Tiwari, S.; Yadav, B.; Deshmukh, U.; Elizabeth, K.E.; Banapurmath, C.R. Prevention of Micronutrient Deficiencies in Young Children: Consensus Statement from Infant and Young Child Feeding Chapter of Indian Academy of Pediatrics. Indian Pediatrics 2019, 56, 577–586. [Google Scholar] [CrossRef] [PubMed]
- Parthasarathy, L.; Chiplonkar, S.; Khadilkar, A.; Khadilkar, V. Dietary modifications to improve micronutrient status of Indian children and adolescents with type 1 diabetes. Asia Pac. J. Clin. Nutr. 2015, 24, 73–82. [Google Scholar] [CrossRef]
- Ebbeling, C.B.; Feldman, H.A.; Chomitz, V.R.; Antonelli, T.A.; Gortmaker, S.L.; Osganian, S.K.; Ludwig, D.S. A randomized trial of sugar-sweetened beverages and adolescent body weight. N. Engl. J. Med. 2012, 367, 1407–1416. [Google Scholar] [CrossRef] [Green Version]
- Evans, S.F.; Meister, M.; Mahmood, M.; Eldoumi, H.; Peterson, S.; Perkins-Veazie, P.; Clarke, S.L.; Payton, M.; Smith, B.J.; Lucas, E.A. Mango supplementation improves blood glucose in obese individuals. Nutr. Metab. Insights 2014, 7, NMI-S17028. [Google Scholar] [CrossRef]
- Seshadri, S.; Nambiar, V. Kanjero (Digera arvensis) and Drumstick Leaves (Moringa oleifera): Nutrient Profile and Potential for Human Consumption. World Rev. Nutr. Diet. 2003, 91, 41–59. [Google Scholar] [CrossRef]
- Nova, E.; Redondo-Useros, N.; Martínez-García, R.M.; Gómez-Martínez, S.; Díaz-Prieto, L.E.; Marcos, A. Potential of Moringa oleifera to Improve Glucose Control for the Prevention of Diabetes and Related Metabolic Alterations: A Systematic Review of Animal and Human Studies. Nutrients 2020, 12, 2050. [Google Scholar] [CrossRef]
- D’Souza, J.; D’Souza, P.; Fazal, F.; Kumar, A.; Bhat, H.; Baliga, S. Anti-diabetic effects of the Indian indigenous fruit Emblica officinalis Gaertn: Active constituents and modes of action. Food Funct. 2014, 5, 635–644. [Google Scholar] [CrossRef]
- Davani-Davari, D.; Negahdaripour, M.; Karimzadeh, I.; Seifan, M.; Mohkam, M.; Masoumi, S.J.; Berenjian, A.; Ghasemi, Y. Prebiotics: Definition, Types, Sources, Mechanisms, and Clinical Applications. Foods. 2019, 8, 92. [Google Scholar] [CrossRef] [Green Version]
- Melse-Boonstra, A. Bioavailability of Micronutrients from Nutrient-Dense Whole Foods: Zooming in on Dairy, Vegetables, and Fruits. Front. Nutr. 2020, 7, 101. [Google Scholar] [CrossRef] [PubMed]
- Sharma, R.; Garg, P.; Kumar, P.; Bhatia, S.K.; Kulshrestha, S. Microbial fermentation and its role in quality improvement of fermented foods. Fermentation 2020, 6, 106. [Google Scholar] [CrossRef]
- Mattson, M.P.; Allison, D.B.; Fontana, L.; Harvie, M.; Longo, V.D.; Malaisse, W.J.; Mosley, M.; Notterpek, L.; Ravussin, E.; Scheer, F.A. Meal frequency and timing in health and disease. Proc. Natl. Acad. Sci. USA 2014, 111, 16647–16653. [Google Scholar] [CrossRef] [Green Version]
- Dennis, M.; Lee, A.R.; McCarthy, T. Nutritional Considerations of the Gluten-Free Diet. Gastroenterol. Clin. N. Am. 2019, 48, 53–72. [Google Scholar] [CrossRef] [PubMed]
- Islas, J.F.; Acosta, E.; G-Buentello, Z.; Delgado-Gallegos, J.L.; Moreno-Treviño, M.G.; Escalante, B.; Moreno-Cuevas, J.E. An overview of Neem (Azadirachta indica) and its potential impact on health. J. Funct. Foods 2020, 74, 104171. [Google Scholar] [CrossRef]
- Ganesan, K.; Xu, B. Polyphenol-Rich Lentils and Their Health Promoting Effects. Int. J. Mol. Sci. 2017, 18, 2390. [Google Scholar] [CrossRef] [Green Version]

| Cooking Methods | Nutrition Highlights |
|---|---|
| Combination of cereal and pulse (Khichdi/pongal/idli/dosa/dhokla/dal baati/zunka or pitla bhakar/dal dhokli/handvo/thalipeet/puttu kadala/dal paratha) | Cereals and pulses, when combined in the ratio of 3:1, complement one another, providing complete protein with all essential amino acids [15,16] Decrease GI of meals [17] |
| Fermentation (idli/dosa/dhokla/enduri pitha/curd/selroti/ambeli/khaman/sez/adai/vada) * | This produces beneficial enzymes which aid in digestion.The microorganisms produce vitamin B12 and β-galactosidase: the enhanced probiotic activity helps maintain gut health and improves immunity. Fermentation of pulses reduces anti-nutritional factors such as phytic acid, thereby increasing nutrient availability. Propensity to flatulence is decreased.Overnight fermentation of idli and dosa batter increases the vitamins B and C content. Fermentation of milk into yogurt converts lactose into lactic acid: beneficial for those with lactose intolerance [18,19]. |
| Cooking and cooling starchy foods | Retrogradation (re-crystallization of starch polymer chains, which occurs after the gelatinization) increases the resistant starch type 3 (RS 3) content, reducing the GI. RS3 is formed when starchy foods are cooked, cooled and stored for several hours. Cooked and cooled potatoes, rice and legumes have been shown to contain significant amounts of RS3 [20,21,22,23] |
| Soaking (pulses and cereals) | Soaking before cooking reduces cooking time, improves digestibility, and reduces anti-nutritional factors like phytates, tannins and enzyme inhibitors [15,24,25]. |
| Steaming (vegetables) | One of the best cooking methods for preserving nutrients, including water-soluble vitamins that are sensitive to heat and water, like Vitamin C and B complex, and phytochemicals [24,25,26] |
| Sand Roasting (Popped, puffed, and flaked rice and maize; roasted cereals and millets; roasted legumes such as groundnut, chickpea, pea, cowpea) | It is the simplest, most inexpensive method of dry heat application. High-temperature short-time treatment in sand results in higher puffing, crispiness, volume, improved color, aroma, flavor, and texture while enhancing shelf life. The sand roasting process enhances carbohydrate and protein digestibility, β-glucan extractability, levels of prebiotic dietary fiber, minerals, and antioxidants; and reduces the inherent anti-nutrient levels in cereals and legumes. The destruction of seed microflora enhances the shelf life and consumer acceptance. These roasted rice, wheat, maize, millets and legume preparations are healthy alternatives to current-day unhealthy snacks [27] |
| Soaking and fermenting cooked rice overnight (pakhala, panta bhaat) | This process increases vitamin B6 and B12 content. This process also increases beneficial bacteria, which helps in digestion, improves gut health and boosts immunity [28]. |
| Pickling (vegetables, fruits, fish, chicken, meat) | Pickling, one of the oldest methods of preservation, imparts unique and desirable changes in flavor, texture and color. It also increases the probiotic potential [29]. |
| Sprouting (green gram, chickpeas, Bengal gram, other legumes) | Sprouting increases the content of vitamin C and some B-group vitamins. This also reduces phytic acid and enhances the absorption of zinc. This also enriches vitamin A [25]. |
| Cooking in earthen pots | Traditionally, Indian households cooked in earthen and iron pots. Benefits include the ability of earthen vessels to absorb moisture (due to their porous nature) and let heat circulate evenly and slowly through the food being cooked, making it aromatic and retaining nutrition. Clay being alkaline balances the pH and neutralizes acidity. It adds many important nutrients like calcium, magnesium, iron and phosphorus. Cooking in a clay pot needs much less oil [28,30]. (Clay pots available today are often glazed with substances containing lead, mercury and others which are hazardous for health. Unglazed, pure earthen pots are used for cooking, after soaking in water for a few hours) |
| Cooking in iron pots | It is a useful method of iron fortification and prevents iron-deficiency anemia [30,31] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Salis, S.; Virmani, A.; Priyambada, L.; Mohan, M.; Hansda, K.; Beaufort, C.d. ‘Old Is Gold’: How Traditional Indian Dietary Practices Can Support Pediatric Diabetes Management. Nutrients 2021, 13, 4427. https://doi.org/10.3390/nu13124427
Salis S, Virmani A, Priyambada L, Mohan M, Hansda K, Beaufort Cd. ‘Old Is Gold’: How Traditional Indian Dietary Practices Can Support Pediatric Diabetes Management. Nutrients. 2021; 13(12):4427. https://doi.org/10.3390/nu13124427
Chicago/Turabian StyleSalis, Sheryl, Anju Virmani, Leena Priyambada, Meena Mohan, Kajal Hansda, and Carine de Beaufort. 2021. "‘Old Is Gold’: How Traditional Indian Dietary Practices Can Support Pediatric Diabetes Management" Nutrients 13, no. 12: 4427. https://doi.org/10.3390/nu13124427
APA StyleSalis, S., Virmani, A., Priyambada, L., Mohan, M., Hansda, K., & Beaufort, C. d. (2021). ‘Old Is Gold’: How Traditional Indian Dietary Practices Can Support Pediatric Diabetes Management. Nutrients, 13(12), 4427. https://doi.org/10.3390/nu13124427






