The Effectiveness of Extra Virgin Olive Oil and the Traditional Brazilian Diet in Reducing the Inflammatory Profile of Individuals with Severe Obesity: A Randomized Clinical Trial
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Design, Data Collection, and Ethical Aspects
2.2. Inclusion and Exclusion Criteria
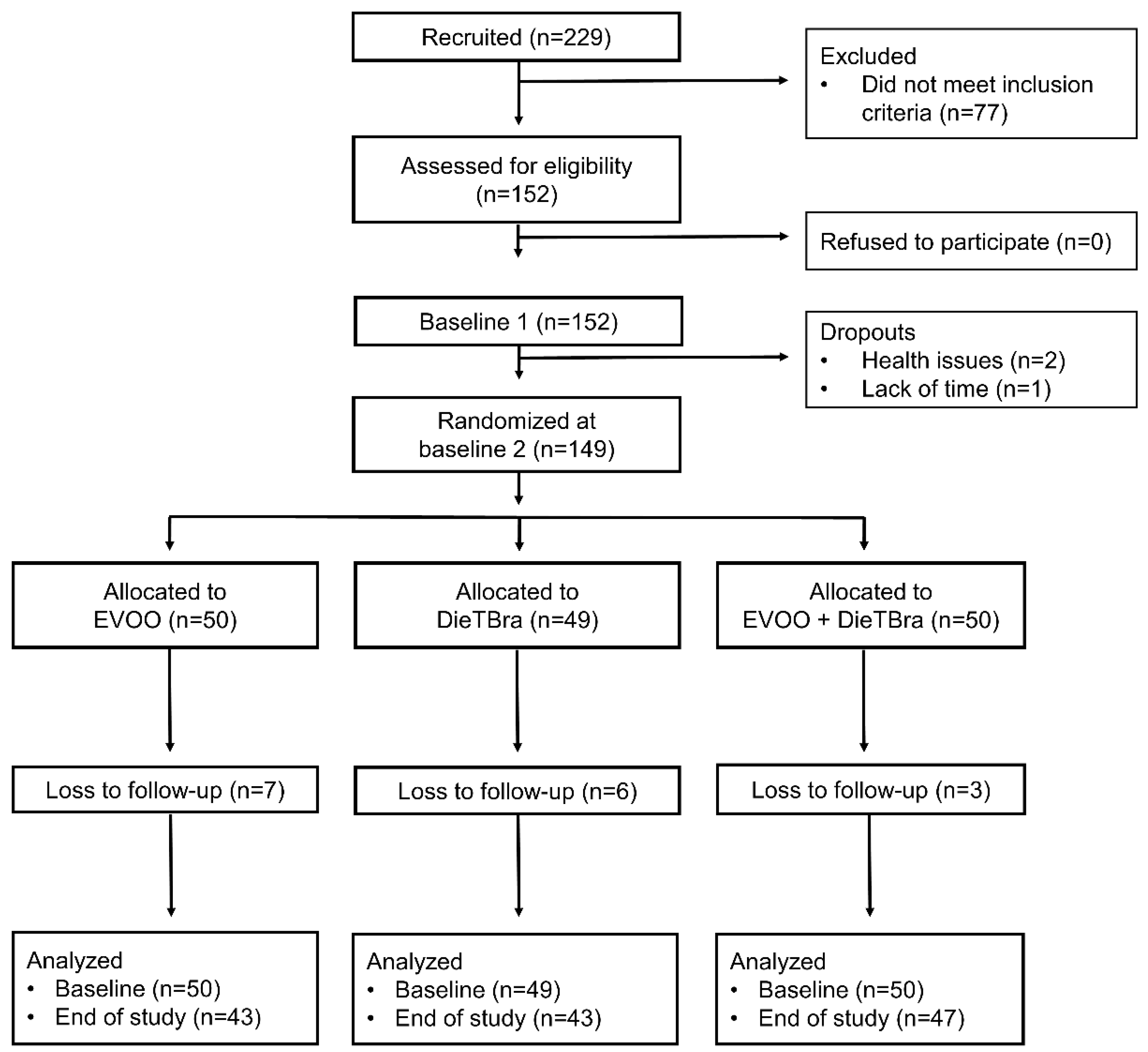
2.3. Participants, Randomization, and Study Phases
2.4. Interventions
2.5. Blinding
2.6. Variables and Outcomes
2.6.1. Anthropometric Variables
2.6.2. Biochemical Analysis
2.7. Quality Control
2.8. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Poirier, P.; Alpert, M.A.; Fleisher, L.A.; Thompson, P.D.; Sugerman, H.J.; Burke, L.E.; Marceau, P.; Franklin, B.A. Cardiovascular evaluation and management of severely obese patients undergoing surgery: A science advisory from the American Heart Association. Circulation 2009, 120, 86–95. [Google Scholar] [CrossRef] [Green Version]
- Calle, E.E.; Rodriguez, C.; Walker-Thurmond, K.; Thun, M.J. Overweight, obesity, and mortality from cancer in a prospectively studied cohort of U.S. adults. N. Engl. J. Med. 2003, 348, 1625–1638. [Google Scholar] [CrossRef] [Green Version]
- Cardoso, C.K.D.S.; Santos, A.S.E.A.D.C.; Rosa, L.P.D.S.; Mendonça, C.R.; Vitorino, P.V.D.O.; Peixoto, M.D.R.G.; Silveira, É.A. Effect of Extra Virgin Olive Oil and Traditional Brazilian Diet on the Bone Health Parameters of Severely Obese Adults: A Randomized Controlled Trial. Nutrients 2020, 12, 403. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Collins, J.; Meng, C.; Eng, A. Psychological Impact of Severe Obesity. Curr. Obes. Rep. 2016, 5, 435–440. [Google Scholar] [CrossRef] [PubMed]
- Prospective Studies, C. Body-mass index and cause-specific mortality in 900000 adults: Collaborative analyses of 57 prospective studies. Lancet 2009, 373, 1083–1096. [Google Scholar] [CrossRef] [Green Version]
- Kivimäki, M.; Kuosma, E.; Ferrie, J.E.; Luukkonen, R.; Nyberg, S.T.; Alfredsson, L.; Batty, G.D.; Brunner, E.J.; Fransson, E.; Goldberg, M.; et al. Overweight, obesity, and risk of cardiometabolic multimorbidity: Pooled analysis of individual-level data for 120,813 adults from 16 cohort studies from the USA and Europe. Lancet Public Health 2017, 2, e277–e285. [Google Scholar] [CrossRef] [Green Version]
- Silveira, E.A.; Kliemann, N.; Noll, M.; Sarrafzadegan, N.; de Oliveira, C. Visceral obesity and incident cancer and cardiovascular disease: An integrative review of the epidemiological evidence. Obes. Rev. 2021, 22, e13088. [Google Scholar] [CrossRef]
- Herder, C.; Schneitler, S.; Rathmann, W.; Haastert, B.; Schneitler, H.; Winkler, H.; Bredahl, R.; Hahnloser, E.; Martin, S. Low-grade inflammation, obesity, and insulin resistance in adolescents. J. Clin. Endocrinol. Metab. 2007, 92, 4569–4574. [Google Scholar] [CrossRef]
- Valle, M.; Martos, R.; Gascón, F.; Cañete, R.; Zafra, M.A.; Morales, R. Low-grade systemic inflammation, hypoadiponectinemia and a high concentration of leptin are present in very young obese children, and correlate with metabolic syndrome. Diabetes Metab. 2005, 31, 55–62. [Google Scholar] [CrossRef]
- Rodríguez-Rodríguez, E.; López-Sobaler, A.M.; Ortega, R.M.; Delgado-Losada, M.L.; López-Parra, A.M.; Aparicio, A. Association between Neutrophil-to-Lymphocyte Ratio with Abdominal Obesity and Healthy Eating Index in a Representative Older Spanish Population. Nutrients 2020, 12, 855. [Google Scholar] [CrossRef] [Green Version]
- Aparecida Silveira, E.; Vaseghi, G.; de Carvalho Santos, A.S.; Kliemann, N.; Masoudkabir, F.; Noll, M.; Mohammadifard, N.; Sarrafzadegan, N.; de Oliveira, C. Visceral Obesity and Its Shared Role in Cancer and Cardiovascular Disease: A Scoping Review of the Pathophysiology and Pharmacological Treatments. Int. J. Mol. Sci. 2020, 21, 9042. [Google Scholar] [CrossRef]
- Kim, J.; Nam, J.-H. Insight into the relationship between obesity-induced low-level chronic inflammation and COVID-19 infection. Int. J. Obes. 2020, 44, 1541–1542. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, J.; Fialho, M.; Santos, R.; Peixoto-Plácido, C.; Madeira, T.; Sousa-Santos, N.; Virgolino, A.; Santos, O.; Vaz Carneiro, A. Is olive oil good for you? A systematic review and meta-analysis on anti-inflammatory benefits from regular dietary intake. Nutrition 2020, 69, 110559. [Google Scholar] [CrossRef]
- Semeniuk-Wojtas, A.; Lubas, A.; Stec, R.; Syrylo, T.; Niemczyk, S.; Szczylik, C. Neutrophil-to-lymphocyte Ratio, Platelet-to-lymphocyte Ratio, and C-reactive Protein as New and Simple Prognostic Factors in Patients with Metastatic Renal Cell Cancer Treated With Tyrosine Kinase Inhibitors: A Systemic Review and Meta-analysis. Clin. Genitourin. Cancer 2018, 16, e685–e693. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Avan, A.; Tavakoly Sany, S.B.; Ghayour-Mobarhan, M.; Rahimi, H.R.; Tajfard, M.; Ferns, G. Serum C-reactive protein in the prediction of cardiovascular diseases: Overview of the latest clinical studies and public health practice. J. Cell. Physiol. 2018, 233, 8508–8525. [Google Scholar] [CrossRef] [PubMed]
- Ryder, E.; Diez-Ewald, M.; Mosquera, J.; Fernández, E.; Pedreañez, A.; Vargas, R.; Peña, C.; Fernández, N. Association of obesity with leukocyte count in obese individuals without metabolic syndrome. Diabetes Metab. Syndr. 2014, 8, 197–204. [Google Scholar] [CrossRef]
- Vahit, D.; Akboga, M.K.; Samet, Y.; Hüseyin, E. Assessment of monocyte to high density lipoprotein cholesterol ratio and lymphocyte-to-monocyte ratio in patients with metabolic syndrome. Biomark. Med. 2017, 11, 535–540. [Google Scholar] [CrossRef]
- Garanty-Bogacka, B.; Syrenicz, M.; Goral, J.; Krupa, B.; Syrenicz, J.; Walczak, M.; Syrenicz, A. Changes in inflammatory biomarkers after successful lifestyle intervention in obese children. Endokrynol. Pol. 2011, 62, 499–505. [Google Scholar]
- Yubero-Serrano, E.M.; Lopez-Moreno, J.; Gomez-Delgado, F.; Lopez-Miranda, J. Extra virgin olive oil: More than a healthy fat. Eur. J. Clin. Nutr. 2019, 72, 8–17. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rees, K.; Takeda, A.; Martin, N.; Ellis, L.; Wijesekara, D.; Vepa, A.; Das, A.; Hartley, L.; Stranges, S. Mediterranean-style diet for the primary and secondary prevention of cardiovascular disease. Cochrane Database Syst. Rev. 2019, 3, Cd009825. [Google Scholar] [CrossRef] [Green Version]
- Rodrigues, A.P.S.; Rosa, L.P.S.; Silveira, E.A. PPARG2 Pro12Ala polymorphism influences body composition changes in severely obese patients consuming extra virgin olive oil: A randomized clinical trial. Nutr. Metab. 2018, 15, 52. [Google Scholar] [CrossRef] [PubMed]
- Canheta, A.B.S.; Santos, A.; Souza, J.D.; Silveira, E.A. Traditional Brazilian diet and extra virgin olive oil reduce symptoms of anxiety and depression in individuals with severe obesity: Randomized clinical trial. Clin. Nutr. 2021, 40, 404–411. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, A.P.; Rosa, L.P.; Noll, M.; Silveira, E.A. Traditional Brazilian Diet and Olive Oil Reduce Cardiometabolic Risk Factors in Severely Obese Individuals: A Randomized Trial. Nutrients 2020, 12, 1413. [Google Scholar] [CrossRef]
- Aparecida Silveira, E.; Danésio de Souza, J.; Dos Santos Rodrigues, A.P.; Lima, R.M.; de Souza Cardoso, C.K.; de Oliveira, C. Effects of Extra Virgin Olive Oil (EVOO) and the Traditional Brazilian Diet on Sarcopenia in Severe Obesity: A Randomized Clinical Trial. Nutrients 2020, 12, 1498. [Google Scholar] [CrossRef]
- Canheta, A.B.S.; Santos, A.; de Souza, J.D.; Silveira, E.A. Adequate statistical analyses and inferences of Randomized Clinical Trial. Clin. Nutr. 2021, 40, 334–335. [Google Scholar] [CrossRef] [PubMed]
- Brasil. Guia Alimentar Para a População Brasileira, 2nd ed.; 1. reimpr; Ministério Da Saúde, Secretaria de Atenção à Saúde, Departamento de Atenção Básica: Brasília, Brazil, 2014; 156p.
- Hall, K.D. What is the required energy deficit per unit weight loss? Int. J. Obes. 2008, 32, 573–576. [Google Scholar] [CrossRef] [Green Version]
- Horie, L.M.; Gonzalez, M.C.; Torrinhas, R.S.; Cecconello, I.; Waitzberg, D.L. New specific equation to estimate resting energy expenditure in severely obese patients. Obesity 2011, 19, 1090–1094. [Google Scholar] [CrossRef]
- World Health Organization. Obesity: Preventing and Managing the Global Epidemic; WHO: Geneva, Switzerland, 2000. [Google Scholar]
- Hill, J.O.; Wyatt, H.R.; Peters, J.C. Energy balance and obesity. Circulation 2012, 126, 126–132. [Google Scholar] [CrossRef]
- Medicine, I.O. Dietary Reference Intakes for Energy, Carbohydrate, Fiber, Fat, Fatty Acids, Cholesterol, Protein, and Amino Acids; The National Academies Press: Washington, DC, USA, 2005; p. 1358. [Google Scholar]
- Satija, A.; Yu, E.; Willett, W.C.; Hu, F.B. Understanding nutritional epidemiology and its role in policy. Adv. Nutr. 2015, 6, 5–18. [Google Scholar] [CrossRef] [Green Version]
- Gupte, A.A.; Lyon, C.J.; Hsueh, W.A. Nuclear factor (erythroid-derived 2)-like-2 factor (Nrf2), a key regulator of the antioxidant response to protect against atherosclerosis and nonalcoholic steatohepatitis. Curr. Diab. Rep. 2013, 13, 362–371. [Google Scholar] [CrossRef] [PubMed]
- Chae, J.S.; Paik, J.K.; Kang, R.; Kim, M.; Choi, Y.; Lee, S.H.; Lee, J.H. Mild weight loss reduces inflammatory cytokines, leukocyte count, and oxidative stress in overweight and moderately obese participants treated for 3 years with dietary modification. Nutr. Res. 2013, 33, 195–203. [Google Scholar] [CrossRef]
- Hernaez, A.; Lassale, C.; Castro-Barquero, S.; Babio, N.; Ros, E.; Castaner, O.; Tresserra-Rimbau, A.; Pinto, X.; Martinez-Gonzalez, M.A.; Corella, D.; et al. Mediterranean Diet and White Blood Cell Count-A Randomized Controlled Trial. Foods 2021, 10, 1268. [Google Scholar] [CrossRef]
- Arpón, A.; Milagro, F.I.; Estruch, R.; Fitó, M.; Marti, A.; Martínez-González, M.A.; Ros, E.; Salas-Salvadó, J.; Riezu-Boj, J.I.; Martínez, J.A.; et al. Impact of Consuming Extra-Virgin Olive Oil or Nuts within a Mediterranean Diet on DNA Methylation in Peripheral White Blood Cells within the PREDIMED-Navarra Randomized Controlled Trial: A Role for Dietary Lipids. Nutrients 2017, 10, 15. [Google Scholar] [CrossRef] [Green Version]
- Widmer, R.J.; Freund, M.A.; Flammer, A.J.; Sexton, J.; Lennon, R.; Romani, A.; Mulinacci, N.; Vinceri, F.F.; Lerman, L.O.; Lerman, A. Beneficial effects of polyphenol-rich olive oil in patients with early atherosclerosis. Eur. J. Nutr. 2013, 52, 1223–1231. [Google Scholar] [CrossRef] [Green Version]
- de Mello, V.D.; Kolehmanien, M.; Schwab, U.; Pulkkinen, L.; Uusitupa, M. Gene expression of peripheral blood mononuclear cells as a tool in dietary intervention studies: What do we know so far? Mol. Nutr. Food Res. 2012, 56, 1160–1172. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Liu, R.; Chang, M.; Zhang, H.; Jin, Q.; Wang, X. Dietary oleic acid supplementation and blood inflammatory markers: A systematic review and meta-analysis of randomized controlled trials. Crit. Rev. Food Sci. Nutr. 2020, 1–18. [Google Scholar] [CrossRef]
- Casas, R.; Sacanella, E.; Urpí-Sardà, M.; Corella, D.; Castañer, O.; Lamuela-Raventos, R.M.; Salas-Salvadó, J.; Martínez-González, M.A.; Ros, E.; Estruch, R. Long-Term Immunomodulatory Effects of a Mediterranean Diet in Adults at High Risk of Cardiovascular Disease in the PREvención con DIeta MEDiterránea (PREDIMED) Randomized Controlled Trial. J. Nutr. 2016, 146, 1684–1693. [Google Scholar] [CrossRef]
- Santos, A.; Rodrigues, A.P.S.; Rosa, L.P.S.; Sarrafzadegan, N.; Silveira, E.A. Cardiometabolic risk factors and Framingham Risk Score in severely obese patients: Baseline data from DieTBra trial. Nutr. Metab. Cardiovasc. Dis. 2020, 30, 474–482. [Google Scholar] [CrossRef] [PubMed]
- Khandouzi, N.; Zahedmehr, A.; Nasrollahzadeh, J. Effect of polyphenol-rich extra-virgin olive oil on lipid profile and inflammatory biomarkers in patients undergoing coronary angiography: A randomised, controlled, clinical trial. Int. J. Food Sci. Nutr. 2021, 72, 548–558. [Google Scholar] [CrossRef] [PubMed]
- Ghavipour, M.; Saedisomeolia, A.; Djalali, M.; Sotoudeh, G.; Eshraghyan, M.R.; Moghadam, A.M.; Wood, L.G. Tomato juice consumption reduces systemic inflammation in overweight and obese females. Br. J. Nutr. 2013, 109, 2031–2035. [Google Scholar] [CrossRef]
- Freitas, P.D.; Ferreira, P.G.; Silva, A.G.; Stelmach, R.; Carvalho-Pinto, R.M.; Fernandes, F.L.; Mancini, M.C.; Sato, M.N.; Martins, M.A.; Carvalho, C.R. The Role of Exercise in a Weight-Loss Program on Clinical Control in Obese Adults with Asthma. A Randomized Controlled Trial. Am. J. Respir. Crit. Care Med. 2017, 195, 32–42. [Google Scholar] [CrossRef] [PubMed]
- Egert, S.; Baxheinrich, A.; Lee-Barkey, Y.H.; Tschoepe, D.; Wahrburg, U.; Stratmann, B. Effects of an energy-restricted diet rich in plant-derived α-linolenic acid on systemic inflammation and endothelial function in overweight-to-obese patients with metabolic syndrome traits. Br. J. Nutr. 2014, 112, 1315–1322. [Google Scholar] [CrossRef] [PubMed] [Green Version]
| Total n = 149 | EVOO n = 50 | DieTBra n = 49 | DieTBra + EVOO n = 50 | |
|---|---|---|---|---|
| Variables | Mean ± SD | Mean ± SD | Mean ± SD | Mean ± SD |
| Age, years | 39.63 ± 8.82 | 38.14 ± 8.14 | 39.14 ± 8.15 | 41.60 ± 9.85 |
| BMI, kg/m2 | 46.03 ± 6.40 | 45.77 ± 6.27 | 46.22 ± 6.26 | 46.13 ± 6.79 |
| NLR | 3.60 ± 2.88 | 3.44 ± 1.35 | 3.46 ± 1.84 | 3.90 ± 4.47 |
| Leucocytes, mm3 | 8199.54 ± 2144.73 | 8027.30 ± 2052.14 | 8299.22 ± 2030.03 | 8274.1 ± 2366.303 |
| LMR | 5.89 ± 4.23 | 6.37 ± 6.64 | 5.65 ± 2.19 | 5.63 ± 2.19 |
| CRP, mg/L | 10.53 ± 7.12 | 10.80 ± 4.90 | 10.40 ± 8.62 | 10.45 ± 7.26 |
| Body weight, kg | 118.81 ± 19.47 | 117.38 ± 18.69 | 120.43 ± 20.85 | 118.64 ± 19.10 |
| Females, n (%) | 127 (85.23) | 45 (35.43) | 40 (31.50) | 42 (33.07) |
| Males, n (%) | 22 (14.77) | 5 (22.73) | 9 (40.91) | 8 (36.36) |
| NLR | Leucocytes, mm3 | LMR | CRP, mg/L | Body Weight | ||
|---|---|---|---|---|---|---|
| EVOO | Baseline (n = 50) | 3.44 ± 1.35 | 8027.30 ± 2052.14 | 6.37 ± 6.64 | 10.80 ± 4.90 | 117.38 ± 18.69 |
| 12 weeks (n = 43) | 3.50 ± 1.16 | 7521.72 ± 2030.45 | 5.13 ± 1.63 | 13.44 ± 18.19 | 118.46 ± 18.93 | |
| P | 0.923 * | 0.088 * | 0.311 † | 0.303 † | 0.092 * | |
| DieTBra | Baseline (n = 49) | 3.46 ± 1.84 | 8299.22 ± 2030.03 | 5.65 ± 2.19 | 10.40 ± 8.62 | 120.43 ± 20.85 |
| 12 weeks (n = 43) | 3.21 ± 1.12 | 7981.59 ± 1930.52 | 5.29 ± 1.59 | 8.60 ± 4.07 | 118.85 ± 19.28 | |
| P | 0.513 * | 0.208 * | 0.365 † | 0.173 † | <0.001 * | |
| DieTBra + EVOO | Baseline (n = 50) | 3.90 ± 4.47 | 8274.10 ± 2366.30 | 5.63 ± 2.19 | 10.45 ± 7.26 | 118.64 ± 19.10 |
| 12 weeks (n = 47) | 3.31 ± 1.05 | 7582.04 ± 1942.68 | 4.90 ± 1.29 | 8.44 ± 3.99 | 115.42 ± 18.68 | |
| P | 0.702 * | 0.037 * | 0.008 † | 1.000 † | <0.001 * |
| Endpoints at the End of Follow Up | EVOO (n = 43) | DieTBra (n = 43) | DieTBra + EVOO (n = 47) | All Groups | EVOO vs. DieTBra | EVOO vs. DieTBra + EVOO | DieTBra vs. DieTBra + EVOO |
|---|---|---|---|---|---|---|---|
| Mean ± SD | Mean ± SD | Mean ± SD | p | P | p | p | |
| NLR | 3.50 ± 1.16 | 3.21 ± 1.12 | 3.31 ± 1.05 | 0.585 * | 0.322 ‡ | 0.502 ‡ | 0.694 ‡ |
| Leucocytes, mm3 | 7521.72 ± 2030.45 | 7981.59 ± 1930.52 | 7582.04 ± 1942.68 | 0.498 * | 0.285 ‡ | 0.886 ‡ | 0.331 ‡ |
| LMR | 5.13 ± 1.63 | 5.29 ± 1.59 | 4.90 ± 1.29 | 0.471 * | 0.651 ‡ | 0.463 ‡ | 0.207 ‡ |
| CRP, mg/L | 13.44 ± 18.19 | 8.60 ± 4.07 | 8.44 ± 3.99 | 0.539 † | 0.387 § | 0.300 § | 0.853 § |
| Endpoints (12 Weeks–Baseline) | EVOO | DieTBra | DieTBra + EVOO | p-Value |
|---|---|---|---|---|
| Mean ± SD | Mean ± SD | Mean ± SD | ||
| ∆ NLR | 0.02 ± 1.08 | −0.22 ± 1.87 | 0.05 ± 0.78 | 0.902 † |
| ∆ Leucocytes, mm3 | −468.84 ± 1757.39 | −297.16 ± 1523.73 | −469.98 ± 1501.62 | 0.844 * |
| ∆ LMR | −1.57 ± 7.29 | −0.35 ± 2.44 | −0.84 ± 2.26 | 0.460 † |
| ∆ CRP, mg/dL | −0.90 ± 18.27 | −1.04 ± 3.46 | 0.00 ± 0.313 | 0.474 † |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Longhi, R.; Santos, A.S.e.A.d.C.; López-Yerena, A.; Rodrigues, A.P.S.; Oliveira, C.d.; Silveira, E.A. The Effectiveness of Extra Virgin Olive Oil and the Traditional Brazilian Diet in Reducing the Inflammatory Profile of Individuals with Severe Obesity: A Randomized Clinical Trial. Nutrients 2021, 13, 4139. https://doi.org/10.3390/nu13114139
Longhi R, Santos ASeAdC, López-Yerena A, Rodrigues APS, Oliveira Cd, Silveira EA. The Effectiveness of Extra Virgin Olive Oil and the Traditional Brazilian Diet in Reducing the Inflammatory Profile of Individuals with Severe Obesity: A Randomized Clinical Trial. Nutrients. 2021; 13(11):4139. https://doi.org/10.3390/nu13114139
Chicago/Turabian StyleLonghi, Rafael, Annelisa Silva e Alves de Carvalho Santos, Anallely López-Yerena, Ana Paula Santos Rodrigues, Cesar de Oliveira, and Erika Aparecida Silveira. 2021. "The Effectiveness of Extra Virgin Olive Oil and the Traditional Brazilian Diet in Reducing the Inflammatory Profile of Individuals with Severe Obesity: A Randomized Clinical Trial" Nutrients 13, no. 11: 4139. https://doi.org/10.3390/nu13114139
APA StyleLonghi, R., Santos, A. S. e. A. d. C., López-Yerena, A., Rodrigues, A. P. S., Oliveira, C. d., & Silveira, E. A. (2021). The Effectiveness of Extra Virgin Olive Oil and the Traditional Brazilian Diet in Reducing the Inflammatory Profile of Individuals with Severe Obesity: A Randomized Clinical Trial. Nutrients, 13(11), 4139. https://doi.org/10.3390/nu13114139








