Animal, Plant, Collagen and Blended Dietary Proteins: Effects on Musculoskeletal Outcomes
Abstract
1. Skeletal Muscle, Bone, Protein Sources and the Notion of Protein “Quality”
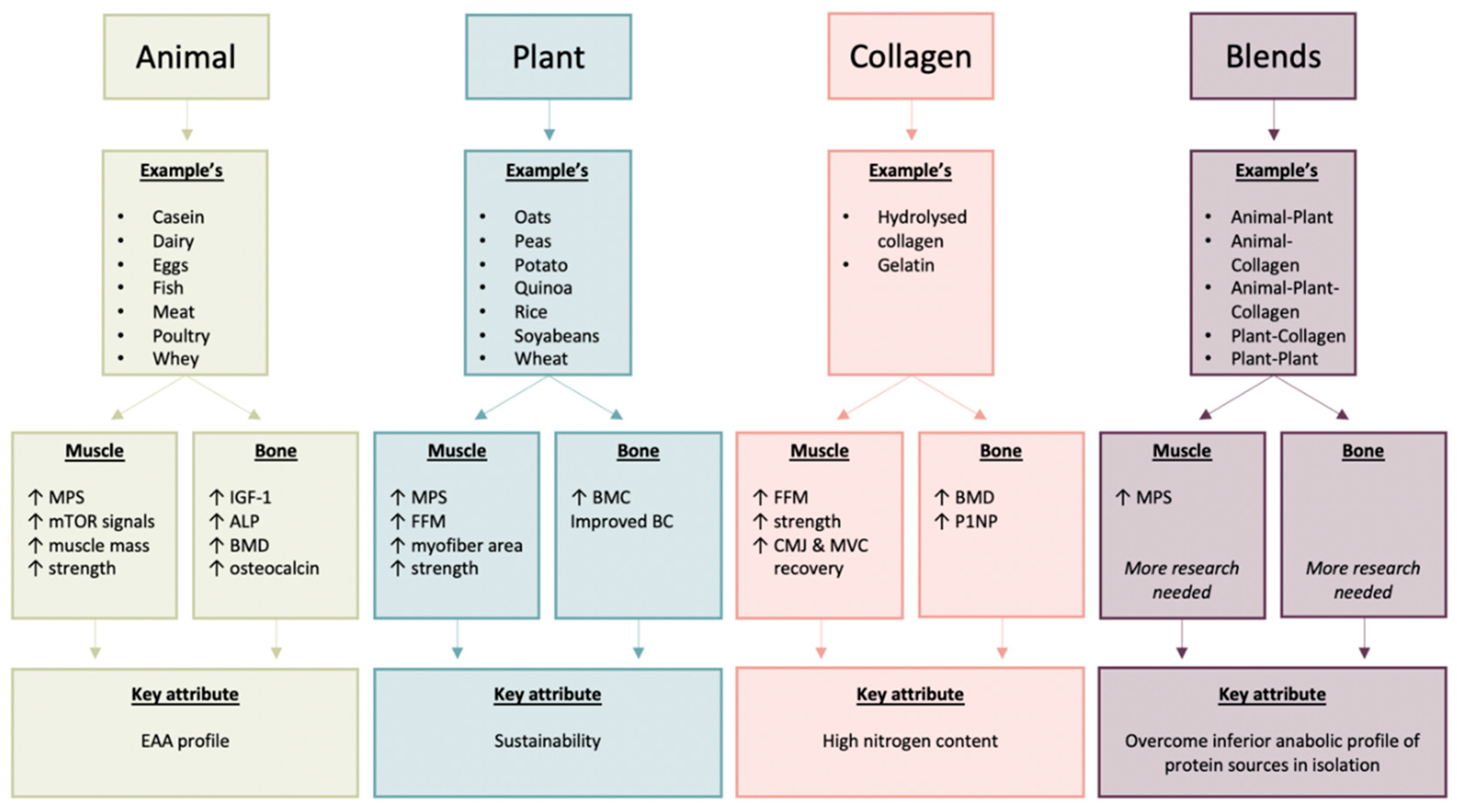
1.1. Definition of Animal, Plant, Collagen and Blended Dietary Protein Sources
1.2. Muscle and Bone Protein Turnover
1.3. Dietary Protein Requirements
1.4. Protein Quality
2. Animal-Derived Proteins: Effects in Relation to Age, Exercise, Energy Restriction and Source
2.1. Skeletal Muscle
2.2. Bone
3. Plant-Derived Proteins: Effects in Relation to Age, Exercise, Energy Restriction and Source
3.1. Skeletal Muscle
3.2. Bone
4. Collagen-Derived Proteins: Effects in Relation to Age, Exercise, Energy Restriction and Source
4.1. Skeletal Muscle
4.2. Bone
5. Animal-, Plant- and/or Collagen-Derived Protein Blends
6. Future Directions
7. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Bradlee, M.L.; Mustafa, J.; Singer, M.R.; Moore, L.L. High-protein foods and physical activity protect against age-related muscle loss and functional decline. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2018, 73, 88–94. [Google Scholar] [CrossRef] [PubMed]
- Dawson-Hughes, B.; Harris, S.S. Calcium intake influences the association of protein intake with rates of bone loss in elderly men and women. Am. J. Clin. Nutr. 2002, 75, 773–779. [Google Scholar] [CrossRef] [PubMed]
- Bowen, J.; Noakes, M.; Clifton, P.M. A high dairy protein, high-calcium diet minimizes bone turnover in overweight adults during weight loss. J. Nutr. 2004, 134, 568–573. [Google Scholar] [CrossRef] [PubMed]
- Hector, A.J.; Marcotte, G.R.; Churchward-Venne, T.A.; Murphy, C.H.; Breen, L.; von Allmen, M.; Baker, S.K.; Phillips, S.M. Whey protein supplementation preserves postprandial myofibrillar protein synthesis during short-term energy restriction in overweight and obese adults. J. Nutr. 2015, 145, 246–252. [Google Scholar] [CrossRef] [PubMed]
- Cermak, N.M.; Res, P.T.; de Groot, L.C.P.G.M.; Saris, W.H.M.; van Loon, L.J.C. Protein supplementation augments the adaptive response of skeletal muscle to resistance-type exercise training: A meta-analysis. Am. J. Clin. Nutr. 2012, 96, 1454–1464. [Google Scholar] [CrossRef]
- Pimentel, D.; Pimentel, M. Sustainability of meat-based and plant-based diets and the environment. Am. J. Clin. Nutr. 2003, 78, 660–663. [Google Scholar] [CrossRef]
- van Vliet, S.; Burd, N.A.; van Loon, L.J. The skeletal muscle anabolic response to plant- versus animal-based protein consumption. J. Nutr. 2015, 145, 1981–1991. [Google Scholar] [CrossRef]
- Castellanos, V.H.; Litchford, M.D.; Campbell, W.W. Modular protein supplements and their application to long-term care. Nutr. Clin. Pract. 2006, 21, 485–504. [Google Scholar] [CrossRef]
- Gorissen, S.H.M.; Witard, O.C. Characterising the muscle anabolic potential of dairy, meat and plant-based protein sources in older adults. Proc. Nutr. Soc. 2018, 77, 20–31. [Google Scholar] [CrossRef]
- Turkiewicz, M. Collagen hydrolysates as a new diet supplement. Sci. Bull. Tech. Univ. Lodz 2009, 73, 83–92. [Google Scholar]
- Etheridge, T.; Oczypok, E.A.; Lehmann, S.; Fields, B.D.; Shephard, F.; Jacobson, L.A.; Szewczyk, N.J. Calpains mediate integrin attachment complex maintenance of adult muscle in Caenorhabditis elegans. PLoS Genet. 2012, 8. [Google Scholar] [CrossRef] [PubMed]
- Demontiero, O.; Vidal, C.; Duque, G. Aging and bone loss: New insights for the clinician. Ther. Adv. Musculoskelet. Dis. 2012, 4, 61–76. [Google Scholar] [CrossRef] [PubMed]
- Henry, Y.M.; Fatayerji, D.; Eastell, R. Attainment of peak bone mass at the lumbar spine, femoral neck and radius in men and women: Relative contributions of bone size and volumetric bone mineral density. Osteoporos. Int. 2004, 15, 263–273. [Google Scholar] [CrossRef] [PubMed]
- Wilkinson, D.J.; Hossain, T.; Hill, D.S.; Phillips, B.E.; Crossland, H.; Williams, J.; Loughna, P.; Churchward-Venne, T.A.; Breen, L.; Phillips, S.M.; et al. Effects of leucine and its metabolite β-hydroxy-β-methylbutyrate on human skeletal muscle protein metabolism. J. Physiol. 2013, 591, 2911–2923. [Google Scholar] [CrossRef] [PubMed]
- Heaney, R.P.; Layman, D.K. Amount and type of protein influences bone health. Am. J. Clin. Nutr. 2008, 87, 1567–1570. [Google Scholar] [CrossRef] [PubMed]
- Layman, D.K.; Anthony, T.G.; Rasmussen, B.B.; Adams, S.H.; Lynch, C.J.; Brinkworth, G.D.; Davis, T.A. Defining meal requirements for protein to optimize metabolic roles of amino acids. Am. J. Clin. Nutr. 2015, 101, 1330S–1338S. [Google Scholar] [CrossRef]
- Traylor, D.A.; Gorissen, S.H.M.; Phillips, S.M. Perspective: Protein requirements and optimal intakes in aging: Arewe ready to recommend more than the recommended daily allowance? Adv. Nutr. 2018, 9, 171–182. [Google Scholar] [CrossRef]
- Deutz, N.E.P.; Bauer, J.M.; Barazzoni, R.; Biolo, G.; Boirie, Y.; Bosy-Westphal, A.; Cederholm, T.; Cruz-Jentoft, A.; Krznariç, Z.; Nair, K.S.; et al. Protein intake and exercise for optimal muscle function with aging: Recommendations from the ESPEN Expert Group. Clin. Nutr. 2014, 33, 929–936. [Google Scholar] [CrossRef]
- Phillips, S.M.; Chevalier, S.; Leidy, H.J. Protein “requirements” beyond the RDA: Implications for optimizing health. Appl. Physiol. Nutr. Metab. 2016, 41, 565–572. [Google Scholar] [CrossRef]
- Lin, P.H.; Miwa, S.; Li, Y.J.; Wang, Y.; Levy, E.; Lastor, K.; Champagne, C. Factors influencing dietary protein sources in the PREMIER trial population. J. Am. Diet. Assoc. 2010, 110, 291–295. [Google Scholar] [CrossRef]
- O’Neil, C.E.; Keast, D.R.; Fulgoni, V.L.; Nicklas, T.A. Food sources of energy and nutrients among adults in the US: NHANES 2003–2006. Nutrients 2012, 4, 2097–2120. [Google Scholar] [CrossRef] [PubMed]
- Song, M.; Fung, T.T.; Hu, F.B.; Willett, W.C.; Longo, V.D.; Chan, A.T.; Giovannucci, E.L. Association of animal and plant protein intake with all-cause and cause-specific mortality. JAMA Intern. Med. 2016, 176, 1453–1463. [Google Scholar] [CrossRef] [PubMed]
- Joy, J.M.; Lowery, R.P.; Wilson, J.M.; Purpura, M.; De Souza, E.O.; Wilson, S.M.; Kalman, D.S.; Dudeck, J.E.; Jäger, R. The effects of 8 weeks of whey or rice protein supplementation on body composition and exercise performance. Nutr. J. 2013, 12. [Google Scholar] [CrossRef] [PubMed]
- Langsetmo, L.; Shikany, J.M.; Cawthon, P.M.; Cauley, J.A.; Taylor, B.C.; Vo, T.N.; Bauer, D.C.; Orwoll, E.S.; Schousboe, J.T.; Ensrud, K.E. The association between protein intake by source and osteoporotic fracture in older men: A prospective cohort study. J. Bone Miner. Res. 2017, 32, 592–600. [Google Scholar] [CrossRef] [PubMed]
- Langsetmo, L.; Shikany, J.M.; Burghardt, A.J.; Cawthon, P.M.; Orwoll, E.S.; Cauley, J.A.; Taylor, B.C.; Schousboe, J.T.; Bauer, D.C.; Vo, T.N.; et al. High dairy protein intake is associated with greater bone strength parameters at the distal radius and tibia in older men: A cross-sectional study. Osteoporos. Int. 2018, 29, 69–77. [Google Scholar] [CrossRef]
- Zdzieblik, D.; Oesser, S.; Baumstark, M.W.; Gollhofer, A.; König, D. Collagen peptide supplementation in combination with resistance training improves body composition and increases muscle strength in elderly sarcopenic men: A randomised controlled trial. Br. J. Nutr. 2015, 114, 1237–1245. [Google Scholar] [CrossRef]
- Oikawa, S.Y.; Kamal, M.J.; Webb, E.K.; McGlory, C.; Baker, S.K.; Phillips, S.M. Whey protein but not collagen peptides stimulate acute and longer-term muscle protein synthesis with and without resistance exercise in healthy older women: A randomized controlled trial. Am. J. Clin. Nutr. 2020, 111, 708–718. [Google Scholar] [CrossRef]
- Oertzen-Hagemann, V.; Kirmse, M.; Eggers, B.; Pfeiffer, K.; Marcus, K.; de Marées, M.; Platen, P. Effects of 12 weeks of hypertrophy resistance exercise training combined with collagen peptide supplementation on the skeletal muscle proteome in recreationally active men. Nutrients 2019, 11, 1072. [Google Scholar] [CrossRef]
- Clifford, T.; Ventress, M.; Allerton, D.M.; Stansfield, S.; Tang, J.C.Y.; Fraser, W.D.; Vanhoecke, B.; Prawitt, J.; Stevenson, E. The effects of collagen peptides on muscle damage, inflammation and bone turnover following exercise: A randomized, controlled trial. Amino Acids 2019, 51, 691–704. [Google Scholar] [CrossRef]
- Reidy, P.T.; Walker, D.K.; Dickinson, J.M.; Gundermann, D.M.; Drummond, M.J.; Timmerman, K.L.; Fry, C.S.; Borack, M.S.; Cope, M.B.; Mukherjea, R.; et al. Protein blend ingestion following resistance exercise promotes human muscle protein synthesis. J. Nutr. 2013, 143, 410–416. [Google Scholar] [CrossRef]
- Reidy, P.T.; Borack, M.S.; Markofski, M.M.; Dickinson, J.M.; Deer, R.R.; Husaini, S.H.; Walker, D.K.; Igbinigie, S.; Robertson, S.M.; Cope, M.B.; et al. Protein supplementation has minimal effects on muscle adaptations during resistance exercise training in young men: A double-blind randomized clinical trial. J. Nutr. 2016, 146, 1660–1669. [Google Scholar] [CrossRef] [PubMed]
- Borack, M.S.; Reidy, P.T.; Husaini, S.H.; Markofski, M.M.; Deer, R.R.; Richison, A.B.; Lambert, B.S.; Cope, M.B.; Mukherjea, R.; Jennings, K.; et al. Soy-dairy protein blend or whey protein isolate ingestion induces similar postexercise muscle mechanistic target of rapamycin complex 1 signaling and protein synthesis responses in older men. J. Nutr. 2016, 146, 2468–2475. [Google Scholar] [CrossRef]
- Mathai, J.K.; Liu, Y.; Stein, H.H. Values for digestible indispensable amino acid scores (DIAAS) for some dairy and plant proteins may better describe protein quality than values calculated using the concept for protein digestibility-corrected amino acid scores (PDCAAS). Br. J. Nutr. 2017, 117, 490–499. [Google Scholar] [CrossRef] [PubMed]
- Schaafsma, G. The protein digestibility–corrected amino acid score. J. Nutr. 2000, 130, 1865S–1867S. [Google Scholar] [CrossRef] [PubMed]
- Phillips, S.M. Nutrient-rich meat proteins in offsetting age-related muscle loss. Meat Sci. 2012, 92, 174–178. [Google Scholar] [CrossRef]
- Moughan, P.J. Holistic properties of foods: A changing paradigm in human nutrition. J. Sci. Food Agric. 2018. [Google Scholar] [CrossRef]
- Burd, N.A.; Beals, J.W.; Martinez, I.G.; Salvador, A.F.; Skinner, S.K. Food-first approach to enhance the regulation of post-exercise skeletal muscle protein synthesis and remodeling. Sport Med. 2019, 49, 59–68. [Google Scholar] [CrossRef]
- Witard, O.C.; Wardle, S.L.; Macnaughton, L.S.; Hodgson, A.B.; Tipton, K.D. Protein considerations for optimising skeletal muscle mass in healthy young and older adults. Nutrients 2016, 8, 181. [Google Scholar] [CrossRef]
- Deane, C.S.; Wilkinson, D.J.; Phillips, B.E.; Smith, K.; Etheridge, T.; Atherton, P.J. “Nutraceuticals” in relation to human skeletal muscle and exercise. Am. J. Physiol. Endocrinol. Metab. 2017, 312, E282–E299. [Google Scholar] [CrossRef]
- Sharples, A.P.; Hughes, D.C.; Deane, C.S.; Saini, A.; Selman, C.; Stewart, C.E. Longevity and skeletal muscle mass: The role of IGF signalling, the sirtuins, dietary restriction and protein intake. Aging Cell 2015, 14, 511–523. [Google Scholar] [CrossRef]
- Symons, T.B.; Sheffield-Moore, M.; Wolfe, R.R.; Paddon-Jones, D. A moderate serving of high-quality protein maximally stimulates skeletal muscle protein synthesis in young and elderly subjects. J. Am. Diet. Assoc. 2009, 109, 1582–1586. [Google Scholar] [CrossRef]
- Smith, K.; Reynolds, N.; Downie, S.; Patel, A.; Rennie, M.J. Effects of flooding amino acids on incorporation of labeled amino acids into human muscle protein. Am. J. Physiol. Metab. 1998, 275, E73–E78. [Google Scholar] [CrossRef] [PubMed]
- Atherton, P.J.; Smith, K.; Etheridge, T.; Rankin, D.; Rennie, M.J. Distinct anabolic signalling responses to amino acids in C2C12 skeletal muscle cells. Amino Acids 2010, 38, 1533–1539. [Google Scholar] [CrossRef]
- Witard, O.C.; Jackman, S.R.; Breen, L.; Smith, K.; Selby, A.; Tipton, K.D. Myofibrillar muscle protein synthesis rates subsequent to a meal in response to increasing doses of whey protein at rest and after resistance exercise. Am. J. Clin. Nutr. 2014, 99, 86–95. [Google Scholar] [CrossRef] [PubMed]
- Moore, D.R.; Robinson, M.J.; Fry, J.L.; Tang, J.E.; Glover, E.I.; Wilkinson, S.B.; Prior, T.; Tarnopolsky, M.A.; Phillips, S.M. Ingested protein dose response of muscle and albumin protein synthesis after resistance exercise in young men. Am. J. Clin. Nutr. 2009, 89, 161–168. [Google Scholar] [CrossRef] [PubMed]
- Cuthbertson, D.; Smith, K.; Babraj, J.; Leese, G.; Waddell, T.; Atherton, P.; Wackerhage, H.; Taylor, P.M.; Rennie, M.J. Anabolic signaling deficits underlie amino acid resistance of wasting, aging muscle. FASEB J. 2005, 19, 422–424. [Google Scholar] [CrossRef] [PubMed]
- Atherton, P.J.; Smith, K. Muscle protein synthesis in response to nutrition and exercise. J. Physiol. 2012, 590, 1049–1057. [Google Scholar] [CrossRef]
- Atherton, P.J.; Etheridge, T.; Watt, P.W.; Wilkinson, D.; Selby, A.; Rankin, D.; Smith, K.; Rennie, M.J. Muscle full effect after oral protein: Time-dependent concordance and discordance between human muscle protein synthesis and mTORC1 signaling. Am. J. Clin. Nutr. 2010, 92, 1080–1088. [Google Scholar] [CrossRef]
- Alexandrov, N.V.; Eelderink, C.; Singh-Povel, C.M.; Navis, G.J.; Bakker, S.J.L.; Corpeleijn, E. Dietary protein sources and muscle mass over the life course: The lifelines cohort study. Nutrients 2018, 10, 1471. [Google Scholar] [CrossRef]
- Koopman, R.; Walrand, S.; Beelen, M.; Gijsen, A.P.; Kies, A.K.; Boirie, Y.; Saris, W.H.M.; van Loon, L.J.C. Dietary protein digestion and absorption rates and the subsequent postprandial muscle protein synthetic response do not differ between young and elderly men. J. Nutr. 2009, 139, 1707–1713. [Google Scholar] [CrossRef]
- Symons, T.B.; Schutzler, S.E.; Cocke, T.L.; Chinkes, D.L.; Wolfe, R.R.; Paddon-Jones, D. Aging does not impair the anabolic response to a protein-rich meal. Am. J. Clin. Nutr. 2007, 86, 451–456. [Google Scholar] [CrossRef] [PubMed]
- Moro, T.; Brightwell, C.R.; Deer, R.R.; Graber, T.G.; Galvan, E.; Fry, C.S.; Volpi, E.; Rasmussen, B.B. Muscle protein anabolic resistance to essential amino acids does not occur in healthy older adults before or after resistance exercise training. J. Nutr. 2018, 148, 900–909. [Google Scholar] [CrossRef]
- Burd, N.A.; Gorissen, S.H.; Van Loon, L.J.C. Anabolic resistance of muscle protein synthesis with aging. Exerc. Sport Sci. Rev. 2013, 41, 169–173. [Google Scholar] [CrossRef]
- Katsanos, C.S.; Kobayashi, H.; Sheffield-Moore, M.; Aarsland, A.; Wolfe, R.R. Aging is associated with diminished accretion of muscle proteins after the ingestion of a small bolus of essential amino acids. Am. J. Clin. Nutr. 2005, 82, 1065–1073. [Google Scholar] [CrossRef] [PubMed]
- Wall, B.T.; Gorissen, S.H.; Pennings, B.; Koopman, R.; Groen, B.B.L.; Verdijk, L.B.; Van Loon, L.J.C. Aging is accompanied by a blunted muscle protein synthetic response to protein ingestion. PLoS ONE 2015, 10, e0140903. [Google Scholar] [CrossRef] [PubMed]
- Boirie, Y.; Gachon, P.; Beaufrère, B. Splanchnic and whole-body leucine kinetics in young and elderly men. Am. J. Clin. Nutr. 1997, 65, 489–495. [Google Scholar] [CrossRef]
- Gorissen, S.H.M.; Rémond, D.; van Loon, L.J.C. The muscle protein synthetic response to food ingestion. Meat Sci. 2015, 109, 96–100. [Google Scholar] [CrossRef]
- Burd, N.A.; Yang, Y.; Moore, D.R.; Tang, J.E.; Tarnopolsky, M.A.; Phillips, S.M. Greater stimulation of myofibrillar protein synthesis with ingestion of whey protein isolate v. micellar casein at rest and after resistance exercise in elderly men. Br. J. Nutr. 2012, 108, 958–962. [Google Scholar] [CrossRef]
- Pennings, B.; Boirie, Y.; Senden, J.M.G.; Gijsen, A.P.; Kuipers, H.; Van Loon, L.J.C. Whey protein stimulates postprandial muscle protein accretion more effectively than do casein and casein hydrolysate in older men. Am. J. Clin. Nutr. 2011, 93, 997–1005. [Google Scholar] [CrossRef]
- Mitchell, W.K.; Phillips, B.E.; Williams, J.P.; Rankin, D.; Lund, J.N.; Wilkinson, D.J.; Smith, K.; Atherton, P.J. The impact of delivery profile of essential amino acids upon skeletal muscle protein synthesis in older men: Clinical efficacy of pulse vs. bolus supply. Am. J. Physiol. Endocrinol. Metab. 2015, 309, E450–E457. [Google Scholar] [CrossRef]
- Pennings, B.; Groen, B.B.L.; Van Dijk, J.W.; De Lange, A.; Kiskini, A.; Kuklinski, M.; Senden, J.M.G.; Van Loon, L.J.C. Minced beef is more rapidly digested and absorbed than beef steak, resulting in greater postprandial protein retention in older men. Am. J. Clin. Nutr. 2013, 98, 121–128. [Google Scholar] [CrossRef]
- Barbé, F.; Ménard, O.; Gouar, Y.L.; Buffière, C.; Famelart, M.H.; Laroche, B.; Feunteun, S.L.; Rémond, D.; Dupont, D. Acid and rennet gels exhibit strong differences in the kinetics of milk protein digestion and amino acid bioavailability. Food Chem. 2014, 143, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Barbé, F.; Ménard, O.; Le Gouar, Y.; Buffière, C.; Famelart, M.H.; Laroche, B.; Le Feunteun, S.; Dupont, D.; Rémond, D. The heat treatment and the gelation are strong determinants of the kinetics of milk proteins digestion and of the peripheral availability of amino acids. Food Chem. 2013, 136, 1203–1212. [Google Scholar] [CrossRef] [PubMed]
- Rémond, D.; Machebeuf, M.; Yven, C.; Buffière, C.; Mioche, L.; Mosoni, L.; Mirand, P.P. Postprandial whole-body protein metabolism after a meat meal is influenced by chewing efficiency in elderly subjects. Am. J. Clin. Nutr. 2007, 85, 1286–1292. [Google Scholar] [CrossRef] [PubMed]
- Bax, M.L.; Buffière, C.; Hafnaoui, N.; Gaudichon, C.; Savary-Auzeloux, I.; Dardevet, D.; Santé-Lhoutellier, V.; Rémond, D. Effects of meat cooking, and of ingested amount, on protein digestion speed and entry of residual proteins into the colon: A study in minipigs. PLoS ONE 2013, 8. [Google Scholar] [CrossRef]
- Alemán-Mateo, H.; Carreón, V.R.; Macías, L.; Astiazaran-García, H.; Gallegos-Aguilar, A.C.; Enríquez, J.R.R. Nutrient-rich dairy proteins improve appendicular skeletal muscle mass and physical performance, And attenuate the loss of muscle strength in older men and women subjects: A single-blind randomized clinical trial. Clin. Interv. Aging 2014, 9, 1517–1525. [Google Scholar] [CrossRef]
- Zhu, K.; Kerr, D.A.; Meng, X.; Devine, A.; Solah, V.; Binns, C.W.; Prince, R.L. Two-year whey protein supplementation did not enhance muscle mass and physical function in well-nourished healthy older postmenopausal women. J. Nutr. 2015, 145, 2520–2526. [Google Scholar] [CrossRef]
- Pennings, B.; Koopman, R.; Beelen, M.; Senden, J.M.G.; Saris, W.H.M.; Van Loon, L.J.C. Exercising before protein intake allows for greater use of dietary protein-derived amino acids for de novo muscle protein synthesis in both young and elderly men. Am. J. Clin. Nutr. 2011, 93, 322–331. [Google Scholar] [CrossRef]
- Boirie, Y.; Dangin, M.; Gachon, P.; Vasson, M.P.; Maubois, J.L.; Beaufrère, B. Slow and fast dietary proteins differently modulate postprandial protein accretion. Proc. Natl. Acad. Sci. USA 1997, 94, 14930–14935. [Google Scholar] [CrossRef]
- Reitelseder, S.; Agergaard, J.; Doessing, S.; Helmark, I.C.; Lund, P.; Kristensen, N.B.; Frystyk, J.; Flyvbjerg, A.; Schjerling, P.; Van Hall, G.; et al. Whey and casein labeled with L-[1-13C]leucine and muscle protein synthesis: Effect of resistance exercise and protein ingestion. Am. J. Physiol. Endocrinol. Metab. 2011, 300, E231–E242. [Google Scholar] [CrossRef]
- Yang, Y.; Breen, L.; Burd, N.A.; Hector, A.J.; Churchward-Venne, T.A.; Josse, A.R.; Tarnopolsky, M.A.; Phillips, S.M. Resistance exercise enhances myofibrillar protein synthesis with graded intakes of whey protein in older men. Br. J. Nutr. 2012, 108, 1780–1788. [Google Scholar] [CrossRef] [PubMed]
- Robinson, M.J.; Burd, N.A.; Breen, L.; Rerecich, T.; Yang, Y.; Hector, A.J.; Baker, S.K.; Phillips, S.M. Dose-dependent responses of myofibrillar protein synthesis with beef ingestion are enhanced with resistance exercise in middle-aged men. Appl. Physiol. Nutr. Metab. 2013, 38, 120–125. [Google Scholar] [CrossRef] [PubMed]
- Luiking, Y.C.; Deutz, N.E.P.; Memelink, R.G.; Verlaan, S.; Wolfe, R.R. Postprandial muscle protein synthesis is higher after a high whey protein, leucine-enriched supplement than after a dairy-like product in healthy older people: A randomized controlled trial. Nutr. J. 2014, 13. [Google Scholar] [CrossRef] [PubMed]
- Farnfield, M.M.; Breen, L.; Carey, K.A.; Garnham, A.; Cameron-Smith, D. Activation of mTOR signalling in young and old human skeletal muscle in response to combined resistance exercise and whey protein ingestion. Appl. Physiol. Nutr. Metab. 2012, 37, 21–30. [Google Scholar] [CrossRef] [PubMed]
- Chalé, A.; Cloutier, G.J.; Hau, C.; Phillips, E.M.; Dallal, G.E.; Fielding, R.A. Efficacy of whey protein supplementation on resistance exercise-induced changes in lean mass, muscle strength, and physical function in mobility-limited older adults. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2013, 68, 682–690. [Google Scholar] [CrossRef]
- Kang, L.; Gao, Y.; Liu, X.; Liang, Y.Y.; Chen, Y.; Liang, Y.Y.; Zhang, L.; Chen, W.; Pang, H.; Peng, L.N. Effects of whey protein nutritional supplement on muscle function among community-dwelling frail older people: A multicenter study in China. Arch. Gerontol. Geriatr. 2019, 83, 7–12. [Google Scholar] [CrossRef]
- Oikawa, S.Y.; McGlory, C.; D’Souza, L.K.; Morgan, A.K.; Saddler, N.I.; Baker, S.K.; Parise, G.; Phillips, S.M. A randomized controlled trial of the impact of protein supplementation on leg lean mass and integrated muscle protein synthesis during inactivity and energy restriction in older persons. Am. J. Clin. Nutr. 2018, 108, 1060–1068. [Google Scholar] [CrossRef]
- Mojtahedi, M.C.; Thorpe, M.P.; Karampinos, D.C.; Johnson, C.L.; Layman, D.K.; Georgiadis, J.G.; Evans, E.M. The effects of a higher protein intake during energy restriction on changes in body composition and physical function in older women. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2011, 66A, 1218–1225. [Google Scholar] [CrossRef]
- Weigle, D.S.; Breen, P.A.; Matthys, C.C.; Callahan, H.S.; Meeuws, K.E.; Burden, V.R.; Purnell, J.Q. A high-protein diet induces sustained reductions in appetite, ad libitum caloric intake, and body weight despite compensatory changes in diurnal plasma leptin and ghrelin concentrations. Am. J. Clin. Nutr. 2005, 82, 41–48. [Google Scholar] [CrossRef]
- Coker, R.H.; Miller, S.; Schutzler, S.; Deutz, N.; Wolfe, R.R. Whey protein and essential amino acids promote the reduction of adipose tissue and increased muscle protein synthesis during caloric restriction-induced weight loss in elderly, obese individuals. Nutr. J. 2012, 11. [Google Scholar] [CrossRef]
- Haub, M.D.; Wells, A.M.; Tarnopolsky, M.A.; Campbell, W.W. Effect of protein source on resistive-training-induced changes in body composition and muscle size in older men. Am. J. Clin. Nutr. 2002, 76, 511–517. [Google Scholar] [CrossRef]
- Durosier-Izart, C.; Biver, E.; Merminod, F.; Van Rietbergen, B.; Chevalley, T.; Herrmann, F.R.; Ferrari, S.L.; Rizzoli, R. Peripheral skeleton bone strength is positively correlated with total and dairy protein intakes in healthy postmenopausal women. Am. J. Clin. Nutr. 2017, 105, 513–525. [Google Scholar] [CrossRef] [PubMed]
- Pistoia, W.; Van Rietbergen, B.; Lochmüller, E.M.; Lill, C.A.; Eckstein, F.; Rüegsegger, P. Estimation of distal radius failure load with micro-finite element analysis models based on three-dimensional peripheral quantitative computed tomography images. Bone 2002, 30, 842–848. [Google Scholar] [CrossRef]
- Frassetto, L.; Banerjee, T.; Powe, N.; Sebastian, A. Acid balance, dietary acid load, and bone effects—A controversial subject. Nutrients 2018, 10, 517. [Google Scholar] [CrossRef] [PubMed]
- Kerstetter, J.E.; Kenny, A.M.; Insogna, K.L. Dietary protein and skeletal health: A review of recent human research. Curr. Opin. Lipidol. 2011, 22, 16–20. [Google Scholar] [CrossRef]
- Hannan, M.T.; Tucker, K.L.; Dawson-Hughes, B.; Cupples, L.A.; Felson, D.T.; Kiel, D.P. Effect of dietary protein on bone loss in elderly men and women: The framingham osteoporosis study. J. Bone Miner. Res. 2000, 15, 2504–2512. [Google Scholar] [CrossRef] [PubMed]
- Roughead, Z.K. (Fariba); Johnson, L.K.; Lykken, G.I.; Hunt, J.R. Controlled High Meat Diets Do Not Affect Calcium Retention or Indices of Bone Status in Healthy Postmenopausal Women. J. Nutr. 2003, 133, 1020–1026. [Google Scholar] [CrossRef]
- Cao, J.J.; Johnson, L.K.; Hunt, J.R. A diet high in meat protein and potential renal acid load increases fractional calcium absorption and urinary calcium excretion without affecting markers of bone resorption or formation in postmenopausal women. J. Nutr. 2011, 141, 391–397. [Google Scholar] [CrossRef]
- Ballard, T.L.P.; Clapper, J.A.; Specker, B.L.; Binkley, T.L.; Vukovich, M.D. Effect of protein supplementation during a 6-mo strength and conditioning program on insulin-like growth factor I and markers of bone turnover in young adults. Am. J. Clin. Nutr. 2005, 81, 1442–1448. [Google Scholar] [CrossRef]
- Mullins, N.M.; Sinning, W.E. Effects of resistance training and protein supplementation on bone turnover in young adult women. Nutr. Metab. 2005, 2. [Google Scholar] [CrossRef]
- Holm, L.; Olesen, J.L.; Matsumoto, K.; Doi, T.; Mizuno, M.; Alsted, T.J.; Mackey, A.L.; Schwarz, P.; Kjær, M. Protein-containing nutrient supplementation following strength training enhances the effect on muscle mass, strength, and bone formation in postmenopausal women. J. Appl. Physiol. 2008, 105, 274–281. [Google Scholar] [CrossRef] [PubMed]
- Wright, C.S.; McMorrow, A.M.; Weinheimer-Haus, E.M.; Campbell, W.W. Whey protein supplementation and higher total protein intake do not influence bone quantity in overweight and obese adults following a 36-week exercise and diet intervention. J. Nutr. 2017, 147, 179–186. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Farnsworth, E.; Luscombe, N.D.; Noakes, M.; Wittert, G.; Argyiou, E.; Clifton, P.M. Effect of a high-protein, energy-restricted diet on body composition, glycemic control, and lipid concentrations in overweight and obese hyperinsulinemic men and women. Am. J. Clin. Nutr. 2003, 78, 31–39. [Google Scholar] [CrossRef] [PubMed]
- Josse, A.R.; Atkinson, S.A.; Tarnopolsky, M.A.; Phillips, S.M. Diets higher in dairy foods and dietary protein support bone health during diet- and exercise-induced weight loss in overweight and obese premenopausal women. J. Clin. Endocrinol. Metab. 2012, 97, 251–260. [Google Scholar] [CrossRef] [PubMed]
- Houston, D.K.; Nicklas, B.J.; Ding, J.; Harris, T.B.; Tylavsky, F.A.; Newman, A.B.; Jung, S.L.; Sahyoun, N.R.; Visser, M.; Kritchevsky, S.B. Dietary protein intake is associated with lean mass change in older, community-dwelling adults: The health, aging, and body composition (health ABC) study. Am. J. Clin. Nutr. 2008, 87, 150–155. [Google Scholar] [CrossRef] [PubMed]
- Sahni, S.; Mangano, K.M.; Hannan, M.T.; Kiel, D.P.; McLean, R.R. Higher protein intake is associated with higher lean mass and quadriceps muscle strength in adult men and women. J. Nutr. 2015, 145, 1569–1575. [Google Scholar] [CrossRef] [PubMed]
- Tang, J.E.; Moore, D.R.; Kujbida, G.W.; Tarnopolsky, M.A.; Phillips, S.M. Ingestion of whey hydrolysate, casein, or soy protein isolate: Effects on mixed muscle protein synthesis at rest and following resistance exercise in young men. J. Appl. Physiol. 2009, 107, 987–992. [Google Scholar] [CrossRef]
- Phillips, S.M. A brief review of critical processes in exercise-induced muscular hypertrophy. Sport Med. 2014, 44, 71–77. [Google Scholar] [CrossRef]
- Gorissen, S.H.; Horstman, A.M.; Franssen, R.; Crombag, J.J.; Langer, H.; Bierau, J.; Respondek, F.; van Loon, L.J. Ingestion of wheat protein increases in vivo muscle protein synthesis rates in healthy older men in a randomized trial. J. Nutr. 2016, 146, 1651–1659. [Google Scholar] [CrossRef]
- Payette, H.; Gray-Donald, K.; Cyr, R.; Boutier, V. Predictors of dietary intake in a functionally dependent elderly population in the community. Am. J. Public Health 1995, 85, 677–683. [Google Scholar] [CrossRef]
- Benelam, B. Satiety and the anorexia of ageing. Br. J. Community Nurs. 2009, 14, 332–335. [Google Scholar] [CrossRef] [PubMed]
- Oikawa, S.Y.; Bahniwal, R.; Holloway, T.M.; Lim, C.; McLeod, J.C.; McGlory, C.; Baker, S.K.; Phillips, S.M. Potato protein isolate stimulates muscle protein synthesis at rest and with resistance exercise in young women. Nutrients 2020, 12, 1235. [Google Scholar] [CrossRef]
- Wilkinson, S.B.; Tarnopolsky, M.A.; MacDonald, M.J.; MacDonald, J.R.; Armstrong, D.; Phillips, S.M. Consumption of fluid skim milk promotes greater muscle protein accretion after resistance exercise than does consumption of an isonitrogenous and isoenergetic soy-protein beverage. Am. J. Clin. Nutr. 2007, 85, 1031–1040. [Google Scholar] [CrossRef] [PubMed]
- Candow, D.G.; Burke, N.C.; Smith-Palmer, T.; Burke, D.G. Effect of whey and soy protein supplementation combined with resistance training in young adults. Int. J. Sport Nutr. Exerc. Metab. 2006, 16, 233–244. [Google Scholar] [CrossRef] [PubMed]
- DeNysschen, C.A.; Burton, H.W.; Horvath, P.J.; Leddy, J.J.; Browne, R.W. Resistance training with soy vs whey protein supplements in hyperlipidemic males. J. Int. Soc. Sports Nutr. 2009, 6, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Hartman, J.W.; Tang, J.E.; Wilkinson, S.B.; Tarnopolsky, M.A.; Lawrence, R.L.; Fullerton, A.V.; Phillips, S.M. Consumption of fat-free fluid milk after resistance exercise promotes greater lean mass accretion than does consumption of soy or carbohydrate in young, novice, male weightlifters. Am. J. Clin. Nutr. 2007, 86, 373–381. [Google Scholar] [CrossRef]
- Babault, N.; Païzis, C.; Deley, G.; Guérin-Deremaux, L.; Saniez, M.-H.; Lefranc-Millot, C.; Allaert, F.A. Pea proteins oral supplementation promotes muscle thickness gains during resistance training: A double-blind, randomized, Placebo-controlled clinical trial vs. Whey protein. J. Int. Soc. Sports Nutr. 2015, 12, 3. [Google Scholar] [CrossRef]
- Luiking, Y.C.; Engelen, M.P.K.J.; Soeters, P.B.; Boirie, Y.; Deutz, N.E.P. Differential metabolic effects of casein and soy protein meals on skeletal muscle in healthy volunteers. Clin. Nutr. 2011, 30, 65–72. [Google Scholar] [CrossRef]
- Deibert, P.; Solleder, F.; König, D.; Vitolins, M.Z.; Dickhuth, H.H.; Gollhofer, A.; Berg, A. Soy protein based supplementation supports metabolic effects of resistance training in previously untrained middle aged males. Aging Male 2011, 14, 273–279. [Google Scholar] [CrossRef]
- Gilbert, J.A.; Bendsen, N.T.; Tremblay, A.; Astrup, A. Effect of proteins from different sources on body composition. Nutr. Metab. Cardiovasc. Dis. 2011, 21, B16–B31. [Google Scholar] [CrossRef]
- Shams-White, M.M.; Chung, M.; Fu, Z.; Insogna, K.L.; Karlsen, M.C.; LeBoff, M.S.; Shapses, S.A.; Sackey, J.; Shi, J.; Wallace, T.C.; et al. Animal versus plant protein and adult bone health: A systematic review and meta-analysis from the National Osteoporosis Foundation. PLoS ONE 2018, 13, e0192459. [Google Scholar] [CrossRef] [PubMed]
- Langsetmo, L.; Barr, S.I.; Berger, C.; Kreiger, N.; Rahme, E.; Adachi, J.D.; Papaioannou, A.; Kaiser, S.M.; Prior, J.C.; Hanley, D.A.; et al. Associations of protein intake and protein source with bone mineral density and fracture risk: A population-based cohort study. J. Nutr. Heal Aging 2015, 19, 861–868. [Google Scholar] [CrossRef] [PubMed]
- Lauderdale, D.S.; Jacobsen, S.J.; Furner, S.E.; Levy, P.S.; Brody, J.A.; Goldberg, J. Hip fracture incidence among elderly Asian-American populations. Am. J. Epidemiol. 1997, 146, 502–509. [Google Scholar] [CrossRef]
- Ma, D.F.; Qin, L.Q.; Wang, P.Y.; Katoh, R. Soy isoflavone intake inhibits bone resorption and stimulates bone formation in menopausal women: Meta-analysis of randomized controlled trials. Eur. J. Clin. Nutr. 2008, 62, 155–161. [Google Scholar] [CrossRef] [PubMed]
- Pawlowski, J.W.; Martin, B.R.; McCabe, G.P.; McCabe, L.; Jackson, G.S.; Peacock, M.; Barnes, S.; Weaver, C.M. Impact of equol-producing capacity and soy-isoflavone profiles of supplements on bone calcium retention in postmenopausal women: A randomized crossover trial. Am. J. Clin. Nutr. 2015, 102, 695–703. [Google Scholar] [CrossRef] [PubMed]
- Alekel, D.L.; St. Germain, A.; Peterson, C.T.; Hanson, K.B.; Stewart, J.W.; Toda, T. Isoflavone-rich soy protein isolate attenuates bone loss in the lumbar spine of perimenopausal women. Am. J. Clin. Nutr. 2000, 72, 844–852. [Google Scholar] [CrossRef]
- Brink, E.; Coxam, V.; Robins, S.; Wahala, K.; Cassidy, A.; Branca, F. Long-term consumption of isoflavone-enriched foods does not affect bone mineral density, bone metabolism, or hormonal status in early postmenopausal women: A randomized, double-blind, placebo controlled study. Am. J. Clin. Nutr. 2008, 87, 761–770. [Google Scholar] [CrossRef]
- Lee, H.; Choue, R.; Lim, H. Effect of soy isoflavones supplement on climacteric symptoms, bone biomarkers, and quality of life in Korean postmenopausal women: A randomized clinical trial. Nutr. Res. Pract. 2017, 11, 223–231. [Google Scholar] [CrossRef]
- Kreijkamp-Kaspers, S.; Kok, L.; Grobbee, D.E.; de Haan, E.H.F.; Aleman, A.; Lampe, J.W.; van der Schouw, Y.T. Effect of Soy Protein Containing Isoflavones on Cognitive Function, Bone Mineral Density, and Plasma Lipids in Postmenopausal WomenA Randomized Controlled Trial. JAMA 2004, 292, 65–74. [Google Scholar] [CrossRef]
- Liu, Z.M.; Ho, S.C.; Chen, Y.M.; Ho, Y.P. A mild favorable effect of soy protein with isoflavones on body compositiona 6-month double-blind randomized placebo-controlled trial among Chinese postmenopausal women. Int. J. Obes. 2010, 34, 309–318. [Google Scholar] [CrossRef]
- Chiang, T.I.; Chang, I.C.; Lee, H.H.; Hsieh, K.H.; Chiu, Y.W.; Lai, T.J.; Liu, J.Y.; Hsu, L.S.; Kao, S.H. Amelioration of estrogen deficiency-induced obesity by collagen hydrolysate. Int. J. Med. Sci. 2016, 13, 853–857. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Kitakaze, T.; Sakamoto, T.; Kitano, T.; Inoue, N.; Sugihara, F.; Harada, N.; Yamaji, R. The collagen derived dipeptide hydroxyprolyl-glycine promotes C2C12 myoblast differentiation and myotube hypertrophy. Biochem. Biophys. Res. Commun. 2016, 478, 1292–1297. [Google Scholar] [CrossRef]
- Phillips, S.M. The impact of protein quality on the promotion of resistance exercise-induced changes in muscle mass. Nutr. Metab. 2016, 13, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Caldow, M.K.; Ham, D.J.; Trieu, J.; Chung, J.D.; Lynch, G.S.; Koopman, R. Glycine protects muscle cells from wasting in vitro via mTORC1 signaling. Front. Nutr. 2019, 6, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Okiura, T.; Oishi, Y.; Takemura, A.; Ishihara, A. Effects of collagen hydrolysate on the tibialis anterior muscle and femur in senescence-accelerated mouse prone 6. J. Musculoskelet. Neuronal Interact. 2016, 16, 161–167. [Google Scholar] [PubMed]
- Shaw, G.; Lee-Barthel, A.; Ross, M.L.; Wang, B.; Baar, K. Vitamin C-enriched gelatin supplementation before intermittent activity augments collagen synthesis. Am. J. Clin. Nutr. 2017, 105, 136–143. [Google Scholar] [CrossRef] [PubMed]
- Hays, N.P.; Kim, H.; Wells, A.M.; Kajkenova, O.; Evans, W.J. Effects of whey and fortified collagen hydrolysate protein supplements on nitrogen balance and body composition in older women. J. Am. Diet. Assoc. 2009, 109, 1082–1087. [Google Scholar] [CrossRef] [PubMed]
- Kirmse, M.; Oertzen-Hagemann, V.; de Marées, M.; Bloch, W.; Platen, P. Prolonged collagen peptide supplementation and resistance exercise training affects body composition in recreationally active men. Nutrients 2019, 11, 1154. [Google Scholar] [CrossRef]
- Impey, S.G.; Hammond, K.M.; Naughton, R.; Langan-Evans, C.; Shepherd, S.O.; Sharples, A.P.; Cegielski, J.; Smith, K.; Jeromson, S.; Hamilton, D.L.; et al. Whey protein augments leucinemia and postexercise p70s6k1 activity compared with a hydrolyzed collagen blend when in recovery from training with low carbohydrate availability. Int. J. Sport Nutr. Exerc. Metab. 2018, 28, 651–659. [Google Scholar] [CrossRef]
- Oikawa, S.Y.; Macinnis, M.J.; Tripp, T.R.; Mcglory, C.; Baker, S.K.; Phillips, S.M. Lactalbumin, not collagen, augments muscle protein synthesis with aerobic exercise. Med. Sci. Sport Exerc. 2020, 52. [Google Scholar] [CrossRef]
- Kjær, M.; Magnusson, P.; Krogsgaard, M.; Møller, J.B.; Olesen, J.; Heinemeier, K.; Hansen, M.; Haraldsson, B.; Koskinen, S.; Esmarck, B.; et al. Extracellular matrix adaptation of tendon and skeletal muscle to exercise. J. Anat. 2006, 208, 445–450. [Google Scholar] [CrossRef]
- Rindom, E.; Nielsen, M.H.; Kececi, K.; Jensen, M.E.; Vissing, K.; Farup, J. Effect of protein quality on recovery after intense resistance training. Eur. J. Appl. Physiol. 2016, 116, 2225–2236. [Google Scholar] [CrossRef] [PubMed]
- Lewis, M.I.; Fournier, M.; Storer, T.W.; Bhasin, S.; Porszasz, J.; Ren, S.G.; Da, X.; Casaburi, R. Skeletal muscle adaptations to testosterone and resistance training in men with COPD. J. Appl. Physiol. 2007, 103, 1299–1310. [Google Scholar] [CrossRef]
- Phillips, S.M.; Tipton, K.D.; Van Loon, L.J.C.; Verdijk, L.B.; Paddon-Jones, D.; Close, G.L. Exceptional body composition changes attributed to collagen peptide supplementation and resistance training in older sarcopenic men. Br. J. Nutr. 2016, 116, 569–570. [Google Scholar] [CrossRef] [PubMed]
- Jendricke, P.; Centner, C.; Zdzieblik, D.; Gollhofer, A.; König, D. Specific collagen peptides in combination with resistance training improve body composition and regional muscle strength in premenopausal women: A randomized controlled trial. Nutrients 2019, 11, 892. [Google Scholar] [CrossRef] [PubMed]
- Vechin, F.C.; Libardi, C.A.; Conceição, M.S.; Damas, F.; Cavaglieri, C.R.; Chacon-Mikahil, M.P.T.; Coutinho, L.L.; Andrade, S.C.S.; Neves, M.T.; Roschel, H.; et al. Low-intensity resistance training with partial blood flow restriction and high-intensity resistance training induce similar changes in skeletal muscle transcriptome in elderly humans. Appl. Physiol. Nutr. Metab. 2019, 44, 216–220. [Google Scholar] [CrossRef] [PubMed]
- Centner, C.; Zdzieblik, D.; Roberts, L.; Gollhofer, A.; König, D. Effects of blood flow restriction training with protein supplementation on muscle mass and strength in older men. J. Sport Sci. Med. 2019, 18, 471–478. [Google Scholar]
- Guillerminet, F.; Beaupied, H.; Fabien-Soulé, V.; Tomé, D.; Benhamou, C.L.; Roux, C.; Blais, A. Hydrolyzed collagen improves bone metabolism and biomechanical parameters in ovariectomized mice: An in vitro and in vivo study. Bone 2010, 46, 827–834. [Google Scholar] [CrossRef]
- Han, X.L.; Xu, Y.J.; Wang, J.B.; Pei, X.R.; Yang, R.Y.; Li, N.; Li, Y. Effects of cod bone gelatin on bone metabolism and bone microarchitecture in ovariectomized rats. Bone 2009, 44, 942–947. [Google Scholar] [CrossRef]
- Elam, M.L.; Johnson, S.A.; Hooshmand, S.; Feresin, R.G.; Payton, M.E.; Gu, J.; Arjmandi, B.H. A calcium-collagen chelate dietary supplement attenuates bone loss in postmenopausal women with osteopenia: A randomized controlled trial. J. Med. Food 2015, 18, 324–331. [Google Scholar] [CrossRef]
- König, D.; Oesser, S.; Scharla, S.; Zdzieblik, D.; Gollhofer, A. Specific collagen peptides improve bone mineral density and bone markers in postmenopausal women—A randomized controlled study. Nutrients 2018, 10, 97. [Google Scholar] [CrossRef] [PubMed]
- Pinnell, S.R. Regulation of collagen biosynthesis by ascorbic acid: A review. Yale J. Biol. Med. 1985, 58, 553–559. [Google Scholar] [PubMed]
| Reference | Study Design | Protein Composition | Measurements | Key Outcomes |
|---|---|---|---|---|
| Alexandrov et al., 2018 [49] | Data analysis of the Lifelines Cohort 31,278 males (M) and 45,355 females (F) (n = 76,633, 44.9 ± 12.8 years, 18–91 years) (mean ± standard deviation (SD)) | Protein type/intake determined through food frequency questionnaire (mean protein intake per day 1 ± 0.3 g/kg) | Protein intake, muscle mass (24 h urinary creatinine excretion) | Increased intake of total and animal protein associated with increased creatinine excretion in M and F |
| Bradlee et al., 2018 [1] | Data analysis of the Framingham Offspring study Diet, physical activity and functional performance data collected from M (n = 1016) and F (n = 1333) to evaluate effects on muscle mass | Protein type/intake determined through 3-day food records | Dietary analysis, physical activity, % muscle mass, functional performance | Higher protein intake associated with higher % muscle mass over a 9-year period Higher intake of animal protein had higher % muscle mass In those less active, only animal protein consumption reduced risk of functional decline |
| Symons et al., 2009 [41] | Healthy young adults (M n = 8, F n = 9, 35 ± 3 years) and older (M n = 10, F n = 7, 68 ± 2 years) randomly assigned to moderate or large protein serving (mean ± SD) | Single moderate serving (113 g; 220 kcal; 30 g protein) of 90% lean beef Large (340 g; 660 kcal; 90 g protein) serving of 90% lean beef | Muscle protein synthesis (MPS) | Moderate serving of beef increased MPS ~50% in young and older adults with no further increase seen after ingestion of a large serving |
| Alemán-Mateo et al., 2014 [66] | Single-blind randomised controlled trial (RCT) Older adults randomised to habitual diet (M n = 25, F n = 25, 69.6 ± 6.4 years) or habitual diet with ricotta cheese (M n = 25, F n = 25, 70.8 ± 7.6 years) for 12 weeks (mean ± SD) | 12 weeks of habitual diet or habitual diet with dairy-rich protein (210 g ricotta cheese) | Lean mass (LM), muscle strength | LM increased in supplemented group relative to normal diet group Both groups lost strength but greater loss of muscle strength in controls |
| Zhu et al., 2015 [67] | Randomised, double-blind, placebo-controlled design F were randomly assigned to a high protein drink (n = 101, 74.2 ± 2.8 years) or placebo (n = 95, 74.3 ± 2.6 years) (mean ± SD) | Over a 2-year period, F consumed either daily high protein drink (30 g of whey protein) or placebo (2.1 g protein) | Appendicular lean mass (ALM), muscle cross-sectional area (CSA), handgrip strength, lower limb muscle strength, dietary analysis | Both groups showed decrease in upper arm and calf muscle area over 2 years, but no change in ALM No effect of protein supplementation on muscle mass or function after 1 or 2 years |
| Luiking et al., 2014 [73] | RCT Healthy older adults were randomised to consume either high whey protein (n = 9, 66.9 ± 4.8 years) or milk protein control (n = 10, 71.1 ± 6.3 years) after unilateral resistance exercise (RE) (mean ± SD) | Single bolus of high whey protein, leucine-enriched supplement containing 20 g whey protein, 3 g total leucine Isocaloric milk protein control containing 6 g milk protein | MPS, dietary analysis | Higher MPS with whey protein supplement than milk protein |
| Witard et al., 2014 [44] | Single-blind parallel design Young, resistance-trained M (n = 48) were randomised to consume 0 (22 ± 3 years), 10 (20 ± 1 years), 20 (22 ± 3 years) or 40 (20 ± 1 years) g protein after a single bout of unilateral RE (mean ± SD) | 0, 10, 20 or 40 g whey protein isolate following RE | MPS, whole-body phenylalanine oxidation, dietary analysis | Ingestion of 20 and 40 g whey protein increased myofibrillar MPS above 0 g 40 g whey protein increased rates of phenylalanine oxidation |
| Farnfield et al., 2012 [74] | Randomised, placebo-controlled design Healthy young and older M completed a 12-week resistance exercise training (RET) and were randomly assigned to consume whey protein (young n = 8, 20.5 ± 0.7 years, older n = 9, 68.1 ± 1.6 years) or placebo (young n = 8, 20.4 ± 0.8 years, older n = 9, 67.4 ± 1.3 years) after each exercise session (mean ± standard error of the mean (SEM)) | Whey protein containing 26.6 g amino acids (AA) per serving Placebo containing same amount of artificial flavour and aspartame sweetener | Strength, protein signalling, dietary analysis | Strength increased in all volunteers Whey protein caused greater increases in mechanistic target of rapamycin phosphorylation than placebo in both age groups |
| Robinson et al., 2013 [72] | RCT 35 M (59 ± 2 years) were randomly assigned to 1 of 4 protein groups with and without RE (n = 7 per group) (mean ± SEM) | Consumed 0, 57 g (12 g protein), 113 g (24 g protein) or 170 g (36 g protein) of ground beef | MPS, leucine oxidation | Ingestion of 170 g beef increased myofibrillar MPS at rest and after RE more than other amounts Higher leucine oxidation with increasing amounts of beef |
| Yang et al., 2012 [71] | RCT Older M (n = 37, 71 ± 4 years) completed a bout of unilateral leg RE prior to ingesting 1 of 4 protein doses (mean ± SD) | 0, 10, 20 or 40 g whey protein isolate | MPS, leucine oxidation | Whole-body leucine oxidation increased in a dose-dependent manner MPS increased with 20 and 40 g whey protein but not lower doses 20 and 40 g whey protein ingestion post-exercise increased MPS above 0 and 10 g exercise rates |
| Haub et al., 2002 [81] | RCT M randomly assigned to beef-containing (n = 10, 63 ± 3 years) or lacto-ovo-vegetarian (n = 11, 67 ± 6 years) diet throughout 12-week RET (mean ± SD) | Beef-containing diet: 0.6 g protein/kg/d from beef Lacto-ovo-vegetarian diet: 0.6 g protein/kg/d from soy | Strength, muscle CSA, dietary analysis | No difference between dietary groups in terms of strength improvements CSA of m. vastus lateralis increased with training similarly in both groups |
| Chalé et al., 2013 [75] | Randomised, double-blind controlled design Older mobility-limited adults were randomised to protein (n = 42, 78 ± 4 years) or isocaloric control (n = 38, 77.3 ± 3.9 years) and high intensity RET for 6 months (mean ± SD) | Whey protein: 40 g/d Isocaloric control | Strength, muscle CSA, LM, dietary analysis | LM, muscle CSA and muscle strength increased in both groups but there was no difference between groups |
| Kang et al., 2019 [76] | Multicentre, interventional, two parallel-group case-control design Frail older adults received daily protein supplementation (n = 49, 78 ± 7 years) or no supplementation (n = 66, 76.8 ± 7 years) combined with RET for 12 weeks (mean ± SD) | Protein containing 32.4 g of whey protein | Handgrip strength, gait speed, chair rise test | Handgrip strength, chair-stand time and gait speed improved to a greater extent in the group that received whey protein |
| Hector et al., 2015 [4] | Randomised, double-blind design Adults were randomised to receive whey protein (n = 14, 52 ± 2 years), soy protein (n = 14, 52 ± 2 years) or carbohydrate (CHO) (n = 12, 48 ± 3 years) during a 14-day hypoenergetic diet (mean ± SEM) | Twice daily supplements of: Whey protein: 27 g/supplement or Soy protein: 26 g/supplement or Isoenergetic CHO Hypoenergetic diet: −750 kcal/d | MPS, dietary analysis | Whey protein stimulated MPS to greater extent than soy protein or CHO pre-intervention Postprandial MPS was reduced by whey protein less than soy protein and CHO post-intervention |
| Mojtahedi et al., 2011 [78] | Randomised, double-blind parallel design Overweight/obese, postmenopausal F prescribed reduced calorie diet and randomised to protein (n = 13, 64.7 ± 4.4 years) or CHO (n = 13, 64.6 ± 5.2 years) for 6 months (mean ± SD) | Reduced calorie diet: 1400 kcal/d, 15%, 65% and 30% energy from protein, CHO and fat, respectively. Protein: 2 × 25 g/d whey protein CHO: 2 × 25 g/d maltodextrin | LM, strength | More weight lost in protein group No differences changes to LM or strength Relative to thigh volume changes, protein group gained more muscle than CHO group |
| Coker et al., 2012 [80] | Older adults (n = 12) randomised to 8-week calorie restriction diet using (i) 7% weight loss with meal replacement (70 ± 2 years) or (ii) competitive meal replacement (68 ± 2 years) (mean ± SEM) | Whey protein (7 g) plus essential amino acids (EAA) formulation (6 g) in form of meal replacement (5 ×/d) or competitive meal replacement | LM, MPS, dietary analysis | Whey protein/EAA did not preserve LM but there was an increase in acute FSR |
| Reference | Study Design | Protein Composition | Measurements | Key Outcomes |
|---|---|---|---|---|
| Hannan et al., 2000 [86] | 615 older adults (75 ± 4.4 years, 391 females (F), 224 males (M) (mean ± standard deviation (SD)) Relationship between dietary protein and subsequent 4-year change in bone health | Protein type/intake determined through food frequency questionnaire | Protein intake, bone mineral density (BMD) | Lower protein intake associated with increased bone loss Higher intake of animal protein not associated with decrease in BMD |
| Roughead et al., 2003 [87] | Randomised crossover design Healthy postmenopausal F (n = 15, 60.5 ± 7.8 years) randomised to 8-week high-meat and 8-week low-meat diet (mean ± SD) | High-meat diet: 20% of energy as protein Low-meat diet: 12% of energy as protein Calcium content similar (~600 mg) in both diets | Calcium excretion, bone markers, dietary analysis | High-meat diet did not adversely affect urinary calcium excretion, calcium retention or markers of bone metabolism |
| Cao et al., 2011 [88] | Randomised crossover design Postmenopausal F (n = 16, 56.9 ± 3.2 years, mean ± SD) randomised to two diets: low protein, low potential renal acid load (PRAL) and high protein, high PRAL diet. | Low protein, low PRAL diet: 10% of energy as protein High protein, high PRAL diet: 20% of energy as protein Each diet was 7 weeks separated by 1 week | Calcium absorption, bone markers, dietary analysis | No effect of high meat/PRAL diet on markers of bone metabolism Increased fractional rate of calcium absorption and urinary calcium excretion |
| Durosier-Izart et al., 2017 [82] | Cross-sectional study design 746 F (65 ± 1.4 years, mean ± SD) Associations between animal (separated into non-dairy and dairy) and vegetable protein sources and bone health | Protein type/intake determined through food frequency questionnaire | Areal BMD, distal radius and tibia bone microstructures, bone strength, protein intake | Predicted failure load and stiffness at distal radius and tibia positively associated with total, animal and dairy protein intake |
| Langsetmo et al., 2018 [25] | Cross-sectional study design Questionnaire data from 1016 M (84.3 ± 4 years, mean ± SD) Association of dairy, non-dairy and plant-derived protein intake on bone health | Protein type/intake determined through food frequency questionnaire | Bone strength, BMD, protein intake | Higher dairy protein associated with higher estimated failure load at the distal radius and distal tibia Higher non-dairy animal protein associated with higher total BMD |
| Ballard et al., 2006 [89] | Randomised controlled trial 51 younger adults (18–25 years, 28 M, 23 F) were randomised to either protein (20.9 ± 2.4 years) or placebo (21.1 ± 2.2 years) supplementation during a 6-month training intervention of alternating resistance exercise training (RET) and aerobic exercise 5 ×/week (mean ± standard error of the mean (SEM)) | Twice daily protein (42 g protein, 24 g carbohydrate (CHO), 2 g fat) Isocaloric CHO supplement (70 g CHO) | Bone markers, protein intake | Increases in plasma insulin-like growth factor-I greater in protein group Serum bone alkaline phosphatase increased over time and tended to be higher in protein group N-terminal telopeptide concentrations greater in protein group |
| Mullins & Sinning, 2005 [90] | Randomised, double-blind, placebo-controlled design 24 healthy, untrained, young adult F (18–29 years) engaged in 12-week RET 3 d/week and were randomised to protein (22.8 ± 0.9 years) or placebo (22.7 ± 1.1 years) during the final 10 days (mean ± SEM) | High-protein diet (during final 10 days): purified whey protein for daily protein intake of 2.4 g/kg/d Control: equivalent dose of isoenergetic CHO | Bone markers, dietary analysis | High protein intake for final 10 days of RET had no effects on bone metabolism |
| Holm et al., 2008 [91] | Randomised, double-blind, placebo-controlled design Postmenopausal F were randomised to a protein-containing nutrient supplement (n = 13, 55 ± 1 years) or placebo (n = 16, 55 ± 1 years) in conjunction with 24-week RET (mean ± SEM) | Nutrient supplement containing: 10 g whey protein, 31 g CHO, 1 g fat, 250 mg calcium and 5 µg vitamin D. 730 kJ in total. Placebo supplement containing: 6 g CHO and 12 mg calcium. 102 kJ in total. Supplements were consumed after each training session | BMD, bone markers, dietary analysis | Nutrient group had greater increase in BMD at the femoral neck than controls Increased bone formation and osteocalcin following training in nutrient group |
| Wright et al., 2017 [92] | Randomised, double-blind, placebo-controlled design Obese/overweight adults were randomised to 0 g protein (n = 68, 50 ± 7 years) 20 g protein (n = 72, 48 ± 8 years) or ≥40 g protein (n = 46, 49 ± 8 years) combined with 36-week RET and aerobic exercise training 3 d/week for 36 weeks (mean ± SD) | Unrestricted diet in combination with whey protein supplementation (0, 20, 40 or 60 g/d) (40 and 60 g group combined to form a ≥40 g group for analysis) | BMD, bone mineral content (BMC), protein intake | Whey protein, regardless of dose, had no effect on BMD or BMC during training |
| Farnsworth et al., 2003 [93] | Parallel design 57 overweight adults randomised to either high protein (M n = 7 51.9 ± 3.3 years, F n = 21, 50.6 ± 2.7 years) or standard protein (M n = 7 48.6 ± 3.2 years, F n = 22, 50.6 ± 2.1 years) diet during 12 weeks of energy restriction and 4 weeks of energy balance (mean ± SEM) | High-protein diet of meat, poultry and dairy foods (27% of energy as protein, 44% as CHO, and 29% as fat) Standard protein diet low in those foods (16% of energy as protein, 57% as CHO, and 27% as fat) Diets during 12 weeks of energy restriction (6–6.3 MJ/d) and 4 weeks of energy balance (≈8.2 MJ/d) | Calcium excretion, bone markers, dietary analysis | Markers of bone turnover and calcium excretion unchanged between diet groups |
| Bowen et al., 2004 [3] | Randomised study design Overweight adults were randomly assigned to isoenergetic diets high in dairy protein (M 49.4 ± 3.2 years, F 46.5 ± 2.4 years) or mixed source protein (M 48.7 ± 4.2 years, F 46.1 ± 2.7 years) during 12 weeks of energy restriction and 4 weeks of energy balance (mean ± SEM) | Isoenergetic diets (34% of energy as protein) high in either dairy protein (~2400 mg calcium/d) or mixed protein sources (~500 mg calcium/d) | Calcium excretion, bone markers, dietary analysis | Urinary calcium excretion decreased independently of diet Greater increase in bone resorption marker deoxypyridinoline with mixed protein Increased osteocalcin in mixed protein group |
| Josse et al., 2012 [94] | Randomised, controlled, parallel intervention design Premenopausal overweight and obese F were randomised into high protein/high dairy (30 ± 1 years), adequate protein/medium dairy (26 ± 1 years) or adequate protein/low dairy protein (28 ± 1 years) (mean ± SEM) | High protein/high dairy: dietary protein (30% of energy), dairy foods (15% energy from protein) and dietary calcium (~1600 mg/d) Adequate protein/medium dairy: dietary protein (15% of energy), dairy foods (7.5% energy from protein) and dietary calcium (~1000 mg/d) Adequate protein/low dairy: dietary protein (15% of energy), dairy foods (<2% energy from protein) and dietary calcium (<500 mg/d) | Bone markers | With low dairy, C-terminal telopeptide of collagen type-I, urinary deoxypyridinoline and osteocalcin increased With high dairy, osteocalcin, amino-terminal propeptide of collagen I increased with resorption markers unchanged |
| Reference | Study Design | Protein Composition | Measurements | Key Outcomes |
|---|---|---|---|---|
| Hartman et al., 2007 [106] | Randomised, controlled, parallel intervention design Soy protein (n = 19) vs. milk (n = 18) vs. carbohydrate (CHO) control (n = 19) Healthy young males (M) (18–30 years). 12 weeks 5 d/week whole-body resistance exercise training (RET) | Soy protein—17.5 g isoenergetic/nitrogenous milk—17.5 g protein CHO—isoenergetic 2 × supplement, post exercise + 1 h | Fat- and bone-free mass (FBFM), fibre cross (CSA), plasma amino acid (AA) profile | No increased FBFM in soy group Soy protein increased type I fibre CSA after 12 weeks, however milk greatly increase type I + II CSA Soy protein increased post-ingestion plasma leucine and EAA profiles similar to milk Increased plasma insulin immediately after ingestion similar to milk |
| Tang et al., 2009 [97] | Randomised, controlled, parallel intervention design Soy vs. whey vs. casein protein All groups n = 6 Healthy young M (22.8 ± 3.9 years, mean ± standard error of the mean (SEM)) Unilateral leg press and knee extension (4 sets, 10–12 repetition maximum (RM)) | Soy protein—22.2 g protein, 1.8 g leucine Whey protein—21.4 g protein, 2.3 g leucine Casein protein—21.9 protein, 1.8 g leucine All provided ~10 g EAA Protein drink post exercise. | Rest and exercise muscle fractional synthesis rates (FSR), plasma AA profile | Soy and whey protein increased rest muscle FSR above casein Soy protein + exercise muscle FSR increased above casein protein, however a greater increase was seen in whey protein + exercise Soy protein ingestion increase EAA + leucine profiles above casein protein, with whey protein ingestion increasing both to a greater degree |
| DeNysschen et al., 2009 [105] | Randomised, double-blind, controlled parallel intervention design Soy protein (n = 10) vs. whey protein (n = 9) vs. CHO placebo (n = 9) Overweight males (21–50 years, mean 38 years, body mass index (BMI) 25–30) 12 weeks 3 d/week whole-body RET | Soy protein—25.8 g Whey protein—26.6 g CHO placebo—0.6 g protein Supplement ingested post-resistance exercise (RE), daily | Body composition, strength, fasting blood measures | All groups increased strength pre to post Total cholesterol decreased in all groups No differences between groups for any measures |
| Wilkinson et al., 2007 [103] | Randomised cross-over intervention design Soy protein vs. milk n = 8 Healthy young M (21.6 ± 0.3 years, mean ± SEM) Unilateral standardised leg workout, 80% 1-RM | Soy protein—18.2 g Isoenergetic/nitrogenous milk—18.2 g protein Protein drink post RE | AV balance-based FSR and fractional breakdown rate (FBR), net balance, plasma AA profile | A significant, but lower increase in total AA and muscle FSR after consumption of soy protein vs. milk Soy protein ingestion resulted in a shorter period of positive net protein balance and area under the curve compared to milk Total AA net balance remained elevated after milk consumption vs. soy protein |
| Luiking et al., 2011 [108] | Randomised, single-blind parallel intervention design Soy protein (n = 10) vs. casein protein (n = 12) Healthy young adults (M/females (F) 50:50, 22 ± 1 years, mean ± SEM) | Soy protein—3.4 g protein/100 mL Isonitrogenous casein protein—2.95 g/100 mL Enteral ingestion (2 mL/kg/bw/h) | AV balance based FSR & FBR, net balance, plasma AA profile | Greater net uptake of glutamate, serine, histidine and lysine from casein vs. soy protein Reduced intramuscular branch AA concentrations from soy ingestion compared to casein No differences in muscle protein synthesis (MPS) or muscle protein breakdown between protein sources |
| Joy et al., 2013 [23] | Randomised, double-blind, parallel intervention design Rice protein vs. whey protein isolate All groups n = 12 Healthy young males (21.3 ± 1.9 years, mean ± standard deviation (SD)) Periodic whole-body RET | Rice protein—48 g protein, 80 mg/g leucine Isonitrogenous whey protein isolate—48 g protein, 115 mg/g leucine Ingested post exercise 3 d/week Control diet provided | Muscle thickness, body composition, strength measures | Both groups increased lean mass (LM), bicep/quadricep thickness, with no differences between groups |
| Babault et al., 2015 [107] | Randomised, double-blind, controlled parallel intervention design Pea protein (n = 53) vs. whey protein (n = 54) vs. placebo (n = 54) Healthy young M (21.9 ± 3.7 years, mean ± SD) 6 weeks 3 d/week progressive strength training, elbow flexor/extensor | Pea protein—26.6 g protein, 2.9 g leucine Whey protein—23.9 g protein Placebo—3.9 g maltodextrin Ingested twice daily morning/afternoon (post exercise) for 6 weeks | Bicep thickness, maximal voluntary torque, 1-RM | All groups increased bicep thickness compared to baseline after 42 and 82 days, no difference between groups Baseline weakest volunteers supplemented with pea protein demonstrated increased bicep thickness between 42 and 84 days |
| Candow et al., 2006 [104] | Randomised, double-blind, controlled parallel intervention design Soy protein vs. whey protein vs. placebo All groups n = 9 Healthy young adults (M/F 1:2, 23 ± 6 years, mean ± SD) 6 weeks 3 d/week whole-body RET | Soy and whey protein—1.2 g/kg Placebo—1.2 g/kg maltodextrin, isocaloric Ingestion split between 3 equal daily doses pre/post-training and evening | Body composition, strength measures, muscle FBR | Both soy and whey protein groups increased LM and strength greater than the placebo group All groups increased muscle FBR similarly |
| Yang et al., 2012 [71] | Parallel intervention, controlled design Soy protein 20 g or 40 g vs. whey protein 20 g or 40 g vs. water All groups n = 10 Healthy older M (71 ± Unilateral knee extension (3 sets, 10-RM). | Soy protein—20 g protein, 1.6 g leucine Soy protein—40 g protein, 3.2 g leucine Whey protein—20 g protein, 2 g leucine Whey protein—40 g, 4 g leucine Water control Protein drink post exercise | Myofibrillar FSR (rest and RE) plasma AA profile, leucine oxidation | No increase in rest myofibrillar FSR in either 20 or 40 g soy protein groups Increased RE myofibrillar FSR in 40 g soy protein group Significant increases in myofibrillar FSR for all whey protein groups, rest + RE 20 and 40 g soy protein increased leucine oxidation to similar degrees |
| Deibert et al., 2011 [109] | Randomised controlled intervention design Whole-body RET with/without soy protein Healthy moderately overweight older M (55.7 ± 4.6 years, BMI 27.7 ± 2.1, mean ± SD) 12 weeks 2 d/week progressive whole-body RET | 50 g soy protein yoghurt—26.7 g protein Control—RET only Consumed after evening training | Skinfold measures, BMI, strength measures, blood biomarkers | Decreased waist circumference and fat mass and increased fat free mass in soy protein supplemented group Improved glycaemic control and metabolic markers in soy protein-supplemented group Both groups increased in strength and coordination |
| Gorissen et al., 2016 [99] | Randomised, double-blind, controlled parallel intervention design 35 g wheat protein vs. 35 g or 60 g wheat protein hydrolysate vs. 35 g micellar casein protein, 35 g whey protein All groups n = 12 Healthy older M (71 ± 1 years, mean ± SEM) Single protein drink ingestion | Wheat protein—35 g Wheat hydrolysate protein—35 g Wheat hydrolysate protein—60 g Micellar casein protein—35 g Whey protein—35 g Single ingestion | Myofibrillar FSR, plasma AA profile | Ingestion of 35 g wheat protein did not increase myofibrillar FSR as much as 35 g whey or 35 g casein protein 60 g wheat hydrolysate stimulated myofibrillar FSR to a greater degree than 35 g whey protein 2–4 h post-ingestion Whey protein ingestion had a greater plasma leucine increase compared to 60 g wheat hydrolysate protein Plasma AA content was more persistent following 60 g wheat hydrolysate ingestion |
| Oikawa et al., 2020 [102] | Single blind, parallel group design 24 F randomised to potato protein (n = 12, 20 ± 3) or control (n = 12, 21 ± 3) diet for 2 weeks plus unilateral RET (3 ×/weeks) (mean ± SD) | Potato protein—25 g 2 ×/d (1.6 g/kg/d total protein) Control—0.8 g/kg/d total protein (breakdown of AA composition within each supplement can be found in original article) | Myofibrillar protein synthesis, cell signalling, baseline body composition and strength, dietary analysis | No difference in total kcals or percentage fat intake between groups Protein intake was significantly greater in the potato protein group compared to control MPS increased above baseline at rest in the potato protein, but not control, group MPS increased similarly above baseline with exercise in both groups In response to exercise, total protein kinase B (PKB/Akt) increased compared to baseline Main effect of time for total mechanistic target of rapamycin and ribosomal protein s6 |
| Reference | Study Design | Protein Composition | Measurements | Key Outcomes |
|---|---|---|---|---|
| Roughead et al., 2005 [87] | Randomised cross-over intervention study design Low meat soy supplemented vs. high meat n = 13 7 weeks, healthy postmenopausal females (F) (59.9 ± 5 years, mean ± standard deviation (SD)) | Low meat soy supplemented—55 g/d meat, 25 g soy protein High meat—170 g/g meat All meals provided | Calcium retention, urine composition, blood biomarkers of bone mineral status | No difference in calcium retention between groups No change in blood biomarkers of bone mineral status (i.e., 25-OH vitamin D, parathyroid hormone, insulin-like growth factor-I. |
| Kreijkamp-Kaspers et al., 2004 [119] | Randomised, double-blind, controlled parallel intervention design Soy protein supplement (n = 88) vs. milk protein supplement (n = 87) 12 months, healthy postmenopausal F (66 ± 5 years) | Soy protein—25.6 g protein Milk protein—25.6 g protein Single daily ingestion | Hip and lumbar spine bone mineral density (BMD), plasma lipid profiles | No difference in BMD from supplementation No change in plasma lipid profiles |
| Alekel et al., 2000 [116] | Randomised, double-blind, controlled parallel intervention design Isoflavone-rich soy protein (n = 24) vs. isoflavone-poor soy protein (n = 24) vs. whey protein control (n = 21) 24 weeks supplementation postmenopausal F (42–62 years, mean 50 years) | All groups 40 g protein/d, 160 mg calcium/d Isoflavone-rich 80.4 mg aglycone Isoflavone-poor 4.4 g aglycone Single daily 500 kcal muffin (20 g protein) as a meal replacement | Lumbar spine BMD and bone mineral content (BMC) | Both soy protein groups did not significantly decrease BMD, whereas the whey control group did BMC increase in the isoflavone-rich soy group and decreased in the whey protein control group. |
| Liu et al., 2010 [120] | Randomised, double-blind, controlled parallel intervention design Soy protein + isoflavone whey protein + isoflavone whey protein control 6 months postmenopausal F (56.1 ± 4.3 years, mean ± SD) | Soy protein—15 g, 100 mg isoflavone Whey protein—15 g, 100 mg isoflavone Whey protein—15 g Daily supplementation | Body composition | Soy protein with isoflavone supplementation demonstrated small but significant improvements in body weight, body mass index and body fat percentage |
| Reference | Study Design | Protein Composition | Measurements | Key Outcomes |
|---|---|---|---|---|
| Oikawa et al., 2020 [27] | Double-blind, parallel group, randomised controlled trial (RCT) within-subject design (unilateral leg—rest, contralateral leg—resistance exercise (RE)) 22 healthy older female (F) (n = 11/group, 69 ± 3 years, mean ± standard deviation (SD)) Randomised to collagen protein or whey protein 2 ×/d for 6 d and unilateral RE twice during 6 d period | Collagen protein—30 g amino acids (AA) of hydrolysed collagen protein Whey protein—30 g AA of whey protein isolate (breakdown of AA composition within each supplement can be found in original article) | Myofibrillar and collagen protein synthesis, cell signalling, baseline body composition and strength | Plasma leucine concentrations increased above baseline post whey protein, but not collagen peptide supplementation Myofibrillar muscle protein synthesis (MPS) increased at rest and post-RE following whey protein, but only increased post-RE following collagen peptide supplementation Collagen peptide supplementation did not influence integrated myofibrillar MPS Rates of integrated myofibrillar MPS significantly greater in whey protein than collagen peptide supplementation |
| Kirmse et al., 2019 [128] (uses data set from Oertzen-Hagemann et al., 2019) | Randomised, double-blind, placebo-controlled design 57 moderately trained males (M) (24 ± 3 years, mean ± SD) were randomised to full-body resistance exercise training (RET) 3 ×/week for 12 weeks and collagen peptide (n = 29) or placebo (n = 28) Supplements taken daily for 12 weeks | Hydrolysed collagen peptide—15 g/d Placebo—15 g/d noncaloric silicon dioxide | Body composition, muscle thickness, strength, muscle fibre cross sectional area (CSA), dietary analysis | Strength and type II CSA increased in both groups Fat free mass (FFM) significantly increased in the collagen peptide group, not placebo Body fat mass (FM) did not change in the collagen peptide group but increase in the placebo group No difference in macronutrient intake between groups Protein intake was 1.81 ± 0.42 and 1.74 ± 0.5 g/kg/d in collagen and placebo groups, respectively |
| Zdzieblik et al., 2015 [26] | Randomised, double-blind, placebo-controlled design 53 older (72.2 ± 4.68 years, mean ± SD) sarcopenic M randomised to full body RET 3 ×/week for 12 weeks and collagen peptide (n = 26) or placebo (n = 27) Supplements taken daily for 12 weeks | Collagen peptide—15 g/d Placebo—15 g/d silicon dioxide (breakdown of AA composition within collagen peptide supplement can be found in original article) | Body composition, strength, dietary analysis | Increase in FFM and strength greater in collagen peptide versus placebo group Decrease in FM was greater in collagen peptide versus placebo group No difference in dietary intake between groups pre or post intervention and neither were protein deficient |
| Jendricke et al., 2019 [135] | Randomised, double-blind, placebo-controlled design 77 premenopausal untrained F were randomised to full body RET 3 ×/week for 12 weeks and collagen peptide (n = 40, 38.3 ± 8.7 years) or placebo (n = 37, 41.6 ± 6.9 years) (mean ± SD) Supplements taken daily for 12 weeks | Collagen peptide—15 g/d Placebo—15 g/d noncaloric silicon dioxide | Body composition, strength | Increase in FFM and hand grip strength was higher in collagen peptide versus placebo group Decrease in percentage body fat was greater in collagen peptide versus placebo group |
| Oertzen-Hagemann et al., 2019 [28] | Randomised, double-blind, placebo-controlled design 25 M (24.2 ± 2.6 years, mean ± SD) were randomised to full body RET 3 ×/week for 12 weeks and collagen peptide (n = 12) or placebo (n = 13) Supplements taken daily for 12 weeks | Hydrolysed collagen peptide—15 g/d Placebo—15 g/d noncaloric silicon dioxide | Body composition, strength, proteome | Collagen peptide is bioactive, demonstrated by increased circulating levels of hydroxyproline 2 h following collagen peptide ingestion Body mass and FFM higher in collagen peptide group versus placebo 221 higher abundant proteins identified in collagen peptide group versus on 44 in placebo (proteomic analysis) Upregulated proteins in the collagen peptide group mostly associated with protein metabolism of contractile fibres |
| Hays et al., 2009 [127] | Double-blind, randomised, cross-over design 9 healthy F (71 ± 1 years, mean ± standard error of the mean (SEM)) completed 2 × 15 d trials (7 d wash-out period in between) Each trial consisted of consuming 0.8 g protein/kg body weight/d with either whey protein or collagen peptide intended to provide ~0.4 g/kg body weight/d | Hydrolysed collagen peptide—~0.4 g/kg body weight/d Whey protein—~0.4 g/kg body weight/d | Body composition, nitrogen balance, dietary analysis | Body weight decreased after whey but not collagen protein intake Nitrogen excretion was higher during whey versus collagen protein intake No difference in macronutrient intake between collagen peptide and whey protein groups (protein intake was 0.82 ± 0.04 g/kg/d) |
| Oikawa et al., 2018 [77] | Double-blind, parallel group, RCT 16 M (69 ± 3 years) and 15 F (68 ± 4 years) were randomised to collagen peptide (n = 15) or whey protein (n = 16) and completed 4 phases: 1. 1-week energy balance 2. 1-week energy restriction (−500 kcal/d) and protein supplementation (1.6.g protein/kg/d with 45 ± 9% from whey protein (30 g 2 ×/d) or collagen peptide (30 g 2 ×/d)) 3. 2-week energy restriction with step reduction (≤750 steps/d) 4. 1-week habitual activity (continuing the high protein supplementation protocol) (mean ± SD) | Hydrolysed collagen peptide—30 g Whey protein isolate—30 g (breakdown of AA composition within each supplement can be found in original article) | Myofibrillar MPS, body composition, fascicle CSA, inflammation, insulin sensitivity | Protein supplementation (whey protein or collagen peptide) did not prevent leg LM loss during energy restriction and energy restriction with step reduction Whey protein, but not collagen peptide, augmented lean body mass, leg LM and MPS during habitual activity MPS remained suppressed during the energy restriction with step reduction and habitual activity phases in the collagen peptide group |
| Impey et al., 2018 [129] | Repeated-measures, counterbalanced design 7–9 d wash-out period 8 recreational M cyclists (25 ± 3 years, mean ± SD) completed an exercise trial in conditions of reduced carbohydrate with hydrolysed collagen or whey protein consumed before, during and after exercise | Hydrolysed collagen blend—22 g (66 g total) taken pre, during and post-exercise Whey protein—22 g (66 g total) taken pre, during and post-exercise | Cell signalling, muscle mitochondria markers | No effect of hydrolysed collagen (or whey protein) on markers of muscle mitochondrial adaptations Hydrolysed collagen supplementation increased anabolic signalling but to a lesser extent than whey protein |
| Clifford et al., 2019 [29] | Double-blind, placebo-controlled, independent group design 24 recreationally active M were randomised to collagen peptide (n = 12, 24.1 ± 4.3 years) or placebo (n = 12, 24.8 ± 4.8 years) supplementation 7 d before and 2 d after exercise (mean ± SD) | Collagen peptide—20 g/d Isoenergetic and isovolumic placebo—20 g/d | Muscle function, dietary analysis | Countermovement jump recovered quicker following collagen peptide supplementation (versus placebo) No difference in macronutrient intake between groups throughout the study Protein intake was 1.26 ± 0.46 and 1.18 ± 0.27 g/kg/bm−1 for collagen peptide and placebo groups, respectively. |
| Rindom et al., 2016 [132] | Double-blind, randomised, cross-over design 12 young M (24.6 ± 2.1 years, mean ± SD) completed 1 week of intense full-body RET (4 RET sessions) whilst consuming collagen protein or whey protein, followed by 3 weeks recovery, then completed another 1-week period of intense RET whilst consuming collagen or whey protein (opposite to the type ingested during the first week) | Collagen protein—20 g/d Whey protein—20 g/d During the intense RET period, all volunteers received 1.4 g protein/kg bodyweight in addition to the study supplement (i.e., whey/collagen protein) | Muscle function | 48 h after the final exercise bout, maximal voluntary contraction had returned to baseline in both groups. No difference was noted between whey or collagen protein groups at any timepoint 48 h after the final exercise bout, counter movement jump (CMJ) height had returned to baseline in the collagen protein, but not whey protein, supplemented group 3 h after the final exercise bout, whey protein supplemented group displayed attenuated losses in CMJ compared to collagen protein |
| Oikawa et al., 2019 [130] | Double-blind, randomised, cross-over design 4 d wash-out 11 endurance trained adults (M n = 5, F n = 6, 24 ± 4 years, mean ± SD) engaged in daily high-intensity interval training with hydrolysed collagen or α-lactalbumin supplementation for 3 d | Hydrolysed collagen peptides—60 g/d α-lactalbumin—60 g/d (breakdown of AA composition within each supplement can be found in original article) | Myofibrillar and sarcoplasmic MPS, dietary analysis | Plasma leucine and tryptophan concentrations were greater following α-lactalbumin compared to hydrolysed collagen supplementation Exercise-induced increased in myofibrillar and sarcoplasmic MPS were greater with α-lactalbumin compared to hydrolysed collagen supplementation No differences in macronutrient intake between groups |
| Centner et al., 2019 [137] | Prospective, randomised, placebo-controlled design Older M randomised to 8 weeks blood flow resistance (BFR) training with collagen hydrolysate (n = 11, 61.7 ± 5.5 years) or 8 weeks BFR training with placebo (n = 11, 56.6 ± 6.1 years) or no training with collagen hydrolysate (control) (n = 8, 62.5 ± 10.5 years) (mean ± SD) | Collagen hydrolysate—15 g/d Placebo—silicon dioxide—15 g/d | CSA and muscle function | Muscle CSA increase in BFR-collagen hydrolysate (+6.7% ± 3.2%) and BFR-placebo (+5.7% ± 2.7%) but not in control 1-repition maximum strength increased in BRF-collagen hydrolysate (+10.2% ± 24.8%), and BFR-placebo (+4.8% ± 11.4%) but not control, relative to pre-study levels |
| Reference | Study Design | Protein Composition | Measurements | Key Outcomes |
|---|---|---|---|---|
| Zdzieblik et al., 2015 [26] (also studied effects on muscle, see Table 5) | Randomised, double-blind, placebo-controlled design 53 older (72.2 ± 4.68 years, mean ± standard deviation (SD)) sarcopenic males (M) randomised to full body resistance exercise training (RET) 3 ×/week for 12 weeks and collagen peptide (n = 26) or placebo (n = 27) Supplements taken daily for 12 weeks | Collagen peptide—15 g/d Placebo—15 g/d silicon dioxide (breakdown of amino acid composition within collagen peptide supplement can be found in original article) | Bone mass, dietary analysis | No potentiating effect of collagen peptide on bone mass (beyond that of RET alone) No difference in dietary intake between groups pre- or post-intervention and neither were protein deficient |
| Clifford et al., 2019 [29] (also studied effects on muscle, see table above) | Double-blind, placebo-controlled, independent group design 24 recreationally active M were randomised to collagen peptide (n = 12, 24.1 ± 4.3 years) or placebo (n = 2, 24.8 ± 4.8 years) supplementation 7 d before and 2 d after exercise (mean ± SD) | Collagen peptide—20 g/d Isoenergetic and isovolumic placebo—20 g/d | Bone turnover markers, dietary analysis | Collagen peptide had no effect on markers of bone turnover No difference in macronutrient intake between groups throughout the study No difference in macronutrient intake between collagen peptide and placebo groups. Protein intake was 1.26 ± 0.46 and 1.18 ± 0.27 g/kg/bm−1 for collagen peptide and placebo groups, respectively |
| König et al., 2018 [141] | Randomised, double-blind, placebo-controlled design Postmenopausal females (F) randomised to collagen peptide (n = 66, 63.8 ± 7.4 years) or placebo (n = 65, 64.9 ± 7.1 years) supplementation for 12 months (mean ± SD) | Collagen peptide—5 g/d Maltodextrin (placebo)—5 g/d | Bone mineral density (BMD), bone turnover markers, dietary analysis | BMD increased following collagen peptide supplementation (no change in placebo) Amino-terminal propeptide of collagen I (P1NP) increased following collagen peptide supplementation C-terminal telopeptide of collagen type-I increased following placebo supplementation No difference in macro or micronutrient intake between the groups pre or post intervention and neither were protein deficient |
| Shaw et al., 2017 [126] | Double-blind, randomised, cross-over design 4 d washout period 8 healthy recreationally active M (27 ± 6 years, mean ± standard error of the mean) provided placebo, 5 or 15 g vitamin-C-enriched gelatin and completed rope skipping exercise, 3 ×/d for 3 days | 5 g vitamin-C (48 mg)-enriched gelatin 15 g vitamin-C (48 mg)-enriched gelatin Maltodextrin (placebo)—weight and calorie matched | Bone turnover marker | 15 g vitamin-C-enriched gelatin increased circulating P1NP more so than placebo and 5 g |
| Elam et al., 2014 [140] | Randomised, double-blind, placebo-controlled design 39 postmenopausal F (55.7 ± 3.3 years, mean ± SD) randomised to daily hydrolysed calcium-collagen chelate or placebo supplementation for 12 months | Hydrolysed calcium-collagen chelate—5 g/d (with 500 mg elemental calcium, 200 IU vitamin D3) Control—500 mg elemental calcium, 200 IU vitamin D3 | Total body, lumbar and hip BMD, bone turnover markers | Loss of total body BMD was lower following 12 months supplementation of hydrolysed calcium-collagen chelate versus control (n = 22 at 12 months follow up) Sclerostin and tartrate-resistant acid phosphatase isoform 5b (TRAP5b) were lower and bone-specific alkaline phosphatase/TRAP5b ratio was higher following 6 months supplementation of hydrolysed calcium-collagen chelate versus control |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Deane, C.S.; Bass, J.J.; Crossland, H.; Phillips, B.E.; Atherton, P.J. Animal, Plant, Collagen and Blended Dietary Proteins: Effects on Musculoskeletal Outcomes. Nutrients 2020, 12, 2670. https://doi.org/10.3390/nu12092670
Deane CS, Bass JJ, Crossland H, Phillips BE, Atherton PJ. Animal, Plant, Collagen and Blended Dietary Proteins: Effects on Musculoskeletal Outcomes. Nutrients. 2020; 12(9):2670. https://doi.org/10.3390/nu12092670
Chicago/Turabian StyleDeane, Colleen S, Joseph J Bass, Hannah Crossland, Bethan E Phillips, and Philip J Atherton. 2020. "Animal, Plant, Collagen and Blended Dietary Proteins: Effects on Musculoskeletal Outcomes" Nutrients 12, no. 9: 2670. https://doi.org/10.3390/nu12092670
APA StyleDeane, C. S., Bass, J. J., Crossland, H., Phillips, B. E., & Atherton, P. J. (2020). Animal, Plant, Collagen and Blended Dietary Proteins: Effects on Musculoskeletal Outcomes. Nutrients, 12(9), 2670. https://doi.org/10.3390/nu12092670







