1. Introduction
Over the past two decades, research on the gut microbiota has revealed that it plays an important role in maintaining health and preventing disease [
1]. Although novel clinical applications of microbiota modulation are mainly restricted to fecal microbiota transplantation for treatment of
Clostridium difficile [
2], gut-targeted dietary supplements are a large and growing segment of the public market [
3] Among these dietary supplements are probiotics, live beneficial microorganisms that confer benefits to the host through a variety of mechanisms [
4]. In addition to commercial availability as over-the-counter dietary supplements, they can also be found as natural components in many fermented foods or supplemented additives in non-fermented food products. Probiotics are being explored as an accessible, convenient, and relatively low-cost strategy to modify the gut microbiota for a variety of human health outcomes ranging from improved gastrointestinal (GI) function to the management of lipid and glucose metabolism [
5]. While there is still minimal scientific evidence to support many of these uses, there are data supporting the efficacy of probiotics for improving GI function. Efficacy appears to be somewhat dependent on the specific species/strains, or the combination, used for a given condition; however, a 2012 meta-analysis of randomized controlled trials showed that 8 of the 11 species/mixtures tested were efficacious for a range of GI conditions, including pouchitis, infectious and
C. difficile associated-diarrhea, Irritable Bowel Syndrome (IBS), and antibiotic-associated diarrhea [
6]. A more recent meta-analysis suggests that probiotics may also be beneficial in alleviating functional constipation in adults [
7]. Even in cases where there were insufficient data to demonstrate probiotic efficacy, adverse effects were minimal [
8], suggesting probiotics are a low-risk intervention to improve GI health in humans.
Bacteriophages, or phages, are also being explored for their potential to selectively modify the gut microbiota. Phages attach to a specific host bacterium, insert their genetic material into the cell, and take over the machinery of the host cell to replicate phage components. Once this process is complete, the cell is destroyed, or lysed, releasing new phage particles. Unlike antibiotics, phages display a narrow host range, limiting global perturbations to the gut microbiota that can promote dysbiosis, reduce intestinal homeostasis, and promote disease. In addition to their narrow host range, phages are ubiquitous in the environment, and many are Generally Regarded as Safe (GRAS) for human consumption. We recently showed that supplemental consumption of a cocktail of
E. coli-targeting bacteriophages, marketed as PreforPro, was considered safe and tolerable [
9]. We also demonstrated that these phages showed bifidogenic effects after 4 weeks of oral supplementation, with no global disruption of the gut microbiota [
10]. Based on these data, we hypothesized that, in addition to reducing target host species, phages may also reduce competition among commensal bacteria for limited resources and produce a potential fuel source in the form of contents released from the dead bacteria. Therefore, phages taken in combination with a probiotic may extend benefits on GI health and intestinal environment.
In the current study, we tested whether a combined probiotic and phage-based intervention extends probiotic impacts on gastrointestinal discomfort and stool consistency in a healthy adult population. Our randomized, double-blind, placebo-controlled intervention included three parallel arms in which participants were assigned to consume either
Bifidobacterium animalis subsp.
lactis BL04 (
B. lactis),
B. lactis with a commercial phage cocktail targeting
E. coli, or a maltodextrin-based placebo. We selected this combination because our previous data showed that PreforPro increased commensal
Bifidobacterium [
10]. The primary outcome measures included subjective digestive health questionnaires and stool consistency measurements based on the Bristol Stool Scale. We hypothesized that probiotic-consumption would improve one or more aspects of digestive health and stool consistency in our participant population and that addition of the phage cocktail would potentiate these effects. Specifically, our primary outcome measures included a digestive health questionnaire, stool consistency measurements, and molecular analysis of the gut microbiota. We hypothesized that consumption (
B. lactis BL04 + PreforPro) would improve one or more aspects of digestive health and stool consistency in our participant population compared to the probiotic alone.
2. Materials and Methods
2.1. Study Design
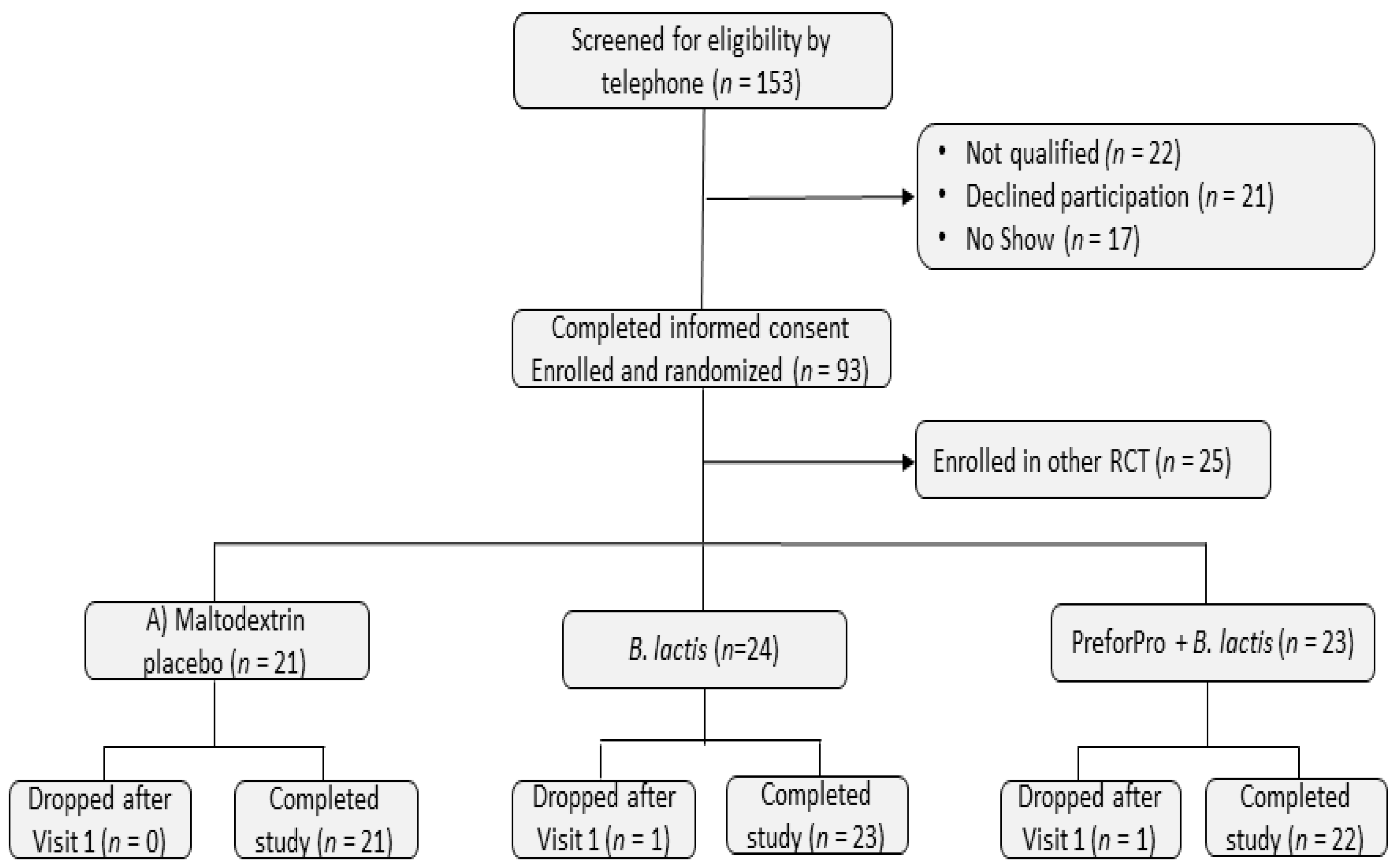
The BacterioPHAGE for Gastrointestinal Health 2 Study (PHAGE-2 Study; #NCT04511221) is a 4-week, randomized, parallel-arm, double-blind, placebo-controlled clinical intervention trial. The PHAGE 2 Study was designed to test whether combining supplemental bacteriophages with a probiotic would provide additional benefits to GI health and gut microbiota compared to consuming a probiotic alone. Data from our previous study [
10] were used to determine sample size, based on 80% power to detect a significant change (alpha = 0.05) in
Bifidobacterium levels between the placebo and treatment groups. The Colorado State University Institutional Review Board approved the study protocol (CSU #19-9145H) and all 68 participants enrolled in one of the three study arms provided written informed consent. This trial is registered at ClinicalTrials.gov under #NCT04511221.
Participants were pre-qualified by phone screening and attended an in-person study visit at Colorado State University’s Food and Nutrition Clinical Research Laboratory (FNCRL) in the Department of Food Science and Human Nutrition. Participants were fasted at least 8 h and asked to abstain from exercise for at least 12 h prior to clinic visits. They were also instructed to refrain from taking any medications or dietary supplements for 24 h prior to their visits. During their initial visit (baseline), eligibility was confirmed by taking anthropometric measurements of height (cm) and weight (kg) to calculate BMI and completing a written medical health questionnaire to determine medical history and current medication use. At both baseline and 4-week (final) visits, participants were asked to complete a digestive health questionnaire, undergo several measures of cardiovascular function (reported in Trotter et al. 2020 in press), and provide a venous blood and stool sample. They were also provided with a stool log and asked to keep a daily record of their bowel movements.
After undergoing sample collections and analysis procedures at the baseline visit, participants were randomly assigned to 1 of 3 treatments groups: (A) 15 mg rice maltodextrin (placebo); (B) 1 × 10
9 Colony Forming Units (CFU)
Bifidobacterium animalis subspecies
lactis strain BL04 (
B. lactis BL04); or (C) 1 × 10
9 CFU
B. lactis BL04 + 1 × 10
6 Plaque Forming Units (PFU) LH01-Myoviridae, LL5-Siphoviridae, T4D-Myoviridae, and LL12-Myoviridae bacteriophages, marketed as PreforPro (
B. lactis BL04 + PreforPro). Participants were asked to consume one 15-mg capsule per day during the 4-week intervention period. All treatment capsules contained maltodextrin from rice and medium chain triglycerides from palm and coconut oil as a filler material. Participants were randomized to treatment groups using the second generator at
www.randomization.com. To assess treatment compliance, participants returned all unused treatment capsules at their final visit.
Participants were asked to maintain their normal eating and exercise habits throughout the course of the study. To aid in determining dietary compliance, participants were completed a 2-day diet record at the beginning and end of the treatment period, including 1 weekday and 1 weekend day, using the National Institutes of Health, National Cancer Institute Automated Self-Administered 24-h dietary assessment tool (ASA24;
https://asa24.nci.nih.gov). For each of the 2-day diet records, participants recorded all foods, drinks, and supplements they consumed in a 24-h period and the time of day they were eating and drinking.
2.2. Participant Characteristics
Healthy, adult participants were recruited from Fort Collins, Colorado and surrounding areas by referral from local healthcare practitioners, flyers, email, and through word of mouth. Initial eligibility was determined by a phone screening questionnaire and confirmed onsite at the FNCRL via interview and BMI assessment by the clinical coordinator. Inclusion and exclusion criteria are presented in
Table 1. Participants were asked to maintain their regular diet and exercise habits as well as to limit alcohol consumption to 1–2 drinks per day or no more than 8 drinks per week during the study. They were also asked to abstain from taking any supplemental pre- or probiotics. A summary of baseline participant characteristics can be found in
Table 2.
2.3. Comprehensive Metabolic Panels
Blood samples (100 uL) were collected from the antecubital vein in a lithium heparin tube and immediately analyzed using a Piccolo Metlyte Plus CRP Reagent Disc, which included glucose, blood urea nitrogen (BUN), creatinine (CRE), creatinine kinase (CK), NA+, K+, Cl- and C-reactive protein (CRP), on the Piccolo Xpress Chemistry Blood Analyzer (Abaxis, Union City, CA, USA).
2.4. Digestive Health and Bowel Movement Assessment
Participants completed a digestive health questionnaire at baseline and after 4 weeks on treatment to assess perceived effects on GI symptoms, as previously described [
9]. Briefly, the questionnaire had 4 sections corresponding to gastric function, GI inflammation, small intestine (SI) and pancreas pain, and colon function (
Supplemental Methods File S1). Participants ranked questions within each section, choosing no/rarely, occasionally, often, or frequently. Symptom severity was then ranked according to their priority status outlined in
Table 3. The percentage of participants that experienced low, moderate, and high priority gastrointestinal symptoms at the initial and follow-up visit were calculated. Initial test scores (baseline) and retest scores (week 4) were compared within treatment groups across sections of the questionnaire to assess the percentage of participants that experienced an improvement, worsening, or no change to the severity of their gastrointestinal symptoms after the 4-week intervention.
Participants were also asked to record all bowel movements during the study and rank them according to the Bristol Stool chart. Participants were given a copy of the Bristol Stool Chart as a reference to help determine stool type. Types included Type 1 (separate hard lumps), Type 2 (lumpy and sausage like), Type 3 (a sausage shape with cracks in the surface), Type 4 (like a smooth, soft sausage or snake), Type 5 (soft blobs with clear-cut edges), Type 6 (mushy consistency with ragged edges) and Type 7 (liquid consistency with no solid pieces). For data analysis, each stool type was coded as either hard (Types 1 or 2), soft (Types 3, 4, or 5), or diarrhea (Type 6 or 7). Stools categorized as soft were considered normal, while hard and diarrhea stools were considered abnormal.
2.5. Stool Sample Processing
Stool samples were collected at home, stored refrigerated, and brought into the clinic within 24 h. In the clinic, they were stored at 4 °C and processed within 24 h in the following manner: two sterile cotton swabs were inserted into the stool at three separate locations and then stored at −80 °C for DNA extraction. Approximately 1 g of stool was mixed with PBS buffer containing 10% glycerol and stored at −80 °C prior to use for phage counts. Remaining stool was transferred to a 100 mL sterile plastic container and stored at −80 °C for possible future analyses.
2.6. Phage Enumeration
Fecal samples stored in glycerol were thawed at room temperature. Samples were vortexed for 5 min with ceramic beads in 1 mL 2× sodium chloride magnesium sulfide (SM) buffer to homogenize [
11]. A 1.5 mL aliquot was removed and centrifuged at 2000×
g for 2 min, and supernatant was removed and centrifuged again at 140,000 rpm for 10 min. An amount of 500 uL of prepared sample was mixed with 100 uL of
E. coli K12 culture (grown overnight in Luria Bertani (LB) broth on a 37 °C shaker to OD
600 = 0.7 − 1.0) and incubated at 37 °C for 5 min. An amount of 3 mL of freshly prepared LB soft agar was then added to the mixture and immediately poured on LB agar plates which were pre-warmed to 37 °C for 30 min. Each sample was plated in duplicate and quantified using plated phage standard dilution plates.
2.7. Microbiota Assessment
Fecal DNA was extracted using the FastDNA
® Kit (MP Biomedicals, #116540400) following manufacturer’s protocol. The V4 region of the 16S rRNA gene was amplified following the Earth Microbiome Project protocol using the 515F-806R primer set [
12] containing a unique 12 bp error correcting barcode included on the forward primer. Cycling and sequencing conditions were as previously described [
13]. DNA extraction controls, no template PCR controls, and the Zymo mock community were included on each sequencing plate. Sequence reads were imported into QIIME2 version 2020.2 for analysis [
14]. Briefly, the sequence reads were demultiplexed, and concatenated. Utilizing a Phred score cutoff of 30, upon examining the demultiplexed data, the reverse reads did not meet the quality filtering parameter. As such analysis proceeded with single-end sequences which were trimmed to 209 base pairs based on the same quality score. Reads were binned into ASVs using the DADA2 pipeline [
15]. Taxonomic assignments were made using GreenGenes version 13.8. Samples with low reads or suspected contamination were removed and mitochondrial and chloroplast sequences were filtered from remaining samples. The resulting feature tables and taxonomy files were imported into MyPhyloDB version v.1.2.0 [
16]. Alpha-diversity was calculated using Faith’s and Shannon metrics through the QIIME2 diversity plugin. Beta diversity was determined by Bray Curtis distance measurements and visualized by Principle Coordinates Analysis (PCoA) in MyPhyloDB. Differences in taxa among treatments were assessed in MyPhyloDB using univariate Analysis of Covariance (ANCoVA) and multivariate DiffAbund.
2.8. Statistical Analysis
Statistical analysis of clinical parameters (GI questionnaires, stool logs, metabolic panel analytes, dietary intakes, anthropometrics) and qPCR data was completed using GraphPad Prism, version 8.3.0. All data were analyzed using a 2-way mixed-model ANOVA with Sidak’s post hoc for multiple comparisons. In addition, baseline adjusted final values were analyzed using a 1-way ANOVA with Tukey’s post hoc for multiple comparisons. Statistical significance was assigned as
p < 0.05 and a statistical trend was defined as between
p < 0.10–0.051. Microbiota alpha-diversity parameters were analyzed using a one-way ANOVA with a non-parametric Kruskal–Wallis post hoc test, and β-diversity was analyzed by PERMANOVA with 1000 iterations. Analysis of Covariance (ANCoVA) with Tukey’s post-hoc for multiple comparisons and a negative binomial general linear model [
17] were used to determine statistically different taxa between time points and treatment groups.
4. Discussion
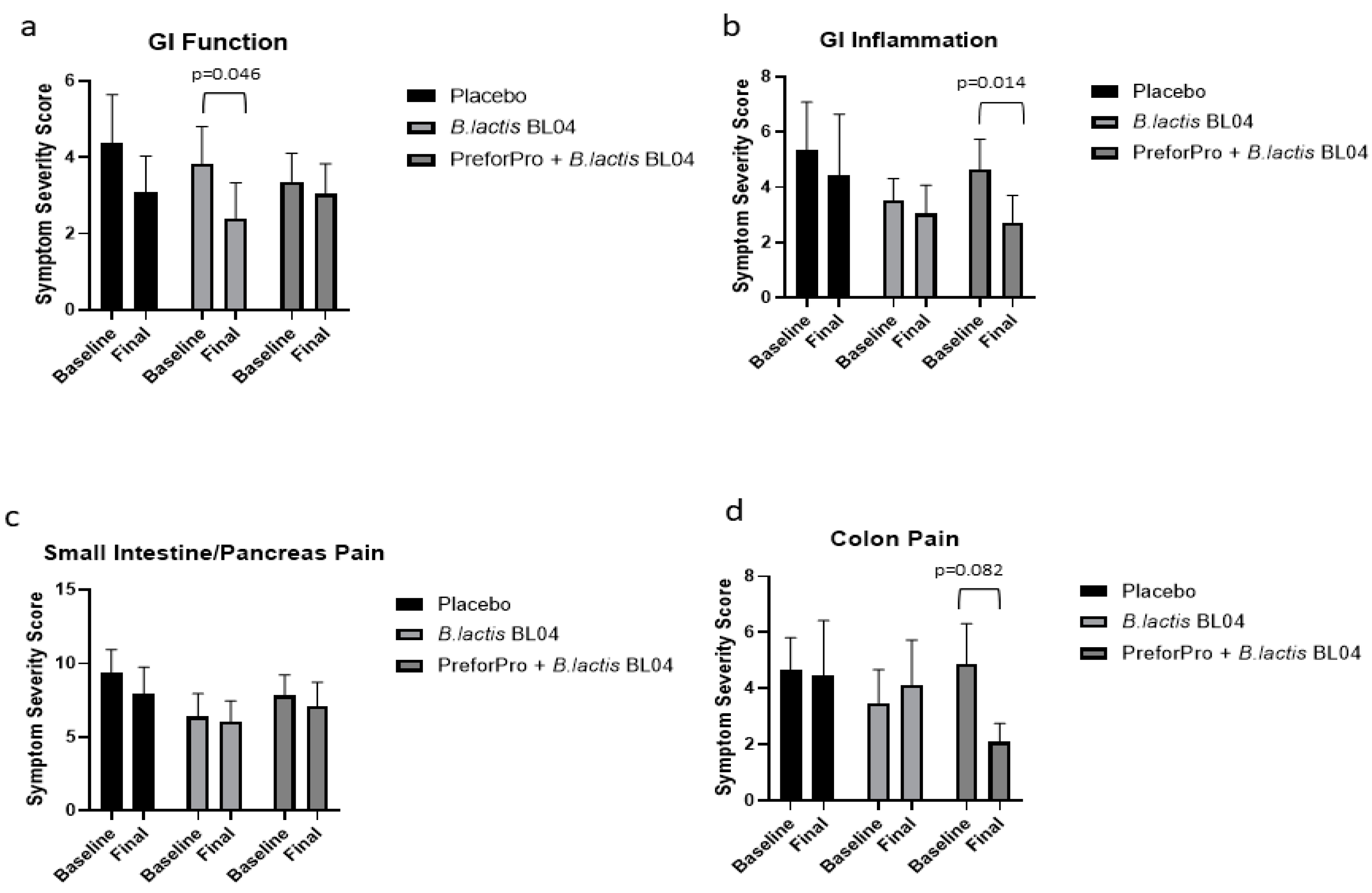
There is growing interest in the incorporation of phages with probiotic dietary supplements. Phages can target specific pro-inflammatory or pathogenic organisms in the gut and potentially enhance the GI benefits of the probiotics, although evidence for these effects in human trials is currently lacking. In this randomized, double-blind, placebo-controlled clinical study, we observed the effects of
B. lactis BL04, with or without the
E.coli-targeting phage mixture marketed as PreforPro, on gastrointestinal well-being and modulation of the gut microbiota. The beneficial effect of supplementation with
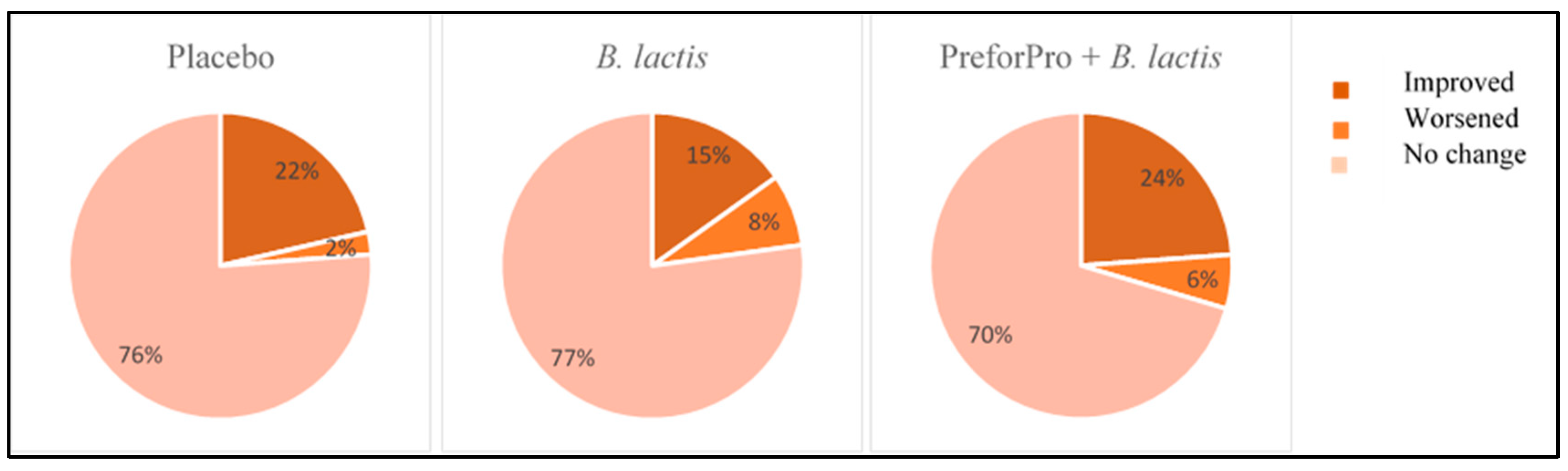
B. lactis BL04 appeared restricted to improvements in symptoms related to gastric function and stool consistency in 52% of participants. Participants who consumed
B. lactis BL04 + PreforPro showed improvements in digestive symptoms related to GI inflammation and colon pain and had the highest percentage of individuals reporting overall reduced symptom severity. This is largely in agreement with a previous study, where Ginden et al. reported that PreforPro alone resulted in an improvement in gastric function and colon pain, but likewise reported a placebo effect [
9]. Additionally, in the population of phage-only consuming participants, a higher percentage of participants reported an overall worsening in symptoms than was reported in the current study. This indicates that there might be some additive benefit to consuming these phages with a probiotic. However, the probiotic used in this study,
B. lactis BL04 alone, elicited minimal effects on our study population, which was largely healthy adults with no diagnosed GI diseases.
Bifidobacterium lactis BL04 is often studied in conjunction with other probiotic strains, and a recent study suggested that a five-strain mixture, containing
B. lactis BL04, improved symptoms of antibiotic-associated diarrhea when given at a high dose (>10
10 CFU) [
21]. Our study used a single strain, lower dose (10
9 CFU), and tested on a healthy population. While most people occasionally experience GI distress or irregular bowel movements, it is likely the effects of this probiotic are more subtle in healthy individuals. It would be interesting to determine whether the benefits we observed are more pronounced with a combination of probiotic and/or in patients with GI disorders.
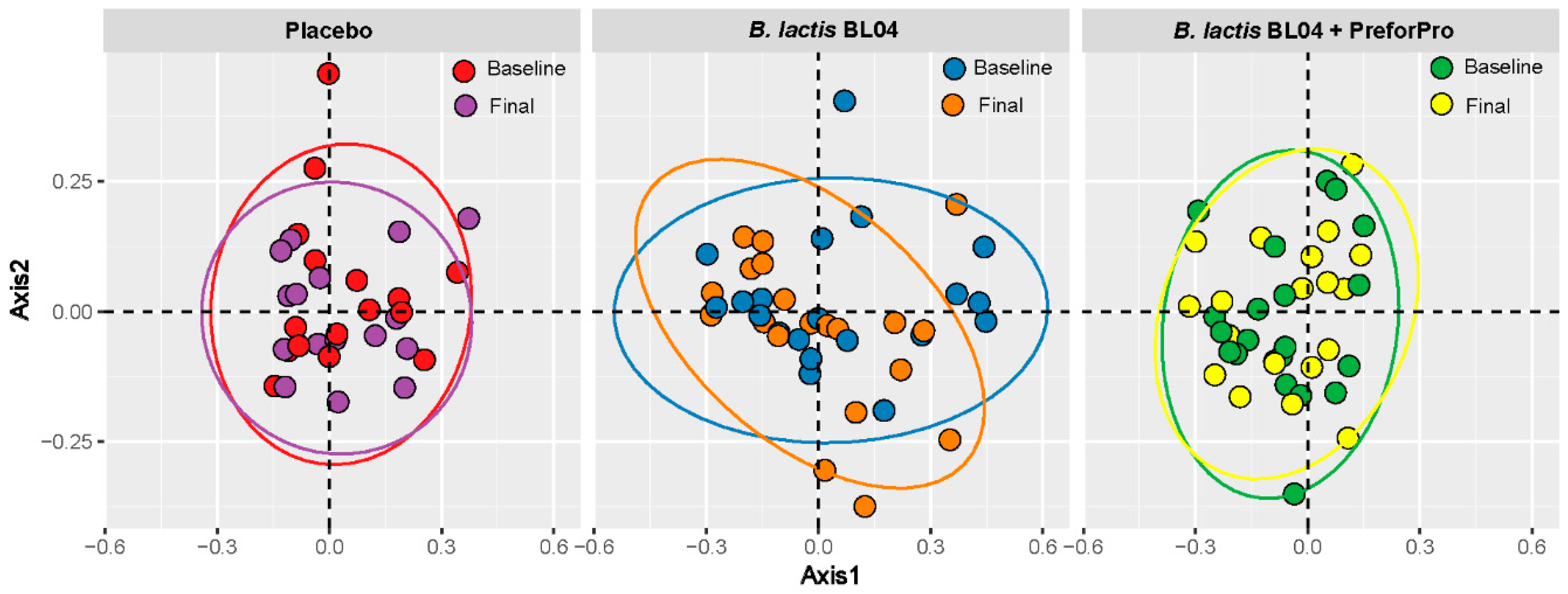
Consistent with other published phage literature, the combination of
B. lactis BL04 + PreforPro did not significantly impact gut ecology, with no significant shifts in either alpha- or beta-diversity parameters or large phyla-level changes in taxa. This concurs with our previous study, which looked at the impacts of the phages alone on gut microbiota [
10]. Similarly,
E. coli-targeting phages added to a simulated gut environment with a defined microbial consortium only showed effects on the target species and did not disrupt commensal organisms [
22]. However, various lytic phages administered to gnotobiotic mice transplanted with a defined human commensal microbiota did demonstrate cascading effects on many of the non-target species, likely mediated by complex ecological interactions between bacteria [
23]. This consortium was made up of only 10 bacterial species, suggesting that effects on these species may have been more profound than if they were observed within the context of an intact human gut containing hundreds of bacterial species.
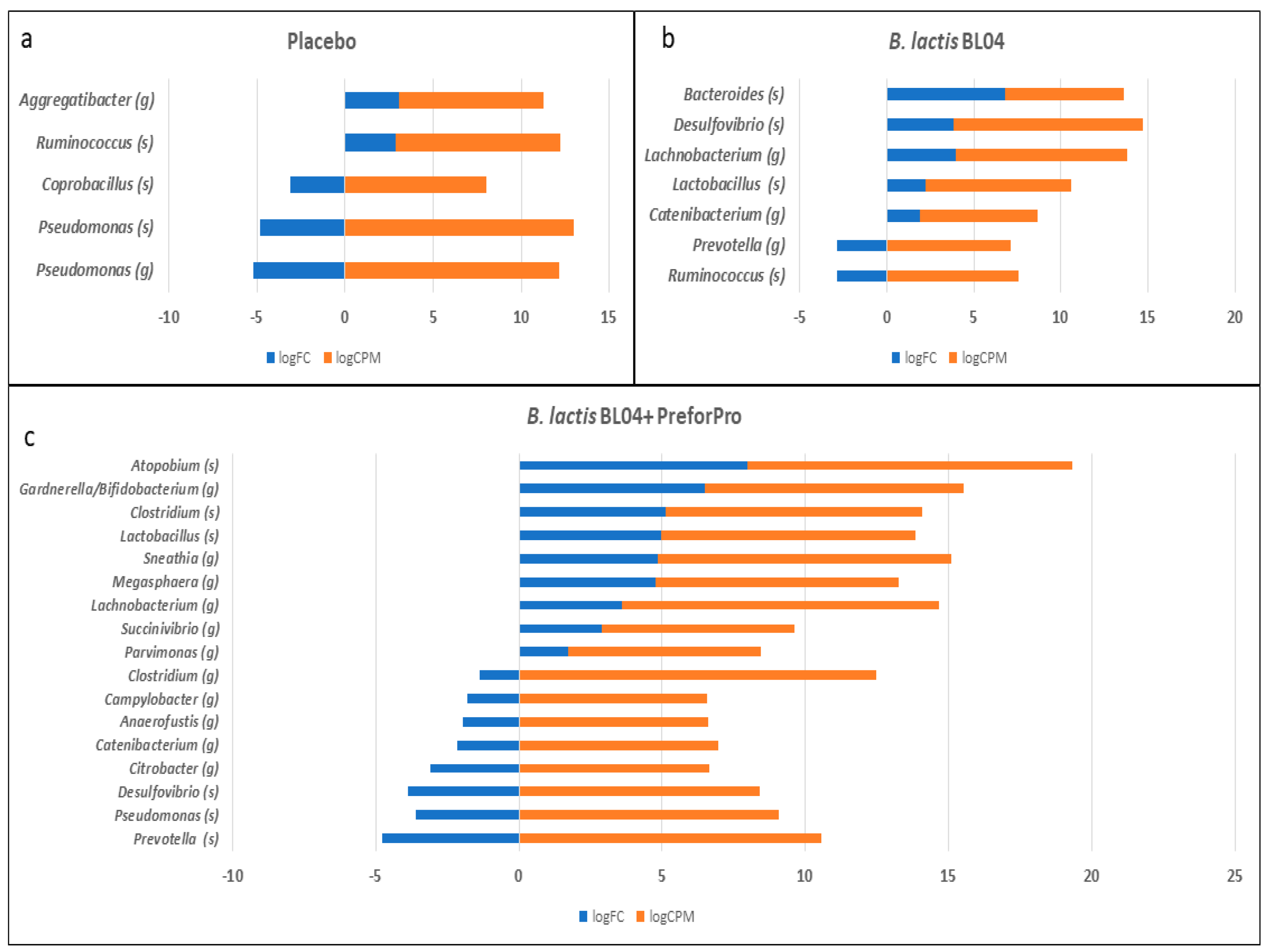
Despite a lack of global alterations in the microbiota, the treatments were associated with changes in a small number of taxa. Most notably, we saw an increase in taxa identified as
Lactobacillus after 4-weeks of both
B. lactis BL04 and
B. lactis BL04 + PreforPro. However, the increase was 10-fold greater than baseline levels when phages were included in the
B. lactis treatment compared to a ~4-fold increase seen with the probiotic alone. Oral administration of a lytic phage induced similar increases in
Lactobacillus in a mouse model, as well as increasing
Bifidobacterium [
24]. It is interesting to note that we did not observe increased
Bifidobacterium in either treatment group, despite daily consumption of 10
9 CFU of
B. lactis BL04. A similar finding was reported in a study of healthy volunteers taking Amoxicillin and a multi-species probiotic, which included
B. lactis,
E. faecalis and several other
Bifidobacteria and
Lactobacilli. Using culture-based methods, the investigators only reported an increase in stool
Enterococci [
25]. We used a sequencing approach, which is not sufficiently sensitive to distinguish bacteria at the species level and it is possible that the baseline levels of
Bifidobacterium across our treatment groups were sufficiently high to obscure any treatment effects. Finally, there were several taxa that have demonstrated inflammatory effects on the gut that were reduced after treatment of
B. lactis BL04 + PreforPro. Specifically, there were decreases in
Citrobacter and
Desulfovibrio. Citrobacter overgrowth is associated with bloating, and both Faber et al. [
26] and Gangi et al. [
19] reported increased levels of
Citrobacter in IBS patients. In the Faber study, symptoms of bloating and abdominal pain were alleviated after reducing
Citrobacter, and other pro-inflammatory bacteria and yeasts, by antibiotic treatment and supplementation with probiotics [
26].
Desulfovibrio is a sulfate-reducing bacteria that is also often associated with IBS [
20] and induces alterations in colonic architecture, cellular infiltration into the lamina propria, and increased inflammatory immune responses in mouse models of colitis [
27]. Therefore, the reduction of these bacteria may have been responsible for the symptom reduction reported in our participant population and suggest that future trials in a population with IBS or colitis may be warranted.
The study has some strengths and limitations worth considering. One major strength is that this study is a randomized, double-blind, placebo-controlled clinical trial. Randomized controlled trials are the gold standard for study design since they eliminate much inherent bias that other study designs introduce. Similarly, compliance across treatments was high at about 95% with no significant dietary differences in total calories, macronutrients, fiber, cholesterol, or saturated fat within any of the treatment groups. However, we recognize that there are several limitations to this study as well. First, although we saw pre- to post-treatment improvements with treatment in several of the GI parameters, there were no significant differences in score change across groups and the effect size, while significant, was small. This may be due to the fact that the study was powered from previously collected data [
10] to detect differences in the detection of
Bifidobacterium populations among groups but was not specifically powered for the GI outcomes. Therefore, the study may not have been sufficiently powered to detect these differences between treatment groups. Another limitation was that true baseline data are lacking for stool consistency measurements, as these were recorded concurrently with the start of the study protocol. To minimize the impact of treatment effects on stool consistency measures, we analyzed Day 1 and 2 as a baseline in comparison to Week 4 in addition to comparing data from Week 1 to Week 4. Another limitation is that participants had the option of numerically assessing their stool using the Bristol Stool Chart or provide a designation of hard (H), soft (S), or diarrhea (D). This mixture in data capture methods prevented the quantitative assessment of stool changes, although it was still useful for functional assessment. Finally, the high participant burden of recording every bowel movement for a month could have led to participants not recording all of their stools or recording only their first bowel movement of the day.
Another limitation of the study was our ability to detect the phages in stool samples post-consumption. Due to blinding during the study, all collected samples were plated for phage enumeration. While no baseline samples, nor any in the probiotic alone and placebo group samples resulted in plaque detection, only about 40% of the samples from the group receiving the bacteriophage cocktail resulted in plaque formation after incubation with
E. coli. There are several possibilities for the low detection of phages in these samples, including both technical challenges and individual participant factors. First, in order to reduce variability due to plating and enumeration techniques, samples were frozen and then batch processed at the end of the study, which may have resulted in a loss of viability, particularly in samples stored for longer periods. In addition, stool samples are heterogenous and few studies have looked at the distribution of phages within stool, so it is possible that our subsample-based approach was insufficient to detect low populations of phages in the stool. Finally, a study looking at the survival of phages in a simulated digestive tract found that meal composition, pH and other factors affected phage survival and detection [
28]. Therefore, it is likely that the timing of capsule consumption, the pH of the individual’s GI tract, and the meal composition prior to sample collection all may have played a role in the survival of phages in the stool. This raises some interesting possibilities for future research in terms of looking at the timing and delivery of phages, as well as interindividual variability in efficacy and stool pH to determine the optimal therapeutic conditions for phage supplementation.
In conclusion, the present study demonstrates that oral supplementation of B. lactis BL04 over the course of 4 weeks has the potential to improve stool consistency, and B. lactis BL04 with PreforPro can significantly improve some GI symptoms. In addition, phage consumption did not significantly disrupt the gut microbial community but was associated with an increased relative abundance of some beneficial species, like Lactobacillus, and decreases in certain pro-inflammatory taxa. While we did not observe significant improvements in all aspects of GI well-being or stool consistency, the findings of this study indicate a low risk for oral supplementation with phages and suggest that probiotic taken with a phage cocktail may offer a safe solution for the management of occasional GI symptoms in healthy adults.