Effects of Habitual Caffeine Intake, Physical Activity Levels, and Sedentary Behavior on the Inflammatory Status in a Healthy Population
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design and Participants
2.2. Laboratory Visit
2.3. Anthropometrical Measurements
2.4. Questionnaires
2.5. Measurement of Inflammatory Markers
2.6. Statistical Analysis
3. Results
3.1. General Characteristics of Participants in the Study
3.2. Sources of Caffeine Consumption
3.3. Concentration of Inflammatory Markers
3.4. Bivariate Correlations between Dependent and Independent Variables
3.5. Multivariable Linear Regression Analysis
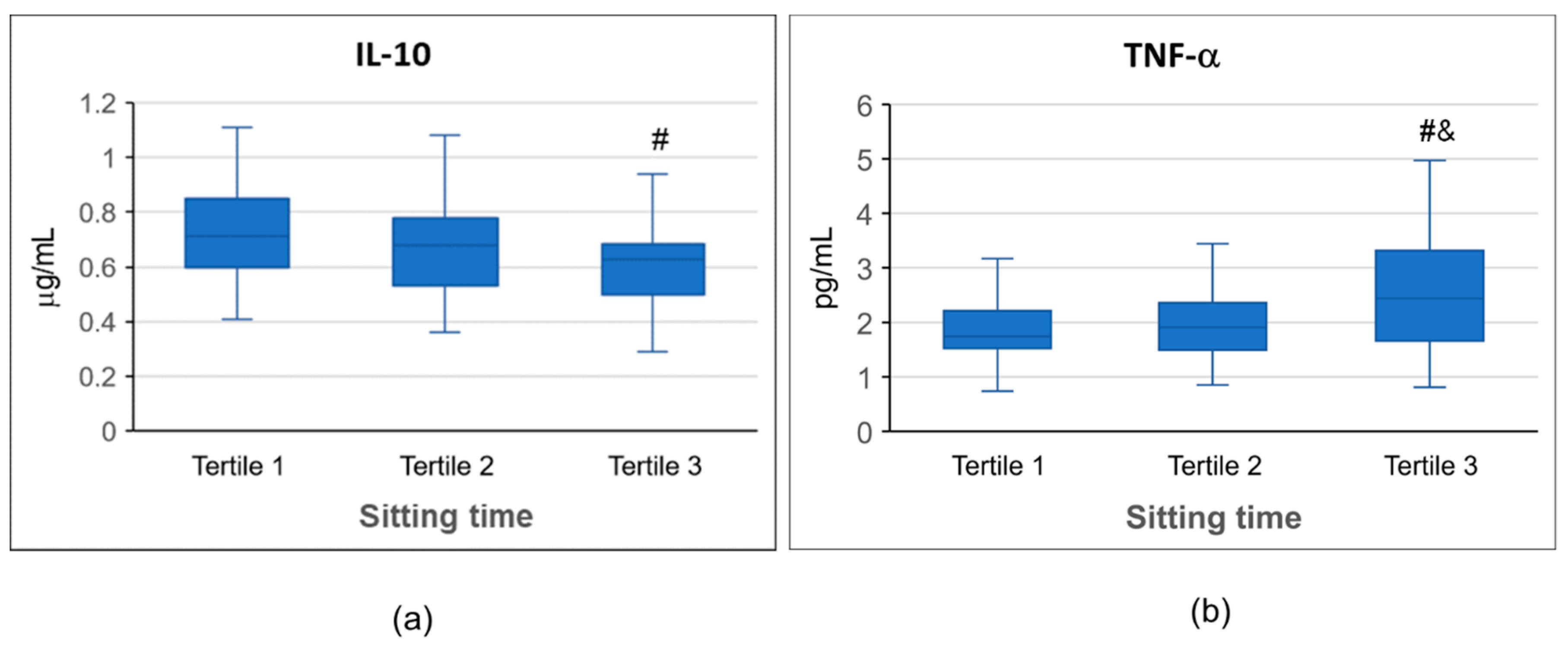
3.6. Effects of Sitting Time on IL-10 and Tumor Necrosis Factor (TNF) -a Concentrations
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ross, R. Atherosclerosis—An inflammatory disease. N. Engl. J. Med. 1999, 340, 115–126. [Google Scholar] [CrossRef] [PubMed]
- Gleeson, M.; Bishop, N.C.; Stensel, D.J.; Lindley, M.R.; Mastana, S.S.; Nimmo, M.A. The anti-inflammatory effects of exercise: Mechanisms and implications for the prevention and treatment of disease. Nat. Rev. Immunol. 2011, 11, 607–615. [Google Scholar] [CrossRef] [PubMed]
- Ouchi, N.; Parker, J.L.; Lugus, J.J.; Walsh, K. Adipokines in inflammation and metabolic disease. Nat. Rev. Immunol. 2011, 11, 85–97. [Google Scholar] [CrossRef] [PubMed]
- Petersen, A.M.W.; Pedersen, B.K. The anti-inflammatory effect of exercise. J. Appl. Physiol. 2005, 98, 1154–1162. [Google Scholar] [CrossRef] [PubMed]
- Pedersen, B.K. Health Benefits Related to Chronic Low-Grade Exercise in Patients with Systemic Inflammation. Am. J. Lifestyle Med. 2007, 1, 289–298. [Google Scholar] [CrossRef]
- Allison, M.A.; Jensky, N.E.; Marshall, S.J.; Bertoni, A.G.; Cushman, M. Sedentary behavior and adiposity-associated inflammation: The multi-ethnic study of atherosclerosis. Am. J. Prev. Med. 2012, 42, 8–13. [Google Scholar] [CrossRef]
- Henson, J.; Yates, T.; Edwardson, C.L.; Khunti, K.; Talbot, D.; Gray, L.J.; Leigh, T.M.; Carter, P.; Davies, M.J. Sedentary time and markers of chronic low-grade inflammation in a high risk population. PLoS ONE 2013, 8, 4–9. [Google Scholar] [CrossRef]
- Healy, G.N.; Matthews, C.E.; Dunstan, D.W.; Winkler, E.A.H.; Owen, N. Sedentary time and cardio-metabolic biomarkers in US adults: NHANES 200306. Eur. Heart J. 2011, 32, 590–597. [Google Scholar] [CrossRef]
- Yates, T.; Khunti, K.; Wilmot, E.G.; Brady, E.; Webb, D.; Srinivasan, B.; Henson, J.; Talbot, D.; Davies, M.J. Self-reported sitting time and markers of inflammation, insulin resistance, and adiposity. Am. J. Prev. Med. 2012, 42, 1–7. [Google Scholar] [CrossRef]
- Horrigan, L.A.; Kelly, J.P.; Connor, T.J. Caffeine suppresses TNF-alpha production via activation of the cyclic AMP/protein kinase A pathway. Int. Immunopharmacol. 2004, 4, 1409–1417. [Google Scholar] [CrossRef]
- Paiva, C.L.R.S.; Beserra, B.T.S.; Reis, C.E.G.; Dorea, J.G.; Da Costa, T.H.M.; Amato, A.A. Consumption of coffee or caffeine and serum concentration of inflammatory markers: A systematic review. Crit. Rev. Food Sci. Nutr. 2019, 59, 652–663. [Google Scholar] [CrossRef] [PubMed]
- Kempf, K.; Herder, C.; Erlund, I.; Kolb, H.; Martin, S.; Carstensen, M.; Koenig, W.; Sundvall, J.; Bidel, S.; Kuha, S.; et al. Effects of coffee consumption on subclinical inflammation and other risk factors for type 2 diabetes: A clinical trial. Am. J. Clin. Nutr. 2010, 91, 950–957. [Google Scholar] [CrossRef] [PubMed]
- Wedick, N.M.; Brennan, A.M.; Sun, Q.; Hu, F.B.; Mantzoros, C.S.; Van Dam, R.M. Effects of caffeinated and decaffeinated coffee on biological risk factors for type 2 diabetes: A randomized controlled trial. Nutr. J. 2011, 10, 93. [Google Scholar] [CrossRef] [PubMed]
- Naveed, M.; Hejazi, V.; Abbas, M.; Kamboh, A.A.; Khan, G.J.; Shumzaid, M.; Ahmad, F.; Babazadeh, D.; FangFang, X.; Modarresi-Ghazani, F.; et al. Chlorogenic acid (CGA): A pharmacological review and call for further research. Biomed. Pharmacother. 2018, 97, 67–74. [Google Scholar] [CrossRef]
- Gómez-Ruiz, J.Á.; Leake, D.S.; Ames, J.M. In vitro antioxidant activity of coffee compounds and their metabolites. J. Agric. Food Chem. 2007, 55, 6962–6969. [Google Scholar] [CrossRef]
- EFSA NDA Panel (EFSA Panel on Dietetic Products, Nutrition and Allergies). Scientific Opinion on the safety of caffeine. EFSA J. 2015, 13, 4102. [Google Scholar]
- Akash, M.S.H.; Rehman, K.; Chen, S. Effects of coffee on type 2 diabetes mellitus. Nutrition 2014, 30, 755–763. [Google Scholar] [CrossRef]
- Ding, M.; Bhupathiraju, S.N.; Chen, M.; van Dam, R.M.; Hu, F.B. Caffeinated and decaffeinated coffee consumption and risk of type 2 diabetes: A systematic review and a dose-response meta-analysis. Diabetes Care 2014, 37, 569–586. [Google Scholar] [CrossRef]
- Shang, F.; Li, X.; Jiang, X. Coffee consumption and risk of the metabolic syndrome: A meta-analysis. Diabetes Metab. 2016, 42, 80–87. [Google Scholar] [CrossRef]
- Williams, C.J.; Fargnoli, J.L.; Hwang, J.J.; Van Dam, R.M.; Blackburn, G.L.; Hu, F.B.; Mantzoros, C.S. Coffee consumption is associated with higher plasma adiponectin concentrations in women with or without type 2 diabetes a prospective cohort study. Diabetes Care 2008, 31, 1434–1437. [Google Scholar] [CrossRef]
- Kotani, K.; Tsuzaki, K.; Sano, Y.; Maekawa, M.; Fujiwara, S.; Hamada, T.; Sakane, N. The relationship between usual coffee consumption and serum C-reactive protein level in a Japanese female population. Clin. Chem. Lab. Med. 2008, 46, 1434–1437. [Google Scholar] [CrossRef] [PubMed]
- Lopez-Garcia, E.; Van Dam, R.M.; Qi, L.; Hu, F.B. Coffee consumption and markers of inflammation and endothelial dysfunction in healthy and diabetic women. Am. J. Clin. Nutr. 2006, 84, 888–893. [Google Scholar] [CrossRef] [PubMed]
- Maki, T.; Pham, N.M.; Yoshida, D.; Yin, G.; Ohnaka, K.; Takayanagi, R.; Kono, S. The relationship of coffee and green tea consumption with high-sensitivity C-reactive protein in Japanese men and women. Clin. Chem. Lab. Med. 2010, 48, 849–854. [Google Scholar] [CrossRef] [PubMed]
- Martínez-González, M.A.; Fernández-Jarne, E.; Serrano-Martínez, M.; Wright, M.; Gomez-Gracia, E. Development of a short dietary intake questionnaire for the quantitative estimation of adherence to a cardioprotective Mediterranean diet. Eur. J. Clin. Nutr. 2004, 58, 1550–1552. [Google Scholar] [CrossRef] [PubMed]
- Tauler, P.; Martinez, S.; Moreno, C.; Monjo, M.; Martinez, P.; Aguilo, A. Effects of caffeine on the inflammatory response induced by a 15-km run competition. Med. Sci. Sports Exerc. 2013, 45, 1269–1276. [Google Scholar] [CrossRef] [PubMed]
- Tauler, P.; Martinez, S.; Martinez, P.; Lozano, L.; Moreno, C.; Aguiló, A. Effects of caffeine supplementation on plasma and blood mononuclear cell IL-10 levels after exercise. Int. J. Sport Nutr. Exerc. Metab. 2016, 26, 8–16. [Google Scholar] [CrossRef]
- Renouf, M.; Marmet, C.; Giuffrida, F.; Lepage, M.; Barron, D.; Beaumont, M.; Williamson, G.; Dionisi, F. Dose-response plasma appearance of coffee chlorogenic and phenolic acids in adults. Mol. Nutr. Food Res. 2014, 58, 301–309. [Google Scholar] [CrossRef]
- Fitt, E.; Pell, D.; Cole, D. Assessing caffeine intake in the United Kingdom diet. Food Chem. 2013, 140, 421–426. [Google Scholar] [CrossRef]
- Rochat, C.; Eap, C.B.; Bochud, M.; Chatelan, A. Caffeine consumption in switzerland: Results from the first national nutrition survey menuCH. Nutrients 2020, 12, 28. [Google Scholar] [CrossRef]
- Sousa, A.G.; da Costa, T.H.M. Usual coffee intake in Brazil: Results from the National Dietary Survey 2008–9. Br. J. Nutr. 2015, 113, 1615–1620. [Google Scholar] [CrossRef]
- Gressner, O.A.; Lahme, B.; Rehbein, K.; Siluschek, M.; Weiskirchen, R.; Gressner, A.M. Pharmacological application of caffeine inhibits TGF-β-stimulated connective tissue growth factor expression in hepatocytes via PPARγ and SMAD2/3-dependent pathways. J. Hepatol. 2008, 49, 758–767. [Google Scholar] [CrossRef] [PubMed]
- Rebello, S.A.; Chen, C.H.; Naidoo, N.; Xu, W.; Lee, J.; Chia, K.S.; Tai, E.S.; Van Dam, R.M. Coffee and tea consumption in relation to inflammation and basal glucose metabolism in a multi-ethnic Asian population: A cross-sectional study. Nutr. J. 2011, 10, 61. [Google Scholar] [CrossRef] [PubMed]
- Pearson, T.A.; Mensah, G.A.; Alexander, R.W.; Anderson, J.L.; Cannon, R.O.; Criqui, M.; Fadl, Y.Y.; Fortmann, S.P.; Hong, Y.; Myers, G.L.; et al. Markers of inflammation and cardiovascular disease: Application to clinical and public health practice: A statement for healthcare professionals from the centers for disease control and prevention and the American Heart Association. Circulation 2003, 107, 499–511. [Google Scholar] [CrossRef] [PubMed]
- Moua, E.D.; Hu, C.; Day, N.; Hord, N.G.; Takata, Y. Coffee consumption and c-reactive protein levels: A systematic review and meta-analysis. Nutrients 2020, 12, 1349. [Google Scholar] [CrossRef]
- Izadi, V.; Larijani, B.; Azadbakht1, L. Is coffee and green tea consumption related to serum levels of adiponectin and leptin? Int. J. Prev. Med. 2018, 9, 106. [Google Scholar] [CrossRef]
- Visser, M.; Bouter, L.M.; McQuillan, G.M.; Wener, M.H.; Harris, T.B. Elevated C-reactive protein levels in overweight and obese adults. J. Am. Med. Assoc. 1999, 282, 2131–2135. [Google Scholar] [CrossRef]
- Pou, K.M.; Massaro, J.M.; Hoffmann, U.; Vasan, R.S.; Maurovich-Horvat, P.; Larson, M.G.; Keaney, J.F.; Meigs, J.B.; Lipinska, I.; Kathiresan, S.; et al. Visceral and subcutaneous adipose tissue volumes are cross-sectionally related to markers of inflammation and oxidative stress: The Framingham Heart Study. Circulation 2007, 116, 1234–1241. [Google Scholar] [CrossRef]
- Scherer, P.E.; Williams, S.; Fogliano, M.; Baldini, G.; Lodish, H.F. A novel serum protein similar to C1q, produced exclusively in adipocytes. J. Biol. Chem. 1995, 270, 26746–26749. [Google Scholar] [CrossRef]
- Kern, P.A.; Di Gregorio, G.B.; Lu, T.; Rassouli, N.; Ranganathan, G. Adiponectin expression from human adipose tissue: Relation to obesity, insulin resistance, and tumor necrosis factor-α expression. Diabetes 2003, 52, 1779–1785. [Google Scholar] [CrossRef]
- Thorand, B.; Baumert, J.; Döring, A.; Herder, C.; Kolb, H.; Rathmann, W.; Giani, G.; Koenig, W.; Wichmann, H.E.; Löwel, H.; et al. Sex differences in the relation of body composition to markers of inflammation. Atherosclerosis 2006, 184, 216–224. [Google Scholar] [CrossRef]
- Panagiotakos, D.B.; Pitsavos, C.; Yannakoulia, M.; Chrysohoou, C.; Stefanadis, C. The implication of obesity and central fat on markers of chronic inflammation: The ATTICA study. Atherosclerosis 2005, 183, 308–315. [Google Scholar] [CrossRef] [PubMed]
- Park, H.S.; Park, J.Y.; Yu, R. Relationship of obesity and visceral adiposity with serum concentrations of CRP, TNF-α and IL-6. Diabetes Res. Clin. Pract. 2005, 69, 29–35. [Google Scholar] [CrossRef] [PubMed]
- Rexrode, K.M.; Pradhan, A.; Manson, J.E.; Buring, J.E.; Ridker, P.M. Relationship of total and abdominal adiposity with CRP and IL-6 in women. Ann. Epidemiol. 2003, 13, 674–682. [Google Scholar] [CrossRef]
- Day, K.; Kwok, A.; Evans, A.; Mata, F.; Verdejo-garcia, A.; Hart, K.; Ward, L.C.; Truby, H. Comparison of a Bioelectrical Impedance Device against the Reference Method Dual Energy X-Ray Absorptiometry and Anthropometry for the Evaluation of Body Composition in Adults. Nutrients 2018, 10, 1469. [Google Scholar] [CrossRef] [PubMed]
| All (n = 244) | Men (n = 112) | Women (n = 132) | p Value | |
|---|---|---|---|---|
| Age (years) | 32.1 ± 10.8 | 33.4 ± 10.8 | 31.1 ± 10.8 | 0.098 |
| Weight (kg) | 66.9 ± 13.3 | 76.3 ± 11.4 | 58.9 ± 8.7 | <0.001 * |
| Height (cm) | 170.0 ± 9.0 | 177.0 ± 6.5 | 164.0 ± 6.0 | <0.001 * |
| BMI (kg·m−2) | 23.0 ± 3.4 | 24.3 ± 3.3 | 22.0 ± 3.2 | <0.001 * |
| Fat mass (%) | 23.3 ± 7.9 | 18.3 ± 6.3 | 27.6 ± 6.5 | <0.001 * |
| Visceral fat rating | 4.01 ± 3.30 | 5.66 ± 3.68 | 2.63 ± 2.12 | <0.001 * |
| Mediterranean diet score | 8.18 ± 1.91 | 8.22 ± 1.93 | 8.15 ± 1.91 | 0.771 |
| Caffeine intake (mg·day−1) | 164.3 ± 143.4 | 174.7 ± 152.4 | 155.5 ± 135.3 | 0.298 |
| Caffeine intake (mg·kg body weight−1·day−1) | 2.48 ± 2.18 | 2.33 ± 2.18 | 2.60 ± 2.18 | 0.346 |
| Coffee intake (servings·day−1) | 1.32 ± 1.35 | 1.41 ± 1.47 | 1.24 ± 1.25 | 0.311 |
| Physical activity levels (METs-hour·week−1) | 43.8 ± 36.1 | 48.7 ± 33.7 | 39.7 ± 37.5 | 0.054 |
| Physical activity (hours·week−1) | 9.18 ± 8.50 | 9.90 ± 7.36 | 8.57 ± 9.35 | 0.224 |
| Sitting time (hours·day−1) | 6.96 ± 2.77 | 7.20 ± 3.09 | 6.77 ± 2.46 | 0.235 |
| Source | All (n = 228) | Men (n = 105) | Women (n = 123) | p Value |
|---|---|---|---|---|
| Coffee (%) | 67.5 ± 37.1 | 69.6 ± 37.4 | 66.0 ± 36.9 | 0.538 |
| Tea (%) | 10.1 ± 19.8 | 8.9 ± 19.4 | 11.0 ± 20.1 | 0.425 |
| Cola drinks (%) | 12.8 ± 24.7 | 14.0 ± 26.8 | 11.8 ± 22.9 | 0.506 |
| Chocolate (%) | 7.9 ± 20.4 | 5.2 ± 14.1 | 10.2 ± 24.2 | 0.054 |
| Energetic drinks (%) | 1.6 ± 7.6 | 2.2 ± 9.0 | 1.0 ± 6.1 | 0.241 |
| Sport products (%) | 0.1 ± 0.7 | 0.1 ± 1.0 | n.d. | 0.175 |
| Inflammatory Marker | All (n = 244) | Men (n = 112) | Women (n = 132) | p Value |
|---|---|---|---|---|
| IL-10 (μg·mL−1) | 0.65 (0.54, 0.77) | 0.65 (0.55, 0.83) | 0.66 (0.54, 0.74) | 0.333 |
| IL-6 (μg·mL−1) | 2.17 (1.44, 3.04) | 2.38 (1.84, 3.12) | 1.89 (1.14, 2.99) | 0.003 * |
| IL-1β (pg·mL−1) | 3.84 (2.66, 6.29) | 4.02 (2.88, 6.40) | 3.58 (2.43, 6.00) | 0.205 |
| TNF-α (pg·mL−1) | 1.98 (1.52, 2.68) | 2.11 (1.64, 3.04) | 1.76 (1.49, 2.51) | 0.005 * |
| CRP (μg·mL−1) | 3.50 (1.81, 5.67) | 2.46 (1.58, 4.28) | 4.26 (2.42, 6.34) | <0.001 * |
| Adiponectin (μg·mL−1) | 5.78 (3.62, 7.96) | 4.59 (3.12, 6.22) | 6.72 (4.77, 8,78) | <0.001 * |
| Age | %Fat | Vis Fat | MD | Caffeine | PA | Sit | |
|---|---|---|---|---|---|---|---|
| IL-10 | −0.138 (0.032 *) | −0.219 (0.001 *) | −0.159 (0.013 *) | −0.083 (0.197) | −0.094 (0.142) | 0.195 (0.002 *) | −0.325 (<0.001*) |
| IL-6 | 0.205 (0.001 *) | 0.063 (0.329) | 0.290 (<0.001 *) | −0.032 (0.615) | 0.089 (0.164) | −0.037 (0.561) | 0.003 (0.957) |
| IL-1β | 0.006 (0.932) | 0.071 (0.267) | 0.052 (0.417) | 0.132 (0.039 *) | −0.006 (0.930) | −0.009 (0.889) | −0.030 (0.639) |
| TNF-α | 0.157 (0.014 *) | 0.086 (0.180) | 0.335 (<0.001 *) | −0.023 (0.717) | 0.067 (0.299) | −0.081 (0.208) | 0.249 (<0.001 *) |
| CRP | 0.009 (0.889) | 0.427 (<0.001 *) | 0.115 (0.074) | −0.109 (0.088) | −0.129 (0.044 *) | −0.037 (0.563) | −0.039 (0.549) |
| Adiponectin | −0.107 (0.095) | −0.222 (<0.001 *) | −0.117 (0.072) | −0.012 (0.429) | −0.087 (0.182) | −0.041 (0.520) | −0.003 (0.963) |
| Age | %Fat | Vis Fat | MD | Caffeine | PA | Sit | |
|---|---|---|---|---|---|---|---|
| IL-10 | −0.203 (0.020 *) | −0.133 (0.128) | −0.170 (0.052) | −0.045 (0.605) | −0.098 (0.265) | 0.185 (0.033 *) | −0.281 (0.001 *) |
| IL-6 | 0.198 (0.023 *) | 0.162 (0.064) | 0.182 (0.037 *) | −0.046 (0.597) | 0.054 (0.539) | −0.100 (0.254) | 0.017 (0.844) |
| IL-1β | −0.005 (0.959) | −0.011 (0.902) | 0.048 (0.588) | 0.157 (0.072) | −0.016 (0.854) | −0.028 (0.754) | −0.091 (0.300) |
| TNF-α | 0.096 (0.274) | 0.164 (0.060) | 0.172 (0.049 *) | −0.062 (0.479) | −0.034 (0.699) | −0.016 (0.856) | 0.297 (0.001 *) |
| CRP | −0.066 (0.455) | 0.330 (<0.001 *) | 0.158 (0.070) | −0.097 (0.267) | −0.155 (0.077) | 0.030 (0.729) | 0.048 (0.582) |
| Adiponectin | −0.111 (0.243) | 0.063 (0.510) | −0.053 (0.579) | 0.152 (0.109) | −0.027 (0.773) | −0.106 (0.268) | −0.062 (0.513) |
| Age | %Fat | Vis Fat | MD | Caffeine | PA | Sit | |
|---|---|---|---|---|---|---|---|
| IL-10 | −0.100 (0.296) | −0.295 (0.002 *) | −0.249 (0.008 *) | −0.120 (0.207) | −0.102 (0.286) | 0.242 (0.010 *) | −0.369 (<0.001 *) |
| IL-6 | 0.180 (0.058) | 0.300 (0.001 *) | 0.293 (0.002 *) | −0.024 (0.803) | 0.130 (0.164) | −0.023 (0.806) | −0.042 (0.662) |
| IL-1β | −0.002 (0.980) | −0.054 (0.577) | −0.011 (0.913) | 0.094 (0.323) | −0.006 (0.953) | 0.004 (0.953) | 0.026 (0.784) |
| TNF-α | 0.193 (0.042 *) | 0.353 (<0.001 *) | 0.378 (<0.001 *) | 0.115 (0.226) | 0.148 (0.118) | −0.262 (0.005 *) | 0.188 (0.047 *) |
| CRP | 0.166 (0.079) | 0.366 (<0.001 *) | 0.381 (<0.001 *) | −0.122 (0.200) | −0.074 (0.440) | −0.051 (0.594) | 0.076 (0.424) |
| Adiponectin | −0.107 (0.095) | −0.222 (<0.001 *) | −0.117 (0.072) | −0.012 (0.429) | −0.087 (0.182) | −0.041 (0.520) | −0.003 (0.963) |
| Variable | B | 95% CI | β | t | p Value |
|---|---|---|---|---|---|
| Age | −0.002 | (−0.006, 0.002) | −0.104 | −1.706 | 0.089 |
| Sex | 0.010 | (−0.060, 0.080) | 0.042 | 0568 | 0.570 |
| Fat mass | −0.003 | (−0.005, −0.001) | −0.189 | −3.171 | 0.002 * |
| Visceral fat rating | −0.002 | (−0.014, 0.010) | −0.066 | −1.346 | 0.180 |
| Adherence to MD | −0.005 | (−0.013, 0.003) | −0.078 | −1.324 | 0.187 |
| Caffeine intake | 0.000 | (−0.001, 0.000) | −0.080 | −1.336 | 0.183 |
| Physical activity | 0.000 | (0.000, 0.001) | 0.136 | 2.218 | 0.028 * |
| Sitting time | −0.012 | (−0.017, −0.007) | −0.281 | −4.607 | <0.001 * |
| Variable | B | 95% CI | β | t | p Value |
|---|---|---|---|---|---|
| Age | 0.000 | (−0.004, 0.004) | −0.019 | −0.285 | 0.776 |
| Sex | 0.194 | (0.124−0.265) | 0.407 | 5.426 | <0.001 * |
| Fat mass | −0.005 | (−0.009, −0.001) | −0.162 | −2.156 | 0.032 * |
| Visceral fat rating | 0.004 | (−0.004−0.012) | 0.050 | 0.443 | 0.658 |
| Adherence to MD | 0.008 | (−0.006, 0.022) | 0.065 | 1.075 | 0.283 |
| Caffeine intake | 0.000 | (−0.001, 0.000) | −0.068 | −1.092 | 0.276 |
| Physical activity | 0.000 | (−0.001, 0.000) | −0.012 | 0.010 | 0.846 |
| Sitting time | 0.003 | (−0.007, 0.013) | 0.039 | 0.637 | 0.585 |
| Variable | B | 95% CI | β | t | p Value |
|---|---|---|---|---|---|
| Age | 0.001 | (−0.003, 0.005) | 0.013 | 0.151 | 0.880 |
| Sex | −0.035 | (−0.075, 0.005) | −0.073 | −1.055 | 0.292 |
| Fat mass | −0.001 | (−0.003, 0.001) | −0.028 | −0.439 | 0.661 |
| Visceral fat rating | 0.021 | (0.012, 0.030) | 0.290 | 4.701 | <0.001 * |
| Adherence to MD | −0.005 | (−0.017, 0.007) | −0.044 | −0.706 | 0.481 |
| Caffeine intake | 0.000 | (0.000, 0.001) | 0.039 | 0.579 | 0.563 |
| Physical activity | −0.001 | (−0.002, 0.001) | −0.059 | −0.948 | 0.344 |
| Sitting time | −0.003 | (−0.011, 0.005) | −0.033 | −0.532 | 0.595 |
| Variable | B | 95% CI | β | t | p Value |
|---|---|---|---|---|---|
| Age | −0.002 | (−0.005, 0.001) | −0.108 | −1.327 | 0.186 |
| Sex | −0.014 | (−0.128, 0.100) | −0.041 | −0.608 | 0.544 |
| Fat mass | −0.001 | (−0.011, 0.010) | −0.023 | −0.367 | 0.714 |
| Visceral fat rating | 0.016 | (0.010, 0.022) | 0.309 | 5.166 | <0.001 * |
| Adherence to MD | 0.001 | (−0.009, 0.012) | 0.007 | 0.119 | 0.905 |
| Caffeine intake | 0.000 | (−0.001, 0.000) | −0.060 | −0.936 | 0.350 |
| Physical activity | 0.000 | (−0.001, 0.000) | −0.025 | −0.984 | 0.326 |
| Sitting time | 0.013 | (0.004, 0.019) | 0.211 | 3.538 | <0.001 * |
| Variable | B | 95% CI | β | t | p Value |
|---|---|---|---|---|---|
| Age | −0.002 | (−0.010, 0.006) | −0.039 | −0.601 | 0.549 |
| Sex | −0.016 | (−0.082, 0.050) | −0.021 | −0.288 | 0.774 |
| Fat mass | 0.018 | (0.013, 0.022) | 0.454 | 7.885 | <0.001 * |
| Visceral fat rating | 0.006 | (−0.022, 0.034) | 0.063 | 0.980 | 0.328 |
| Adherence to MD | −0.011 | (−0.029, 0.007) | −0.070 | −1.219 | 0.224 |
| Caffeine intake | −0.002 | (−0.003, −0.001) | −0.195 | −3.379 | 0.001 * |
| Physical activity | 0.000 | (−0.004, 0.004) | 0.023 | 0.407 | 0.684 |
| Sitting time | 0.000 | (−0.013, 0.013) | 0.011 | 0.187 | 0.852 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rodas, L.; Riera-Sampol, A.; Aguilo, A.; Martínez, S.; Tauler, P. Effects of Habitual Caffeine Intake, Physical Activity Levels, and Sedentary Behavior on the Inflammatory Status in a Healthy Population. Nutrients 2020, 12, 2325. https://doi.org/10.3390/nu12082325
Rodas L, Riera-Sampol A, Aguilo A, Martínez S, Tauler P. Effects of Habitual Caffeine Intake, Physical Activity Levels, and Sedentary Behavior on the Inflammatory Status in a Healthy Population. Nutrients. 2020; 12(8):2325. https://doi.org/10.3390/nu12082325
Chicago/Turabian StyleRodas, Lluis, Aina Riera-Sampol, Antoni Aguilo, Sonia Martínez, and Pedro Tauler. 2020. "Effects of Habitual Caffeine Intake, Physical Activity Levels, and Sedentary Behavior on the Inflammatory Status in a Healthy Population" Nutrients 12, no. 8: 2325. https://doi.org/10.3390/nu12082325
APA StyleRodas, L., Riera-Sampol, A., Aguilo, A., Martínez, S., & Tauler, P. (2020). Effects of Habitual Caffeine Intake, Physical Activity Levels, and Sedentary Behavior on the Inflammatory Status in a Healthy Population. Nutrients, 12(8), 2325. https://doi.org/10.3390/nu12082325






