The Effects and Potential Mechanism of Oil Palm Phenolics in Cardiovascular Health: A Review on Current Evidence
Abstract
1. Introduction
2. The Effects of OPP in Cardiovascular Health
3. Mechanism of Actions of OPP in Cardiovascular Health
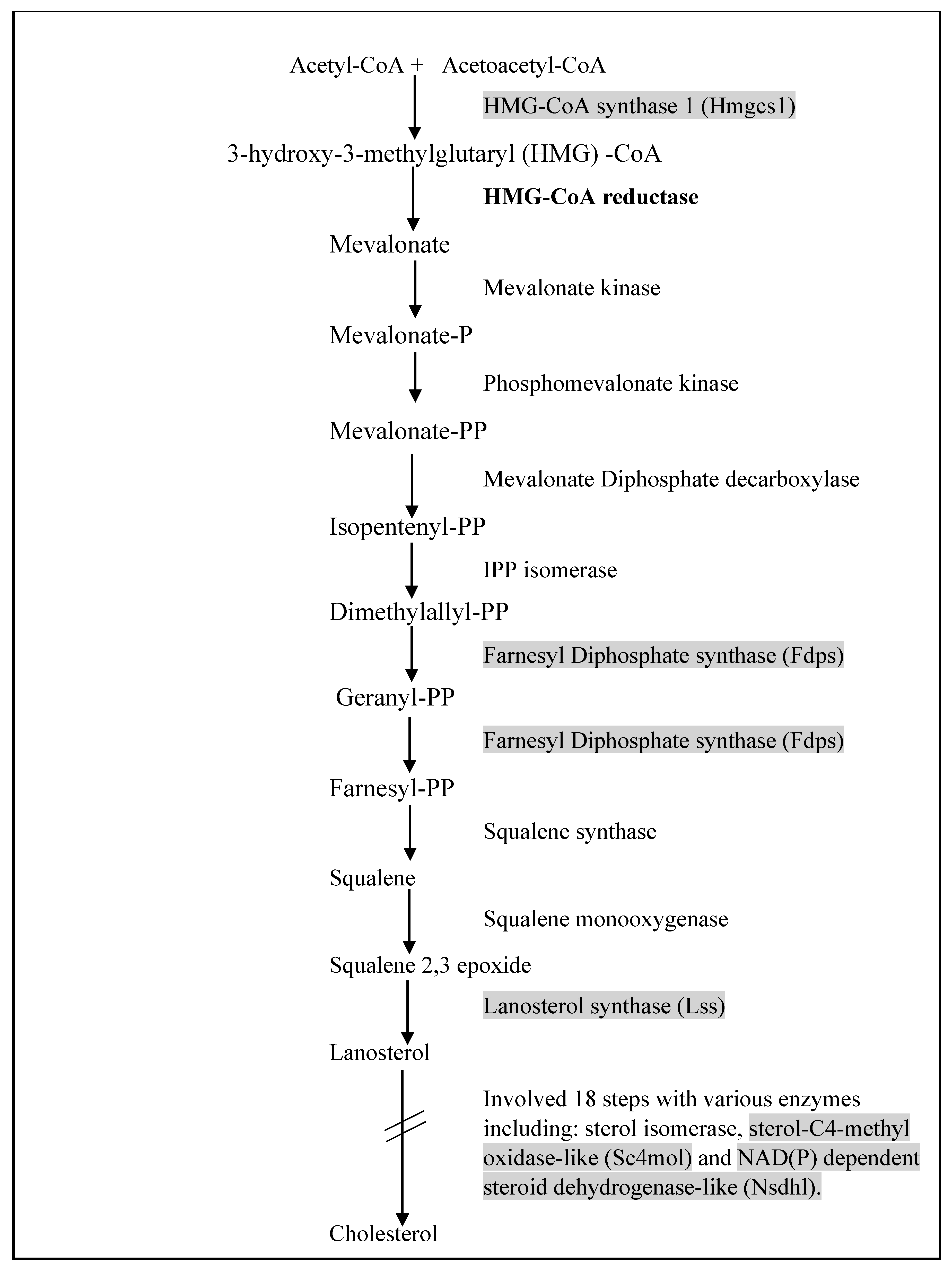
3.1. Cholesterol Biosynthesis
3.2. Antioxidant Property
3.3. Anti-Inflammatory Effects
3.4. Other Mechanisms: Fatty Acid Beta Oxidation, Tricarboxylic Acid (TCA) cycle and Electron Transport Chain (ETC)
4. Safety and Toxicity of OPP
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| Acadl | acetyl-CoA dehydrogenase |
| Acads | acyl-CoA dehydrogenase |
| Ar | androgen receptor |
| Cxcl12 | chemokine (C-X-C motif) ligand 12 |
| Fdps | farnesyl diphosphate synthase |
| Gjb1 | gap junction membrane channel protein beta 1 |
| Gstm2, Gstm5, Gstm6 | glutathione S transferases |
| Gpx1 | glutathione peroxidase 1 |
| Hadhb, Hadhsc | hydroxyacyl-CoA dehydrogenases |
| Hmgcs1 | 3-hydroxy-3-methylglutaryl-CoA synthase 1 |
| Lss | lanosterol synthase |
| Mgst1 | microsomal glutathione S-transferase 1 |
| Nsdhl | NAD(P) dependent steroid dehydrogenase-like |
| Sc4mol | sterol-C4-methyl oxidase-like |
References
- World Health Organization. Cardiovascular Diseases (CVDs). 2017. Available online: https://www.who.int/news-room/fact-sheets/detail/cardiovascular-diseases-(cvds) (accessed on 22 July 2019).
- World Health Organization. Cardiovascular Diseases (CVDs). 2019. Available online: https://www.who.int/health-topics/cardiovascular-diseases/ (accessed on 22 July 2019).
- Nelson, R.H. Hyperlipidemia as a risk factor for cardiovascular disease. Prim. Care 2013, 40, 195–211. [Google Scholar] [CrossRef] [PubMed]
- Wilson, P.W.; D’Agostino, R.B.; Levy, D.; Belanger, A.M.; Silbershatz, H.; Kannel, W.B. Prediction of coronary heart disease using risk factor categories. Circulation 1998, 97, 1837–1847. [Google Scholar] [CrossRef] [PubMed]
- Safeer, R.S.; Lacivita, C.L. Choosing drug therapy for patients with hyperlipidemia. Am. Fam. Physician. 2000, 61, 3371–3382. [Google Scholar]
- Reiner, Z.; Catapano, A.L.; de Backer, G.; Graham, I.; Taskinen, M.A.; Wiklund, O.; Agewall, S.; Alegria, E.; Chapman, M.J.; Durrington, P. ESC/EAS Guidelines for the management of dyslipidaemias. Eur. Heart J. 2011, 32, 1769–1818. [Google Scholar] [CrossRef]
- Dragos, D.; Pruteanu, D.; Constantin, R. High-Dose Statin Associated with Rhabdomyolysis, Acute Kidney Injury, Cholestatic Liver Injury, and Thrombocytopenia. SM J. Nephrol. Kidney Dis. 2017, 1, 1–7. [Google Scholar] [CrossRef]
- Sattar, N.; Preiss, D.; Murray, H.M.; Welsh, P.; Buckley, B.M.; de Craen, A.J.M.; Seshasai, S.R.K.; McMurray, J.J.; Freeman, D.J.; Jukema, J.W. Statins and risk of incident diabetes: A collaborative meta-analysis of randomised statin trials. Lancet 2010, 375, 735–742. [Google Scholar] [CrossRef]
- Preiss, D.; Seshasai, S.R.K.; Welsh, P.; Murphy, S.A.; Ho, J.E.; Waters, D.D.; DeMicco, D.A.; Cannon, C.P.; Sabatine, M.S.; Braunwald, E.; et al. Risk of incident diabetes with intensive-dose compared with moderate-dose statin therapy: A meta-analysis. JAMA 2011, 305, 2556–2564. [Google Scholar] [CrossRef]
- Sharma, M.; Ansari, M.T.; Abou-Setta, A.M.; Soares-Weiser, K.; Ooi, T.C.; Sears, M.; Yazdi, F.; Tsertsvadze, A.; Moher, D. Systematic review: Comparative effectiveness and harms of combination therapy and monotherapy for dyslipidemia. Ann. Intern Med. 2009, 151, 622–630. [Google Scholar] [CrossRef]
- Mohanty, I.R.; Borde, M.; Kumar, C.S.; Maheshwari, U. Dipeptidyl peptidase IV Inhibitory activity of Terminalia arjuna attributes to its cardioprotective effects in experimental diabetes: In silico, in vitro and in vivo analyses. Phytomedicine 2019, 57, 158–165. [Google Scholar] [CrossRef]
- Shang, X.-F.; Morris-Natschke, S.L.; Yang, G.Z.; Liu, Y.Q.; Guo, X.; Xu, X.S.; Goto, M.; Li, J.C.; Zhang, J.Y.; Lee, K.H. Biologically active quinoline and quinazoline alkaloids part II. Med. Res. Rev. 2018, 38, 1614–1660. [Google Scholar] [CrossRef]
- Latypova, G.M.; Bychenkova, M.A.; Katayev, V.A.; Perfilova, V.N.; Tyurenkov, I.N.; Mokrousov, I.S.; Prokofiev, I.I.; Salikhov, S.M.; Iksanova, G.R. Composition and cardioprotective effects of Primula veris L. solid herbal extract in experimental chronic heart failure. Phytomedicine 2019, 54, 17–26. [Google Scholar] [PubMed]
- Vincent, A.; Covinhes, A.; Barrère, C.; Gallot, L.; Thoumala, S.; Piot, C.; Heurteaux, C.; Lazdunski, M.; Nargeot, J.; Barrère-Lemaire, S. Acute and long-term cardioprotective effects of the Traditional Chinese Medicine MLC901 against myocardial ischemia-reperfusion injury in mice. Sci. Rep. 2017, 7. [Google Scholar] [CrossRef] [PubMed]
- Sundaram, R.L.; Sali, V.K.; Vasanthi, H.R. Protective effect of rutin isolated from Spermococe hispida against cobalt chloride-induced hypoxic injury in H9c2 cells by inhibiting oxidative stress and inducing apoptosis. Phytomedicine 2018, 51, 196–204. [Google Scholar] [CrossRef]
- Sun, L.; Jia, H.; Ma, L.; Yu, M.; Yang, Y.; Liu, Y.; Zhang, H.; Zou, Z. Metabolic profiling of hypoxia/reoxygenation injury in H9c2 cells reveals the accumulation of phytosphingosine and the vital role of Dan-Shen in Xin-Ke-Shu. Phytomedicine 2018, 49, 83–94. [Google Scholar] [CrossRef] [PubMed]
- Vijay, V.; Pimm, S.L.; Jenkins, C.N.; Smith, S.J. The Impacts of Oil Palm on Recent Deforestation and Biodiversity Loss. PLoS ONE 2016, 11, e0159668. [Google Scholar] [CrossRef]
- Shahbandeh, M. Production Volume of Palm Oil Worldwide From 2012/13 to 2018/19 (in Million Metric Tons). Available online: https://www.statista.com/statistics/613471/palm-oil-production-volume-worldwide/ (accessed on 22 August 2019).
- Edem, D.O. Palm oil: Biochemical, physiological, nutritional, hematological, and toxicological aspects: A review. Plant Foods Hum. Nutr. 2002, 57, 319–341. [Google Scholar] [CrossRef]
- Owoyele, B.; Owolabi, G. Traditional oil palm (Elaeis guineensis jacq.) and its medicinal uses: A review. TANG 2014, 4, 16.1–16.8. [Google Scholar]
- Basiron, Y.; Kook Weng, C. The oil palm and its sustainability. J. Oil Palm Res. 2004, 16, 1–10. [Google Scholar]
- Tan, A. By-products of palm oil extraction and refining. Oléagineuxcorps Graslipides 2006, 13, 9–11. [Google Scholar] [CrossRef][Green Version]
- Sambanthamurthi, R.; Tan, Y.A.; Sundram, K.; Abeywardena, M.; Sambandan, T.G.; Rha, C.; Sinskey, A.J.; Subramaniam, K.; Leow, S.S.; Hayes, K.C.; et al. Oil palm vegetation liquor: A new source of phenolic bioactives. Br. J. Nutr. 2011, 106, 1655–1663. [Google Scholar] [CrossRef]
- Sambanthamurthi, R.; Tan, Y.A.; Sundram, K. Treatment of Vegetation Liquors Derived from Oil-Bearing Fruit. U.S. Patent No. 7,387,802 B2, 17 June 2008. [Google Scholar]
- Sundram, K.; Sambanthamurthi, R.; Tan, Y.A. Palm fruit chemistry and nutrition. Asia Pac. J. Clin. Nutr. 2003, 12, 355–362. [Google Scholar] [PubMed]
- Ji, X.; Usman, A.; Razalli, N.H.; Sambanthamurthi, R.; Gupta, S.V. Oil palm phenolics (OPP) inhibit pancreatic cancer cell proliferation via suppression of NF-kappaB pathway. Anticancer Res. 2015, 35, 97–106. [Google Scholar] [PubMed]
- Leow, S.S.; Sekaran, S.D.; Tan, Y.A.; Sundram, K.; Sambanthamurthi, R. Oil palm phenolics confer neuroprotective effects involving cognitive and motor functions in mice. Nutr. Neurosci. 2013, 16, 207–217. [Google Scholar] [CrossRef]
- Che Idris, C.A.; Karupaiah, T.; Sundram, K.; Tan, Y.A.; Balasundram, A.; Leow, S.S.; Nasruddin, N.S.; Sambanthamurthi, R. Oil palm phenolics and vitamin E reduce atherosclerosis in rabbits. J. Funct. Foods 2014, 7, 541–550. [Google Scholar] [CrossRef]
- Katengua-Thamahane, E.; Marnewick, J.L.; Ajuwon, O.R.; Chegou, N.N.; Szűcs, G.; Ferdinandy, P.; Csont, T.; Csonka, C.; Van Rooyen, J. The combination of red palm oil and rooibos show anti-inflammatory effects in rats. J. Inflamm. 2014, 11, 41. [Google Scholar] [CrossRef]
- Auger, C.; Gérain, P.; Laurent-Bichon, F.; Portet, K.; Bornet, A.; Caporiccio, B.; Cros, G.; Teissédre, P.L.; Rouanet, J.M. Phenolics from commercialized grape extracts prevent early atherosclerotic lesions in hamsters by mechanisms other than antioxidant effect. J. Agric. Food Chem. 2004, 52, 5297–5302. [Google Scholar] [CrossRef]
- Morton, L.W.; Caccetta, R.A.M.; Puddey, I.B.; Croft, K.D. Chemistry and Biological Effects of Dietary Phenolic Compounds: Relevance to Cardiovascular Disease. Clin. Exp. Pharmacol. Physiol. 2000, 27, 152–159. [Google Scholar] [CrossRef]
- Leow, S.S.; Sekaran, S.D.; Sundram, K.; Tan, Y.A.; Sambanthamurthi, R. Differential transcriptomic profiles effected by oil palm phenolics indicate novel health outcomes. BMC Genom. 2011, 12, 432. [Google Scholar] [CrossRef]
- Abeywardena, M.; Sundram, K.; Sambanthamurthi, R.; Tan, Y.A. Oil palm phenolics as a bioactive ingredient in promoting cardiovascular health. J. Oil Palmenvironment Health 2014, 5, 38–48. [Google Scholar]
- Fairus, S.; Leow, S.S.; Mohamed, I.N.; Tan, Y.A.; Sundram, K.; Sambanthamurthi, R. A phase I single-blind clinical trial to evaluate the safety of oil palm phenolics (OPP) supplementation in healthy volunteers. Sci. Rep. 2018, 8, 8217. [Google Scholar] [CrossRef]
- Paulis, L.; Zicha, J.; Kunes, J.; Hojna, S.; Behuliak, M.; Celec, P.; Kojsova, S.; Pechanova, O.; Simko, F. Regression of L-NAME-induced hypertension: The role of nitric oxide and endothelium-derived constricting factor. Hypertens Res. 2008, 31, 793–803. [Google Scholar] [CrossRef]
- Morel, I.; Lescoat, G.; Cillard, P.; Cillard, J. Role of flavonoids and iron chelation in antioxidant action. Methods Enzym. 1994, 234, 437–443. [Google Scholar]
- Salah, N.; Miller, N.J.; Paganga, G.; Tijburg, L.; Bolwell, G.P.; Rice-Evans, C. Polyphenolic flavanols as scavengers of aqueous phase radicals and as chain-breaking antioxidants. Arch Biochem. Biophys. 1995, 322, 339–346. [Google Scholar] [CrossRef] [PubMed]
- Osakabe, N.; Yasuda, A.; Natsume, M.; Takizawa, T.; Terao, J.; Kondo, K. Catechins and their oligomers linked by C4 --> C8 bonds are major cacao polyphenols and protect low-density lipoprotein from oxidation in vitro. Exp. Biol. Med. 2002, 227, 51–56. [Google Scholar] [CrossRef] [PubMed]
- Poloni, D.M.; Dangles, O. Binding of Plant Polyphenols to Serum Albumin and LDL: Healthy Implications for Heart Disease. J. Agric. Food Chem. 2019, 67, 9139–9147. [Google Scholar] [CrossRef]
- Vinson, J.A.; Jang, J.; Yang, J.; Dabbagh, Y.; Liang, X.; Serry, M.; Proch, J.; Cai, S. Vitamins and especially flavonoids in common beverages are powerful in vitro antioxidants which enrich lower density lipoproteins and increase their oxidative resistance after ex vivo spiking in human plasma. J. Agric. Food Chem. 1999, 47, 2502–2504. [Google Scholar] [CrossRef]
- Gofman, J.W.; Lindgren, F.; Elliott, H.; Mantz, W.; Hewitt, J.; Strisower, B.; Herring, V.; Lyon, T.P. The Role of Lipids and Lipoproteins in Atherosclerosis. Available online: https://www.ncbi.nlm.nih.gov/books/NBK343489/ (accessed on 27 September 2019).
- Steinberg, D.; Parthasarathy, S.; Carew, T.E.; Khoo, J.C.; Witztum, J.L. Beyond cholesterol. Modifications of low-density lipoprotein that increase its atherogenicity. N. Engl. J. Med. 1989, 320, 915–924. [Google Scholar]
- Chong, M.F.; Macdonald, R.; Lovegrove, J.A. Fruit polyphenols and CVD risk: A review of human intervention studies. Br. J. Nutr. 2010, 104 (Suppl. 3), S28–S39. [Google Scholar] [CrossRef]
- Micallef, M.; Lexis, L.; Lewandowski, P. Red wine consumption increases antioxidant status and decreases oxidative stress in the circulation of both young and old humans. Nutr. J. 2007, 6, 27. [Google Scholar] [CrossRef]
- Duthie, S.J.; Jenkinson, A.M.; Crozier, A.; Mullen, W.; Pirie, L.; Kyle, J.; Yap, L.S.; Christen, P.; Duthie, G.G. The effects of cranberry juice consumption on antioxidant status and biomarkers relating to heart disease and cancer in healthy human volunteers. Eur. J. Nutr. 2006, 45, 113–122. [Google Scholar] [CrossRef]
- Heber, D.; Seeram, N.P.; Wyatt, H.; Henning, S.M.; Zhang, Y.; Ogden, L.G.; Dreher, M.; Hill, J.O. Safety and antioxidant activity of a pomegranate ellagitannin-enriched polyphenol dietary supplement in overweight individuals with increased waist size. J. Agric. Food Chem. 2007, 55, 10050–10054. [Google Scholar] [CrossRef] [PubMed]
- Syarifah-Noratiqah, S.-B.; Zulfarina, M.S.; Ahmad, S.U.; Fairus, S.; Naina-Mohamed, I. The Pharmacological Potential of Oil Palm Phenolics (OPP) Individual Components. Int. J. Med Sci. 2019, 16, 711–719. [Google Scholar] [CrossRef] [PubMed]
- Masella, R.; Cantafora, A.; Modesti, D.; Cardilli, A.; Gennaro, L.; Bocca, A.; Coni, E. Antioxidant activity of 3,4-DHPEA-EA and protocatechuic acid: A comparative assessment with other olive oil biophenols. Redox Rep. 1999, 4, 113–121. [Google Scholar] [CrossRef] [PubMed]
- Li, P.; Wang, X.Q.; Wang, H.Z.; Wu, Y.N. High performance liquid chromatographic determination of phenolic acids in fruits and vegetables. Biomed. Environ. Sci. 1993, 6, 389–398. [Google Scholar]
- Ali, B.H.; al Wabel, N.; Blunden, G. Phytochemical, pharmacological and toxicological aspects of Hibiscus sabdariffa L.: A review. Phytother Res. 2005, 19, 369–375. [Google Scholar] [CrossRef]
- Lin, C.Y.; Huang, C.Y.; Huang, C.Y.; Yin, M.C. Anticoagulatory, antiinflammatory, and antioxidative effects of protocatechuic acid in diabetic mice. J. Agric. Food Chem. 2009, 57, 6661–6667. [Google Scholar] [CrossRef]
- Borate, A.R.; Suralkar, A.A.; Birje, S.S.; Malusare, P.V.; Bangale, P.A. Antihyperlipidemic effect of protocatechuic acid in fructose induced hyperlipidemia in rats. Int. J. Pharma Bio. Sci. 2011, 2, 456–460. [Google Scholar]
- Wang, D.; Wei, X.; Yan, X.; Jin, T.; Ling, W. Protocatechuic acid, a metabolite of anthocyanins, inhibits monocyte adhesion and reduces atherosclerosis in apolipoprotein E-deficient mice. J. Agric. Food Chem. 2010, 58, 12722–12728. [Google Scholar] [CrossRef]
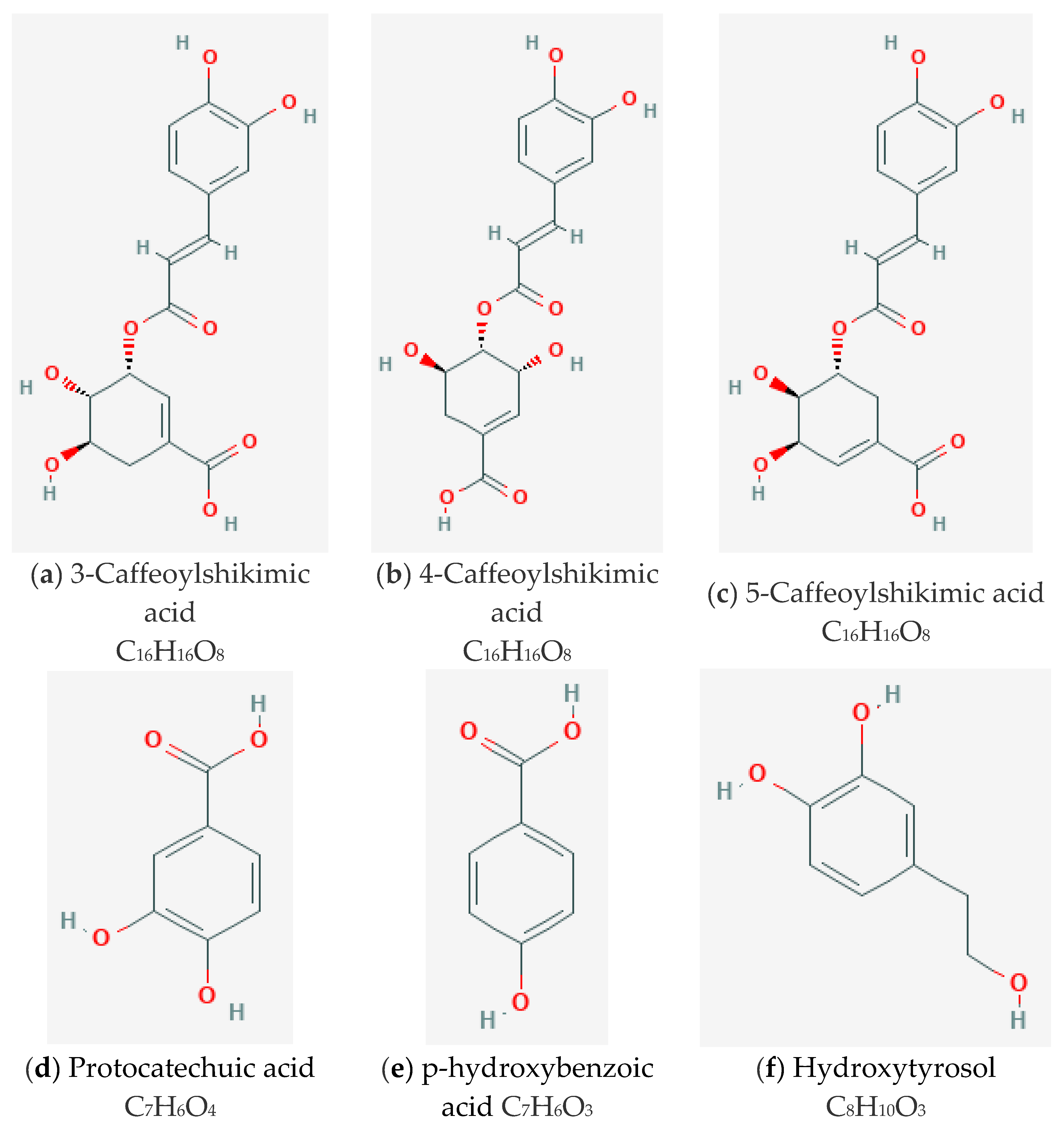
- Sambandan, T.G.; Rha, C.; Sinskey, A.J.; Sambanthamurthi, R.; Tan, Y.A.; Manickam, K.S.P.; Wahid, M.B. Composition Comprising Caffeoylshikimic Acids, Protocatechuic Acid, Hydroxytyrosol, Hydroxybenzoic Acid and Their Derivatives and Method of Preparation Thereof. U.S. Patent 9,962,421, 8 May 2018. [Google Scholar]
- Reboredo-Rodriguez, P.; Rey-Salgueiro, L.; Regueiro, J.; González-Barreiro, C.; Cancho-Grande, B.; Simal-Gándara, J. Ultrasound-assisted emulsification-microextraction for the determination of phenolic compounds in olive oils. Food Chem. 2014, 150, 128–136. [Google Scholar] [CrossRef]
- Borzì, A.M.; Biondi, A.; Basile, F.; Luca, S.; Vicari, E.S.D.; Vacante, M. Olive Oil Effects on Colorectal Cancer. Nutrients 2019, 11, 32. [Google Scholar] [CrossRef]
- Visioli, F.; Davalos, A.; de Las Hazas, M.L.; Crespo, M.C.; Tomé-Carneiro, J. An overview of the pharmacology of olive oil and its active ingredients. Br. J. Pharmacol. 2019, 177, 1316–1330. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Santiago, M.; Martín-Bautista, E.; Carrero, J.J.; Fonollá, J.; Baró, L.; Bartolomé, M.V.; Gil-Loyzaga, P.; López-Huertas, E. One-month administration of hydroxytyrosol, a phenolic antioxidant present in olive oil, to hyperlipemic rabbits improves blood lipid profile, antioxidant status and reduces atherosclerosis development. Atherosclerosis 2006, 188, 35–42. [Google Scholar] [CrossRef]
- Covas, M.I.; Nyyssönen, K.; Poulsen, H.E.; Kaikkonen, J.; Zunft, H.J.F.; Kiesewetter, H.; Gaddi, A.; de la Torre, R.; Mursu, J.; Bäumler, H.; et al. The effect of polyphenols in olive oil on heart disease risk factors: A randomized trial. Ann. Intern Med. 2006, 145, 333–341. [Google Scholar] [CrossRef] [PubMed]
- Carluccio, M.A.; Siculella, L.; Ancora, M.A.; Massaro, M.; Scoditti, E.; Storelli, C.; Visioli, F.; Distante, A.; De Caterina, R. Olive oil and red wine antioxidant polyphenols inhibit endothelial activation: Antiatherogenic properties of Mediterranean diet phytochemicals. Arter. Thromb Vasc. Biol. 2003, 23, 622–629. [Google Scholar] [CrossRef] [PubMed]
- Bulotta, S.; Celano, M.; Lepore, S.M.; Montalcini, T.; Pujia, A.; Russo, D. Beneficial effects of the olive oil phenolic components oleuropein and hydroxytyrosol: Focus on protection against cardiovascular and metabolic diseases. J. Transl. Med. 2014, 12, 219. [Google Scholar] [CrossRef]
- Cerqueira, N.M.; Oliveira, E.F.; Gesto, D.S.; Santos-Martins, D.; Moreira, C.; Moorthy, H.N.; Ramos, M.J.; Fernandes, F.A. Cholesterol Biosynthesis: A Mechanistic Overview. Biochemistry 2016, 55, 5483–5506. [Google Scholar] [CrossRef]
- Lehninger, A.L.; Nelson, D.L.; Cox, M.M. Lipid biosynthesis. In Lehninger’s Principles of Biochemistry, 4th ed.; W. H. Freeman: New York, NY, USA, 2005. [Google Scholar]
- Sharpe, L.J.; Brown, A.J. Controlling cholesterol synthesis beyond 3-hydroxy-3-methylglutaryl-CoA reductase (HMGCR). J. Biol. Chem. 2013, 288, 18707–18715. [Google Scholar] [CrossRef]
- Le Jossic-Corcos, C.; Gonthier, C.; Zaghini, I.; Logette, E.; Shechter, I.; Bournot, P. Hepatic farnesyl diphosphate synthase expression is suppressed by polyunsaturated fatty acids. Biochem. J. 2005, 385, 787–794. [Google Scholar] [CrossRef]
- Harris, W.S. n-3 fatty acids and serum lipoproteins: Animal studies. Am. J. Clin. Nutr. 1997, 65 (Suppl. 5), 1611s–1616s. [Google Scholar] [CrossRef]
- Ribeiro, A.; Mangeney, M.; Cardot, P.; Loriette, C.; Rayssiguier, Y.; Chambaz, J.; Bereziat, G. Effect of dietary fish oil and corn oil on lipid metabolism and apolipoprotein gene expression by rat liver. Eur. J. Biochem. 1991, 196, 499–507. [Google Scholar] [CrossRef]
- Alvaro, A.; Solà, R.; Rosales, R.; Ribalta, J.; Anguera, A.; Masana, L.; Vallvé, J.C. Gene expression analysis of a human enterocyte cell line reveals downregulation of cholesterol biosynthesis in response to short-chain fatty acids. Iubmb Life 2008, 60, 757–764. [Google Scholar]
- Cervantes Gracia, K.; Llanas-Cornejo, D.; Husi, H. CVD and Oxidative Stress. J. Clin. Med. 2017, 6, 22. [Google Scholar] [CrossRef] [PubMed]
- Austin, V.; Crack, P.J.; Bozinovski, S.; Miller, A.A.; Vlahos, R. COPD and stroke: Are systemic inflammation and oxidative stress the missing links? Clin. Sci. 2016, 130, 1039–1050. [Google Scholar] [CrossRef] [PubMed]
- Witztum, J.L.; Steinberg, D. Role of oxidized low density lipoprotein in atherogenesis. J. Clin. Investig. 1991, 88, 1785–1792. [Google Scholar] [CrossRef]
- Berliner, J.A.; Heinecke, J.W. The role of oxidized lipoproteins in atherogenesis. Free Radic Biol. Med. 1996, 20, 707–727. [Google Scholar] [CrossRef]
- Ross, R. The pathogenesis of atherosclerosis: A perspective for the 1990s. Nature 1993, 362, 801–809. [Google Scholar] [CrossRef]
- Tedgui, A.; Mallat, Z. Cytokines in atherosclerosis: Pathogenic and regulatory pathways. Physiol Rev. 2006, 86, 515–581. [Google Scholar] [CrossRef]
- Pandey, K.B.; Rizvi, S.I. Plant polyphenols as dietary antioxidants in human health and disease. Oxidative Med. Cell. Longev. 2009, 2, 270–278. [Google Scholar] [CrossRef]
- Xu, R.; Yokoyama, W.H.; Irving, D.; Rein, D.; Walzem, R.L.; German, J.B. Effect of dietary catechin and vitamin E on aortic fatty streak accumulation in hypercholesterolemic hamsters. Atherosclerosis 1998, 137, 29–36. [Google Scholar] [CrossRef]
- Poljsak Borut, F.R. The Protective Role of Antioxidants in the Defence against ROS/RNS-Mediated Environmental Pollution. Oxidative Med. Cell. Longev. 2014, 2014, 22. [Google Scholar]
- Kinsella, J.E.; Frankel, E.N.; German, J.B.; Kanner, J. Possible mechanism for the protective role of antioxidants in wine and plant foods. Food Technol. 1993, 47, 85–89. [Google Scholar]
- Goszcz, K.; Duthie, G.G.; Stewart, D.; Leslie, S.J.; Megson, L.J. Bioactive polyphenols and cardiovascular disease: Chemical antagonists, pharmacological agents or xenobiotics that drive an adaptive response? Br. J. Pharm. 2017, 174, 1209–1225. [Google Scholar] [CrossRef]
- Pasinetti, G.M.; Singh, R.; Westfall, S.; Herman, F.; Faith, J.; Ho, L. The Role of the Gut Microbiota in the Metabolism of Polyphenols as Characterized by Gnotobiotic Mice. J. Alzheimers Dis. 2018, 63, 409–421. [Google Scholar] [CrossRef] [PubMed]
- Marín, L.; Miguélez, E.M.; Villar, C.J.; Lombó, F. Bioavailability of dietary polyphenols and gut microbiota metabolism: Antimicrobial properties. BioMed Res. Int. 2015, 2015, 905215. [Google Scholar] [CrossRef] [PubMed]
- Kawabata, K.; Yoshioka, Y.; Terao, J. Role of Intestinal Microbiota in the Bioavailability and Physiological Functions of Dietary Polyphenols. Molecules 2019, 24, 370. [Google Scholar] [CrossRef] [PubMed]
- Moskaug, J.O.; Carlsen, H.; Myhrstad, M.C.W.; Blomhoff, R. Polyphenols and glutathione synthesis regulation. Am. J. Clin. Nutr. 2005, 81 (Suppl. 1), 277s–283s. [Google Scholar] [CrossRef]
- Rice-Evans, C.A.; Miller, N.J.; Paganga, G. Structure-antioxidant activity relationships of flavonoids and phenolic acids. Free Radic. Biol. Med. 1996, 20, 933–956. [Google Scholar] [CrossRef]
- Grootveld, M.; Halliwell, B. Aromatic hydroxylation as a potential measure of hydroxyl-radical formation in vivo. Identification of hydroxylated derivatives of salicylate in human body fluids. Biochem. J. 1986, 237, 499–504. [Google Scholar]
- Rodriguez, J.C.; Gómez, D.; Pacetti, D.; Núñez, O.; Gagliardi, R.; Frega, N.G.; Ojeda, M.L.; Loizzo, M.R.; Tundis, R.; Lucci, P. Effects of the Fruit Ripening Stage on Antioxidant Capacity, Total Phenolics, and Polyphenolic Composition of Crude Palm Oil from Interspecific Hybrid Elaeis oleifera x Elaeis guineensis. J. Agric. Food Chem. 2016, 64, 852–859. [Google Scholar] [CrossRef]
- Selby-Pham, S.N.B.; Siow, L.F.; Bennett, L.E. Characterising absorption and health-related properties of phytochemicals extracted from Malaysian palm fruit biomass after oil extraction. Food Funct. 2020, 11, 907–920. [Google Scholar] [CrossRef]
- Mehra, V.C.; Ramgolam, V.S.; Bender, J.R. Cytokines and cardiovascular disease. J. Leukoc. Biol. 2005, 78, 805–818. [Google Scholar] [CrossRef]
- Berger, A. Th1 and Th2 responses: What are they? BMJ 2000, 321, 424. [Google Scholar] [CrossRef] [PubMed]
- Frostegard, J.; Ulfgren, A.K.; Nyberg, P.; Hedin, U.; Swedenborg, J.; Andersson, U.; Hansson, G.K. Cytokine expression in advanced human atherosclerotic plaques: Dominance of pro-inflammatory (Th1) and macrophage-stimulating cytokines. Atherosclerosis 1999, 145, 33–43. [Google Scholar] [CrossRef]
- Engelbertsen, D.; Andersson, L.; Ljungcrantz, I.; Wigren, M.; Hedblad, B.; Nilsson, J.; Björkbacka, H. T-helper 2 immunity is associated with reduced risk of myocardial infarction and stroke. Arter. Thromb. Vasc. Biol. 2013, 33, 637–644. [Google Scholar] [CrossRef] [PubMed]
- Kleemann, R.; Zadelaar, S.; Kooistra, T. Cytokines and atherosclerosis: A comprehensive review of studies in mice. Cardiovasc Res. 2008, 79, 360–376. [Google Scholar] [CrossRef] [PubMed]
- Abbas, A.K.; Lichtman, A.H.; Pillai, S. Cellular and Molecular Immunology; Elsevier: Amsterdam, The Netherlands, 2005. [Google Scholar]
- Patel, N.S.; Paris, D.; Mathura, V.; Quadros, A.N.; Crawford, F.C.; Mullan, M.J. Inflammatory cytokine levels correlate with amyloid load in transgenic mouse models of Alzheimer’s disease. J. Neuroinflammation 2005, 2, 9. [Google Scholar] [CrossRef] [PubMed]
- Bartlett, K.; Eaton, S. Mitochondrial β-oxidation. Eur. J. Biochem. 2004, 271, 462–469. [Google Scholar] [CrossRef] [PubMed]
- Nagy, K.; Tiuca, I.-D. Importance of Fatty Acids in Physiopathology of Human Body. In Fatty Acids; Intech Open: London, UK, 2017. [Google Scholar] [CrossRef]
- Houten, S.M.; Wanders, R.J.A. A general introduction to the biochemistry of mitochondrial fatty acid β-oxidation. J. Inherit. Metab. Dis. 2010, 33, 469–477. [Google Scholar] [CrossRef]
- Stocker, R.; Keaney, J.F., Jr. Role of oxidative modifications in atherosclerosis. Physiol. Rev. 2004, 84, 1381–1478. [Google Scholar] [CrossRef]
- Murase, T.; Nagasawa, A.; Suzuki, J.; Hase, T.; Tokimitsu, I. Beneficial effects of tea catechins on diet-induced obesity: Stimulation of lipid catabolism in the liver. Int. J. Obes. Relat. Metab. Disord 2002, 26, 1459–1464. [Google Scholar] [CrossRef]
- Shimoda, H.; Seki, E.; Aitani, M. Inhibitory effect of green coffee bean extract on fat accumulation and body weight gain in mice. BMC Complementary Altern. Med. 2006, 6, 9. [Google Scholar] [CrossRef]
- Bender, D.A. Tricarboxylic Acid Cycle, in Encyclopedia of Food Sciences and Nutrition, 2nd ed.; Caballero, B., Ed.; Academic Press: Oxford, UK, 2003; pp. 5851–5856. [Google Scholar]
- Czibik, G.; Steeples, V.; Yavari, A.; Ashrafian, H. Citric Acid Cycle Intermediates in Cardioprotection. Circ. Cardiovasc. Genet. 2014, 7, 711–719. [Google Scholar] [CrossRef] [PubMed]
- Doenst, T.; Nguyen, T.D.; Abel, E.D. Cardiac metabolism in heart failure: Implications beyond ATP production. Circ. Res. 2013, 113, 709–724. [Google Scholar] [CrossRef] [PubMed]
- Opie, L.H. Heart Physiology: From Cell to Circulation; Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2004. [Google Scholar]
- Heather, L.C.; Cole, M.A.; Lygate, C.A.; Evans, R.D.; Stuckey, D.J.; Murray, A.J.; Neubauer, S.; Clarke, K. Fatty acid transporter levels and palmitate oxidation rate correlate with ejection fraction in the infarcted rat heart. Cardiovasc Res. 2006, 72, 430–437. [Google Scholar] [CrossRef] [PubMed]
- Lundsgaard, A.-M.; Fritzen, A.M.; Kiens, B. Chapter 36—Exercise Physiology in Men and Women, in Principles of Gender-Specific Medicine, 3rd ed.; Legato, M.J., Ed.; Academic Press: Cambridge, MA, USA, 2017; pp. 525–542. [Google Scholar]
- Laskowski, K.R.; Russell, R.R., 3rd. Uncoupling proteins in heart failure. Curr. Heart Fail. Rep. 2008, 5, 75–79. [Google Scholar] [CrossRef] [PubMed]
- Fisler, J.S.; Warden, C.H. Uncoupling proteins, dietary fat and the metabolic syndrome. Nutr. Metab. 2006, 3, 38. [Google Scholar] [CrossRef] [PubMed]
- Adams, P.L.; Turnbull, D.M. Disorders of the electron transport chain. J. Inherit. Metab. Dis. 1996, 19, 463–469. [Google Scholar] [CrossRef]
- Mei, N.; Guo, L.; Zhang, L.; Shi, L.; Sun, Y.A.; Fung, C.; Moland, C.L.; Dial, S.L.; Fuscoe, J.C.; Chen, T. Analysis of gene expression changes in relation to toxicity and tumorigenesis in the livers of Big Blue transgenic rats fed comfrey (Symphytum officinale). BMC Bioinform. 2006, 7 (Suppl. 2), S16. [Google Scholar] [CrossRef]
- Sambanthamurthi, R.; Tan, Y.A.; Sundram, K.; Hayes, K.C.; Abeywardena, M.; Leow, S.S.; Sekaran, S.K.; Sambandan, T.G.; Rha, C.; Sinskey, A.J.; et al. Positive outcomes of oil palm phenolics on degenerative diseases in animal models. Br. J. Nutr. 2011, 106, 1664–1675. [Google Scholar] [CrossRef]
- Bolsinger, J.; Pronczuk, A.; Sambanthamurthi, R.; Hayes, K.C. Anti-diabetic effects of palm fruit juice in the Nile rat (Arvicanthis niloticus). J. Nutr. Sci. 2014, 3, e5. [Google Scholar] [CrossRef]
- Reagan-Shaw, S.; Nihal, M.; Ahmad, N. Dose translation from animal to human studies revisited. FASEB J. 2008, 22, 659–661. [Google Scholar] [CrossRef] [PubMed]
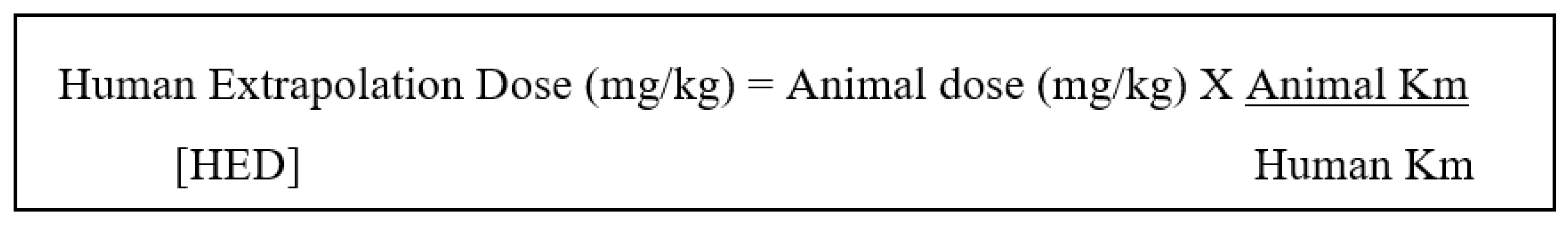
| Study Type | Sample/Population | Intervention Dose and Route | Findings | Reference |
|---|---|---|---|---|
| Animal study | Male New Zealand white rabbits induced with an atherogenic diet for 100 days. | OPP (1500 mg GAE/L) in drinking fluid. | OPP treatment showed no significant difference in plasma lipid profile, while slightly higher in the high-density lipoprotein cholesterol (HDL-C) level compared to control. OPP treatment resulted in significantly lower fatty streaks development compared to the control (p < 0.05). OPP treatment indicated proliferation of smooth muscle, development of intimal fibrosis and extracellular lipid. However, lipid core or necrosis was absent. The formation of foam cell was at lesser degree. | [28] |
| Animal study | Male spontaneously hypertensive rats (SHR). | OPP at 1500 and 3000 GAE for 20 weeks, as a beverage (30 mL/rat/day). | The prolonged OPP supplementation at 1500 mg/L GAE did not affect blood pressure in this model. | [33] |
| Animal study | Sprague-Dawley rats induced hypertension with L-NAME. | OPP at 1500 mg/L and 3000 mg/L GAE) as a beverage (30 mL/rat/day) for 4 weeks prior to L-NAME induction. Treatments were continued for a further two months thereafter. | OPP treatment at 3000 mg/L GAE significantly lowered the blood pressure in the L-NAME hypertension model (p < 0.001) compared to OPP dosed at 1500 mg/L GAE (p < 0.01). OPP treatment at both doses did not return BP to a complete normalization to the baseline level of the control group. OPP treatment at 3000 mg/L GAE significantly reduced the duration of ventricular tachycardia (p < 0.01). There were no episodes of ventricular fibrillation occurred when compared to the control group. | |
| Animal study | Male SHR. | Fractionated OPP (as a single oral dose, 250 mg/kg) was introduced via pipette positioned at the back of the tongue of SHR. | The OPP fractions reduced mean BP (7–27 mmHg) within 6 h post-administration. Both OPP fractions showed the highest BP reduction at 6 h post-administration. | |
| Animal study | Male Wistar Kyoto rats fed with pro-arrhythmic diet. | OPP (1500 mg/L GAE) was given as beverage (30 mL/rat/day). | OPP significantly reduced the ventricular fibrillation (VF) incidences when compared to the control group (p < 0.05). OPP-treated group had a lower percentage of VF (52%) compared to the control group (90%) OPP-treated group had lower mortality (20%) compared to control rats (40%) There was no difference in ischemic-affected myocardium area (zone-at-risk) between OPP and control groups. | |
| Ex vivo | Isolated segments (3 mm) of the thoracic aorta and mesenteric arterial bed from male normotensive Wistar Kyoto rats and SHR. | OPP was introduced at the following doses: 0.25, 0.50, and 1.00 mg/kg to pre-contracted vascular preparations in the organ bath chamber. | In a dose-dependently manner, OPP enhanced vascular relaxation in both ex vivo systems; isolated aortic rings (conductance vessels) and perfused mesenteric vascular bed (resistance vessels). | [23] |
| In vitro | Conjugated dienes. | OPP extracts were added to low-density lipoprotein cholesterols (LDL-C) immediately before the addition of oxidant (copper sulphate). | In a dose-dependently manner, OPP prevented the Cu-mediated LDL oxidation. OPP have delayed the duration of conjugated diene formation when compared to the control. | |
| Animal study | Male inbred BALB/c mice. | OPP in drinking fluids ad libitum 1500 GAE mg/L. | OPP have upregulated four lipid catabolism genes (Acadl, Acads, Hadhb, Hadhsc) and downregulated five cholesterol biosynthesis genes (Hmgcs1, Lss, Sc4mol, Fdps, Nsdhl) | [32] |
| Animal study | Male inbred BALB/c mice. | OPP in drinking fluids ad libitum at 1500 ppm GAE mg/L. | OPP-treated group have significantly increased the total cholersterol (TC), LDL and HDL levels OPP have downregulated the genes expressed in the presentation of endogenous antigen, metabolism of fatty acids, enzymatic activities of NADH dehydrogenase (ubiquinone) and oxidoreductase. OPP have upregulated genes expressed in the heart antioxidant activity; Gpx1 and Mgst1 | [27] |
| Human study | 25 volunteers Normolipidemic, nonsmokers, and no clinical symptoms associated with CVD. | OPP was supplemented as 300 mL beverage (containing 450 mg/GAE/day). | Following the 60 days OPP supplementation, plasma TC and LDL-C levels were significantly lower compared to the control treatment, with p = 0.025 and p = 0.04, respectively. However, the OPP-supplemented group showed insignificant changes in HDL-C, triacylglycerol (TAG) and TC/HDL ratio when compared to control treatments. | [34] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ibrahim, N.‘I.; Fairus, S.; Naina Mohamed, I. The Effects and Potential Mechanism of Oil Palm Phenolics in Cardiovascular Health: A Review on Current Evidence. Nutrients 2020, 12, 2055. https://doi.org/10.3390/nu12072055
Ibrahim N‘I, Fairus S, Naina Mohamed I. The Effects and Potential Mechanism of Oil Palm Phenolics in Cardiovascular Health: A Review on Current Evidence. Nutrients. 2020; 12(7):2055. https://doi.org/10.3390/nu12072055
Chicago/Turabian StyleIbrahim, Nurul ‘Izzah, Syed Fairus, and Isa Naina Mohamed. 2020. "The Effects and Potential Mechanism of Oil Palm Phenolics in Cardiovascular Health: A Review on Current Evidence" Nutrients 12, no. 7: 2055. https://doi.org/10.3390/nu12072055
APA StyleIbrahim, N. ‘I., Fairus, S., & Naina Mohamed, I. (2020). The Effects and Potential Mechanism of Oil Palm Phenolics in Cardiovascular Health: A Review on Current Evidence. Nutrients, 12(7), 2055. https://doi.org/10.3390/nu12072055





