Cognitive Impairment in Coeliac Disease with Respect to Disease Duration and Gluten-Free Diet Adherence: A Pilot Study
Abstract
1. Introduction
- (1)
- Do newly diagnosed patients with CD have cognitive difficulties when compared to healthy controls?
- (2)
- Do patients with a longstanding diagnosis of CD differ from newly diagnosed patients with regard to their cognitive profile?
- (3)
- Do patients who comply with a gluten-free diet have better cognition than those who do not?
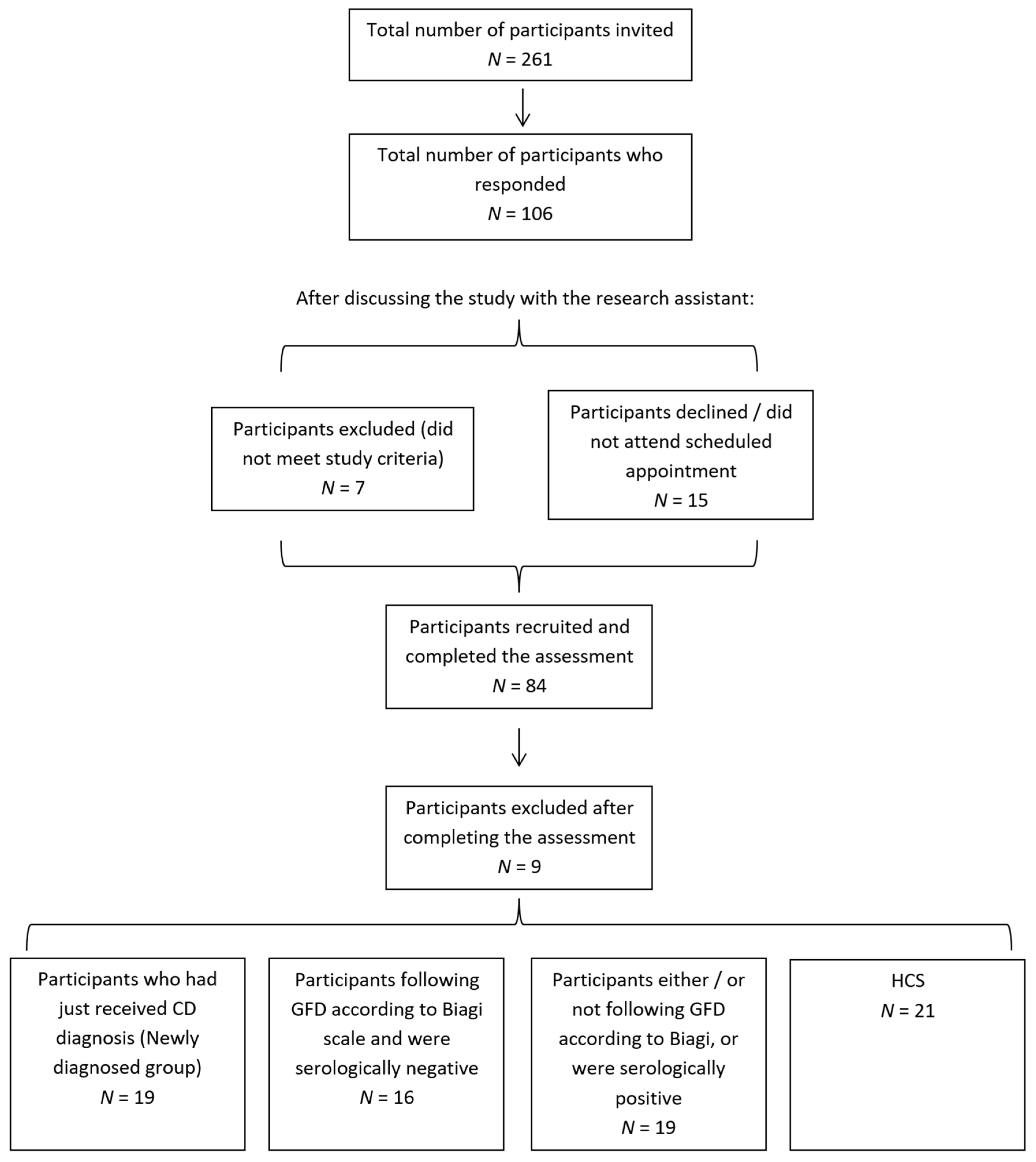
2. Materials and Methods
2.1. Participants
2.2. Study Power
2.3. Design
2.4. Assessment Procedure
2.5. Testing Battery and Initial Data Handling
2.6. Statistical Analysis
3. Results
3.1. Analyses of Samples’ Demographics
3.2. Analyses of Cognitive Performance
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ludvigsson, J.F.; Leffler, D.A.; Bai, J.C.; Biagi, F.; Fasano, A.; Green, P.H.R.; Hadjivassiliou, M.; Kaukinen, K.; Kelly, C.P.; Leonard, J.N.; et al. The Oslo definitions for coeliac disease and related terms. Gut 2012, 62, 43–52. [Google Scholar] [CrossRef] [PubMed]
- Mooney, P.; Hadjivassiliou, M.; Sanders, D.S. Coeliac disease. BMJ 2014, 348, g1561. [Google Scholar] [CrossRef] [PubMed]
- Halfdanarson, T.R.; Litzow, M.R.; Murray, J.A. Hematologic manifestations of celiac disease. Blood 2006, 109, 412–421. [Google Scholar] [CrossRef] [PubMed]
- Collin, P.; Salmi, T.; Hervonen, K.; Kaukinen, K.; Reunala, T. Dermatitis herpetiformis: A cutaneous manifestation of coeliac disease. Ann. Med. 2016, 49, 23–31. [Google Scholar] [CrossRef]
- Nikpour, S. Neurological manifestations, diagnosis, and treatment of celiac disease: A comprehensive review. Iran. J. Neurol. 2012, 11, 59–64. [Google Scholar]
- Burk, K.; Bösch, S.; Müller, C.A.; Melms, A.; Zühlke, C.; Stern, M.; Besenthal, I.; Skalej, M.; Ruck, P.; Ferber, S.; et al. Sporadic cerebellar ataxia associated with gluten sensitivity. Brain 2001, 124, 1013–1019. [Google Scholar] [CrossRef] [PubMed]
- Hadjivassiliou, M.; Grunewald, R.; Chattopadhyay, A.; Davies-Jones, G.; Gibson, A.; Jarratt, J.; Kandler, R.; Lobo, A.; Powell, T.; Smith, C. Clinical, radiological, neurophysiological, and neuropathological characteristics of gluten ataxia. Lancet 1998, 352, 1582–1585. [Google Scholar] [CrossRef]
- Hadjivassiliou, M.; Sanders, D.S.; Grünewald, R.A.; Woodroofe, N.; Boscolo, S.; Aeschlimann, D. Gluten sensitivity: From gut to brain. Lancet Neurol. 2010, 9, 318–330. [Google Scholar] [CrossRef]
- Lebwohl, B.; Luchsinger, J.; Freedberg, D.E.; Green, P.H.; Ludvigsson, J.F. Risk of Dementia in Patients with Celiac Disease: A Population-Based Cohort Study. J. Alzheimer’s Dis. 2015, 49, 179–185. [Google Scholar] [CrossRef] [PubMed]
- Collin, P.; Pirttila, T.; Nurmikko, T.; Somer, H.; Erilä, T.; Keyriläinen, O. Celiac disease, brain atrophy, and dementia. Neurology 1991, 41, 372. [Google Scholar] [CrossRef] [PubMed]
- Hallert, C.; Åström, J. Intellectual ability of adults after lifelong intestinal malabsorption due to coeliac disease. J. Neurol. Neurosurg. Psychiatry 1983, 46, 87–89. [Google Scholar] [CrossRef] [PubMed]
- Casella, S.; Zanini, B.; Lanzarotto, F.; Ricci, C.; Marengoni, A.; Romanelli, G.; Lanzini, A. Cognitive performance is impaired in coeliac patients on gluten free diet: A case–control study in patients older than 65 years of age. Dig. Liver Dis. 2012, 44, 729–735. [Google Scholar] [CrossRef] [PubMed]
- Lichtwark, I.T.; Newnham, E.D.; Robinson, S.R.; Shepherd, S.J.; Hosking, P.; Gibson, P.R.; Yelland, G. Cognitive impairment in coeliac disease improves on a gluten-free diet and correlates with histological and serological indices of disease severity. Aliment. Pharmacol. Ther. 2014, 40, 160–170. [Google Scholar] [CrossRef] [PubMed]
- Croall, I.D.; Sanders, D.S.; Hadjivassiliou, M.; Hoggard, N. Cognitive Deficit and White Matter Changes in Persons with Celiac Disease: A Population-Based Study. Gastroenterology 2020, 158, 2112–2122. [Google Scholar] [CrossRef] [PubMed]
- Longarini, G.I.; Richly, P.; Temprano, M.D.L.P.; Costa, A.F.; Vázquez, H.; Moreno, M.L.; Niveloni, S.I.; Jer, H.H.; López, P.L.; Smecuol, E.; et al. Su1441 Cognitive Performance in Patients With Active Celiac Disease. Preliminary Results of a Prospective Study. Gastroenterology 2014, 146. [Google Scholar] [CrossRef]
- Hadjivassiliou, M.; Davies-Jones, G.A.B.; Sanders, D.S.; Grunewald, R. Dietary treatment of gluten ataxia. J. Neurol. Neurosurg. Psychiatry 2003, 74, 1221–1224. [Google Scholar] [CrossRef]
- Rashtak, S.; Murray, J.A. Review article: Coealic disease, new approaches to therapy. Aliment. Pharmacol. Ther. 2012, 35, 768–781. [Google Scholar] [CrossRef]
- Biagi, F.; Bianchi, P.I.; Marchese, A.; Trotta, L.; Vattiato, C.; Balduzzi, D.; Brusco, G.; Andrealli, A.; Cisarò, F.; Astegiano, M.; et al. A score that verifies adherence to a gluten-free diet: A cross-sectional, multicentre validation in real clinical life. Br. J. Nutr. 2012, 108, 1884–1888. [Google Scholar] [CrossRef] [PubMed]
- Lezak, M.D.; Howieson, D.B.; Bigler, E.D.; Tranel, D. Neuropsychological Assessment., 5th ed.; Oxford University Press: Oxford, UK, 2012. [Google Scholar]
- Ware, J.E.; Sherbourne, C.D. The MOS 36-item short-form health survey (SF-36). I. Conceptual framework and item selection. Med Care 1992, 30, 473–483. [Google Scholar] [CrossRef] [PubMed]
- Hu, W.T.; Murray, J.A.; Greenaway, M.C.; Parisi, J.E.; Josephs, K.A. Cognitive Impairment and Celiac Disease. Arch. Neurol. 2006, 63, 1440–1446. [Google Scholar] [CrossRef]
- Volta, U.; De Giorgio, R.; Petrolini, N.; Stanghellini, V.; Barbara, G.; Granito, A.; De Ponti, F.; Corinaldesi, R.; Bianchi, F.B. Clinical Findings and Anti-Neuronal Antibodies in Coeliac Disease with Neurological Disorders. Scand. J. Gastroenterol. 2002, 37, 1276–1281. [Google Scholar] [CrossRef] [PubMed]
- Ismail, Z.; Rajji, T.K.; Shulman, K.I. Brief cognitive screening instruments: An update. Int. J. Geriatr. Psychiatry 2010, 25, 111–120. [Google Scholar] [CrossRef] [PubMed]
- Hadjivassiliou, M.; Croall, I.D.; Zis, P.; Sarrigiannis, P.G.; Sanders, D.S.; Aeschlimann, P.; Grünewald, R.A.; Armitage, P.A.; Connolly, D.; Aeschlimann, D.; et al. Neurologic Deficits in Patients With Newly Diagnosed Celiac Disease Are Frequent and Linked With Autoimmunity to Transglutaminase 6. Clin. Gastroenterol. Hepatol. 2019, 17, 2678–2686.e2. [Google Scholar] [CrossRef]
- Liu, Y.-T.; Tang, B.-S.; Lan, W.; Song, N.-N.; Huang, Y.; Zhang, L.; Guan, W.-J.; Shi, Y.-T.; Shen, L.; Jiang, H.; et al. Distribution of Transglutaminase 6 in the Central Nervous System of Adult Mice. Anat. Rec. Adv. Integr. Anat. Evol. Boil. 2013, 296, 1576–1587. [Google Scholar] [CrossRef]
- Pratesi, R.; Gandolfi, L.; Riedman, H.F.G.; Farage, L.; de Castro, C.A.; Catassi, C. Serum IgA Antibodies from Patients with Coeliac Disease React Strongly with Human Brain Blood-Vessel Structures. Scand. J. Gastroenterol. 1998, 33, 817–821. [Google Scholar] [CrossRef] [PubMed]
- Rondanelli, M.; Faliva, M.A.; Gasparri, C.; Peroni, G.; Naso, M.; Picciotto, G.; Riva, A.; Nichetti, M.; Infantino, V.; Alalwan, T.A.; et al. Micronutrients Dietary Supplementation Advices for Celiac Patients on Long-Term Gluten-Free Diet with Good Compliance: A Review. Medical 2019, 55, 337. [Google Scholar] [CrossRef] [PubMed]
- Reynolds, E.H. Folic acid, ageing, depression, and dementia. BMJ 2002, 324, 1512–1515. [Google Scholar] [CrossRef]
- Landel, V.; Annweiler, C.; Millet, P.; Morello, M.; Féron, F. Vitamin D, Cognition and Alzheimer’s Disease: The Therapeutic Benefit is in the D-Tails. J. Alzheimer’s Dis. 2016, 53, 419–444. [Google Scholar] [CrossRef] [PubMed]
- Schmahmann, J.D. The cerebellum and cognition. Neurosci. Lett. 2018, 688, 62–75. [Google Scholar] [CrossRef]
- Fama, R.; Sullivan, E.V. Thalamic structures and associated cognitive functions: Relations with age and aging. Neurosci. Biobehav. Rev. 2015, 54, 29–37. [Google Scholar] [CrossRef] [PubMed]
- Lau, M.S.; Mooney, P.D.; White, W.; Rees, M.A.; Wong, S.H.; Kurien, M.; Trott, N.; Leffler, D.A.; Hadjivassiliou, M.; Sanders, D.S. The Role of an IgA/IgG-Deamidated Gliadin Peptide Point-of-Care Test in Predicting Persistent Villous Atrophy in Patients With Celiac Disease on a Gluten-Free Diet. Am. J. Gastroenterol. 2017, 112, 1859–1867. [Google Scholar] [CrossRef] [PubMed]
- Casellas, F.; Rodrigo, L.; Vivancos, J.L.; Riestra, S.; Pantiga, C.; Baudet, J.; Junquera, F.; Diví, V.P.; Abadia, C.; Papo, M.; et al. Factors that impact health-related quality of life in adults with celiac disease: A multicenter study. World J. Gastroenterol. 2008, 14, 46–52. [Google Scholar] [CrossRef] [PubMed]
- Barratt, S.M.; Leeds, J.S.; Sanders, D.S. Quality of life in Coeliac Disease is determined by perceived degree of difficulty adhering to a gluten-free diet, not the level of dietary adherence ultimately achieved. J. Gastrointest. Liver Dis. 2011, 20, 241–245. [Google Scholar]
- Zingone, F.; Siniscalchi, M.; Capone, P.; Tortora, R.; Andreozzi, P.; Capone, E.; Ciacci, C. The quality of sleep in patients with coeliac disease. Aliment. Pharmacol. Ther. 2010, 32, 1031–1036. [Google Scholar] [CrossRef] [PubMed]
- Murman, D.L. The Impact of Age on Cognition. Semin. Hear. 2015, 36, 111–121. [Google Scholar] [CrossRef] [PubMed]
| Variable | HCS | Newly Diagnosed | All Established Cases | Results of Statistical Comparison between Major Groups | Subgroup: Established CD and Diet Adherent | Subgroup: Established Case and Not Diet Adherent | Results of Test Comparing Established CD Subgroups |
|---|---|---|---|---|---|---|---|
| Sample Size | 21 | 19 | 35 | - | 16 | 19 | - |
| Age | 43.5 ± 16.2 | 45.1 ± 17.3 | 55.5 ± 12.7 | p = 0.007 1 F = 5.329 | 57.8 ± 10.6 | 53.6 ± 14.3 | p = 0.481 |
| % Female | 76.2% (N = 16) | 84.2% (N = 16) | 88.6 (N = 31) | p = 0.473 | 93.8% (N = 15) | 84.2% (N = 16) | p = 0.377 |
| Years of Education | 15.2 ± 2.0 | 14.1 ± 3.0 | 13.8 ± 3.0 | p = 0.172 F = 1.802 | 14.2 ± 3.2 | 13.4 ± 2.8 | p = 0.452 |
| ToPF IQ | 106.4 ± 5.6 | 104.4 ± 8.8 | 104.8 ± 8.2 | p = 0.662 F = 0.415 | 106.6 ± 6.8 | 103.3 ± 9.1 | p = 0.239 |
| Years since coeliac diagnosis | - | 0.0 | 11.8 ± 7.8 | - | 11.4 ± 4.4 | 12.1 ± 9.8 | p = 0.803 |
| Cognitive Outcome | HCS Mean ± SD | Newly Diagnosed Mean ± SD | Established CD Mean ± SD | p Value F Value |
|---|---|---|---|---|
| Mental Flexibility | 0.00 ± 0.60 | −0.14 ± 1.05 | −0.80 ± 0.95 | p = 0.064 F = 2.850 |
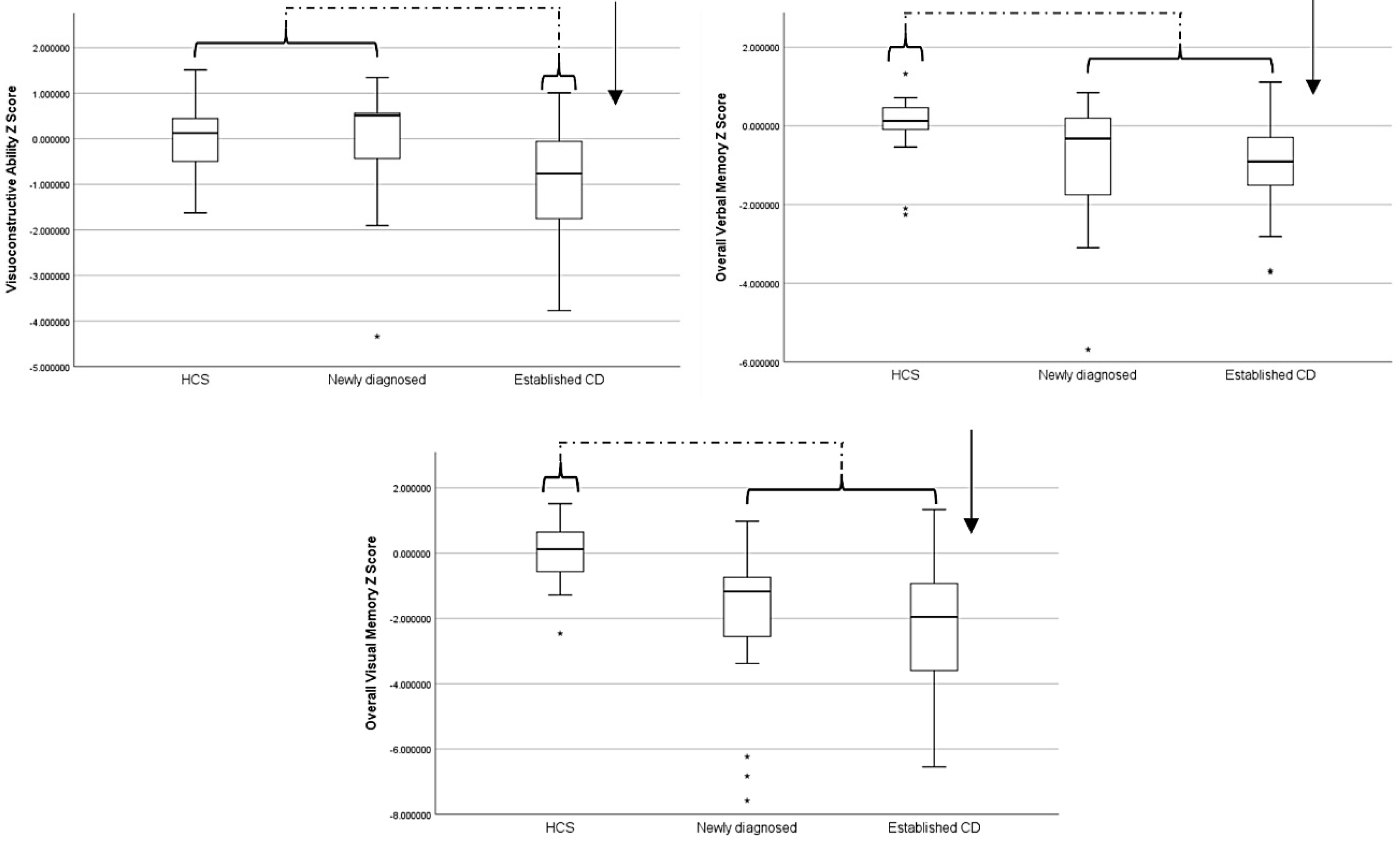
| Visuoconstructive Ability | 0.00 ± 0.79 | −0.11 ± 1.38 | −0.91 ± 1.09 | p = 0.050 1 F = 3.130 |
| Verbal Ability | 0.00 ± 0.87 | −0.07 ± 1.67 | −0.59 ± 1.03 | p = 0.129 F = 2.105 |
| Overall Verbal Memory | 0.00 ±0.83 | −0.97 ± 1.65 | −0.93 ± 1.19 | p = 0.046 2 F = 3.216 |
| Overall Visual Memory | 0.00 ± 0.96 | −2.04 ± 2.40 | −2.49 ± 2.02 | p < 0.001 3 F = 8.517 |
| Working Memory | 0.00 ± 1.02 | −0.26 ± 1.51 | −0.18 ± 1.66 | p = 0.762 F = 0.273 |
| Processing Speed | 0.00 ± 0.74 | 0.31 ± 1.02 | −0.13 ± 1.03 | p = 0.332 F = 1.121 |
| Cognitive Outcome | HCS Mean ± SD | Newly Diagnosed Mean ± SD | Established CD Mean ± SD | p Value F Value |
|---|---|---|---|---|
| ST Verbal Memory | 0.00 ± 0.59 | −0.29 ± 0.80 | −0.29 ± 0.74 | p = 0.478 F = 0.745 |
| LT Verbal Memory | 0.00 ± 0.85 | −1.23 ± 1.91 | −1.18 ± 1.34 | p = 0.019 F = 4.206 |
| ST Visual Memory | 0.00 ± 1.02 | −2.13 ± 2.31 | −2.65 ± 2.09 | p < 0.001 F = 9.472 |
| LT Visual Memory | 0.00 ± 1.02 | −1.95 ± 2.51 | −2.33 ± 2.01 | p = 0.002 F = 7.089 |
| SF-36 Outcome | HCS Mean ± SD | Newly Diagnosed Mean ± SD | Established CD Mean ± SD | Model Results |
|---|---|---|---|---|
| Physical Functioning | 51.33 ± 10.21 | 51.50 ± 8.24 | 47.49 ± 10.18 | p = 0.732 F = 0.313 |
| Physical Role | 52.85 ± 7.26 | 50.64 ± 10.40 | 46.93 ± 10.10 | p = 0.204 F = 1.627 |
| Bodily Pain | 54.39 ± 10.51 | 50.82 ± 9.06 | 44.14 ± 11.56 | p = 0.009 F = 4.977 |
| General Health | 50.51 ± 6.21 | 46.91 ± 9.01 | 44.80 ± 10.33 | p = 0.127 F = 2.126 |
| Vitality | 55.16 ± 5.81 | 47.83 ± 10.24 | 51.10 ± 8.67 | p = 0.030 F = 3.700 |
| Social Functioning | 54.27 ± 4.71 | 48.85 ± 7.83 | 49.06 ± 9.60 | p = 0.094 F = 2.449 |
| Emotional Role | 51.80 ± 9.01 | 48.66 ± 11.21 | 51.10 ± 8.14 | p = 0.534 F = 0.632 |
| Mental Health | 50.44 ± 6.89 | 49.14 ± 10.37 | 50.90 ± 9.69 | p = 0.841 F = 0.173 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Croall, I.D.; Tooth, C.; Venneri, A.; Poyser, C.; Sanders, D.S.; Hoggard, N.; Hadjivassiliou, M. Cognitive Impairment in Coeliac Disease with Respect to Disease Duration and Gluten-Free Diet Adherence: A Pilot Study. Nutrients 2020, 12, 2028. https://doi.org/10.3390/nu12072028
Croall ID, Tooth C, Venneri A, Poyser C, Sanders DS, Hoggard N, Hadjivassiliou M. Cognitive Impairment in Coeliac Disease with Respect to Disease Duration and Gluten-Free Diet Adherence: A Pilot Study. Nutrients. 2020; 12(7):2028. https://doi.org/10.3390/nu12072028
Chicago/Turabian StyleCroall, Iain D, Claire Tooth, Annalena Venneri, Charlotte Poyser, David S Sanders, Nigel Hoggard, and Marios Hadjivassiliou. 2020. "Cognitive Impairment in Coeliac Disease with Respect to Disease Duration and Gluten-Free Diet Adherence: A Pilot Study" Nutrients 12, no. 7: 2028. https://doi.org/10.3390/nu12072028
APA StyleCroall, I. D., Tooth, C., Venneri, A., Poyser, C., Sanders, D. S., Hoggard, N., & Hadjivassiliou, M. (2020). Cognitive Impairment in Coeliac Disease with Respect to Disease Duration and Gluten-Free Diet Adherence: A Pilot Study. Nutrients, 12(7), 2028. https://doi.org/10.3390/nu12072028






