Hydration Efficacy of a Milk Permeate-Based Oral Hydration Solution
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Population
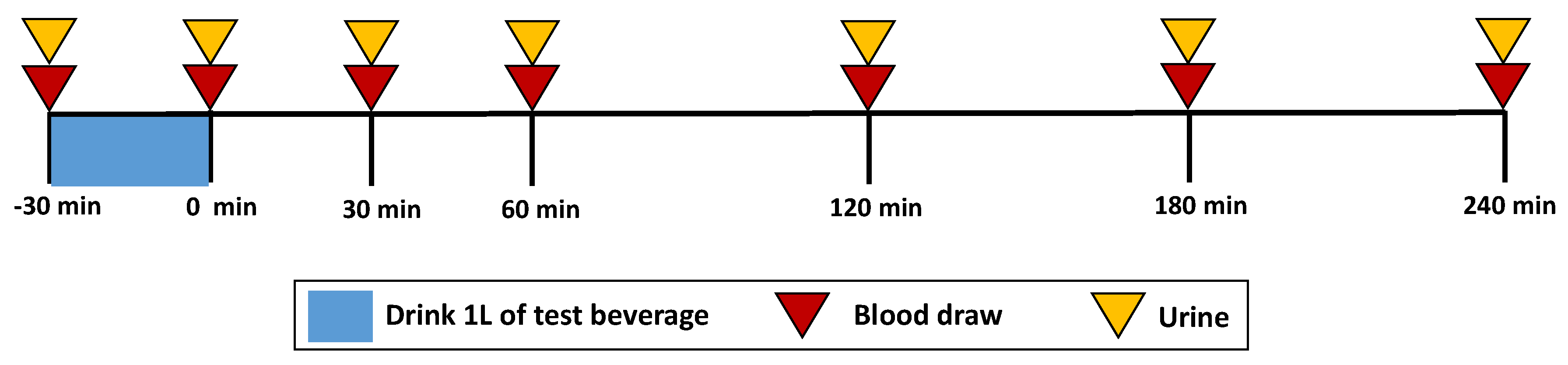
2.2. Study Design
2.3. Test Beverages
2.4. Urine and Serum Analysis
2.5. Data and Statistical Analysis
3. Results
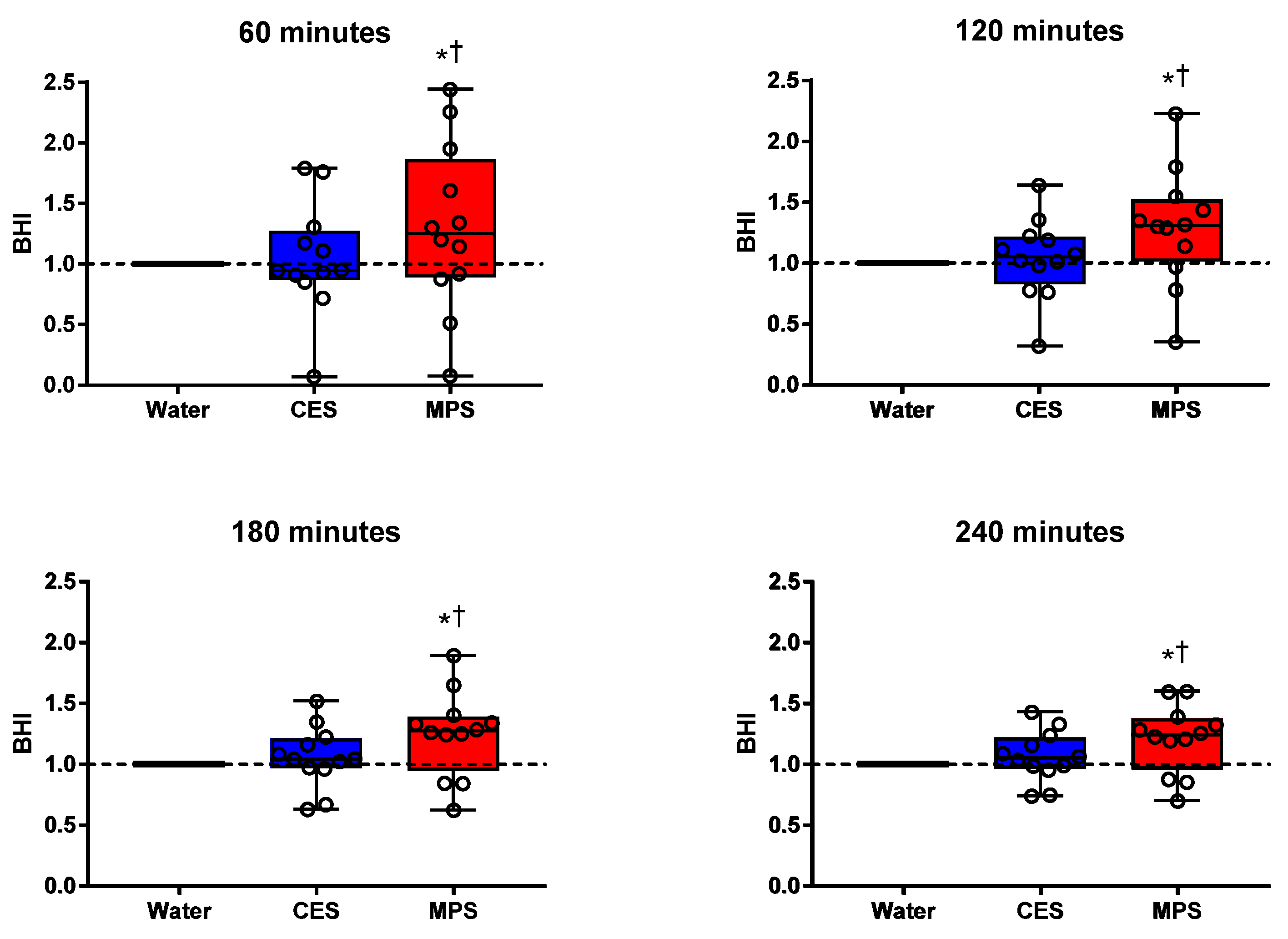
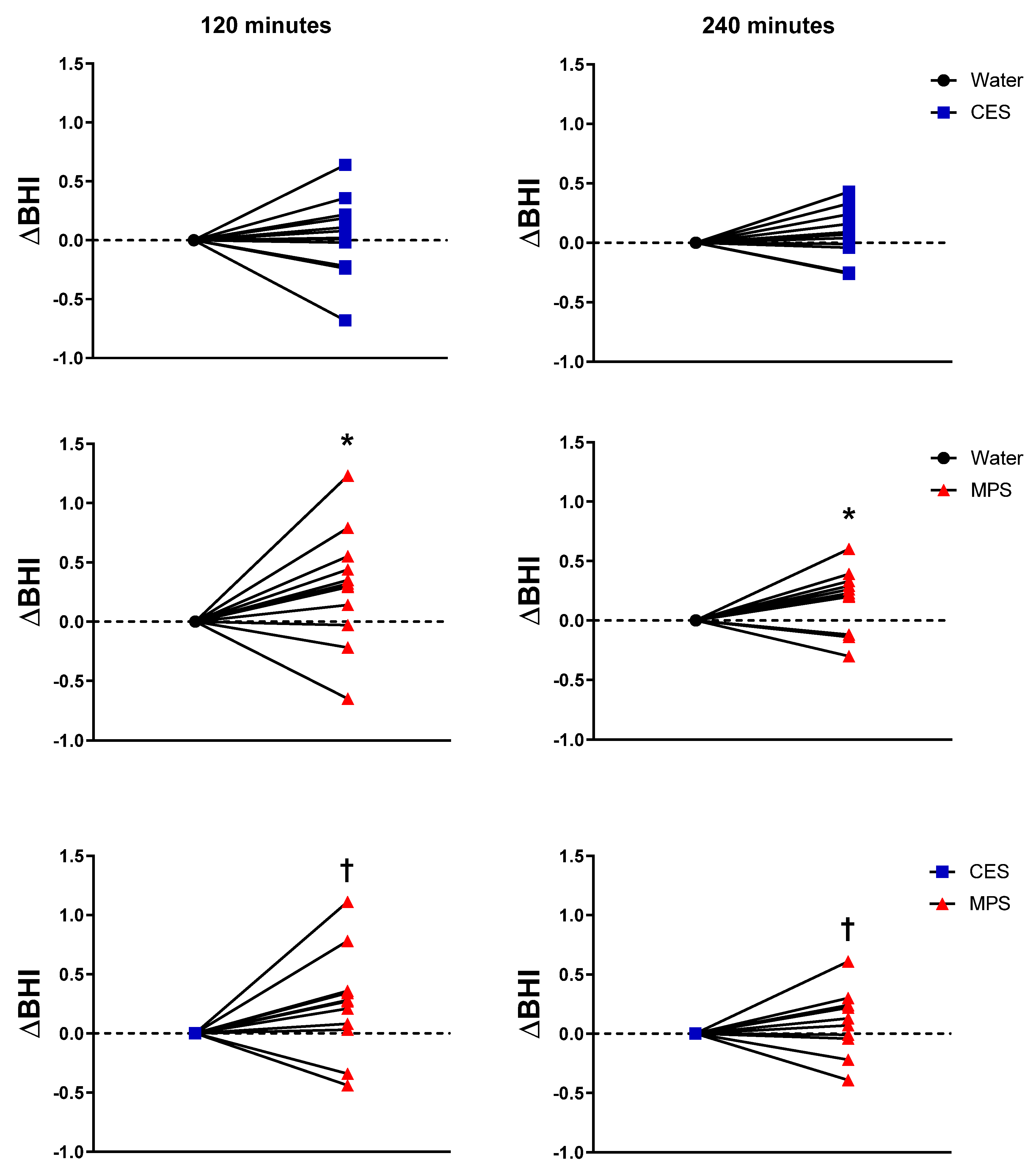
3.1. Cumulative Urine Output, Net Fluid Balance, and Beverage Hydration Index
3.2. Serum and Urine Electrolyte Concentrations and Osmolalities
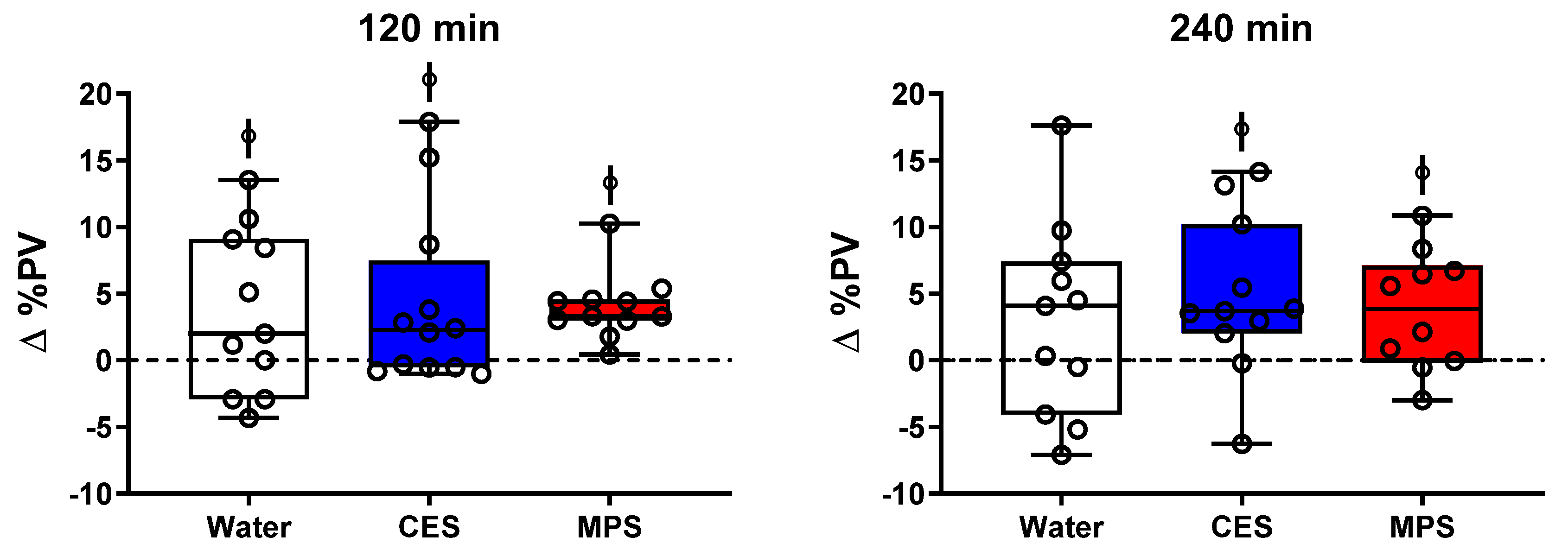
3.3. Plasma Volume
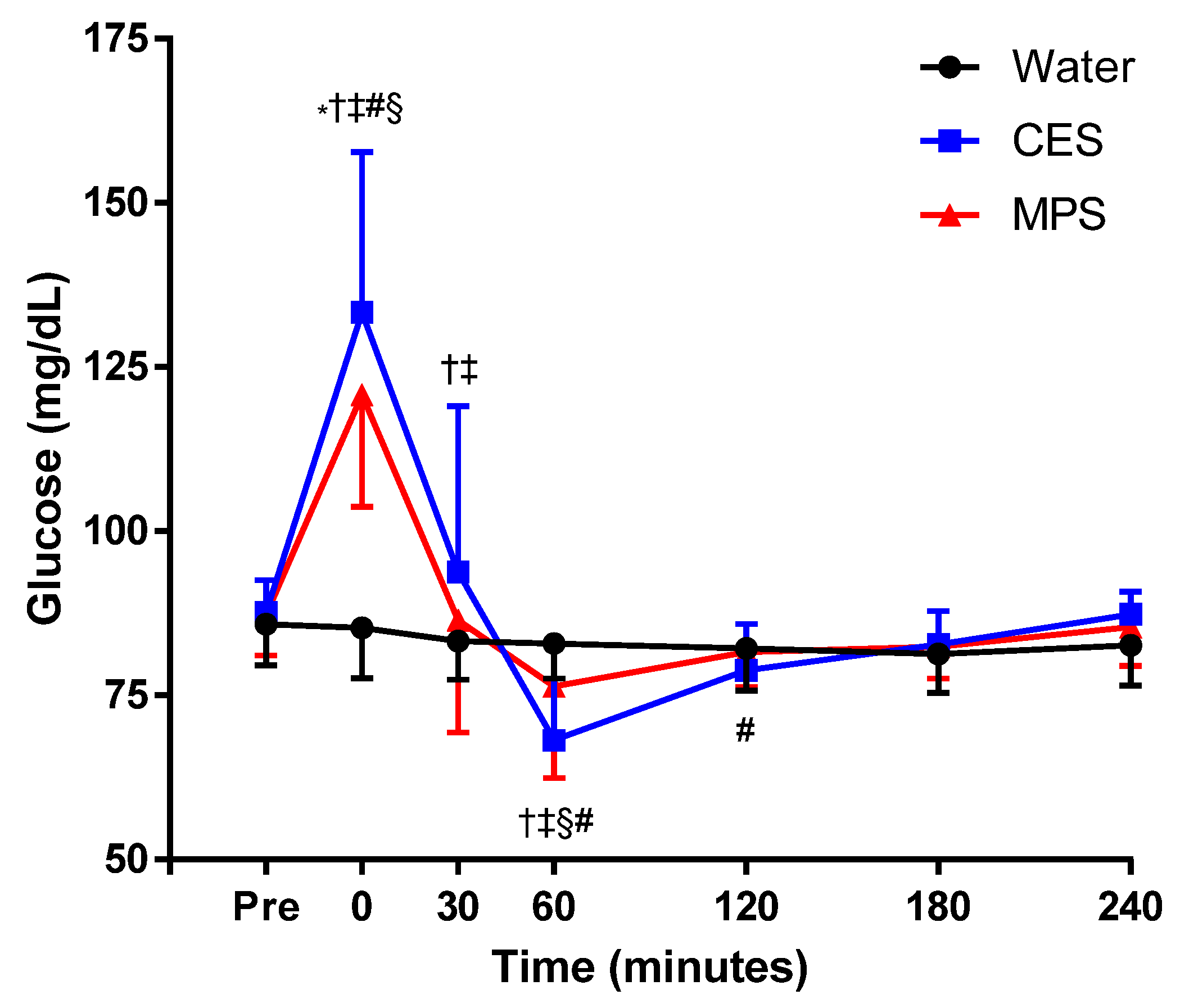
3.4. Plasma Glucose Responses
4. Discussion
Limitations
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Manz, F.; Wentz, A. The Importance of Good Hydration for the Prevention of Chronic Diseases. Nutr. Rev. 2005, 63, S2–S5. [Google Scholar] [CrossRef] [PubMed]
- Bethancourt, H.J.; Kenney, W.L.; Almeida, D.M.; Rosinger, A.Y. Cognitive performance in relation to hydration status and water intake among older adults, NHANES 2011–2014. Eur. J. Nutr. 2019, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Ganio, M.S.; Armstrong, L.E.; Casa, U.J.; McDermott, B.P.; Lee, E.C.; Yamamoto, L.M.; Marzano, S.; Lopez, R.; Jimenez, L.; Le Bellego, L.; et al. Mild dehydration impairs cognitive performance and mood of men. Br. J. Nutr. 2011, 106, 1535–1543. [Google Scholar] [CrossRef] [PubMed]
- Watson, P.; Whale, A.; Mears, S.A.; Reyner, L.A.; Maughan, R.J. Mild hypohydration increases the frequency of driver errors during a prolonged, monotonous driving task. Physiol. Behav. 2015, 147, 313–318. [Google Scholar] [CrossRef]
- Armstrong, L.E.; Costill, D.L.; Fink, W.J. Influence of diuretic-induced dehydration on competitive running performance. Med. Sci. Sports Exerc. 1985, 17, 456–461. [Google Scholar] [CrossRef]
- Walsh, R.; Noakes, T.D.; Hawley, J.A.; Dennis, S. Impaired High-Intensity Cycling Performance Time at Low Levels of Dehydration. Int. J. Sports Med. 1994, 15, 392–398. [Google Scholar] [CrossRef]
- Passe, D.; Horn, M.; Murray, R. Impact of beverage acceptability on fluid intake during exercise. Appetite 2000, 35, 219–229. [Google Scholar] [CrossRef]
- Rivera-Brown, A.M.; Gutiérrez, R.; Gutiérrez, J.C.; Frontera, W.R.; Bar-Or, O. Drink composition, voluntary drinking, and fluid balance in exercising, trained, heat-acclimatized boys. J. Appl. Physiol. 1999, 86, 78–84. [Google Scholar] [CrossRef]
- Wilk, B.; Bar-Or, O. Effect of drink flavor and NaCL on voluntary drinking and hydration in boys exercising in the heat. J. Appl. Physiol. 1996, 80, 1112–1117. [Google Scholar] [CrossRef]
- Gisolfi, C.V.; Summers, R.W.; Lambert, G.P.; Xia, T. Effect of beverage osmolality on intestinal fluid absorption during exercise. J. Appl. Physiol. 1998, 85, 1941–1948. [Google Scholar] [CrossRef]
- Nose, H.; Mack, G.W.; Shi, X.R.; Nadel, E.R. Role of osmolality and plasma volume during rehydration in humans. J. Appl. Physiol. 1988, 65, 325–331. [Google Scholar] [CrossRef] [PubMed]
- Shirreffs, S.M.; Aragón, L.F.; Keil, M.; Love, T.D.; Phillips, S. Rehydration after exercise in the heat: A comparison of 4 commonly used drinks. Int. J. Sport Nutr. Exerc. Metab. 2007, 17, 244–258. [Google Scholar] [CrossRef] [PubMed]
- Maughan, R.J.; Leiper, J.B.; Shirreffs, S.M. Factors influencing the restoration of fluid and electrolyte balance after exercise in the heat. Br. J. Sports Med. 1997, 31, 175–182. [Google Scholar] [CrossRef] [PubMed]
- Maughan, R.J.; Watson, P.; Cordery, P.A.; Walsh, N.P.; Oliver, S.J.; Dolci, A.; Rodriguez-Sanchez, N.; Galloway, S.D. A randomized trial to assess the potential of different beverages to affect hydration status: Development of a beverage hydration index. Am. J. Clin. Nutr. 2015, 103, 717–723. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, B.; Sjøgaard, G.; Ugelvig, J.; Knudsen, B.; Dohlmann, B. Fluid balance in exercise dehydration and rehydration with different glucose-electrolyte drinks. Eur. J. Appl. Physiol. Occup. Physiol. 1986, 55, 318–325. [Google Scholar] [CrossRef]
- Shirreffs, S.M.; Maughan, R.J. Rehydration and recovery of fluid balance after exercise. Exerc. Sport Sci. Rev. 2000, 28, 27–32. [Google Scholar]
- Shirreffs, S.M.; Taylor, A.J.; Leiper, J.B.; Maughan, R.J. Post-exercise rehydration in man: Effects of volume consumed and drink sodium content. Med. Sci. Sports Exerc. 1996, 28, 1260–1271. [Google Scholar] [CrossRef]
- Clarke, M.M.; E Stanhewicz, A.; Wolf, S.T.; Cheuvront, S.N.; Kenefick, R.W.; Kenney, W.L. A randomized trial to assess beverage hydration index in healthy older adults. Am. J. Clin. Nutr. 2019, 109, 1640–1647. [Google Scholar] [CrossRef]
- Sollanek, K.J.; Tsurumoto, M.; Vidyasagar, S.; Kenefick, R.W.; Cheuvront, S.N. Neither body mass nor sex influences beverage hydration index outcomes during randomized trial when comparing 3 commercial beverages. Am. J. Clin. Nutr. 2018, 107, 544–549. [Google Scholar] [CrossRef]
- Wolf, S.T.; Stanhewicz, A.E.; Clarke, M.M.; Cheuvront, S.N.; Kenefick, R.W.; Kenney, W.L. Age-related differences in water and sodium handling after commercial hydration beverage ingestion. J. Appl. Physiol. 2019, 126, 1042–1048. [Google Scholar] [CrossRef]
- Roy, B.D. Milk: The new sports drink? A Review. J. Int. Soc. Sports Nutr. 2008, 5, 15. [Google Scholar] [CrossRef] [PubMed]
- Beucler, J.; Drake, M.; Foegeding, E.A. Design of a Beverage from Whey Permeate. J. Food Sci. 2006, 70, S277–S285. [Google Scholar] [CrossRef]
- El-Khair, A.A. Formulation of milk permeate for utilization as electrolyte beverages. Aust. J. Basic Appl. Sci. 2009, 3, 572–578. [Google Scholar]
- Geilman, W.; Schmidt, D.; Herfurth-Kennedy, C.; Path, J. Potential of milk permeate as an electrolyte beverage. J. Dairy Sci. 1990, 73 (Suppl. 1), 94. [Google Scholar]
- Geilman, W.G.; Schmidt, D.; Herfurth-Kennedy, C.; Path, J.; Cullor, J. Production of an Electrolyte Beverage from Milk Permeate. J. Dairy Sci. 1992, 75, 2364–2369. [Google Scholar] [CrossRef]
- Hill, T.; Lewicki, P.; Lewicki, P. Statistics: Methods and Applications: A Comprehensive Reference for Science, Industry, and Data Mining; StatSoft, Inc.: Tulsa, Oklahoma, 2006. [Google Scholar]
- Dill, D.B.; Costill, D.L. Calculation of percentage changes in volumes of blood, plasma, and red cells in dehydration. J. Appl. Physiol. 1974, 37, 247–248. [Google Scholar] [CrossRef]
- Desbrow, B.; Jansen, S.; Barrett, A.; Leveritt, M.; Irwin, C. Comparing the rehydration potential of different milk-based drinks to a carbohydrate–electrolyte beverage. Appl. Physiol. Nutr. Metab. 2014, 39, 1366–1372. [Google Scholar] [CrossRef]
- Volterman, K.A.; Obeid, J.; Wilk, B.; Timmons, B.W. Effect of milk consumption on rehydration in youth following exercise in the heat. Appl. Physiol. Nutr. Metab. 2014, 39, 1257–1264. [Google Scholar] [CrossRef]
- Maughan, R.J.; Leiper, J.B.; Vist, G.E. Gastric emptying and fluid availability after ingestion of glucose and soy protein hydrolysate solutions in man. Exp. Physiol. 2003, 89, 101–108. [Google Scholar] [CrossRef]
- Okabe, T.; Terashima, H.; Sakamoto, A. Determinants of liquid gastric emptying: Comparisons between milk and isocalorically adjusted clear fluids. Br. J. Anaesth. 2015, 114, 77–82. [Google Scholar] [CrossRef]
- Shi, X.; Osterberg, K.L.; Petrie, H.; Stofan, J.R.; Murray, R. Effect of Different Osmolalities, CHO Types, and [CHO] on Gastric Emptying in Humans. Med. Sci. Sports Exerc. 2017, 49, 1015–1021. [Google Scholar] [CrossRef] [PubMed]
- Hill, R.; Bluck, L.J.; Davies, P.S. The hydration ability of three commercially available sports drinks and water. J. Sci. Med. Sport 2008, 11, 116–123. [Google Scholar] [CrossRef] [PubMed]
- Nose, H.; Mack, G.W.; Shi, X.R.; Nadel, E.R. Shift in body fluid compartments after dehydration in humans. J. Appl. Physiol. 1988, 65, 318–324. [Google Scholar] [CrossRef] [PubMed]
- Cheuvront, S.N.; Kenefick, R.W.; Charkoudian, N.; Mitchell, K.M.; Luippold, A.J.; Bradbury, K.E.; Vidyasagar, S. Efficacy of Glucose or Amino Acid-Based Commercial Beverages in Meeting Oral Rehydration Therapy Goals After Acute Hypertonic and Isotonic Dehydration. J. Parenter. Enter. Nutr. 2018, 42, 1185–1193. [Google Scholar] [CrossRef]
- Girona, M.; Grasser, E.K.; Dulloo, A.G.; Montani, J.P. Cardiovascular and metabolic responses to tap water ingestion in young humans: Does the water temperature matter? Acta Physiol. 2014, 211, 358–370. [Google Scholar] [CrossRef]
- Boulze, D.; Montastruc, P.; Cabanac, M. Water intake, pleasure and water temperature in humans. Physiol. Behav. 1983, 30, 97–102. [Google Scholar] [CrossRef]
- Graham, C.; Morris, N.B.; Harwood, A.E.; Jay, O. Ad libitum water consumption off-sets the thermal and cardiovascular strain exacerbated by dehydration during a 3-h simulated heatwave. Eur. J. Appl. Physiol. 2019, 120, 391–399. [Google Scholar] [CrossRef]
- American College of Sports Medicine; Sawka, M.N.; Burke, L.M.; Eichner, E.R.; Maughan, R.J.; Montain, S.J.; Stachenfeld, N.S. American College of Sports Medicine Position Stand. Exercise and Fluid Replacement. Med. Sci. Sports Exerc. 2007, 39, 377–390. [Google Scholar] [CrossRef]
- Appelgren, B.H.; Thrasher, T.N.; Keil, L.C.; Ramsay, D.J. Mechanism of drinking-induced inhibition of vasopressin secretion in dehydrated dogs. Am. J. Physiol. Integr. Comp. Physiol. 1991, 261, R1226–R1233. [Google Scholar] [CrossRef]
- Figaro, M.K.; Mack, G.W. Regulation of fluid intake in dehydrated humans: Role of oropharyngeal stimulation. Am. J. Physiol. Content 1997, 272, R1740–R1746. [Google Scholar] [CrossRef]
- Mandelblat-Cerf, Y.; Kim, A.; Burgess, C.; Subramanian, S.; Tannous, B.A.; Lowell, B.B.; Andermann, M.L. Bidirectional Anticipation of Future Osmotic Challenges by Vasopressin Neurons. Neuron 2016, 93, 57–65. [Google Scholar] [CrossRef] [PubMed]
- Zimmerman, C.A.; Huey, E.L.; Ahn, J.S.; Beutler, L.; Tan, C.L.; Kosar, S.; Bai, L.; Chen, Y.; Corpuz, T.V.; Madisen, L.; et al. A gut-to-brain signal of fluid osmolarity controls thirst satiation. Nature 2019, 568, 98–102. [Google Scholar] [CrossRef] [PubMed]
- Baertschi, A.J.; Pence, R.A. Gut-brain signaling of water absorption inhibits vasopressin in rats. Am. J. Physiol. Integr. Comp. Physiol. 1995, 268, R236–R247. [Google Scholar] [CrossRef] [PubMed]
- Geelen, G.; Keil, L.C.; Kravik, S.E.; Wade, C.E.; Thrasher, T.N.; Barnes, P.R.; Pyka, G.; Nesvig, C.; Greenleaf, J.E. Inhibition of plasma vasopressin after drinking in dehydrated humans. Am. J. Physiol. Integr. Comp. Physiol. 1984, 247, R968–R971. [Google Scholar] [CrossRef]
- Criswell, D.; Renshler, K.; Powers, S.K.; Tulley, R.; Cicale, M. Fluid replacement beverages and maintenance of plasma volume during exercise: Role of aldosterone and vasopressin. Eur. J. Appl. Physiol. Occup. Physiol. 1992, 65, 445–451. [Google Scholar] [CrossRef] [PubMed]
- Storhaug, C.L.; Fosse, S.K.; Fadnes, L.T. Country, regional, and global estimates for lactose malabsorption in adults: A systematic review and meta-analysis. Lancet Gastroenterol. Hepatol. 2017, 2, 738–746. [Google Scholar] [CrossRef]
- James, L.J. Milk Protein and the Restoration of Fluid Balance after Exercise. Epidemiol. Pediatr. Sports Inj. 2012, 59, 120–126. [Google Scholar] [CrossRef]
- Shirreffs, S.M.; Watson, P.; Maughan, R.J. Milk as an effective post-exercise rehydration drink. Br. J. Nutr. 2007, 98, 173–180. [Google Scholar] [CrossRef]

| Mean | Range | |
|---|---|---|
| n (M/F) | 12 (6/6) | |
| Age (years) | 23 | 20–26 |
| Weight (kg) | 69.6 | 52.9–94.6 |
| BMI (kg·m−2) | 23.7 | 20.5–29.9 |
| Systolic BP (mmHg) | 116 | 100–130 |
| Diastolic BP (mmHg) | 75 | 62–82 |
| HR (beats·min−1) | 65 | 56–72 |
| Total cholesterol (mg·dL−1) | 169 | 123–264 |
| HDL-C (mg·dL−1) | 56 | 41–74 |
| LDL-C (mg·dL−1) | 94 | 56–192 |
| HbA1C (%) | 5.0 | 4.8–5.5 |
| Beverage | CHO (%) | Energy (kcal/L) | Sodium, mmol/L | Potassium, mmol/L | Osmolality, mosm/kg |
|---|---|---|---|---|---|
| Water | 0 | 0 | 0 | 0 | 0 |
| CES | 6 | 220 | 20 | 3.2 | 326 ± 3 |
| MPS | 4 | 160 | 21 | 28 | 621 ± 5 |
| Water | CES | MPS | |
|---|---|---|---|
| Mean ± SD | Mean ± SD | Mean ± SD | |
| Fasting glucose (mg·dL−1) | 87 ± 6 | 88 ± 5 | 88 ± 6 |
| Creatinine (mg·dL−1) | 0.86 ± 0.17 | 0.87 ± 0.18 | 0.89 ± 0.21 |
| Serum Na+ (mmol·L−1) | 138 ± 2 | 137 ± 2 | 138 ± 2 |
| Urine Na+ (mmol·L−1) | 84 ± 48 | 91 ± 79 | 75 ± 49 |
| Sosm (mOsm·kg−1) | 291 ± 5 | 291 ± 5 | 291 ± 5 |
| Uosm (mOsm·kg−1) | 511 ± 296 | 566 ± 301 | 574 ± 342 |
| Urine Specific Gravity | 1.015 ± 0.008 | 1.017 ± 0.008 | 1.017 ± 0.009 |
| Pre | 0 min | 30 min | 60 min | 120 min | 180 min | 240 min | |||
|---|---|---|---|---|---|---|---|---|---|
| Serum | |||||||||
| Urine | Sodium (mmol/L) | Water | 138 ± 2 | 136 ± 3 ‡ | 135 ± 2 ‡ | 136 ± 2 | 137 ± 2 | 138 ± 2 | 138 ± 3 |
| CES | 137 ± 2 | 136 ± 1 ‡ | 137 ± 2 * | 138 ± 1 * | 138 ± 1 | 138 ± 2 | 138 ± 1 | ||
| MPS | 138 ± 2 | 135 ± 5 ‡ | 137 ± 2 * | 136 ± 4 †‡ | 137 ± 1 | 138 ± 2 | 138 ± 2 | ||
| Potassium (mmol/L) | Water | 4.1 ± 0.1 | 4.3 ± 0.2 ‡ | 4.2 ± 0.3 | 4.3 ± 0.3 ‡ | 4.2 ± 0.4 ‡ | 4.1 ± 0.4 | 4.1 ± 0.3 | |
| CES | 4.0 ± 0.2 | 4.0 ± 0.3 * | 3.9 ± 0.2 * | 4.1 ± 0.2 * | 4.3 ± 0.2 ‡ | 4.4 ± 0.3 *‡ | 4.3 ± 0.3 *‡ | ||
| MPS | 4.0 ± 0.3 | 4.3 ± 0.4 †‡ | 4.4 ± 0.4 *†‡ | 4.4 ± 0.4 *†‡ | 4.5 ± 0.3 *†‡ | 4.4 ± 0.2 *‡ | 4.4 ± 0.2 *‡ | ||
| Osmolality (mosm/kg) | Water | 291 ± 5 | 288 ± 6 ‡ | 287 ± 6 ‡ | 287 ± 9 ‡ | 291 ± 6 | 291 ± 5 | 292 ± 6 | |
| CES | 291 ± 5 | 292 ± 4 * | 290 ± 5 * | 290 ± 4 * | 292 ± 3 | 292 ± 5 | 293 ± 5 | ||
| MPS | 291 ± 5 | 293 ± 6 *‡ | 293 ± 7 * | 294 ± 6 *†‡ | 293 ± 6 | 294 ± 3 ‡ | 293 ± 3 ‡ | ||
| Sodium (mmol/L) | Water | 72 ± 49 | 42 ± 36 | 28 ± 47 | 24 ± 19 | 62 ± 20 | 95 ± 42 | ||
| CES | 87 ± 73 | 38 ± 30 ‡ | 29 ± 37 ‡ | 26 ± 17 ‡ | 59 ± 30 ‡ | 103 ± 28 | |||
| MPS | 71 ± 50 | 61 ± 47 ‡ | 46 ± 32 ‡ | 78 ± 42 *†‡ | 100 ± 36 *†‡ | 115 ± 40 ‡ | |||
| Potassium (mmol/L) | Water | 28.5 ± 23.2 | 29.0 ± 18.2 | 10.2 ± 2.8 | 11.9 ± 6.8 | 29.6 ± 22.7 | 42.6 ± 33.5 | ||
| CES | 33.3 ± 20.8 | 21.4 ± 21.7 | 3.1 ± 1.5 ‡ | 10.2 ± 4.9 ‡ | 40.5 ± 46.7 | 55.3 ± 32.5 ‡ | |||
| MPS | 28.5 ± 20.7 | 32.3 ± 33.5 | 14.5 ± 13.5 ‡ | 35.5 ± 27.0 *†‡ | 51.3 ± 29.8 *‡ | 63.5 ± 33.3 *‡ | |||
| Osmolality (mosm/kg) | Water | 511 ± 282 | 308 ± 265 ‡ | 144 ± 217 ‡ | 113 ± 23 ‡ | 305 ± 305 ‡ | 465 ± 465 | ||
| CES | 564 ± 288 | 352 ± 298 ‡ | 71 ± 18 ‡ | 130 ± 45 ‡ | 309 ± 68 ‡ | 540 ± 115 | |||
| MPS | 563 ± 345 | 387 ± 305 ‡ | 226 ± 147 †‡ | 370 ± 166 *†‡ | 537 ± 138 *† | 635 ± 134 * | |||
| CH2O | Water | 3 ± 5 | 6 ± 2 | 4 ± 1 | 1 ± 2 | −1 ± 1 | |||
| (mL/min) | CES | 2 ± 4 | 7 ± 2 | 4 ± 2 | 0 ± 1 | −1 ± 0 | |||
| MPS | 1 ± 5 *† | 2 ± 3 *† | 0 ± 3 *† | −2 ± 1 * | −2 ± 1 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Berry, C.W.; Wolf, S.T.; Murray, B.; Kenney, W.L. Hydration Efficacy of a Milk Permeate-Based Oral Hydration Solution. Nutrients 2020, 12, 1502. https://doi.org/10.3390/nu12051502
Berry CW, Wolf ST, Murray B, Kenney WL. Hydration Efficacy of a Milk Permeate-Based Oral Hydration Solution. Nutrients. 2020; 12(5):1502. https://doi.org/10.3390/nu12051502
Chicago/Turabian StyleBerry, Craig W., S. Tony Wolf, Bob Murray, and W. Larry Kenney. 2020. "Hydration Efficacy of a Milk Permeate-Based Oral Hydration Solution" Nutrients 12, no. 5: 1502. https://doi.org/10.3390/nu12051502
APA StyleBerry, C. W., Wolf, S. T., Murray, B., & Kenney, W. L. (2020). Hydration Efficacy of a Milk Permeate-Based Oral Hydration Solution. Nutrients, 12(5), 1502. https://doi.org/10.3390/nu12051502




