Recipe for a Healthy Gut: Intake of Unpasteurised Milk Is Associated with Increased Lactobacillus Abundance in the Human Gut Microbiome
Abstract
1. Introduction
2. Methods
2.1. Study Site and Subjects
2.2. Subject Metadata
2.3. Diet Quantification
2.4. Faecal Sample Collection and 16S rRNA Gene Sequencing and Processing
2.5. Sequence Table Generation
2.6. Short Chain Fatty Acid (SCFA) Measurements
2.7. Statistical Analysis
3. Results
3.1. Participant Characteristics
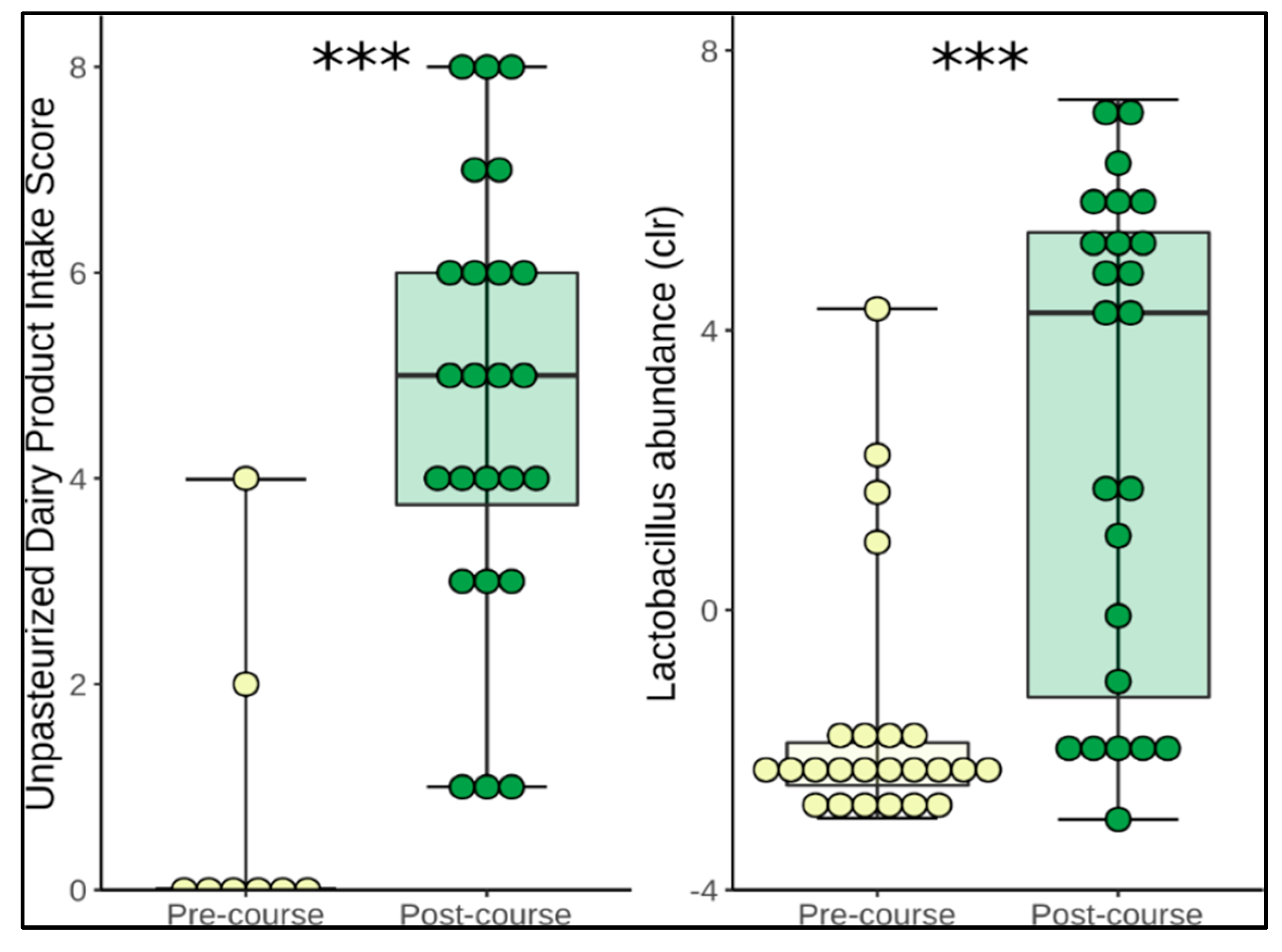
3.2. Changes in Diet
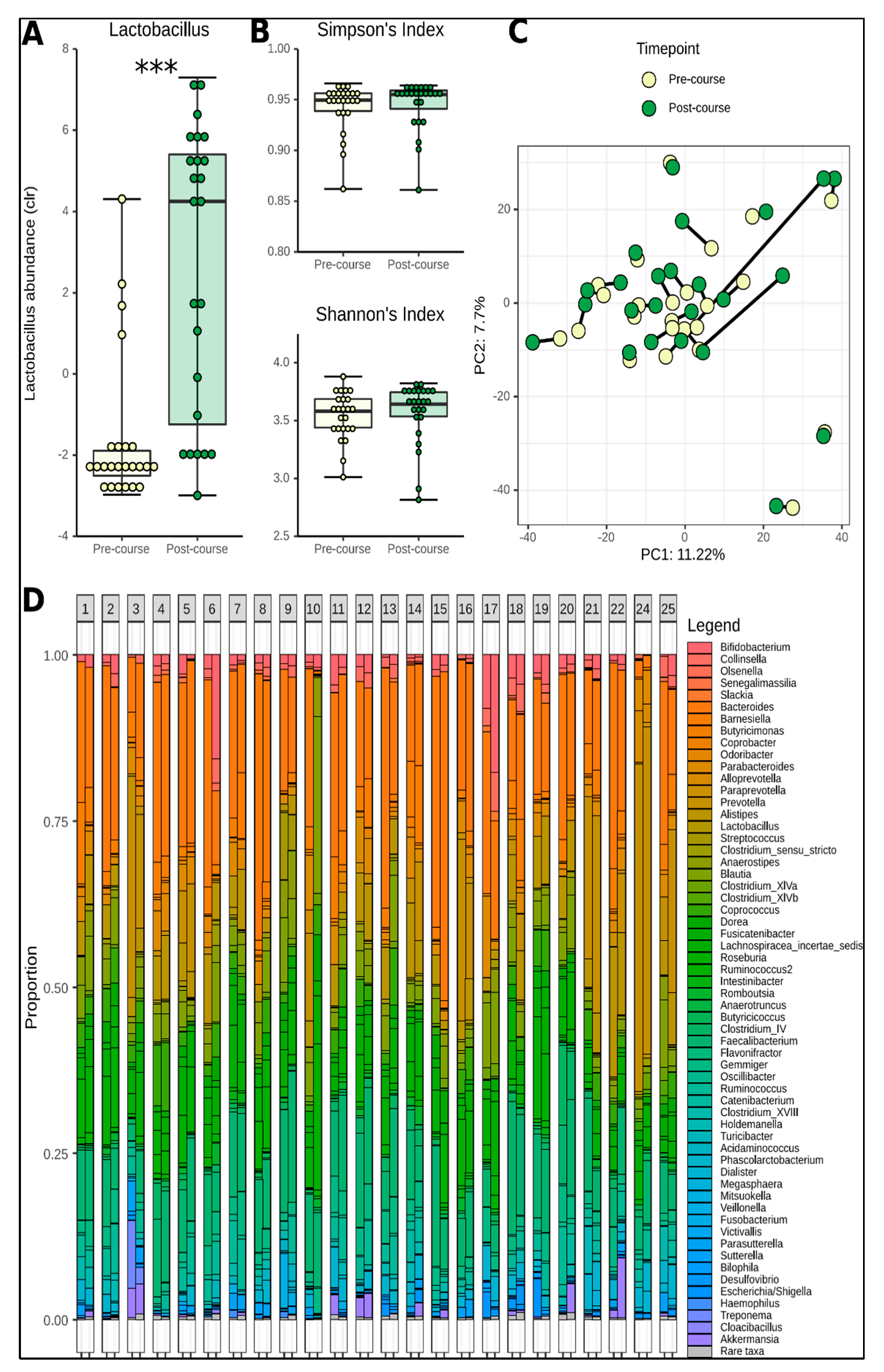
3.3. Change in Microbiome Composition
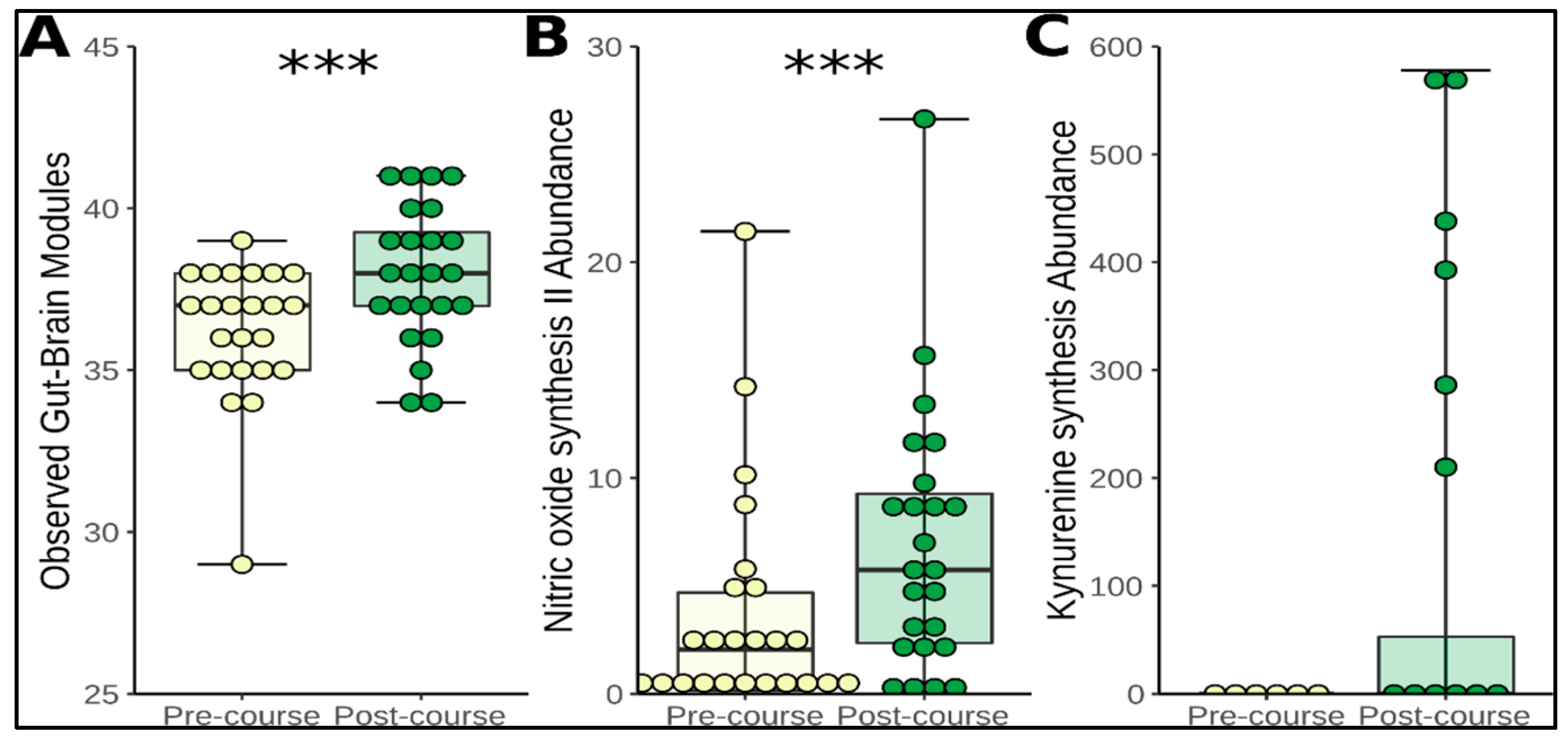
3.4. Functional Prediction and Application of Gut-Brain Modules
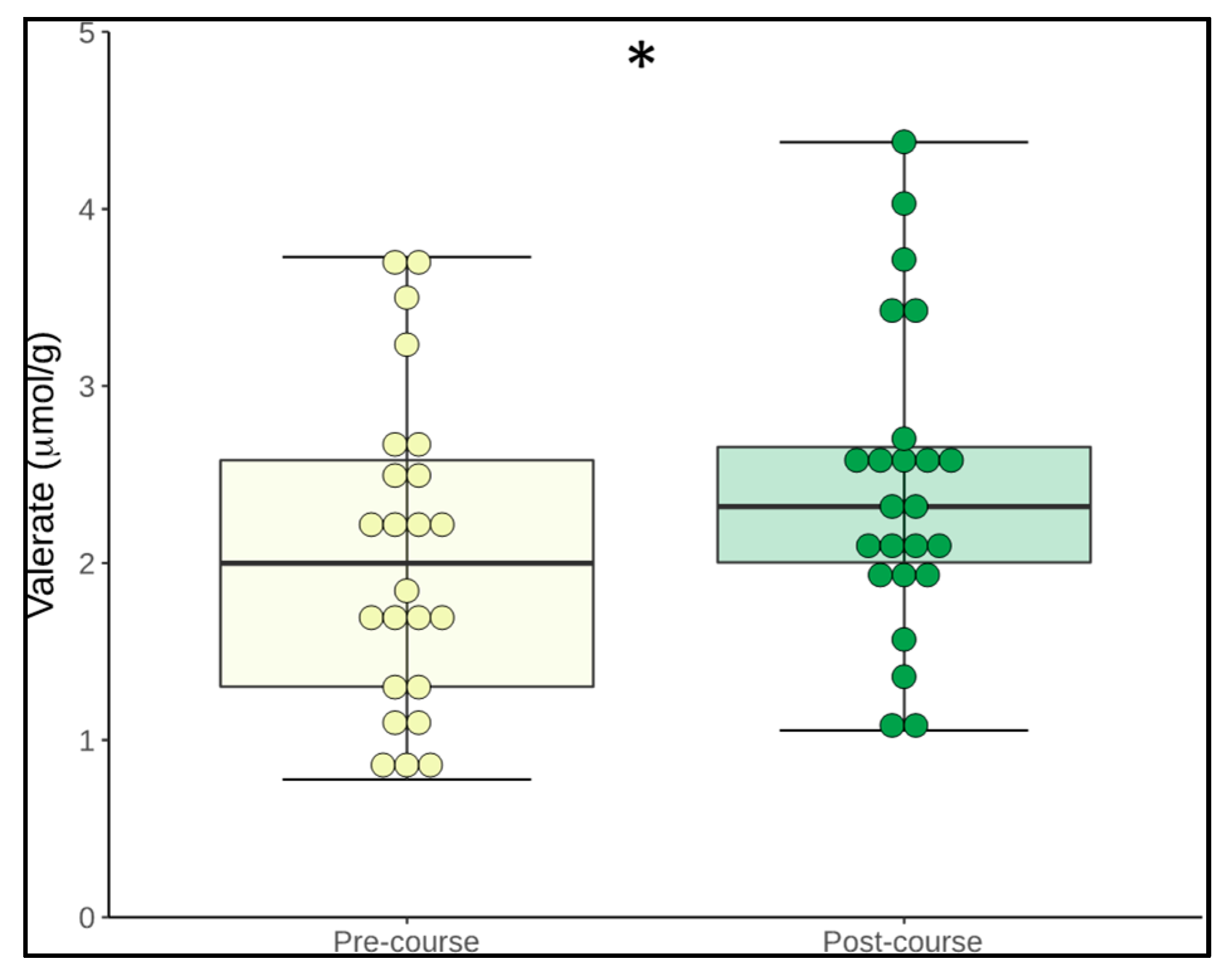
3.5. Change in Microbiome Metabolites
3.6. Change in Psychological Measures
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Di Cerbo, A.; Palmieri, B.; Aponte, M.; Morales-Medina, J.C.; Iannitti, T. Mechanisms and therapeutic effectiveness of lactobacilli. J. Clin. Pathol. 2016, 69, 187–203. [Google Scholar] [CrossRef] [PubMed]
- Hill, C.; Guarner, F.; Reid, G.; Gibson, G.R.; Merenstein, D.J.; Pot, B.; Morelli, L.; Canani, R.B.; Flint, H.J.; Salminen, S.; et al. Expert consensus document. The International Scientific Association for Probiotics and Prebiotics consensus statement on the scope and appropriate use of the term probiotic. Nat. Rev. Gastroenterol. Hepatol. 2014, 11, 506–514. [Google Scholar] [CrossRef] [PubMed]
- Dinan, T.G.; Stanton, C.; Cryan, J.F. Psychobiotics: A novel class of psychotropic. Biol. Psychiatry 2013, 74, 720–726. [Google Scholar] [CrossRef] [PubMed]
- John, G.K.; Wang, L.; Nanavati, J.; Twose, C.; Singh, R.; Mullin, G. Dietary Alteration of the Gut Microbiome and Its Impact on Weight and Fat Mass: A Systematic Review and Meta-Analysis. Genes 2018, 9, 167. [Google Scholar] [CrossRef]
- Hsieh, M.-C.; Tsai, W.-H.; Jheng, Y.-P.; Su, S.-L.; Wang, S.-Y.; Lin, C.-C.; Chen, Y.-H.; Chang, W.-W. The beneficial effects of Lactobacillus reuteri ADR-1 or ADR-3 consumption on type 2 diabetes mellitus: A randomized, double-blinded, placebo-controlled trial. Sci. Rep. 2018, 8. [Google Scholar] [CrossRef] [PubMed]
- Wong, V.W.; Won, G.L.; Chim, A.M.; Chu, W.C.; Yeung, D.K.; Li, K.C.; Chan, H.L. Treatment of nonalcoholic steatohepatitis with probiotics. A proof-of-concept study. Ann. Hepatol. 2013, 12, 256–262. [Google Scholar] [CrossRef]
- Di Rienzo, D.B. Effect of probiotics on biomarkers of cardiovascular disease: Implications for heart-healthy diets. Nutr. Rev. 2014, 72, 18–29. [Google Scholar] [CrossRef]
- Wilkins, T.; Sequoia, J. Probiotics for Gastrointestinal Conditions: A Summary of the Evidence. Am. Fam. Phys. 2017, 96, 170–178. [Google Scholar]
- Butler, M.I.; Cryan, J.F.; Dinan, T.G. Man and the Microbiome: A New Theory of Everything? Annu. Rev. Clin. Psychol. 2019, 15, 371–398. [Google Scholar] [CrossRef]
- Kolacek, S.; Hojsak, I.; Berni Canani, R.; Guarino, A.; Indrio, F.; Orel, R.; Pot, B.; Shamir, R.; Szajewska, H.; Vandenplas, Y.; et al. Commercial Probiotic Products: A Call for Improved Quality Control. A Position Paper by the ESPGHAN Working Group for Probiotics and Prebiotics. J. Pediatr. Gastroenterol. Nutr. 2017, 65, 117–124. [Google Scholar] [CrossRef]
- Johnson, A.J.; Vangay, P.; Al-Ghalith, G.A.; Hillmann, B.M.; Ward, T.L.; Shields-Cutler, R.R.; Kim, A.D.; Shmagel, A.K.; Syed, A.N.; Walter, J.; et al. Daily Sampling Reveals Personalized Diet-Microbiome Associations in Humans. Cell Host Microbe 2019, 25, 789–802. [Google Scholar] [CrossRef]
- Yatsunenko, T.; Rey, F.E.; Manary, M.J.; Trehan, I.; Dominguez-Bello, M.G.; Contreras, M.; Magris, M.; Hidalgo, G.; Baldassano, R.N.; Anokhin, A.P.; et al. Human gut microbiome viewed across age and geography. Nature 2012, 486, 222–227. [Google Scholar] [CrossRef]
- Clemente, J.C.; Pehrsson, E.C.; Blaser, M.J.; Sandhu, K.; Gao, Z.; Wang, B.; Magris, M.; Hidalgo, G.; Contreras, M.; Noya-Alarcon, O.; et al. The microbiome of uncontacted Amerindians. Sci. Adv. 2015, 1. [Google Scholar] [CrossRef]
- De Filippo, C.; Cavalieri, D.; Di Paola, M.; Ramazzotti, M.; Poullet, J.B.; Massart, S.; Collini, S.; Pieraccini, G.; Lionetti, P. Impact of diet in shaping gut microbiota revealed by a comparative study in children from Europe and rural Africa. Proc. Natl. Acad. Sci. USA 2010, 107, 14691–14696. [Google Scholar] [CrossRef]
- Jha, A.R.; Davenport, E.R.; Gautam, Y.; Bhandari, D.; Tandukar, S.; Ng, K.M.; Fragiadakis, G.K.; Holmes, S.; Gautam, G.P.; Leach, J.; et al. Gut microbiome transition across a lifestyle gradient in Himalaya. PLoS Biol. 2018, 16, e2005396. [Google Scholar] [CrossRef]
- Sonnenburg, E.D.; Sonnenburg, J.L. The ancestral and industrialized gut microbiota and implications for human health. Nat. Rev. Microbiol. 2019, 17, 383–390. [Google Scholar] [CrossRef]
- Central Statistics Office. Census 2016 Summary of Results; Central Statistics Office: Cork, Ireland, 2016. [Google Scholar]
- Braun-Fahrlander, C.; von Mutius, E. Can farm milk consumption prevent allergic diseases? Clin. Exp. Allergy 2011, 41, 29–35. [Google Scholar] [CrossRef]
- Buzby, J.C.; Hannah Gould, L.; Kendall, M.E.; Jones, T.F.; Robinson, T.; Blayney, D.P. Characteristics of Consumers of Unpasteurized Milk in the United States. J. Consum. Aff. 2013, 47, 153–166. [Google Scholar] [CrossRef]
- Fagnani, R.; Ito Eleodoro, J.; Osti Zanon, E. Milk-borne infections awareness and the health status of consumers: An on-line survey. Int. Dairy J. 2019, 96, 85–92. [Google Scholar] [CrossRef]
- Quigley, L.; McCarthy, R.; O’Sullivan, O.; Beresford, T.P.; Fitzgerald, G.F.; Ross, R.P.; Stanton, C.; Cotter, P.D. The microbial content of raw and pasteurized cow milk as determined by molecular approaches. J. Dairy Sci. 2013, 96, 4928–4937. [Google Scholar] [CrossRef]
- Tannock, G.W. A special fondness for lactobacilli. Appl. Environ. Microbiol. 2004, 70, 3189–3194. [Google Scholar] [CrossRef]
- Harrington, J.; Lutomski, J.; Morgan, K.; McGee, H.; Shelley, E.; Watson, D. SLÁN 2007: Survey of Lifestyle, Attitudes and Nutrition in Ireland. Dietary Habits of the Irish Population; Department of Health and Children: Dublin, Ireland, 2008.
- Riboli, E.; Kaaks, R. The EPIC Project: Rationale and study design. European Prospective Investigation into Cancer and Nutrition. Int. J. Epidemiol. 1997, 26, S6–S14. [Google Scholar] [CrossRef]
- Mulligan, A.A.; Luben, R.N.; Bhaniani, A.; Parry-Smith, D.J.; O’Connor, L.; Khawaja, A.P.; Forouhi, N.G.; Khaw, K.T. A new tool for converting food frequency questionnaire data into nutrient and food group values: FETA research methods and availability. BMJ Open 2014, 4, e004503. [Google Scholar] [CrossRef]
- Arthur, A.E.; Peterson, K.E.; Rozek, L.S.; Taylor, J.M.G.; Light, E.; Chepeha, D.B.; Hébert, J.R.; Terrell, J.E.; Wolf, G.T.; Duffy, S.A.; et al. Pretreatment dietary patterns, weight status, and head and neck squamous cell carcinoma prognosis. Am. J. Clin. Nutr. 2013, 97, 360–368. [Google Scholar] [CrossRef]
- Callahan, B.J.; McMurdie, P.J.; Rosen, M.J.; Han, A.W.; Johnson, A.J.; Holmes, S.P. DADA2: High-resolution sample inference from Illumina amplicon data. Nat. Methods 2016, 13, 581–583. [Google Scholar] [CrossRef]
- Hsieh, T.C.; Ma, K.H.; Chao, A. iNEXT: An R package for rarefaction and extrapolation of species diversity (Hill numbers). Methods Ecol. Evol. 2016, 7, 1451–1456. [Google Scholar] [CrossRef]
- Fernandes, A.D.; Reid, J.N.; Macklaim, J.M.; McMurrough, T.A.; Edgell, D.R.; Gloor, G.B. Unifying the analysis of high-throughput sequencing datasets: Characterizing RNA-seq, 16S rRNA gene sequencing and selective growth experiments by compositional data analysis. Microbiome 2014, 2, 15. [Google Scholar] [CrossRef]
- Iwai, S.; Weinmaier, T.; Schmidt, B.L.; Albertson, D.G.; Poloso, N.J.; Dabbagh, K.; DeSantis, T.Z. Piphillin: Improved Prediction of Metagenomic Content by Direct Inference from Human Microbiomes. PLoS ONE 2016, 11, e0166104. [Google Scholar] [CrossRef]
- Darzi, Y.; Falony, G.; Vieira-Silva, S.; Raes, J. Towards biome-specific analysis of meta-omics data. ISME J. 2016, 10, 1025–1028. [Google Scholar] [CrossRef]
- IBastiaanssen, T. Tjazi: Microbiome Oriented Compositional Data Toolkit. Available online: https://github.com/thomazbastiaanssen/Tjazi (accessed on 12 December 2019). [CrossRef]
- Dabney, A.; Storey, J.D.; Warnes, G.R. qvalue: Q-value estimation for false discovery rate control. R Package Vers. 2019, 2. [Google Scholar] [CrossRef]
- Valles-Colomer, M.; Falony, G.; Darzi, Y.; Tigchelaar, E.F.; Wang, J.; Tito, R.Y.; Schiweck, C.; Kurilshikov, A.; Joossens, M.; Wijmenga, C.; et al. The neuroactive potential of the human gut microbiota in quality of life and depression. Nat. Microbiol. 2019. [Google Scholar] [CrossRef]
- Govender, M.; Choonara, Y.E.; Kumar, P.; du Toit, L.C.; van Vuuren, S.; Pillay, V. A review of the advancements in probiotic delivery: Conventional vs. non-conventional formulations for intestinal flora supplementation. AAPS Pharm. Sci. Tech. 2014, 15, 29–43. [Google Scholar] [CrossRef] [PubMed]
- De Simone, C. The Unregulated Probiotic Market. Clin. Gastroenterol. Hepatol. 2019, 17, 809–817. [Google Scholar] [CrossRef] [PubMed]
- Sanders, M.E.; Klaenhammer, T.R.; Ouwehand, A.C.; Pot, B.; Johansen, E.; Heimbach, J.T.; Marco, M.L.; Tennila, J.; Ross, R.P.; Franz, C.; et al. Effects of genetic, processing, or product formulation changes on efficacy and safety of probiotics. Ann. N. Y. Acad. Sci. 2014, 1309, 1–18. [Google Scholar] [CrossRef]
- Cordain, L.; Eaton, S.B.; Sebastian, A.; Mann, N.; Lindeberg, S.; Watkins, B.A.; O’Keefe, J.H.; Brand-Miller, J. Origins and evolution of the Western diet: Health implications for the 21st century. Am. J. Clin. Nutr. 2005, 81, 341–354. [Google Scholar] [CrossRef]
- Zinocker, M.K.; Lindseth, I.A. The Western Diet-Microbiome-Host Interaction and Its Role in Metabolic Disease. Nutrients 2018, 10, 365. [Google Scholar] [CrossRef]
- Estruch, R.; Ros, E.; Salas-Salvadó, J.; Covas, M.-I.; Corella, D.; Arós, F.; Gómez-Gracia, E.; Ruiz-Gutiérrez, V.; Fiol, M.; Lapetra, J.; et al. Primary Prevention of Cardiovascular Disease with a Mediterranean Diet Supplemented with Extra-Virgin Olive Oil or Nuts. N. Engl. J. Med. 2018, 378, e34. [Google Scholar] [CrossRef]
- Lassale, C.; Batty, G.D.; Baghdadli, A.; Jacka, F.; Sanchez-Villegas, A.; Kivimaki, M.; Akbaraly, T. Healthy dietary indices and risk of depressive outcomes: A systematic review and meta-analysis of observational studies. Mol. Psychiatry 2018. [Google Scholar] [CrossRef]
- De Filippis, F.; Pellegrini, N.; Vannini, L.; Jeffery, I.B.; La Storia, A.; Laghi, L.; Serrazanetti, D.I.; Di Cagno, R.; Ferrocino, I.; Lazzi, C.; et al. High-level adherence to a Mediterranean diet beneficially impacts the gut microbiota and associated metabolome. Gut 2016, 65, 1812–1821. [Google Scholar] [CrossRef]
- David, L.A.; Maurice, C.F.; Carmody, R.N.; Gootenberg, D.B.; Button, J.E.; Wolfe, B.E.; Ling, A.V.; Devlin, A.S.; Varma, Y.; Fischbach, M.A.; et al. Diet rapidly and reproducibly alters the human gut microbiome. Nature 2014, 505, 559–563. [Google Scholar] [CrossRef]
- Haug, A.; Høstmark, A.T.; Harstad, O.M. Bovine milk in human nutrition—A review. Lipids Health Dis. 2007, 6, 25. [Google Scholar] [CrossRef] [PubMed]
- Quigley, L.; O’Sullivan, O.; Stanton, C.; Beresford, T.P.; Ross, R.P.; Fitzgerald, G.F.; Cotter, P.D. The complex microbiota of raw milk. FEMS Microbiol. Rev. 2013, 37, 664–698. [Google Scholar] [CrossRef] [PubMed]
- Raats, D.; Offek, M.; Minz, D.; Halpern, M. Molecular analysis of bacterial communities in raw cow milk and the impact of refrigeration on its structure and dynamics. Food Microbiol. 2011, 28, 465–471. [Google Scholar] [CrossRef] [PubMed]
- Rankin, S.A.; Bradley, R.L.; Miller, G.; Mildenhall, K.B. A 100-Year Review: A century of dairy processing advancements—Pasteurization, cleaning and sanitation, and sanitary equipment design. J. Dairy Sci. 2017, 100, 9903–9915. [Google Scholar] [CrossRef] [PubMed]
- Freedberg, D.E.; Toussaint, N.C.; Chen, S.P.; Ratner, A.J.; Whittier, S.; Wang, T.C.; Wang, H.H.; Abrams, J.A. Proton Pump Inhibitors Alter Specific Taxa in the Human Gastrointestinal Microbiome: A Crossover Trial. Gastroenterology 2015, 149, 883–885. [Google Scholar] [CrossRef]
- Imhann, F.; Vich Vila, A.; Bonder, M.J.; Lopez Manosalva, A.G.; Koonen, D.P.Y.; Fu, J.; Wijmenga, C.; Zhernakova, A.; Weersma, R.K. The influence of proton pump inhibitors and other commonly used medication on the gut microbiota. Gut Microbes 2017, 8, 351–358. [Google Scholar] [CrossRef]
- Lucey, J.A. Raw Milk Consumption: Risks and Benefits. Nutr. Today 2015, 50, 189–193. [Google Scholar] [CrossRef]
- Beals, T. Pilot Survey of Cow Share Consumer/Owners Lactose Intolerance Section. Available online: http://www.realmilk.com/health/lactose-intolerance-survey/ (accessed on 2 January 2020).
- Mummah, S.; Oelrich, B.; Hope, J.; Vu, Q.; Gardner, C.D. Effect of raw milk on lactose intolerance: A randomized controlled pilot study. Ann. Fam. Med. 2014, 12, 134–141. [Google Scholar] [CrossRef]
- Mullin, G.E.; Belkoff, S.M. Survey to determine why people drink raw milk. Glob. Adv. Health Med. 2014, 3, 19–24. [Google Scholar] [CrossRef]
- Macdonald, L.E.; Brett, J.; Kelton, D.; Majowicz, S.E.; Snedeker, K.; Sargeant, J.M. A systematic review and meta-analysis of the effects of pasteurization on milk vitamins, and evidence for raw milk consumption and other health-related outcomes. J. Food Prot. 2011, 74, 1814–1832. [Google Scholar] [CrossRef]
- Johansson, I.; Esberg, A.; Eriksson, L.; Haworth, S.; Lif Holgerson, P. Self-reported bovine milk intake is associated with oral microbiota composition. PLoS ONE 2018, 13, e0193504. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Yin, J.; Zhu, Y.; Wang, X.; Hu, X.; Bao, W.; Huang, Y.; Chen, L.; Chen, S.; Yang, W.; et al. Effects of Whole Milk Supplementation on Gut Microbiota and Cardiometabolic Biomarkers in Subjects with and without Lactose Malabsorption. Nutrients 2018, 10, 1403. [Google Scholar] [CrossRef] [PubMed]
- Herrero, C.; Granado, F.; Blanco, I.; Olmedilla, B. Vitamin A and E content in dairy products: Their contribution to the recommended dietary allowances (RDA) for elderly people. J. Nutr. Health Aging 2002, 6, 57–59. [Google Scholar] [PubMed]
- Herrero-Barbudo, C.; Olmedilla-Alonso, B.; Granado-Lorencio, F.; Blanco-Navarro, I. Bioavailability of vitamins A and E from whole and vitamin-fortified milks in control subjects. Eur. J. Nutr. 2006, 45, 391–398. [Google Scholar] [CrossRef] [PubMed]
- Manzi, P.; Di Costanzo, M.G.; Mattera, M. Updating Nutritional Data and Evaluation of Technological Parameters of Italian Milk. Foods 2013, 2, 254–273. [Google Scholar] [CrossRef]
- Schnorr, S.L.; Candela, M.; Rampelli, S.; Centanni, M.; Consolandi, C.; Basaglia, G.; Turroni, S.; Biagi, E.; Peano, C.; Severgnini, M.; et al. Gut microbiome of the Hadza hunter-gatherers. Nat. Commun. 2014, 5, 3654. [Google Scholar] [CrossRef]
- Martínez, I.; Stegen James, C.; Maldonado-Gómez Maria, X.; Eren, A.M.; Siba Peter, M.; Greenhill Andrew, R.; Walter, J. The Gut Microbiota of Rural Papua New Guineans: Composition, Diversity Patterns, and Ecological Processes. Cell Rep. 2015, 11, 527–538. [Google Scholar] [CrossRef]
- Thorsen, J.; McCauley, K.; Fadrosh, D.; Lynch, K.; Barnes, K.L.; Bendixsen, C.G.; Seroogy, C.M.; Lynch, S.V.; Gern, J.E. Evaluating the Effects of Farm Exposure on Infant Gut Microbiome. J. Allergy Clin. Immunol. 2019, 143, AB299. [Google Scholar] [CrossRef]
- He, L.; Prodhan, M.A.I.; Yuan, F.; Yin, X.; Lorkiewicz, P.K.; Wei, X.; Feng, W.; McClain, C.; Zhang, X. Simultaneous quantification of straight-chain and branched-chain short chain fatty acids by gas chromatography mass spectrometry. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2018, 1092, 359–367. [Google Scholar] [CrossRef]
- Tan, J.; McKenzie, C.; Potamitis, M.; Thorburn, A.N.; Mackay, C.R.; Macia, L. The role of short-chain fatty acids in health and disease. Adv. Immunol. 2014, 121, 91–119. [Google Scholar] [CrossRef]
- Van de Wouw, M.; Boehme, M.; Lyte, J.M.; Wiley, N.; Strain, C.; O’Sullivan, O.; Clarke, G.; Stanton, C.; Dinan, T.G.; Cryan, J.F. Short-chain fatty acids: Microbial metabolites that alleviate stress-induced brain-gut axis alterations. J. Physiol. 2018, 596, 4923–4944. [Google Scholar] [CrossRef] [PubMed]
- Francois, I.E.; Lescroart, O.; Veraverbeke, W.S.; Marzorati, M.; Possemiers, S.; Evenepoel, P.; Hamer, H.; Houben, E.; Windey, K.; Welling, G.W.; et al. Effects of a wheat bran extract containing arabinoxylan oligosaccharides on gastrointestinal health parameters in healthy adult human volunteers: A double-blind, randomised, placebo-controlled, cross-over trial. Br. J. Nutr. 2012, 108, 2229–2242. [Google Scholar] [CrossRef] [PubMed]
- Russell, W.R.; Gratz, S.W.; Duncan, S.H.; Holtrop, G.; Ince, J.; Scobbie, L.; Duncan, G.; Johnstone, A.M.; Lobley, G.E.; Wallace, R.J.; et al. High-protein, reduced-carbohydrate weight-loss diets promote metabolite profiles likely to be detrimental to colonic health. Am. J. Clin. Nutr. 2011, 93, 1062–1072. [Google Scholar] [CrossRef]
- Le Blanc, J.G.; Chain, F.; Martín, R.; Bermúdez-Humarán, L.G.; Courau, S.; Langella, P. Beneficial effects on host energy metabolism of short-chain fatty acids and vitamins produced by commensal and probiotic bacteria. Microbial. Cell Fact. 2017, 16, 79. [Google Scholar] [CrossRef] [PubMed]
- Bartolomaeus, H.; Balogh, A.; Yakoub, M.; Homann, S.; Markó, L.; Höges, S.; Tsvetkov, D.; Krannich, A.; Wundersitz, S.; Avery Ellen, G.; et al. Short-Chain Fatty Acid Propionate Protects From Hypertensive Cardiovascular Damage. Circulation 2019, 139, 1407–1421. [Google Scholar] [CrossRef] [PubMed]
- Chambers, E.S.; Viardot, A.; Psichas, A.; Morrison, D.J.; Murphy, K.G.; Zac-Varghese, S.E.K.; MacDougall, K.; Preston, T.; Tedford, C.; Finlayson, G.S.; et al. Effects of targeted delivery of propionate to the human colon on appetite regulation, body weight maintenance and adiposity in overweight adults. Gut 2015, 64, 1744–1754. [Google Scholar] [CrossRef]
- Demigne, C.; Morand, C.; Levrat, M.A.; Besson, C.; Moundras, C.; Remesy, C. Effect of propionate on fatty acid and cholesterol synthesis and on acetate metabolism in isolated rat hepatocytes. Br. J. Nutr. 1995, 74, 209–219. [Google Scholar] [CrossRef]
- Luu, M.; Pautz, S.; Kohl, V.; Singh, R.; Romero, R.; Lucas, S.; Hofmann, J.; Raifer, H.; Vachharajani, N.; Carrascosa, L.C.; et al. The short-chain fatty acid pentanoate suppresses autoimmunity by modulating the metabolic-epigenetic crosstalk in lymphocytes. Nat. Commun. 2019, 10, 1–12. [Google Scholar] [CrossRef]
- Li, Z.; Wang, W.; Liu, D.; Guo, Y. Effects of Lactobacillus acidophilus on gut microbiota composition in broilers challenged with Clostridium perfringens. PLoS ONE 2017, 12, e0188634. [Google Scholar] [CrossRef]
- McDonald, J.A.K.; Mullish, B.H.; Pechlivanis, A.; Liu, Z.; Brignardello, J.; Kao, D.; Holmes, E.; Li, J.V.; Clarke, T.B.; Thursz, M.R.; et al. Inhibiting Growth of Clostridioides difficile by Restoring Valerate, Produced by the Intestinal Microbiota. Gastroenterology 2018, 155, 1495–1507. [Google Scholar] [CrossRef]
- Liu, D.-M.; Wang, P.; Zhang, X.-Y.; Xu, X.-L.; Wu, H.; Li, L. Characterization of nitrite degradation by Lactobacillus casei subsp. rhamnosus LCR 6013. PLoS ONE 2014, 9, e93308. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Verstraete, W. Evaluation of nitric oxide production by lactobacilli. Appl. Microbiol. Biotechnol. 2001, 56, 504–507. [Google Scholar] [CrossRef] [PubMed]
- McLeod, T.M.; Lopez-Figueroa, A.L.; Lopez-Figueroa, M.O. Nitric oxide, stress, and depression. Psychopharmacol. Bull. 2001, 35, 24–41. [Google Scholar] [PubMed]
- Chiu, H.-C.; Levy, R.; Borenstein, E. Emergent Biosynthetic Capacity in Simple Microbial Communities. PLoS Comput. Biol. 2014, 10, e1003695. [Google Scholar] [CrossRef] [PubMed]
- Perisin, M.A.; Sund, C.J. Human gut microbe co-cultures have greater potential than monocultures for food waste remediation to commodity chemicals. Sci. Rep. 2018, 8, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Liu, R.T.; Walsh, R.F.L.; Sheehan, A.E. Prebiotics and probiotics for depression and anxiety: A systematic review and meta-analysis of controlled clinical trials. Neurosci. Biobehav. Rev. 2019, 102, 13–23. [Google Scholar] [CrossRef] [PubMed]
- Ng, Q.X.; Peters, C.; Ho, C.Y.X.; Lim, D.Y.; Yeo, W.S. A meta-analysis of the use of probiotics to alleviate depressive symptoms. J. Affect. Disord. 2018, 228, 13–19. [Google Scholar] [CrossRef]
- Bravo, J.A.; Forsythe, P.; Chew, M.V.; Escaravage, E.; Savignac, H.M.; Dinan, T.G.; Bienenstock, J.; Cryan, J.F. Ingestion of Lactobacillus strain regulates emotional behavior and central GABA receptor expression in a mouse via the vagus nerve. Proc. Natl. Acad. Sci. USA 2011, 108, 16050–16055. [Google Scholar] [CrossRef]
- Nishihira, J.; Kagami-Katsuyama, H.; Tanaka, A.; Nishimura, M.; Kobayashi, T.; Kawasaki, Y. Elevation of natural killer cell activity and alleviation of mental stress by the consumption of yogurt containing Lactobacillus gasseri SBT2055 and Bifidobacterium longum SBT2928 in a double-blind, placebo-controlled clinical trial. J. Funct. Foods 2014, 11, 261–268. [Google Scholar] [CrossRef]
- Messaoudi, M.; Lalonde, R.; Violle, N.; Javelot, H.; Desor, D.; Nejdi, A.; Bisson, J.F.; Rougeot, C.; Pichelin, M.; Cazaubiel, M.; et al. Assessment of psychotropic-like properties of a probiotic formulation (Lactobacillus helveticus R0052 and Bifidobacterium longum R0175) in rats and human subjects. Br. J. Nutr. 2011, 105, 755–764. [Google Scholar] [CrossRef]
- Takada, M.; Nishida, K.; Kataoka-Kato, A.; Gondo, Y.; Ishikawa, H.; Suda, K.; Kawai, M.; Hoshi, R.; Watanabe, O.; Igarashi, T.; et al. Probiotic Lactobacillus casei strain Shirota relieves stress-associated symptoms by modulating the gut-brain interaction in human and animal models. Neurogastroenterol. Motil. 2016, 28, 1027–1036. [Google Scholar] [CrossRef] [PubMed]
- Rao, A.V.; Bested, A.C.; Beaulne, T.M.; Katzman, M.A.; Iorio, C.; Berardi, J.M.; Logan, A.C. A randomized, double-blind, placebo-controlled pilot study of a probiotic in emotional symptoms of chronic fatigue syndrome. Gut Pathog. 2009, 1, 6. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.; Zhao, X.; Tang, S.; Huang, H.; Ning, Z.; Fu, X.; Zhang, C. Probiotics reduce psychological stress in patients before laryngeal cancer surgery. Asia Pac. J. Clin. Oncol. 2016, 12, e92–e96. [Google Scholar] [CrossRef] [PubMed]
- Weaver, C.M.; Miller, J.W. Challenges in conducting clinical nutrition research. Nutr. Rev. 2017, 75, 491–499. [Google Scholar] [CrossRef]
- Ruggles, K.V.; Wang, J.; Volkova, A.; Contreras, M.; Noya-Alarcon, O.; Lander, O.; Caballero, H.; Dominguez-Bello, M.G. Changes in the Gut Microbiota of Urban Subjects during an Immersion in the Traditional Diet and Lifestyle of a Rainforest Village. mSphere 2018, 3. [Google Scholar] [CrossRef]
- Suez, J.; Korem, T.; Zilberman-Schapira, G.; Segal, E.; Elinav, E. Non-caloric artificial sweeteners and the microbiome: Findings and challenges. Gut Microbes 2015, 6, 149–155. [Google Scholar] [CrossRef]
| Pre-Course | Post-Course | p-Value | |
|---|---|---|---|
| Number of participants | 24 | ||
| Female; n (%) | 13 (54) | ||
| Mean age; n (range) | 30.25 (18–59) | ||
| Smoking status; n (%) | 7 (29) | ||
| BMI (kg/m) | 24.87 (3.42) | 25.33 (3.61) | 0.1 |
| Physical activity (as measured by IPAQ score) | 4757.52 (4614.74) | 3271.52 (7280.05) | 0.32 |
| Sleep quality (as measured by PSQI) | 5.36 (2.87) | 4.95 (2.91) | 0.25 |
| Bristol stool scale score | 3.78 (1.085) | 4.04 (0.706) | 0.39 |
| GI-Visual Analogue Scale; Satisfaction with bowel habit | 38.37 (33.757) | 27.29 (27.98) | 0.25 |
| Nutrient | Recommended Daily Intake | Pre-Course | Post-Course | p-Value |
|---|---|---|---|---|
| Kilocalories | 2000–2400 (males; depending on activity level) | 2264 ± 1006 | 2723 ± 1494 | 0.47 |
| Protein (g) | 10%–35% of total energy | 97 ± 40 (17%) | 109 ± 57 (16%) | 0.54 |
| Fat (g) | 20%–35% of total calories | 94 ± 35 (37%) | 128 ± 66 (42%) | 0.08 |
| Carbohydrate (g) | 45%–65% of total calories | 246 ± 158 (43%) | 275 ± 178 (40%) | 0.77 |
| Alcohol (mL) | 21 standard drinks (1/2 pint of beer, small glass of wine, one measure of spirits) | 15 ± 13 | 14 ± 13 | 0.98 |
| Monounsaturated fatty acids (g) | >12% of total energy | 38 ± 17 (15%) | 51 ± 27 (17%) | 0.13 |
| Polyunsaturated fatty acids (g) | >6% of total energy | 16 ± 7 (6%) | 22 ± 13 (7%) | 0.20 |
| Saturated fatty acids (g) | <10% of total energy | 34 ± 13 (14%) | 49 ± 25 (16%) | 0.04 * |
| Cholesterol (mg) | 300 mg | 381 ± 173 | 469 ± 236 | 0.13 |
| Total sugar (g) | <10% of total energy | 115 ± 67 (20%) | 125 ± 76 (18%) | 0.81 |
| Starch (g) | 128 ± 96 | 147 ± 104 | 0.62 | |
| Fibre (g) | >25 g | 19 ± 15 | 20 ± 15 | 0.88 |
| Vitamin A (µg) | 800 µg | 715 ± 577 | 1505 ± 975 | 0.005 * |
| Thiamine (mg) | 1.1 mg | 1.8 ± 1.2 | 1.9 ± 1.2 | 0.87 |
| Riboflavin (mg) | 1.4 mg | 2.3 ± 1.5 | 2.6 ± 1.7 | 0.45 |
| Niacin (mg) | 16 mg | 27 ± 15 | 29 ± 19 | 1.00 |
| Vitamin B6 (mg) | 1.4 mg | 3.1 ± 3.0 | 2.9 ± 1.9 | 0.81 |
| Vitamin B12 (µg) | 2.5 µg | 7.8 ± 3.6 | 11 ± 5.8 | 0.04 * |
| Folate (µg) | 200 µg | 339 ± 284 | 328 ± 236 | 0.58 |
| Vitamin C (mg) | 80 mg | 104 ± 60 | 79 ± 41 | 0.16 |
| Vitamin D (µg) | 5 µg | 3.6 ± 2.3 | 5.1 ± 3.5 | 0.09 |
| Vitamin E (mg) | 12 mg | 14 ± 7 | 16.6 ± 9.7 | 0.41 |
| Phosphorous (mg) | 700 mg | 1612 ± 763 | 1787 ± 942 | 0.64 |
| Calcium (mg) | 1000 mg | 914 ± 387 | 1062 ± 548 | 0.34 |
| Iron (mg) | 7 mg | 15 ± 11 | 16 ± 11 | 0.81 |
| Selenium (µg) | 55 µg | 67 ± 30 | 79 ± 43 | 0.49 |
| Zinc (mg) | 10 mg | 12 ± 6 | 13 ± 7 | 0.58 |
| Sodium (mg) | 1600 mg | 2983 ± 1559 | 3385 ± 1964 | 0.59 |
| Potassium (mg) | 2000 mg | 3798 ± 1603 | 4015 ± 1958 | 0.85 |
| Magnesium (mg) | 375 mg | 359 ± 200 | 343 ± 189 | 0.67 |
| Copper (mg) | 1 mg | 1.2 ± 0.6 | 1.5 ± 0.8 | 0.31 |
| Chloride (mg) | 800 mg | 4407 ± 2407 | 4885 ± 2886 | 0.64 |
| Manganese (mg) | 2 mg | 3.1 ± 1.5 | 3.2 ± 1.8 | 0.85 |
| Iodine (µg) | 15 µg | 169 ± 76 | 201 ± 100 | 0.28 |
| Food Group | Pre-Course | Post-Course | p-Value |
|---|---|---|---|
| Red meats | 0.62 ± 0.35 | 0.85 ± 0.56 | 0.07 |
| Processed meats | 0.58 ± 0.77 | 0.33 ± 0.27 | 0.08 |
| Poultry | 0.31 ± 0.26 | 0.25 ± 0.22 | 0.73 |
| Organ meats | 0.04 ± 0.07 | 0.11 ± 0.09 | 0.01 * |
| Fish | 0.55 ± 0.43 | 0.68 ± 0.49 | 0.04 * |
| Fried foods | 0.21 ± 0.14 | 0.30 ± 0.16 | 0.03 * |
| Refined carbohydrates | 0.84 ± 0.56 | 1.28 ± 1.17 | 0.26 |
| Whole grains | 0.66 ± 0.41 | 0.95 ± 0.75 | 0.29 |
| Cereal | 0.69 ± 1.45 | 0.47 ± 0.55 | 0.76 |
| Potatoes | 0.34 ± 0.26 | 0.49 ± 0.34 | 0.06 |
| Pasta meals | 0.42 ± 0.31 | 0.34 ± 0.25 | 0.66 |
| High-fat dairy products | 2.03 ± 1.15 | 3.59 ± 3.07 | 0.09 |
| Low-fat dairy products | 0.21 ± 0.27 | 0.36 ± 0.28 | 0.02 * |
| Egg dishes | 0.61 ± 0.49 | 0.43 ± 0.29 | 0.34 |
| Fruit | 2.02 ± 1.2 | 1.38 ± 0.84 | 0.04 * |
| Green leafy vegetables | 0.77 ± 0.42 | 1.25 ± 1.03 | 0.11 |
| Cruciferous vegetables | 0.68 ± 0.61 | 0.44 ± 0.32 | 0.34 |
| Starchy vegetables | 0.42 ± 0.56 | 0.37 ± 0.24 | 0.16 |
| Other vegetables | 3.98 ± 2.17 | 3.72 ± 2.03 | 0.66 |
| Legumes | 0.30 ± 0.24 | 0.22 ± 0.25 | 0.10 |
| Sweets | 1.77 ± 1.29 | 2.45 ± 2.00 | 0.31 |
| Snacks | 0.48 ± 1.00 | 0.37 ± 0.66 | 0.40 |
| Soups | 1.15 ± 1.24 | 1.11 ± 1.17 | 0.48 |
| Sauces | 0.19 ± 0.17 | 0.27 ± 0.28 | 0.31 |
| Condiments | 2.68 ± 1.65 | 3.17 ± 2.12 | 0.58 |
| Non-alcoholic beverages | 2.39 ± 1.71 | 1.99 ± 1.42 | 0.45 |
| Alcoholic beverages | 1.43 ± 1.21 | 1.42 ± 1.29 | 0.91 |
| Fruit Juice | 0.42 ± 0.71 | 0.39 ± 0.53 | 0.50 |
| Sweetened beverages | 0.81 ± 1.03 | 0.78 ± 0.74 | 0.56 |
| Dairy Intake | Pre-Course | Post-Course | p-Value |
|---|---|---|---|
| Total Milk (mL) | 177 ± 120 mls | 192 ± 134 mls | 0.6 |
| High-fat dairy products (servings/day) | 2.03 ± 1.15 | 3.59 ± 3.07 | 0.09 |
| Low-fat dairy products (servings/day) | 0.21 ± 0.27 | 0.36 ± 0.28 | 0.02 * |
| Total dairy products | 2.24 ± 1.23 | 3.35 ± 3.16 | 0.07 |
| Unpasteurised milk (mL) | 23 ± 116 | 239 ± 51 | <0.0001 * |
| Unpasteurised dairy products (servings/day) | 0.01 ± 0.04 | 1.2 ± 1.4 | <0.0001 * |
| SCFA (µmol/g) | Pre-Course Mean (SD) | Post-Course Mean (SD) | p-Value |
|---|---|---|---|
| Acetate | 27.0 (8.6) | 29.3 (10.3) | 0.268 |
| Propionate | 14.0 (7.0) | 16.3 (7.6) | 0.091 |
| Iso-butyrate | 2.4 (1.1) | 2.6 (0.9) | 0.485 |
| Butyrate | 17.6 (9.6) | 19.0 (10.8) | 0.156 |
| Iso-valerate | 3.2 (1.9) | 3.4 (1.7) | 0.498 |
| Valerate | 2.3 (0.8) | 2.5 (0.8) | 0.049 * |
| Total BCFA | 5.6 (2.9) | 6.0 (2.5) | 0.44 |
| Total SCFA | 66.6 (24.7) | 73.1 (27.7) | 0.113 |
| Scale | Pre-Course Mean (SD) | Post-Course Mean (SD) | p-Value | |
|---|---|---|---|---|
| PSS | 14.96 (6.23) | 13.13 (5.12) | 0.149 | |
| HADS-A | 5.61 (3.72) | 4.83 (3.21) | 0.274 | |
| HADS-D | 3.04 (2.82) | 3.83 (3.42) | 0.198 | |
| HADS-T | 8.65 (5.37) | 8.65 (5.93) | 1 | |
| Subgroup Analysis | Pre-Course Mean (SD) | Post-Course Mean (SD) | Change (Delta) between Pre- and Post-Course Scores Mean (SD) | p-Value † |
| PSS-Highest | 19.58 (3.12) | 15.17(5.15) | −4.42 (5.9) | 0.026 * |
| PSS-Lowest | 10.08 (4.42) | 10.91 (4.25) | 1.0 (4.58) | |
| HADS-A-Highest | 7.67 (2.85) | 5.56(3.44) | −2.11 (2.30) | 0.004 * |
| HADS-A-Lowest | 1.75 (1.17) | 4 (2.35) | 2.25 (2.30) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Butler, M.I.; Bastiaanssen, T.F.S.; Long-Smith, C.; Berding, K.; Morkl, S.; Cusack, A.-M.; Strain, C.; Busca, K.; Porteous-Allen, P.; Claesson, M.J.; et al. Recipe for a Healthy Gut: Intake of Unpasteurised Milk Is Associated with Increased Lactobacillus Abundance in the Human Gut Microbiome. Nutrients 2020, 12, 1468. https://doi.org/10.3390/nu12051468
Butler MI, Bastiaanssen TFS, Long-Smith C, Berding K, Morkl S, Cusack A-M, Strain C, Busca K, Porteous-Allen P, Claesson MJ, et al. Recipe for a Healthy Gut: Intake of Unpasteurised Milk Is Associated with Increased Lactobacillus Abundance in the Human Gut Microbiome. Nutrients. 2020; 12(5):1468. https://doi.org/10.3390/nu12051468
Chicago/Turabian StyleButler, Mary I., Thomaz F. S. Bastiaanssen, Caitriona Long-Smith, Kirsten Berding, Sabrina Morkl, Anne-Marie Cusack, Conall Strain, Kizkitza Busca, Penny Porteous-Allen, Marcus J. Claesson, and et al. 2020. "Recipe for a Healthy Gut: Intake of Unpasteurised Milk Is Associated with Increased Lactobacillus Abundance in the Human Gut Microbiome" Nutrients 12, no. 5: 1468. https://doi.org/10.3390/nu12051468
APA StyleButler, M. I., Bastiaanssen, T. F. S., Long-Smith, C., Berding, K., Morkl, S., Cusack, A.-M., Strain, C., Busca, K., Porteous-Allen, P., Claesson, M. J., Stanton, C., Cryan, J. F., Allen, D., & Dinan, T. G. (2020). Recipe for a Healthy Gut: Intake of Unpasteurised Milk Is Associated with Increased Lactobacillus Abundance in the Human Gut Microbiome. Nutrients, 12(5), 1468. https://doi.org/10.3390/nu12051468





