COVID-19: The Inflammation Link and the Role of Nutrition in Potential Mitigation
Abstract
1. Introduction—Current Status
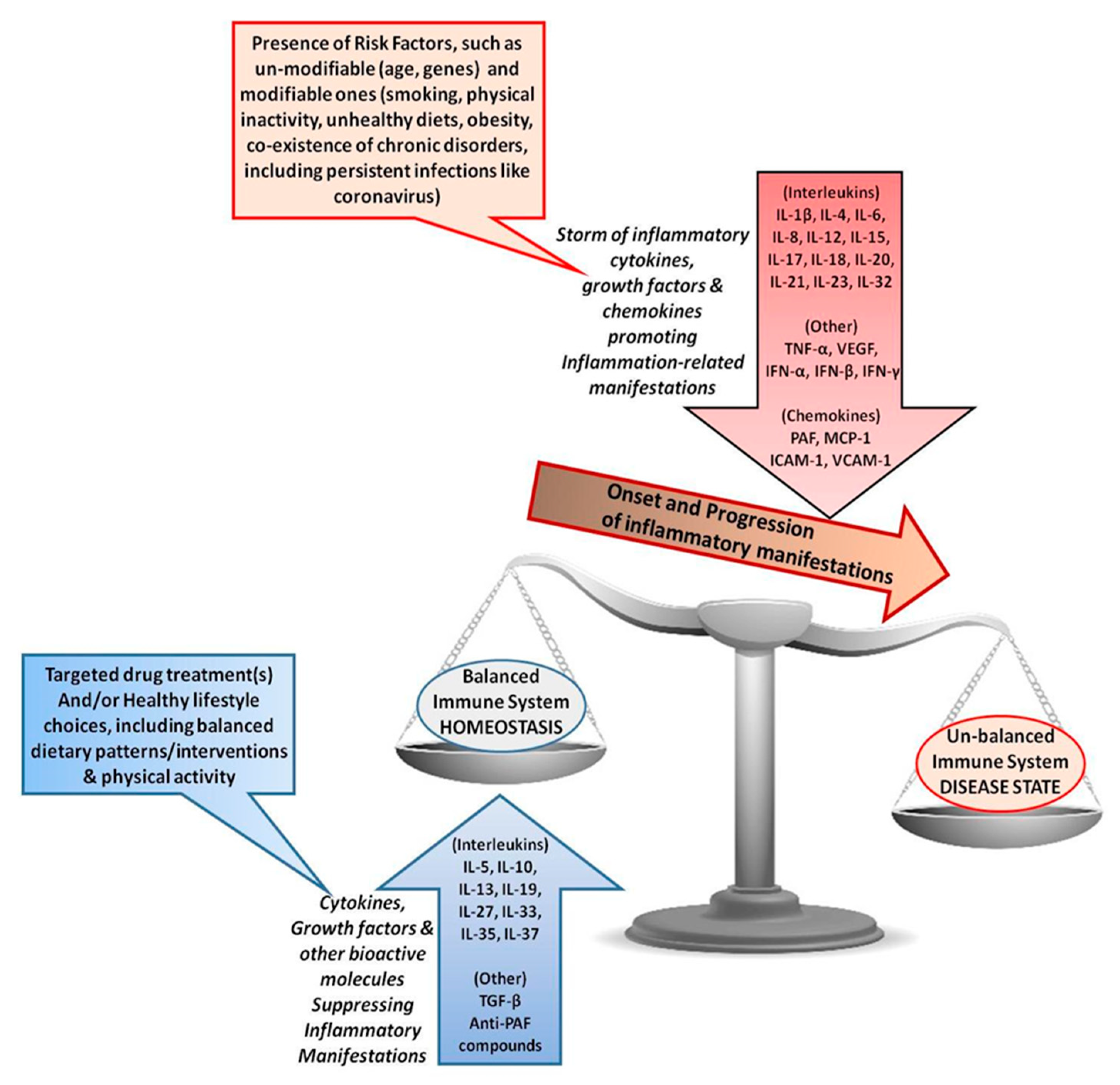
2. Mechanisms of the Uncontrolled Inflammatory Response and Cytokine Storm during Disease Progress
3. Current Knowledge of COVID-19 Treatment and Anti-Inflammatory Approaches
4. Noncommunicable Diseases and COVID-19—The Link between Immunity and Nutrition
5. Immunomodulatory and Anti-Inflammatory Potential of Maintaining a Healthy Nutritional Status
5.1. Dietary Patterns
5.1.1. Mediterranean Diet
5.1.2. Western Diet
5.2. Foods, Food Groups, and Individual Nutrients
5.2.1. Fruit and Vegetables
5.2.2. Fish and Fish Oils
5.2.3. Vitamin C
- Reduced cold severity: They reduced the symptoms of a cold, making it less severe.
- Reduced cold duration: Supplements decreased the recovery time by 8% in adults and 14% in children, on average [187].
5.2.4. Vitamin D
5.2.5. Vitamin E
5.2.6. Zinc
5.2.7. Copper
5.2.8. Fiber
5.3. General Dietary Advice during the COVID-19 Pandemic
6. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- John Hopkins University. John Hopkins University & Medicine: Coronavirus Resource Center. Available online: https://coronavirus.jhu.edu/map.html (accessed on 10 May 2020).
- Wang, L.-S.; Wang, Y.-R.; Ye, D.-W.; Liu, Q.-Q. A review of the 2019 novel coronavirus (COVID-19) based on current evidence. Int. J. Antimicrob. Agents 2020, in press. [Google Scholar] [CrossRef] [PubMed]
- Lai, C.-C.; Shih, T.-P.; Ko, W.-C.; Tang, H.-J.; Hsueh, P.-R. Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) and coronavirus disease-2019 (COVID-19): The epidemic and the challenges. Int. J. Antimicrob. Agents 2020, 55, 105924. [Google Scholar] [CrossRef] [PubMed]
- Zhou, P.; Yang, X.-L.; Wang, X.-G.; Hu, B.; Zhang, L.; Zhang, W.; Si, H.-R.; Zhu, Y.; Li, B.; Huang, C.-L.; et al. A pneumonia outbreak associated with a new coronavirus of probable bat origin. Nature 2020, 579, 270–273. [Google Scholar] [CrossRef] [PubMed]
- Wan, Y.; Shang, J.; Graham, R.; Baric, R.S.; Li, F. Receptor recognition by the novel coronavirus from wuhan: An analysis based on decade-long structural studies of SARS coronavirus. J. Virol. 2020, 94, e00120–e00127. [Google Scholar] [CrossRef]
- Yan, R.; Zhang, Y.; Li, Y.; Xia, L.; Guo, Y.; Zhou, Q. Structural basis for the recognition of SARS-CoV-2 by full-length human ACE2. Science 2020, 367, 1444–1448. [Google Scholar] [CrossRef]
- Gheblawi, M.; Wang, K.; Viveiros, A.; Nguyen, Q.; Zhong, J.-C.; Turner, A.J.; Raizada, M.K.; Grant, M.B.; Oudit, G.Y. Angiotensin converting enzyme 2: SARS-CoV-2 receptor and regulator of the renin-angiotensin system. Circ. Res. 2020, 126, 1456–1474. [Google Scholar] [CrossRef]
- Chen, L.; Hao, G. The role of angiotensin-converting enzyme 2 in coronaviruses/influenza viruses and cardiovascular disease. Cardiovasc. Res. 2020, cvaa093. [Google Scholar] [CrossRef]
- Touyz, R.M.; Li, H.; Delles, C. Ace2 the janus-faced protein—From cardiovascular protection to severe acute respiratory syndrome-coronavirus and covid-19. Clin. Sci. (Lond. Engl. 1979) 2020, 134, 747–750. [Google Scholar] [CrossRef]
- Li, G.; Fan, Y.; Lai, Y.; Han, T.; Li, Z.; Zhou, P.; Pan, P.; Wang, W.; Hu, D.; Liu, X.; et al. Coronavirus infections and immune responses. J. Med. Virol. 2020, 92, 424–432. [Google Scholar] [CrossRef]
- Zhang, W.; Zhao, Y.; Zhang, F.; Wang, Q.; Li, T.; Liu, Z.; Wang, J.; Qin, Y.; Zhang, X.; Yan, X.; et al. The use of anti-inflammatory drugs in the treatment of people with severe coronavirus disease 2019 (COVID-19): The perspectives of clinical immunologists from China. Clin. Immunol. 2020, 214, 108393. [Google Scholar] [CrossRef]
- Conti, P.; Ronconi, G.; Caraffa, A.; Gallenga, C.E.; Ross, R.; Frydas, I.; Kritas, S.K. Induction of pro-inflammatory cytokines (IL-1 and IL-6) and lung inflammation by coronavirus-19 (COVID-19 or SARS-CoV-2): Anti-inflammatory strategies. J. Biol. Regul. Homeost. Agents 2020, 34, 1. [Google Scholar] [PubMed]
- Jiang, F.; Deng, L.; Zhang, L.; Cai, Y.; Cheung, C.W.; Xia, Z. Review of the clinical characteristics of coronavirus disease 2019 (COVID-19). J. Gen. Intern. Med. 2020, 35, 1545–1549. [Google Scholar] [CrossRef] [PubMed]
- Wu, Z.; McGoogan, J.M. Characteristics of and important lessons from the coronavirus disease 2019 (COVID-19) outbreak in China: Summary of a report of 72,314 cases from the Chinese center for disease control and prevention. JAMA 2020, 323, 1239–1242. [Google Scholar] [CrossRef] [PubMed]
- Xu, Z.; Shi, L.; Wang, Y.; Zhang, J.; Huang, L.; Zhang, C.; Liu, S.; Zhao, P.; Liu, H.; Zhu, L.; et al. Pathological findings of COVID-19 associated with acute respiratory distress syndrome. Lancet Respir. Med. 2020, 8, 420–422. [Google Scholar] [CrossRef]
- Zhou, F.; Yu, T.; Du, R.; Fan, G.; Liu, Y.; Liu, Z.; Xiang, J.; Wang, Y.; Song, B.; Gu, X.; et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: A retrospective cohort study. Lancet 2020, 395, 1054–1062. [Google Scholar] [CrossRef]
- Kritas, S.K.; Ronconi, G.; Caraffa, A.; Gallenga, C.E.; Ross, R.; Conti, P. Mast cells contribute to coronavirus-induced inflammation: New anti-inflammatory strategy. J. Biol. Regul. Homeost. Agents 2020, 34, 34. [Google Scholar]
- Mehta, P.; McAuley, D.F.; Brown, M.; Sanchez, E.; Tattersall, R.S.; Manson, J.J. COVID-19: Consider cytokine storm syndromes and immunosuppression. Lancet 2020, 395, 1033–1034. [Google Scholar] [CrossRef]
- Qin, C.; Zhou, L.; Hu, Z.; Zhang, S.; Yang, S.; Tao, Y.; Xie, C.; Ma, K.; Shang, K.; Wang, W.; et al. Dysregulation of immune response in patients with COVID-19 in Wuhan, China. Clin. Infect. Dis. 2020, ciaa248. [Google Scholar] [CrossRef]
- de Masson, A.; Bouaziz, J.-D.; Sulimovic, L.; Cassius, C.; Jachiet, M.; Ionescu, M.-A.; Rybojad, M.; Bagot, M.; Duong, T.-A. Chilblains are a common cutaneous finding during the COVID-19 pandemic: A retrospective nationwide study from France. J. Am. Acad. Dermatol. 2020, in press. [Google Scholar] [CrossRef]
- Lechien, J.R.; Chiesa-Estomba, C.M.; De Siati, D.R.; Horoi, M.; Le Bon, S.D.; Rodriguez, A.; Dequanter, D.; Blecic, S.; El Afia, F.; Distinguin, L.; et al. Olfactory and gustatory dysfunctions as a clinical presentation of mild-to-moderate forms of the coronavirus disease (COVID-19): A multicenter european study. Eur. Arch. Otorhinolaryngol. 2020. Available online: https://link.springer.com/article/10.1007%2Fs00405-020-05965-1 (accessed on 1 April 2020). [CrossRef]
- Avula, A.; Nalleballe, K.; Narula, N.; Sapozhnikov, S.; Dandu, V.; Toom, S.; Glaser, A.; Elsayegh, D. COVID-19 presenting as stroke. Brain. Behav. Immun. 2020, in press. [Google Scholar] [CrossRef] [PubMed]
- NIAID-RML. New Images of Novel Coronavirus SARS-CoV-2 Now Available. Available online: https://www.niaid.nih.gov/news-events/novel-coronavirus-sarscov2-images (accessed on 9 May 2020).
- Li, T.; Lu, H.; Zhang, W. Clinical observation and management of COVID-19 patients. Emerg. Microbes Infect. 2020, 9, 687–690. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. COVID-19 and NCDs. Available online: https://www.who.int/internal-publications-detail/covid-19-and-ncds (accessed on 23 March 2020).
- World Health Organization. Coronavirus Disease 2019 (COVID-19) Situation Report-72. Available online: https://www.who.int/docs/default-source/coronaviruse/situation-reports/20200401-sitrep-72-covid-19.pdf?sfvrsn=3dd8971b_2 (accessed on 1 April 2020).
- Cascella, M.; Rajnik, M.; Cuomo, A.; Dulebohn, S.C.; Di Napoli, R. Features, evaluation and treatment coronavirus (COVID-19). In Statpearls [Internet]; StatPearls Publishing: Treasure Island, FL, USA, 2020. [Google Scholar]
- Cheng, V.C.-C.; Wong, S.-C.; Chuang, V.W.-M.; So, S.Y.-C.; Chen, J.H.-K.; Sridhar, S.; To, K.K.-W.; Chan, J.F.-W.; Hung, I.F.-N.; Ho, P.-L.; et al. The role of community-wide wearing of face mask for control of coronavirus disease 2019 (COVID-19) epidemic due to SARS-CoV-2. J. Infect. 2020, in press. [Google Scholar] [CrossRef] [PubMed]
- Centers for Disease Control and Prevention. How to Protect Yourself & Others. Available online: https://www.cdc.gov/coronavirus/2019-ncov/prevent-getting-sick/prevention.html (accessed on 1 April 2020).
- Park, S.W.; Sun, K.; Viboud, C.; Grenfell, B.T.; Dushoff, J. Potential roles of social distancing in mitigating the spread of coronavirus disease 2019 (COVID-19) in South Korea. medRxiv 2020, 2020, 20045815. [Google Scholar]
- Matrajt, L.; Leung, T. Evaluating the effectiveness of social distancing interventions against COVID-19. medRxiv 2020, 2020, 20044891. [Google Scholar]
- Wu, D.; Lewis, E.D.; Pae, M.; Meydani, S.N. Nutritional modulation of immune function: Analysis of evidence, mechanisms, and clinical relevance. Front. Immunol. 2019, 9, 9. [Google Scholar] [CrossRef]
- Grant, W.B.; Lahore, H.; McDonnell, S.L.; Baggerly, C.A.; French, C.B.; Aliano, J.L.; Bhattoa, H.P. Evidence that vitamin d supplementation could reduce risk of influenza and COVID-19 infections and deaths. Nutrients 2020, 12, 988. [Google Scholar] [CrossRef]
- Childs, C.E.; Calder, P.C.; Miles, E.A. Diet and immune function. Nutrients 2019, 11, 1933. [Google Scholar] [CrossRef]
- Calder, P.C.; Carr, A.C.; Gombart, A.F.; Eggersdorfer, M. Optimal nutritional status for a well-functioning immune system is an important factor to protect against viral infections. Preprints 2020, 12, 1181. [Google Scholar]
- Hoffmann, M.; Kleine-Weber, H.; Schroeder, S.; Krüger, N.; Herrler, T.; Erichsen, S.; Schiergens, T.S.; Herrler, G.; Wu, N.-H.; Nitsche, A.; et al. SARS-CoV-2 Cell Entry Depends on ACE2 and TMPRSS2 and Is Blocked by a Clinically Proven Protease Inhibitor. Cell 2020, 181, 271–280.e8. [Google Scholar] [CrossRef]
- Sommerstein, R.; Kochen, M.M.; Messerli, F.H.; Gräni, C. Coronavirus disease 2019 (COVID-19): Do angiotensin-converting enzyme inhibitors/angiotensin receptor blockers have a biphasic effect? J. Am. Heart Assoc. 2020, 9, e016509. [Google Scholar] [CrossRef] [PubMed]
- Dai, X.; Hakizimana, O.; Zhang, X.; Kaushik, A.C.; Zhang, J. Orchestrated efforts on host network hijacking: Processes governing virus replication. Virulence 2020, 11, 183–198. [Google Scholar] [CrossRef] [PubMed]
- Akira, S. Pathogen recognition by innate immunity and its signaling. Proc. Jpn. Acad. 2009, 85, 143–156. [Google Scholar] [CrossRef] [PubMed]
- Pennock, N.D.; White, J.T.; Cross, E.W.; Cheney, E.E.; Tamburini, B.A.; Kedl, R.M. T cell responses: Naive to memory and everything in between. Adv. Physiol. Educ. 2013, 37, 273–283. [Google Scholar] [CrossRef] [PubMed]
- Cecere, T.E.; Todd, S.M.; Leroith, T. Regulatory t cells in arterivirus and coronavirus infections: Do they protect against disease or enhance it? Viruses 2012, 4, 833–846. [Google Scholar] [CrossRef] [PubMed]
- Julkunen, I.; Melén, K.; Nyqvist, M.; Pirhonen, J.; Sareneva, T.; Matikainen, S. Inflammatory responses in influenza a virus infection. Vaccine 2000, 19, S32–S37. [Google Scholar] [CrossRef]
- Huang, C.; Wang, Y.; Li, X.; Ren, L.; Zhao, J.; Hu, Y.; Zhang, L.; Fan, G.; Xu, J.; Gu, X.; et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet 2020, 395, 497–506. [Google Scholar] [CrossRef]
- Siddiqi, H.K.; Mehra, M.R. COVID-19 illness in native and immunosuppressed states: A clinical-therapeutic staging proposal. J. Heart Lung Transplant. 2020, 39, 405–407. [Google Scholar] [CrossRef]
- Blanco-Melo, D.; Nilsson-Payant, B.E.; Liu, W.C.; Uhl, S.; Hoagland, D.; Møller, R.; Jordan, T.X.; Oishi, K.; Pais, M.; Sachs, D.; et al. Imbalanced host response to SARS-CoV-2 drives development of COVID-19. Cell 2020. [Google Scholar] [CrossRef]
- Wadman, M.; Couzin-Frankel, J.; Kaiser, J.; Matacic, C. A rampage through the body. Science 2020, 368, 356–360. [Google Scholar]
- Klok, F.A.; Kruip, M.J.H.A.; van der Meer, N.J.M.; Arbous, M.S.; Gommers, D.A.M.P.J.; Kant, K.M.; Kaptein, F.H.J.; van Paassen, J.; Stals, M.A.M.; Huisman, M.V.; et al. Incidence of thrombotic complications in critically ill ICU patients with COVID-19. Thromb. Res. 2020. Available online: https://www.ncbi.nlm.nih.gov/pubmed/32291094 (accessed on 1 April 2020). [CrossRef] [PubMed]
- Elfiky, A.A. Ribavirin, Remdesivir, Sofosbuvir, Galidesivir, and Tenofovir against SARS-CoV-2 RNA dependent rna polymerase (RDRP): A molecular docking study. Life Sci. 2020, 253, 117592. [Google Scholar] [CrossRef] [PubMed]
- Papakonstantinou, V.D.; Chini, M.; Mangafas, N.; Stamatakis, G.M.; Tsogas, N.; Tsoupras, A.B.; Psarra, K.; Fragopoulou, E.; Antonopoulou, S.; Gargalianos, P.; et al. In vivo effect of two first-line art regimens on inflammatory mediators in male HIV patients. Lipids Health Dis. 2014, 13, 90. [Google Scholar] [CrossRef] [PubMed]
- Tsoupras, A.B.; Chini, M.; Tsogas, N.; Fragopoulou, E.; Nomikos, T.; Lioni, A.; Mangafas, N.; Demopoulos, C.A.; Antonopoulou, S.; Lazanas, M.C. Anti-platelet-activating factor effects of highly active antiretroviral therapy (HAART): A new insight in the drug therapy of HIV infection? AIDS Res. Hum. Retrovir. 2008, 24, 1079–1086. [Google Scholar] [CrossRef]
- Fedson, D.S. Confronting the next influenza pandemic with anti-inflammatory and immunomodulatory agents: Why they are needed and how they might work. Influenza Other Respir. Viruses 2009, 3, 129–142. [Google Scholar] [CrossRef]
- Ritchie, A.I.; Singanayagam, A. Immunosuppression for hyperinflammation in COVID-19: A double-edged sword? Lancet 2020, 395, 1111. [Google Scholar] [CrossRef]
- Thomas, B.J.; Porritt, R.A.; Hertzog, P.J.; Bardin, P.G.; Tate, M.D. Glucocorticosteroids enhance replication of respiratory viruses: Effect of adjuvant interferon. Sci. Rep. 2014, 4, 7176. [Google Scholar] [CrossRef]
- Samuel, C.E. Antiviral actions of interferons. Clin. Microbiol. Rev. 2001, 14, 778–809. [Google Scholar] [CrossRef]
- Zhang, C.; Huang, S.; Zheng, F.; Dai, Y. Controversial treatments: An updated understanding of the coronavirus disease 2019. J. Med. Virol. 2020. [Google Scholar] [CrossRef]
- Colson, P.; Rolain, J.-M.; Lagier, J.-C.; Brouqui, P.; Raoult, D. Chloroquine and hydroxychloroquine as available weapons to fight COVID-19. Int. J. Antimicrob. Agents 2020, 55, 105932. [Google Scholar] [CrossRef]
- Wang, M.; Cao, R.; Zhang, L.; Yang, X.; Liu, J.; Xu, M.; Shi, Z.; Hu, Z.; Zhong, W.; Xiao, G. Remdesivir and Chloroquine effectively inhibit the recently emerged novel coronavirus (2019-nCoV) in vitro. Cell Res. 2020, 30, 269–271. [Google Scholar] [CrossRef]
- Yao, X.; Ye, F.; Zhang, M.; Cui, C.; Huang, B.; Niu, P.; Liu, X.; Zhao, L.; Dong, E.; Song, C.; et al. In vitro antiviral activity and projection of optimized dosing design of hydroxychloroquine for the treatment of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). Clin. Infect. Dis. 2020, ciaa237. [Google Scholar] [CrossRef] [PubMed]
- Schrezenmeier, E.; Dörner, T. Mechanisms of action of hydroxychloroquine and chloroquine: Implications for rheumatology. Nat. Rev. Rheumatol. 2020, 16, 155–166. [Google Scholar] [CrossRef] [PubMed]
- Zhou, D.; Dai, S.M.; Tong, Q. Covid-19: A recommendation to examine the effect of hydroxychloroquine in preventing infection and progression. J. Antimicrob. Chemother. 2020, dkaa114. [Google Scholar] [CrossRef]
- Čulić, O.; Eraković, V.; Čepelak, I.; Barišić, K.; Brajša, K.; Ferenčić, Ž.; Galović, R.; Glojnarić, I.; Manojlović, Z.; Munić, V.; et al. Azithromycin modulates neutrophil function and circulating inflammatory mediators in healthy human subjects. Eur. J. Pharmacol. 2002, 450, 277–289. [Google Scholar] [CrossRef]
- Beigelman, A.; Mikols, C.L.; Gunsten, S.P.; Cannon, C.L.; Brody, S.L.; Walter, M.J. Azithromycin attenuates airway inflammation in a mouse model of viral bronchiolitis. Respir. Res. 2010, 11, 90. [Google Scholar] [CrossRef]
- Takatori, H.; Kanno, Y.; Chen, Z.; O′Shea, J.J. New complexities in helper T cell fate determination and the implications for autoimmune diseases. Mod. Rheumatol. 2008, 18, 533–541. [Google Scholar] [CrossRef][Green Version]
- Gautret, P.; Lagier, J.-C.; Parola, P.; Hoang, V.T.; Meddeb, L.; Mailhe, M.; Doudier, B.; Courjon, J.; Giordanengo, V.; Vieira, V.E.; et al. Hydroxychloroquine and azithromycin as a treatment of COVID-19: Results of an open-label non-randomized clinical trial. Int. J. Antimicrob. Agents 2020, in press. [Google Scholar] [CrossRef]
- Trump, D.J. White House Coronavirus Task Force Press Conference. Available online: https://www.politico.com/video/2020/04/04/trump-says-he-may-take-hydroxychloroquine-070784 (accessed on 4 April 2020).
- Kalil, A.C. Treating COVID-19-off-label drug use, compassionate use, and randomized clinical trials during pandemics. JAMA 2020, 2763802. Available online: https://jamanetwork.com/journals/jama/fullarticle/2763802 (accessed on 4 April 2020). [CrossRef]
- Owens, B. Excitement around hydroxychloroquine for treating COVID-19 causes challenges for rheumatology. Lancet Rheumatol. 2020, 2, e257. [Google Scholar] [CrossRef]
- Singh, R. Need for abundant caution in prophylactic application of chloroquine and hydroxychloroquine for viral infections including COVID-19: Possibility of increased susceptibility. Soc. Sci. Res. Netw. 2020, 3570607. Available online: http://dx.doi.org/10.2139/ssrn.3570607 (accessed on 4 April 2020). [CrossRef]
- Chen, Z.; Hu, J.; Zhang, Z.; Jiang, S.; Han, S.; Yan, D.; Zhuang, R.; Hu, B.; Zhang, Z. Efficacy of hydroxychloroquine in patients with COVID-19: Results of a randomized clinical trial. medRxiv 2020, 2020, 20040758. [Google Scholar]
- Cortegiani, A.; Ingoglia, G.; Ippolito, M.; Giarratano, A.; Einav, S. A systematic review on the efficacy and safety of chloroquine for the treatment of COVID-19. J. Crit. Care 2020, 57, 279–283. [Google Scholar] [CrossRef] [PubMed]
- Magagnoli, J.; Narendran, S.; Pereira, F.; Cummings, T.; Hardin, J.W.; Sutton, S.S.; Ambati, J. Outcomes of hydroxychloroquine usage in United States veterans hospitalized with COVID-19. medRxiv 2020, 2020, 20065920. [Google Scholar]
- Devaux, C.A.; Rolain, J.-M.; Colson, P.; Raoult, D. New insights on the antiviral effects of chloroquine against coronavirus: What to expect for COVID-19? Int. J. Antimicrob. Agents 2020, in press. [Google Scholar] [CrossRef]
- Esler, M.; Esler, D. Can angiotensin receptor-blocking drugs perhaps be harmful in the COVID-19 pandemic? J. Hypertens. 2020, 38, 781–782. [Google Scholar] [CrossRef]
- Patel, A.B.; Verma, A. COVID-19 and angiotensin-converting enzyme inhibitors and angiotensin receptor blockers: What is the evidence? JAMA 2020, 323, 1769–1770. [Google Scholar] [CrossRef]
- European Society of Cardiology. Position Statement of the ESC Council on Hypertension on ACE-Inhibitors and Angiotensin Receptor Blockers. Available online: https://www.escardio.org/Councils/Council-on-Hypertension-(CHT)/News/position-statement-of-the-esc-council-on-hypertension-on-ace-inhibitors-and-ang (accessed on 11 April 2020).
- Murray, B.A.; FitzGerald, R.J. Angiotensin converting enzyme inhibitory peptides derived from food proteins: Biochemistry, bioactivity and production. Curr. Pharm. Des. 2007, 13, 773–791. [Google Scholar] [CrossRef]
- Pareek, M.; Bangash, M.N.; Pareek, N.; Pan, D.; Sze, S.; Minhas, J.S.; Hanif, W.; Khunti, K. Ethnicity and COVID-19: An urgent public health research priority. Lancet 2020, 395, 1421–1422. [Google Scholar] [CrossRef]
- Hastie, C.E.; Mackay, D.F.; Ho, F.; Celis-Morales, C.A.; Katikireddi, S.V.; Niedzwiedz, C.L.; Jani, B.D.; Welsh, P.; Mair, F.S.; Gray, S.R.; et al. Vitamin D concentrations and COVID-19 infection in UK biobank. Diabetes Metab. Syndr. Clin. Res. Rev. 2020, 14, 561–565. [Google Scholar] [CrossRef] [PubMed]
- Lordan, R.; Tsoupras, A.; Zabetakis, I. Platelet activation and prothrombotic mediators at the nexus of inflammation and atherosclerosis: Potential role of antiplatelet agents. Blood Rev. 2020, in press. [Google Scholar] [CrossRef] [PubMed]
- Luo, P.; Liu, D.; Li, J. Pharmacological perspective: Glycyrrhizin may be an efficacious therapeutic agent for covid-19. Int. J. Antimicrob. Agents 2020, in press. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.-Z.; Zhang, R.-Y.; Bai, J. An anti-oxidative therapy for ameliorating cardiac injuries of critically ill COVID-19-infected patients. Int. J. Cardiol. 2020. Available online: https://www.ncbi.nlm.nih.gov/pubmed/32321655 (accessed on 1 April 2020). [CrossRef] [PubMed]
- Zhang, L.; Liu, Y. Potential interventions for novel coronavirus in China: A systematic review. J. Med. Virol. 2020, 92, 479–490. [Google Scholar] [CrossRef] [PubMed]
- Caccialanza, R.; Laviano, A.; Lobascio, F.; Montagna, E.; Bruno, R.; Ludovisi, S.; Corsico, A.G.; Di Sabatino, A.; Belliato, M.; Calvi, M.; et al. Early nutritional supplementation in non-critically ill patients hospitalized for the 2019 novel coronavirus disease (COVID-19): Rationale and feasibility of a shared pragmatic protocol. Nutrition 2020, 74, 110835. [Google Scholar] [CrossRef] [PubMed]
- Barazzoni, R.; Bischoff, S.C.; Krznaric, Z.; Pirlich, M.; Singer, P. ESPEN expert statements and practical guidance for nutritional management of individuals with SARS-CoV-2 infection. Clin. Nutr. 2020, in press. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Zheng, Y.; Gou, X.; Pu, K.; Chen, Z.; Guo, Q.; Ji, R.; Wang, H.; Wang, Y.; Zhou, Y. Prevalence of comorbidities in the novel wuhan coronavirus (COVID-19) infection: A systematic review and meta-analysis. Int. J. Infect. Dis. 2020, 94, 91–95. [Google Scholar] [CrossRef]
- Van Kerkhove, M.D.; Vandemaele, K.A.H.; Shinde, V.; Jaramillo-Gutierrez, G.; Koukounari, A.; Donnelly, C.A.; Carlino, L.O.; Owen, R.; Paterson, B.; Pelletier, L.; et al. Risk factors for severe outcomes following 2009 influenza a (H1N1) infection: A global pooled analysis. PLoS Med. 2011, 8, e1001053. [Google Scholar] [CrossRef]
- Tsoupras, A.; Lordan, R.; Zabetakis, I. Inflammation, not cholesterol, is a cause of chronic disease. Nutrients 2018, 10, 604. [Google Scholar] [CrossRef]
- Hotamisligil, G.S. Inflammation, metaflammation and immunometabolic disorders. Nature 2017, 542, 177–185. [Google Scholar] [CrossRef]
- Zheng, Y.-Y.; Ma, Y.-T.; Zhang, J.-Y.; Xie, X. COVID-19 and the cardiovascular system. Nature Rev. Cardiol. 2020, 17, 259–260. [Google Scholar] [CrossRef] [PubMed]
- Centers for Disease Control and Prevention. Morbidity and Mortality Weekly Report (mmwr): Preliminary Estimates of the Prevalence of Selected Underlying Health Conditions among Patients with Coronavirus Disease 2019-February 12-March 28. 2020. Available online: https://www.cdc.gov/mmwr/volumes/69/wr/mm6913e2.htm?s_cid=mm6913e2_w (accessed on 3 April 2020).
- Pal, R.; Bhadada, S.K. COVID-19 and diabetes mellitus: An unholy interaction of two pandemics. Diabetes Metab. Syndr. Clin. Res. Rev. 2020, 14, 513–517. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Arora, A.; Sharma, P.; Anil Anikhindi, S.; Bansal, N.; Singla, V.; Khare, S.; Srivastava, A. Is diabetes mellitus associated with mortality and severity of COVID-19? A meta-analysis. Diabetes Metab. Syndr. Clin. Res. Rev. 2020, 14, 535–545. [Google Scholar] [CrossRef] [PubMed]
- Yu, E.; Malik, V.S.; Hu, F.B. Cardiovascular disease prevention by diet modification: JACC health promotion series. J. Am. Coll. Cardiol. 2018, 72, 914–926. [Google Scholar] [CrossRef]
- Wu, G. Important roles of dietary taurine, creatine, carnosine, anserine and 4-hydroxyproline in human nutrition and health. Amino Acids 2020, 52, 329–360. [Google Scholar] [CrossRef]
- Chandra, R.K. Nutrition, immunity and infection: From basic knowledge of dietary manipulation of immune responses to practical application of ameliorating suffering and improving survival. Proc. Natl. Acad. Sci. USA 1996, 93, 14304–14307. [Google Scholar] [CrossRef]
- Hulsewé, K.W.E.; van Acker, B.A.C.; von Meyenfeldt, M.F.; Soeters, P.B. Nutritional depletion and dietary manipulation: Effects on the immune response. World J. Surg. 1999, 23, 536–544. [Google Scholar] [CrossRef]
- Costa, D.; Barbalho, M.C.; Miguel, G.P.S.; Forti, E.M.P.; Azevedo, J.L.M.C. The impact of obesity on pulmonary function in adult women. Clinics 2008, 63, 719–724. [Google Scholar] [CrossRef]
- de Onis, M.; Habicht, J.P. Anthropometric reference data for international use: Recommendations from a World Health Organization Expert Committee. Am. J. Clin. Nutr. 1996, 64, 650–658. [Google Scholar] [CrossRef]
- World Health Organization. Off-label Use of Medicines for COVID-19. Available online: https://www.who.int/news-room/commentaries/detail/off-label-use-of-medicines-for-covid-19 (accessed on 31 March 2020).
- Beck, M.A.; Handy, J.; Levander, O.A. Host nutritional status: The neglected virulence factor. Trends Microbiol. 2004, 12, 417–423. [Google Scholar] [CrossRef]
- Georgousopoulou, E.N.; Kouli, G.-M.; Panagiotakos, D.B.; Kalogeropoulou, A.; Zana, A.; Chrysohoou, C.; Tsigos, C.; Tousoulis, D.; Stefanadis, C.; Pitsavos, C. Anti-inflammatory diet and 10-year (2002–2012) cardiovascular disease incidence: The ATTICA study. Int. J. Cardiol. 2016, 222, 473–478. [Google Scholar] [CrossRef] [PubMed]
- de Boer, A.; van de Worp, W.R.P.H.; Hageman, G.J.; Bast, A. The effect of dietary components on inflammatory lung diseases – a literature review. Int. J. Food Sci. Nutr. 2017, 68, 771–787. [Google Scholar] [CrossRef] [PubMed]
- Lago, J.H.G.; Toledo-Arruda, A.C.; Mernak, M.; Barrosa, K.H.; Martins, M.A.; Tibério, I.F.L.C.; Prado, C.M. Structure-activity association of flavonoids in lung diseases. Molecules 2014, 19, 3570–3595. [Google Scholar] [CrossRef] [PubMed]
- Phillips, C.M.; Chen, L.-W.; Heude, B.; Bernard, J.Y.; Harvey, N.C.; Duijts, L.; Mensink-Bout, S.M.; Polanska, K.; Mancano, G.; Suderman, M.; et al. Dietary inflammatory index and non-communicable disease risk: A narrative review. Nutrients 2019, 11, 1873. [Google Scholar] [CrossRef] [PubMed]
- Melaku, Y.A.; Renzaho, A.; Gill, T.K.; Taylor, A.W.; Dal Grande, E.; de Courten, B.; Baye, E.; Gonzalez-Chica, D.; Hyppönen, E.; Shi, Z.; et al. Burden and trend of diet-related non-communicable diseases in Australia and comparison with 34 OECD countries, 1990–2015: Findings from the global burden of disease study 2015. Eur. J. Nut. 2019, 58, 1299–1313. [Google Scholar] [CrossRef] [PubMed]
- Mattioli, A.V.; Ballerini Puviani, M. Lifestyle at time of COVID-19: How could quarantine affect cardiovascular risk. Am. J. Lifestyle Med. 2020, 14, 240–242. [Google Scholar] [CrossRef]
- Wu, C.; Chen, X.; Cai, Y.; Xia, J.; Zhou, X.; Xu, S.; Huang, H.; Zhang, L.; Zhou, X.; Du, C.; et al. Risk factors associated with acute respiratory distress syndrome and death in patients with coronavirus disease 2019 pneumonia in Wuhan, China. JAMA Intern. Med. 2020. Available online: https://www.practiceupdate.com/content/risk-factors-associated-with-ards-and-death-in-patients-with-covid-19-in-wuhan/97961 (accessed on 1 April 2020). [CrossRef]
- Pae, M.; Meydani, S.N.; Wu, D. The role of nutrition in enhancing immunity in aging. Aging Dis. 2012, 3, 91–129. [Google Scholar]
- Meyer, K.C. The Role of Immunity and Inflammation in Lung Senescence and Susceptibility to Infection in the Elderly. Semin. Respir. Crit. Care Med. 2010, 31, 561–574. [Google Scholar] [CrossRef]
- Volkert, D.; Visser, M.; Corish, C.A.; Geisler, C.; de Groot, L.; Cruz-Jentoft, A.J.; Lohrmann, C.; O′Connor, E.M.; Schindler, K.; de van der Schueren, M.A.E.; et al. Joint action malnutrition in the elderly (MANUEL) knowledge hub: Summary of project findings. Eur. Geriatr. Med. 2020, 11, 169–177. [Google Scholar] [CrossRef]
- Power, S.E.; Jeffery, I.B.; Ross, R.P.; Stanton, C.; O′Toole, P.W.; O′Connor, E.M.; Fitzgerald, G.F. Food and nutrient intake of Irish community-dwelling elderly subjects: Who is at nutritional risk? J. Nutr. Health Aging 2014, 18, 561–572. [Google Scholar] [CrossRef] [PubMed]
- Haase, H.; Rink, L. The immune system and the impact of zinc during aging. Immun. Ageing 2009, 6, 9. [Google Scholar] [CrossRef] [PubMed]
- Gammoh, N.Z.; Rink, L. Zinc in infection and inflammation. Nutrients 2017, 9, 624. [Google Scholar] [CrossRef] [PubMed]
- Jayawardena, R.; Sooriyaarachchi, P.; Chourdakis, M.; Jeewandara, C.; Ranasinghe, P. Enhancing immunity in viral infections, with special emphasis on COVID-19: A review. Diabetes Metab. Syndr. Clin. Res. Rev. 2020, 14, 367–382. [Google Scholar] [CrossRef] [PubMed]
- McCarty, M.F.; DiNicolantonio, J.J. Nutraceuticals have potential for boosting the type 1 interferon response to RNA viruses including influenza and coronavirus. Prog. Cardiovasc. Dis. 2020. Available online: https://www.ncbi.nlm.nih.gov/pubmed/32061635 (accessed on 1 April 2020). [CrossRef] [PubMed]
- Chrysohoou, C.; Panagiotakos, D.B.; Pitsavos, C.; Das, U.N.; Stefanadis, C. Adherence to the Mediterranean diet attenuates inflammation and coagulation process in healthy adults: The ATTICA study. J. Am. Coll. Cardiol. 2004, 44, 152–158. [Google Scholar] [CrossRef]
- Saadeh, D.; Salameh, P.; Baldi, I.; Raherison, C. Diet and allergic diseases among population aged 0 to 18 years: Myth or reality? Nutrients 2013, 5, 3399–3423. [Google Scholar] [CrossRef]
- Martinez-Gonzalez, M.A.; Bes-Rastrollo, M. Dietary patterns, Mediterranean diet, and cardiovascular disease. Curr. Opin. Lipidol. 2014, 25, 20–26. [Google Scholar] [CrossRef]
- Casas, R.; Sacanella, E.; Estruch, R. The immune protective effect of the Mediterranean diet against chronic low-grade inflammatory diseases. Endocr. Metab. Immune Disord. Drug Targets 2014, 14, 245–254. [Google Scholar] [CrossRef]
- Estruch, R.; Ros, E.; Salas-Salvadó, J.; Covas, M.-I.; Corella, D.; Arós, F.; Gómez-Gracia, E.; Ruiz-Gutiérrez, V.; Fiol, M.; Lapetra, J.; et al. Primary prevention of cardiovascular disease with a Mediterranean diet supplemented with extra-virgin olive oil or nuts. N. Engl. J. Med. 2018, 378, e34. [Google Scholar] [CrossRef]
- Kapiszewska, M.; Soltys, E.; Visioli, F.; Cierniak, A.; Zajac, G. The protective ability of the Mediterranean plant extracts against the oxidative DNA damage. The role of the radical oxygen species and the polyphenol content. J. Physiol. Pharmacol. Suppl. 2005, 56, 183–197. [Google Scholar]
- Bonaccio, M.; Pounis, G.; Cerletti, C.; Donati, M.B.; Iacoviello, L.; de Gaetano, G. Mediterranean diet, dietary polyphenols and low grade inflammation: Results from the MOLI-SANI study. Br. J. Clin. Pharmacol. 2017, 83, 107–113. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Y.-C.; Sheen, J.-M.; Hu, W.L.; Hung, Y.-C. Polyphenols and oxidative stress in atherosclerosis-related ischemic heart disease and stroke. Oxid. Med. Cell. Longev. 2017, 2017, 16. [Google Scholar] [CrossRef] [PubMed]
- Cheng, S.; Tu, M.; Liu, H.; Zhao, G.; Du, M. Food-derived antithrombotic peptides: Preparation, identification, and interactions with thrombin. Crit. Rev. Food Sci. Nutr. 2019, 59, S81–S95. [Google Scholar] [CrossRef]
- Shah, R.; Makarem, N.; Emin, M.; Liao, M.; Jelic, S.; Aggarwal, B. Mediterranean diet components are linked to greater endothelial function and lower inflammation in a pilot study of ethnically diverse women. Nutr. Res. 2020, 75, 77–84. [Google Scholar] [CrossRef]
- Muscogiuri, G.; Barrea, L.; Savastano, S.; Colao, A. Nutritional recommendations for COVID-19 quarantine. Eur. J. Clin. Nutr. 2020. Available online: https://www.ncbi.nlm.nih.gov/pubmed/32286533 (accessed on 1 April 2020). [CrossRef]
- Willett, W.C.; Sacks, F.; Trichopoulou, A.; Drescher, G.; Ferro-Luzzi, A.; Helsing, E.; Trichopoulos, D. Mediterranean diet pyramid: A cultural model for healthy eating. Am. J. Clin. Nutr. 1995, 61, 1402s–1406s. [Google Scholar] [CrossRef]
- Galland, L. Diet and inflammation. Nutr. Clin. Pract. 2010, 25, 634–640. [Google Scholar] [CrossRef]
- Tierney, A.; Lordan, R.; Tsoupras, A.; Zabetakis, I. Chapter 8—Diet and cardiovascular disease: The Mediterranean diet. In The Impact of Nutrition and Statins on Cardiovascular Diseases; Zabetakis, I., Lordan, R., Tsoupras, A., Eds.; Academic Press: Cambridge, MA, USA, 2019; pp. 267–288. [Google Scholar]
- Panagiotakos, D.B.; Georgousopoulou, E.N.; Pitsavos, C.; Chrysohoou, C.; Skoumas, I.; Pitaraki, E.; Georgiopoulos, G.A.; Ntertimani, M.; Christou, A.; Stefanadis, C. Exploring the path of Mediterranean diet on 10-year incidence of cardiovascular disease: The ATTICA study (2002–2012). Nutr. Metab. Cardiovasc. Dis. 2015, 25, 327–335. [Google Scholar] [CrossRef]
- O′Keefe, J.H.; Gheewala, N.M.; O′Keefe, J.O. Dietary strategies for improving post-prandial glucose, lipids, inflammation, and cardiovascular health. J. Am. Coll. Cardiol. 2008, 51, 249–255. [Google Scholar] [CrossRef]
- Varraso, R.; Fung, T.T.; Barr, R.G.; Hu, F.B.; Willett, W.; Camargo, C.A., Jr. Prospective study of dietary patterns and chronic obstructive pulmonary disease among us women. Am. J. Clin. Nutr. 2007, 86, 488–495. [Google Scholar] [CrossRef] [PubMed]
- Butler, M.J.; Barrientos, R.M. The impact of nutrition on COVID-19 susceptibility and long-term consequences. Brain Behav. Immun. 2020, in press. [Google Scholar] [CrossRef] [PubMed]
- Sergi, D.; Boulestin, H.; Campbell, F.M.; Williams, L.M. The role of dietary advanced glycation end products in metabolic dysfunction. Mol. Nutr. Food Res. 2020, 1900934. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Xu, S.; Yu, M.; Wang, K.; Tao, Y.; Zhou, Y.; Shi, J.; Zhou, M.; Wu, B.; Yang, Z.; et al. Risk factors for severity and mortality in adult COVID-19 inpatients in Wuhan. J. Allergy Clin. Immunol. 2020, in press. [Google Scholar] [CrossRef] [PubMed]
- Carey, O.J.; Cookson, J.B.; Britton, J.; Tattersfield, A.E. The effect of lifestyle on wheeze, atopy, and bronchial hyperreactivity in asian and white children. Am. J. Respir. Crit. Care Med. 1996, 154, 537–540. [Google Scholar] [CrossRef]
- Wickens, K.; Barry, D.; Friezema, A.; Rhodius, R.; Bone, N.; Purdie, G.; Crane, J. Fast foods—Are they a risk factor for asthma? Allergy 2005, 60, 1537–1541. [Google Scholar] [CrossRef]
- Alencar, L. The role of Western diet on low-grade inflammation. Immunology 2018, 78. Available online: http://www.imedpub.com/proceedings/the-role-of-western-diet-on-lowgrade-inflammation-3221.html (accessed on 1 April 2020).
- Kaluza, J.; Harris, H.R.; Linden, A.; Wolk, A. Long-term consumption of fruits and vegetables and risk of chronic obstructive pulmonary disease: A prospective cohort study of women. Int. J. Epidemiol. 2018, 47, 1897–1909. [Google Scholar] [CrossRef]
- Holt, E.M.; Steffen, L.M.; Moran, A.; Basu, S.; Steinberger, J.; Ross, J.A.; Hong, C.P.; Sinaiko, A.R. Fruit and vegetable consumption and its relation to markers of inflammation and oxidative stress in adolescents. J. Am. Diet. Assoc. 2009, 109, 414–421. [Google Scholar] [CrossRef]
- Serino, A.; Salazar, G. Protective role of polyphenols against vascular inflammation, aging and cardiovascular disease. Nutrients 2018, 11, 53. [Google Scholar] [CrossRef]
- Lichota, A.; Gwozdzinski, L.; Gwozdzinski, K. Therapeutic potential of natural compounds in inflammation and chronic venous insufficiency. Eur. J. Med. Chem. 2019, 176, 68–91. [Google Scholar] [CrossRef]
- Jasso-Miranda, C.; Herrera-Camacho, I.; Flores-Mendoza, L.K.; Dominguez, F.; Vallejo-Ruiz, V.; Sanchez-Burgos, G.G.; Pando-Robles, V.; Santos-Lopez, G.; Reyes-Leyva, J. Antiviral and immunomodulatory effects of polyphenols on macrophages infected with dengue virus serotypes 2 and 3 enhanced or not with antibodies. Infect. Drug Resist. 2019, 12, 1833–1852. [Google Scholar] [CrossRef]
- Vázquez-Calvo, Á.; Jiménez de Oya, N.; Martín-Acebes, M.A.; Garcia-Moruno, E.; Saiz, J.-C. Antiviral properties of the natural polyphenols delphinidin and epigallocatechin gallate against the flaviviruses West Nile virus, Zika virus, and Dengue virus. Front. Microbiol. 2017, 8, 8. [Google Scholar] [CrossRef]
- Wallace, T.C.; Bailey, R.L.; Blumberg, J.B.; Burton-Freeman, B.; Chen, C.y.O.; Crowe-White, K.M.; Drewnowski, A.; Hooshmand, S.; Johnson, E.; Lewis, R.; et al. Fruits, vegetables, and health: A comprehensive narrative, umbrella review of the science and recommendations for enhanced public policy to improve intake. Crit. Rev. Food Sci. Nutr. 2019, 60, 1–38. [Google Scholar] [CrossRef] [PubMed]
- Lordan, R.; Redfern, S.; Tsoupras, A.; Zabetakis, I. Inflammation and cardiovascular disease: Are marine phospholipids the answer? Food Funct. 2020, 11, 2861–2885. [Google Scholar] [CrossRef] [PubMed]
- Lordan, R.; Tsoupras, A.; Zabetakis, I. Phospholipids of animal and marine origin: Structure, function, and anti-inflammatory properties. Molecules 2017, 22, 1964. [Google Scholar] [CrossRef] [PubMed]
- Li, N.; Wu, X.; Zhuang, W.; Xia, L.; Chen, Y.; Wu, C.; Rao, Z.; Du, L.; Zhao, R.; Yi, M.; et al. Fish consumption and multiple health outcomes: Umbrella review. Trends Food Sci. Technol. 2020, 99, 273–283. [Google Scholar] [CrossRef]
- Moro, K.; Nagahashi, M.; Ramanathan, R.; Takabe, K.; Wakai, T. Resolvins and omega-3 polyunsaturated fatty acids: Clinical implications in inflammatory diseases and cancer. World J. Clin. Cases 2016, 4, 155–164. [Google Scholar] [CrossRef] [PubMed]
- Schwerbrock, N.M.J.; Karlsson, E.A.; Shi, Q.; Sheridan, P.A.; Beck, M.A. Fish oil-fed mice have impaired resistance to influenza infection. J. Nutr. 2009, 139, 1588–1594. [Google Scholar] [CrossRef] [PubMed]
- Byleveld, P.M.; Pang, G.T.; Clancy, R.L.; Roberts, D.C.K. Fish oil feeding delays influenza virus clearance and impairs production of interferon-γ and virus-specific immunoglobulin a in the lungs of mice. J. Nutr. 1999, 129, 328–335. [Google Scholar] [CrossRef]
- Byleveld, M.; Pang, G.T.; Clancy, R.L.; Roberts, D.C. Fish oil feeding enhances lymphocyte proliferation but impairs virus-specific t lymphocyte cytotoxicity in mice following challenge with influenza virus. Clin. Exp. Immunol. 2000, 119, 287–292. [Google Scholar] [CrossRef] [PubMed]
- Morita, M.; Kuba, K.; Ichikawa, A.; Nakayama, M.; Katahira, J.; Iwamoto, R.; Watanebe, T.; Sakabe, S.; Daidoji, T.; Nakamura, S.; et al. The lipid mediator protectin D1 inhibits influenza virus replication and improves severe influenza. Cell 2013, 153, 112–125. [Google Scholar] [CrossRef] [PubMed]
- Russell, C.D.; Schwarze, J. The role of pro-resolution lipid mediators in infectious disease. Immunology 2013, 141, 166–173. [Google Scholar] [CrossRef] [PubMed]
- Skarke, C.; Alamuddin, N.; Lawson, J.A.; Li, X.; Ferguson, J.F.; Reilly, M.P.; FitzGerald, G.A. Bioactive products formed in humans from fish oils. J. Lipid Res. 2015, 56, 1808–1820. [Google Scholar] [CrossRef] [PubMed]
- Ramirez, J.L.; Gasper, W.J.; Khetani, S.A.; Zahner, G.J.; Hills, N.K.; Mitchell, P.T.; Sansbury, B.E.; Conte, M.S.; Spite, M.; Grenon, S.M. Fish oil increases specialized pro-resolving lipid mediators in PAD (the omega-pad II trial). J. Surg. Res. 2019, 238, 164–174. [Google Scholar] [CrossRef] [PubMed]
- Carey, M.A.; Bradbury, J.A.; Rebolloso, Y.D.; Graves, J.P.; Zeldin, D.C.; Germolec, D.R. Pharmacologic inhibition of COX-1 and COX-2 in influenza a viral infection in mice. PLoS ONE 2010, 5, e11610. [Google Scholar] [CrossRef]
- Yan, X.; Hao, Q.; Mu, Y.; Timani, K.A.; Ye, L.; Zhu, Y.; Wu, J. Nucleocapsid protein of SARS-CoV activates the expression of cyclooxygenase-2 by binding directly to regulatory elements for nuclear factor-kappa b and ccaat/enhancer binding protein. Int. J. Biochem. Cell Bio. 2006, 38, 1417–1428. [Google Scholar] [CrossRef]
- FitzGerald, G.A. Misguided drug advice for COVID-19. Science 2020, 367, 1434. [Google Scholar]
- Smeitink, J.; Jiang, X.; Pecheritsyna, S.; Renkema, H.; van Maanen, R.; Beyrath, J. Hypothesis: mPGES-1-derived prostaglandin E2, a so far missing link in COVID-19 pathophysiology? Preprints 2020, 2020040180. [Google Scholar] [CrossRef]
- Ye, J.; Ghosh, S. Omega-3 PUFA vs. NSAIDs for preventing cardiac inflammation. Front. Cardiovasc. Med. 2018, 5, 5. [Google Scholar] [CrossRef]
- Das, U.N. Can bioactive lipids inactivate coronavirus (COVID-19)? Arch. Med. Res. 2020, in press. [Google Scholar] [CrossRef] [PubMed]
- Das, U.N. Arachidonic acid and other unsaturated fatty acids and some of their metabolites function as endogenous antimicrobial molecules: A review. J. Adv. Res. 2018, 11, 57–66. [Google Scholar] [CrossRef] [PubMed]
- Kohn, A.; Gitelman, J.; Inbar, M. Unsaturated free fatty acids inactivate animal enveloped viruses. Arch. Virol. 1980, 66, 301–307. [Google Scholar] [CrossRef] [PubMed]
- Hilmarsson, H.; Larusson, L.V.; Thormar, H. Virucidal effect of lipids on visna virus, a lentivirus related to HIV. Arch. Virol. 2006, 151, 1217–1224. [Google Scholar] [CrossRef]
- Yan, B.; Chu, H.; Yang, D.; Sze, K.-H.; Lai, P.-M.; Yuan, S.; Shuai, H.; Wang, Y.; Kao, R.Y.-T.; Chan, J.F.-W.; et al. Characterization of the lipidomic profile of human coronavirus-infected cells: Implications for lipid metabolism remodeling upon coronavirus replication. Viruses 2019, 11, 73. [Google Scholar] [CrossRef]
- Yan, B.; Zou, Z.; Chu, H.; Chan, G.; Tsang, J.O.-L.; Lai, P.-M.; Yuan, S.; Yip, C.C.-Y.; Yin, F.; Kao, R.Y.-T.; et al. Lipidomic profiling reveals significant perturbations of intracellular lipid homeostasis in enterovirus-infected cells. Int. J. Mol. Sci. 2019, 20, 5952. [Google Scholar] [CrossRef]
- Innes, J.K.; Calder, P.C. Omega-6 fatty acids and inflammation. Prostaglandins Leukot. Essent. Fatty Acids 2018, 132, 41–48. [Google Scholar] [CrossRef]
- Miles, E.A.; Calder, P.C. Influence of marine n-3 polyunsaturated fatty acids on immune function and a systematic review of their effects on clinical outcomes in rheumatoid arthritis. Br. J. Nutr. 2012, 107, S171–S184. [Google Scholar] [CrossRef]
- Tsoupras, A.; Lordan, R.; Shiels, K.; Saha, S.K.; Nasopoulou, C.; Zabetakis, I. In vitro antithrombotic properties of salmon (Salmo salar) phospholipids in a novel food-grade extract. Mar. Drugs 2019, 17, 62. [Google Scholar] [CrossRef]
- Souza, D.G.; Fagundes, C.T.; Sousa, L.P.; Amaral, F.A.; Souza, R.S.; Souza, A.L.; Kroon, E.G.; Sachs, D.; Cunha, F.Q.; Bukin, E.; et al. Essential role of platelet-activating factor receptor in the pathogenesis of dengue virus infection. Proc. Natl. Acad. Sci. USA 2009, 106, 14138–14143. [Google Scholar] [CrossRef]
- Jeewandara, C.; Gomes, L.; Wickramasinghe, N.; Gutowska-Owsiak, D.; Waithe, D.; Paranavitane, S.A.; Shyamali, N.L.A.; Ogg, G.S.; Malavige, G.N. Platelet-activating factor contributes to vascular leak in acute Dengue infection. PLoS Negl. Trop. Dis. 2015, 9, e0003459. [Google Scholar] [CrossRef] [PubMed]
- Villani, A.; Cirino, N.; Baldi, E.; Kester, M.; McFadden, E.; Panuska, J. Respiratory syncytial virus infection of human mononuclear phagocytes stimulates synthesis of platelet-activating factor. J. Biol. Chem. 1991, 266, 5472–5479. [Google Scholar] [PubMed]
- Garcia, C.C.; Russo, R.C.; Guabiraba, R.; Fagundes, C.T.; Polidoro, R.B.; Tavares, L.P.; Salgado, A.P.C.; Cassali, G.D.; Sousa, L.P.; Machado, A.V.; et al. Platelet-activating factor receptor plays a role in lung injury and death caused by influenza a in mice. PLoS Pathog. 2010, 6, e1001171. [Google Scholar] [CrossRef] [PubMed]
- Eggert, D.; Dash, P.K.; Serradji, N.; Dong, C.-Z.; Clayette, P.; Heymans, F.; Dou, H.; Gorantla, S.; Gelbard, H.A.; Poluektova, L.; et al. Development of a platelet-activating factor antagonist for HIV-1 associated neurocognitive disorders. J. Neuroimmunol. 2009, 213, 47–59. [Google Scholar] [CrossRef] [PubMed]
- Lordan, R.; Zabetakis, I. Invited review: The anti-inflammatory properties of dairy lipids. J. Dairy Sci. 2017, 100, 4197–4212. [Google Scholar] [CrossRef] [PubMed]
- Lordan, R.; Walsh, A.M.; Crispie, F.; Finnegan, L.; Cotter, P.D.; Zabetakis, I. The effect of ovine milk fermentation on the antithrombotic properties of polar lipids. J. Funct. Foods 2019, 54, 289–300. [Google Scholar] [CrossRef]
- Tsoupras, A.; Lordan, R.; Harrington, J.; Pienaar, R.; Devaney, K.; Heaney, S.; Koidis, A.; Zabetakis, I. The effects of oxidation on the antithrombotic properties of tea lipids against PAF, thrombin, collagen, and ADP. Foods 2020, 9, 385. [Google Scholar] [CrossRef]
- Tsoupras, A.; O′Keeffe, E.; Lordan, R.; Redfern, S.; Zabetakis, I. Bioprospecting for antithrombotic polar lipids from salmon, herring, and boarfish by-products. Foods 2019, 8, 416. [Google Scholar] [CrossRef]
- Lordan, R.; Nasopoulou, C.; Tsoupras, A.; Zabetakis, I. The anti-inflammatory properties of food polar lipids. In Bioactive Molecules in Food; Mérillon, J.M., Ramawat, K.G., Eds.; Springer International Publishing: Cham, Switzerland, 2018; pp. 1–34. [Google Scholar]
- Bhat, Z.F.; Kumar, S.; Bhat, H.F. Antihypertensive peptides of animal origin: A review. Crit. Rev. Food Sci. Nutr. 2017, 57, 566–578. [Google Scholar] [CrossRef]
- Yang, B.; Ren, X.; Li, Z.; Shi, M.; Ding, F.; Su, K.-P.; Guo, X.; Li, D. Lowering effects of fish oil supplementation on proinflammatory markers in hypertension: Results from a randomized controlled trial. Food Funct. 2020, 11, 1779–1789. [Google Scholar] [CrossRef]
- Knapp, H.R.; FitzGerald, G.A. The antihypertensive effects of fish oil. N. Engl. J. Med. 1989, 320, 1037–1043. [Google Scholar] [CrossRef] [PubMed]
- Knapp, H.R.; Reilly, I.A.G.; Alessandrini, P.; FitzGerald, G.A. In vivo indexes of platelet and vascular function during fish-oil administration in patients with atherosclerosis. N. Engl. J. Med. 1986, 314, 937–942. [Google Scholar] [CrossRef] [PubMed]
- Pauling, L. Vitamin c and the common cold. Can. Med. Assoc. J. 1971, 105, 448. [Google Scholar]
- Hemilä, H.; Chalker, E. Vitamin C for preventing and treating the common cold. Cochrane Database Syst. Rev. 2013, 1, CD000980. [Google Scholar] [CrossRef] [PubMed]
- Douglas, R.M.; Hemilä, H.; Chalker, E.; Treacy, B. Vitamin C for preventing and treating the common cold. Cochrane Database Syst. Rev. 2007, 3, CD000980. [Google Scholar]
- Forastiere, F.; Pistelli, R.; Sestini, P.; Fortes, C.; Renzoni, E.; Rusconi, F.; Dell′Orco, V.; Ciccone, G.; Bisanti, L. Consumption of fresh fruit rich in vitamin C and wheezing symptoms in children. Thorax 2000, 55, 283–288. [Google Scholar] [CrossRef]
- Heimer, K.A.; Hart, A.M.; Martin, L.G.; Rubio-Wallace, S. Examining the evidence for the use of vitamin C in the prophylaxis and treatment of the common cold. J. Am. Acad. Nurse Pract. 2009, 21, 295–300. [Google Scholar] [CrossRef]
- Hemilä, H. Vitamin C and infections. Nutrients 2017, 9, 339. [Google Scholar] [CrossRef]
- Cheng, R.Z. Can early and high intravenous dose of vitamin C prevent and treat coronavirus disease 2019 (COVID-19)? Med. Drug Discov 2020, 5, 100028. [Google Scholar] [CrossRef]
- Krinsky, N.I.; Beecher, G.; Burk, R.; Chan, A.; Erdman, j.J.; Jacob, R.; Jialal, I.; Kolonel, L.; Marshall, J.; Taylor Mayne, P.R. Dietary Reference Intakes for Vitamin C, Vitamin E, Selenium, and Carotenoids; A Report of the Panel on Dietary Antioxidants and Related Compounds, Subcommittees on Upper Reference Levels of Nutrients and Interpretation and Uses of Dietary Reference Intakes, and the Standing Committee on the Scientific Evaluation of Dietary Reference Intakes, Institute of Medicine; The National Academies Press: Washington, DC, USA, 2000. [Google Scholar]
- Martineau, A.R.; Jolliffe, D.A.; Hooper, R.L.; Greenberg, L.; Aloia, J.F.; Bergman, P.; Dubnov-Raz, G.; Esposito, S.; Ganmaa, D.; Ginde, A.A.; et al. Vitamin D supplementation to prevent acute respiratory tract infections: Systematic review and meta-analysis of individual participant data. BMJ 2017, 356, i6583. [Google Scholar] [CrossRef]
- Rhodes, J.M.; Subramanian, S.; Laird, E.; Kenny, R.A. Editorial: Low population mortality from COVID-19 in countries south of latitude 35 degrees north supports vitamin d as a factor determining severity. Aliment. Pharmacol. Ther. 2020. Available online: https://onlinelibrary.wiley.com/doi/full/10.1111/apt.15777?af=R (accessed on 1 April 2020). [CrossRef] [PubMed]
- McCartney, D.M.; Byrne, D.G. Optimisation of vitamin D status for enhanced immuno-protection against COVID-19. Ir. Med. J. 2020, 113, 58. [Google Scholar] [PubMed]
- Laird, E.; Kenny, R.A. Vitamin D Deficiency in Ireland—Implications forCOVID-19. Results from the Irish Longitudinal Study on Ageing (TILDA); Trinity College Dublin: Dublin, Ireland, 10 April 2020. [Google Scholar]
- Teymoori-Rad, M.; Shokri, F.; Salimi, V.; Marashi, S.M. The interplay between vitamin D and viral infections. Rev. Med. Virol. 2019, 29, e2032. [Google Scholar] [CrossRef] [PubMed]
- Jakovac, H. COVID-19 and vitamin D-is there a link and an opportunity for intervention? Am. J. Physiol. Endocrinol. Metab. 2020, 318, E589. [Google Scholar] [CrossRef]
- D′Avolio, A.; Avataneo, V.; Manca, A.; Cusato, J.; De Nicolò, A.; Lucchini, R.; Keller, F.; Cantù, M. 25-hydroxyvitamin D concentrations are lower in patients with positive PCR for SARS-CoV-2. Nutrients 2020, 12, 1359. [Google Scholar] [CrossRef] [PubMed]
- Del Valle, H.B.; Yaktine, A.L.; Taylor, C.L.; Ross, A.C. Dietary Reference Intakes for Calcium and \vitamin D. In Institute of Medicine (US) Committee to Review Dietary Reference Intakes for Vitamin D and Calcium; National Academies Press: Cambridge, MA, USA, 2011. [Google Scholar]
- Lee, G.Y.; Han, S.N. The role of vitamin E in immunity. Nutrients 2018, 10, 1614. [Google Scholar] [CrossRef]
- Wu, D.; Meydani, S.N. Vitamin E, immunity, and infection. In Nutrition, Immunity, and Infection; CRC Press: Boca Raton, FL, USA, 2017; pp. 197–212. [Google Scholar]
- Coquette, A.; Vray, B.; Vanderpas, J. Role of vitamin E in the protection of the resident macrophage membrane against oxidative damage. Arch. Int. Physiol. Biochim. 1986, 94, S29–S34. [Google Scholar]
- Meydani, S.N.; Lewis, E.D.; Wu, D. Perspective: Should vitamin E recommendations for older adults be increased? Adv. Nutr. 2018, 9, 533–543. [Google Scholar] [CrossRef]
- Gavazzi, C.; Colatruglio, S.; Sironi, A.; Mazzaferro, V.; Miceli, R. Importance of early nutritional screening in patients with gastric cancer. Br. J. Nutr. 2011, 106, 1773–1778. [Google Scholar] [CrossRef]
- Hemilä, H. Vitamin E administration may decrease the incidence of pneumonia in elderly males. Clin. Interv. Aging 2016, 11, 1379–1385. [Google Scholar] [CrossRef]
- Ginaldi, L.; Loreto, M.F.; Corsi, M.P.; Modesti, M.; De Martinis, M. Immunosenescence and infectious diseases. Microbes Infect. 2001, 3, 851–857. [Google Scholar] [CrossRef]
- Prasad, A.S. Zinc in human health: Effect of zinc on immune cells. Mol. Med. 2008, 14, 353–357. [Google Scholar] [CrossRef] [PubMed]
- Walker, C.F.; Black, R.E. Zinc and the risk for infectious disease. Annu. Rev. Nutr. 2004, 24, 255–275. [Google Scholar] [CrossRef] [PubMed]
- Hess, S.Y.; Lönnerdal, B.; Hotz, C.; Rivera, J.A.; Brown, K.H. Recent advances in knowledge of zinc nutrition and human health. Food Nutr. Bull. 2009, 30, S5–S11. [Google Scholar] [CrossRef]
- Sahuquillo, A.; Barberá, R.; Farré, R. Bioaccessibility of calcium, iron and zinc from three legume samples. Food/Nahrung 2003, 47, 438–441. [Google Scholar] [CrossRef]
- Poutzalis, S.; Lordan, R.; Nasopoulou, C.; Zabetakis, I. Phospholipids of goat and sheep origin: Structural and functional studies. Small Rumin. Res. 2018, 167, 39–47. [Google Scholar] [CrossRef]
- Lordan, R.; Tsoupras, A.; Mitra, B.; Zabetakis, I. Dairy fats and cardiovascular disease: Do we really need to be concerned? Foods 2018, 7, 29. [Google Scholar] [CrossRef]
- Science, M.; Johnstone, J.; Roth, D.E.; Guyatt, G.; Loeb, M. Zinc for the treatment of the common cold: A systematic review and meta-analysis of randomized controlled trials. CMAJ 2012, 184, E551–E561. [Google Scholar] [CrossRef]
- Russell, R.M.; Beard, J.L.; Cousins, R.J.; Dunn, J.T.; Ferland, G.; Hambidge, K.M.; Lynch, S.; Penland, J.G.; Ross, A.C.; Stoecker, B.J. Dietary Reference Intakes for Vitamin A, Vitamin K, Arsenic, Boron, Chromium, Copper, Iodine, Iron, Manganese, Molybdenum, Nickel, Silicon, Vanadium, and Zinc; A Report of the Panel on Micronutrients, Subcommittees on Upper Reference Levels of Nutrients and of Interpretation and Uses of Dietary Reference Intakes, and the Standing Committee on the Scientific Evaluation of Dietary Reference Intakes Food and Nutrition Board Institute of Medicine; National Academies Press: Washington, DC, USA, 2001. [Google Scholar]
- Bost, M.; Houdart, S.; Oberli, M.; Kalonji, E.; Huneau, J.-F.; Margaritis, I. Dietary copper and human health: Current evidence and unresolved issues. J. Trace Elem. Med. Biol. 2016, 35, 107–115. [Google Scholar] [CrossRef]
- Uriu-Adams, J.Y.; Keen, C.L. Copper, oxidative stress, and human health. Mol. Aspects Med. 2005, 26, 268–298. [Google Scholar] [CrossRef]
- Bonham, M.; O′Connor, J.M.; Hannigan, B.M.; Strain, J.J. The immune system as a physiological indicator of marginal copper status? Br. J. Nutr. 2007, 87, 393–403. [Google Scholar] [CrossRef]
- Liu, L.; Geng, X.; McDermott, J.; Shen, J.; Corbin, C.; Xuan, S.; Kim, J.; Zuo, L.; Liu, Z. Copper deficiency in the lungs of TNF-α transgenic mice. Front. Physiol. 2016, 7, 543. [Google Scholar] [CrossRef] [PubMed]
- Pearson, P.; Britton, J.; McKeever, T.; Lewis, S.A.; Weiss, S.; Pavord, I.; Fogarty, A. Lung function and blood levels of copper, selenium, vitamin C and vitamin E in the general population. Eur. J. Clin. Nutr. 2005, 59, 1043–1048. [Google Scholar] [CrossRef] [PubMed]
- Viñas, B.R.; Roman Viñas, B.; Ribas Barba, L.; Ngo, J.; Gurinovic, M.; Novakovic, R.; Cavelaars, A.; de Groot, L.C.P.G.M.; van′t Veer, P.; Matthys, C.; et al. Projected prevalence of inadequate nutrient intakes in Europe. Ann. Nutr. Metab. 2011, 59, 84–95. [Google Scholar] [CrossRef] [PubMed]
- Turnlund, J.R.; Jacob, R.A.; Keen, C.L.; Strain, J.; Kelley, D.S.; Domek, J.M.; Keyes, W.R.; Ensunsa, J.L.; Lykkesfeldt, J.; Coulter, J. Long-term high copper intake: Effects on indexes of copper status, antioxidant status, and immune function in young men. Am. J. Clin. Nutr. 2004, 79, 1037–1044. [Google Scholar] [CrossRef]
- Schley, P.D.; Field, C.J. The immune-enhancing effects of dietary fibres and prebiotics. Br. J. Nutr. 2007, 87, S221–S230. [Google Scholar] [CrossRef]
- Arrieta, M.C.; Meddings, J.; Field, C.J. The immunomodulatory effects of dietary fiber and prebiotics in the gastrointestinal tract. In Nondigestible Carbohydrates and Digestive Health; Paeschke, T.M., Aimutis, W.R., Eds.; Blackwell Publishing Ltd. and Institute of Food Technologists: Ames, IA, USA, 2011; pp. 37–77. [Google Scholar]
- Roberfroid, M.; Gibson, G.R.; Hoyles, L.; McCartney, A.L.; Rastall, R.; Rowland, I.; Wolvers, D.; Watzl, B.; Szajewska, H.; Stahl, B.; et al. Prebiotic effects: Metabolic and health benefits. Br. J. Nutr. 2010, 104, S1–S63. [Google Scholar] [CrossRef]
- Trompette, A.; Gollwitzer, E.S.; Pattaroni, C.; Lopez-Mejia, I.C.; Riva, E.; Pernot, J.; Ubags, N.; Fajas, L.; Nicod, L.P.; Marsland, B.J. Dietary fiber confers protection against flu by shaping ly6c—Patrolling monocyte hematopoiesis and CD8+ t cell metabolism. Immunity 2018, 48, 992–1005. [Google Scholar] [CrossRef]
- King, D.E.; Mainous, A.G.; Lambourne, C.A. Trends in dietary fiber intake in the United States, 1999–2008. J. Acad. Nutr. Diet. 2012, 112, 642–648. [Google Scholar] [CrossRef]
- Simpson, R.J.; Campbell, J.P.; Gleeson, M.; Krüger, K.; Nieman, D.C.; Pyne, D.B.; Turner, J.E.; Walsh, N.P. Can exercise affect immune function to increase susceptibility to infection? Exerc. Immunol. Rev. 2020, 26, 8–22. [Google Scholar]
- Halabchi, F.; Ahmadinejad, Z.; Selk-Ghaffari, M. COVID-19 epidemic: Exercise or not to exercise; that is the question! Asian J. Sports Med. 2020, 11, e102630. [Google Scholar] [CrossRef]
- Yılmaz, C.; Gökmen, V. Neuroactive compounds in foods: Occurrence, mechanism and potential health effects. Food Res. Int. 2020, 128, 108744. [Google Scholar] [CrossRef] [PubMed]
- Moynihan, A.B.; Tilburg, W.A.P.v.; Igou, E.R.; Wisman, A.; Donnelly, A.E.; Mulcaire, J.B. Eaten up by boredom: Consuming food to escape awareness of the bored self. Front. Psychol. 2015, 6, 6. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Food and Nutrition Tips during Self-Quarantine. Available online: http://www.euro.who.int/en/health-topics/health-emergencies/coronavirus-covid-19/novel-coronavirus-2019-ncov-technical-guidance/food-and-nutrition-tips-during-self-quarantine (accessed on 21 April 2020).
- Lovesley, D.; Parasuraman, R.; Ramamurthy, A. Combating hospital malnutrition: Dietitian-led quality improvement initiative. Clin. Nutr. ESPEN 2019, 30, 19–25. [Google Scholar] [CrossRef] [PubMed]
- Curtis, L.J.; Bernier, P.; Jeejeebhoy, K.; Allard, J.; Duerksen, D.; Gramlich, L.; Laporte, M.; Keller, H.H. Costs of hospital malnutrition. Clin. Nutr. 2017, 36, 1391–1396. [Google Scholar] [CrossRef]
- Cena, H.; Maffoni, S.; Braschi, V.; Brazzo, S.; Pallavicini, C.; Vietti, I.; Portale, S.; Corradi, E. Position paper of the Italian Association of Medical Specialists in Dietetics and Clinical Nutrition (ANSISA) on nutritional management of patients with COVID-19 disease. Med. J. Nutr. Metab. 2020. [Google Scholar] [CrossRef]
- Kalantar-Zadeh, K.; Moore, L.W. Impact of nutrition and diet on COVID-19 infection and implications for kidney health and kidney disease management. J. Ren. Nutr. 2020, 30, 179–181. [Google Scholar] [CrossRef]

© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zabetakis, I.; Lordan, R.; Norton, C.; Tsoupras, A. COVID-19: The Inflammation Link and the Role of Nutrition in Potential Mitigation. Nutrients 2020, 12, 1466. https://doi.org/10.3390/nu12051466
Zabetakis I, Lordan R, Norton C, Tsoupras A. COVID-19: The Inflammation Link and the Role of Nutrition in Potential Mitigation. Nutrients. 2020; 12(5):1466. https://doi.org/10.3390/nu12051466
Chicago/Turabian StyleZabetakis, Ioannis, Ronan Lordan, Catherine Norton, and Alexandros Tsoupras. 2020. "COVID-19: The Inflammation Link and the Role of Nutrition in Potential Mitigation" Nutrients 12, no. 5: 1466. https://doi.org/10.3390/nu12051466
APA StyleZabetakis, I., Lordan, R., Norton, C., & Tsoupras, A. (2020). COVID-19: The Inflammation Link and the Role of Nutrition in Potential Mitigation. Nutrients, 12(5), 1466. https://doi.org/10.3390/nu12051466







