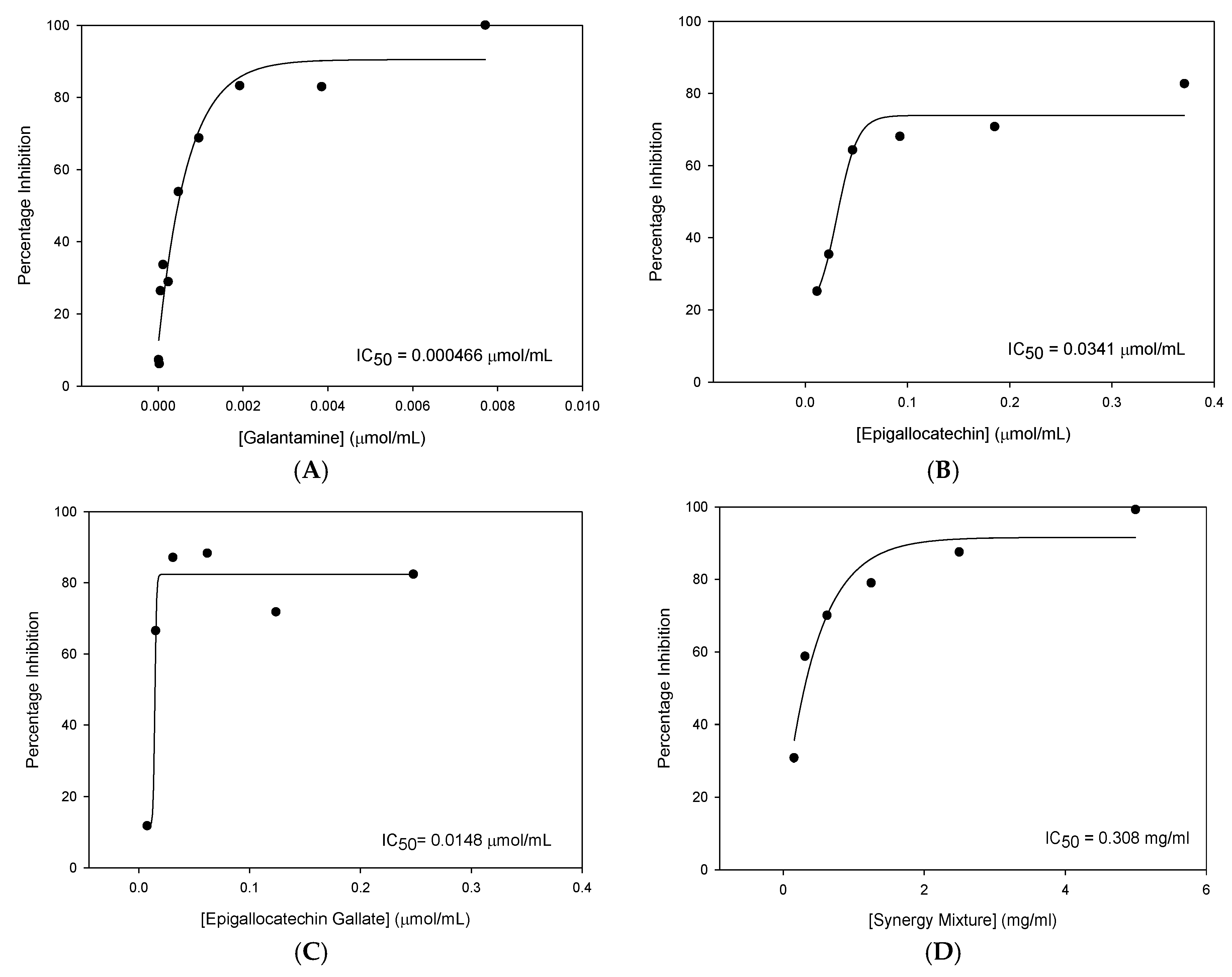1. Introduction
Alzheimer’s disease is the most common form of dementia in the UK accounting for 50–75% of cases [
1]. Indeed, the number of people with AD in the UK is expected to nearly double by 2040 to 1.6 million patients, with the total cost of treating dementia expected to reach £94.1 bn by 2040 [
2]. AD is an irreversible neurodegenerative disorder in which there is a progressive and continual deterioration in cognitive function [
3]. Initial symptoms typically include confusion, repetitive questioning and changes in mood [
4]. These symptoms progressively worsen with patients beginning to experience delusion and aphasia until eventually suffering from difficulties with breathing, eating and moving, especially without significant assistance from carers [
4].
The aetiology and pathophysiology of AD is complex and is characterized by the apoptosis of cholinergic neurons, especially those in the limbic and neocortical regions [
5] and the consequent attenuation of acetylcholine mediated neurotransmission. This cholinergic neuronal apoptosis is thought to result primarily from the abnormal production and processing of the β-amyloid and tau proteins [
6,
7,
8,
9,
10]. In addition to this, there is increasing recognition that the immune system, and the subsequent inflammatory cascades it stimulates, are also of significant importance [
11]. Genetic susceptibility in the form of polymorphism at the APOE gene locus also appears to modulate risk with the ε4 variant conferring greater risk of AD development [
12].
Consequently, there is considerable interest in the development of effective treatments to alleviate the symptoms of AD and potentially halt the underlying neurodegeneration the disease causes. Such treatment should lead to an improved quality of life for patients whilst also reducing the financial burden that health systems experience as a result of the disease. The loss of cholinergic neurons, especially those in the limbic and neocortical regions (which are vital for cognitive functions such as memory, learning and attention [
13] leads to diminished acetylcholine (ACh) production [
5]. Therefore, inhibition of Acetylcholinesterase (AChE), the enzyme responsible for ACh metabolism [
14], is a key target in the treatment of AD. Effective inhibition of AChE allows synaptic levels of ACh to be restored, thus alleviating the symptoms of AD.
In addition, Butyrylcholinesterase (BuChE) has also been identified as another important cholinesterase relevant to AD [
15]. Levels of BuChE expression are increased by up to 120% in patients with AD, possibly due to compensatory mechanisms which relate to AChE expression which can be reduced by 55–67% [
15]. Consequently, the majority of ACh is metabolised by BuChE in the later stages of AD.
Therefore, dual inhibition of BuChE, as well as AChE, would be an attractive property for any potential novel cholinesterase inhibitor. Several AChE inhibitors including galantamine (Gal), donepezil and rivastigmine have been developed and approved for the treatment of mild AD [
16]. Only rivastigmine has been approved for the inhibition of both AChE and BuChE [
16], although Gal has been shown to inhibit BuChE to a certain extent [
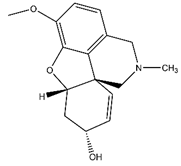
17]. Gal is currently the treatment of choice for AD and has a dual mechanism of action, increasing the sensitivity of postsynaptic NMDA receptors as well as inhibiting AChE [
18]. Both of these factors work to enhance cholinergic neurotransmission. However, despite its efficacy, Gal results in unpleasant side-effects which include cardiac arrhythmias, GI irritation and tremor [
19]. These side-effects combined with the fact that Gal is unable to halt the underlying progression of AD means that there is significant interest in the identification of superior alternatives concerning both efficacy and side-effect profile.
Multiple compounds with anti-cholinesterase activity have been derived from natural plant-based sources [
20]. These natural compounds often have multiple, wide-ranging health benefits beyond cholinesterase inhibition such as antioxidation and anti-inflammation.
Consequently, natural compounds offer an attractive proposition for the identification of effective AD treatments since they have the potential to improve symptoms by targeting multiple parts of AD pathology, including inflammation and the generation of reactive oxygen species [
11].
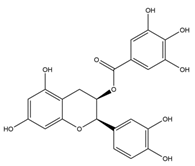
Flavan-3-ols are an example of a class of naturally occurring compounds, found mostly in green tea, which have been shown to possess antioxidant and anti-inflammatory properties [
21]. The average cup of green tea is estimated to contain a total of 67 mg of these flavan-3-ol compounds [
22].
In addition, studies in human clinical trials have demonstrated the ability of flavan-3-ol compounds to slow cognitive decline [
23]. Consequently, flavan-3-ols are an attractive source from which to identify novel cholinesterase inhibitors.
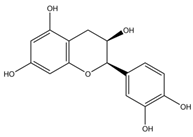
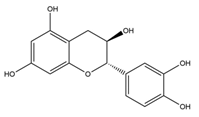
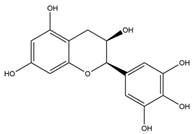
Table 1 summarises the five flavan-3-ol compounds which were investigated in this study along with Gal, the gold standard for AChE inhibition.
Based on this information, it was hypothesised that the beneficial effects of these flavan-3-ol compounds extended to cholinesterase inhibition.
2. Materials and Methods
Acetylcholinesterase (AChE) (EC 3.1.1.7) from electric eel, Butyrylcholinesterase (BuChE) (EC 3.1.18), from equine serum, acetylthiocholine iodide (ATC), butyrylthiocholine iodide (BTC), 5:5-dithiobis-2-nitrobenzoic acid (DTNB), sodium phosphate (mono and dibasic), sodium bicarbonate, Galanthamine hydrobromide from Lycoris sp, (-)-epicatechin (EC), catechin, (-)-epicatechin-3-gallate (ECG), (-)-epigallocatechin (EGC) and (-)-epigallocatechin-3-gallate (EGCG) were purchased from Sigma-Aldrich (UK).
2.1. Ellman Essay
The Ellman assay was used to quantify inhibition of the cholinesterase enzymes [
26] with some modification [
27]. In brief, 5 µL of AChE isolated from the electric eel (0.03 U/mL, pH 8 Na
3PO
4 buffer) or 5 µL of BuChE isolated from equine serum (0.03 U/mL, pH 8 Na
3PO
4 buffer) was added to a generic 96 well plate followed by 200 µL of pH 8 Na
3PO
4 buffer, 5 µL of DTNB (0.3 mM in pH 7 Na
3PO
4 buffer) and finally, 5 µL of the inhibitor under investigation (dissolved in de-ionised H
2O). Blank (no inhibitor or enzyme) and control (no inhibitor) samples were also set up with the total volume made up to 215 µL using pH 8 phosphate buffer prior to incubation. Samples were then mixed for 10 s at 600 rpm and incubated at 30 °C for 10 min in the Ascent Multiskan Plate Reader. Following this, 5 µL of the substrate ATC (0.5 mM in pH 8 Na
3PO
4 buffer) or BTC was added (0.5 mM in pH 8 Na
3PO
4 buffer) to each well.
Average absorbance was then measured every 30 s for 6 min at 405 nm. The concentration of each inhibitor used to initially test for cholinesterase inhibition varied according to solubility (
Table 2).
A test for synergy between four of the flavan-3-ols under investigation was also carried out. Epicatechin (EC), catechin, epigallocatechin (EGC) and epigallocatechin gallate (EGCG) were mixed in a 1:1:1:1 ratio resulting in a test concentration for each inhibitor of 1.25 mg/mL in the synergy mixture.
Average absorbance from the blank wells was calculated and subtracted from each control and inhibitor concentration triplicate absorbance average. Percentage inhibition was then calculated using the following equation: (mean absorbance with inhibitor/negative control mean absorbance) × 100.
If statistically significant inhibition was observed, a serial dilution was carried out to produce a range of concentrations from which a concentration-percentage inhibition curve could be constructed using SigmaPlotTM (Systat Software Inc., San Jose, CA, USA) From these plots, the IC50 value was calculated.
2.2. Kinetic Analysis: Lineweaver–Burk Plots (L-B)
Kinetics of inhibition was determined using L-B reciprocal plots which were constructed using SigmaPlotTM. Two concentrations of each inhibitor, as well as a control, were tested for their anti-cholinesterase activity using four different substrate concentrations (0.5, 0.25, 0.125 and 0.0625 mM).
Results were then plotted on a graph of 1/absorbance against 1/[substrate] and lines were extrapolated backwards to determine the point at which the three lines intersected. The point of intersection allows the type of inhibition present to be identified. From these plots, the KM and VMAX for each inhibitor concentration were calculated by obtaining the gradient and y-intercept for each trend line from Excel. VMAX was calculated by 1/Y-intercept and KM was calculated by multiplying the gradient and the VMAX.
Therefore, for non-competitive inhibitors, the V
MAX will be reduced, and the K
M will remain the same—the opposite is true for competitive inhibition [
28]. For uncompetitive inhibition, there is a reduction in both the V
MAX and the K
M values whilst for mixed inhibition, V
MAX decreases and the K
M can either increase or decrease [
28].
3. Results
3.1. AChE Inhibition
Percentage inhibition of AChE in the presence of the compounds under investigation is shown in
Table 3.
Of the seven inhibitors tested, five exhibited statistically significant inhibition (
p < 0.01), with Gal showing the most potent AChE inhibitory activity (
Table 3). In addition to Gal, ECG, EGC and EGCG were found to possess significant AChE inhibitory properties and were studied further.
Synergism between four of the flavan-3-ol compounds (EC, catechin, EGC and EGCG) was observed despite two of the flavan-3-ols, EC and catechin, being identified as inactive when administered alone. EC and catechin and were found to have no significant inhibition of AChE even at the highest available concentrations. Therefore, these compounds were not investigated any further.
Serial dilutions for each of the active compounds and the synergistic mixture yielded a range of concentrations from which IC
50 values were calculated (
Table 4). Whilst ECG did show statistically significant inhibition, its potency was too low for an IC
50 value to be accurately calculated.
Gal was by far the most potent inhibitor tested with an IC
50 value 73.2× and 31.8× more potent than EGC and EGCG respectively. The IC
50 value for the synergistic mixture is expressed in mg/mL and compared with Gal in
Table 5.
Again, Gal demonstrates much greater potency with an IC50 value 40.8-fold smaller than that of the synergy mixture.
A one-way ANOVA with post-hoc Tukey analysis was then used to reveal the minimum inhibitor concentration required for each compound to achieve statistically significant inhibition of AChE (
Table 6).
The inhibitor concentration threshold required for AChE inhibition varied between compounds with Gal producing the lowest concentration required for AChE inhibition. Below these concentrations, no statistically significant inhibition of AChE was detected. The following dose-response curves for each of the active compounds along with the corresponding IC
50 values are shown in
Figure 1.
From this data, it was concluded that of the compounds tested, EGCG was the most efficacious novel cholinesterase inhibitor with the most potent IC50 value.
Some synergistic effects were observed with similar percentage inhibition obtained for the synergistic mixture (99.22%) and EGCG (82.35%) despite concentrations of the inhibitors being four times lower in the synergy mixture than when the compounds were tested individually. Despite these findings, none of the novel compounds or the synergy mixture was found to inhibit AChE to the same extent as Gal.
3.2. AChE Inhibition Kinetics
The kinetics of AChE inhibition was then determined for each of the active compounds using the reciprocal L-B plots shown in
Figure 2.
From these plots, the V
MAX and K
M were calculated (
Table 7), allowing the type of enzyme inhibition present to be confirmed.
Gal and EGCG were found to competitively inhibit AChE (
Figure 1,
Table 7), whereas ECG and EGC were found to be uncompetitive inhibitors of AChE.
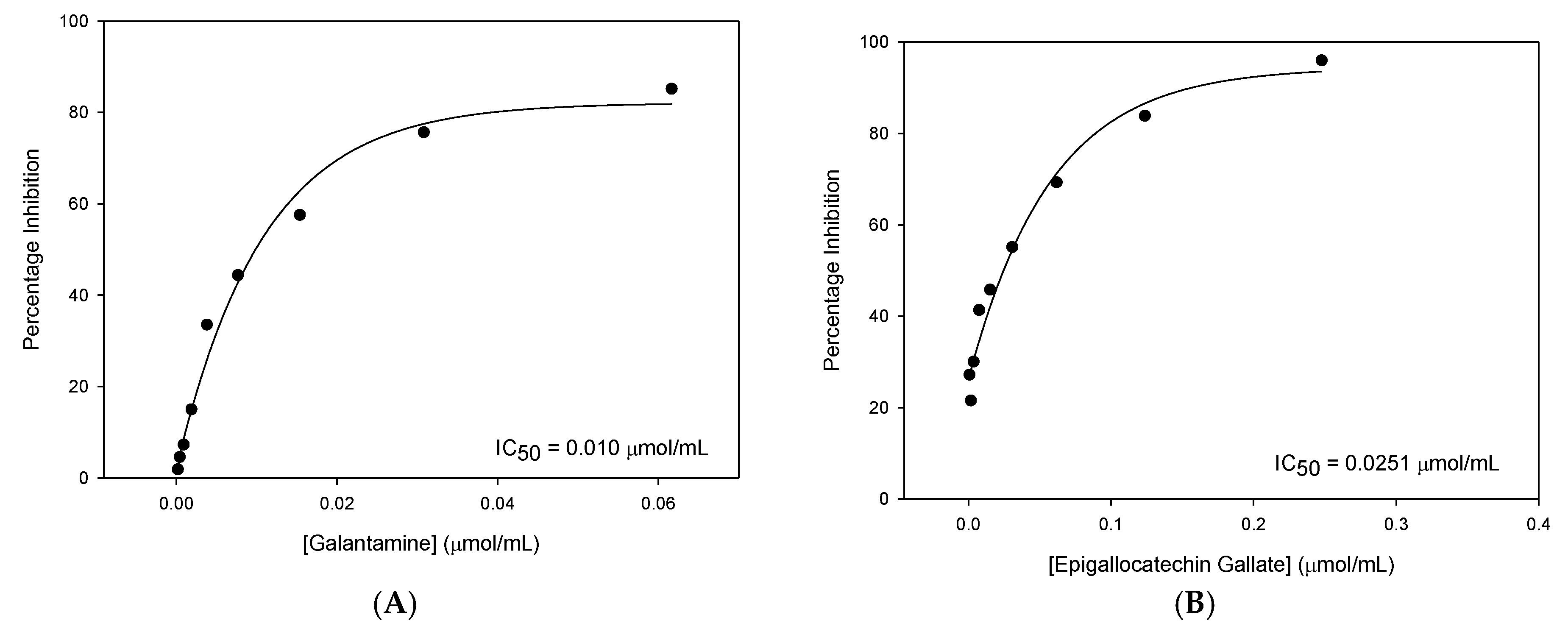
3.3. BuChE Inhibition
Compounds were then tested for their ability to inhibit BuChE. Percentage inhibition and the corresponding
p-values obtained from two-sample t-tests are shown in
Table 8.
Gal and EGCG were the only compounds that showed extensive inhibition of BuChE, which was high enough for IC50 values to be calculated. Whilst ECG (45.65%), EGC (47.72%) and the synergy mixture (56.02%) did show statistically significant inhibition, this extent of inhibition at high concentrations is unlikely to have any relevance clinically. Consequently, these inhibitors were not investigated any further concerning BuChE inhibition.
IC
50 values were then calculated and again Gal was found to be more potent than EGCG in terms of BuChE inhibition (
Table 9).
Gal was 2.51× more potent than EGCG. This is a much smaller difference, suggesting reduced Gal affinity for the BuChE enzyme.
A one-way ANOVA with post-hoc Tukey analysis revealed the minimum inhibitor concentration required for statistically significant inhibition BuChE to be achieved (
Table 10).
The lowest concentration of EGCG tested was sufficient to produce significant inhibition. Consequently, a higher minimum concentration of Gal was required to achieve statistically significant inhibition of BuChE. The IC
50 plots for BuChE inhibition with Gal and EGCG are shown in
Figure 3.
EGCG was identified as the most effective novel inhibitor of BuChE. Although EGCG was less potent than Gal, the difference in the amount of BuChE inhibition produced is relatively small.
3.4. BuChE Inhibition Kinetics
The kinetics of BuChE inhibition was again determined using L-B plots (
Figure 4).
3.5. Cholinesterase Affinity Comparison
EGCG was the only novel compound identified as a significant inhibitor of both cholinesterase enzymes. Varying affinity of EGCG for the AChE and BuChE enzymes was identified (
Table 12).
Calculated IC50 values demonstrated that Gal was 21.5-fold more selective for AChE than BuChE. A similar but much smaller difference in selectivity was identified for EGCG which had an AChE IC50 value 1.7-fold smaller than the corresponding BuChE IC50 value.
Finally, synergism was observed between flavan-3-ol compounds, producing statistically significant inhibition of both AChE and BuChE (
Table 13).
Synergism was more pronounced for inhibition of AChE, resulting in 43.20% more inhibition than for BuChE.