Oxidative Stress Biomarkers, Nut-Related Antioxidants, and Cardiovascular Disease
Abstract
1. Introduction
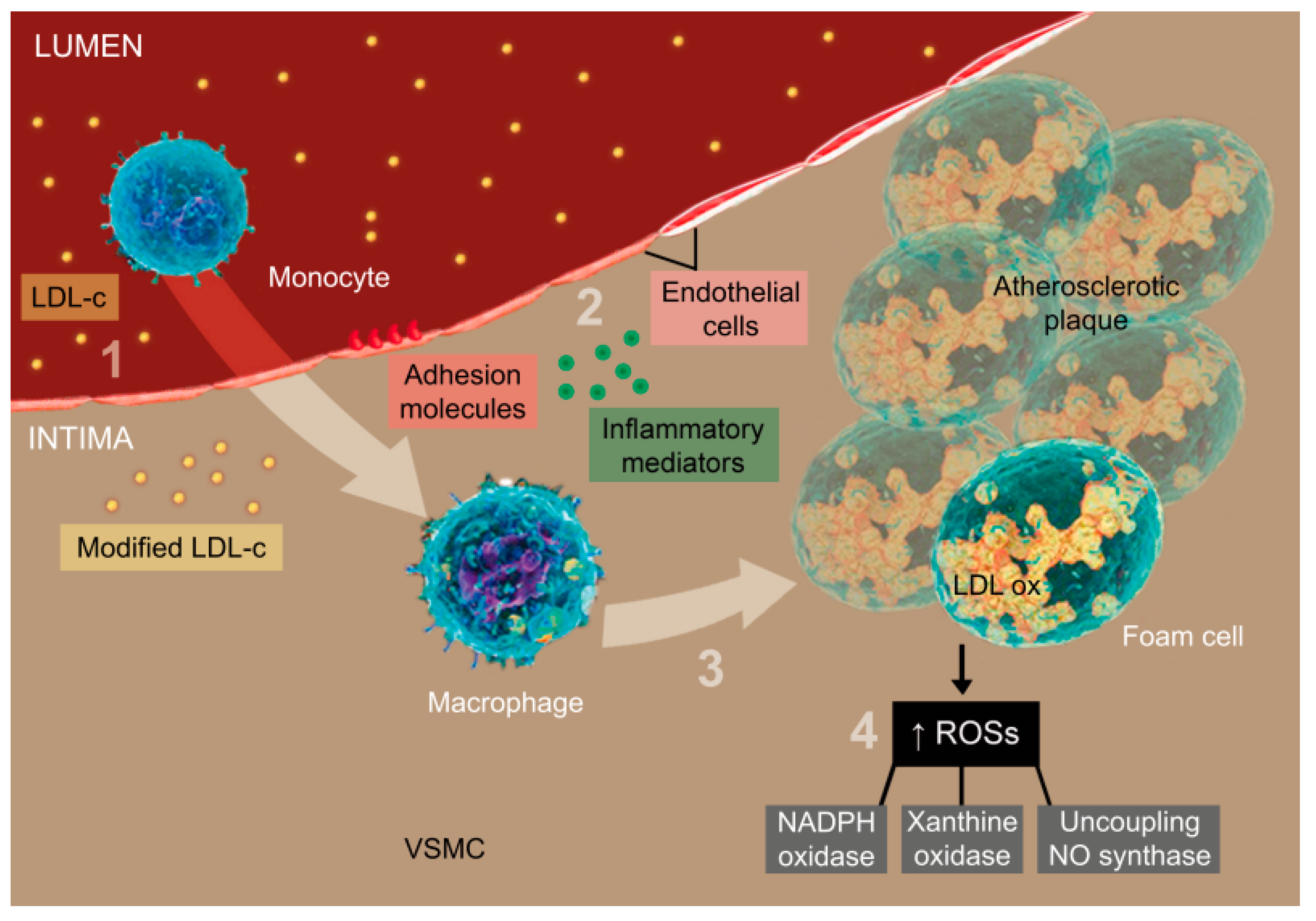
2. The Role of Oxidative Stress in the Development of Atherosclerosis and ACS
3. Nuts and Their Antioxidant Properties
Definition and Nutritional Composition of Different Nuts
4. Nuts, Antioxidants and Oxidative Stress: Studies In Vitro and in Animal Models
4.1. Studies In Vitro
4.2. Studies in Animal Models
5. Nuts and Oxidative Stress Biomarkers: Studies in Primary Cardiovascular Prevention
6. Nuts, Oxidative Stress Biomarkers, and Secondary Cardiovascular Prevention
7. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Aschoff, L. Introduction. In Arteriosclerosis: A Survey of Problem; Cowdry, E.V., Ed.; Macmillan: New York, NY, USA, 1933; pp. 1–18. [Google Scholar]
- Lippi, G.; Sanchis-Gomar, F.; Cervellin, G. Chest pain, dyspnea and other symptoms in patients with type 1 and 2 myocardial infarction. A literature review. Int. J. Cardiol. 2016, 215, 20–22. [Google Scholar] [CrossRef]
- Ambrose, J.; Singh, M. Pathophysiology of coronary artery disease leading to acute coronary syndromes. F1000Prime Rep. 2015, 7. [Google Scholar] [CrossRef]
- World Health Organization (WHO). The Top 10 Causes of Death. Available online: https://www.who.int/news-room/fact-sheets/detail/the-top-10-causes-of-death (accessed on 10 January 2020).
- Liguori, I.; Russo, G.; Francesco, C.; Bulli, G.; Della-Morte, D.; Gargiulo, G.; Testa, G.; Cacciatore, F.; Bonaduce, D.; Abete, P. Oxidative stress, aging, and diseases. Clin. Interv. Aging 2018, 13, 757–772. [Google Scholar] [CrossRef]
- Lefer, D.J.; Granger, D.N. Oxidative stress and cardiac disease. Am. J. Med. 2000, 109, 315–323. [Google Scholar] [CrossRef]
- Barbosa, K.B.F.; Costa, N.M.B.; Alfenas, R.C.G.; Paula, S.O.; Minim, V.P.R.; Bressan, J. Estresse oxidativo: Conceito, implicações e fatores modulatórios. Rev. Nut. 2010, 23, 629–643. [Google Scholar] [CrossRef]
- Kim, H.; Yun, J.; Kwon, S.M. Therapeutic Strategies for Oxidative Stress-Related Cardiovascular Diseases: Removal of Excess Reactive Oxygen Species in Adult Stem Cells. Oxidative Med. Cell. Longev. 2016, 2016, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Siti, H.N.; Kamisah, Y.; Kamsiah, J. The role of oxidative stress, antioxidants and vascular inflammation in cardiovascular disease (a review). Vascul. Pharmacol. 2015, 71, 40–56. [Google Scholar] [CrossRef] [PubMed]
- Chaalal, M.; Ouchemoukh, S.; Mehenni, C.; Salhi, N.; Soufi, O.; Ydjedd, S.; Louaileche, H. Phenolic contents and in vitro antioxidant activity of four commonly consumed nuts in algeria. Acta Aliment. 2018, 48, 1–7. [Google Scholar] [CrossRef]
- Morel, A.; Hamed, A.I.; Oleszek, W.; Stochmal, A.; Głowacki, R.; Olas, B. Protective action of proanthocyanidin fraction fromMedemia argunnuts against oxidative/nitrative damages of blood platelet and plasma components. Platelets 2014, 25, 75–80. [Google Scholar] [CrossRef]
- Yang, L.; Xian, D.; Xiong, X.; Lai, R.; Song, J.; Zhong, J. Proanthocyanidins against Oxidative Stress: From Molecular Mechanisms to Clinical Applications. BioMed Res. Int. 2018, 85844136, 1–11. [Google Scholar] [CrossRef]
- Macedo, T.J.S.; Barros, V.R.P.; Monte, A.P.O.; Gouveia, B.B.; Bezerra, M.É.S.; Cavalcante, A.Y.P.; Matos, M.H.T. Resveratrol has dose-dependent effects on DNA fragmentation and mitochondrial activity of ovine secondary follicles cultured in vitro. Zygote 2017, 25, 434–442. [Google Scholar] [CrossRef] [PubMed]
- Lux, S.; Scharlau, D.; Schlörmann, W.; Birringer, M.; Glei, M. In vitro fermented nuts exhibit chemopreventive effects in HT29 colon cancer cells. Br. J. Nut. 2012, 108, 1177–1186. [Google Scholar] [CrossRef] [PubMed]
- Paterniti, I.; Impellizzeri, D.; Cordaro, M.; Siracusa, R.; Bisignano, C.; Gugliandolo, E.; Cuzzocrea, S. The Anti-Inflammatory and Antioxidant Potential of Pistachios (Pistacia vera L.) In Vitro and In Vivo. Nutrients 2017, 9, 915. [Google Scholar] [CrossRef] [PubMed]
- Laubertová, L.; Koňariková, K.; Gbelcová, H.; Ďuračková, Z.; Žitňanová, I. Effect of walnut oil on hyperglycemia-induced oxidative stress and pro-inflammatory cytokines production. Eur. J. Nut. 2015, 54, 291–299. [Google Scholar] [CrossRef]
- Sheng, J.; Yang, X.; Chen, J.; Peng, T.; Yin, X.; Liu, W.; Yang, X. Antioxidative effects and mechanism study of bioactive peptides from defatted walnut (Juglans regia L.) meal hydrolysate. J. Agric. Food Chem. 2019, 67, 3305–3312. [Google Scholar] [CrossRef]
- Anderson, K.J.; Teuber, S.S.; Gobeille, A.; Cremin, P.; Waterhouse, A.L.; Steinberg, F.M. Walnut Polyphenolics Inhibit In Vitro Human Plasma and LDL Oxidation. J. Nutr. 2001, 131, 2837–2842. [Google Scholar] [CrossRef]
- Anselmo, N.A.; Paskakulis, L.C.; Garcias, R.C.; Botelho, F.F.R.; Toledo, G.Q.; Cury, M.F.R. Prior intake of Brazil nuts attenuates renal injury induced by ischemia and reperfusion. Braz. J. Nephrol. 2018, 40, 10–17. [Google Scholar] [CrossRef]
- Choi, Y.; Abdelmegeed, M.A.; Song, B.-J. Preventive effects of dietary walnuts on high-fat-induced hepatic fat accumulation, oxidative stress and apoptosis in mice. J. Nut. Biochem. 2016, 38, 70–80. [Google Scholar] [CrossRef]
- Hong, M.; Groven, S.; Marx, A.; Rasmussen, C.; Beidler, J. Anti-Inflammatory, Antioxidant, and Hypolipidemic Effects of Mixed Nuts in Atherogenic Diet-Fed Rats. Molecules 2018, 23, 3126. [Google Scholar] [CrossRef]
- Surra, J.C.; Barranquero, C.; Torcal, M.P.; Orman, I.; Segovia, J.C.; Guillén, N.; Navarro, M.A.; Arnal, C.; Osada, J. In comparison with pal oil, dietary nut supplementation delays the progression of atherosclerotic lesions in famale apoE-deficient mice. Br. J. Nutr. 2013, 109, 202–209. [Google Scholar] [CrossRef]
- Avila, J.A.D.; Alvarez-Parrilla, E.; López-Díaz, J.A.; Maldonado-Mendoza, I.; Gómez-García, M.D.C.; De La Rosa, L.A. The pecan nut (Carya illinoinensis) and its oil and polyphenolic fractions differentially modulate lipid metabolism and the antioxidant enzyme activities in rats fed high-fat diets. Food Chem. 2015, 168, 529–537. [Google Scholar] [CrossRef] [PubMed]
- Alturfan, A.A.; Emekeli-Alturfan, E.; Uslu, E. Consumption of pistachio nuts beneficially affect blood lipids and total antioxidant activity in rats fed a high-cholesterol diet. Folia Biol. (Praha) 2009, 55, 132–136. [Google Scholar] [PubMed]
- Bitok, E.; Sabaté, J. Nuts and Cardiovascular Disease. Prog. Cardiovasc. Dis. 2018, 61, 33–37. [Google Scholar] [CrossRef] [PubMed]
- De Souza, R.G.M.; Gomes, A.C.; Navarro, A.M.; Cunha, L.C.; da Silva, M.A.C.; Junior, F.B.; Mota, J.F. Baru Almonds Increase the Activity of Glutathione Peroxidase in Overweight and Obese Women: A Randomized, Placebo-Controlled Trial. Nutrients 2019, 11, 1750. [Google Scholar] [CrossRef] [PubMed]
- Duarte, G.B.S.; Reis, B.Z.; Rogero, M.M.; Vargas-Mendez, E.; Barbosa, F.; Cercato, C.; Cozzolino, S.M.F. Consumption of Brazil nuts with high selenium levels increased inflammation biomarkers in obese women: A randomized controlled trial. Nutrition 2019, 63, 162–168. [Google Scholar] [CrossRef] [PubMed]
- Huguenin, G.V.; Oliveira, G.M.M.; Moreira, A.S.B.; Saint’Pierre, T.D.; Gonçalves, R.A.; Pinheiro-Mulder, A.R.; Rosa, G. Improvement of antioxidant status after Brazil nut intake in hypertensive and dyslipidemic subjects. Nutr. J. 2015, 14, 1–10. [Google Scholar] [CrossRef]
- Gulati, S.; Misra, A.; Pandey, R.M.; Bhatt, S.P.; Saluja, S. Effects of pistachio nuts on body composition, metabolic, inflammatory and oxidative stress parameters in Asian Indians with metabolic syndrome: A 24-wk, randomized control trial. Nutrition 2015, 30, 192–197. [Google Scholar] [CrossRef]
- Liu, J.F.; Liu, Y.-H.; Chen, C.-M.; Chang, W.-H.; Chen, C.-Y.O. The effect of almonds on inflammation and oxidative stress in Chinese patients with type 2 diabetes mellitus: A randomized crossover controlled feeding trial. Eur. J. Nut. 2012, 52, 927–935. [Google Scholar] [CrossRef]
- Maranhão, P.A.; Kraemer-Aguiar, L.G.; de Oliveira, C.L.; Kuschnir, M.C.; Vieira, Y.R.; Souza, M.G.; Bouskela, E. Brazil nuts intake improves lipid profile, oxidative stress and microvascular function in obese adolescents: A randomized controlled trial. Nutr. Metab. 2011, 8, 1–8. [Google Scholar] [CrossRef]
- López-Uriarte, P.; Nogués, R.; Saez, G.; Bulló, M.; Romeu, M.; Masana, L.; Salas-Salvadó, J. Effect of nut consumption on oxidative stress and the endothelial function in metabolic syndrome. Clin. Nutr. 2010, 29, 373–380. [Google Scholar] [CrossRef]
- Campisi, J. Senescent cells, tumor suppression, and organismal aging: Good citizens, bad neighbors. Cell 2005, 120, 513–522. [Google Scholar] [CrossRef] [PubMed]
- Gardner, S.E.; Humphry, M.; Bennett, M.R.; Clarke, M.C. Senescent vascular smooth muscle cells drive inflammation through an interleukin-1α-dependent senescence-associated secretory phenotype. Arterioscler. Thromb. Vasc. Biol. 2015, 35, 1963–1974. [Google Scholar] [CrossRef] [PubMed]
- Acosta, J.C.; Banito, A.; Wuestefeld, T.; Georgilis, A.; Janich, P.; Morton, J.P.; Athineos, D.; Kang, T.W.; Lasitschka, F.; Andrulis, M.; et al. A complex secretory program orchestrated by the inflammasome controls paracrine senescence. Nat. Cell Biol. 2013, 15, 978–990. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Horke, S.; Förstermann, U. Vascular oxidative stress, nitric oxide and atherosclerosis. Atherosclerosis 2014, 237, 208–219. [Google Scholar] [CrossRef]
- Sakakura, K.; Nakano, M.; Otsuka, F.; Ladich, E.; Kolodgie, F.D.; Virmani, R. Pathophysiology of atherosclerosis plaque progression. Heart Lung Circ. 2013, 22, 399–411. [Google Scholar] [CrossRef]
- Förstermann, U. Oxidative stress in vascular disease: Causes, defense mechanisms and potential therapies. Nat. Clin. Pract. Cardiovasc. Med. 2008, 5, 338–349. [Google Scholar] [CrossRef]
- Förstermann, U. Nitric oxide and oxidative stress in vascular disease. Pflügers Arch.-Eur. J. Physiol. 2010, 459, 923–939. [Google Scholar] [CrossRef]
- Glaudemans, A.W.J.M.; Slart, R.H.J.A.; Bozzao, A.; Bonanno, E.; Arca, M.; Dierckx, R.A.J.O.; Signore, A. Molecular imaging in atherosclerosis. Eur. J. Nucl. Med. Mol. Imaging 2010, 37, 2381–2397. [Google Scholar] [CrossRef][Green Version]
- Judd, W.S.; Campbell, C.S.; Kellogg, E.A.; Stevens, P.F.; Donoghue, M.J. Taxonomic evidence: Structural and biochemical characters. In Plant Systematics: A Phylogentetic Approach, 2nd ed.; Judd, W.S., Campbell, C.S., Kellogg, E.A., Stevens, P.F., Donoghue, M.J., Eds.; Massachusetts: Sunderland, UK, 2002; pp. 55–104. [Google Scholar]
- Freitas, J.; Fernandes, D.; Czeder, L.; Lima, J.; Sousa, A.; Naves, M. Edible Seeds and Nuts Grown in Brazil as Sources of Protein for Human Nutrition. Food Nutr. Sci. 2012, 3, 857–862. [Google Scholar] [CrossRef]
- Venkatachalam, M.; Sathe, S.K. Chemical composition of selected edible nut seeds. J. Agric. Food Chem. 2006, 54, 4705–4714. [Google Scholar] [CrossRef]
- US Department of Agriculture. USDA National Nutrition Database for Standard Reference; Release 26; USDA: Springfield, VA, USA, 2013. [Google Scholar]
- Miraliakbari, H.; Shahidi, F. Lipid class compositions, tocopherols and sterols of tree nut oils extracted with different solvents. J. Food Lipids 2008, 15, 81–96. [Google Scholar] [CrossRef]
- Bolling, B.W.; Chen, C.Y.; McKay, D.L.; Blumberg, J.B. Tree nut phytochemicals: Composition, antioxidant capacity, bioactivity, impact factors: A systematic review of almonds, Brazils, cashews, hazelnuts, macadamias, pecans, pine nuts, pistachios and walnuts. Nutr. Res. Rev. 2011, 24, 244–275. [Google Scholar] [CrossRef] [PubMed]
- Alasalvar, C.; Bolling, B.W. Review of nut phytochemicals, fat-soluble bioactives, antioxidant components and health effects. Br. J. Nutr. 2015, 113, 68–78. [Google Scholar] [CrossRef] [PubMed]
- Cardoso, B.R.; Duarte, G.B.S.; Reis, B.Z.; Cozzolino, S.M.F. Brazil nuts: Nutritional composition, health benefits and safety aspects. Food Res Int. 2017, 100, 9–18. [Google Scholar] [CrossRef]
- De Souza, R.G.M.; Schincaglia, R.M.; Pimentel, G.D.; Mota, J.F. Nuts and Human Health Outcomes: A Systematic Review. Nutrients 2017, 9, 1311. [Google Scholar] [CrossRef]
- Sánchez-González, C.; Ciudad, C.J.; Noé, V.; Izquierdo-Pulido, M. Health benefits of walnut polyphenols: An exploration beyond their lipid profile. Crit. Rev. Food Sci. Nutr. 2017, 57, 3373–3383. [Google Scholar] [CrossRef]
- Kim, Y.; Keogh, J.B.; Clifton, P.M. Benefits of Nut Consumption on Insulin Resistance and Cardiovascular Risk Factors: Multiple Potential Mechanisms of Actions. Nutrients 2017, 9, 1271. [Google Scholar] [CrossRef]
- Freitas, J.B.; Naves, M.M.V. Chemical composition of nuts and edible seeds and their relation to nutrition and health. Rev. Nutr. 2010, 23, 269–279. [Google Scholar] [CrossRef]
- Vera, R.; Soares Junior, M.S.; Naves, R.V.; Souza, E.R.B.; Fernandes, E.P.; Caliari, M.; Leandro, W.M. Características químicas de amêndoas de barueiros (dipteryx alata vog.) de ocorrência natural no cerrado do estado de Goiás, Brasil. Rev. Bras. Frutic. 2009, 31, 112–118. [Google Scholar] [CrossRef]
- Alasalvar, C.; Shahidi, F. Natural antioxidants in tree nuts. Eur. J. Lipid Technol. 2009, 111, 1056–1062. [Google Scholar] [CrossRef]
- Pérez-Jiménez, J.; Neveu, V.; Vos, F.; Scalbert, A. Identification of the 100 richest dietary sources of polyphenols: An application of the Phenol-Explorer database. Eur. J. Clin. Nutr. 2010, 64, S112–S120. [Google Scholar] [CrossRef] [PubMed]
- Bolling, B.W.; McKay, D.L.; Blumberg, J.B. The phytochemical composition and antioxidant actions of tree nuts. Asia Pac. J. Clin. Nutr. 2010, 19, 117–123. [Google Scholar] [PubMed]
- Ryan, E.; Galvin, K.; O’Connor, T.P.; Maguire, A.R.; O’Brien, N.M. Fatty acid profile, tocopherol, squalene and phytosterol content of brazil, pecan, pine, pistachio and cashew nuts. Int. J. Food Sci. Nutr. 2006, 54, 219–228. [Google Scholar] [CrossRef] [PubMed]
- Dugo, G.; La Pera, L.; Lo Turco, V.; Mavrogeni, E.; Alfa, M. Determination of selenium in nuts by cathodictripping potentiometry (CSP). J. Agric. Food Chem. 2003, 51, 3722–3725. [Google Scholar] [CrossRef] [PubMed]
- Stevens-Barrón, J.C.; de la Rosa, L.A.; Wall-Medrano, A.; Álvarez-Parrilla, E.; Rodríguez-Ramirez, R.; Robles-Zepeda, R.E.; Astiazaran-García, H. Chemical Composition and In Vitro Bioaccessibility of Antioxidant Phytochemicals from Selected Edible Nuts. Nutrients 2019, 11, 2303. [Google Scholar] [CrossRef]
- USDA—United States Department of Agriculture. Available online: https://fdc.nal.usda.gov/ (accessed on 31 December 2019).
- Amaral, J.S.; Alves, M.R.; Seabra, R.M.; Oliveira, B.P.P. Vitamin E compositions of walnuts (Juglans regia L.): A 3-year comparative study of different cultivars. J. Agric. Food Chem. 2005, 53, 5467–5472. [Google Scholar] [CrossRef] [PubMed]
- Köksal, A.I.; Artik, N.A.; Şimşek, A.; Güne, N. Nutrient composition of hazelnut (Corylus avellana L.) varieties cultivated in Turkey. Food Chem. 2006, 99, 509–515. [Google Scholar] [CrossRef]
- Shin, E.C.; Huang, Y.Z.; Pegg, R.B.; Phillips, R.D.; Eitenmiller, R.R. Commercial runner peanut cultivars in the United States: Tocopherol composition. J. Agric. Food Chem. 2009, 57, 10289–10295. [Google Scholar] [CrossRef]
- Estruch, R.; Ros, E.; Salas-Salvadó, J.; Covas, M.I.; Corella, D.; Arós, F.; Gómez-Gracia, E.; Ruiz-Gutiérrez, V.; Fiol, M.; Lapetra, J.; et al. Primary Prevention of Cardiovascular Disease with a Mediterranean Diet Supplemented with Extra-Virgin Olive Oil or Nuts. N. Engl. J. Med. 2018, 378, e34. [Google Scholar] [CrossRef]
- Arnett, D.K.; Blumenthal, R.S.; Albert, M.A.; Buroker, A.B.; Goldberger, Z.D.; Hahn, E.J.; Himmelfarb, C.D.; Khera, A.; Lloyd-Jones, D.; McEvoy, J.W.; et al. 2019 ACC/AHA Guideline on the Primary Prevention of Cardiovascular Disease: A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Circulation 2019, 140, e596–e646. [Google Scholar]
- Fraser, G.E.; Sabaté, J.; Beeson, W.L.; Strahan, T.M. A possible protective effect of nut consumption on risk of coronary heart disease. The Adventist Health Study. Arch. Intern. Med. 1992, 152, 1416–1424. [Google Scholar] [CrossRef] [PubMed]
- Ellsworth, J.L.; Kushi, L.H.; Folsom, A.R. Frequent nut intake and risk of death from coronary heart disease and all causes in postmenopausal women: The Iowa Women’s Health Study. Nutr. Metab. Cardiovasc. Dis. 2001, 11, 372–377. [Google Scholar] [PubMed]
- Bao, Y. Association of nut consumption with total and cause-specific mortality. Indian Heart J. 2014, 66, 388–389. [Google Scholar] [CrossRef]
- Hshieh, T.T.; Petrone, A.B.; Djoussé, L. Nut consupmtion and risk of mortality in the Physicians Health Study. Am. J. Clin. Nutr. 2015, 101, 407–412. [Google Scholar] [CrossRef] [PubMed]
- Sureda, A.; Bibiloni, M.D.; Martorell, M.; Buil-Cosiales, P.; Marti, A.; Pons, A.; Tur, J.A.; Martinez-Gonzalez, M.Á.; PREDIMED Study Investigators. Mediterranean diets supplemented with virgin olive oil and nuts enhance plasmatic antioxidant capabilities and decrease xanthine oxidase activity in people with metabolic syndrome: The PREDIMED study. Mol. Nutr. Food Res. 2016, 60, 2654–2664. [Google Scholar] [CrossRef] [PubMed]
- Zamora-Ros, R.; Serafini, M.; Estruch, R.; Lamuela-Raventós, R.M.; Martínez-González, M.A.; Salas-Salvadó, J.; Andres-Lacueva, C. Mediterranean diet and non enzymatic antioxidant capacity in the PREDIMED study: Evidence for a mechanism of antioxidant tuning. Nutr. Metab. Cardiovasc. Dis. 2013, 23, 1167–1174. [Google Scholar] [CrossRef]
- Berryman, C.E.; Grieger, J.A.; West, S.G.; Chen, C.-Y.O.; Blumberg, J.B.; Rothblat, G.H.; Kris-Etherton, P.M. Acute Consumption of Walnuts and Walnut Components Differentially Affect Postprandial Lipemia, Endothelial Function, Oxidative Stress, and Cholesterol Efflux in Humans with Mild Hypercholesterolemia. J. Nutr. 2013, 143, 788–794. [Google Scholar] [CrossRef]
- Stockler-Pinto, M.B.; Mafra, D.; Moraes, C.; Lobo, J.; Boaventura, G.T.; Farage, N.E.; Malm, O. Brazil Nut (Bertholletia excelsa, H.B.K.) Improves Oxidative Stress and Inflammation Biomarkers in Hemodialysis Patients. Biol. Trace Elem. Res. 2014, 158, 105–112. [Google Scholar]
- Bento, A.P.N.; Cominetti, C.; Simões Filho, A.; Naves, M.M.V. Baru almond improves lipid profile in mildly hypercholesterolemic subjects: A randomized, controlled, crossover study. Nutr. Metab. Cardiovasc. Dis. 2014, 24, 1330–1336. [Google Scholar] [CrossRef]
- Darvish, D.R.; Mousavi, S.N.; Shidfar, F.; Mohammadi, V.; Rajab, A. Effects of Daily Consumption of Cashews on Oxidative Stress and Atherogenic Indices in Patients with Type 2 Diabetes: A Randomized, Controlled-Feeding Trial. Int. J. Endocrinol. Metab. 2019, 17, e70744. [Google Scholar]
- Guaraldi, F.; Deon, V.; Del Bo’, C.; Vendrame, S.; Porrini, M.; Riso, P.; Guardamagna, O. Effect of short-term hazelnut consumption on DNA damage and oxidized LDL in children and adolescents with primary hyperlipidemia: A randomized controlled trial. J. Nutr. Biochem. 2018, 57, 206–211. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.J.; Nam, G.E.; Seo, J.A.; Yoon, T.; Seo, I.; Lee, J.H.; Kim, N.H. Nut consumption has favorable effects on lipid profiles of Korean women with metabolic syndrome. Nutr. Res. 2014, 34, 814–820. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.Y.; Holbrook, M.; Duess, M.A.; Dohadwala, M.M.; Hamburg, N.M.; Asztalos, B.F.; Milbury, P.E.; Blumberg, J.B.; Vita, J.A. Effect of almond consumption on vascular function in patients with coronary artery disease: A randomized, controlled, cross-over trial. Nutr. J. 2015, 17, 61. [Google Scholar] [CrossRef] [PubMed]
- Thomazella, M.C.; Góes, M.F.; Andrade, C.R.; Debbas, V.; Barbeiro, D.F.; Correia, R.L.; Marie, S.K.; Cardounel, A.J.; daLuz, P.L.; Laurindo, F.R. Effects of high adherence to mediterranean or low-fat diets in medicated secondary prevention patients. Am. J. Cardiol. 2011, 108, 1523–1529. [Google Scholar] [CrossRef] [PubMed]
- Stephens, N.G.; Parsons, A.; Schofield, P.M.; Kelly, F.; Cheeseman, K.; Mitchinson, M.J. Randomised controlled trial of vitamina E in patients with coronary disease: Cambridge Heart Antioxidant Study (CHAOS). Lancet 1996, 23, 781–786. [Google Scholar] [CrossRef]
- Rapola, J.M.; Virtamo, J.; Ripatti, S.; Huttunen, J.K.; Albanes, D.; Taylor, P.R.; Heinonen, O.P. Randomised trial of alpha-tocopherol and beta-carotene supplements on incidence of major coronary events in men with previous myocardial infarction. Lancet 1997, 349, 1715–1720. [Google Scholar] [CrossRef]
- GISSI-Prevenzione Investigators. Dietary supplementation with n-3 polyunsaturated fatty acids and vitamin E after myocardial infarction: Results of the GISSI-Prevenzione trial. Gruppo Italiano per lo Studio della Sopravvivenza nell’Infarto miocardico. Lancet 1999, 354, 447–455. [Google Scholar]
- Wang, T.; Lin, X. Circulating Vitamina E Levels and Risk of Coronary Artery Disease and Myocardial Infarction: A Mendelian Randomization Study. Nutrients 2019, 11, 2153. [Google Scholar] [CrossRef]
| Fatty Acids (g/100g of Lipids) | Nuts | Edible Seeds | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Almond | Hazelnut | Macadamia | Walnut | Pecan | Pistachio | Brazil Nut | Cashew Nut | Peanut | Baru Almond | |
| Saturated | 9.19 | 8.25 | 16.09 | 9.81 | 7.33 | 14.6 | 25.35 | 21.12 | 14.81 | 18.77 |
| Palmitic C16:0 | 7.43 | 5.57 | 8.88 | 7.15 | 5.09 | 12.61 | 15.11 | 10.7 | 7.2 | 7.39 |
| Stearic C18:0 | 1.7 | 2.5 | 4.26 | 2.55 | 2.02 | 1.42 | 9.51 | 9.33 | 1.84 | 4.62 |
| Arachid C20:0 | 0.06 | 0.14 | 2.95 | 0.07 | 0.06 | 0.35 | 0.25 | 0.63 | 1.19 | 1.10 |
| MUFA | 65.89 | 80.62 | 58.51 | 16.30 | 54.26 | 56.28 | 29.04 | 61.68 | 43.93 | 51.07 |
| Palmitoleic C16:1 | 0.66 | 0.15 | 18.69 | 0.09 | 0.07 | 1.18 | 0.29 | 0.54 | 0.07 | - |
| Oleic C18:1 | 65.89 | 80.52 | 58.51 | 16.14 | 53.65 | 55.98 | 28.75 | 61.15 | 42.48 | 48.37 |
| Gadoleic C20:1 | 0.00 | 0.10 | 0.00 | 0.16 | 0.61 | 0.3 | - | - | 1.45 | 2.70 |
| PUFA | 23.95 | 10.57 | 4.39 | 72.79 | 37.95 | 27.11 | 45.61 | 17.19 | 37.81 | 32.35 |
| Linoleic C18:2 | 23.85 | 10.43 | 1.81 | 60.23 | 37 | 26.55 | 45.43 | 16.88 | 37.52 | 30.13 |
| Linolenic C 18:3 | 0.10 | 0.14 | 2.58 | 12.56 | 0.95 | 0.56 | 0.18 | 0.32 | 0.29 | 2.22 |
| Phytochemical | Nuts | |||||||
|---|---|---|---|---|---|---|---|---|
| Almond | Hazelnut | Macadamia | Walnut | Pecan | Pistachio | Brazil Nut | Cashew Nut | |
| Total phenolics (mg/100 g) | 261 | 447 | 233 | 1602 | 1588 | 703 | 197 | 242 |
| Proanthocyanidins (mg/100 g) | 184.1 | 500.6 | 10 | 67.2 | 493.9 | 252.71 | 10 | 8.7 |
| Flavonoids (mg/100 g) | 25.01 | 13.21 | 137.9 | 0.54 | 2713.49 | 136.45 | 0.85 | 1.12 |
| Phenolic acids and aldehydes (mg/100 g) | 0.44 | 1.87 | 3.69 | 39.11 | 2052 | 1.27 | 11.35 | - |
| Carotenoids (µg/100 g) | 2 | 106 | - | 21 | 55 | 22832 | - | 31 |
| Sterols (mg/100 g) | 192.37 | 132.47 | 105.7 | 197.89 | 233.52 | 189.43 | 160.19 | 154 |
| Cashew Nut [60] | Brazil Nut [57,60] | Almond [60] | Walnut [60,61] | Pecan [60] | Pistachio [60] | Macadamia [60] | Hazelnut [60,62] | Peanut [60,63] | |
|---|---|---|---|---|---|---|---|---|---|
| Selenium (µg) | 19.9 | 1917 | 4.1 | 4.9 | 3.8 | 7 | 3.6 | 2.4 | 9.3 |
| Zinc (mg) | 5.78 | 4.06 | 3.12 | 3.09 | 4.53 | 2.2 | 1.3 | 2.45 | 2.77 |
| Vitamin A (μg) | 0 | 0 | 0 | 1 | 3 | 26 | 0 | 1 | 0 |
| Vitamin C (mg) | 0.5 | 0.7 | 0 | 1.3 | 1.1 | 5.6 | 1.2 | 6.3 | 0 |
| α-tocopherol (mg) | 0.9 | 5.65 | 25.63 | 0.7 | 1.4 | 2.86 | 0.54 | 15.03 | 4.93 |
| β-tocopherol (mg) | 0.03 | - | 0.23 | 0.10 | 0.39 | 0 | 0 | - | 0.33 |
| γ-tocopherol (mg) | 5.31 | 116.2 | 0.64 | 22.65 | 24.44 | 1.67 | 0 | 1.36 | 10.4 |
| Author; Location | RCT Design | Population (n: Intervention/Control) | Type of Nut | Duration | Intervention | Control | Biomarkers | Main Results | Conclusion |
|---|---|---|---|---|---|---|---|---|---|
| de Souza RGM et al. [26]; Brazil | Placebo-controlled | Women overweight and obese (n: 24/22) | Baru nut | 8 weeks | Normocaloric and isocaloric diet-based Guideline + 20g Baru/day | Normocaloric and isocaloric diet-based Guideline | MDA, catalase, GPx, SOD | GPx intervention: +0.08; GPx control: -0.07. p <0.01 | Baru nut supplementation increased GPx activity in women with excess of weight. |
| GBS Duarte et al. [27]; Brazil | Controlled | Women obese (n: 36/36) | Brazil nut | 2 months | Usual diet + 1 unit Brazil nut/day | Usual diet | GPx1 | GPx1 intervention: Δ8.5; GPx1 control: Δ 2.5. p =0.03 | Brazil nut supplementation increased GPx1 activity in obese women. |
| Darvish Damavandi R et al. [75]; Iran | Controlled | Individuals with T2DM (n: 22/21) | Cashew nut | 8 weeks | Adjusted calorie diet with 10% of cashew and reduced consumption of visible fat | Usual diet | TAC and PON-1 | Intervention vs control: TAC (p= 0.34); PON-1 (p= 0.41). | Cashew nut did not improve TAC and PON-1 plasma activities in individuals with T2DM. |
| Guaraldi F et al. [76]; Italy | Controlled | Children and adolescents with hyperlipidemia (n: 42/18) | Hazelnut with or without skin | 8 weeks | Nutritional recommendations based on CHILD-1 + hazelnut with or out skin 15 to 30g/day | Nutritional recommendations based on CHILD-1 | oxLDL | oxLDL intervention vs control: p= 0.462. | Hazelnuts did not improve the oxLDL in children and adolescents with hyperlipidemia. |
| Huguenin GV et al. [28]; Brazil | Cross, double-blind, placebo-controlled | Individuals hypertension and dyslipidemia (n: 52/48) | Brazil nut partially defatted | 12 weeks | Nutritional counseling for dyslipidemia and hypertension + Brazil nut 13g/day | Nutritional counseling for dyslipidemia and hypertension | GPx3, TAC, 8-epi PGF2α, oxLDL | GPx3 and oxLDL intervention: 128.32±38.31 60.68±20.88, respectively. GPx3 and oxLDL control: 115.06±38.09 and 63.76 ±23.03, respectively. p <0.05 | Brazil nut intake increased GPx3 actitity and reduced oxLDL in individuals with hypertension and dyslipidemia. |
| Lee YJ et al. [77]; South Korea | Controlled | Individuals with MS (n: 30/30) | Mixed nuts (walnut, peanuts, and pine nuts) | 6 weeks | Recommendations-based dietary guidelines + mix nuts 30g / day | Recommendations-based dietary guidelines | MDA, oxLDL | Serum and urine MDA and oxLDL intervention vs control: p= 0.445, p= 0.394, and p= 0.885, respectively. | Mixed nuts did not improve MDA and oxLDL in individuals with MS. |
| Bento AP et al. [74]; Brazil | Placebo-controlled, crossover | Mildly hypercholesterolemic (n: 20/20) | Baru nut | 6 weeks | Usual diet + Baru 20g /day | Usual diet | TBARS, SOD, FRAP | TBARS, SOD, FRAP intervention vs control: p= 0.82, p =0.34, and p =0.33, respectively. | Baru nuts did not improve TBARS, SOD and FRAP in mildly hypercholesterolemic individuals. |
| Gulati S et al. [29]; India | Controlled | Asian Indian with MS (n: 33/35) | Pistachio nut unsalted | 24 weeks | Guideline based on the standard diet with 20% of TEV in pistachio | Guideline based on the standard diet | TBARS | TBARS intervention: 2.4±1.3; TBARS control: 3.1±1.3. p= 0.01 | Pistachio nut improved plasma TBARS in Asian Indian with MS. |
| Liu JF et al. [30]; Taiwan | Cross and controlled | Individuals with T2DM and mild hyperlipidemia (n: 20/10) | Almond nut | 12 weeks | Diet for obtaining or maintaining weight with 20% of TEV in almond | Diet for obtaining or maintaining weight | carbonyl protein, oxLDL and MDA | Carbonyl protein intervention: 1.59±0.16; carbonyl protein control: 2.16 ±0.23 (p= 0.0003). oxLDL: reduced 6,9% with intervention as compared control. p ≤0.05 | Almond reduced plasma carbonyl protein and oxLDL in individuals with T2DM and mild hyperlipidemia. |
| Maranhao PA et al. [31]; Brazil | Controlled | Obese adolescents (n: 8/9) | Brazil nuts | 16 weeks | Usual diet + Brazil nut 15-25g/day | Usual diet | GPx3, oxLDL, 8-epi PGF2α | oxLDL: reduced in intervention group (622.4 to 514.9). p=0.02 | Brazil nuts improved oxLDL in adolescents with obesity. |
| López-Uriarte P et al. [32]; Spain | Controlled | Individuals with MS (n: 25/25) | Mixed nuts (walnut, almond and hazelnut) | 12 weeks | Nutritional guidelines + mix nuts 30g/day | Nutritional guidelines | plasma antioxidant capacity, oxLDL,8-oxo-dG, 8-isoprostane | Mean difference in final 8-oxo-dG (intervention vs control): Δ -2.42; p ≤0.001 | Mixed nuts improved urinary 8-oxo-dG in individuals with MS. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lorenzon dos Santos, J.; Schaan de Quadros, A.; Weschenfelder, C.; Bueno Garofallo, S.; Marcadenti, A. Oxidative Stress Biomarkers, Nut-Related Antioxidants, and Cardiovascular Disease. Nutrients 2020, 12, 682. https://doi.org/10.3390/nu12030682
Lorenzon dos Santos J, Schaan de Quadros A, Weschenfelder C, Bueno Garofallo S, Marcadenti A. Oxidative Stress Biomarkers, Nut-Related Antioxidants, and Cardiovascular Disease. Nutrients. 2020; 12(3):682. https://doi.org/10.3390/nu12030682
Chicago/Turabian StyleLorenzon dos Santos, Julia, Alexandre Schaan de Quadros, Camila Weschenfelder, Silvia Bueno Garofallo, and Aline Marcadenti. 2020. "Oxidative Stress Biomarkers, Nut-Related Antioxidants, and Cardiovascular Disease" Nutrients 12, no. 3: 682. https://doi.org/10.3390/nu12030682
APA StyleLorenzon dos Santos, J., Schaan de Quadros, A., Weschenfelder, C., Bueno Garofallo, S., & Marcadenti, A. (2020). Oxidative Stress Biomarkers, Nut-Related Antioxidants, and Cardiovascular Disease. Nutrients, 12(3), 682. https://doi.org/10.3390/nu12030682





