Impacts of Dietary Macronutrient Pattern on Adolescent Body Composition and Metabolic Risk: Current and Future Health Status—A Narrative Review
Abstract
1. Introduction
2. Trends in Obesity and Related Metabolic Disease in Childhood and Adolescence
3. Factors Influencing Adolescent Obesity and Body Composition
4. Association between Dietary Macronutrient Intake Pattern, Obesity, and Related Metabolic Status in Children and Adolescents
4.1. Definition of Low- or High-Macronutrient Dietary Compositions
4.2. Beneficial Effect of Dietary Macronutrient Modulation (DMM) on Body Composition and Metabolic Outcomes
4.3. Non-Effectiveness of DMM on Body Composition and Metabolic Outcomes
5. Additional Factors Determining the Effectiveness of DMM on Body Adiposity and Metabolic Improvement
5.1. Study Period
5.2. Typical Eating Habits
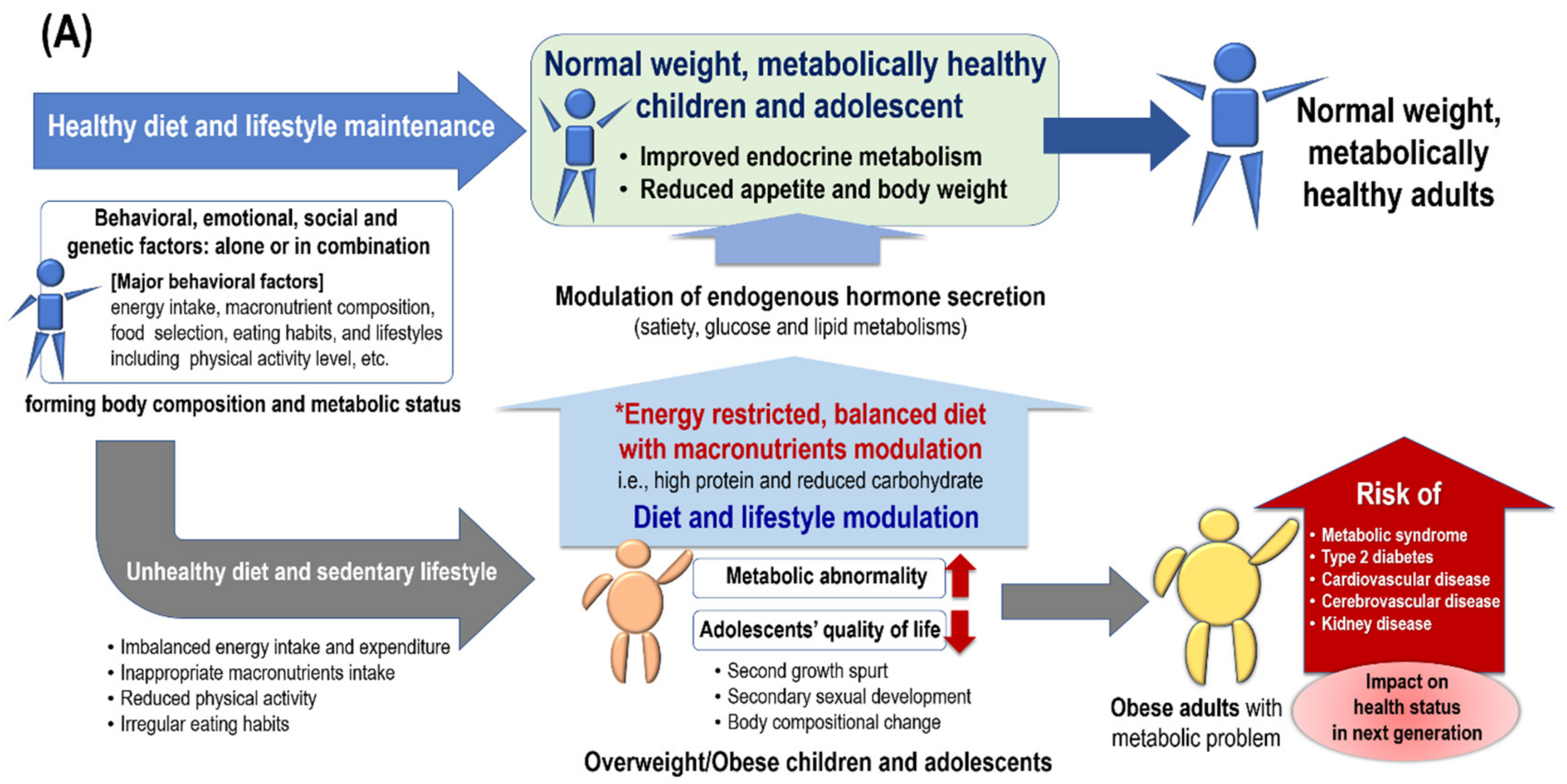
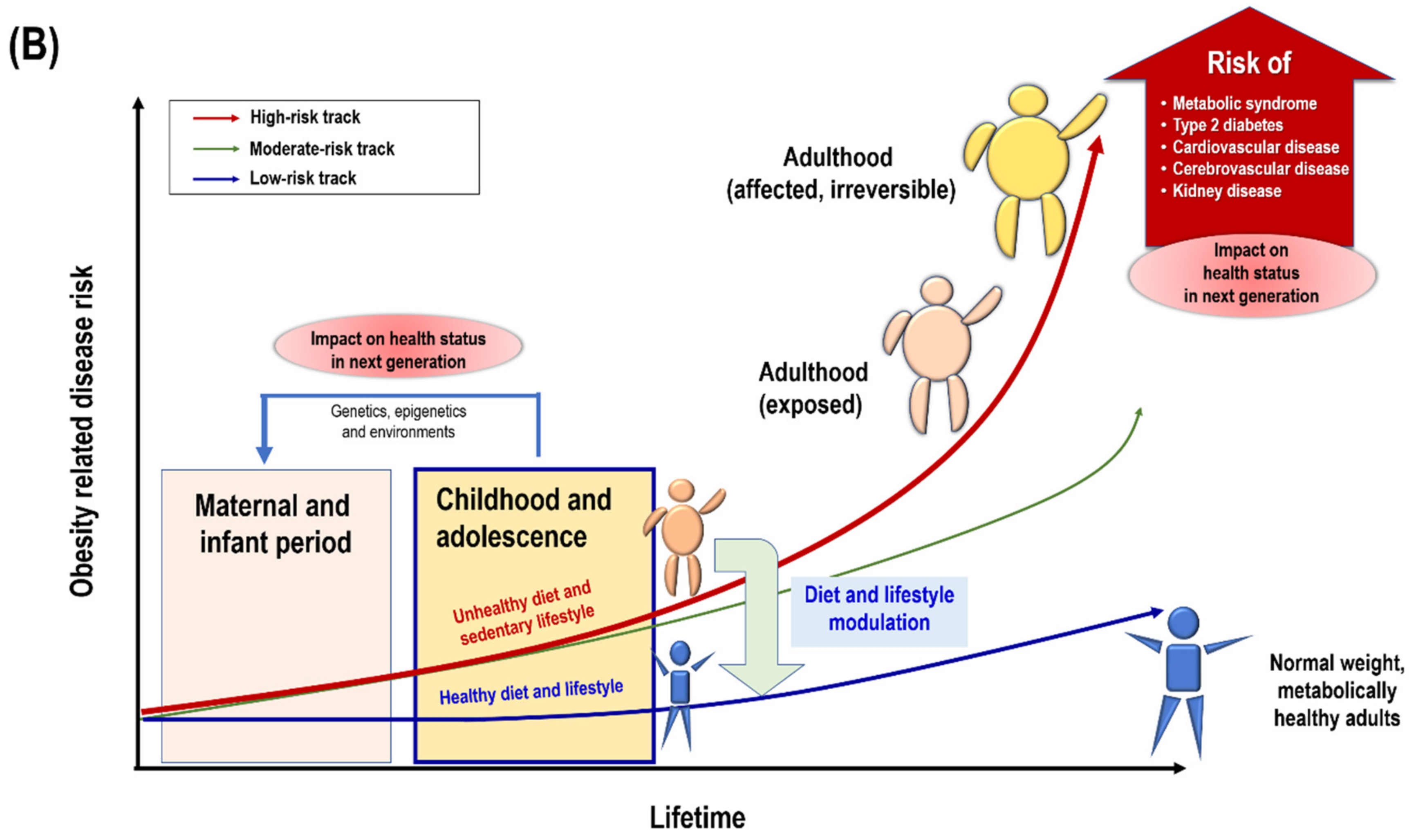
6. Effects of Adolescent Macronutrient Intake on Body Composition and Metabolic Status in Adulthood and Consequent Effects on the Health of the Next Generation
7. Summary
Author Contributions
Funding
Conflicts of Interest
References
- World Health Organization. WHO Obesity and Overweight Fact Sheet. No. 311; World Health Organization: Geneva, Switzerland, 2016. [Google Scholar]
- Bhupathiraju, S.N.; Hu, F.B. Epidemiology of Obesity and Diabetes and Their Cardiovascular Complications. Circ. Res. 2016, 118, 1723–1735. [Google Scholar] [CrossRef]
- Gupta, N.; Goel, K.; Shah, P.; Misra, A. Childhood Obesity in Developing Countries: Epidemiology, Determinants, and Prevention. Endocr. Rev. 2012, 33, 48–70. [Google Scholar] [CrossRef] [PubMed]
- Inchley, J.; Currie, D.; Jewell, J.; Breda, J.; Barnekow, V. (Eds.) Adolescent Obesity and Related Behaviours: Trends and Inequalities in the WHO European Region, 2002–2014. Observation from the Health Behavior in School-Aged Children (HBSC) WHO Collaborative Cross-National Study; World Health Organization Regional Office for Europe: Copenhagen, Denmark, 2017. [Google Scholar]
- Biro, F.M.; Wien, M. Childhood obesity and adult morbidities. Am. J. Clin. Nutr. 2010, 91, 1499S–1505S. [Google Scholar] [CrossRef] [PubMed]
- Freedman, D.S.; Khan, L.K.; Serdula, M.K.; Dietz, W.H.; Srinivasan, S.R.; Berenson, G.S. The Relation of Childhood BMI to Adult Adiposity: The Bogalusa Heart Study. Pediatrics 2005, 115, 22–27. [Google Scholar] [CrossRef]
- Mustillo, S.; Worthman, C.; Erkanli, A.; Keeler, G.; Angold, A.; Costello, E.J. Obesity and Psychiatric Disorder: Developmental Trajectories. Pediatrics 2003, 111, 851–859. [Google Scholar] [CrossRef]
- Strauss, R.S. Childhood obesity and self-esteem. Pediatrics 2000, 105, e15. [Google Scholar] [CrossRef]
- Swallen, K.C.; Reither, E.N.; Haas, S.A.; Meier, A.M. Overweight, Obesity, and Health-Related Quality of Life among Adolescents: The National Longitudinal Study of Adolescent Health. Pediatrics 2005, 115, 340–347. [Google Scholar] [CrossRef]
- Collins, C.; Warren, J.; Neve, M.; McCoy, P.; Stokes, B. Measuring effectiveness of dietetic interventions in child obesity: A systematic review of randomized trials. Arch. Pediatr. Adolesc. Med. 2006, 160, 906–922. [Google Scholar] [CrossRef]
- Wycherley, T.P.; Moran, L.; Clifton, P.M.; Noakes, M.; Brinkworth, G.D. Effects of energy-restricted high-protein, low-fat compared with standard-protein, low-fat diets: A meta-analysis of randomized controlled trials. Am. J. Clin. Nutr. 2012, 96, 1281–1298. [Google Scholar] [CrossRef]
- Gow, M.L.; Ho, M.; Burrows, T.L.; Baur, L.A.; Stewart, L.; Hutchesson, M.J.; Cowell, C.T.; Collins, C.E.; Garnett, S.P. Impact of dietary macronutrient distribution on BMI and cardiometabolic outcomes in overweight and obese children and adolescents: A systematic review. Nutr. Rev. 2014, 72, 453–470. [Google Scholar] [CrossRef]
- Ervin, R.B.; Ogden, C.L. Trends in intake of energy and macronutrients in children and adolescents from 1999-2000 through 2009–2010. NCHS Data Brief 2013, 113, 1–8. [Google Scholar]
- Sunehag, A.L.; Toffolo, G.; Campioni, M.; Bier, D.M.; Haymond, M.W. Effects of Dietary Macronutrient Intake on Insulin Sensitivity and Secretion and Glucose and Lipid Metabolism in Healthy, Obese Adolescents. J. Clin. Endocrinol. Metab. 2005, 90, 4496–4502. [Google Scholar] [CrossRef] [PubMed]
- Nguo, K.; Bonham, M.; Truby, H.; Barber, E.; Brown, J.; Huggins, C.E. Effect of Macronutrient Composition on Appetite Hormone Responses in Adolescents with Obesity. Nutrients 2019, 11, 340. [Google Scholar] [CrossRef] [PubMed]
- Nguo, K.; Huggins, C.E.; Truby, H.; Brown, J.; Bonham, M.P. Effect of macronutrient composition on meal-induced thermogenesis in adolescents with obesity. Eur. J. Nutr. 2019, 58, 2327–2333. [Google Scholar] [CrossRef] [PubMed]
- Truby, H.; Baxter, K.; Ware, R.S.; Jensen, D.E.; Cardinal, J.W.; Warren, J.M.; Daniels, L.; Davies, P.S.W.; Barrett, P.; Blumfield, M.L.; et al. A Randomized Controlled Trial of Two Different Macronutrient Profiles on Weight, Body Composition and Metabolic Parameters in Obese Adolescents Seeking Weight Loss. PLoS ONE 2016, 11, e0151787. [Google Scholar] [CrossRef]
- An, J.; Yoon, S.R.; Lee, J.H.; Kim, H.; Kim, O.Y. Importance of Adherence to Personalized Diet Intervention in Obesity Related Metabolic Improvement in Overweight and Obese Korean Adults. Clin. Nutr. Res. 2019, 8, 171–183. [Google Scholar] [CrossRef]
- Figueroa-Colon, R.; Von Almen, T.K.; Franklin, F.A.; Schuftan, C.; Suskind, R.M. Comparison of Two Hypocaloric Diets in Obese Children. Am. J. Dis. Child. 1993, 147, 160–166. [Google Scholar] [CrossRef]
- Schwartz, C.; King, N.A.; Perreira, B.; Blundell, J.E.; Thivel, D. A systematic review and meta-analysis of energy and macronutrient intake responses to physical activity interventions in children and adolescents with obesity. Pediatr. Obes. 2017, 12, 179–194. [Google Scholar] [CrossRef]
- Demol, S.; Yackobovitch-Gavan, M.; Shalitin, S.; Nagelberg, N.; Gillon-Keren, M.; Phillip, M. Low-carbohydrate (low & high-fat) versus high-carbohydrate low-fat diets in the treatment of obesity in adolescents. Acta Paediatr. 2008, 98, 346–351. [Google Scholar] [CrossRef]
- Duckworth, L.C.; Gately, P.J.; Radley, D.; Cooke, C.B.; King, R.F.; Hill, A.J. RCT of a High-protein Diet on Hunger Motivation and Weight-loss in Obese Children: An Extension and Replication. Obesity 2009, 17, 1808–1810. [Google Scholar] [CrossRef]
- Mirza, N.M.; Palmer, M.G.; Sinclair, K.B.; McCarter, R.; He, J.; Ebbeling, C.B.; Ludwig, D.S.; A Yanovski, J. Effects of a low glycemic load or a low-fat dietary intervention on body weight in obese Hispanic American children and adolescents: A randomized controlled trial. Am. J. Clin. Nutr. 2013, 97, 276–285. [Google Scholar] [CrossRef]
- Garnett, S.P.; Gow, M.L.; Ho, M.; Baur, L.A.; Noakes, M.; Woodhead, H.J.; Broderick, C.R.; Burrell, S.; Chisholm, K.; Halim, J.; et al. Optimal Macronutrient Content of the Diet for Adolescents with Prediabetes; RESIST a Randomised Control Trial. J. Clin. Endocrinol. Metab. 2013, 98, 2116–2125. [Google Scholar] [CrossRef]
- Lomenick, J.P.; Clasey, J.L.; Anderson, J.W. Meal-related Changes in Ghrelin, Peptide YY, and Appetite in Normal Weight and Overweight Children. Obesity 2008, 16, 547–552. [Google Scholar] [CrossRef]
- Smeets, A.J.; Soenen, S.; Luscombe-Marsh, N.D.; Ueland, O.; Westerterp-Plantenga, M.S. Energy Expenditure, Satiety, and Plasma Ghrelin, Glucagon-Like Peptide 1, and Peptide Tyrosine-Tyrosine Concentrations following a Single High-Protein Lunch. J. Nutr. 2008, 138, 698–702. [Google Scholar] [CrossRef]
- Haroun, D.; Croker, H.; Viner, R.M.; Williams, J.E.; Darch, T.S.; Fewtrell, M.S.; Eaton, S.; Wells, J.C.K. Validation of BIA in Obese Children and Adolescents and Re-evaluation in a Longitudinal Study. Obesity 2009, 17, 2245–2250. [Google Scholar] [CrossRef]
- Institute of Medicine, Subcommittee on Nutritional Status Weight Gain during Pregnancy, Subcommittee on Dietary Intake and Nutrient Supplements during Pregnancy. Nutrition during Pregnancy: Part I, Weight Gain: Part II, Nutrient Supplements; The National Academies Press: Washington, DC, USA, 1990.
- Gray, C.A.; Bartol, F.F.; Tarleton, B.J.; Wiley, A.A.; Johnson, G.A.; Bazer, F.W.; Spencer, T.E. Developmental Biology of Uterine Glands. Biol. Reprod. 2001, 65, 1311–1323. [Google Scholar] [CrossRef]
- Hales, C.M.; Carroll, M.D.; Fryar, C.D.; Ogden, C.L. Prevalence of Obesity Among Adults and Youth: United States, 2015–2016. NCHS Data Brief 2017, 288, 1–8. [Google Scholar]
- Health at a Glance 2017. OECD Indicators. OECD Library. Available online: https://www.oecd-ilibrary.org/social-issues-migration-health/health-at-a-glance-2017_health_glance-2017-en (accessed on 16 May 2020).
- Korean Health Promotion Institute. Korea Health Promotion Forum; Korean Health Promotion Institute: Wonju City, Korea, 2018.
- Ministry of Education. 2018 School Health Statistics Report; Ministry of Education: Sejong City, Korea, 2019.
- Webb, V.L.; Wadden, T.A. Intensive Lifestyle Intervention for Obesity: Principles, Practices, and Results. Gastroenterology 2017, 152, 1752–1764. [Google Scholar] [CrossRef]
- Fock, K.M.; Khoo, J. Diet and exercise in management of obesity and overweight. J. Gastroenterol. Hepatol. 2013, 28, 59–63. [Google Scholar] [CrossRef]
- Ryan, D.H.; Frühbeck, G.; Ryan, D.H.; Wilding, J.P.H. Management of obesity. Lancet 2016, 387, 1947–1956. [Google Scholar] [CrossRef]
- Report of the Commission on Ending Childhood Obesity; World Health Organization: Geneva, Switzerland, 2016. Available online: http://www.who.int/end-childhood-obesity/news/launch-final-report/en/ (accessed on 16 August 2020).
- Bowen, J.; Noakes, M.; Trenerry, C.; Clifton, P.M. Energy Intake, Ghrelin, and Cholecystokinin after Different Carbohydrate and Protein Preloads in Overweight Men. J. Clin. Endocrinol. Metab. 2006, 91, 1477–1483. [Google Scholar] [CrossRef]
- Institute of Medicine Food and Nutrition Board. Dietary Reference Intakes for Energy, Carbohydrate, Fiber, Fat, Fatty Acids, Cholesterol, Protein, and Amino Acids (Macronutrients); National Academies Press: Washington, DC, USA, 2005.
- European Food Safety Authority. Dietary Reference Values for Nutrients. Summary Report. EFSA Supporting Publication. 2017, e15121. Available online: https://www.efsa.europa.eu/en/supporting/pub/e15121 (accessed on 16 August 2020).
- NNR Project Group. Nordic Nutrition Recommendations 2004: Integrating Nutrition And Physical Activit, 4th ed.; Nordic Council of Ministers: Copenhagen, Denmark, 2005.
- Becker, W.; Lyhne, N.; Pedersen, A.N.; Aro, A.; Fogelholm, M.; Phorsdottir, I.; Alexander, J.; Anderssen, S.A.; Meltzer, H.M.; Pedersen, J.I. Nordic Nutrition Recommendations 2004—Integrating nutrition and physical activity. Scandi J. Nutr. 2004, 48, 178–187. [Google Scholar] [CrossRef]
- Australian National Health and Medical Research Council and the New Zealand Ministry of Health. Nutrient Reference Values for Australia and New Zealand: Including Recommended Dietary Intakes; Australian National Health and Medical Research Council and the New Zealand Ministry of Health: Canberra, Australia, 2006.
- Atkins Nutritionals. New Atkins. Atkins Nutritionals 2011. Available online: http://sa.atkins.com/ (accessed on 16 August 2020).
- WebMD. The Atkins Diet. WebMD, 2012. Available online: http://www.webmd.com/diet/atkins-dietwhat-it-is (accessed on 19 November 2020).
- Zone Labs. Dr Sears Zone Diet. Zone Labs Inc. 2012. Available online: http://www.zonediet.com/ (accessed on 16 August 2020).
- Barnett, J. The Zone Diet Explained. Crossfit Impulse, 2009. Available online: http://crossfitimpulse.com/thezone-diet-explained-edited (accessed on 17 May 2020).
- Ministry of Health and Welfare. Korean Nutrition Society. Dietary Reference Intake for Koreans; Ministry of Health and Welfare: Sejong City, Korea, 2015.
- Westerterp-Plantenga, M.S.; Nieuwenhuizen, A.; Tomé, D.; Soenen, S.; Westerterp, K. Dietary Protein, Weight Loss, and Weight Maintenance. Annu. Rev. Nutr. 2009, 29, 21–41. [Google Scholar] [CrossRef] [PubMed]
- Skov, A.R.; Toubro, S.; Rønn, B.; Holm, L.; Astrup, A. Randomized trial on protein vs carbohydrate in ad libitum fat reduced diet for the treatment of obesity. Int. J. Obes. 1999, 23, 528–536. [Google Scholar] [CrossRef]
- Westerterp-Plantenga, M.S.; Lejeune, M.P.; Nijs, I.; Van Ooijen, A.; Kovacs, E. High protein intake sustains weight maintenance after body weight loss in humans. Int. J. Obes. Relat. Metab. Disord. 2004, 28, 57–64. [Google Scholar] [CrossRef]
- Larsen, T.M.; Dalskov, S.M.; van Baak, M.; Jebb, S.A.; Papadaki, A.; Pfeiffer, A.F.; Martinez, J.A.; Handjieva-Darlenska, T.; Kunešová, M.; Pihlsgård, M.; et al. Diets with High or Low Protein Content and Glycemic Index for Weight-Loss Maintenance. N. Engl. J. Med. 2010, 363, 2102–2113. [Google Scholar] [CrossRef]
- Gögebakan, Ö.; Kohl, A.; Osterhoff, M.A.; van Baak, M.A.; Jebb, S.A.; Papadaki, A.; Martinez, J.A.; Handjieva-Darlenska, T.; Hlavaty, P.; Weickert, M.O.; et al. Effects of weight loss and long-term weight maintenance with diets varying in protein and glycemic index on cardiovascular risk factors: The diet, obesity, and genes (DiOGenes) study: A randomized, controlled trial. Circulation 2011, 124, 2829–2838. [Google Scholar] [CrossRef]
- Foster-Schubert, K.E.; Overduin, J.; Prudom, C.E.; Liu, J.; Callahan, H.S.; Gaylinn, B.D.; Thorner, M.O.; Cummings, D.E. Acyl and Total Ghrelin Are Suppressed Strongly by Ingested Proteins, Weakly by Lipids, and Biphasically by Carbohydrates. J. Clin. Endocrinol. Metab. 2008, 93, 1971–1979. [Google Scholar] [CrossRef]
- Hrolfsdottir, L.; Halldorsson, T.I.; Rytter, R.; Bech, B.H.; Birgisdottir, B.E.; Gunnarsdottir, I.; Granström, C.; Henriksen, T.B.; Olsen, S.F.; Maslova, E. Maternal Macronutrient Intake and Offspring Blood Pressure 20 Years Later. J. Am. Heart Assoc. 2017, 6, e005808. [Google Scholar] [CrossRef]
- Kereliuk, S.M.; Brawerman, G.M.; Dolinsky, V.W. Maternal Macronutrient Consumption and the Developmental Origins of Metabolic Disease in the Offspring. Int. J. Mol. Sci. 2017, 18, 1451. [Google Scholar] [CrossRef]
- Correa-Rodríguez, M.; Rueda-Medina, B.; González-Jiménez, E.; Schmidt-RioValle, J. Associations between body composition, nutrition, and physical activity in young adults. Am. J. Hum. Biol. 2017, 29, e22903. [Google Scholar] [CrossRef] [PubMed]
- Cameron, J.D.; Sigal, R.J.; Kenny, G.P.; Alberga, A.S.; Prud’Homme, D.; Phillips, P.; Doucette, S.; Goldfield, G.S. Body composition and energy intake—Skeletal muscle mass is the strongest predictor of food intake in obese adolescents: The HEARTY trial. Appl. Physiol. Nutr. Metab. 2016, 41, 611–617. [Google Scholar] [CrossRef] [PubMed]
- Verreijen, A.M.; Engberink, M.F.; Memelink, R.G.; Van Der Plas, S.E.; Visser, M.; Weijs, P.J.M. Effect of a high protein diet and/or resistance exercise on the preservation of fat free mass during weight loss in overweight and obese older adults: A randomized controlled trial. Nutr. J. 2017, 16, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Layman, D.K.; Evans, E.M.; Erickson, D.; Seyler, J.; Weber, J.; Bagshaw, D.; Griel, A.; Psota, T.; Kris-Etherton, P. A Moderate-Protein Diet Produces Sustained Weight Loss and Long-Term Changes in Body Composition and Blood Lipids in Obese Adults. J. Nutr. 2009, 139, 514–521. [Google Scholar] [CrossRef]
- Layman, D.K.; Evans, E.; Baum, J.I.; Seyler, J.; Erickson, D.J.; Boileau, R.A. Dietary Protein and Exercise Have Additive Effects on Body Composition during Weight Loss in Adult Women. J. Nutr. 2005, 135, 1903–1910. [Google Scholar] [CrossRef]
- Helms, E.R.; Zinn, C.; Rowlands, D.S.; Brown, S.R. A Systematic Review of Dietary Protein during Caloric Restriction in Resistance Trained Lean Athletes: A Case for Higher Intakes. Int. J. Sport Nutr. Exerc. Metab. 2014, 24, 127–138. [Google Scholar] [CrossRef]
- Rolland-Cachera, M.F.; Thibault, H.; Souberbielle, J.C.; Soulié, D.; Carbonel, P.; Deheeger, M.; Roinsol, D.; Longueville, E.; Bellisle, F.; Serog, P. Massive obesity in adolescents: Dietary interventions and behaviours associated with weight regain at 2 y follow-up. Int. J. Obes. Relat. Metab. Disord. 2004, 28, 514–519. [Google Scholar] [CrossRef]
- Naude, C.E.; Schoonees, A.; Senekal, M.; Young, T.; Garner, P.; Volmink, J. Low carbohydrate versus isoenergetic balanced diets for reducing weight and cardiovascular risk: A systematic review and meta-analysis. PLoS ONE 2014, 9, e100652. [Google Scholar] [CrossRef]
- Alhussain, M.H.; Macdonald, I.A.; Taylor, M.A. Irregular meal-pattern effects on energy expenditure, metabolism, and appetite regulation: A randomized controlled trial in healthy normal-weight women. Am. J. Clin. Nutr. 2016, 104, 21–32. [Google Scholar] [CrossRef]
- Pot, G.K.; Hardy, R.; Stephen, A.M. Irregularity of energy intake at meals: Prospective associations with the metabolic syndrome in adults of the 1946 British birth cohort. Br. J. Nutr. 2016, 115, 315–323. [Google Scholar] [CrossRef]
- Thayer, Z.M.; Rutherford, J.; Kuzawa, C.W. The Maternal Nutritional Buffering Model: An evolutionary framework for pregnancy nutritional intervention. Evol. Med. Public Health 2020, 2020, 14–27. [Google Scholar] [CrossRef]
- Chen, L.-W.; Aris, I.M.; Bernard, J.Y.; Tint, M.-T.; Colega, M.; Gluckman, P.D.; Tan, K.H.; Shek, L.P.-C.; Chong, Y.-S.; Yap, F.; et al. Associations of maternal macronutrient intake during pregnancy with infant BMI peak characteristics and childhood BMI. Am. J. Clin. Nutr. 2017, 105, 705–713. [Google Scholar] [CrossRef]
- Fernandez-Twinn, D.S.; Wayman, A.P.; Ekizoglou, S.; Martin, M.S.; Hales, C.N.; Ozanne, S.E. Maternal protein restriction leads to hyperinsulinemia and reduced insulin-signaling protein expression in 21-mo-old female rat offspring. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2005, 288, R368–R373. [Google Scholar] [CrossRef]
- Gonzalez, P.N.; Gasperowicz, M.; Barbeito-Andres, J.; Klenin, N.; Cross, J.C.; Hallgrimsson, B. Chronicprotein restriction in mice impacts placental function and maternal body weight before fetal growth. PLoS ONE 2016, 11, e0152227. [Google Scholar] [CrossRef]
- Ozanne, S.E.; Hales, C.N. For Debate: Fetal and early postnatal growth restriction lead to diabetes, the metabolic syndrome and renal failure. Diabetologia 2003, 46, 1013–1019. [Google Scholar] [CrossRef]
- De Brito Alves, J.L.; de Oliveira, J.M.D.; Ferreira, D.J.S.; de V Barros, M.A.; Nogueira, V.O.; Alves, D.S.; Vida, H.; Leandro, C.G.; Lagranha, C.J.; Pirola, L.; et al. Maternal protein restriction induced-hypertension is associated to oxidative disruption at transcriptional and functional levels in the medullaoblongata. Clin. Exp. Pharmacol. Physiol. 2016, 43, 1177–1184. [Google Scholar] [CrossRef]
- Zambrano, E.; Bautista, C.J.; Deás, M.; Martínez-Samayoa, P.M.; González-Zamorano, M.; Ledesma, H.; Morales, J.; Larrea, F.; Nathanielsz, P.W. A low maternal protein diet during pregnancy and lactation has sex-and window of exposure-specific effects on offspring growth and food intake, glucose metabolism and serum leptin in the rat. J. Physiol. 2006, 571, 221–230. [Google Scholar] [CrossRef]
- Blumfield, M.L.; Nowson, C.A.; Hure, A.J.; Smith, R.; Simpson, S.J.; Raubenheimer, D.; MacDonald-Wicks, L.; Collins, C.E. Lower Protein-to-Carbohydrate Ratio in Maternal Diet is Associated with Higher Childhood Systolic Blood Pressure up to Age Four Years. Nutrients 2015, 7, 3078–3093. [Google Scholar] [CrossRef]
- Pereira, T.J.; Fonseca, M.A.; Campbell, K.E.; Moyce, B.L.; Cole, L.K.; Hatch, G.M.; Doucette, C.A.; Klein, J.; Aliani, M.; Dolinsky, V.W. Maternal obesity characterized by gestational diabetes increases the susceptibility of rat offspring to hepatic steatosis via a disrupted liver metabolome. J. Physiol. 2015, 593, 3181–3197. [Google Scholar] [CrossRef]
- Clausen, T.D.; Mathiesen, E.R.; Hansen, T.; Pedersen, O.; Jensen, D.M.; Lauenborg, J.; Schmidt, L.; Damm, P. Overweight and the Metabolic Syndrome in Adult Offspring of Women with Diet-Treated Gestational Diabetes Mellitus or Type 1 Diabetes. J. Clin. Endocrinol. Metab. 2009, 94, 2464–2470. [Google Scholar] [CrossRef]
- Shapiro, A.L.B.; Ringham, B.M.; Glueck, D.H.; Norris, J.M.; Barbour, L.A.; Friedman, J.E.; Dabelea, D.; Glueck, D.H. Infant Adiposity is Independently Associated with a Maternal High Fat Diet but not Related to Niacin Intake: The Healthy Start Study. Matern. Child Health J. 2017, 21, 1662–1668. [Google Scholar] [CrossRef]
- Shapiro, A.L.B.; Schmiege, S.J.; Brinton, J.T.; Glueck, D.; Crume, T.L.; Friedman, J.E.; Dabelea, D. Testing the fuel-mediated hypothesis: Maternal insulin resistance and glucose mediate the association between maternal and neonatal adiposity, the Healthy Start study. Diabetologia 2015, 58, 937–941. [Google Scholar] [CrossRef] [PubMed]
| Ref | Sources | Region | Subjects Characteristics | Study Design | Dietary Macronutrient Information | Main Outcome |
|---|---|---|---|---|---|---|
| [14] | J Clin Endocrinol Metab; 90: 4496–4502, 2005 | Italy | Healthy obesity (boys n = 6; girls n = 7, 14.7 ± 0.3 y, BMI: 34 ± 1 kg/m2, body fat: 42 ± 1%); normal weight adolescents in previous studies | Randomized crossover design (7-d) | Isocaloric/isonitrogenous diet; High-CHO: 60% CHO, 25% FAT; low-CHO: 30% CHO, 55% FAT | Increased total cholesterol, β-OH butyrate, and gluconeogenesis in the low-CHO group |
| [15] | Nutrients; 11: 340, 2019 | Australia | Obesity (n = 8, 16.1 ± 0.4 y, BMI ≥ 95th percentile); healthy weight (n = 12, 16.0 ± 0.6 y, BMI: 5–85th percentile) | Double-blind crossover design | High-CHO: 79% CHO, 5% PRO, 16% FAT. High-PRO: 55% PRO, 30% CHO, 15% FAT | Higher ghrelin reduction in high-PRO group than high-CHO group; no differences in postprandial GLP-1 and PYY between the groups; higher postprandial glucose and insulin in high-CHO group than high-PRO group, particularly among obese subjects than normal weight subjects |
| [16] | Eur J Nutr; 58: 2327–2333, 2019 | Australia | Obesity (n = 13, 15.4 ± 0.5 y, BMI ≥ 95th percentile); healthy weight (n = 13, 16.0 ± 0.6 y, BMI: 5–85th percentile) | Double-blind crossover design | High-CHO: 79% CHO, 5% PRO, 16% FAT. High-PRO: 55% PRO, 30% CHO, 15% FAT | Higher meal-induced thermogenesis in high-PRO group than high-CHO group; higher hunger in high-CHO group than high-PRO group |
| [17] | PLoS ONE; 11: e0151787, 2016 | Australia | Hospitalized overweight /obesity (10–17 y, BMI ≥ 90th percentile): SMC (n = 37, 13.2 y), SLF (n = 36, 13.2 y), control (n = 14, 13.6 y) | Randomized, clinical trial (12-wk) | SMC: 35% CHO, 30% PRO, 35% FAT; SLF: 55% CHO, 20% PRO, 25% FAT | BMI z-score improvement in both SMC and, SLF groups; improvement of insulin resistance, total and LDL-cholesterol, and C-reactive protein in SLF group; improvement of adiponectin and interleukin-6 in SMC group |
| [19] | Am J Dis Child; 147: 160–166, 1993 | USA | Obesity (n = 19, 7.5–16.9 y) | Intervention (14.5-m) | PSMFD: 2520 to 3360 J; HBD: 3360 to 4200 J | Significant weight reduction in both diet groups after 6 months; more weight reduction and fat-free mass maintenance in PSMFD group at 10-wk and 6-m |
| [21] | Pediatr Obes; 12; 179–194, 2017 | Israel | Health obesity (boys n = 21, girls n = 34); 12–18 y (14.4 ± 1.7 y); BMI ≥ 90th percentile | Randomized clinical trial (12-wk) and follow-up (9-m) | LCLF (n = 18): 60g CHO (up to 20%), 30% FAT, 50% PRO; LCHF (n = 17): 60 g CHO (up to 20%), 20% FAT, 60% PRO; HCLF (n = 20): 50–60% CHO, 30% FAT, 20% PRO | No differences in the changes of BMI, BMI percentile, body fat %, and metabolic parameters among the all groups; reduction of insulin and insulin resistance in LCLF and LCHF groups |
| [22] | Obesity; 17;1808–1810, 2009 | USA | Healthy obesity (boys n = 34, girls n = 61, 9–18 y) | Randomized trials (31-d) | SD (n = 49): 50–55% CHO, 15% PRO, 30–35% FAT; High-PRO (n = 46): 40–45% CHO, 25% PRO, 30–35% FAT | Reduction of BMI (−2 ± 0.25 kg/m2) and other anthropometric parameters except body fat and systolic blood pressure in both groups; no differences of height or appetite between the two groups; increased desire for appetite in both groups |
| [23] | Am J Clin Nutr; 97; 276–285, 2013 | Spain | Healthy obesity (boys n = 58, girls n = 55, 7–15 y), BMI ≥ 90th percentile | Randomized clinical trial (12-wk) and follow-up (2-y) | LGD (n = 57): 45–50% CHO-LG), 20–25% PRO, 30–35% FAT; LFD (n = 56): 55–60% CHO-no LGI, 15–20% PRO, 25–30% FAT | Significant reduction of BMI z-score in both groups; no significant differences in the changes of weight loss, insulin resistance, and metabolic syndrome risk factors between LGD and LFD groups |
| [24] | J Clin Endocrinol Metab; 98; 2116–2125 | Australia | Overweight/obesity with prediabetes or insulin resistance (n = 111, boys 45, girls 66, 10–17 y) | Randomized clinical trials (12-m) and follow-up (24-m) * | HCLF (n = 55): 55–60% CHO-MGL, 30% FAT (≤10% saturated fat), 15% PRO; MCIP (n = 56): 40–45% CHO-MGL, 30% FAT(≤10% saturated fat), 25–30% PRO | Increased insulin sensitivity and reduced insulin to glucose in MCIP after 6-m; significant reductions of fasting insulin, SBP z-score, and DBP z-score in HCLF and MCIP groups, but not different between the two groups |
| [63] | Intern J Obes; 28; 514–519, 2004 | France | Healthy obesity (boys n = 32, girls n = 89, 11–16 y, BMI ≥ 97 percentile) | Intervention (9-m) and follow-up (2-y) | Low-PRO (n = 53), 54% CHO, 15% PRO; High-PRO (n = 46), 50% CHO, 19% PRO with energy by breakfast 20%, lunch 31%, afternoon snack 16%, and dinner 33% | Reduction of BMI and BMI z-score in Low-PRO and High-PRO groups after the intervention, but yoyo effect observed after 2-y follow-up; no significant differences in the changes of weight loss (BMI z-score) between the two groups |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, O.Y.; Kim, E.M.; Chung, S. Impacts of Dietary Macronutrient Pattern on Adolescent Body Composition and Metabolic Risk: Current and Future Health Status—A Narrative Review. Nutrients 2020, 12, 3722. https://doi.org/10.3390/nu12123722
Kim OY, Kim EM, Chung S. Impacts of Dietary Macronutrient Pattern on Adolescent Body Composition and Metabolic Risk: Current and Future Health Status—A Narrative Review. Nutrients. 2020; 12(12):3722. https://doi.org/10.3390/nu12123722
Chicago/Turabian StyleKim, Oh Yoen, Eun Mi Kim, and Sochung Chung. 2020. "Impacts of Dietary Macronutrient Pattern on Adolescent Body Composition and Metabolic Risk: Current and Future Health Status—A Narrative Review" Nutrients 12, no. 12: 3722. https://doi.org/10.3390/nu12123722
APA StyleKim, O. Y., Kim, E. M., & Chung, S. (2020). Impacts of Dietary Macronutrient Pattern on Adolescent Body Composition and Metabolic Risk: Current and Future Health Status—A Narrative Review. Nutrients, 12(12), 3722. https://doi.org/10.3390/nu12123722





