Higher Omega-3 Index Is Associated with Better Asthma Control and Lower Medication Dose: A Cross-Sectional Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Subjects
2.2. Sputum Induction and Analysis
2.3. Plasma Inflammatory Markers
2.4. Erythrocyte Membrane Fatty Acid Preparation
2.5. Fatty Acid Determination
2.6. Fatty Acid Calculations
2.7. Statistical Analysis
3. Results
3.1. Comparison of Nonasthmatic and Asthmatic Subjects
3.2. Asthma Clinical Markers, Systemic Inflammation and O3I
3.3. Asthma, O3I and Obesity
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Australian Bureau of Statistics. National Health Survey First Results 2014-15; 4364.0.55.001; Australian Bureau of Statistics: Canberra, Australia, 2015. [Google Scholar]
- Global Asthma Network. The Global Asthma Report; Global Asthma Network: Auckland, New Zealand, 2014. [Google Scholar]
- Beermann, C.; Neumann, S.; Fussbroich, D.; Zielen, S.; Schubert, R. Combinations of distinct long-chain polyunsaturated fatty acid species for improved dietary treatment against allergic bronchial asthma. Nutr. Burbank Los Angeles Cty. Calif. 2016, 32, 1165–1170. [Google Scholar] [CrossRef] [PubMed]
- Wood, L.G.; Baines, K.J.; Fu, J.; Scott, H.A.; Gibson, P.G. The neutrophilic inflammatory phenotype is associated with systemic inflammation in asthma. Chest 2012, 142, 86–93. [Google Scholar] [CrossRef] [PubMed]
- Olafsdottir, I.S.; Gislason, T.; Thjodleifsson, B.; Olafsson, I.; Gislason, D.; Jogi, R.; Janson, C. C reactive protein levels are increased in non-allergic but not allergic asthma: A multicentre epidemiological study. Thorax 2005, 60, 451–454. [Google Scholar] [CrossRef] [PubMed]
- Takemura, M.; Matsumoto, H.; Niimi, A.; Ueda, T.; Matsuoka, H.; Yamaguchi, M.; Jinnai, M.; Muro, S.; Hirai, T.; Ito, Y.; et al. High sensitivity C-reactive protein in asthma. Eur. Respir. J. 2006, 27, 908–912. [Google Scholar] [CrossRef] [PubMed]
- Scott, H.A.; Jensen, M.E.; Wood, L.G. Dietary interventions in asthma. Curr. Pharm. Des. 2014, 20, 1003–1010. [Google Scholar] [CrossRef]
- The Global Initiative for Asthma. GINA Report: Global Strategy for Asthma Management and Prevention. 2007. Available online: http://www.ginasthma.org (accessed on 8 October 2019).
- Barros, R.; Moreira, A.; Padrao, P.; Teixeira, V.H.; Carvalho, P.; Delgado, L.; Lopes, C.; Severo, M.; Moreira, P. Dietary patterns and asthma prevalence, incidence and control. Clin. Exp. Allergy J. Br. Soc. Allergy Clin. Immunol. 2015, 45, 1673–1680. [Google Scholar] [CrossRef]
- Calder, P.C. Marine omega-3 fatty acids and inflammatory processes: Effects, mechanisms and clinical relevance. Biochim. Biophys. Acta 2015, 1851, 469–484. [Google Scholar] [CrossRef]
- Endo, J.; Arita, M. Cardioprotective mechanism of omega-3 polyunsaturated fatty acids. J. Cardiol. 2016, 67, 22–27. [Google Scholar] [CrossRef]
- Olliver, M.; Veysey, M.; Lucock, M.; Niblett, S.; King, K.; MacDonald-Wicks, L.; Garg, M.L. Erythrocyte omega-3 polyunsaturated fatty acid levels are associated with biomarkers of inflammation in older Australians. J. Nutr. Intermed. Metab. 2016, 5, 61–69. [Google Scholar] [CrossRef][Green Version]
- Yokoyama, A.; Hamazaki, T.; Ohshita, A.; Kohno, N.; Sakai, K.; Zhao, G.D.; Katayama, H.; Hiwada, K. Effect of aerosolized docosahexaenoic acid in a mouse model of atopic asthma. Int. Arch. Allergy Immunol. 2000, 123, 327–332. [Google Scholar] [CrossRef]
- Bilal, S.; Haworth, O.; Wu, L.; Weylandt, K.H.; Levy, B.D.; Kang, J.X. Fat-1 transgenic mice with elevated omega-3 fatty acids are protected from allergic airway responses. Biochim. Biophys. Acta 2011, 1812, 1164–1169. [Google Scholar] [CrossRef] [PubMed]
- Farjadian, S.; Moghtaderi, M.; Kalani, M.; Gholami, T.; Hosseini Teshnizi, S. Effects of omega-3 fatty acids on serum levels of T-helper cytokines in children with asthma. Cytokine 2016, 85, 61–66. [Google Scholar] [CrossRef] [PubMed]
- Mickleborough, T.D.; Murray, R.L.; Ionescu, A.A.; Lindley, M.R. Fish oil supplementation reduces severity of exercise-induced bronchoconstriction in elite athletes. Am. J. Respir. Crit. Care Med. 2003, 168, 1181–1189. [Google Scholar] [CrossRef] [PubMed]
- Brannan, J.D.; Bood, J.; Alkhabaz, A.; Balgoma, D.; Otis, J.; Delin, I.; Dahlen, B.; Wheelock, C.E.; Nair, P.; Dahlen, S.E.; et al. The effect of omega-3 fatty acids on bronchial hyperresponsiveness, sputum eosinophilia, and mast cell mediators in asthma. Chest 2015, 147, 397–405. [Google Scholar] [CrossRef]
- Woods, R.K.; Thien, F.C.; Abramson, M.J. Dietary marine fatty acids (fish oil) for asthma in adults and children. Cochrane Database Syst. Rev. 2002. [Google Scholar] [CrossRef]
- Thien, F.C.K.; De Luca, S.; Woods, R.K.; Abramson, M.J. Cochrane Review: Dietary marine fatty acids (fish oil) for asthma in adults and children. Evid. Based Child Health Cochrane Rev. J. 2011, 6, 984–1012. [Google Scholar] [CrossRef]
- Venter, C.; Meyer, R.W.; Nwaru, B.I.; Roduit, C.; Untersmayr, E.; Adel-Patient, K.; Agache, I.; Agostoni, C.; Akdis, C.A.; Bischoff, S.C.; et al. EAACI position paper: Influence of dietary fatty acids on asthma, food allergy and atopic dermatitis. Allergy 2019, 74, 1429–1444. [Google Scholar] [CrossRef]
- Harris, W.S.; Pottala, J.V.; Vasan, R.S.; Larson, M.G.; Robins, S.J. Changes in erythrocyte membrane trans and marine fatty acids between 1999 and 2006 in older Americans. J. Nutr. 2012, 142, 1297–1303. [Google Scholar] [CrossRef]
- Harris, W.S.; Von Schacky, C. The Omega-3 Index: A new risk factor for death from coronary heart disease? Prev. Med. 2004, 39, 212–220. [Google Scholar] [CrossRef]
- Ferguson, J.J.; Veysey, M.; Lucock, M.; Niblett, S.; King, K.; MacDonald-Wicks, L.; Garg, M.L. Association between omega-3 index and blood lipids in older Australians. J. Nutr. Biochem. 2016, 27, 233–240. [Google Scholar] [CrossRef]
- Stoodley, I.; Williams, L.; Thompson, C.; Scott, H.; Wood, L. Evidence for lifestyle interventions in asthma. Breathe 2019, 15, e50. [Google Scholar] [CrossRef] [PubMed]
- Scott, H.A.; Gibson, P.G.; Garg, M.L.; Wood, L.G. Airway inflammation is augmented by obesity and fatty acids in asthma. Eur. Respir. J. 2011, 38, 594–602. [Google Scholar] [CrossRef] [PubMed]
- Wood, L.G. Diet, Obesity, and Asthma. Ann. Am. Thorac. Soc. 2017, 14, S332–S338. [Google Scholar] [CrossRef] [PubMed]
- Berthon, B.S.; Gibson, P.G.; McElduff, P.; MacDonald-Wicks, L.K.; Wood, L.G. Effects of short-term oral corticosteroid intake on dietary intake, body weight and body composition in adults with asthma—A randomized controlled trial. Clin. Exp. Allergy J. Br. Soc. Allergy Clin. Immunol. 2015, 45, 908–919. [Google Scholar] [CrossRef] [PubMed]
- Williams, E.J.; Baines, K.J.; Berthon, B.S.; Wood, L.G. Effects of an Encapsulated Fruit and Vegetable Juice Concentrate on Obesity-Induced Systemic Inflammation: A Randomised Controlled Trial. Nutrients 2017, 9, 116. [Google Scholar] [CrossRef] [PubMed]
- Scott, H.A.; Gibson, P.G.; Garg, M.L.; Pretto, J.J.; Morgan, P.J.; Callister, R.; Wood, L.G. Dietary restriction and exercise improve airway inflammation and clinical outcomes in overweight and obese asthma: A randomized trial. Clin. Exp. Allergy J. Br. Soc. Allergy Clin. Immunol. 2013, 43, 36–49. [Google Scholar] [CrossRef]
- Wood, L.G.; Garg, M.L.; Smart, J.M.; Scott, H.A.; Barker, D.; Gibson, P.G. Manipulating antioxidant intake in asthma: A randomized controlled trial. Am. J. Clin. Nutr. 2012, 96, 534–543. [Google Scholar] [CrossRef]
- Periyalil, H.A.; Wood, L.G.; Wright, T.A.; Karihaloo, C.; Starkey, M.R.; Miu, A.S.; Baines, K.J.; Hansbro, P.M.; Gibson, P.G. Obese asthmatics are characterized by altered adipose tissue macrophage activation. Clin. Exp. Allergy J. Br. Soc. Allergy Clin. Immunol. 2018, 48, 641–649. [Google Scholar] [CrossRef]
- Wanger, J.; Clausen, J.L.; Coates, A.; Pedersen, O.F.; Brusasco, V.; Burgos, F.; Casaburi, R.; Crapo, R.; Enright, P.; van der Grinten, C.P.; et al. Standardisation of the measurement of lung volumes. Eur. Respir. J. 2005, 26, 511–522. [Google Scholar] [CrossRef]
- Miller, M.R.; Hankinson, J.; Brusasco, V.; Burgos, F.; Casaburi, R.; Coates, A.; Crapo, R.; Enright, P.; van der Grinten, C.P.; Gustafsson, P.; et al. Standardisation of spirometry. Eur. Respir. J. 2005, 26, 319–338. [Google Scholar] [CrossRef]
- Juniper, E.F.; O’Byrne, P.M.; Guyatt, G.H.; Ferrie, P.J.; King, D.R. Development and validation of a questionnaire to measure asthma control. Eur. Respir. J. 1999, 14, 902–907. [Google Scholar] [CrossRef] [PubMed]
- Gibson, P.G.; Wlodarczyk, J.W.; Hensley, M.J.; Gleeson, M.; Henry, R.L.; Cripps, A.W.; Clancy, R.L. Epidemiological association of airway inflammation with asthma symptoms and airway hyperresponsiveness in childhood. Am. J. Respir. Crit. Care Med. 1998, 158, 36–41. [Google Scholar] [CrossRef] [PubMed]
- Tomoda, A.; Kodaira, K.; Taketo, A.; Tanimoto, K.; Yoneyama, Y. Isolation of human erythrocyte membranes in glucose solution. Anal. Biochem. 1984, 140, 386–390. [Google Scholar] [CrossRef]
- Lepage, G.; Roy, C.C. Direct transesterification of all classes of lipids in a one-step reaction. J. Lipid Res. 1986, 27, 114–120. [Google Scholar] [PubMed]
- Zhou, J.; Chen, L.; Liu, Z.; Sang, L.; Li, Y.; Yuan, D. Changes in erythrocyte polyunsaturated fatty acids and plasma eicosanoids level in patients with asthma. Lipids Health Dis. 2018, 17, 206. [Google Scholar] [CrossRef] [PubMed]
- Miyata, J.; Arita, M. Role of omega-3 fatty acids and their metabolites in asthma and allergic diseases. Allergol. Int. 2015, 64, 27–34. [Google Scholar] [CrossRef]
- Barros, R.; Moreira, A.; Fonseca, J.; Delgado, L.; Castel-Branco, M.G.; Haahtela, T.; Lopes, C.; Moreira, P. Dietary intake of alpha-linolenic acid and low ratio of n-6:n-3 PUFA are associated with decreased exhaled NO and improved asthma control. Br. J. Nutr. 2011, 106, 441–450. [Google Scholar] [CrossRef]
- Papamichael, M.M.; Katsardis, C.; Lambert, K.; Tsoukalas, D.; Koutsilieris, M.; Erbas, B.; Itsiopoulos, C. Efficacy of a Mediterranean diet supplemented with fatty fish in ameliorating inflammation in paediatric asthma: A randomised controlled trial. J. Hum. Nutr. Diet. Off. J. Br. Diet. Assoc. 2019, 32, 185–197. [Google Scholar] [CrossRef]
- Mickleborough, T.D.; Lindley, M.R.; Ionescu, A.A.; Fly, A.D. Protective effect of fish oil supplementation on exercise-induced bronchoconstriction in asthma. Chest 2006, 129, 39–49. [Google Scholar] [CrossRef]
- Reisman, J.; Schachter, H.M.; Dales, R.E.; Tran, K.; Kourad, K.; Barnes, D.; Sampson, M.; Morrison, A.; Gaboury, I.; Blackman, J. Treating asthma with omega-3 fatty acids: Where is the evidence? A systematic review. BMC Complementary Altern. Med. 2006, 6, 26. [Google Scholar] [CrossRef]
- AUSNUT 2011–13–Australian Food Composition Database; Food Standards Australia New Zealand: Canberra, Australia, 2014.
- Peters, U.; Dixon, A.E.; Forno, E. Obesity and asthma. J. Allergy Clin. Immunol. 2018, 141, 1169–1179. [Google Scholar] [CrossRef] [PubMed]
- Lang, J.E.; Mougey, E.B.; Hossain, M.J.; Livingston, F.; Balagopal, P.B.; Langdon, S.; Lima, J.J. Fish Oil Supplementation in Overweight/Obese Patients with Uncontrolled Asthma. A Randomized Trial. Ann. Am. Thorac. Soc. 2019, 16, 554–562. [Google Scholar] [CrossRef] [PubMed]
- Haidari, F.; Mohammadshahi, M.; Borsi, S.H.; Haghighizadeh, M.H.; Malgard, S. Comparison of essential fatty acid intakes and serum levels of inflammatory factors between asthmatic and healthy adults: A case-control study. Iran. J. Allergy Asthma Immunol. 2014, 13, 335–342. [Google Scholar] [PubMed]
- Skulas-Ray, A.C.; Kris-Etherton, P.M.; Harris, W.S.; Vanden Heuvel, J.P.; Wagner, P.R.; West, S.G. Dose-response effects of omega-3 fatty acids on triglycerides, inflammation, and endothelial function in healthy persons with moderate hypertriglyceridemia. Am. J. Clin. Nutr. 2011, 93, 243–252. [Google Scholar] [CrossRef]
- Geelen, A.; Brouwer, I.A.; Schouten, E.G.; Kluft, C.; Katan, M.B.; Zock, P.L. Intake of n-3 fatty acids from fish does not lower serum concentrations of C-reactive protein in healthy subjects. Eur. J. Clin. Nutr. 2004, 58, 1440–1442. [Google Scholar] [CrossRef]
- Khorsan, R.; Crawford, C.; Ives, J.A.; Walter, A.R.; Jonas, W.B. The effect of omega-3 fatty acids on biomarkers of inflammation: A rapid evidence assessment of the literature. Mil. Med. 2014, 179, 2–60. [Google Scholar] [CrossRef]
- Kumar, A.; Mastana, S.S.; Lindley, M.R. n-3 Fatty acids and asthma. Nutr. Res. Rev. 2016, 29, 1–16. [Google Scholar] [CrossRef]
- Wood, L.G.; Powell, H.; Gibson, P.G. Mannitol challenge for assessment of airway responsiveness, airway inflammation and inflammatory phenotype in asthma. Clin. Exp. Allergy J. Br. Soc. Allergy Clin. Immunol. 2010, 40, 232–241. [Google Scholar] [CrossRef]
- Wagner, A.; Simon, C.; Morio, B.; Dallongeville, J.; Ruidavets, J.B.; Haas, B.; Laillet, B.; Cottel, D.; Ferrieres, J.; Arveiler, D. Omega-3 index levels and associated factors in a middle-aged French population: The MONA LISA-NUT Study. Eur. J. Clin. Nutr. 2015, 69, 436–441. [Google Scholar] [CrossRef]
- Cohen, B.E.; Garg, S.K.; Ali, S.; Harris, W.S.; Whooley, M.A. Red Blood Cell Docosahexaenoic Acid and Eicosapentaenoic Acid Concentrations Are Positively Associated with Socioeconomic Status in Patients with Established Coronary Artery Disease: Data from the Heart and Soul Study. J. Nutr. 2008, 138, 1135–1140. [Google Scholar] [CrossRef]
- Meyer, B.J. Australians are not Meeting the Recommended Intakes for Omega-3 Long Chain Polyunsaturated Fatty Acids: Results of an Analysis from the 2011-2012 National Nutrition and Physical Activity Survey. Nutrients 2016, 8, 111. [Google Scholar] [CrossRef] [PubMed]
| No Asthma | Asthma | p-Value * | Odds Ratio (95% CI) | |
|---|---|---|---|---|
| Subjects | 137 | 255 | ||
| Age | 53.5 (45.2–64.35) | 57.1 (40.9–66.0) | 0.8473 ^ | |
| Gender (% female) | 39.4 (n = 54) | 50.6 (n = 129) | 0.0435 # | |
| BMI (kg/m2) | 33.5 (28.9–41.45) | 31.0 (26.9–36.2) | 0.0011 ^ | |
| Smokers (% Ex) | 46.72 (n = 64) | 43.14 (n = 110) | 0.854 | 0.98 (0.79–1.22) |
| Smoking history (pack years) | 4.0 (0.0–11.0) | 5.5 (0.0–20.0) | 0.011 | 1.03 (1.00–1.06) |
| ACQ6 | 0.7 (0.2–1.3) (n = 206) | |||
| GINA Classification (%1/2/3/4) | 25/20/39/16 (n = 243) | |||
| FEV1 (% predicted) | 98.54 ± 13.04 (n = 71) | 79.42 ± 18.77 (n = 235) | <0.001 | 0.93 (0.90–0.95) |
| FVC (% predicted) | 102.8 ± 14.07 (n = 71) | 91.46 ± 16.11 (n = 235) | <0.001 | 0.95 (0.93–0.97) |
| FEV1/FVC (%) | 77.0 (74.0–81.0) (n = 71) | 70.0 (63.1–77.0) (n = 235) | <0.001 | 0.88 (0.84–0.92) |
| Airway markers | ||||
| Neutrophils (%) | 30.5 (12.0–47.25) (n = 51) | 34.75 (10.25–54.75) (n = 206) | 0.782 | 1.00 (0.99–1.02) |
| Eosinophils (%) | 0.75 (0.25–1.25) (n = 51) | 2.5 (0.72–14.4) (n = 206) | 0.003 | 1.25 (1.08–1.44) |
| Macrophages (%) | 61.5 (46.0–74.0) (n = 51) | 38.31 (18.9–61.31) (n = 206) | <0.001 | 0.97 (0.96–0.98) |
| Lymphocytes (%) | 1.75 (0.75–3.0) (n = 51) | 1.0 (0.25–3.25) (n = 206) | 0.032 | 1.06 (1.01–1.11) |
| Systemic markers | ||||
| CRP (mg/L) | 3.0 (1.5–5.5) (n = 135) | 3.41 (1.2–8.18) (n = 196) | 0.018 | 1.06 (1.01–1.11) |
| IL-6 (pg/mL) | 1.45 (1.05–1.94) (n = 34) | 1.51 (0.89–2.58) (n = 171) | 0.289 | 1.19 (0.87–1.63) |
| TNF-a (pg/mL) | 1.05 (0.88–1.34) (n = 44) | 1.18 (0.64–1.77) (n = 143) | 0.168 | 1.43 (0.86–2.38) |
| Erythrocyte fatty acids | ||||
| SFA (%) | 43.29 (42.54–44.26) (n = 127) | 41.92 (40.87–43.11) (n = 242) | 0.001 | 0.89 (0.83–0.96) |
| MUFA (%) | 18.27 (17.29–19.28) (n = 127) | 19.06 (17.67–20.13) (n = 242) | 0.526 | 1.02 (0.96–1.09) |
| PUFA (%) | 29.13 (27.27–30.44) (n = 127) | 28.77 (27.29–30.39) (n = 242) | 0.995 | 1.00 (0.91–1.10) |
| n-3 PUFA (%) | 8.91 (7.85–10.25) (n = 127) | 10.03 (8.74–12.54) (n = 242) | <0.001 | 1.24 (1.12–1.37) |
| O3I (%) | 6.1 (4.9–7.4) (n = 127) | 6.2 (5.4–7.9) (n = 242) | 0.089 | 1.10 (0.99–1.23) |
| n-6:n-3 | 3.31 (2.72–3.74) (n = 127) | 2.82 (2.30–3.40) (n = 242) | <0.001 | 0.58 (0.44–0.78) |
| Asthma, Low O3I (<8%) | Asthma, High O3I (≥8%) | p-Value * | Odds Ratio (95% CI) | |
|---|---|---|---|---|
| n | 185 | 57 | ||
| Age | 57.9 (42.8–66.4) | 54.6 (36.5–65.6) | 0.1938 ^ | |
| BMI (kg/m2) | 30.6 (26.9–37.7) | 31.7 (28.1–35.5) | 0.9591 ^ | |
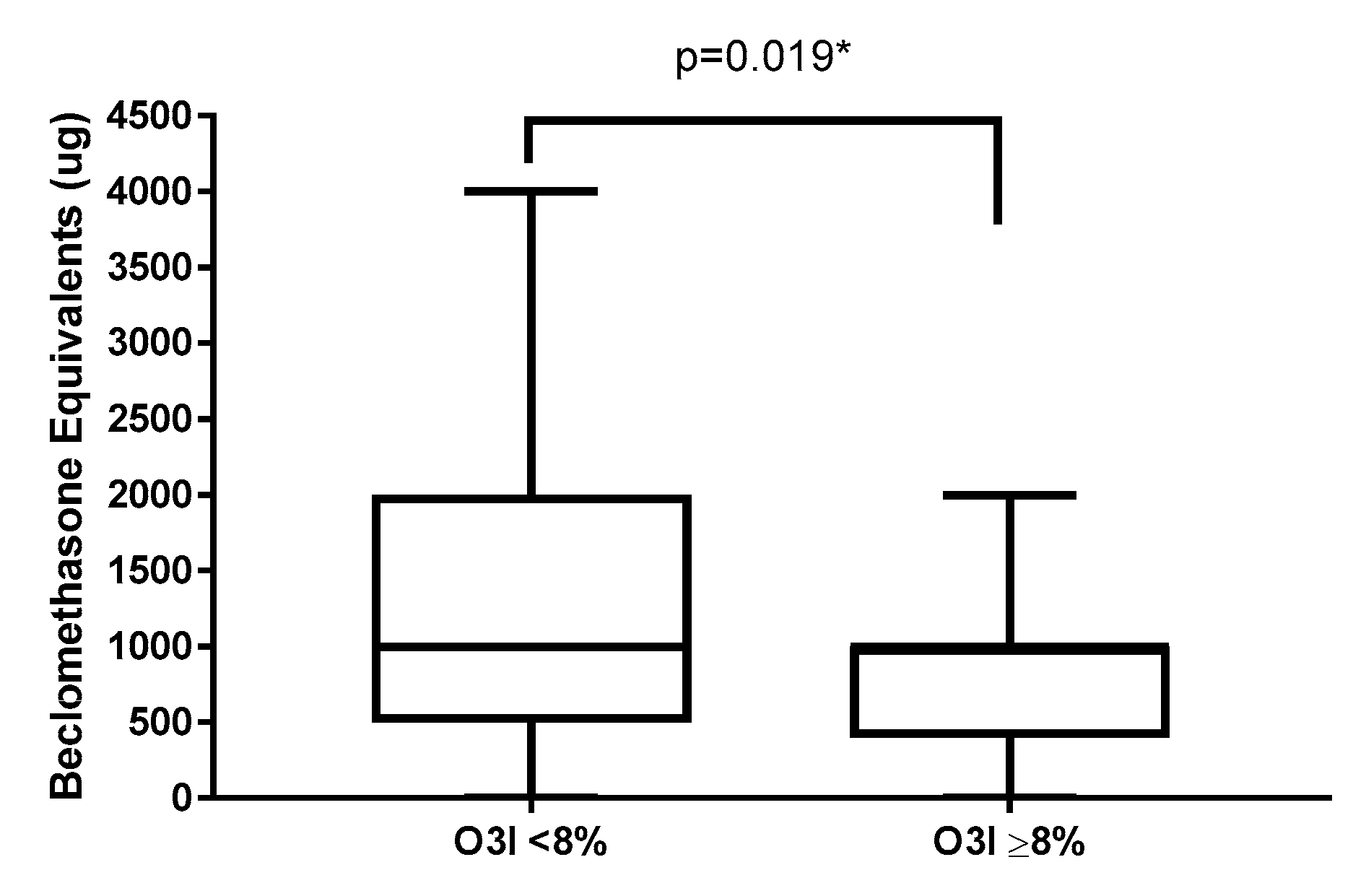
| ICS (ug beclomethasone eq/d) | 1000 (500–2000) (n = 120) | 1000 (400–1000) (n = 51) | 0.019 | 0.999 (0.9989–0.9999) |
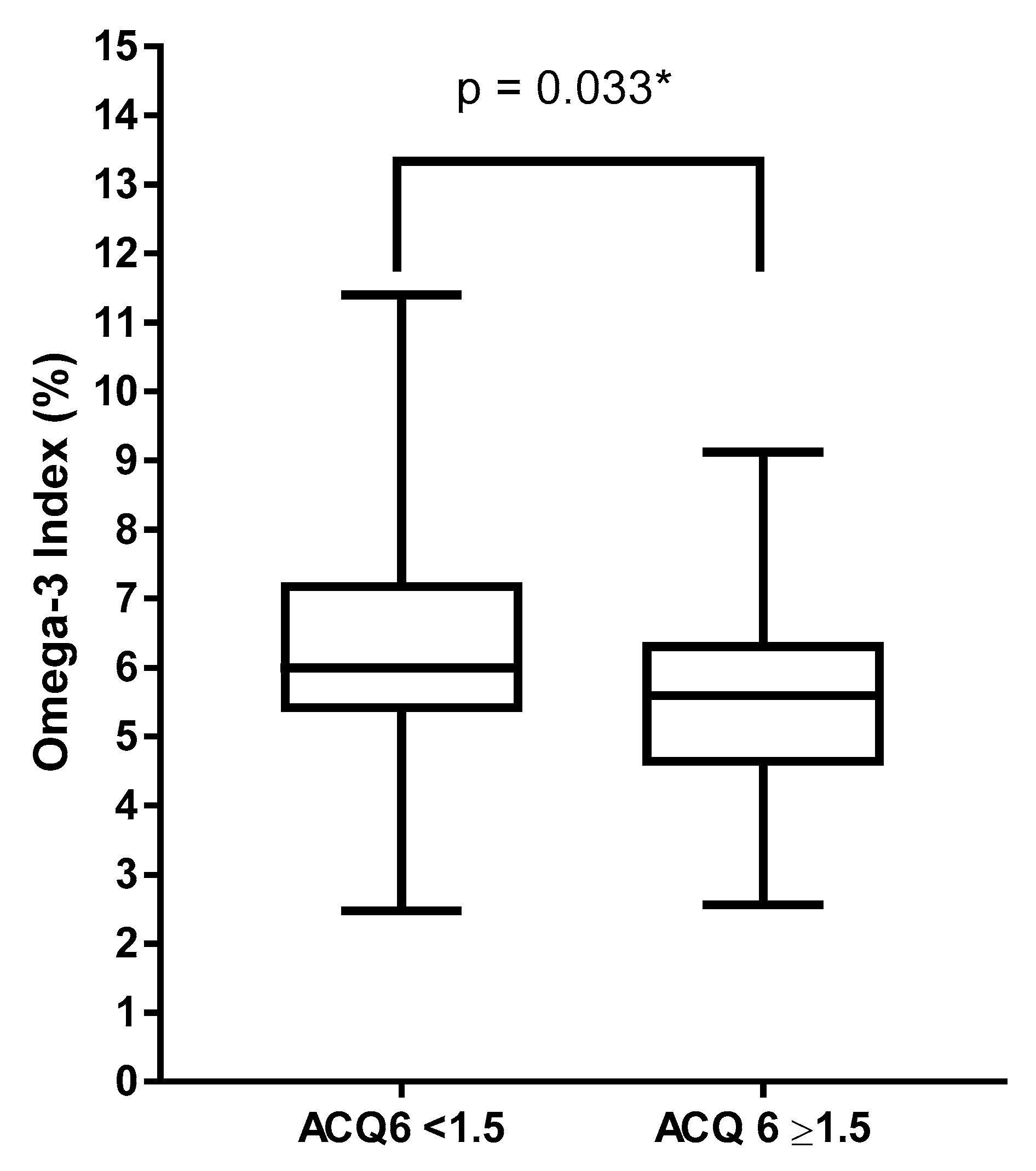
| ACQ6 | 0.7 (0.2–1.3) (n = 170) | 0.42 (0.2–1.11) (n = 24) | 0.311 | 0.71 (0.37–1.37) |
| GINA Classification: 1/2/3/4 (%) | 24/22/38/16 (n = 176) | 19/25/41/15 (n = 54) | 0.7638 # | |
| FEV1 (% predicted) | 79.11 ± 19.4 (n = 169) | 78.93 ± 16.78 (n = 54) | 0.610 | 1.00 (0.98–1.01) |
| FVC (% predicted) | 91.01 ± 16.82 (n = 169) | 91.71 ± 13.43 (n = 54) | 0.760 | 1.00 (0.98–1.02) |
| FEV1/FVC (%) | 70 (63–77) (n = 169) | 69.55 (59.78–75.33) (n = 54) | 0.249 | 0.98 (0.95–1.01) |
| CRP (mg/mL) | 3.8 (1.3–8.7) (n = 140) | 2.9 (1.1–7.8) (n = 44) | 0.124 | 0.94 (0.87–1.02) |
| IL-6 (pg/mL) | 1.8 (1.3–8.7) (n = 41) | 0.9 (0.1–1.7) (n = 41) | 0.212 | 0.85 (0.65–1.10) |
| TNF (pg/mL) | 1.3 (0.9–1.8) (n = 99) | 0.4 (0.2–1.2) (n = 37) | 0.944 | 1.01 (0.85–1.19) |
| Obese Asthma | Nonobese Asthma | |||||||
|---|---|---|---|---|---|---|---|---|
| Obese Asthma | >8% O3I | <8% O3I | >8% O3I | <8% O3I | p-Value * | Obesity × O3I Interaction | High vs. Low O3I | Obese vs. Nonobese |
| n | 37 | 98 | 20 | 87 | ||||
| ICS (μg beclomethasone eq/d) | 1000 (212.5–1000) (n = 36) | 1000 (1000–2000) (n = 61) | 1000 (500–1000) (n = 15) | 500 (500–1000) (n = 59) | 0.0002 | 0.1556 | 0.0104 | 0.0077 |
| ACQ6 | 0.33 (0–1.8) (n = 7) | 0.75 (0.3–1.5) (n = 86) | 0.5 (0.3–1.0) (n = 17) | 0.7 (0.2–1.3) (n = 84) | 0.6981 | 0.8864 | 0.4235 | 0.4948 |
| FEV1 (% predicted) | 82.49 ± 15.08 (n = 35) | 80.18 ± 19.37 (n = 84) | 72.37 ± 18.15 (n = 19) | 78.05 ± 19.48 (n = 85) | 0.0001 | 0.6616 | 0.4851 | 0.3445 |
| FVC (% predicted) | 94.04 ± 13.87 (n = 35) | 88.61 ± 15.84 (n = 84) | 87.42 ± 11.74 (n = 19) | 93.38 ± 17 (n = 85) | <0.0001 | 0.2197 | 0.8011 | 0.3537 |
| FEV1/FVC (5) | 71.5 (63.9–77.7) (n = 35) | 74 (66.25–80) (n = 84) | 66 (55–71.6) (n = 19) | 68 (60–74) (n = 85) | <0.0001 | 0.6012 | 0.1146 | 0.0035 |
| CRP (mg/mL) | 3.1 (1.25–8) (n = 33) | 5.59 (2.67–12.42) (n = 74) | 1.57 (0.9–7.28) (n = 11) | 1.77 (0.97–5) (n = 67) | 0.0003 | 0.0475 | ||
| IL-6 (pg/mL) | 0.30 (0.04–1.13) (n = 30) | 2.35 (1.61–3.37) (n = 53) | 1.77 (1.16–2.42) (n = 11) | 1.38 (0.9–2.11) (n = 65) | 0.0326 | 0.6909 | 0.1857 | 0.3207 |
| TNF (pg/mL) | 0.35 (0.18–0.9) (n = 27) | 1.41 (0.96–1.92) (n = 50) | 1 (0.46–1.54) (n = 10) | 1.23 (0.82–1.75) (n = 49) | 0.4769 | 0.3315 | 0.8142 | 0.2666 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Stoodley, I.; Garg, M.; Scott, H.; Macdonald-Wicks, L.; Berthon, B.; Wood, L. Higher Omega-3 Index Is Associated with Better Asthma Control and Lower Medication Dose: A Cross-Sectional Study. Nutrients 2020, 12, 74. https://doi.org/10.3390/nu12010074
Stoodley I, Garg M, Scott H, Macdonald-Wicks L, Berthon B, Wood L. Higher Omega-3 Index Is Associated with Better Asthma Control and Lower Medication Dose: A Cross-Sectional Study. Nutrients. 2020; 12(1):74. https://doi.org/10.3390/nu12010074
Chicago/Turabian StyleStoodley, Isobel, Manohar Garg, Hayley Scott, Lesley Macdonald-Wicks, Bronwyn Berthon, and Lisa Wood. 2020. "Higher Omega-3 Index Is Associated with Better Asthma Control and Lower Medication Dose: A Cross-Sectional Study" Nutrients 12, no. 1: 74. https://doi.org/10.3390/nu12010074
APA StyleStoodley, I., Garg, M., Scott, H., Macdonald-Wicks, L., Berthon, B., & Wood, L. (2020). Higher Omega-3 Index Is Associated with Better Asthma Control and Lower Medication Dose: A Cross-Sectional Study. Nutrients, 12(1), 74. https://doi.org/10.3390/nu12010074








