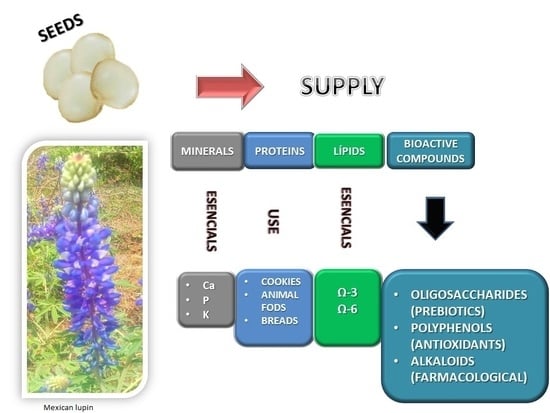
Nutritional and Bioactive Compounds in Mexican Lupin Beans Species: A Mini-Review
Abstract
1. Introduction
2. Domesticated Lupins
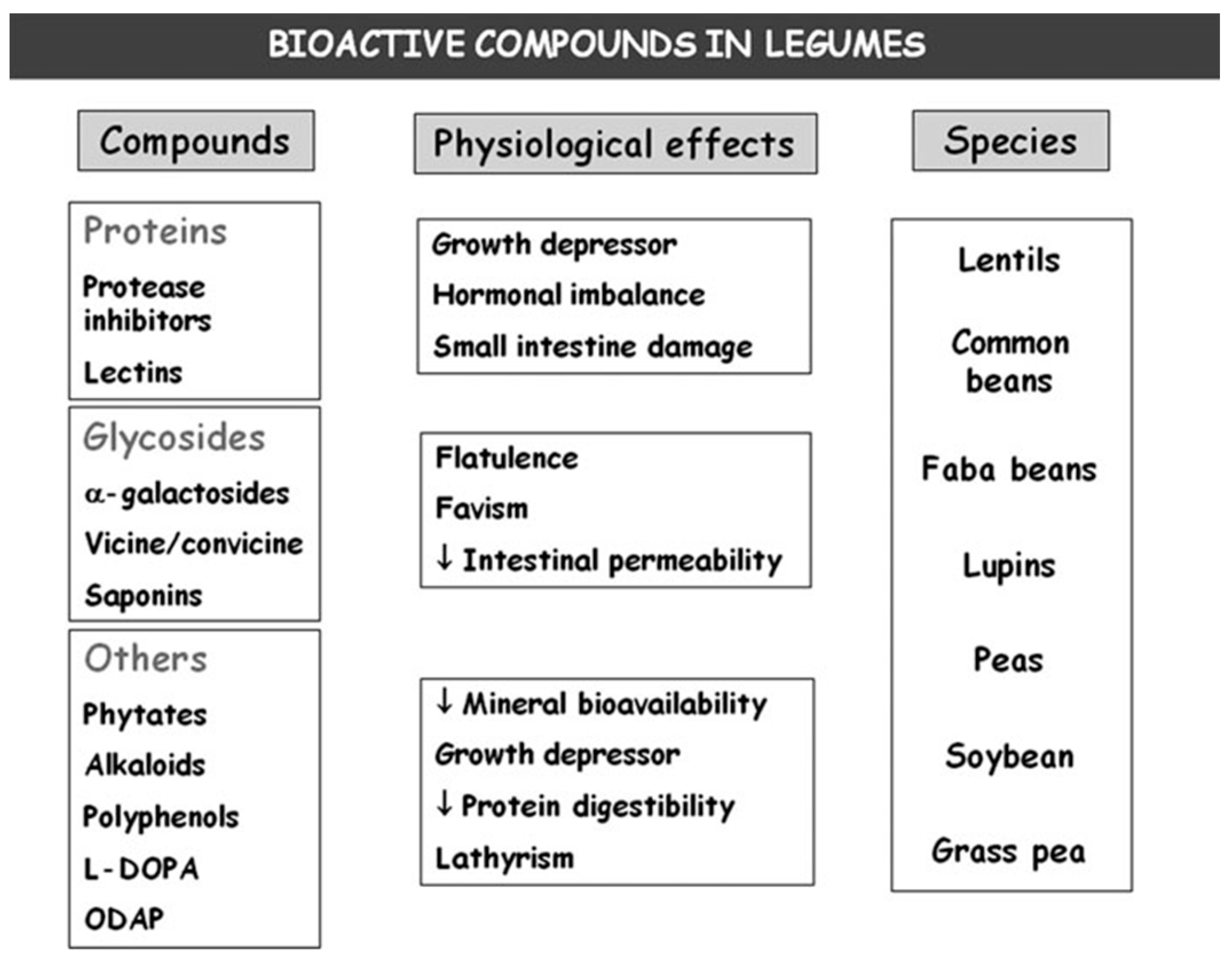
3. Bioactive Compounds
4. Mexican Wild Lupinus
4.1. Distribution in Mexico
4.2. Nutritional Composition
4.3. Protein Isolates and Their Functionality
4.4. Dietary Fiber
4.5. Minerals and Bioavailability
5. Bioactive Compounds
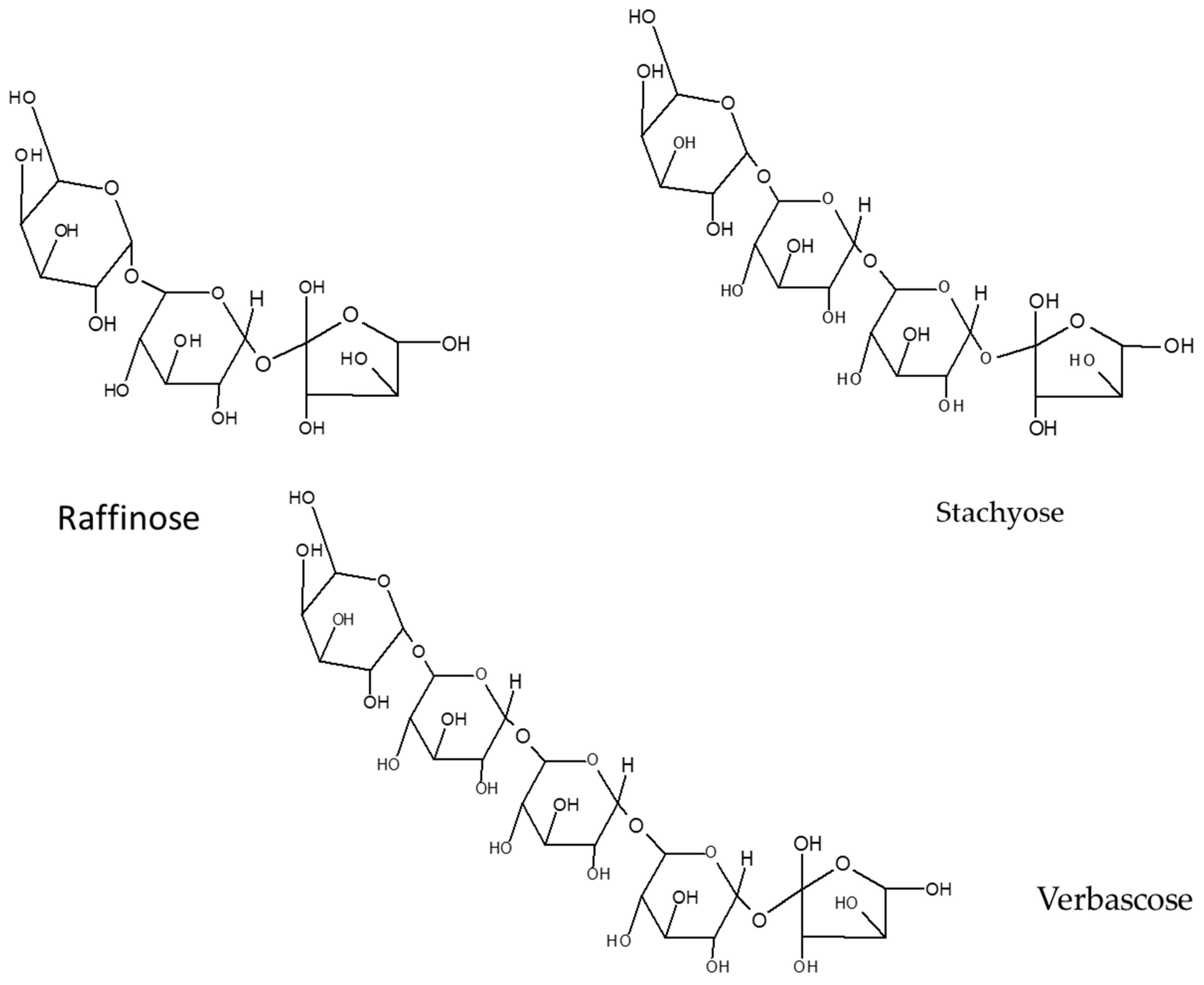
5.1. Oligosaccharides
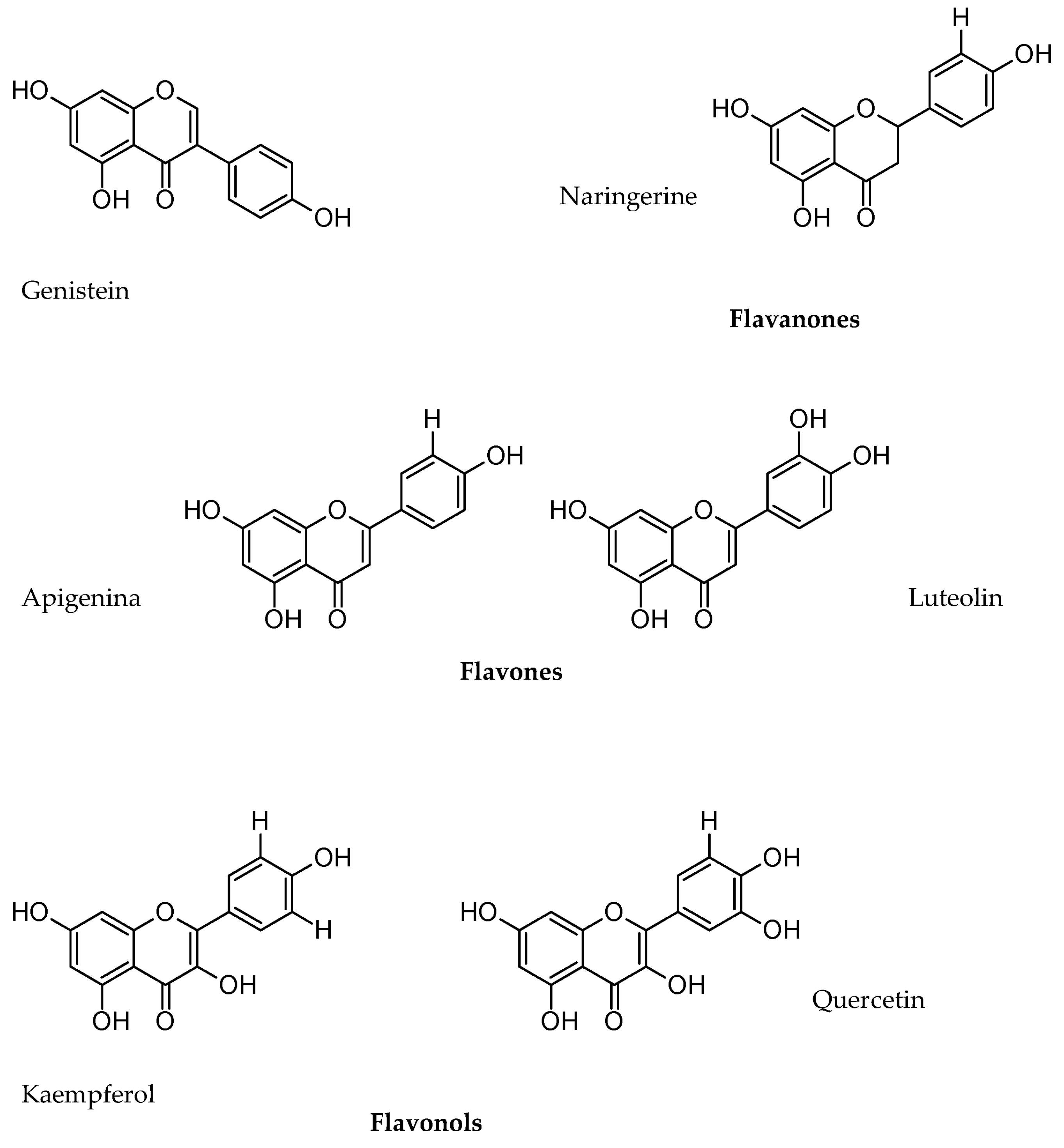
5.2. Phenols
5.3. Alkaloids
6. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Dillard, C.J.; German, J.B. Phytochemicals: Nutraceuticals and human health. J. Sci. Food Agric. 2000, 80, 1744–1756. [Google Scholar] [CrossRef]
- Rochfort, S.; Panozzo, J. Phytochemicals for health, the role of pulses. J. Agric. Food Chem. 2007, 55, 7981–7994. [Google Scholar] [CrossRef] [PubMed]
- Dilis, V.; Trichopoulou, A. Nutritional and health properties of pulses. Mediterr. J. Nutr. Metab. 2009, 1, 149–157. [Google Scholar] [CrossRef]
- Muzquiz, M.; Varela, A.; Burbano, C.; Cuadrado, C.; Guillamón, E.; Pedrosa, M.M. Bioactive compounds in legumes: Pronutritive and antinutritive actions. Implications for nutrition and health. Phytochem. Rev. 2012, 11, 227–244. [Google Scholar] [CrossRef]
- Villarino, C.; Jayasena, V.; Coorey, R.; Chakrabarti-Bell, S.; Johnson, S. Nutritional, health, and technological functionality of lupin flour addition to bread and other baked products: Benefits and challenges. Crit. Rev. Food Sci. Nutr. 2016, 56, 835–857. [Google Scholar] [CrossRef]
- Petterson, D.S. Composition and food uses of Lupinus. In Lupin as Crop Plants. Biology, Production and Utilization; Gladstones, J.S., Atkins, C., Hamblin, J., Eds.; CAB International: Wallingford, UK, 1998; pp. 353–384. [Google Scholar]
- Sujak, A.; Kotlarz, A.; Strobel, W. Compositional and nutritional evaluation of several lupin seeds. Food Chem. 2006, 98, 711–719. [Google Scholar] [CrossRef]
- Arnoldi, A.; Greco, S. Nutritional and nutraceutical characteristics of lupin protein. Nutrafoods 2011, 10, 23–29. [Google Scholar] [CrossRef]
- Lovati, M.A.; Manzoni, C.; Castiglioni, S.; Parolari, A.; Magni, C.; Duranti, M. Lupin seed g-conglutin lowers blood glucose in hyperglycemic rats and increases glucose consumption of HepG2 cells. Br. J. Nutr. 2012, 107, 67–73. [Google Scholar] [CrossRef]
- Marchesi, M.; Parolini, C.; Diani, E.; Rigamonti, E.; Cornelli, L.; Arnoldi, A.; Sirtori, C.R.; Chiesa, G. Hypolipidaemic and anti-atherosclerotic effects of lupin proteins in a rabbit model. Br. J. Nutr. 2008, 100, 707–710. [Google Scholar] [CrossRef]
- Sirtori, C.R.; Lovati, M.R.; Manzoni, C.; Castiglioni, S.; Duranti, M.; Magni, C.; Morandi, S.; D’Agostina, A.; Arnoldi, A. Proteins of white lupin seed, a naturally isoflavone-poor legume, reduce cholesterolemia in rats and increase LDL receptor activity in HepG2 cells. J. Nutr. 2004, 134, 18–23. [Google Scholar] [CrossRef]
- Viveros, A.; Centeno, C.; Arija, I.; Brenes, A. Cholesterol-Lowering Effects of Dietary Lupin (Lupinus albus var Multolupa) in Chicken Diets. Poult. Sci. 2007, 86, 2631–2638. [Google Scholar] [CrossRef]
- Pihlanto, A.; Mattila, P.; Mäkinen, S.; Pajari, A.M. Bioactivities of alternative protein sources and their potential health benefits. Food Funct. 2017, 8, 3443–3458. [Google Scholar] [CrossRef]
- Lizarazo, C.I.; Lampi, A.; Liu, J.; Sontag-Strohm, T.; Piironen, V.; Stoddard, F.L. Nutritive quality and protein production from grain legumes in a boreal climate. J. Sci. Food Agric. 2015, 95, 2053–2064. [Google Scholar] [CrossRef]
- Simopoulos, A.P. Importance of the ratio of omega-6/omega-3 essential fatty acids: Evolutionary aspects. World Rev. Nutr. Diet. 2003, 92, 1–22. [Google Scholar]
- Hall, R.S.; Thomas, S.J.; Johnson, S.K. Australian sweet lupin flour addition reduces the glycaemic index of a white bread breakfast without affecting palatability in healthy human volunteers. Asia Pac. J. Clin. Nutr. 2005, 14, 91–97. [Google Scholar]
- Johnson, S.K.; Chua, V.; Hall, S.R.; Baxter, L.A. Lupin kernel fibre foods improve bowel function and beneficially modify some putative faecal risk factors for colon cancer in men. Br. J. Nutr. 2006, 95, 372–378. [Google Scholar] [CrossRef]
- Písaříková, B.; ZralýA, Z. Dietary fibre content in lupine (Lupinus albus L.) and soya (Glycine max L.) seeds. Acta Vet. Brno 2010, 79, 211–216. [Google Scholar] [CrossRef]
- Porres, J.M.; Aranda, P.; Lopez-Jurado, M.A.; Urbano, G. Nitrogen Fractions and Mineral Content in Different Lupin Species (Lupinus albus, Lupinus angustifolius, and Lupinus luteus). Changes induced by the a-Galactoside extraction process. J. Agric. Food Chem. 2007, 55, 7445−7452. [Google Scholar] [CrossRef]
- Donangelo, C.M.; Pedersen, B.; Eggum, O.B. Protein energy and mineral utilization in rats fed rice: Legume diets. Plant Foods Hum. Nutr. 1986, 36, 119–137. [Google Scholar] [CrossRef]
- Trugo, L.C.; Donangelo, C.M.; Duarte, Y.A.; Tavares, C.L. Phytic acid and selected mineral composition of seed from wild species and cultivated varieties of lupin. Food Chem. 1993, 47, 391–394. [Google Scholar] [CrossRef]
- Jayasena, V.; James, A.P. Total phenolic and phytosterol compounds and the radical scavenging activity of germinated Australian sweet lupin flour. Plant Foods Hum. Nutr. 2013, 68, 352–357. [Google Scholar]
- Champ, M.M. Non-nutrient bioactive substances of pulses. Br. J. Nutr. 2002, 88, 307–319. [Google Scholar] [CrossRef]
- Muzquiz, M.; Wood, J.A. Antinutritional Factors. In Chickpea Breeding Management; Yadav, S.S., Redden, R., Chen, W., Sharma, B., Eds.; CABI: Wallingford, UK, 2007; pp. 143–166. [Google Scholar]
- Del Rio, D.; Rodriguez-Mateos, A.; Spencer, J.P.; Tognolini, M.; Borges, G.; Crozier, A. Dietary (poly) phenolics in human health: Structures, bioavailability, and evidence of protective effects against chronic diseases. Antioxid. Redox Signal. 2013, 18, 1818–1892. [Google Scholar] [CrossRef]
- Campos-Vega, R.; Loarca-Piña, G.F.; Oomah, B.D. Minor components of pulses and their potential impact on human health. Food Res. Int. 2010, 43, 461–482. [Google Scholar] [CrossRef]
- Kadlec, P.; Bjergegaard, C.; Gulewicz, K.; Horbowicz, M.; Jones, A.; Kintia, P.; Kratchanov, C.; Kratchanova, M.; Lewandowicz, G.; Soral-Smietana, M.; et al. Carbohydrate chemistry. In Carbohydrates in Grain Legume Seeds: Improving Nutritional Quality and Agronomic Characteristics; Hedley, C.L., Ed.; CAB International: Wallingford, UK, 2000; pp. 15–59. [Google Scholar]
- Martínez-Villaluenga, C.; Frías, J.; Vidal-Valverde, C. Raffinose family oligosaccharides and sucrose contents in 13 Spanish lupin cultivars. Food Chem. 2005, 91, 645–649. [Google Scholar] [CrossRef]
- Balasundram, N.; Sundram, K.; Samman, S. Phenolic compounds in plants and agro-industrial by-products: Antioxidant activity, occurrence, and potential uses. Food Chem. 2006, 99, 191–203. [Google Scholar] [CrossRef]
- Siger, A.l.; Czubinski, J.; Kachlicki, P.; Dwiecki, K.; Lampart-Szczapa, E.; Nogala-Kalucka, M. Antioxidant activity and phenolic content in three Lupin species. J. Food Compos. Anal. 2012, 25, 190–197. [Google Scholar] [CrossRef]
- Song, F.L.; Gan, R.Y.; Zhang, Y.; Xiao, Q.; Kuang, L.; Li, H.B. Total Phenolic Contents and Antioxidant Capacities of Selected Chinese Medicinal Plants. Int. J. Mol. Sci. 2010, 11, 2362–2372. [Google Scholar] [CrossRef]
- Bordoloi, M.; Bordoloi, P.K.; Dutta, P.P.; Singh, V.; Nath, S.; Narzary, B.; Bhuyan, P.D.; Rao, P.G.; Barua, I.C. Studies on some edible herbs: Antioxidant activity, phenolic content, mineral content and antifungal properties. J. Funct. Foods 2016, 23, 220–229. [Google Scholar] [CrossRef]
- Vahid, F.; Gominho, J.; Pereira, H.; Carvalho, I.S. Screening of the Antioxidant and Enzyme Inhibition Potentials of Portuguese Pimpinella anisum L. Seeds by GC-MS. Food Anal. Method 2018, 11, 2645–2656. [Google Scholar]
- Martínez-Villaluenga, C.; Zielinski, H.; Frias, J.; Piskula, M.K.; Kozlowska, H.; Vidal-Valverde, C. Antioxidante capacity and polyphenolic content of high-protein lupin products. Food Chem. 2009, 112, 84–88. [Google Scholar] [CrossRef]
- Khan, M.K.; Karnpanit, W.; Nasar-Abbas, S.M.; Huma, Z.; Jayasena, V. Phytochemical composition and bioactivities of lupin: A review. Int. J. Food Sci. Technol. 2015, 50, 2004–2012. [Google Scholar] [CrossRef]
- Rajeev, B.; Karim, A.A. Exploring the Nutritional Potential of Wild and Underutilized Legumes. Compr. Rev. Food. Sci. Saf. 2009, 8, 305–331. [Google Scholar]
- Ruiz, L.M.A.; Garcia-López, P.M.; Castañeda-Vazquez, H.; Zamora-Natera, J.F.; Garzón-De la Mora, P.; Bañuelos-Pineda, J.; Burbano, C.; Pedrosa, M.M.; Cuadrado, C.; Muzquiz, M. Chemical composition and antinutrient content of three Lupinus species from Jalisco, Mexico. J. Food Compos. Anal. 2000, 13, 193–199. [Google Scholar] [CrossRef]
- Medina-Gonzalez, E.; Martinez-Herrera, J.; Ruiz-López, M.A.; Martinez-Ayala, A.L. Análisis de alcaloides quinolizidinicos en harina y aislados proteicos de L. exaltatus y L. elegans. Rev. Latinoam. Quím. 2000, 28, 137–142. [Google Scholar]
- Ruiz-Moreno, J.J.; Ruiz-Lopez, M.A.; Zamora-Natera, J.F. The genus Lupinus: Taxonomy and distribution in Jalisco, Mexico. In Proceedings of the 9th International Lupin Conference, Klink/Muritz, Germany, 20–24 June 1999; pp. 297–300. [Google Scholar]
- Ruiz, M.A.; Sotelo, A. Chemical composition, nutritive value, and toxicology evaluation of Mexican wild lupins. J. Agric. Food Chem. 2001, 49, 5336–5339. [Google Scholar] [CrossRef]
- Ruiz-López, M.A.; Rodríguez-Macías, R.; Navarro-Pérez, S. Evaluación químico-nutricional de Lupinus exaltatus Zucc, del nevado de colima, México, como fuente potencial de forraje. Interciencia 2006, 31, 758–761. [Google Scholar]
- Pablo-Pérez, M.; Lagunes-Espinoza, L.C.; López-Upton, J.; Aranda-Ibáñez, E.M.; Ramos-Juárez, J. Composición química de especies silvestres del género Lupinus del estado de Puebla, México. Rev. Fitotec. Mex. 2015, 38, 49–55. [Google Scholar]
- Juárez-Fuentes, B.; Lagunes-Espinoza, L.C.; Bucio-Galindo, A.; Delgado-Alvarado, A.; Pérez-Flores, J.; López-Upton, J. Efecto de tratamientos hidrotérmico, remojo y germinación en la composición química de semillas de Lupinus silvestres. Agroproductividad 2018, 11, 41–47. [Google Scholar]
- Porras Saavedra, J.; Guemez-Vera, N.; Montañez-Soto, J.L.; Fernández-Martínez, M.C.; Yañez-Fernández, J. Comparative study of functional properties of protein isolates obtained from three Lupinus species. Adv. Bioresour. 2013, 4, 106–116. [Google Scholar]
- Rodríguez-Ambriz, S.L.; Martínez-Ayala, A.L.; Millan, F.; Davila-Ortíz, G. Composition and functional properties of Lupinus campestris protein isolates. Plant Foods Hum. Nutr. 2005, 60, 99–107. [Google Scholar] [CrossRef]
- Garcia-López, P.M.; Caro-Ojeda, A.; Ruiz-López, M.A.; Bañuelos-Pineda, J.; Zamora-Natera, F.; Ruiz-Moreno, J. Functional properties of protein isolate obtained from Lupinus exaltatus Zucc. In Opportunieties for High Quality, Healthy and Added-Value Crops to Meet European Demands, Proceeding of 3rd European Conference on Grain Legumes, Valladolid, Spain, 14–19 November 1998; European Association for Grain Legume Research: Valladolid, Espana, 1998. [Google Scholar]
- García-Lopez, P.M.; Muzquiz, M.; Ruiz-Lopez, M.A.; Zamora-Natera, J.F.; Burbano, C.; Pedrosa, M.M.; Cuadrado, C.; Garzon-De la Mora, P. Chemical composition and fatty acid profile of several Mexican wild lupins. J. Food Compos. Anal. 2001, 14, 645–651. [Google Scholar]
- Whelton, S.P.; Hyre, D.A.; Pedersen, B.; Yi, Y.; Whelton, P.K.; He, J. Effect of dietary fiber intake on blood pressure: A meta-analysis of randomized, controlled clinical trials. J. Hypertens. 2005, 23, 475–481. [Google Scholar] [CrossRef]
- Valdés-Miramontes, E.H.; López-Espinoza, A.; Rodríguez-Macías, R.; Salcedo-Pérez, E.; Ruiz-López, M.A. Efecto del tratamiento térmico sobre la composición química y minerales en semillas de lupinos silvestres. Rev. Chil. Nutr. 2015, 42, 186–190. [Google Scholar] [CrossRef]
- Pablo-Pérez, M.; Lagunes-Espinoza, L.D.C.; López-Upton, J.; Ramos-Juárez, J.; Aranda-Ibáñez, E.M. Morfometría, germinación y composición mineral de semillas de Lupinus silvestres. Bioagro 2013, 25, 101–108. [Google Scholar]
- Valdes-Miramontes, E.H.; López-Espinoza, A.; Martínez, M.A.G.; Zamora-Natera, J.F.; Rodriguez-Macias, R.; Ruiz-López, M.A. Iron bioavailability of Lupinus rotundiflorus seeds and roots inlow-iron-diet treated rats. Rev. Nutr. 2017, 30, 827–834. [Google Scholar] [CrossRef][Green Version]
- Gibson, G.R.; Roberfroid, M.B. Handbook of Prebiotics; CRC Press: Boca Raton, FL, USA, 2008; p. 504. [Google Scholar]
- Maya-Zepeda, L.; Hernández-Gobora, J.; Rodríguez-Macías, R.; García-López, P.M.; Ruiz-López, M.A. Contenido de oligosacáridos en semillas de leguminosas silvestres mexicanas. Chil. J. Agric. Anim. Sci. 2013, 29, 161–167. [Google Scholar]
- Jiménez, M.C.; Loarca-Piña, G.; Dávila, O.G. Antimutagenic activity of phenolic compounds, oligosaccharides and quinolizidinic alkaloids from Lupinus campestris seeds. Food Addit. Contam. 2003, 20, 940–948. [Google Scholar]
- Jimenez-Martınez, C.; Cardador-Martınez, A.; Martinez-Ayala, A.L.; Muzquiz, M.; Pedrosa, M.M.; Davila-Ortiz, G. Changes in protein, nonnutritional factors, and antioxidant capacity during germination of L. campestris Seeds. Int. J. Agron. 2012, 2012, 1–7. [Google Scholar] [CrossRef]
- Oleszek, W. Dietary phytochemicals and human health. Phytochem. Rev. 2002, 1, 163–166. [Google Scholar] [CrossRef]
- Stobiecki, M.; Staszkow, A.; Piasecka, A.; Garcia-Lopez, P.M.; Zamora-Natera, F.; Kachlicki, P. LC-MSMS Profiling of Flavonoid Conjugates in Wild Mexican Lupine, Lupinus reflexus. J. Nat. Prod. 2010, 73, 1254–1260. [Google Scholar] [CrossRef]
- Wojakowska, A.; Piasecka, A.; García-López, P.M.; Zamora-Natera, F.; Krajewski, P.; Marczak, L.; Kachlicki, P.; Stobiecki, M. Structural analysis and profiling of phenolic secondary metabolites of Mexican lupine species using LC–MS techniques. Phytochemistry 2013, 92, 71–86. [Google Scholar] [CrossRef]
- Farzaneh, V.; Carvalho, I.S. A review of the health benefit potentials of herbal plant infusions and their mechanism of actions. Ind. Crops Prod. 2015, 65, 247–258. [Google Scholar] [CrossRef]
- Hymowitz, T. Grain legumes. In Advances in New Crops; Janick, J., Simon, J.E., Eds.; Timber Press: Portland, OR, USA, 1990; pp. 54–57. [Google Scholar]
- Ruiz-López, M.M.; García-López, P.M.; Rodríguez-Macias, R.; Zamora Natera, J.F. Mexican wild lupines as a source of quinolizidine alkaloids of economic potential. Polibotanica 2010, 29, 159–164. [Google Scholar]
- Martínez-Herrera, J.; Robledo-Quintos, N.; Mora-Escobedo, R.; Dávila-Ortiz, G. Alkaloid composition of Lupinus campestris from México. J. Food Biochem. 2001, 25, 117–125. [Google Scholar] [CrossRef]
- Montes, H.E.; Corona, R.M.L.; Encarnación, C.A.; Cantor, A.J.A.; Sánchez, L.A.; Sporer, F.; Wink, M.; Bermúdez, T.K. Quinolizidine alkaloid composition in different organs of Lupinus aschenbornii. Rev. Bras. Farmacogn. 2011, 21, 824–828. [Google Scholar]
- Zamora-Natera, F.; García-López, P.; Ruiz-López, M.A.; Salcedo-Pérez, E. Composición de alcaloides en semillas de Lupinus mexicanus (fabaceae) y evaluación antifúngica y alelopática del extracto alcaloideo. Agrociencia 2008, 42, 185–192. [Google Scholar]
- Schmeller, T.; Wink, M. Utilization of alkaloids in modern medicine. In Alkaloids. Biochemistry, Ecology and Medicinal Applications; Roberts, M.F., Wink, M., Eds.; Plenum Press: New York, NY, USA, 1998; pp. 435–459. [Google Scholar]
- Szczawinska, K.; Bobkiewicz, K.T.; Kozaryn, M.; Peretiatkowicz, K.; Gulewicz, K. Some pharmacological properties of an extract from bitter Lupin (L. angustifolius) seeds. In Advances in Lupin Research, Proceeding of the VII th International Lupin Conference, Evora, Portugal, 18–23 April 1993; Neves-Martin, J.M., Beirão da Costa, M.L., Eds.; Instituto Superior de Agronomia: Lisbon, Portugal, 1994; pp. 297–300. [Google Scholar]
- Hatzold, T.; Elmadfa, I.; Gross, R.; Wink, M.; Hartmann, T.; Witte, L. Quinolizidine alkaloids in seeds of Lupinus mutabilis. J. Agric. Food Chem. 1983, 31, 934–938. [Google Scholar] [CrossRef]
- Kubo, H.; Inoue, M.; Kamei, J.; Higashiyama, K. Hypoglycemic effect of multiflorine derivatives in normal mice. Biol. Pharm. Bull. 2006, 29, 2046–2050. [Google Scholar] [CrossRef]
- García-López, P.M.; Garzon de la Mora, P.; Wysocka, W.; Maiztegui, B.; Alzugaray, M.E.; Del Zotto, H.; Borelli, M.I. Quinolizidine alkaloids isolated from Lupinus enhance insulin secretion. Eur. J. Pharm. 2004, 504, 139–142. [Google Scholar] [CrossRef]
- Bobkiewicz, K.T.; Dworacka, M.; Kuczynski, S.; Abramczyk, M.; Kolanos, R.; Wysocka, W.; García-López, P.M.; Winiarska, H. Hypoglycaemic effect of quinolizidine alkaloids-lupanine and 2-tionosparteine on non diabetic and streptozotocin-induced diabetic rats. Eur. J. Pharm. 2007, 565, 240–244. [Google Scholar] [CrossRef]
- El-Seedi, H.R.; Maram, H.Z.; Goransson, U.; Verpoorte, R. Cyclopeptide alkaloids. Phytochem. Rev. 2017, 6, 143–165. [Google Scholar] [CrossRef]









| Component | L. albus | L. angustifolius | L. luteus | L. mutabilis |
|---|---|---|---|---|
| Dry matter | 90.4 | 90.6 | 91.7 | 62.0 |
| Proteins | 36.3 | 33.0 | 46.5 | 44.7 |
| Ashes | 3.9 | 3.7 | 3.7 | 3.0 |
| Crude fat | 11.5 | 6.8 | 4.6 | 14.07 |
| Crude fiber | 14.4 | 14.0 | 13.9 | 7.04 |
| Alkaloids | 0.04 | 0.06 | 0.1 | 1.27 |
| Fatty Acid | L. albus | L. luteus | L. angustifolius |
|---|---|---|---|
| 16:0 | 78 | 48 | 110 |
| 18:0 | 16 | 25 | 382 |
| 18:1 | 530 | 210 | 382 |
| 18:2 | 172 | 475 | 371 |
| 18:3 | 95 | 75 | 53 |
| 20:0 | 55 | 45 | 12 |
| 22:0 | 58 | 79 | 19 |
| 22:1 | 58 | 79 | 19 |
| n3/n6 | 0.55 | 0.16 | 0.14 |
| Lupines | Ca | Mg | P | Cu | Fe | Mn | Zn |
|---|---|---|---|---|---|---|---|
| L. albus var. multolupa | 139.0 ± 2.72 | 145.0 ± 1.65 | 332.1 ± 5.12 | 0.72 ± 0.06 | 3.80 ± 0.05 | 90.1 ± 1.34 | 4.30 ± 0.09 |
| L. albus var. marta | 133.6 ± 0.53 | 193.2 ± 0.86 | 468.1 ± 1.76 | 0.75 ± 0.03 | 6.20 ± 0.06 | 35.0 ± 0.32 | 5.24 ± 0.02 |
| L. angustifolius var. troll | 161.8 ± 1.49 | 191.9 ± 1.00 | 543.3 ± 2.25 | 1.02 ± 0.11 | 4.15 ± 0.03 | 7.6 ± 0.06 | 3.65 ± 0.03 |
| L. angustifolius var. emir | 143.0 ± 1.05 | 219.1 ± 1.25 | 613.4 ± 2.05 | 0.95 ± 0.05 | 4.26 ± 0.03 | 8.4 ± 0.04 | 3.79 ± 0.02 |
| L. luteus var. 4486 | 134.8 ± 0.61 | 294.0 ± 2.39 | 715.5 ± 1.39 | 1.10 ± 0.03 | 5.84 ± 0.03 | 5.6 ± 0.03 | 5.90 ± 0.02 |
| L. luteus var. 4492 | 110.4 ± 0.66 | 308.8 ± 1.84 | 845.7 ± 2.72 | 1.25 ± 0.04 | 7.05 ± 0.05 | 6.8 ± 0.02 | 6.42 ± 0.02 |
| Lupine cultivar | Sucrose | Raffinose | Stachyose | Verbascose | Total |
|---|---|---|---|---|---|
| Lupinus albus var. marta | 0.73 ± 0.0 | 5.5 ± 0.01 | 1.1 ± 0.0 | ||
| Lupinus albus var. multolupa | 0.63 ± 0.0 | 5.9 ± 0.01 | 1.2 ± 0.03 | ||
| L. albus cv. multolupa | 2.58 ± 0.06 | 0.62 ± 0.03 | 5.7 ± 0.06 | 0.19 ± 0.10 | 7.56 ± 0.10 |
| L. albus cv. marta | 3.09 ± 0.08 | 0.33 ± 0.02 | 7.2 ± 0.11 | 0.94 ± 0.01 | 8.51 ± 0.13 |
| L. albus LO-3844 | 2.16 ± 0.20 | 0.44 ± 0.03 | 7.2 ± 0.31 | ND | 7.71 ± 0.33 |
| L. albus LO-3846 | 3.13 ± 0.06 | 0.54 ± 0.02 | 6.85 ± 0.03 | ND | 7.39 ± 0.03 |
| L. albus LO-3848 | 2.41 ± 0.19 | 0.36 ± 0.02 | 5.71 ± 0.57 | ND | 6.07 ± 0.55 |
| L. albus LO-3855 | 2.84 ± 0.09 | 0.48 ± 0.02 | 4.98 ± 0.22 | ND | 5.46 ± 0.24 |
| L. luteus LO-4486 | 1.38 ± 0.13 | 0.56 ± 0.11 | 7.01 ± 1.22 | 3.54 ± 0.37 | 11.1 ± 1.69 |
| L. luteus LO-4492 | 1.21 ± 0.04 | 0.64 ± 0.04 | 8.61 ± 0.20 | 3.04 ± 0.02 | 12.3 ± 0.20 |
| L. luteus LO-4500 | 1.01 ± 0.09 | 0.54 ± 0.03 | 6.13 ± 0.31 | 2.79 ± 0.14 | 9.46 ± 0.41 |
| L. angustifolius LO-4817 | 5.05 ± 0.22 | 1.24 ± 0.09 | 5.11 ± 0.36 | 2.48 ± 0.07 | 8.82 ± 0.43 |
| L. angustifolius LO-4820 | 2.55 ± 0.24 | 0.89 ± 0.07 | 3.62 ± 0.06 | 0.79 ± 0.03 | 5.30 ± 0.12 |
| L. angustifolius LO-4822 | 2.91 ± 0.19 | 1.15 ± 0.07 | 5.19 ± 0.14 | 1.36 ± 0.01 | 7.70 ± 0.11 |
| L. angustifolius cv. zapaton | 3.16 ± 0.17 | 0.63 ± 0.04 | 4.52 ± 0.18 | 1.39 ± 0.13 | 6.54 ± 0.34 |
| Sample | Cultivar | Total Phenolic Compounds (mg GAE/100 g DM) | Total Flavonoids (µg Catechin/g DM) |
|---|---|---|---|
| Lupinus albus | Butan | 212.12 ± 2.24 | |
| Boros | 271.25 ± 3.75 | ||
| Multolupa | 1100 ± 17.6 | ||
| Lupinus luteus | Lord | 249.32 ± 4.72 | |
| Parys | 317.88 ± 2.69 | ||
| Lupinus angustifolius | Bojar | 269.72 ± 9.97 | |
| Zeus | 258.42 ± 7.21 | ||
| Troll | 133 ± 12.6 | ||
| Emir | 362 ± 9.00 |
| Sample | Lupinus albus | Lupinus luteus | Lupinus angustifolius | |||
|---|---|---|---|---|---|---|
| Butan | Boros | Lord | Parys | Bojar | Zeus | |
| Apigenin-6,8-di-C-b-glucopyranoside (mg/100 g DM) | 11.90 ± 0.33 | 14.3 ± 0.33 | 53.63 ± 0.44 | 63.14 ± 0.14 | 30.25 ± 0.22 | 27.78 ± 0.07 |
| Gallic acid (mg/kg DM) | 3.53 ± 0.24 | 3.43 ± 0.1 | 3.50 ± 0.33 | 4.21 ± 0.27 | 0.63 ± 0.01 | 0.62 ± 0.04 |
| Protocatechuic (mg/kg DM) | 12.96 ± 0.14 | 14.69 ± 0.36 | 35.90 ± 0.54 | 73.60 ± 1.71 | 12.50 ± 0.39 | 13.77 ± 0.39 |
| p-Hydroxybenzoic (mg/kg DM) | 22.77 ± 0.30 | 27.82 ± 0.68 | 1.06 ± 0.11 | 2.24 ± 0.21 | 43.73 ± 0.48 | 42.73 ± 0.31 |
| Caffeic acid (mg/kg DM) | 0.58 ± 0.05 | 0.09 ± 0.01 | 1.02 ± 0.06 | 1.22 ± 0.11 | 0.84 ± 0.09 | 0.56 ± 0.07 |
| p-Coumaric acid (mg/kg DM) | 0.11 ± 0.01 | 0.18 ± 0.01 | 0.03 ± 0.01 | 0.68 ± 0.14 | 0.42 ± 0.08 | 0.34 ± 0.06 |
| Total (mg/kg DM) | 39.96 ± 0.15 | 46.23 ± 1.22 | 41.52 ± 0.68 | 82.06 ± 1.52 | 58.14 ± 1.02 | 58.03 ± 0.87 |
| Sample | Cultivar | DPPH (mM Trolox) Eq/g | TRAP (mMTrolox) Eq/g |
|---|---|---|---|
| Lupinus albus | Butan | 3.51 ± 0.2 | 0.33 ± 0.01 |
| Boros | 6.78 ± 0.28 | 0.71 ± 0.01 | |
| Lupinus luteus | Lord | 9.03 ± 0.33 | 1.44 ± 0.02 |
| Parys | 8.12 ± 0.10 | 0.96 ± 0.07 | |
| Lupinus angustifolius | Bojar | 6.89 ± 0.35 | 1.60 ± 0.05 |
| Zeus | 7.47 ± 0.29 | 1.78 ± 0.04 |
| Component | Ash | Lipids | Crude Fiber | Crude Protein (n = 6.25) | Carbohydrates |
|---|---|---|---|---|---|
| L. elegans | 4.2 | 5.8–7.3 | 12.9 | 43.6–45.4 | 31.7 |
| L. exaltatus | 3.4–5.2 | 5.8–8.7 | 14.6–27.0 | 32.1–43.9 | 22.9–32.8 |
| L. reflexus | 3.6–7.2 | 6.6–7.9 | 15.2–16.6 | 37.3–38.8 | 32.1–34.6 |
| L. rotundiflorus | 3.1–4.1 | 5.5–6.4 | 151 | 41.9–42.8 | 32.5 |
| L. simulans | 3.6 | 6.3 | 14.4 | 40.7 | 35.0 |
| L. splendens | 3.3–4.3 | 8.9–13.0 | 12.7–16.4 | 34.1–37.2 | 32.1–38.1 |
| L. mexicanus | 3.8–4.1 | 6.1–8.0 | 16.8 | 34.7–36.7 | 34.3 |
| L. madrensis | 3.5 | 6.8 | 15.4 | 41.4 | 32.8 |
| L. campestris | 4.4 | 7.5 | 40.5 | 39.3 | |
| L. montanus | 3.6–4.3 | 7.1–10.0 | 26.5 | 42.4–45.9 | 28.3 |
| L. hintonni | 6.3 | 7.0 | 32.5 | 24.4 |
| Protein | Lipids | Ash | Fiber | Carbohydrates | Alkaloids | |
|---|---|---|---|---|---|---|
| L. campestris | 93.2 | -- | 2.4 | 0.5 | 3.9 | 0.005 |
| L. exaltatus | 95.0 | 0.57 | 2.5 | 0.0 | 1.97 | 0.09 |
| L. elegans | 95 | 0.6 | 1.2 | 0.0 | 1.2 | 0.003 |
| State Location | L. exaltatus, Jalisco | L. montanus, Jalisco | L. stipulatus, Zacatecas | ||||
|---|---|---|---|---|---|---|---|
| CG 1 | CG 2 | Z 3 | T 4 | T 5 | B 6 | ME 7 | |
| C14:0 | 2 | 2 | 2 | 8 | 9 | 7 | 8 |
| C16:0 | 200 | 202 | 206 | 259 | 257 | 273 | 264 |
| C18:0 | 48 | 56 | 66 | 36 | 37 | 69 | 76 |
| C20:0 | - | - | - | - | - | - | - |
| C22:0 | - | - | - | 23 | 22 | 23 | 25 |
| C16:1 | 2 | 2 | 1 | 7 | 6 | 7 | 7 |
| C18:1 | 138 | 124 | 138 | 96 | 99 | 171 | 137 |
| C18:2 | 534 | 544 | 520 | 430 | 423 | 324 | 370 |
| C18:3 | 76 | 71 | 67 | 142 | 146 | 126 | 113 |
| C22:1 | - | - | - | - | - | - | - |
| n6/n3 | 7/1 | 7.5/1 | 7.7/1 | 3/1 | 2.9/1 | 2.6/1 | 3.2/1 |
| Ratio I/S | 3.0 | 2.8 | 2.6 | 2.1 | 2.1 | 1.7 | 1.7 |
| Species | Dietary Fiber |
|---|---|
| L. exaltatus | 17.72 ± 0.1 |
| L. elegans | 21.07 ± 0.0 |
| L. mexicanus | 20.90 ± 0.5 |
| L. montanus | 24.63 ± 0.1 |
| L. rotundiflorus | 27.93 ± 0.1 |
| Species | Ca | P | Mg | Fe | Zn | Cu |
|---|---|---|---|---|---|---|
| L. exaltatus | 1600–2052 | 584–5600 | 2300–2330 | 61.8–81.3 | 46.97–89.6 | 74.4–184 |
| L. elegans | 1777 | 6441 | 2656 | 70.9 | 73.6 | 64.8 |
| L. mexicanus | 3252 | 5865 | 2651 | 63.1 | 73.7 | 70.8 |
| L. montanus | 800–2074 | 6500–7690 | 2443–3000 | 77.7–73.7 | 46.87–73.3 | 56.2–89.7 |
| L. rotundiflorus | 1887 | 6166 | 2213 | 82.8 | 79.0 | 64.9 |
| L. campestris | 1200 | 6300 | 2300 | 70.74 | 46.97 | 93.3 |
| L. hintonii | 1000 | 5400 | 2600 | 49.39 | 38.18 | 67.7 |
| Species | Sucrose | Raffinose | Stachyose | Verbascose | Totals |
|---|---|---|---|---|---|
| L. campestris | 21.5 ± 0.76 | 11.7–13.65 | 57.16 ± 0.95 | 19.45 ± 0.59 | 90.26 ± 0.73 |
| Lupinus elegans | 7.82 ± 0.42 | 25.50 ± 1.21 | 37.20 ± 2.08 | 70.50 ± 3.71 | |
| Lupinus montanus | 14.55 ± 0.92 | 37.32 ± 2.52 | 12.44 ± 0.94 | 64.31 ± 4.38 | |
| Lupinus rotundiflorus | 5.60 ± 0.34 | 19.34 ± 0.82 | 29.57 ± 1.87 | 54.50 ± 3.03 |
| Lupinus species | Sparteine | Lupanine | 3-hydroxy lupanine | 13-hydroxy lupanine | Multiflorine | Hydroxyaphylline | Hydroxyaphyllidine |
|---|---|---|---|---|---|---|---|
| L. exaltatus | 0.03 | 1.47–5.83 | 1.53 | 0.015 | 0.004 | ||
| L. elegans | nd | 0.03 | 3.73 | nd | |||
| L. splendens | 0.29 | 0.89 | 1.05 | 1.00 | |||
| L. reflexus | 26.63 | 2.91 | 0.16 | 0.08 | |||
| L. rotundiflorus | 0.11 | 11.50 | 4.19 | nd | |||
| L. simulans | 0.40 | 8.87 | 2.76 | 0.09 | |||
| L. madrensis | 0.02 | 10.63 | 2.08 | 0.03 | |||
| L. montanus | 3.97 | 1.65 | 0.10 | 0.07 | |||
| L. stipulatus | 0.04 | 0.1 | 0.12 | 0.2 | |||
| L. mexicanus | 5.05–21.2 | 0.015 | 0.096–1.09 | ||||
| L. campestris | 1.2 | 498.0 | 1475.0 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ruiz-López, M.A.; Barrientos-Ramírez, L.; García-López, P.M.; Valdés-Miramontes, E.H.; Zamora-Natera, J.F.; Rodríguez-Macias, R.; Salcedo-Pérez, E.; Bañuelos-Pineda, J.; Vargas-Radillo, J.J. Nutritional and Bioactive Compounds in Mexican Lupin Beans Species: A Mini-Review. Nutrients 2019, 11, 1785. https://doi.org/10.3390/nu11081785
Ruiz-López MA, Barrientos-Ramírez L, García-López PM, Valdés-Miramontes EH, Zamora-Natera JF, Rodríguez-Macias R, Salcedo-Pérez E, Bañuelos-Pineda J, Vargas-Radillo JJ. Nutritional and Bioactive Compounds in Mexican Lupin Beans Species: A Mini-Review. Nutrients. 2019; 11(8):1785. https://doi.org/10.3390/nu11081785
Chicago/Turabian StyleRuiz-López, Mario Alberto, Lucia Barrientos-Ramírez, Pedro Macedonio García-López, Elia Herminia Valdés-Miramontes, Juan Francisco Zamora-Natera, Ramón Rodríguez-Macias, Eduardo Salcedo-Pérez, Jacinto Bañuelos-Pineda, and J. Jesús Vargas-Radillo. 2019. "Nutritional and Bioactive Compounds in Mexican Lupin Beans Species: A Mini-Review" Nutrients 11, no. 8: 1785. https://doi.org/10.3390/nu11081785
APA StyleRuiz-López, M. A., Barrientos-Ramírez, L., García-López, P. M., Valdés-Miramontes, E. H., Zamora-Natera, J. F., Rodríguez-Macias, R., Salcedo-Pérez, E., Bañuelos-Pineda, J., & Vargas-Radillo, J. J. (2019). Nutritional and Bioactive Compounds in Mexican Lupin Beans Species: A Mini-Review. Nutrients, 11(8), 1785. https://doi.org/10.3390/nu11081785