Age-Associated Changes in Gut Microbiota and Dietary Components Related with the Immune System in Adulthood and Old Age: A Cross-Sectional Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Volunteers and Samples
2.2. Quantification of Fecal Microbiota Groups by Quantitative Polymerase Chain Reaction (q PCR)
2.3. Analysis of SCFAs in Feces by Gas Chromatography-Mass Spectrometry/Flame Injection Detector (MS/FID)
2.4. Nutritional Assessment
2.5. Statistical Analyses
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Rinninellla, E.; Raoul, P.; Cintoni, M.; Franceschi, F.; Miggiano, G.A.D.; Gasbarrini, A.; Mele, M.C. What is the healthy gut microbiota composition? A changing ecosystem across age, environment, diet, and diseases. Microorganisms 2019, 7, 14. [Google Scholar] [CrossRef] [PubMed]
- Arumugam, M.; Raes, J.; Pelletier, E.; Le Paslier, D.; Yamada, T.; Mende, D.R.; Fernandes, G.R.; Tap, J.; Bruls, T.; Batto, J.M.; et al. Enterotypes of the human gut microbiome. Nature 2011, 473, 174–180. [Google Scholar] [CrossRef] [PubMed]
- Eckburg, P.B.; Bik, E.M.; Bernstein, C.N.; Purdom, E.; Dethlefsen, L.; Sargent, M.; Gill, S.R.; Nelson, K.E.; Relman, D.A. Diversity of the human intestinal microbial flora. Science 2005, 308, 1635–1638. [Google Scholar] [CrossRef] [PubMed]
- Salazar, L.; Valdés-Varela, L.; Gonzalez, S.; Gueimonde, M.; de los Reyes-Gavilán, C.G. Nutrition and the gut microbiome in the elderly. Gut Microbes 2017, 8, 82–97. [Google Scholar] [CrossRef] [PubMed]
- United Nations, Department of Economic and Social Affairs, Population Division. World Population Ageing. 2015. Available online: http://papersmart.unmeetings.org/media2/2827891/cpd2014_side-event-for-website_nos-03042014.pdf (accessed on 25 June 2019).
- Biagi, E.; Nylund, L.; Candela, M.; Ostan, R.; Bucci, L.; Pini, E.; Nikkïla, J.; Monti, D.; Satokari, R.; Franceschi, C.; et al. Through ageing, and beyond: Gut microbiota and inflammatory status in seniors and centenarians. PLoS ONE 2010, 5, e10667. [Google Scholar] [CrossRef]
- Biagi, E.; Franceschi, C.; Rampelli, S.; Capri, M.; Brigidi, P.; Candela, M.; Consolandi, C.; Quercia, S.; Scurti, M.; Monti, D.; et al. Gut microbiota and extreme longevity. Curr. Biol. 2016, 26, 1480–1485. [Google Scholar] [CrossRef]
- Claesson, M.J.; Cusack, S.; O’Sullivan, O.; Greene-Diniz, R.; de Weerd, H.; Flannery, E.; Marchesi, J.R.; Falush, D.; Dinan, T.; Fitzgerald, G.; et al. Composition, variability, and temporal stability of the intestinal microbiota of the elderly. Proc. Natl. Acad. Sci. USA 2011, 108 (Suppl. 1), 4586–4591. [Google Scholar] [CrossRef]
- Claesson, M.J.; Jeffery, I.B.; Conde, S.; Power, S.E.; O’Connor, E.M.; Cusack, S.; Harris, H.; Coakley, M.; Lakshminarayanan, B.; O’Sullivan, O.; et al. Gut microbiota composition correlates with diet and health in the elderly. Nature 2012, 488, 178–184. [Google Scholar] [CrossRef]
- Salazar, N.; Lopez, P.; Valdes, L.; Margolles, A.; Suarez, A.; Patterson, A.M.; Cuervo, A.; de los Reyes-Gavilán, C.G.; Ruas-Madiedo, P.; Gonzalez, S.; et al. Microbial targets for the development of functional foods accordingly with nutritional and immune parameters altered in the elderly. J. Am. Coll. Nutr. 2013, 32, 399–406. [Google Scholar] [CrossRef]
- Makivuokko, H.; Tiihonen, K.; Tynkkynen, S.; Paulin, L.; Rautonen, N. The effect of age and non-steroidal anti-inflammatory drugs on human intestinal microbiota composition. Br. J. Nutr. 2010, 103, 227–234. [Google Scholar] [CrossRef]
- Woodmansey, E.J. Intestinal bacteria and ageing. J. Appl. Microbiol. 2007, 102, 1178–1186. [Google Scholar] [CrossRef] [PubMed]
- Gueimonde, M.; Ouwehand, A.; Pitkala, K.; Standberg, T.; Finne-Soveri, H.; Salminen, S. Fecal Bifidobacterium levels in elderly nursing home patients- Are levels as expected? Biosci. Microflora 2010, 29, 111–113. [Google Scholar] [CrossRef]
- Hopkins, M.J.; Sharp, R.; Macfarlane, G.T. Age and disease related changes in intestinal bacterial populations assessed by cell culture, 16S rRNA abundance, and community cellular fatty acid profiles. Gut 2001, 48, 198–205. [Google Scholar] [CrossRef] [PubMed]
- Candore, G.; Caruso, C.; Jirillo, E.; Magrone, T.; Vasto, S. Low Grade Inflammation as a Common Pathogenic Denominator in Age-Related Diseases: Novel Drug Targets for Anti-Ageing Strategies and Successfully Ageing Achievement. Curr. Pharm. Des. 2010, 16, 584–596. [Google Scholar] [CrossRef] [PubMed]
- Kong, F.; Hua, Y.; Zeng, B.; Ning, R.; Li, Y.; Zhao, J. Gut microbiota signatures of longevity. Curr. Biol. 2016, 26, R832–R833. [Google Scholar] [CrossRef] [PubMed]
- Santoro, A.; Ostan, R.; Candela, M.; Biagi, E.; Brigidi, P.; Capri, M.; Franceschi, C. Gut microbiota changes in the extreme decades of human life: A focus on centenarians. Cell Mol. Life Sci. 2018, 75, 129–148. [Google Scholar] [CrossRef]
- Franceschi, C.; Ostan, R.; Santoro, A. Nutrition and Inflammation: Are Centenarians Similar to Individuals on Calorie-Restricted Diets? Annu. Rev. Nutr. 2018, 38, 329–356. [Google Scholar] [CrossRef] [PubMed]
- Nogacka, A.; Salazar, N.; Suarez, M.; Milani, C.; Arboleya, S.; Solis, G.; Fernandez, N.; Alaez, L.; Hernandez-Barranco, A.M.; de los Reyes-Gavilan, C.G.; et al. Impact of intrapartum antimicrobial prophylaxis upon the intestinal microbiota and the prevalence of antibiotic resistance genes in vaginally delivered full-term neonates. Microbiome 2017, 5, 93. [Google Scholar] [CrossRef]
- Valdes, L.; Salazar, N.; Gonzalez, S.; Arboleya, S.; Ríos-Covian, D.; Genovés, S.; Ramón, D.; de Los Reyes-Gavilán, C.G.; Ruas-Madiedo, P.; Gueimonde, M. Selection of potential probiotic bifidobacteria and prebiotics for elderly by using in vitro faecal batch cultures. Eur. Food Res. Technol. 2017, 243, 157–165. [Google Scholar] [CrossRef]
- Arboleya, S.; Binetti, A.; Salazar, N.; Fernández, N.; Solís, G.; Hernández-Barranco, A.; Margolles, A.; de Los Reyes-Gavilán, C.G.; Gueimonde, M. Establishment and development of intestinal microbiota in preterm neonates. FEMS Microbiol. Ecol. 2012, 79, 763–772. [Google Scholar] [CrossRef]
- Cuervo, A.; Valdés, L.; Salazar, N.; de los Reyes-Gavilán, C.G.; Ruas-Madiedo, P.; Gueimonde, M.; González, S. Pilot study of diet and microbiota: Interactive associations of fibers and polyphenols with human intestinal bacteria. J. Agric. Food Chem. 2014, 62, 5330–5336. [Google Scholar] [CrossRef] [PubMed]
- Palma, I.; Farran, A.; Cervera, S.P. Tablas de composición de alimentos por medidas caseras de consumo habitual en españa. Actividad Dietética 2008, 12, 85. [Google Scholar] [CrossRef]
- Neveu, V.; Perez-Jiménez, J.; Vos, F.; Crespy, V.; du Chaffaut, L.; Mennen, L.; Knox, C.; Eisner, R.; Cruz, J.; Wishart, D.; et al. Phenol-Explorer: An online comprehensive database on polyphenol contents in foods. Database 2010. [Google Scholar] [CrossRef] [PubMed]
- Forssten, S.D.; Salazar, N.; López, P.; Nikkilä, J.; Ouwehand, A.; Paterson, A.; Ruas-Madiedo, P.; Suárez, A.L.; González, S.F.; Gueimonde, M. Influence of a probiotic milk drink, containing Lactobacillus paracasei Lpc-37, on immune function and gut microbiota in elderly subjects. Eur. J. Food Res. Rev. 2011, 1, 159–172. [Google Scholar] [CrossRef]
- O’Toole, P.W.; Claesson, M.J. Gut microbiota: Changes throughout the lifespan from infancy to elderly. Int. Dairy J. 2010, 20, 281–291. [Google Scholar] [CrossRef]
- Candela, M.; Biagi, E.; Brigidi, P.; O’Toole, P.W.; de Vos, W.M. Maintenance of a healthy trajectory of the intestinal microbiome during aging: A dietary approach. Mech. Ageing Dev. 2014, 136–137, 70–75. [Google Scholar] [CrossRef]
- Odamaki, T.; Kato, K.; Sugahara, H.; Hashikura, N.; Takahashi, S.; Xiao, J.-Z.; Abe, F.; Osawa, R. Age-related changes in gut microbiota composition from newborn to centenarian: A cross-sectional study. BMC Microbiol. 2016, 16, 90. [Google Scholar] [CrossRef]
- Barcena, C.; Valdés-Mas, R.; Mayoral, P.; Garabaya, C.; Durand, S.; Rodriguez, T.; Fernández-García, M.T.; Salazar, N.; Nogacka, A.M.; Garatachea, N. Healthspan and Lifespan extensión by fecal microbiota transplantation into progeroid mice. Nat. Med. 2019. [Google Scholar] [CrossRef]
- Collado, M.C.; Derrien, M.; Isolauri, E.; de Vos, W.M.; Salminen, S. Intestinal integrity and Akkermansia muciniphila, a mucin-degrading member of the intestinal microbiota present in infants, adults, and the elderly. Appl. Environ. Microbiol. 2007, 73, 7767–7770. [Google Scholar] [CrossRef]
- Woodmansey, E.J.; McMurdo, M.E.T.; Macfarlane, G.T.; Macfarlane, S. Comparison of compositions and metabolic activities of fecal microbiotas in young adults and in antibiotic-treated and non-antibiotic-treated elderly subjects. Appl. Environ. Microbiol. 2004, 70, 6113–6122. [Google Scholar] [CrossRef]
- Ríos-Covian, D.; Ruas-Madiedo, P.; Margolles, A.; Gueimonde, M.; de los Reyes-Gavilan, C.G.; Salazar, N. Intestinal short chain fatty acids and their link with diet and human health. Front. Microbiol. 2016, 7, 185. [Google Scholar] [CrossRef] [PubMed]
- Cuervo, A.; Salazar, N.; Ruas-Madiedo, P.; Gueimonde, M.; González, S. Fiber from a regular diet is directly associated with fecal short-chain fatty acid concentrations in the elderly. Nutr. Res. 2013, 33, 811–816. [Google Scholar] [CrossRef] [PubMed]
- Ding, S.; Jiang, H.; Fang, J. Regulation of Immune Function by Polyphenols. J. Immunol. Res. 2018, 1264074. [Google Scholar] [CrossRef] [PubMed]
- Valdés, L.; Cuervo, A.; Salazar, N.; Ruas-Madiedo, P.; Gueimonde, M.; González, S. The relationship between phenolic compounds from diet and microbiota: Impact on human health. Food Funct. 2015, 6, 2424–2439. [Google Scholar] [CrossRef] [PubMed]
- González, S.; López, P.; Margolles, A.; Suárez, A.; Patterson, A.M.; Cuervo, A.; de los Reyes-Gavilán, C.G.; Gueimonde, M. Fatty acids intake and immune parameters in the elderly. Nutr. Hosp. 2013, 28, 474–478. [Google Scholar] [CrossRef] [PubMed]
- Kuikka, L.K.; Salminen, S.; Ouwehand, A.; Gueimonde, M.; Strandberg, T.E.; Finne-Soveri, U.H.; Sintonen, H.; Pitkälä, K.H. Inflammation markers and malnutrition as risk factors for infections and impaired health-related quality of life among older nursing home residents. J. Am. Med. Dir. Assoc. 2009, 10, 348–353. [Google Scholar] [CrossRef]
- Maggini, S.; Pierre, A.; Calder, P.C. Immune Function and Micronutrient Requirements Change over the Life Course. Nutrients 2018, 10, 1531. [Google Scholar] [CrossRef]
- Pae, M.; Wu, D. Nutritional modulation of age-related changes in the immune system and risk of infection. Nutr. Res. 2017, 41, 14–35. [Google Scholar] [CrossRef]
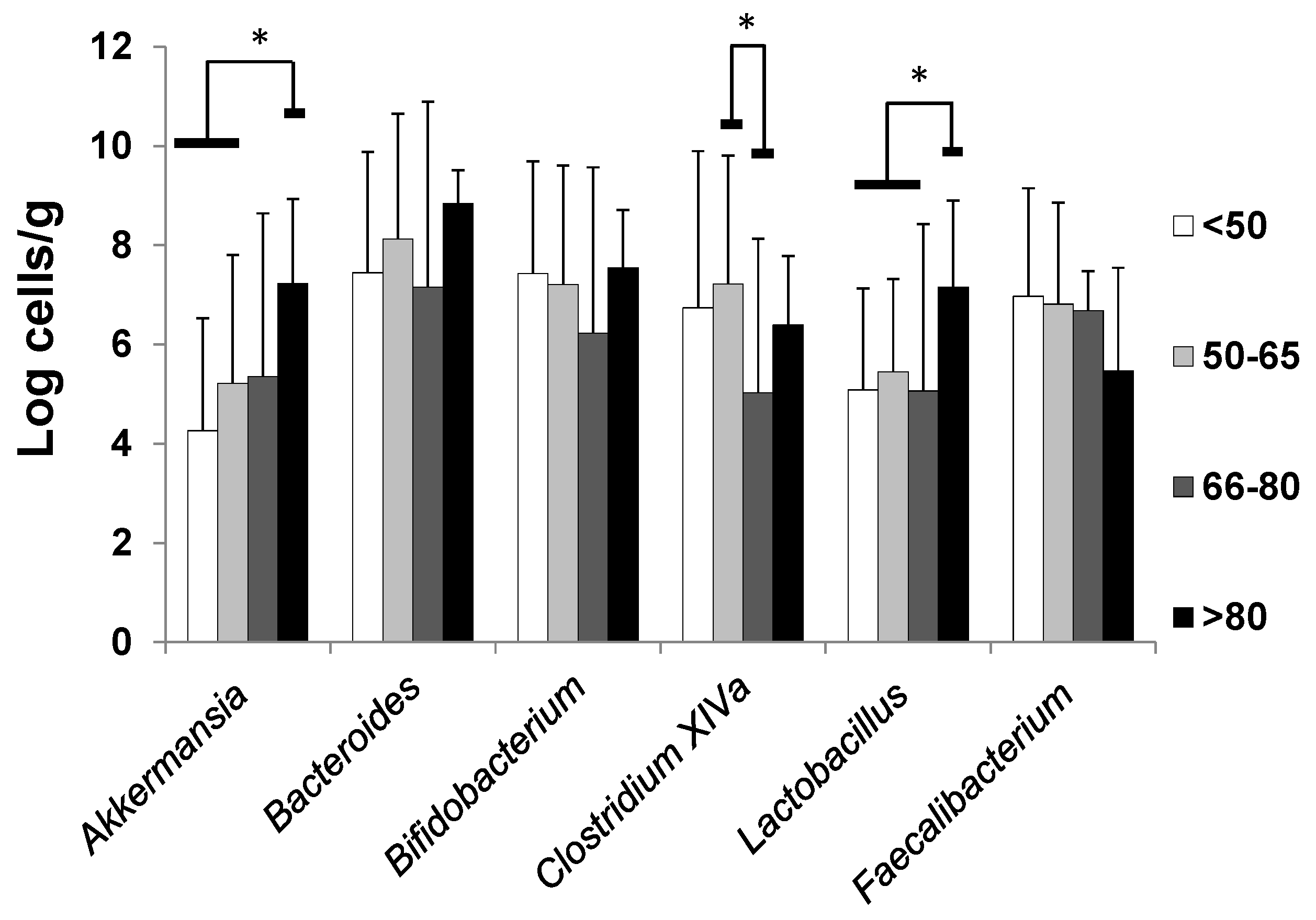
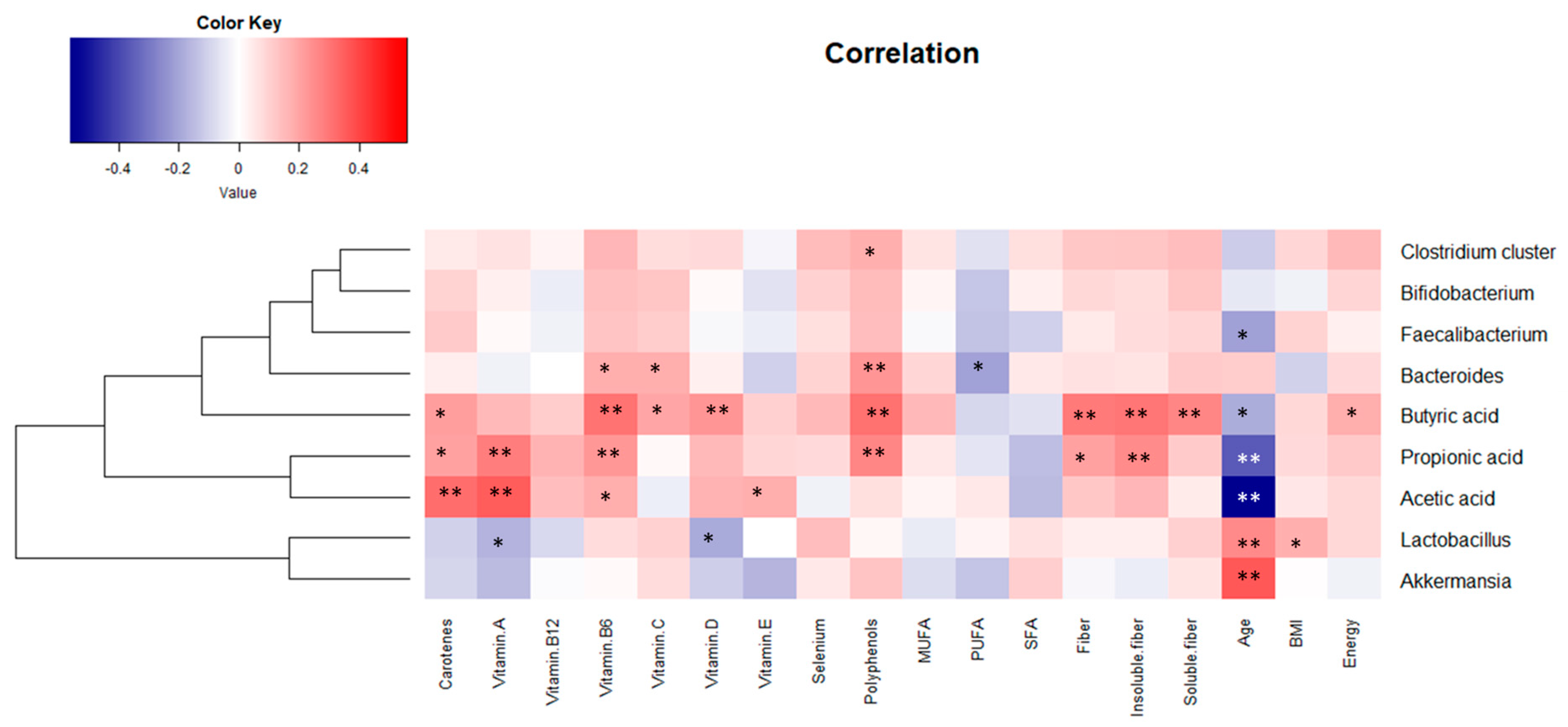
| Variables | Age Group (Years) | |||
|---|---|---|---|---|
| <50 (n = 49) | 50–65 (n = 58) | 66–80 (n = 19) | >80 (n = 27) | |
| Female (%) | 65.3 | 69.0 | 78.9 | 77.8 |
| Age | 37.6 ± 6.4 | 58.1 ± 4.0 | 72.8 ± 5.2 | 85.7 ± 4.4 |
| BMI (kg/m2) | 27.89 ± 5.21 a | 25.64 ± 3.19 b | 29.69 ± 4.31 a | 28.33 ± 3.98 a,b |
| SCFA | Age Group (Years) | |||
|---|---|---|---|---|
| <50 | 50–65 | 66–80 | >80 | |
| Acetate | 49.4 ± 18.3 a | 32.9 ± 14.7 b | 31.6 ± 17.7 bc | 18.8 ± 11.4 c |
| Propionate | 16.0 ± 7.8 a | 12.6 ± 5.9 ab | 12.1 ± 8.1 ab | 8.1 ± 6.8 b |
| Butyrate | 10.9 ± 5.3 a | 10.4 ± 8.1 ab | 12.1 ± 8.3 ab | 6.3 ± 6.4 b |
| TOTAL | 76.4 ± 27.9 a | 56.0 ± 25.4 b | 55.8 ± 32.3 bc | 32.3 ± 24.1 c |
| Age Group (Years) | ||||
|---|---|---|---|---|
| <50 | 50–65 | 66–80 | >80 | |
| Energy (kcal/day) # Food groups (g) | 1922 ± 575 a,b | 2033 ± 581 a | 1624 ± 435 b | 1728 ± 400 a,b |
| Cereals | 176.3 ± 9.7 a | 185.0 ± 9.0 a | 143.9 ± 15.8 a | 153.5 ± 13.4 a |
| Dairy products | 311.7 ± 29.8 a | 396.7 ± 27.7 a | 410.8 ± 48.6 a | 408.0 ± 41.3 a |
| Fats and oils | 25.2 ± 2.4 a | 30.1 ± 2.2 a | 23.8 ± 3.9 a | 24.0 ± 3.3 a |
| Sugar/sugary products ** | 29.7 ± 2.8 a | 11.0 ± 2.6 b | 13.8 ± 4.6 b | 11.2 ± 3.9 b |
| Vegetables * | 157.6 ± 19.0 a,b | 205.2 ± 17.6a | 164.0 ± 31.0 a,b | 105.1 ± 26.2 b |
| Potatoes ** | 45.8 ± 6.8 a,b | 43.1 ± 6.3 a | 69.8 ± 11.3 b | 68.6 ± 9.4 a,b |
| Legumes | 28.7 ± 12.4 a | 37.8 ± 11.5 a | 27.5 ± 20.1 a | 21.5 ± 17.1 a |
| Fruit ** | 193.8 ± 27.6 a | 343.9 ± 25.6 b | 271.4 ± 45.0 a,b | 205.2 ± 38.2 a |
| Meat and derivate | 134.8 ± 7.9 a | 115.5 ± 7.3 a | 110.9 ± 12.9 a | 122.9 ± 10.9 a |
| Fish and derivate ** | 42.2 ± 5.5 a | 60.7 ± 5.1 a,b | 88.2 ± 8.9 b | 89.1 ± 7.6 b |
| Non-alcoholic drinks ** | 307.3 ± 27.6 a | 167.3 ± 25.7 b | 182.8 ± 45.0 b | 145.0 ± 38.2 b |
| Dietary components | ||||
| Carbohydrates (g) | 206.9 ± 4.9 a | 199.2 ± 4.6 a | 196.8 ± 8.1 a | 188.9 ± 6.8 a |
| Total fiber ** | 17.5 ± 0.7 a | 22.1 ± 0.7 b | 19.3 ± 1.2 a | 17.5 ± 1.0 a |
| Insoluble fiber ** | 11.6 ± 5.6 a | 14.3 ± 5.4 b | 12.4 ± 4.0 a,b | 10.2 ± 3.8 a |
| Soluble fiber ** | 2.1 ± 0.8 a | 2.9 ± 1.3 b | 2.6 ± 0.8 a,b | 2.1 ± 0.9 a |
| Proteins (g) | 83.8 ± 2.5 a | 89.6 ± 2.3 a | 89.7 ± 4.0 a | 90.9 ± 3.4 a |
| Animal | 55.3 ± 2.5 a | 58.6 ± 2.3 a | 59.9 ± 4.0 a | 61.1 ± 3.4 a |
| Vegetal | 25.0 ± 1.1 a | 29.2 ± 1.0 a | 27.6 ± 1.8 a | 27.9 ± 1.6 a |
| Lipids (g) | 78.3 ± 2.1 a | 78.3 ± 2.0 a | 79.2 ± 3.5 a | 85.4 ± 3.0 a |
| SFAs ** | 24.8 ± 1.0 a | 25.1 ± 0.9 a | 28.0 ± 1.6 a,b | 30.4 ± 1.4 b |
| MUFAs ** | 31.0 ± 1.4 a,b | 34.9 ± 1.3 a | 28.9 ± 2.3 b | 28.5 ± 2.0 b |
| PUFAs ** | 16.1 ± 1.0 a,b | 11.9 ± 0.9 a | 15.7 ± 1.7 a,b | 19.6 ± 1.4 b |
| Vitamins | ||||
| Carotenes (μg) ** | 2048 ± 1583 a | 2464 ± 1821 a | 1899 ± 2278 a,b | 1014 ± 315 b |
| Folic acid (μg) ** | 308.3± 128.1 a | 390.8 ± 177.6 b | 330.3 ± 128.4 a | 275.3 ± 82.64 a |
| Vitamin A (μg) ** | 822.2 ± 477.8 a | 743.2 ± 435.5 a | 619.7 ± 425.3 b | 430.7 ± 129.8 b |
| Vitamin B12 (μg) | 7.7 ± 7.0 a | 7.1 ± 4.3 a | 8.2 ± 3.1 a | 8.7 ± 2.6 a |
| Vitamin B6 (mg) ** | 1.9 ± 0.7 a | 2.2 ± 0.7 b | 2.1 ± 0.6 a,b | 1.8 ± 0.4 a |
| Vitamin C (mg) ** | 123.7 ± 72.2 a | 199.6 ± 165.2 b | 187.2 ± 97.2 a,b | 155.2 ± 74.8 a |
| Vitamin D (μg) ** | 2.8 ± 1.9 a | 3.8 ± 2.6 b | 1.7 ± 1.7 a,c | 0.8 ± 0.3 c |
| Vitamin E (mg) ** | 12.7 ± 6.8 a | 10.5 ± 5.0 a | 12.1 ± 5.1 a | 13.7 ± 5.5 a |
| Minerals | ||||
| Copper (mg) | 1.4 ± 0.7 a | 1.3 ± 0.5 a | 1.3 ± 0.4 a | 1.1± 0.3 a |
| Iron (mg) | 12.2 ± 5.2 a | 12.8 ± 3.9 a | 13.2 ± 3.7 a | 12.6 ± 2.7 a |
| Selenium (μg) * | 118.9 ± 52.8 a | 122.0 ± 42.4 a | 136.5 ± 38.3 a | 137.6 ± 33.9 a |
| Total polyphenols (mg) ** | 1202 ± 651 a | 1721 ± 938 b | 1265 ± 1324 a | 841 ± 658 a |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Salazar, N.; Arboleya, S.; Fernández-Navarro, T.; de los Reyes-Gavilán, C.G.; Gonzalez, S.; Gueimonde, M. Age-Associated Changes in Gut Microbiota and Dietary Components Related with the Immune System in Adulthood and Old Age: A Cross-Sectional Study. Nutrients 2019, 11, 1765. https://doi.org/10.3390/nu11081765
Salazar N, Arboleya S, Fernández-Navarro T, de los Reyes-Gavilán CG, Gonzalez S, Gueimonde M. Age-Associated Changes in Gut Microbiota and Dietary Components Related with the Immune System in Adulthood and Old Age: A Cross-Sectional Study. Nutrients. 2019; 11(8):1765. https://doi.org/10.3390/nu11081765
Chicago/Turabian StyleSalazar, Nuria, Silvia Arboleya, Tania Fernández-Navarro, Clara G. de los Reyes-Gavilán, Sonia Gonzalez, and Miguel Gueimonde. 2019. "Age-Associated Changes in Gut Microbiota and Dietary Components Related with the Immune System in Adulthood and Old Age: A Cross-Sectional Study" Nutrients 11, no. 8: 1765. https://doi.org/10.3390/nu11081765
APA StyleSalazar, N., Arboleya, S., Fernández-Navarro, T., de los Reyes-Gavilán, C. G., Gonzalez, S., & Gueimonde, M. (2019). Age-Associated Changes in Gut Microbiota and Dietary Components Related with the Immune System in Adulthood and Old Age: A Cross-Sectional Study. Nutrients, 11(8), 1765. https://doi.org/10.3390/nu11081765





