Food Intolerances
Abstract
1. Introduction
2. Sensitivities (Non-Allergic)
2.1. FODMAPs
2.1.1. FODMAP Induced Response
2.1.2. Proposed Mechanisms of FODMAP Induced Symptoms
2.1.3. Diagnosis and Management of FODMAP Related Intolerances
2.2. Wheat
2.2.1. Wheat Induced Response
2.2.2. Proposed Mechanisms of Wheat Related Intolerances
2.2.3. Diagnosis and Management of Wheat Related Intolerances
2.3. Histamine
2.3.1. Histamine Induced Response
2.3.2. Proposed Mechanisms of Histamine Intolerance
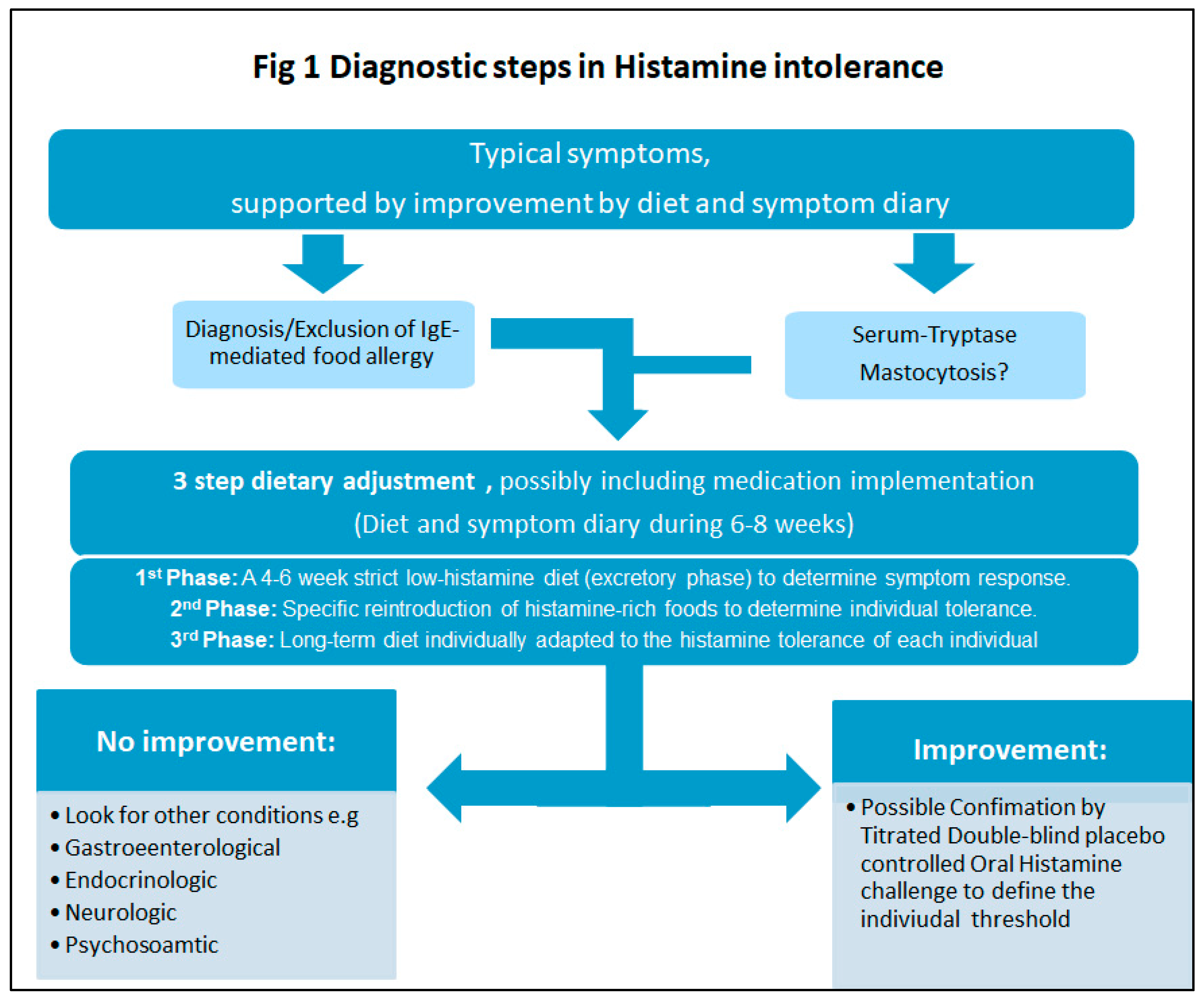
2.3.3. Diagnosis and Management of Histamine Intolerance
2.4. Food Additives and Bioactive Food Chemicals
2.4.1. Food Additives and Chemical Induced Response
2.4.2. Proposed Mechanisms of Food Additives and Chemicals
2.4.3. Diagnosis and Management of Food Additive and Chemical Sensitivity
3. Genetic
3.1. Sucrose and Starch (Sucrase-Isomaltase Deficiency)
3.1.1. Sucrase-Isomaltase Deficiency Response
3.1.2. Proposed Mechanisms of Sucrase-Isomaltase Deficiency
3.1.3. Diagnosis and Management of Sucrase-Isomaltase Deficiency
4. Discussion
4.1. Recommendations for Future Research
4.2. Implications and Recommendations for Clinical Practice
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Rozin, P.; Zellner, D. The role of Pavlovian conditioning in the acquisition of food likes and dislikes. Ann. N. Y. Acad. Sci. 1985, 443, 189–202. [Google Scholar] [CrossRef] [PubMed]
- Turnbull, J.L.; Adams, H.N.; Gorard, D.A. Review article: The diagnosis and management of food allergy and food intolerances. Aliment. Pharmacol. Ther. 2015, 41, 3–25. [Google Scholar] [CrossRef] [PubMed]
- Lomer, M.C. Review article: The aetiology, diagnosis, mechanisms and clinical evidence for food intolerance. Aliment. Pharmacol. Ther. 2015, 41, 262–275. [Google Scholar] [CrossRef] [PubMed]
- Halmos, E.P.; Power, V.A.; Shepherd, S.J.; Gibson, P.R.; Muir, J.G. A Diet Low in FODMAPs Reduces Symptoms of Irritable Bowel Syndrome. Gastroenterology 2014, 146, 67–75. [Google Scholar] [CrossRef] [PubMed]
- Varjú, P.; Farkas, N.; Hegyi, P.; Garami, A.; Szabó, I.; Illés, A.; Solymár, M.; Vincze, Á.; Balaskó, M.; Pár, G.; et al. Low fermentable oligosaccharides, disaccharides, monosaccharides and polyols (FODMAP) diet improves symptoms in adults suffering from irritable bowel syndrome (IBS) compared to standard IBS diet: A meta-analysis of clinical studies. PLoS ONE 2017, 12, e0182942. [Google Scholar] [CrossRef]
- Schumann, D.; Klose, P.; Lauche, R.; Dobos, G.; Langhorst, J.; Cramer, H. Low FODMAP Diet in the Treatment of Irritable Bowel Syndrome: A Systematic Review and Meta-Analysis. Nutrition 2018, 45, 24–31. [Google Scholar] [CrossRef] [PubMed]
- McKenzie, Y.A.; Bowyer, R.K.; Leach, H.; Gulia, P.; Horobin, J.; O’Sullivan, N.A.; Pettitt, C.; Reeves, L.B.; Seamark, L.; Williams, M.; et al. British Dietetic Association systematic review and evidence-based practice guidelines for the dietary management of irritable bowel syndrome in adults (2016 update). J. Hum. Nutr. Diet. 2016, 29, 549–575. [Google Scholar] [CrossRef]
- Muir, J.G.; Shepherd, S.J.; Rosella, O.; Rose, R.; Barrett, J.S.; Gibson, P.R. Fructan and free fructose content of common Australian vegetables and fruit. J. Agr. Food. Chem. 2007, 55, 6619. [Google Scholar] [CrossRef]
- Muir, J.G.; Rose, R.; Rosella, O.; Liels, K.; Barrett, J.S.; Shepherd, S.J.; Gibson, P.R. Measurement of short-chain carbohydrates in common Australian vegetables and fruits by high-performance liquid chromatography (HPLC). J. Agr. Food. Chem. 2009, 57, 554. [Google Scholar] [CrossRef]
- Biesiekierski, J.R.; Rosella, O.; Rose, R.; Liels, K.; Barrett, J.S.; Shepherd, S.J.; Gibson, P.R.; Muir, J.G. Quantification of fructans, galacto-oligosacharides and other short-chain carbohydrates in processed grains and cereals. J. Hum. Nutr. Diet. 2011, 24, 154–176. [Google Scholar] [CrossRef]
- Yao, C.; Tan, H.L.; Langenberg, D.; Barrett, J.; Rose, R.; Liels, K.; Gibson, P.; Muir, J. Dietary sorbitol and mannitol: Food content and distinct absorption patterns between healthy individuals and patients with irritable bowel syndrome. J. Hum. Nutr. Diet. 2013, 27, 264. [Google Scholar] [CrossRef] [PubMed]
- Tuck, C.J.; Ly, E.; Bogatyrev, A.; Costetsou, I.; Gibson, P.R.; Barrett, J.S.; Muir, J. Fermentable short chain carbohydrate (FODMAPs) content of common plant-based foods and processed foods suitable for vegetarian- and vegan-based eating patterns. J. Hum. Nutr. Diet. 2018, 31, 422–435. [Google Scholar] [CrossRef] [PubMed]
- Prichard, R.; Rossi, M.; Muir, J.; Yao, C.; Whelan, K.; Lomer, M. Fermentable oligosaccharide, disaccharide, monosaccharide and polyol content of foods commonly consumed by ethnic minority groups in the United Kingdom. Int. J. Food Sci. Nutr. 2016, 67, 383–390. [Google Scholar] [CrossRef] [PubMed]
- Monash University. The Monash University Low FODMAP App. Available online: http://www.med.monash.edu/cecs/gastro/fodmap/ (accessed on 26 April 2019).
- Staudacher, H.M.; Kurien, M.; Whelan, K. Nutritional implications of dietary interventions for managing gastrointestinal disorders. Curr. Opin. Gastroenterol. 2018, 34, 105–111. [Google Scholar] [CrossRef] [PubMed]
- Staudacher, H.M.; Lomer, M.C.E.; Farquharson, F.M.; Louis, P.; Fava, F.; Franciosi, E.; Scholz, M.; Tuohy, K.M.; Lindsay, J.O.; Irving, P.M.; et al. Diet Low in FODMAPs Reduces Symptoms in Patients with Irritable Bowel Syndrome and Probiotic Restores Bifidobacterium Species: A Randomized Controlled Trial. Gastroenterology 2017, 153, 936–947. [Google Scholar] [CrossRef] [PubMed]
- Harvie, R.; Chisholm, A.; Bisanz, J.; Burton, J.; Herbison, P.; Schultz, K.; Schultz, M. Long-term irritable bowel syndrome symptom control with reintroduction of selected FODMAPs. World J. Gastroenterol. 2017, 23, 4632–4643. [Google Scholar] [CrossRef] [PubMed]
- Böhn, L.; Störsrud, S.; Liljebo, T.; Collin, L.; Lindfors, P.; Törnblom, H.; Simrén, M. Diet low in FODMAPs reduces symptoms of irritable bowel syndrome as well as traditional dietary advice: A randomized controlled trial. Gastroenterology 2015, 149, 1399–1407. [Google Scholar] [CrossRef] [PubMed]
- Eswaran, S.L.; Chey, W.D.; Han-Markey, T.; Ball, S.; Jackson, K. A Randomized Controlled Trial Comparing the Low FODMAP Diet vs. Modified NICE Guidelines in US Adults with IBS-D. Am. J. Gastroenterol. 2016, 111, 1824–1832. [Google Scholar] [CrossRef] [PubMed]
- Masuy, I.; Van Oudenhove, L.; Tack, J.; Biesiekierski, J.R. Effect of intragastric FODMAP infusion on upper gastrointestinal motility, gastrointestinal, and psychological symptoms in irritable bowel syndrome vs healthy controls. Neurogastroenterol. Motil. 2018, 30, e13167. [Google Scholar] [CrossRef] [PubMed]
- Tan, V.P. The low-FODMAP diet in the management of functional dyspepsia in East and Southeast Asia. J. Gastroenterol. Hepatol. 2017, 32, 46–52. [Google Scholar] [CrossRef]
- Ong, D.K.; Mitchell, S.B.; Barrett, J.S.; Shepherd, S.J.; Irving, P.M.; Biesiekierski, J.R.; Smith, S.; Gibson, P.R.; Muir, J.G. Manipulation of dietary short chain carbohydrates alters the pattern of gas production and genesis of symptoms in irritable bowel syndrome. J. Gastroenterol. Hepatol. 2010, 25, 1366–1373. [Google Scholar] [CrossRef] [PubMed]
- Staudacher, H.M.; Whelan, K. The low FODMAP diet: Recent advances in understanding its mechanisms and efficacy in IBS. Gut 2017, 66, 1517–1527. [Google Scholar] [CrossRef]
- Barrett, J.S.; Gearry, R.B.; Muir, J.G.; Irving, P.M.; Rose, R.; Rosella, O.; Haines, M.L.; Shepherd, S.J.; Gibson, P.R. Dietary poorly absorbed, short-chain carbohydrates increase delivery of water and fermentable substrates to the proximal colon. Aliment. Pharmacol. Ther. 2010, 31, 874–882. [Google Scholar] [CrossRef] [PubMed]
- Murray, K.; Wilkinson-Smith, V.; Hoad, C.; Costigan, C.; Cox, E.; Lam, C.; Marciani, L.; Gowland, P.; Spiller, R.C. Differential Effects of FODMAPs (Fermentable Oligo-, Di-, Mono-Saccharides and Polyols) on Small and Large Intestinal Contents in Healthy Subjects Shown by MRI. Am. J. Gastroenterol. 2013, 109, 110–119. [Google Scholar] [CrossRef] [PubMed]
- Major, G.; Pritchard, S.; Murray, K.; Alappadan, J.P.; Hoad, C.L.; Marciani, L.; Gowland, P.; Spiller, R. Colon Hypersensitivity to Distension, Rather Than Excessive Gas Production, Produces Carbohydrate-Related Symptoms in Individuals With Irritable Bowel Syndrome. Gastroenterology 2017, 152, 124–133. [Google Scholar] [CrossRef] [PubMed]
- McIntosh, K.; Reed, D.E.; Schneider, T.; Dang, F.; Keshteli, A.H.; De Palma, G.; Madsen, K.; Bercik, P.; Vanner, S. FODMAPs alter symptoms and the metabolome of patients with IBS: A randomised controlled trial. Gut 2016, 66, 1241–1251. [Google Scholar] [CrossRef]
- Valeur, J.; Røseth, A.G.; Knudsen, T.; Malmstrøm, G.H.; Fiennes, J.T.; Midtvedt, T.; Berstad, A. Fecal Fermentation in Irritable Bowel Syndrome: Influence of Dietary Restriction of Fermentable Oligosaccharides, Disaccharides, Monosaccharides and Polyols. Digestion 2016, 94, 50–56. [Google Scholar] [CrossRef]
- Drossman, D.A. Functional Gastrointestinal Disorders: History, Pathophysiology, Clinical Features, and Rome IV. Gastroenterology 2016, 150, 1262–1279. [Google Scholar] [CrossRef]
- Barrett, J.S. How to institute the low-FODMAP diet. J. Gastroenterol. Hepatol. 2017, 32, 8–10. [Google Scholar] [CrossRef]
- Tuck, C.J.; Barrett, J.S. Re-challenging FODMAPs: The low FODMAP diet phase two. J. Gastroenterol. Hepatol. 2017, 32, 11–15. [Google Scholar] [CrossRef]
- Whelan, K.; Martin, L.; Staudacher, H.; Lomer, M. The low FODMAP diet in the management of irritable bowel syndrome: An evidence-based review of FODMAP restriction, reintroduction and personalisation in clinical practice. J. Hum. Nutr. Diet. 2018, 31, 239–255. [Google Scholar] [CrossRef] [PubMed]
- Hookway, C.; Buckner, S.; Crosland, P.; Longson, D. Irritable bowel syndrome in adults in primary care: summary of updated NICE guidance. BMJ 2015, 350, h701. [Google Scholar] [CrossRef] [PubMed]
- Staudacher, H.M.; Lomer, M.C.E.; Anderson, J.L.; Barrett, J.S.; Muir, J.G.; Irving, P.M.; Whelan, K. Fermentable carbohydrate restriction reduces luminal bifidobacteria and gastrointestinal symptoms in patients with irritable bowel syndrome. J. Nutr. 2012, 142, 1510–1518. [Google Scholar] [CrossRef] [PubMed]
- Halmos, E.P.; Christophersen, C.T.; Bird, A.R.; Shepherd, S.J.; Gibson, P.R.; Muir, J.G. Diets that differ in their FODMAP content alter the colonic luminal microenvironment. Gut 2015, 64, 93–100. [Google Scholar] [CrossRef] [PubMed]
- Biesiekierski, J.R.; Peters, S.L.; Newnham, E.D.; Rosella, O.; Muir, J.G.; Gibson, P.R. No Effects of Gluten in Patients with Self-Reported Non-Celiac Gluten Sensitivity Following Dietary Reduction of Low-Fermentable, Poorly-Absorbed, Short-Chain Carbohydrates. Gastroenterology 2013, 145, 320–328. [Google Scholar] [CrossRef] [PubMed]
- Staudacher, H.M.; Ralph, F.S.E.; Irving, P.M.; Whelan, K.; Lomer, M.C.E. Nutrient Intake, Diet Quality, and Diet Diversity in Irritable Bowel Syndrome and the Impact of the Low FODMAP Diet. J. Acad. Nutr. Diet. 2019, in press. [Google Scholar] [CrossRef] [PubMed]
- Trott, N.; Aziz, I.; Rej, A.; Surendran Sanders, D. How Patients with IBS Use Low FODMAP Dietary Information Provided by General Practitioners and Gastroenterologists: A Qualitative Study. Nutrients 2019, 11, 1313. [Google Scholar] [CrossRef]
- Altenbach, S.B.; Vensel, W.H.; Dupont, F.M. The spectrum of low molecular weight alpha-amylase/protease inhibitor genes expressed in the US bread wheat cultivar Butte 86. BMC Res. Notes 2011, 4, 242. [Google Scholar] [CrossRef]
- Sharon, N.; Lis, H. Lectins: Cell-agglutinating and sugar-specific proteins. Science 1972, 177, 949–959. [Google Scholar] [CrossRef]
- Catassi, C.; Elli, L.; Bonaz, B.; Bouma, G.; Carroccio, A.; Castillejo, G.; Cellier, C.; Cristofori, F.; de Magistris, L.; Dolinsek, J.; et al. Diagnosis of Non-Celiac Gluten Sensitivity (NCGS): The Salerno Experts’ Criteria. Nutrients 2015, 7, 4966–4977. [Google Scholar] [CrossRef]
- Biesiekierski, J.R.; Newnham, E.D.; Shepherd, S.J.; Muir, J.G.; Gibson, P.R. Characterization of Adults With a Self-Diagnosis of Nonceliac Gluten Sensitivity. Nutr. Clin. Pract. 2014, 29, 504–509. [Google Scholar] [CrossRef] [PubMed]
- Molina-Infante, J.; Carroccio, A. Suspected Nonceliac Gluten Sensitivity Confirmed in Few Patients After Gluten Challenge in Double-Blind, Placebo-Controlled Trials. Clin. Gastroenterol. Hepatol. 2017, 15, 339–348. [Google Scholar] [CrossRef] [PubMed]
- Di Sabatino, A.; Volta, U.; Salvatore, C.; Biancheri, P.; Caio, G.; De Giorgio, R.; Di Stefano, M.; Corazza, G.R. Small Amounts of Gluten in Subjects With Suspected Nonceliac Gluten Sensitivity: A Randomized, Double-Blind, Placebo-Controlled, Cross-Over Trial. Clin. Gastroenterol. Hepatol. 2015, 13, 1604–1612.e3. [Google Scholar] [CrossRef] [PubMed]
- Shahbazkhani, B.; Sadeghi, A.; Malekzadeh, R.; Khatavi, F.; Etemadi, M.; Kalantri, E.; Rostami-Nejad, M.; Rostami, K. Non-Celiac Gluten Sensitivity Has Narrowed the Spectrum of Irritable Bowel Syndrome: A Double-Blind Randomized Placebo-Controlled Trial. Nutrients 2015, 7, 4542–4554. [Google Scholar] [CrossRef] [PubMed]
- Uhde, M.; Ajamian, M.; Caio, G.; De Giorgio, R.; Indart, A.; Green, P.H.; Verna, E.C.; Volta, U.; Alaedini, A. Intestinal cell damage and systemic immune activation in individuals reporting sensitivity to wheat in the absence of coeliac disease. Gut 2016, 65, 1930–1937. [Google Scholar] [CrossRef] [PubMed]
- Carroccio, A.; Giannone, G.; Mansueto, P.; Soresi, M.; La Blasca, F.; Fayer, F.; Iacobucci, R.; Porcasi, R.; Catalano, T.; Geraci, G.; et al. Duodenal and Rectal Mucosa Inflammation in Patients With Non-celiac Wheat Sensitivity. Clin. Gastroenterol. Hepatol. 2019, 17, 682–690.e3. [Google Scholar] [CrossRef] [PubMed]
- Sapone, A.; Lammers, K.M.; Casolaro, V.; Cammarota, M.; Giuliano, M.T.; De Rosa, M.; Stefanile, R.; Mazzarella, G.; Tolone, C.; Russo, M.I.; et al. Divergence of gut permeability and mucosal immune gene expression in two gluten-associated conditions: Celiac disease and gluten sensitivity. BMC Med. 2011, 9, 23. [Google Scholar] [CrossRef]
- Volta, U.; Tovoli, F.; Cicola, R.; Parisi, C.; Fabbri, A.; Piscaglia, M.; Fiorini, E.; Caio, G. Serological tests in gluten sensitivity (nonceliac gluten intolerance). J. Clin. Gastroenterol. 2012, 46, 680–685. [Google Scholar] [CrossRef]
- Pozo-Rubio, T.; Olivares, M.; Nova, E.; De Palma, G.; Mujico, J.R.; Ferrer, M.D.; Marcos, A.; Sanz, Y. Immune development and intestinal microbiota in celiac disease. Clin. Dev. Immunol. 2012, 2012, 12. [Google Scholar] [CrossRef][Green Version]
- Junker, Y.; Zeissig, S.; Kim, S.J.; Barisani, D.; Wieser, H.; Leffler, D.A.; Zevallos, V.; Libermann, T.A.; Dillon, S.; Freitag, T.L.; et al. Wheat amylase trypsin inhibitors drive intestinal inflammation via activation of toll-like receptor 4. J. Exp. Med. 2012, 209, 2395–2408. [Google Scholar] [CrossRef]
- Zevallos, V.F.; Raker, V.; Tenzer, S.; Jimenez-Calvente, C.; Ashfaq-Khan, M.; Rüssel, N.; Pickert, G.; Schild, H.; Steinbrink, K.; Schuppan, D. Nutritional Wheat Amylase-Trypsin Inhibitors Promote Intestinal Inflammation via Activation of Myeloid Cells. Gastroenterology 2017, 152, 1100–1113.e12. [Google Scholar] [CrossRef] [PubMed]
- De Punder, K.; Pruimboom, L. The dietary intake of wheat and other cereal grains and their role in inflammation. Nutrients 2013, 5, 771–787. [Google Scholar] [CrossRef] [PubMed]
- Skodje, G.I.; Sarna, V.K.; Minelle, I.H.; Rolfsen, K.L.; Muir, J.G.; Gibson, P.R.; Veierod, M.B.; Henriksen, C.; Lundin, K.E.A. Fructan, Rather Than Gluten, Induces Symptoms in Patients With Self-Reported Non-Celiac Gluten Sensitivity. Gastroenterology 2018, 154, 529–539.e2. [Google Scholar] [CrossRef] [PubMed]
- Maintz, L.; Novak, N. Histamine and histamine intolerance. Am. J. Clin. Nutr. 2007, 85, 1185–1196. [Google Scholar] [CrossRef] [PubMed]
- Martin, I.S.M.; Brachero, S.; Vilar, E.G. Histamine intolerance and dietary management: A complete review. Allergol. Immunopathol. 2016, 44, 475–483. [Google Scholar] [CrossRef]
- Schnedl, W.J.; Lackner, S.; Enko, D.; Schenk, M.; Holasek, S.J.; Mangge, H. Evaluation of symptoms and symptom combinations in histamine intolerance. Intest. Res. 2019. [Google Scholar] [CrossRef] [PubMed]
- Reese, I.; Ballmer-Weber, B.; Beyer, K.; Fuchs, T.; Kleine-Tebbe, J.; Klimek, L.; Lepp, U.; Niggemann, B.; Saloga, J.; Schäfer, C. German guideline for the management of adverse reactions to ingested histamine. Allergo J. Int. 2017, 26, 72–79. [Google Scholar] [CrossRef]
- Smolinska, S.; Jutel, M.; Crameri, R.; O’mahony, L. Histamine and gut mucosal immune regulation. Allergy 2014, 69, 273–281. [Google Scholar] [CrossRef]
- Maintz, L.; Yu, C.F.; Rodríguez, E.; Baurecht, H.; Bieber, T.; Illig, T.; Weidinger, S.; Novak, N. Association of single nucleotide polymorphisms in the diamine oxidase gene with diamine oxidase serum activities. Allergy 2011, 66, 893–902. [Google Scholar] [CrossRef]
- Castells, M.; Butterfield, J. Mast cell activation syndrome and mastocytosis: Initial treatment options and long-term management. J. Allergy Clin. Immunol. Pract. 2019, 7, 1097–1106. [Google Scholar] [CrossRef]
- Valent, P.; Akin, C.; Bonadonna, P.; Hartmann, K.; Brockow, K.; Niedoszytko, M.; Nedoszytko, B.; Siebenhaar, F.; Sperr, W.R.; Elberink, J.N.O. Proposed Diagnostic Algorithm for Patients With Suspected Mast Cell Activation Syndrome. J. Allergy Clin. Immunol. Pract. 2019, 7, 1125–1133. [Google Scholar] [CrossRef] [PubMed]
- Lackner, S.; Malcher, V.; Enko, D.; Mangge, H.; Holasek, S.J.; Schnedl, W.J. Histamine-reduced diet and increase of serum diamine oxidase correlating to diet compliance in histamine intolerance. Eur. J. Clin. Nutr. 2019, 73, 102. [Google Scholar] [CrossRef] [PubMed]
- Swain, A.; Soutter, V.; Loblay, R. RPAH Elimination Diet. Handbook; Allergy unit Royal Prince Alfred Hospital; Allergy Unit, Royal Prince Alfred Hospital: Sydney, Australia, 2009. [Google Scholar]
- Malakar, S.; Gibson, P.R.; Barrett, J.S.; Muir, J.G. Naturally occurring dietary salicylates: A closer look at common Australian foods. J. Food Comp. Anal. 2017, 57, 31–39. [Google Scholar] [CrossRef]
- Bellisle, F. Effects of diet on behaviour and cognition in children. Br. J. Nutr. 2004, 92, S227–S232. [Google Scholar] [CrossRef]
- Mabin, D.C.; Sykes, A.E.; David, T.J. Controlled trial of a few foods diet in severe atopic dermatitis. Arch. Dis. Child. 1995, 73, 202–207. [Google Scholar] [CrossRef]
- Fuglsang, G.; Madsen, G.; Halken, S.; Jorgensen, S.; Ostergaard, P.A.; Osterballe, O. Adverse reactions to food additives in children with atopic symptoms. Allergy 1994, 49, 31–37. [Google Scholar] [CrossRef] [PubMed]
- Young, E.; Patel, S.; Stoneham, M.; Rona, R.; Wilkinson, J.D. The prevalence of reaction to food additives in a survey population. J. R. Coll. Phys. Lond. 1987, 21, 241–247. [Google Scholar]
- Simon, R.A. Adverse reactions to food additives. Curr. Allergy Asthma Rep. 2003, 3, 62–66. [Google Scholar] [CrossRef]
- Barrett, J.S.; Gibson, P.R. Fermentable oligosaccharides, disaccharides, monosaccharides and polyols (FODMAPs) and nonallergic food intolerance: FODMAPs or food chemicals? Therap. Adv. Gastroenterol. 2012, 5, 261–268. [Google Scholar] [CrossRef] [PubMed]
- Holton, K.F.; Taren, D.L.; Thomson, C.A.; Bennett, R.M.; Jones, K.D. The effect of dietary glutamate on fibromyalgia and irritable bowel symptoms. Clin. Exp. Rheumatol. 2012, 30, 10–17. [Google Scholar]
- Malakar, S. Bioactive food chemicals and gastrointestinal symptoms: A focus of salicylates. J. Gastroenterol. Hepatol. 2017, 32, 73–77. [Google Scholar] [CrossRef] [PubMed]
- Stevenson, D.D.; Simon, R.A.; Lumry, W.R.; Mathison, D.A. Adverse reactions to tartrazine. J. Allergy Clin. Immunol. 1986, 78, 182–191. [Google Scholar] [CrossRef]
- Simon, R.A. Update on sulfite sensitivity. Allergy 1998, 53, 78–79. [Google Scholar] [CrossRef] [PubMed]
- Tarlo, S.M.; Broder, I. Tartrazine and benzoate challenge and dietary avoidance in chronic asthma. Clin. Allergy 1982, 12, 303–312. [Google Scholar] [CrossRef] [PubMed]
- Woessner, K.M.; Simon, R.A.; Stevenson, D.D. Monosodium glutamate sensitivity in asthma. J. Allergy Clin. Immunol. 1999, 104, 305–310. [Google Scholar] [CrossRef]
- Vally, H.; Misso, N.L.A. Adverse reactions to the sulphite additives. Gastroenterol. Hepatol. Bed Bench 2012, 5, 16–23. [Google Scholar] [PubMed]
- Chassaing, B.; Koren, O.; Goodrich, J.K.; Poole, A.C.; Srinivasan, S.; Ley, R.E.; Gewirtz, A.T. Dietary emulsifiers impact the mouse gut microbiota promoting colitis and metabolic syndrome. Nature 2015, 519, 92–96. [Google Scholar] [CrossRef]
- Roberts, C.L.; Rushworth, S.L.; Richman, E.; Rhodes, J.M. Hypothesis: Increased consumption of emulsifiers as an explanation for the rising incidence of Crohn’s disease. J. Crohn’s Colitis 2013, 7, 338–341. [Google Scholar] [CrossRef]
- Pinget, G.; Tan, J.; Janac, B.; Kaakoush, N.O.; Angelatos, A.S.; O’Sullivan, J.; Koay, Y.C.; Sierro, F.; Davis, J.; Divakarla, S.K.; et al. Impact of the Food Additive Titanium Dioxide (E171) on Gut Microbiota-Host Interaction. Front. Nutr. 2019, 6. [Google Scholar] [CrossRef]
- Huerta-García, E.; Pérez-Arizti, J.A.; Márquez-Ramírez, S.G.; Delgado-Buenrostro, N.L.; Chirino, Y.I.; Iglesias, G.G.; López-Marure, R. Titanium dioxide nanoparticles induce strong oxidative stress and mitochondrial damage in glial cells. Free Radic. Biol. Med. 2014, 73, 84–94. [Google Scholar] [CrossRef]
- Raithel, M.; Baenkler, H.; Naegel, A.; Buchwald, F.; Schultis, H.; Backhaus, B.; Kimpel, S.; Koch, H.; Mach, K.; Hahn, E. Significance of salicylate intolerance in diseases. J. Physiol. Pharmacol. 2005, 56, 89–102. [Google Scholar]
- Gray, P.; Mehr, S.; Katelaris, C.H.; Wainstein, B.K.; Star, A.; Campbell, D.; Joshi, P.; Wong, M.; Frankum, B.; Keat, K. Salicylate elimination diets in children: Is food restriction supported by the evidence. Med. J. Aust. 2013, 198, 600–602. [Google Scholar] [CrossRef][Green Version]
- Cohen, S.A. The clinical consequences of sucrase-isomaltase deficiency. Mol. Cell. Pediatr. 2016, 3, 5. [Google Scholar] [CrossRef]
- Henström, M.; Diekmann, L.; Bonfiglio, F.; Hadizadeh, F.; Kuech, E.-M.; von Köckritz-Blickwede, M.; Thingholm, L.B.; Zheng, T.; Assadi, G.; Dierks, C. Functional variants in the sucrase–isomaltase gene associate with increased risk of irritable bowel syndrome. Gut 2016, 67, 263–270. [Google Scholar] [CrossRef] [PubMed]
- Puntis, J.; Zamvar, V. Congenital sucrase–isomaltase deficiency: Diagnostic challenges and response to enzyme replacement therapy. Arch. Dis. Child. 2015, 100, 869–871. [Google Scholar] [CrossRef]
- Harms, H.-K.; Bertele-Harms, R.-M.; Bruer-Kleis, D. Enzyme-substitution therapy with the yeast Saccharomyces cerevisiae in congenital sucrase-isomaltase deficiency. N. Engl. J. Med. 1987, 316, 1306–1309. [Google Scholar] [CrossRef] [PubMed]
- Yao, C.K.; Gibson, P.R.; Shepherd, S.J. Design of clinical trials evaluating dietary interventions in patients with functional gastrointestinal disorders. Am. J. Gastroenterol. 2013, 108, 748–758. [Google Scholar] [CrossRef] [PubMed]
- Tuck, C.; Vanner, S. Dietary therapies for functional bowel symptoms: Recent advances, challenges, and future directions. Neurogastroenterol. Motil. 2018, 30, e13238. [Google Scholar] [CrossRef] [PubMed]
- Yao, C.K.; Tuck, C.J.; Barrett, J.S.; Canale, K.E.; Philpott, H.L.; Gibson, P.R. Poor reproducibility of breath hydrogen testing: Implications for its application in functional bowel disorders. U. Eur. Gastroenterol. J. 2017, 5, 284–292. [Google Scholar] [CrossRef] [PubMed]
- Chumpitazi, B.; Cope, J.; Hollister, E.; Tsai, C.; McMeans, A.; Luna, R.; Versalovic, J.; Shulman, R. Randomised clinical trial: Gut microbiome biomarkers are associated with clinical response to a low FODMAP diet in children with the irritable bowel syndrome. Aliment. Pharmacol. Ther. 2015, 42, 418–427. [Google Scholar] [CrossRef]
- Bennet, S.M.; Böhn, L.; Störsrud, S.; Liljebo, T.; Collin, L.; Lindfors, P.; Törnblom, H.; Öhman, L.; Simrén, M. Multivariate modelling of faecal bacterial profiles of patients with IBS predicts responsiveness to a diet low in FODMAPs. Gut 2017, 67, 872–881. [Google Scholar] [CrossRef] [PubMed]
- Rossi, M.; Aggio, R.; Staudacher, H.M.; Lomer, M.C.; Lindsay, J.O.; Irving, P.; Probert, C.; Whelan, K. Volatile Organic Compounds in Feces Associate With Response to Dietary Intervention in Patients With Irritable Bowel Syndrome. Clin. Gastroenterol. Hepatol. 2017, 16, 385–391. [Google Scholar] [CrossRef] [PubMed]
- Bellini, M.; Rossi, A. Is a low FODMAP diet dangerous? Tech. Coloproctol. 2018, 22, 569–571. [Google Scholar] [CrossRef] [PubMed]
- Zheng, T.; Eswaran, S.; Photenhauer, A.L.; Merchant, J.L.; Chey, W.D.; D’Amato, M. Reduced efficacy of low FODMAPs diet in patients with IBS-D carrying sucrase-isomaltase (SI) hypomorphic variants. Gut 2019. [Google Scholar] [CrossRef] [PubMed]
- Tuck, C.J.; Taylor, K.M.; Gibson, P.R.; Barrett, J.S.; Muir, J.G. Increasing symptoms in irritable bowel symptoms with ingestion of galacto-oligosaccharides are mitigated by [alpha]-galactosidase treatment. Am. J. Gastroenterol. 2018, 113, 124. [Google Scholar] [CrossRef]
| Meat | Sausages of any kind, salami, air-dried and corked meat, ham, etc. |
| Fish | Dried or preserved fish, such as herring, tuna, mackerel, sardines and anchovies, seafood, fish sauces. |
| Cheese | All types of hard, soft and processed cheese |
| Vegetables | Eggplant, avocado, sauerkraut, spinach, tomatoes incl. tomato juice/ketchup |
| Drinks and liquids | Vinegar or alcohol of all kinds, mainly red wine, beers, champagne, whisky and cognac; alcohol in general reduces degradation of histamine and increases the permeability of the intestine and can therefore worsen the symptoms of histamine intolerance in general |
| Diagnostic criteria [58] | |
The diagnosis of histamine intolerance is made by a combination of the following criteria:
| |
| Symptom types [57,58] | |
| Skin | Itching, sudden reddening of the skin (flush symptoms) on the face and/or body, very rarely hives, angioedema (different to urticaria) and other exanthemas |
| Digestion | Nausea, vomiting, diarrhea, abdominal pain |
| Circulation | Tachycardia, drop in blood pressure, dizziness |
| Respiratory | Chronic nasal flow, sneezing attacks |
| Neurological | Headaches, migraines |
| Gynecological | Menstrual cramps |
| FOOD CHEMICAL | FOOD SOURCES [64,65] | |
|---|---|---|
| Natural food chemicals | Amines | Cheese, chocolate, banana, ham, fish |
| Glutamate | Tomato | |
| Salicylates | Apples, tomatoes | |
| Added food chemicals | Antioxidants | Oils, margarine |
| Benzoates | Soft drinks, cordials | |
| Colors | Confectionary, jelly | |
| Monosodium glutamate (MSG) | Chinese take-away, packaged foods | |
| Nitrates | Deli meats | |
| Propionates | Breads | |
| Sorbic acid | Processed cheese slices | |
| Sulfites | Soft drink, cordials, dried fruit |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tuck, C.J.; Biesiekierski, J.R.; Schmid-Grendelmeier, P.; Pohl, D. Food Intolerances. Nutrients 2019, 11, 1684. https://doi.org/10.3390/nu11071684
Tuck CJ, Biesiekierski JR, Schmid-Grendelmeier P, Pohl D. Food Intolerances. Nutrients. 2019; 11(7):1684. https://doi.org/10.3390/nu11071684
Chicago/Turabian StyleTuck, Caroline J, Jessica R Biesiekierski, Peter Schmid-Grendelmeier, and Daniel Pohl. 2019. "Food Intolerances" Nutrients 11, no. 7: 1684. https://doi.org/10.3390/nu11071684
APA StyleTuck, C. J., Biesiekierski, J. R., Schmid-Grendelmeier, P., & Pohl, D. (2019). Food Intolerances. Nutrients, 11(7), 1684. https://doi.org/10.3390/nu11071684





