Dietary Anthocyanins and Stroke: A Review of Pharmacokinetic and Pharmacodynamic Studies
Abstract
1. Introduction
2. Anthocyanins (ACNs): Chemical Structures and Dietary Sources
3. Pharmacokinetic Properties of ACNs
3.1. Anthocyanin Uptake
3.1.1. Oral Uptake
3.1.2. Stomach Absorption
3.1.3. Intestinal Absorption
3.2. Bioavailability
3.3. Distribution
3.3.1. Anthocyanins in Blood Circulation
3.3.2. Interactions between ACN and Blood-brain Barrier (BBB)
3.4. Metabolism
3.5. Elimination
4. Molecular Mechanisms of Action
5. Evidence for Anthocyanins’ Effects in the Prevention and Treatment of Stroke
5.1. Epidemiological Studies
5.2. In Vitro Studies
5.3. In Vivo Studies
5.3.1. Clinical Studies on Animals
5.3.2. Clinical Studies in Humans
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Ghosh, D.; Konishi, T. Anthocyanins and anthocyanin-rich extracts: Role in diabetes and eye function. Asia Pac. J. Clin. Nutr. 2007, 16, 200. [Google Scholar] [PubMed]
- Tsuda, T. Recent Progress in Anti-Obesity and Anti-Diabetes Effect of Berries. Antioxidants 2016, 5, 13. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.; Yoon, Y.; Yoon, H.; Park, H.; Song, S.; Yeum, K. Dietary Anthocyanins against Obesity and Inflammation. Nutrients 2017, 9, 1089. [Google Scholar] [CrossRef] [PubMed]
- Lin, B.; Gong, C.; Song, H.; Cui, Y. Effects of anthocyanins on the prevention and treatment of cancer. Br. J. Pharmacol. 2017, 174, 1226. [Google Scholar] [CrossRef] [PubMed]
- Silva, S.; Costa, E.; Mendes, M.; Morais, R.; Calhau, C.; Pintado, M. Antimicrobial, antiadhesive and antibiofilm activity of an ethanolic, anthocyanin-rich blueberry extract purified by solid phase extraction. J. Appl. Microbiol. 2016, 121, 693. [Google Scholar] [CrossRef]
- Reis, J.; Monteiro, V.; de Souza Gomes, R.; do Carmo, M.; da Costa, G.; Ribera, P.; Monteiro, M. Action mechanism and cardiovascular effect of anthocyanins: A systematic review of animal and human studies. J. Transl. Med. 2016, 14, 315. [Google Scholar] [CrossRef] [PubMed]
- Zhao, C.; Meng, X.; Li, Y.; Li, S.; Liu, Q.; Tang, G.; Li, H. Fruits for Prevention and Treatment of Cardiovascular Diseases. Nutrients 2017, 9, 598. [Google Scholar] [CrossRef]
- Tang, Z.; Li, M.; Zhang, X.; Hou, W. Dietary flavonoid intake and the risk of stroke: A dose-response meta-analysis of prospective cohort studies. BMJ Open 2016, 6, e008680. [Google Scholar] [CrossRef]
- He, F.; Nowson, C.; MacGregor, G. Fruit and vegetable consumption and stroke: Meta-analysis of cohort studies. Lancet 2006, 367, 320. [Google Scholar] [CrossRef]
- Castañeda-Ovando, A.; de Lourdes Pacheco-Hernández, M.; Páez-Hernández, E.; Rodríguez, J.-V.C. Chemical studies of anthocyanins: A review. Food Chem. 2009, 113, 859. [Google Scholar] [CrossRef]
- Mazza, G.; Miniati, E. Anthocyanins in Fruits, Vegetables, and Grains; CRC Press: Boca Raton, FL, USA, 1993; ISBN 0-8493-0172-6. [Google Scholar]
- Haytowitz, D.B.; Wu, X.; Bhagwat, S. USDA Database for the Flavonoid Content of Selected Foods, Release 3.3. U.S. Department of Agriculture, Agricultural Research Service. Nutrient Data Laboratory. Available online: http://www.ars.usda.gov/nutrientdata/flav (accessed on 5 May 2019).
- Sweeney, M.I.; Kalt, W.; MacKinnon, S.L.; Ashby, J.; Gottschall-Pass, K.T. Feeding Rats Diets Enriched in Lowbush Blueberries for Six Weeks Decreases Ischemia-induced Brain Damage. Nutr. Neurosci. 2002, 5, 427–431. [Google Scholar] [CrossRef] [PubMed]
- Neveu, V.; Perez-Jimenez, J.; Vos, F.; Crespy, V.; du Chaffaut, L.; Mennen, L.; Knox, C.; Eisner, R.; Cruz, J.; Wishart, D.; et al. Phenol-Explorer: An online comprehensive database on polyphenol contents in foods. Database 2010. [Google Scholar] [CrossRef] [PubMed]
- Rashid, K.; Wachira, F.; Nyabuga, J.; Wanyonyi, B.; Murilla, G.; Isaac, A. Kenyan purple tea anthocyanins ability to cross the blood brain barrier and reinforce brain antioxidant capacity in mice. Nutr. Neurosci. 2014, 17, 178. [Google Scholar] [CrossRef] [PubMed]
- Kamiloglu, S.; Serali, O.; Unal, N.; Capanoglu, E. Antioxidant activity and polyphenol composition of black mulberry (Morus nigra L.) products. J. Berry Res. 2013, 3, 41. [Google Scholar]
- Kim, M.; Kim, H.; Koh, K.; Kim, H.; Lee, Y.; Kim, Y. Identification and quantification of anthocyanin pigments in colored rice. Nutr. Res. Pract. 2008, 2, 46. [Google Scholar] [CrossRef] [PubMed]
- Lila, M.A.; Burton-Freeman, B.; Grace, M.; Kalt, W. Unraveling Anthocyanin Bioavailability for Human Health. Annu. Rev. Food Sci. Technol. 2016, 7, 375–393. [Google Scholar] [CrossRef] [PubMed]
- Ferrer-Gallego, R.; Soares, S.; Mateus, N.; Rivas-Gonzalo, J.; Escribano-Bailón, M.; de Freitas, V. New Anthocyanin-Human Salivary Protein Complexes. Langmuir 2015, 31, 8392. [Google Scholar] [CrossRef]
- Xiao, D.; Sandhu, A.; Huang, Y.; Park, E.; Edirisinghe, I.; Burton-Freeman, B. The effect of dietary factors on strawberry anthocyanins oral bioavailability. Food Funct. 2017, 8, 3970. [Google Scholar] [CrossRef]
- Kamonpatana, K.; Failla, M.L.; Kumar, P.S.; Giusti, M.M. Anthocyanin structure determines susceptibility to microbial degradation and bioavailability to the buccal mucosa. J. Agric. Food Chem. 2014, 62, 6903. [Google Scholar] [CrossRef]
- Passamonti, S.; Vrhovsek, U.; Vanzo, A.; Mattivi, F. The stomach as a site for anthocyanins absorption from food 1. FEBS Lett. 2003, 544, 210–213. [Google Scholar] [CrossRef]
- Talavéra, S.; Felgines, C.; Texier, O.; Besson, C.; Lamaison, J.-L.; Rémésy, C. Anthocyanins Are Efficiently Absorbed from the Stomach in Anesthetized Rats. J. Nutr. 2003, 133, 4178–4182. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, I.; de Freitas, V.; Reis, C.; Mateus, N. A new approach on the gastric absorption of anthocyanins. Food Funct. 2012, 3, 508. [Google Scholar] [CrossRef] [PubMed]
- Mueller, D.; Jung, K.; Winter, M.; Rogoll, D.; Melcher, R.; Richling, E. Human intervention study to investigate the intestinal accessibility and bioavailability of anthocyanins from bilberries. Food Chem. 2017, 231, 275. [Google Scholar] [CrossRef] [PubMed]
- Vanzo, A.; Terdoslavich, M.; Brandoni, A.; Torres, A.; Vrhovsek, U.; Passamonti, S. Uptake of grape anthocyanins into the rat kidney and the involvement of bilitranslocase. Mol. Nutr. Food Res. 2008, 52, 1106. [Google Scholar] [CrossRef] [PubMed]
- Smeriglio, A.; Barreca, D.; Bellocco, E.; Trombetta, D. Chemistry, Pharmacology and Health Benefits of Anthocyanins. Phyther. Res. 2016, 30, 1265–1286. [Google Scholar] [CrossRef] [PubMed]
- Passamonti, S.; Vrhovsek, U.; Mattivi, F. The Interaction of Anthocyanins with Bilitranslocase. Biochem. Biophys. Res. Commun. 2002, 296, 631. [Google Scholar] [CrossRef]
- Fernandes, I.; Faria, A.; Calhaub, C.; de Freitasa, V.; Mateusa, N. Bioavailability of anthocyanins and derivatives. J. Funct. Foods 2014, 7, 54. [Google Scholar] [CrossRef]
- Krga, I.; Milenkovic, D. Anthocyanins: From Sources and Bioavailability to Cardiovascular-Health Benefits and Molecular Mechanisms of Action. J. Agric. Food Chem. 2019, 67, 1771. [Google Scholar] [CrossRef]
- Oliveira, H.; Roma-Rodrigues, C.; Santos, A.; Veigas, B.; Brás, N.; Faria, A.; Calhau, C.; de Freitas, V.; Baptista, P.V.; Mateus, N.; et al. GLUT1 and GLUT3 involvement in anthocyanin gastric transport- Nanobased targeted approach. Sci. Rep. 2019, 9, 789. [Google Scholar] [CrossRef]
- Shahidi, F.; Peng, H. Bioaccessibility and bioavailability of phenolic compounds. J. Food Bioact. 2018, 4, 11. [Google Scholar] [CrossRef]
- Kamiloglu, S.; Capanoglu, E.; Grootaert, C.; Van Camp, J. Anthocyanin Absorption and Metabolism by Human Intestinal Caco-2 Cells-A Review. Int. J. Mol. Sci. 2015, 16, 21555–21574. [Google Scholar] [CrossRef] [PubMed]
- Zou, T.; Feng, D.; Song, G.; Li, H.; Tang, H.; Ling, W. The Role of Sodium-Dependent Glucose Transporter 1 and Glucose Transporter 2 in the Absorption of Cyanidin-3-O-β-Glucoside in Caco-2 Cells. Nutrients 2014, 6, 4165. [Google Scholar] [CrossRef] [PubMed]
- Passamonti, S.; Terdoslavich, M.; Franca, R.; Vanzo, A.; Tramer, F.; Braidot, E.; Petrussa, E.; Vianello, A. Bioavailability of flavonoids: A review of their membrane transport and the function of bilitranslocase in animal and plant organisms. Curr. Drug Metab. 2009, 10, 369. [Google Scholar] [CrossRef] [PubMed]
- Figueira, I.; Menezes, R.; Macedo, D.; Costa, I.; Dos Santos, C. Polyphenols Beyond Barriers: A Glimpse into the Brain. Curr. Neuropharmacol. 2017, 15, 562. [Google Scholar] [CrossRef] [PubMed]
- Fang, J. Bioavailability of anthocyanins. Drug Metab. Rev. 2014, 46, 508–520. [Google Scholar] [CrossRef]
- Zhang, T.; Lv, C.; Chen, L.; Bai, G.; Zhao, G.; Xu, C. Encapsulation of anthocyanin molecules within a ferritin nanocage increases their stability and cell uptake efficiency. Food Res. Int. 2014, 62, 183. [Google Scholar] [CrossRef]
- Kuntz, S.; Asseburg, H.; Dold, S.; Römpp, A.; Fröhling, B.; Kunz, C.; Rudloff, S. Inhibition of low-grade inflammation by anthocyanins from grape extract in an in vitro epithelial-endothelial co-culture model. Food Funct. 2015, 6, 1136–1149. [Google Scholar] [CrossRef] [PubMed]
- Steinert, R.E.; Ditscheid, B.; Netzel, M.; Jahreis, G. Absorption of black currant anthocyanins by monolayers of human intestinal epithelial Caco-2 cells mounted in ussing type chambers. J. Agric. Food Chem. 2008, 59, 4995. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Hassan, Y.; Renaud, J.; Liu, R.; Yang, C.; Sun, Y.; Tsao, R. Bioaccessibility, bioavailability, and anti-inflammatory effects of anthocyanins from purple root vegetables using mono- and co-culture cell models. Mol. Nutr. Food Res. 2017, 61, 1600928. [Google Scholar] [CrossRef]
- Faria, A.; Pestana, D.; Azevedo, J.; Martel, F.; de Freitas, V.; Azevedo, I.; Mateus, N.; Calhau, C. Absorption of anthocyanins through intestinal epithelial cells—Putative involvement of GLUT2. Mol. Nutr. Food Res. 2009, 53, 1430–1437. [Google Scholar] [CrossRef]
- Mueller, D.; Jung, K.; Winter, M.; Rogoll, D.; Melcher, R.; Kulozik, U.; Schwarz, K.; Richling, E. Encapsulation of anthocyanins from bilberries–Effects on bioavailability and intestinal accessibility in humans. Food Chem. 2018, 248, 217–224. [Google Scholar] [CrossRef] [PubMed]
- González-Barrio, R.; Borges, G.; Mullen, W.; Crozier, A. Bioavailability of anthocyanins and ellagitannins following consumption of raspberries by healthy humans and subjects with an ileostomy. J. Agric. Food Chem. 2010, 58, 3933. [Google Scholar] [CrossRef] [PubMed]
- Talavéra, S.; Felgines, C.; Texier, O.; Besson, C.; Manach, C.; Lamaison, J.; Rémésy, C. Anthocyanins are efficiently absorbed from the small intestine in rats. J. Nutr. 2004, 134, 2275. [Google Scholar] [CrossRef] [PubMed]
- Milbury, P.; Kalt, W. Xenobiotic metabolism and berry flavonoid transport across the blood–brain barrier. J. Agric. Food Chem. 2010, 58, 3950. [Google Scholar] [CrossRef] [PubMed]
- Esposito, D.; Damsud, T.; Wilson, M.; Grace, M.; Strauch, R.; Li, X.; Lila, M.; Komarnytsky, S.B. Currant Anthocyanins Attenuate Weight Gain and Improve Glucose Metabolism in Diet-Induced Obese Mice with Intact, but Not Disrupted Gut Microbiome. J. Agric. Food Chem. 2015, 63, 6172. [Google Scholar] [CrossRef] [PubMed]
- Hanske, L.; Engst, W.; Loh, G.; Sczesny, S.; Blaut, M.; Braune, A. Contribution of gut bacteria to the metabolism of cyanidin 3-glucoside in human microbiota-associated rats. Br. J. Nutr. 2013, 109, 1433. [Google Scholar] [CrossRef]
- Mayta-Apaza, A.C.; Pottgen, E.; De Bodt, J.; Papp, N.; Marasini, D.; Howard, L.; Abranko, L.; Van de Wiele, T.; Lee, S.O.; Carbonero, F. Impact of tart cherries polyphenols on the human gut microbiota and phenolic metabolites in vitro and in vivo. J. Nutr. Biochem. 2018, 59, 160. [Google Scholar] [CrossRef]
- Bresciani, L.; Angelino, D.; Vivas, E.; Kerby, R.; Garcia-Viguera, C.; Del Rio, D.; Rey, F.; Mena, P. Differential catabolism of an anthocyanin-rich elderberry extract by three gut microbiota bacterial species. J. Agric. Food Chem. 2019. [Google Scholar] [CrossRef]
- He, J.; Magnuson, B.; Giusti, M. Analysis of anthocyanins in rat intestinal contents: Impact of anthocyanin chemical structure on fecal excretion. J. Agric. Food Chem. 2005, 53, 2859. [Google Scholar] [CrossRef]
- Chen, Y.; Li, Q.; Zhao, T.; Zhang, Z.; Mao, G.; Feng, W.; Wu, X.; Yang, L. Biotransformation and metabolism of three mulberry anthocyanin monomers by rat gut microflora. Food Chem. 2017, 237, 887. [Google Scholar] [CrossRef]
- Flores, G.; del Castillo, M.; Costabile, A.; Klee, A.; Guergoletto, K.; Gibson, G. In vitro fermentation of anthocyanins encapsulated with cyclodextrins: Release, metabolism and influence on gut microbiota growth. J. Funct. Food 2015, 16, 57. [Google Scholar] [CrossRef]
- Aura, A.; Martin-Lopez, P.; O’Leary, K.; Williamson, G.; Oksman-Caldentey, K.; Poutanen, K.; Santos-Buelga, C. In vitro metabolism of anthocyanins by human gut microflora. Eur. J. Nutr. 2005, 44, 133. [Google Scholar] [CrossRef] [PubMed]
- Ávila, M.; Hidalgo, M.; Sánchez-Moreno, C.; Pelaez, C.; Requena, T.; de Pascual-Teresa, S. Bioconversion of anthocyanin glycosides by Bifidobacteria and Lactobacillus. Food Res. Int. 2009, 42, 1453. [Google Scholar] [CrossRef]
- Williamson, G.; Clifford, M. Colonic metabolites of berry polyphenols: The missing link to biological activity? Br. J. Nutr. 2010, 104, S48. [Google Scholar] [CrossRef] [PubMed]
- Finkel, R.; Clark, M.A.; Cubeddu, L.X. Lippincott Illustrated Reviews: Pharmacology, 4th ed.; Lippincott Williams & Wilkins (LWW): Philadelphia, PA, USA, 2009. [Google Scholar]
- Galanakis, C. Nutraceutical and Functional Food Components 1st Edition Effects of Innovative Processing Techniques. In Phytochemistry Reviews; Galanakis, C., Ed.; Academic Press: London, UK, 2017; p. 5. ISBN 9780128096505. [Google Scholar]
- Fernandes, I.; Faria, A.; de Freitas, V.; Calhau, C.; Mateus, N. Multiple-approach studies to assess anthocyanin bioavailability. Phytochem. Rev. 2015, 14, 899–919. [Google Scholar] [CrossRef]
- Kalt, W.; Liu, Y.; McDonald, J.; Vinqvist-Tymchuk, M.; Fillmore, S. Anthocyanin metabolites are abundant and persistent in human urine. J. Agric. Food Chem. 2014, 62, 3926. [Google Scholar] [CrossRef] [PubMed]
- Czank, C.; Cassidy, A.; Zhang, Q.; Morrison, D.; Preston, T.; Kroon, P.; Botting, N.; Kay, C. Human metabolism and elimination of the anthocyanin, cyanidin-3-glucoside: A 13C-tracer study. Am. J. Clin. Nutr. 2013, 97, 995. [Google Scholar] [CrossRef] [PubMed]
- Wallace, T. Anthocyanins in cardiovascular disease. Adv. Nutr. 2011, 2, 1. [Google Scholar] [CrossRef]
- Lucioli, S. Anthocyanins: Mechanism of action and therapeutic efficacy (Chapter 3). In Medicinal Plants as Antioxidant Agents: Understanding Their Mechanism of Action and Therapeutic Efficacy; Capasso, A., Ed.; Research Signpost: Kerala, India, 2012; pp. 27–57. ISBN 97881-308-0509-2. [Google Scholar]
- Kalt, W.; Blumberg, J.; McDonald, J.; Vinqvist-Tymchuk, M.; Fillmore, S.; Graf, B.; O’Leary, J.; Milbury, P. Identification of anthocyanins in the liver, eye, and brain of blueberry-fed pigs. J. Agric. Food Chem. 2008, 56, 705. [Google Scholar] [CrossRef]
- Matsumoto, H.; Nakamura, Y.; Iida, H.; Ito, K.; Ohguro, H. Comparative assessment of distribution of blackcurrant anthocyanins in rabbit and rat ocular tissues. Exp. Eye Res. 2006, 83, 348. [Google Scholar] [CrossRef]
- Ichiyanagi, T.; Shida, Y.; Rahman, M.; Hatano, Y.; Konishi, T. Bioavailability and tissue distribution of anthocyanins in bilberry (Vaccinium myrtillus L.) extract in rats. J. Agric. Food Chem. 2006, 54, 6578. [Google Scholar] [CrossRef] [PubMed]
- El Mohsen, M.; Marks, J.; Kuhnle, G.; Moore, K.; Debnam, E.; Kaila Srai, S.; Rice-Evans, C.; Spencer, J. Absorption, tissue distribution and excretion of pelargonidin and its metabolites following oral administration to rats. Br. J. Nutr. 2006, 95, 51. [Google Scholar] [CrossRef] [PubMed]
- De Ferrars, R.; Czank, C.; Zhang, Q.; Botting, N.; Kroon, P.; Cassidy, A.; Kay, C. The pharmacokinetics of anthocyanins and their metabolites in humans. Br. J. Pharmacol. 2014, 171, 3268. [Google Scholar] [CrossRef] [PubMed]
- Faria, A.; Meireles, M.; Fernandes, I.; Santos-Buelga, C.; Gonzalez-Manzano, S.; Dueñas, M.; de Freitas, V.; Mateus, N.; Calhau, C. Flavonoid metabolites transport across a human BBB model. Food Chem. 2014, 149, 190. [Google Scholar] [CrossRef] [PubMed]
- Helms, H.C.; Abbott, N.J.; Burek, M.; Cecchelli, R.; Couraud, P.-O.; Deli, M.A.; Förster, C.; Galla, H.J.; Romero, I.A.; Shusta, E.V.; et al. In vitro models of the blood–brain barrier: An overview of commonly used brain endothelial cell culture models and guidelines for their use. J. Cereb. Blood Flow Metab. 2016, 36, 862–890. [Google Scholar] [CrossRef] [PubMed]
- Youdim, K.; Dobbie, M.; Kuhnle, G.; Proteggente, A.; Abbott, N.; Rice-Evans, C. Interaction between flavonoids and the blood-brain barrier: In vitro studies. J. Neurochem. 2003, 85, 180. [Google Scholar] [CrossRef] [PubMed]
- Steiner, O.; Coisne, C.; Engelhardt, B.; Lyck, R. Comparison of Immortalized bEnd5 and Primary Mouse Brain Microvascular Endothelial Cells as in vitro Blood–Brain Barrier Models for the Study of T Cell Extravasation. J. Cereb. Blood Flow Metab. 2011, 31, 315–327. [Google Scholar] [CrossRef]
- Faria, A.; Pestana, D.; Teixeira, D.; Azevedo, J.; De Freitas, V.; Mateus, N.; Calhau, C. Flavonoid transport across RBE4 cells: A blood-brain barrier model. Cell. Mol. Biol. Lett. 2010, 15, 234. [Google Scholar] [CrossRef]
- Wilhelm, I.; Fazakas, C.; Krizbai, I.A. In vitro models of the blood-brain barrier. Acta Neurobiol. Exp. 2011, 71, 113–128. [Google Scholar]
- Maki, T.; Hayakawa, K.; Pham, L.-D.D.; Xing, C.; Lo, E.H.; Arai, K. Biphasic Mechanisms of Neurovascular Unit Injury and Protection in CNS Diseases. CNS Neurol. Disord. Drug Targets 2013, 12, 302–315. [Google Scholar] [CrossRef]
- Maoz, B.M.; Herland, A.; FitzGerald, E.A.; Grevesse, T.; Vidoudez, C.; Pacheco, A.R.; Sheehy, S.P.; Park, T.-E.; Dauth, S.; Mannix, R.; et al. A linked organ-on-chip model of the human neurovascular unit reveals the metabolic coupling of endothelial and neuronal cells. Nat. Biotechnol. 2018, 36, 865–874. [Google Scholar] [CrossRef] [PubMed]
- Wevers, N.R.; Kasi, D.G.; Gray, T.; Wilschut, K.J.; Smith, B.; van Vught, R.; Shimizu, F.; Sano, Y.; Kanda, T.; Marsh, G.; et al. A perfused human blood–brain barrier on-a-chip for high-throughput assessment of barrier function and antibody transport. Fluids Barriers CNS 2018, 15, 23. [Google Scholar] [CrossRef] [PubMed]
- Vatine, G.D.; Barrile, R.; Workman, M.J.; Sances, S.; Barriga, B.K.; Rahnama, M.; Barthakur, S.; Kasendra, M.; Lucchesi, C.; Kerns, J.; et al. Human iPSC-Derived Blood-Brain Barrier Chips Enable Disease Modeling and Personalized Medicine Applications. Cell Stem Cell 2019, 24, 995–1005. [Google Scholar] [CrossRef] [PubMed]
- Turner, R.J.; Sharp, F.R. Implications of MMP9 for Blood Brain Barrier Disruption and Hemorrhagic Transformation Following Ischemic Stroke. Front. Cell. Neurosci. 2016, 10, 56. [Google Scholar] [CrossRef] [PubMed]
- Rosenberg, G.A.; Yang, Y. Vasogenic edema due to tight junction disruption by matrix metalloproteinases in cerebral ischemia. Neurosurg. Focus 2007, 22, 1–9. [Google Scholar] [CrossRef]
- Lapi, D.; Chiurazzi, M.; Di Maro, M.; Mastantuono, T.; Battiloro, L.; Sabatino, L.; Ricci, S.; Di Carlo, A.; Starita, N.; Guida, B.; et al. Malvidin’s Effects on Rat Pial Microvascular Permeability Changes Due to Hypoperfusion and Reperfusion Injury. Front. Cell. Neurosci. 2016, 10, 153. [Google Scholar] [CrossRef]
- Pan, Z.; Cui, M.; Dai, G.; Yuan, T.; Li, Y.; Ji, T.; Pan, Y. Protective Effect of Anthocyanin on Neurovascular Unit in Cerebral Ischemia/Reperfusion Injury in Rats. Front. Neurosci. 2018, 12, 947. [Google Scholar] [CrossRef]
- Patching, S. Glucose Transporters at the Blood-Brain Barrier: Function, Regulation and Gateways for Drug Delivery. Mol. Neurobiol. 2017, 54, 1046. [Google Scholar] [CrossRef]
- Dreiseitel, A.; Oosterhui, B.; Vukman, K.; Schreier, P.; Oehme, A.; Locher, S.; Hajak, G.; Sand, P. Berry anthocyanins and anthocyanidins exhibit distinct affinities for the efflux transporters BCRP and MDR1. Br. J. Pharmacol. 2009, 158, 1942. [Google Scholar] [CrossRef]
- Janle, E.; Lila, M.; Grannan, M.; Wood, L.; Higgins, A.; Yousef, G.; Rogers, R.; Kim, H.; Jackson, G.; Weaver, C. Method for evaluating the potential of C labeled plant polyphenols to cross the blood-brain barrier using accelerator mass spectrometry. Nucl. Instrum. Methods Phys. Res. B 2010, 268, 1313. [Google Scholar] [CrossRef]
- Andres-Lacueva, C.; Shukitt-Hale, B.; Galli, R.; Jauregui, O.; Lamuela-Raventos, R.; Joseph, J. Anthocyanins in aged blueberry-fed rats are found centrally and may enhance memory. Nutr. Neurosci. 2005, 8, 111. [Google Scholar] [CrossRef] [PubMed]
- Chen, T.Y.; Kritchevsky, J.; Hargett, K.; Feller, K.; Klobusnik, R.; Song, B.J.; Cooper, B.; Jouni, Z.; Ferruzzi, M.G.; Janle, E.M. Plasma Bioavailability and Regional Brain Distribution of Polyphenols from Apple/Grape Seed and Bilberry Extracts in a Young Swine Model. Mol. Nutr. Food Res. 2015, 59, 2432. [Google Scholar] [CrossRef] [PubMed]
- Zimman, A.; Waterhouse, A.L. Enzymatic synthesis of [3′-O-methyl-3H]malvidin-3-glucoside from petunidin-3-glucoside. J. Agric. Food Chem. 2002, 50, 2429. [Google Scholar] [CrossRef] [PubMed]
- Sandoval-Ramírez, B.; Catalán, Ú.; Fernández-Castillejo, S.; Rubió, L.; Macià, A.; Solà, R. Anthocyanin Tissue Bioavailability in Animals: Possible Implications for Human Health. A Systematic Review. J. Agric. Food Chem. 2018, 66, 11531. [Google Scholar] [CrossRef] [PubMed]
- Talavéra, S.; Felgines, C.; Texier, O.; Besson, C.; Gil-Izquierdo, A.; Lamaison, J.-L.; Rémésy, C. Anthocyanin Metabolism in Rats and Their Distribution to Digestive Area, Kidney, and Brain. J. Agric. Food Chem. 2005, 53, 3902–3908. [Google Scholar] [CrossRef]
- Kirakosyan, A.; Seymour, E.M.; Wolforth, J.; McNish, R.; Kaufman, P.; Bolling, S.F. Tissue Bioavailability of Anthocyanins from Whole Tart Cherry in Healthy Rats. Food Chem. 2015, 171, 26. [Google Scholar] [CrossRef] [PubMed]
- Passamonti, S.; Vrhovsek, U.; Vanzo, A.; Mattivi, F. Fast Access of Some Grape Pigments to the Brain. J. Agric. Food Chem. 2005, 53, 7029–7034. [Google Scholar] [CrossRef]
- Fornasaro, S.; Ziberna, L.; Gasperotti, M.; Tramer, F.; Vrhovšek, U.; Mattivi, F.; Passamonti, S. Determination of cyanidin 3-glucoside in rat brain, liver and kidneys by UPLC/MS-MS and its application to a short-term pharmacokinetic study. Sci. Rep. 2016, 6, 22815. [Google Scholar] [CrossRef]
- Gasperotti, M.; Passamonti, S.; Tramer, F.; Masuero, D.; Guella, G.; Mattivi, F.; Vrhovsek, U. Fate of microbial metabolites of dietary polyphenols in rats: Is the brain their target destination? ACS Chem. Neurosci. 2015, 6, 1341. [Google Scholar] [CrossRef]
- Sakakibara, H.; Ogawa, T.; Koyanagi, A.; Kobayashi, S.; Goda, T.; Kumazawa, S.; Kobayashi, H.; Shimoi, K. Distribution and excretion of bilberry anthocyanins in mice. J. Agric. Food Chem. 2009, 57, 7681. [Google Scholar] [CrossRef]
- Del Bò, C.; Ciappellano, S.; Klimis-Zacas, D.; Martini, D.; Gardana, C.; Riso, P.; Porrini, M. Anthocyanin absorption, metabolism, and distribution from a wild blueberry-enriched diet (Vaccinium angustifolium) is affected by diet duration in the Sprague-Dawley rat. J. Agric. Food Chem. 2010, 58, 2491. [Google Scholar] [PubMed]
- Wu, B.; Kulkarni, K.; Basu, S.; Zhang, S.; Hu, M. First-pass metabolism via UDP-glucuronosyltransferase: A barrier to oral bioavailability of phenolics. J. Pharm. Sci. 2011, 100, 3655. [Google Scholar] [CrossRef] [PubMed]
- Young, J.; Nielsen, S.; Haraldsdottir, J.; Daneshva, B.; Lauridsen, S.T.; Knuthsen, P.; Crozier, A.; Sandstrom, B.; Dragsted, L. Effect of fruit juice intake on urinary quercetin excretion and biomarkers of antioxidative status. Am. J. Clin. Nutr. 1999, 69, 87. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Jiménez, J.; Hubert, J.; Hooper, L.; Cassidy, A.; Manach, C.; Williamson, G.; Scalbert, A. Urinary metabolites as biomarkers of polyphenol intake in humans: A systematic review. Am. J. Clin. Nutr. 2010, 92, 801. [Google Scholar] [CrossRef] [PubMed]
- Elias, M.; Lunazzi, G.; Passamonti, S.; Gazzin, B.; Miccio, M.; Stanta, G.; Sottocasa, G.; Tiribelli, C.; Elias, M.M.; Lunazzi, G.C.; et al. Bilitranslocase localization and function in basolateral plasma membrane of renal proximal tubule in rat. Am. J. Physiol. 1990, 259, F559. [Google Scholar] [CrossRef] [PubMed]
- Milenkovic, D.; Deval, C.; Gouranton, E.; Landrier, J.; Scalbert, A.; Morand, C.; Mazur, A. Modulation of miRNA expression by dietary polyphenols in apoE deficient mice: A new mechanism of the action of polyphenols. PLoS ONE 2012, 7, e29837. [Google Scholar] [CrossRef]
- Krga, I.; Tamaian, R.; Mercier, S.; Boby, C.; Monfoulet, L.; Glibetic, M.; Morand, C.; Milenkovic, D. Anthocyanins and their gut metabolites attenuate monocyte adhesion and transendothelial migration through nutrigenomic mechanisms regulating endothelial cell permeability. Free Radic. Biol. Med. 2018, 124, 364. [Google Scholar] [CrossRef]
- Di Meo, S.; Reed, T.; Venditti, P.; Victor, V. Role of ROS and RNS sources in physiological and pathological conditions. Oxid. Med. Cell. Longev. 2016, 2016, 1245049. [Google Scholar] [CrossRef]
- Valko, M.; Leibfritz, D.; Moncol, J.; Cronin, M.; Mazur, M.; Telser, J. Free radicals and antioxidants in normal physiological functions and human disease. Int. J. Biochem. Cell. Biol. 2006, 39, 44. [Google Scholar] [CrossRef]
- Sies, H.; Berndt, C.; Jones, D. Oxidative stress. Annu. Rev. Biochem. 2017, 86, 715. [Google Scholar] [CrossRef]
- Sies, H.; Jones, D. Oxidative Stress; Elsevier: Amsterdam, The Netherlands, 2007; Volume 3, p. 45. [Google Scholar]
- Liguori, I.; Russo, G.; Curcio, F.; Bulli, G.; Aran, L.; Della-Morte, D.; Gargiulo, G.; Testa, G.; Cacciatore, F.; Bonaduce, D.; et al. Oxidative stress, aging, and diseases. Clin. Interv. Aging 2018, 13, 757. [Google Scholar] [CrossRef] [PubMed]
- Cai, H.; Harrison, D. Endothelial dysfunction in cardiovascular diseases: The role of oxidant stress. Circ. Res. 2000, 87, 840. [Google Scholar] [CrossRef] [PubMed]
- Higashi, Y.; Maruhashi, T.; Noma, K.; Kihara, Y. Oxidative stress and endothelial dysfunction: Clinical evidence and therapeutic implications. Trends Cardiovasc. Med. 2014, 24, 165. [Google Scholar] [CrossRef] [PubMed]
- Incalza, M.; D’Oria, R.; Natalicchio, A.; Perrini, S.; Laviola, L.; Giorgino, F. Oxidative stress and reactive oxygen species in endothelial dysfunction associated with cardiovascular and metabolic diseases. Vasc. Pharmacol. 2018, 100, 1. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.; Chen, G.; Liao, D.; Zhu, Y.; Xue, X. Effects of berries consumption on cardiovascular risk factors: A meta-analysis with trial sequential analysis of randomized controlled trials. Sci. Rep. 2016, 6, 23625. [Google Scholar] [CrossRef]
- Wallace, T.; Slavin, M.; Frankenfeld, C. Systematic review of anthocyanins and markers of cardiovascular disease. Nutrients 2016, 8, 32. [Google Scholar] [CrossRef] [PubMed]
- Fairlie-Jones, L.; Davison, K.; Fromentin, E.; Hill, A. The effect of anthocyanin-rich foods or extracts on vascular function in adults: A systematic review and meta-analysis of randomised controlled trials. Nutrients 2017, 9, 908. [Google Scholar] [CrossRef] [PubMed]
- Ma, Z.; Hong, Q.; Wang, Y.; Tan, H.; Xiao, C.; Liang, Q.; Zhang, B.; Gao, Y. Ferulic acid protects human umbilical vein endothelial cells from radiation induced oxidative stress by phosphatidylinositol 3-kinase and extracellular signal-regulated kinase pathway. Biol. Pharm. Bull. 2010, 33, 29. [Google Scholar] [CrossRef]
- Amin, H.; Czank, C.; Raheem, S.; Zhang, Q.; Botting, N.; Cassidy, A.; Kay, C. Anthocyanins and their physiologically relevant metabolites alter the expression of IL-6 and VCAM-1 in CD40L and oxidized LDL challenged vascular endothelial cells. Mol. Nutr. Food Res. 2015, 59, 1095. [Google Scholar] [CrossRef]
- Krga, I.; Monfoulet, L.; Konic-Ristic, A.; Mercier, S.; Glibetic, M.; Morand, C.; Milenkovic, D. Anthocyanins and their gut metabolites reduce the adhesion of monocyte to TNFα-activated endothelial cells at physiologically relevant concentrations. Arch. Biochem. Biophys. 2016, 599, 51. [Google Scholar] [CrossRef]
- Goszcz, K.; Deakin, S.; Duthie, G.; Stewart, D.; Megson, I. Bioavailable concentrations of delphinidine and its metabolite, gallic acid, induce antioxidant protection associated with increased intracellular glutathione in cultured endothelial cells. Oxid. Med. Cell. Longev. 2017. [Google Scholar] [CrossRef] [PubMed]
- Warner, E.; Smith, M.; Zhang, Q.; Raheem, K.; O’Hagan, D.; O’Connell, M.; Kay, C. Signatures of anthocyanin metabolites identified in humans inhibit biomarkers of vascular inflammation in human endothelial cells. Mol. Nutr. Food Res. 2017, 61, 9. [Google Scholar] [CrossRef] [PubMed]
- Tang, J.; Vissers, M.; Anderson, R.; Sreebhavan, S.; Bozonet, S.; Scheepens, A.; Melton, L. Bioavailable blueberry-derived phenolic acids at physiological concentrations enhance Nrf2-regulated antioxidant responses in human vascular endothelial cells. Mol. Nutr. Food Res. 2018, 62, 1700647. [Google Scholar] [CrossRef] [PubMed]
- Warner, E.; Rodriguez-Ramiro, I.; O’Connell, M.; Kay, C. Cardiovascular mechanisms of action of anthocyanins may be associated with the impact of microbial metabolites on heme oxygenase-1 in vascular smooth muscle cells. Molecules 2018, 23, 898. [Google Scholar] [CrossRef] [PubMed]
- Tsuda, T.; Kato, Y.; Osawa, T. Mechanism for peroxynitrite scavenging activity by anthocyanins. FEBS Lett. 2000, 484, 207. [Google Scholar] [CrossRef]
- Rahman, M.; Ichiyanagi, T.; Komiyama, T.; Hatano, Y.; Konishi, T. Superoxide radical- and peroxynitrite-scavenging activity of anthocyanins; structure-activity relationshjip and their synergism. Free Radic. Res. 2006, 40, 993. [Google Scholar] [CrossRef] [PubMed]
- Dimitrić Marković, J.; Pejin, B.; Milenković, D.; Amić, D.; Begović, N.; Mojović, M.; Marković, Z. Antiradical activity of delphinidin, pelargonidin, and malvidin towards hydroxyl and nitric oxide radicals: The energy requirements calculations as a prediction of the possible antiradical mechanisms. Food Chem. 2017, 218, 440. [Google Scholar] [CrossRef]
- Liu, L.; Lee, H.; Shih, Y.; Chyau, C.; Wang, C. Mulberry anthocyanin extract inhibit LDL oxidation and macrophage-derived foam cell formation induced by oxidative LDL. J. Food Sci. 2008, 73, 113. [Google Scholar] [CrossRef]
- Kamiyama, M.; Kishimoto, Y.; Tani, M.; Andoh, K.; Utsunomiya, K.; Kondo, K. Inhibition of low-density lipoprotein oxidation by Nagano purple grape (Vitis viniferas Vitis labrusca). J. Nutr. Sci. Vitaminol. 2009, 55, 471. [Google Scholar] [CrossRef][Green Version]
- Aboonabi, A.; Singh, I. Chemopreventive role of anthocyanins in atherosclerosis via activation of Nrf2-ARE as an indicator and modulator redox. Biomed. Pharmacother. 2015, 72, 30. [Google Scholar] [CrossRef]
- Ma, Q. Role of Nrf2 in oxidative stress and toxicity. Annu. Rev. Pharmacol. Toxicol. 2013, 53, 401. [Google Scholar] [CrossRef] [PubMed]
- Zhang, B.; Kang, M.; Xie, Q.; Xu, B.; Sun, C.; Chen, K.; Wu, Y. Anthocyanins from Chinese bayberry extract protect β cells from oxidative stress-mediated injury via HO-1 upregulation. J. Agric. Food Chem. 2011, 59, 537. [Google Scholar] [CrossRef] [PubMed]
- Poynten, A.; Gan, S.; Kriketos, A.; Campbell, L.; Chisholm, D. Circulating fatty acids, non-high density lipoproteins cholesterol, and insulin-infused fat oxidation acutely influence whole body insulin sensitivity in nondiabetic men. J. Clin. Endocrinol. Metab. 2005, 90, 1035. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, A.; Gao, L.; Thakur, A.; Siu, P.; Lai, C. Role of free fatty acids in endothelial dysfunction. J. Biomed. Sci. 2017, 24, 50. [Google Scholar] [CrossRef]
- Fratantonio, D.; Speciale, A.; Ferrari, D.; Cristani, M.; Saija, A.; Cimino, F. Palmitate-induced endothelial dysfunction is attenuated by cyanidin-3-O-glucoside through modulation of Nrf2/Bach1 and NF-kB pathways. Toxicol. Lett. 2015, 239, 152. [Google Scholar] [CrossRef]
- Fratantonio, D.; Cimino, F.; Molonia, M.; Ferrari, D.; Saija, A.; Virgili, F.; Speciale, A. Cyanidin-3-O-glucoside ameliorates palmitate-induced insulin resistance by modulating IRS-a phosphorylation and release of endothelial derived vasoactive factors. Biochim. Biophys. Acta Mol. Cell. Biol., Lipids 2017, 1862, 351. [Google Scholar] [CrossRef]
- Bryan, H.; Olayanju, A.; Goldring, C.; Kevin Park, B. The Nrf2 cell defence pathway: Keap1-dependent and -independent mechanisms of regulation. Biochem. Pharmacol. 2013, 85, 705. [Google Scholar] [CrossRef]
- Cimino, F.; Speciale, A.; Anwar, S.; Canali, R.; Ricciardi, E.; Virgili, F.; Trombetta, D.; Saija, A. Anthocyanins protect human endothelial cells from mild hyperoxia damage through modulation of Nrf2 pathway. Genes Nutr. 2013, 8, 391. [Google Scholar] [CrossRef]
- Li, D.; Mehta, J. Oxidized LDL, a critical factor in atherogenesis. Cardiovasc. Res. 2005, 68, 353. [Google Scholar] [CrossRef]
- Xie, X.; Zhao, R.; Shen, G. Influence of delphinidin-3-glucoside on oxidized low-density lipoprotein-induced oxidative stress and apoptosis in cultured endothelial cells. J. Agric. Food Chem. 2012, 60, 1850. [Google Scholar] [CrossRef]
- Jin, X.; Chen, M.; Yi, L.; Chang, H.; Zhang, T.; Wang, L.; Ma, W.; Peng, X.; Zhou, Y.; Mi, M. Delphinidin-3-glucoside protects human umbilical vein endothelial cells against oxidized low-density lipoprotein-induced injury by autophagy upregulation via the AMPK/SIRT1 signaling pathway. Mol. Nutr. Food Res. 2014, 58, 1941. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Ikeda, K.; Yamori, Y. Upregulation of endothelial nitric oxide synthase by cyanidin-3-glucoside, a typical anthocyanin pigment. Hypertension 2004, 44, 217. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Ikeda, K.; Yamori, Y. Cyanidin-3-glucoside regulates phosphorylation of endothelial nitric oxide synthase. FEBS Lett. 2004, 574, 176. [Google Scholar] [CrossRef] [PubMed]
- Martin, S.; Giannone, G.; Andriantsitohaina, R.; Martinez, M. Delphinidin, an active compound of red wine, inhibits endothelial cell apoptosis via nitric oxide pathway and regulation of calcium homeostasis. Br. J. Pharmacol. 2003, 139, 1095. [Google Scholar] [CrossRef] [PubMed]
- Paixão, J.; Dinis, T.; Almeida, L. Malvidin-3-glucoside protects endothelial cells up-regulating endothelial NO synthase and inhibiting peroxynitrite-induced NF-kB activation. Chem. Biol. Interact. 2012, 199, 192. [Google Scholar] [CrossRef] [PubMed]
- Lazzè, M.; Pizzala, R.; Perucca, P.; Cazzalini, O.; Savio, M.; Forti, L.; Vannini, V.; Bianchi, L. Anthocyanidins decrease endothelin-1 production and increase endothelial nitric oxide synthase in human endothelial cells. Mol. Nutr. Food Res. 2006, 50, 44. [Google Scholar] [CrossRef] [PubMed]
- Horie, K.; Nanashima, N.; Maeda, H. Phytoestrogenic effects of blckcurrant anthocyanins increased endothelial nitric oxide synthase (eNOS) expression in human endothelial cells and ovariectomized rats. Molecules 2019, 24, 1259. [Google Scholar] [CrossRef] [PubMed]
- Liu, T.; Zhang, L.; Joo, D.; Sun, S. NF-kB signaling in inflammation. Signal. Transduct. Target Ther. 2017, 2, 17023. [Google Scholar] [CrossRef]
- Chen, C.; Yi, L.; Jin, X.; Zhang, T.; Fu, Y.; Zhu, J.; Mi, M.; Zhang, Q.; Ling, W.; Yu, B. Inhibitory effect of delphinidin on monocyte-endothelial cell adhesion induced by oxidized low-density lipoprotein via ROS/p38MAPK/NF-kB pathway. Cell Biochem. Biophys. 2011, 61, 337. [Google Scholar] [CrossRef]
- Fung, T.; Chiuve, S.; McCullough, M.; Rexrode, K.; Logroscino, G.; Hu, F. Adherence to a DASH-style diet and risk of coronary heart disease and stroke in women. Arch. Intern. Med. 2008, 168, 713. [Google Scholar] [CrossRef]
- Kimble, R.; Keane, K.; Lodge, J.K.; Howatson, G. Dietary intake of anthocyanins and risk of cardiovascular disease: A systematic review and metaanalysis of prospective cohort studies. Crit. Rev. Food Sci. Nutr. 2018, 2, 1. [Google Scholar] [CrossRef]
- Grosso, G.; Micek, A.; Godos, J.; Pajak, A.; Sciacca, S.; Galvano, F.; Giovannucci, E. Dietary flavonoid and lignin intake and mortality in prospective cohort studies: Systematic review and dose response Meta-analysis. Am. J. Epidemiol. 2017, 185, 1304. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Ouyang, Y.; Liu, J.; Zhao, G. Flavonoid intake and risk of CVD: A systematic review and meta-analysis of prospective cohort studies. Br. J. Nutr. 2014, 111, 1. [Google Scholar] [CrossRef] [PubMed]
- Zamora-Ros, R.; Knaze, V.; Lujan-Barroso, L.; Slimani, N.; Romieu, I.; Touillaud, M.; Kaaks, R.; Teucher, B.; Mattiello, A.; Grioni, S.; et al. Estimation of the intake of anthocyanidins and their food sources in the European Prospective Investigation into Cancer and Nutrition (EPIC) Study. Br. J. Nutr. 2011, 106, 1090. [Google Scholar] [CrossRef] [PubMed]
- Aune, D.; Giovannucci, E.; Boffetta, P.; Fadnes, L.; Keum, N.; Norat, T.; Greenwood, D.; Riboli, E.; Vatten, L.; Tonstad, S. Fruit and vegetable intake and the risk of cardiovascular disease, total cancer and all-cause mortality-a systematic review and dose-response meta-analysis of prospective studies. Int. J. Epidemiol. 2017, 46, 1029. [Google Scholar] [CrossRef] [PubMed]
- Horbowicz, M.; Kosson, R.; Grzesiuk, A.; Derbski, H. Anthocyanins of fruits and vegetables-their occurrence, analysis and role in human nutrition. Veg. Crop. Res. Bull. 2008, 68, 22. [Google Scholar] [CrossRef]
- McCullough, M.; Peterson, J.; Patel, R.; Jacques, P.; Shah, R.; Dwyer, J. Flavonoid intake and cardiovascular disease mortality in a prospective cohort of US adults. Am. J. Clin. Nutr. 2012, 95, 454. [Google Scholar] [CrossRef] [PubMed]
- Mink, P.; Scrafford, C.; Barraj, L.; Harnack, L.; Hong, C.; Nettleton, J.; Jacobs, D.J. Flavonoid intake and cardiovascular disease mortality: A prospective study in postmenopausal women. Am. J. Clin. Nutr. 2007, 85, 895. [Google Scholar] [CrossRef] [PubMed]
- Cassidy, A. Berry anthocyanin intake and cardiovascular health. Mol. Asp. Med. 2018, 61, 76. [Google Scholar] [CrossRef] [PubMed]
- Vyas, P.; Kalidindi, S.; Chibrikova, L.; Igamberdiev, A.; Weber, J. Chemical analysis and effect of blueberry and lingonberry fruits and leaves against glutamate-mediated excitotoxicity. J. Agric. Food Chem. 2013, 61, 7759. [Google Scholar] [CrossRef]
- Bhuiyan, M.; Kim, J.; Ha, T.; Kim, S.; Cho, K. Anthocyanins extracted from black soybean seed coat protect primary cortical neurons against in vitro ischemia. Biol. Pharm. Bull. 2012, 35, 999. [Google Scholar] [CrossRef] [PubMed]
- Williams, A.J. ABC of oxygen: Assessing and interpreting arterial blood gases and acid-base balance. BMJ 1998, 317, 1213–1216. [Google Scholar] [CrossRef] [PubMed]
- Rosenberger, J.; Petrovics, G.; Buzas, B. Oxidative stress induces proorphanin FQ and proenkephalin gene expression in astrocytes through p38- and ERK-MAP kinases and NF-kappaB. J. Neurochem. 2001, 79, 35–44. [Google Scholar] [CrossRef] [PubMed]
- Jäkel, S.; Dimou, L. Glial Cells and Their Function in the Adult Brain: A Journey through the History of Their Ablation. Front. Cell. Neurosci. 2017, 11, 24. [Google Scholar] [CrossRef] [PubMed]
- Salman, M.M.; Kitchen, P.; Woodroofe, M.N.; Bill, R.M.; Conner, A.C.; Heath, P.R.; Conner, M.T. Transcriptome Analysis of Gene Expression Provides New Insights into the Effect of Mild Therapeutic Hypothermia on Primary Human Cortical Astrocytes Cultured under Hypoxia. Front. Cell. Neurosci. 2017, 11, 386. [Google Scholar] [CrossRef] [PubMed]
- Barreto, G.; White, R.E.; Ouyang, Y.; Xu, L.; Giffard, R.G. Astrocytes: Targets for neuroprotection in stroke. Cent. Nerv. Syst. Agents Med. Chem. 2011, 11, 164–173. [Google Scholar] [CrossRef]
- Paixão, J.; Dinis, T.; Almeida, L. Dietary anthocyanins protect endothelial cells against peroxynitrite-induced mitochondrial apoptosis pathway and Bax nuclear translocation: An in vitro approach. Apoptosis 2011, 16, 976. [Google Scholar] [CrossRef]
- Kim, Y.; Yoon, H.; Lee, Y.; Youn, D.; Ha, T.; Kim, H.; Lee, J. Anthocyanin Extracts from Black Soybean (Glycine max L.) Protect Human Glial Cells Against Oxygen-Glucose Deprivation by Promoting Autophagy. Biomol. Ther. 2012, 20, 68. [Google Scholar] [CrossRef]
- Kang, T.; Hur, J.; Kim, H.; Ryu, J.; Kim, S. Neuroprotective effects of the cyanidin-3-O-beta-d-glucopyranoside isolated from mulberry fruit against cerebral ischemia. Neurosci. Lett. 2006, 391, 122. [Google Scholar] [CrossRef]
- Lau, F.; Bielinski, D.; Joseph, J. Inhibitory effects of blueberry extract on the production of inflammatory mediators in lipopolysaccharide-activated BV2 microglia. J. Neurosci. Res. 2007, 85, 1010. [Google Scholar] [CrossRef]
- Tarozzi, A.; Morroni, F.; Hrelia, S.; Angeloni, C.; Marchesi, A.; Cantelli-Forti, G.; Hrelia, P. Neuroprotective effects of anthocyanins and their in vivo metabolites in SH-SY5Y cells. Neurosci. Lett. 2007, 424, 36. [Google Scholar] [CrossRef] [PubMed]
- Shui, G.; Bao, Y.; Bo, J.; An, L. Protective effect of protocatechuic acid from Alpinia oxyphylla on hydrogen peroxide-induced oxidative PC12 cell death. Eur. J. Pharmacol. 2006, 538, 73. [Google Scholar] [CrossRef] [PubMed]
- Wiseman, W.; Egan, J.; Slemmer, J.; Shaughnessy, K.; Ballem, K.; Gottschall-Pass, K.T.; Sweeney, M.I. Feeding blueberry diets inhibits angiotensin II-converting enzyme (ACE) activity in spontaneously hypertensive stroke-prone rats. Can. J. Physiol. Pharmacol. 2011, 89, 67. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.; Kang, J.; Xie, C.; Burris, R.; Ferguson, M.; Badger, T.; Nagarajan, S. Dietary blueberries attenuate atherosclerosis in apolipoprotein E-deficient mice by upregulating antioxidant enzyme expression. J. Nutr. 2010, 140, 1628. [Google Scholar] [CrossRef] [PubMed]
- Farrell, N.; Norris, G.; Lee, S.; Chu, O.; Blesso, C. Anthocyanin-rich black elderberry extract improves markers of HDL function and reduces aortic cholesterol in hyperlipidemic mice. Food Funct. 2015, 6, 1278. [Google Scholar] [CrossRef] [PubMed]
- Bacigaluppi, M.; Comi, G.; Hermann, D. Animal models of ischemic stroke. Part One: Modeling Risk Factors. Open Neurol. J. 2010, 4, 26. [Google Scholar] [CrossRef]
- Bailey, E.; Smith, C.; Sudlow, C.; Wardlaw, J. Is the spontaneously hypertensive stroke prone rat a pertinent model of sub cortical ischemic stroke? A systematic review. Int. J. Stroke 2011, 6, 434. [Google Scholar] [CrossRef]
- Shaughnessy, K.; Boswall, I.; Scanlan, A.; Gottschall-Pass, K.; Sweeney, M. Diets containing blueberry extract lower blood pressure in spontaneously hypertensive stroke-prone rats. Nutr. Res. 2009, 29, 130. [Google Scholar] [CrossRef]
- Papandreou, M.; Dimakopoulou, A.; Linardaki, Z.; Cordopatis, P.; Klimis-Zacas, D.; Margarity, M.; Lamari, F. Effect of a polyphenol-rich wild blueberry extract on cognitive performance of mice, brain antioxidant markers and acetylcholinesterase activity. Behav. Brain Res. 2009, 198, 352. [Google Scholar] [CrossRef]
- Ramirez, M.; Izquierdo, I.; do Carmo Bassols Raseira, M.; Zuanazzi, J.; Barros, D.; Henriques, A. Effect of lyophilised Vaccinium berries on memory, anxiety and locomotion in adult rats. Pharmacol. Res. 2005, 52, 457. [Google Scholar] [CrossRef]
- Fluri, F.; Schuhmann, M.; Kleinschnitz, C. Animal models of ischemic stroke and their application in clinical research. Drug Des. Dev. Ther. 2015, 2, 3445. [Google Scholar]
- Bacigaluppi, M.; Comi, G.; Hermann, D. Animal models of ischemic stroke. Part two: Modeling cerebral ischemia. Open Neurol. J. 2010, 4, 34. [Google Scholar] [CrossRef] [PubMed]
- Woodruff, T.; Thundyil, J.; Tang, S.; Sobey, C.; Taylor, S.; Arumugam, T. Pathophysiology, treatment, and animal and cellular models of human ischemic stroke. Mol. Neurodegener. 2011, 6, 11. [Google Scholar] [CrossRef] [PubMed]
- Dewi, L.; Adnyana, M.; Mahdi, C.; Prasetyawan, S.; Srihardyastutie, A.; Aulanni’am, A. Study of Antocyanins Activity from Purple Sweet Potato for Reducing Apoptotic Cells Expression of The Cerebellum On Ischemic Stroke Rats. J. Pure App. Chem. Res. 2018, 7, 94. [Google Scholar] [CrossRef][Green Version]
- Adnyana, I.; Sudewi, A.; Samatra, D.; Suprapta, D. Neuroprotective Effects of Purple Sweet Potato Balinese Cultivar in Wistar Rats With Ischemic Stroke. Open Access Maced. J. Med. Sci. 2018, 6, 1959. [Google Scholar] [CrossRef] [PubMed]
- Cui, H.; Chen, J.; Li, J.; Cheng, F.; Yuan, K. Protection of Anthocyanin from Myrica rubra against Cerebral Ischemia-Reperfusion Injury via Modulation of the TLR4/NF-κB and NLRP3 Pathways. Molecules 2018, 23, 1788. [Google Scholar] [CrossRef]
- Shin, W.; Park, S.; Kim, E. Protective effect of anthocyanins in middle cerebral artery occlusion and reperfusion model of cerebral ischemia in rats. Life Sci. 2006, 79, 130. [Google Scholar] [CrossRef] [PubMed]
- Safaeian, L.; Tameh, A.; Ghannadi, A.; Naghani, E.; Tavazoei, H.; Alavi, S. Protective effects of Echium amoenum Fisch. and C.A. Mey. against cerebral ischemia in the rats. Adv. Biomed. Res. 2015, 4, 107. [Google Scholar]
- Adnyana, I.; Sudewi, R.; Samatra, P.; Suprapta, S. Balinese Cultivar of Purple Sweet Potato Improved Neurological Score and BDNF and Reduced Caspase-Independent Apoptosis among Wistar Rats with Ischemic Stroke. Open Access Maced. J. Med. Sci. 2019, 7, 38. [Google Scholar] [CrossRef]
- Ritz, M.; Curin, Y.; Mendelowitsch, A.; Andriantsitohaina, R. Acute treatment with red wine polyphenols protects from ischemia-induced excitotoxicity, energy failure and oxidative stress in rats. Brain Res. 2008, 1239, 226. [Google Scholar] [CrossRef]
- Di Giacomo, C.; Acquaviva, R.; Santangelo, R.; Sorrenti, V.; Vanella, L.; Li Volti, G.; D’Orazio, N.; Vanella, A.; Galvano, F. Effect of Treatment with Cyanidin-3-O-β-d-Glucoside on Rat Ischemic/Reperfusion Brain Damage. Evid. Based Complement. Altern. Med. 2012, 2012, 285750. [Google Scholar] [CrossRef] [PubMed]
- Min, J.; Yu, S.; Baek, S.; Nair, K.; Bae, O.; Bhatt, A.; Kassab, M.; Nair, M.; Majid, A. Neuroprotective effect of cyanidin-3-O-glucoside anthocyanin in mice with focal cerebral ischemia. Neurosci. Lett. 2011, 500, 157. [Google Scholar] [CrossRef] [PubMed]
- Mastantuono, T.; Di Maro, M.; Chiurazzi, M.; Battiloro, L.; Muscariello, E.; Nasti, G.; Starita, N.; Colantuoni, A.; Lapi, D. Rat Pial Microvascular Changes During Cerebral Blood Flow Decrease and Recovery: Effects of Cyanidin Administration. Front. Physiol. 2018, 9, 540. [Google Scholar] [CrossRef] [PubMed]
- Rasouli Vani, J.; Taghi Mohammadi, M.; Sarami Foroshani, M.; Rezazade, E. Evaluation of the neuroprotective and antioxidant effects of Dorema aucheri extract on cerebral ischaemia-reperfusion injury in rats. Pharm. Biol. 2019, 57, 255. [Google Scholar] [CrossRef] [PubMed]
- Mastantuono, T.; Starita, N.; Sapio, D.; D’Avanzo, S.; Di Maro, M.; Muscariello, E.; Lapi, D. The effects of Vaccinium myrtillus extract on hamster pial microcirculation during hypoperfusion-reperfusion injury. PLoS ONE 2016, 11, e0150659. [Google Scholar] [CrossRef]
- Rodriguez-Mateos, A.; Rendeiro, C.; Bergillos-Meca, T.; Tabatabaee, S.; George, T.; Heiss, C.; Spencer, J. Intake and time dependence of blueberry flavonoid-induced improvements in vascular function: A randomized, controlled, double-blind, crossover intervention study with mechanistic insights into biological activity. Am. J. Clin. Nutr. 2013, 98, 1179. [Google Scholar] [CrossRef]
- Rodriguez-Mateos, A.; Feliciano, R.; Boeres, A.; Weber, T.; Dos Santos, C.; Ventura, M.; Heiss, C. Cranberry (poly)phenol metabolites correlate with improvements in vascular function: A double-blind, randomized, controlled, dose-response, crossover study. Mol. Nutr. Food Res. 2016, 60, 2130. [Google Scholar] [CrossRef]
- Barona, J.; Aristizabal, J.; Blesso, C.; Volek, J.; Fernandez, M. Grape polyphenols reduce blood pressure and increase flow-mediated vasodilation in men with metabolic syndrome. J. Nutr. 2012, 142, 1626. [Google Scholar] [CrossRef]
- Van Mierlo, L.; Zock, P.; van der Knaap, H.; Draijer, R. Grape polyphenols do not affect vascular function in healthy men. J. Nutr. 2010, 140, 1769. [Google Scholar] [CrossRef]
- Rees, A.; Dodd, G.; Spencer, J. The Effects of Flavonoids on Cardiovascular Health: A Review of Human Intervention Trials and Implications for Cerebrovascular Function. Nutrients 2018, 10, 1852. [Google Scholar] [CrossRef]
- Bowtell, J.; Aboo-Bakkar, Z.; Conway, M.; Adlam, A.; Fulford, J. Enhanced task related brain activation and resting perfusion in healthy older adults after chronic blueberry supplementation. Appl. Physiol. Nutr. Metab. 2017, 42, 773. [Google Scholar] [CrossRef] [PubMed]
- Cassidy, A.; O’Reilly, E.; Kay, C.; Sampson, L.; Franz, M.; Forman, J.; Curhan, G.; Rimm, E. Habitual intake of flavonoid subclasses and incident hypertension in adults. Am. J. Clin. Nutr. 2011, 93, 338. [Google Scholar] [CrossRef] [PubMed]
- Igwe, E.; Charlton, K.; Roodenrys, S.; Kent, K.; Fanning, K.; Netzel, M. Anthocyanin-rich plum juice reduces ambulatory blood pressure but not acute cognitive function in younger and older adults: A pilot crossover dose-timing study. Nutr. Res. 2017, 47, 28. [Google Scholar] [CrossRef] [PubMed]
- Herrera-Arellano, A.; Miranda-Sánchez, J.; Avila-Castro, P.; Herrera-Alvarez, S.; Jiménez-Ferrer, J.; Zamilpa, A.; Román-Ramos, R.; Ponce-Monter, H.; Tortoriello, J. Clinical effects produced by a standardized herbal medicinal product of Hibiscus sabdariffa on patients with hypertension. A randomized, double-blind, lisinopril-controlled clinical trial. Planta Med. 2007, 73, 6. [Google Scholar] [CrossRef] [PubMed]
- Vinson, J.; Demkosky, C.; Navarre, D.; Smyda, M.; Vinson, J.A.; Demkosky, C.A.; Navarre, D.A.; Smyda, M.A. High-antioxidant potatoes: Acute in vivo antioxidant source and hypotensive agent in humans after supplementation to hypertensive subjects. J. Agric. Food Chem. 2012, 60, 6749. [Google Scholar] [CrossRef] [PubMed]
- Khoshnam, S.; Sarkaki, A.; Rashno, M.; Farbood, Y. Memory deficits and hippocampal inflammation in cerebral hypoperfusion and reperfusion in male rats: Neuroprotective role of vanillic acid. Life Sci. 2018, 211, 126. [Google Scholar] [CrossRef] [PubMed]
- Asano, T.; Matsuzaki, H.; Iwata, N.; Xuan, M.; Kamiuchi, S.; Hibino, Y.; Sakamoto, T.; Okazaki, M. Protective Effects of Ferulic Acid against Chronic Cerebral Hypoperfusion-Induced Swallowing Dysfunction in Rats. Int. J. Mol. Sci. 2017, 18, 550. [Google Scholar] [CrossRef]
- Cao, Y.; Zhang, L.; Sun, S.; Yi, Z.; Jiang, X.; Jia, D. Neuroprotective effects of syringic acid against OGD/R-induced injury in cultured hippocampal neuronal cells. Int. J. Mol. Med. 2016, 38, 567. [Google Scholar] [CrossRef]
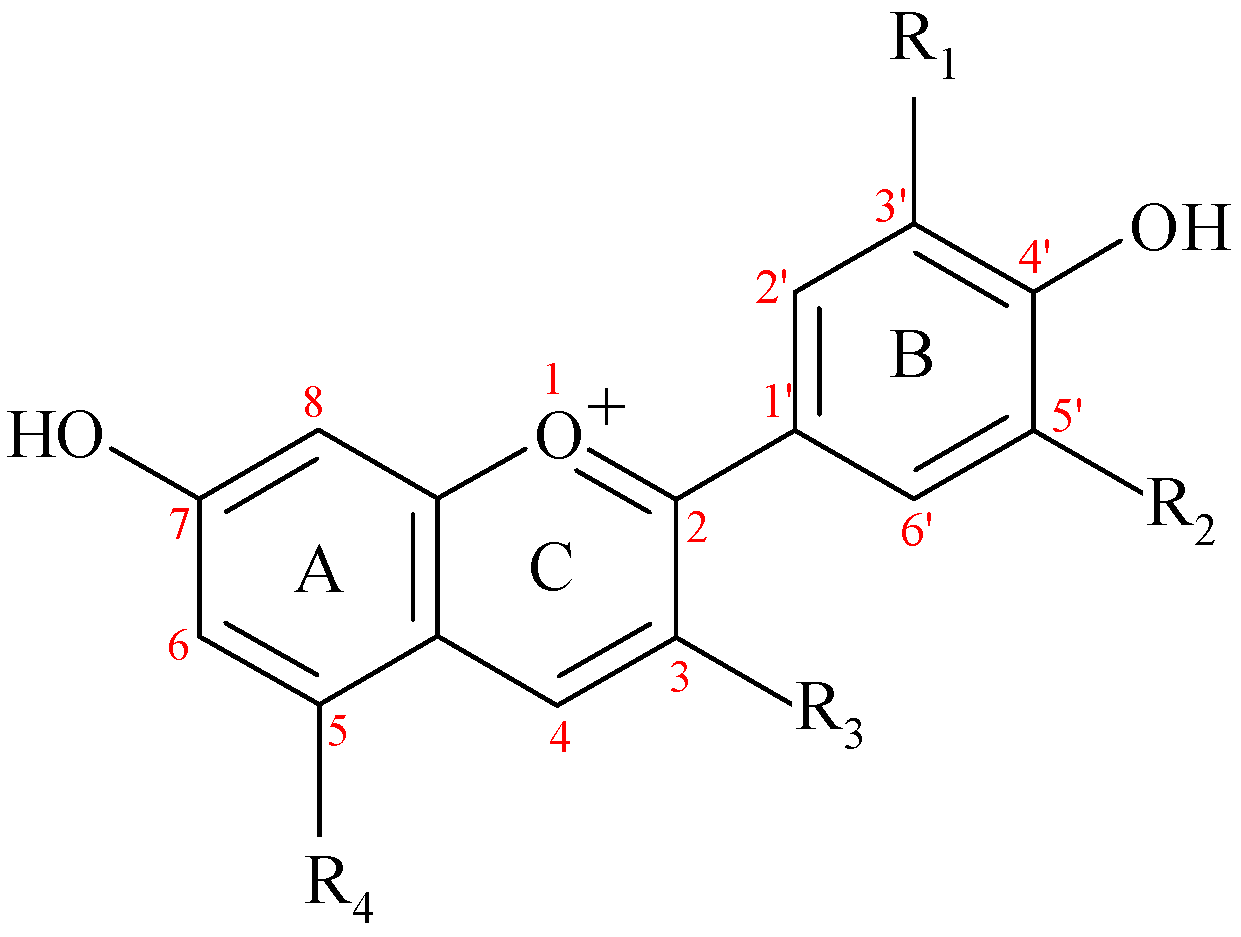
| ACND/ACN (Abbreviation) | R1 | R2 | R3 | R4 | Latin Name of Plant Sources | Main Sources | Mean Content mg/100 g | Reference |
|---|---|---|---|---|---|---|---|---|
| Pelargonidin (Pg) | -H | -H | -OH | -OH | Fragaria sp. Phaseolus vulgaris Camellia sinensis | Strawberry Common bean (black) Tea (Kenyan purple leaf) | 4.31 0.95 0.841 | [14] [14] [15] |
| Cyanidin (Cy) | -OH | -H | -OH | -OH | Camellia sinensis Phaseolus vulgaris Rubus idaeus Fragaria sp. | Tea (Kenyan purple leaf) Common bean (black) Red raspberry Strawberry | 1.751 1.63 0.53 0.50 | [15] [14] [14] [14] |
| Delphinidin (De) | -OH | -OH | -OH | -OH | Camellia sinensis | Tea (Kenyan purple leaf) | 0.121 | [15] |
| Peonidin (Pn) | -OCH3 | -H | -OH | -OH | Phaseolus vulgaris | Common bean (black) | 1.36 | [14] |
| Petunidin (Pt) | -OCH3 | -OH | -OH | -OH | ||||
| Malvidin (Mv) | -OCH3 | -OCH3 | -OH | -OH | Camellia sinensis | Tea (Kenyan purple leaf) | 0.301 | [15] |
| Pelargonidin-3-glucoside (Pg3-glc) | -H | -H | glc | -OH | Fragaria sp. Phaseolus vulgaris Morus nigra | Strawberry Common bean (black) Mulberry | 47.14 12.60 27.8 ± 2.1 | [14] [14] [16] |
| Pelargonidin-3-rutinoside (Pg3-rut) | -H | -H | rut | -OH | Ribes nigrum Morus nigra | Blackcurrant Mulberry | 2.48 1.4 ± 0.2 | [14] [16] |
| Pelargonidin-3-(6′′-succinyl-glucoside) (Pg3-(6′′-succ-glc) | -H | -H | 6′′-succ-glc | -OH | Fragaria sp. | Strawberry | 10.44 | [14] |
| Cyanidin-3-glucoside (Cy3-glc) | -OH | -H | glc | -OH | Aronia melanocarpa Sambucus nigra Rubus fruticosus 2 Ribes nigrum Prunus domestica Rubus idaeus Ribes rubrum Prunus avium Phaseolus vulgaris Oryza sativa 3 Morus nigra | Black chokeberry Black elderberry Blackberry Blackcurrant Plum Red raspberry Redcurrant Sweet cherry Common bean (black) Black rice (some varieties) 3 Mulberry | 19.64 797.13 138.72 25.07 8.63 14.89 3.37 18.73 3.99 0–470 704.1 ± 20.4 | [14] [14] [14] [14] [14] [14] [14] [14] [14] [17] [16] |
| Cyanidin-3-galactoside (Cy3-gal) | -OH | -H | gal | -OH | Aronia melanocarpa Vaccinium vitis-idaea Malus domestica | Black chokeberry Lingonberry Apple | 557.67 48.69 0.81 | [14] [14] [14] |
| Cyanidin-3-rutinoside (Cy3-rut) | -OH | -H | rut | -OH | Rubus fruticosus 2 Ribes nigrum Prunus domestica Rubus idaeus Ribes rubrum Prunus cerasus Prunus avium Morus nigra | Blackberry Blackcurrant Plum Red raspberry Redcurrant Sour cherry Sweet cherry Mulberry | 8.86 160.78 33.85 5.20 2.10 6.98 143.27 572.1 ± 22.5 | [14] [14] [14] [14] [14] [14] [14] [16] |
| Cyanidin-3-sambubioside (Cy3-samb) | -OH | -H | samb | -OH | Sambucus nigra Ribes rubrum | Black elderberry Redcurrant | 462.96 9.47 | [14] [14] |
| Cyanidin-3-sophoroside (Cy3-soph) | -OH | -H | soph | -OH | Rubus idaeus Ribes rubrum | Red raspberry Redcurrant | 37.61 2.62 | [14] [14] |
| Cyanidin-3-arabinoside (Cy3-ara) | -OH | -H | ara | -OH | Aronia melanocarpa Sambucus nigra Vaccinium vitis-idaea | Black chokeberry Black elderberry Lingonberry | 252.76 252.76 5.85 | [14] [14] [14] |
| Cyanidin-3-xyloside (Cy3-xyl) | -OH | -H | xyl | -OH | Aronia melanocarpa Rubus fruticosus 2 | Black chokeberry Blackberry | 45.90 9.74 | [14] [14] |
| Cyanidin-3- glucosyl-rutinoside (Cy3-glc-rut) | -OH | -H | glc-rut | -OH | Rubus idaeus Ribes rubrum Prunus cerasus | Red raspberry Redcurrant Sour cherry | 7.06 4.23 43.63 | [14] [14] [14] |
| Cyanidin-3- xylosyl-rutinoside (Cy3,5-xyl-rut) | -OH | -H | xyl-rut | -OH | Ribes rubrum | Redcurrant | 11.22 | [14] |
| Cyanidin-3,5-diglucoside (Cy3,5-diglc) | -OH | -H | glc | glc | Sambucus nigra | Black elderberry | 17.46 | [14] |
| Delphinidin-3-glucoside (De3-glc) | -OH | -OH | glc | -OH | Ribes nigrum Vitis vinifera Vaccinium augustifolium Phaseolus vulgaris | Blackcurrant Grape (black) Lowbush blueberry Common bean (black) | 86.68 2.63 15.17 14.50 | [14] [14] [14] [14] |
| Delphinidin-3-galactoside (De3-gal) | -OH | -OH | gal | -OH | Vaccinium augustifolium | Lowbush blueberry | 16.14 | [14] |
| Delphinidin-3-rutinoside (De3-rut) | -OH | -OH | rut | -OH | Ribes nigrum | Blackcurrant | 304.91 | [14] |
| Delphinidin-3-glucosyl-glucoside (De3-glc-glc) | -OH | -OH | glc-glc | -OH | Allium cepa L. var. cepa | Onion (red) | 6.50 | [14] |
| Peonidin 3-glucoside (Pn3-glc) | -OCH3 | -H | glc | -OH | Vitis vinifera Vaccinium vitis-idaea | Grape (black) Lingonberry | 5.80 4.25 | [14] [14] |
| Peonidin 3-rutinoside (Pn3-glc) | -OCH3 | -H | rut | -OH | Prunus domestica Prunus cerasus Prunus avium | Plum Sour cherry Sweet cherry | 4.85 2.70 7.42 | [14] [14] [14] |
| Petunidin-3-glucoside (Pt3-glc) | -OCH3 | -OH | glc | -OH | Vaccinium augustifolium Oryza sativa 3 | Lowbush blueberry Black rice (some varieties) 3 | 11.20 0–40 | [14] [17] |
| Malvidin 3-glucoside (Mv3-glc) | -OCH3 | -OCH3 | glc | -OH | Vitis vinifera Vaccinium augustifolium Vitis vinifera | Grape (black) Lowbush blueberry Red wine from grape | 39.23 26.06 9.97 (1) | [14] [14] [14] |
| Malvidin 3-galactoside (Mv3-gal) | -OCH3 | -OCH3 | gal | -OH | Vaccinium augustifolium | Lowbush blueberry | 21.43 | [14] |
| Malvidin 3-O-(6′′-acetyl-glucoside) (Mv3-(6′′-Ac-glc) | -OCH3 | -OCH3 | 6′′-Ac-glc | -OH | Vitis vinifera Vaccinium augustifolium Vitis vinifera | Grape (black) Lowbush blueberry Red wine from grape | 9.66 14.74 3.52 (1) | [14] [14] [14] |
| ACN or Metabolite | Final Concentration of Compound before Incubation (µM) | Transport Efficiency/Uptake/Recovery | Method Applied | References |
|---|---|---|---|---|
| Cy3-glc | 100 | 11.4 ± 2.6, 1 h; 13.72 ± 2.66, 3 h; 21.07 ± 5.2, 18 h 1 | HPLC-DAD | [71] |
| Cy3-glc in presence of 0.1% ethanol | 100 | 5.99 ± 1.18, 1 h; 13.1 ± 0.68, 3 h; 17.8 ± 0.31, 18 h 1 | HPLC-DAD | |
| De3-glc | 100 | 5.0 ± 0.7, 1 h; 8.8 ± 1.1, 3 h; 11.6 ± 0.6, 18 h 2 | HPLC-DAD/MS | [69] |
| 4′Me-De3-glc | 100 | 5.5 ± 1.3, 1 h; 11.5 ± 1.0, 3 h; 17.6 ± 1.7, 18 h 2 | HPLC-DAD/MS | |
| Cy3-glc | 100 | 8.0 ± 1.1, 1 h; 12.6 ± 0.9, 3 h; 16.0 ± 0.6, 18 h 2 | HPLC-DAD/MS | |
| 4′/3′Me-Cy3-glc | 100 | 9.2 ± 2.0, 1 h; 13.4 ± 1.2, 3 h; 19.0 ± 1.4, 18 h 2 | HPLC-DAD/MS | |
| Mv3-glc | 100 | 5.3 ± 0.1, 1 h; 13.3 ± 2.4, 3 h; 20.0 ± 3.3, 18 h 2 | HPLC-DAD/MS | |
| Cy3-rut | 30 | n.d., 2 h; 5.1±6.5, 6 h; 25.6 ± 8.5 18 h 3 n.d., 2 h and 6 h; 18.7 ± 3.2, 18 h 4 | LC/MS-MS | [73] |
| Pg3-glc | 30 | n.d., 2 h; 9.9 ± 3.2, 6 h; 18.1 ± 6.6, 18 h 3 n.d., 2 h and 6 h; 11.23 ± 4.3, 18 h 4 | LC/MS-MS | |
| Cy3-rut | 30 | 83.2 ± 3.1 5 | LC/MS-MS | |
| Pg3-glc | 30 | 84.3 ± 6.6 5 | LC/MS-MS |
| Source Composition | Dose Administrated, Administration Route | ACN/Metabolites in Brain Tissue | Method Applied | Experimental Model | References |
|---|---|---|---|---|---|
| Powdered blueberry 2% (w/w) in diet supplemented 1 | ad libitum in food, 8 weeks, p.o. | Mv3-glc: 279 fmol/g cortex tissue, 432 fmol/g midbrain and diencephalon tissue 2 | LC-MS/MS | 15 healthy neutered 32–41-day-oldmale pigs (Yorkshire Landrace) | [46] |
| Powdered whole blueberry fruit (with 7.97 mg Cy3-glc equivalents/g) 3 | ad libitum in food, 4 weeks, 0, 10, 20, 40 g/kg diet, 4 groups (around 1.48 mmol ANCs of dose/kg bw/day) | 0.81 pmoli Cy3-glc equivalents/g FW (cortex) 0.36 pmoli Cy3-glc\equivalents/g FW (cerebellum) | LC-MS/MS | 20 healthy neutered 32–41-day-oldmale pigs (Yorkshire Landrace) | [64] |
| Bilberry extract contained, (as mg aglycone/g extract): 69.69% De3-glc and others 4 | 82.5 mg/kg bw/day, 3 weeks, p.o. | Mv3-glc: 4.43 pmol/g 5 | LC/MS-MS | 40 commercial, newly weaned piglets | [87] |
| Blackberry extract with 47.9% ACN, Cy3-glc (91.2%) | 14.8 mmol ACN/kg diet (with 13.5 mmol of Cy3-glc/kg diet/day) ad libitum, 15 days, p.o. | Cy3-glc: 0.21 ± 0.05 nmol/g tissue (84.0%) | HPLC-ESI-MS-MS and HPLC-DAD | 12 male Wistar rats, (Iffa-Credo, L’Arbresle, France) | [90] |
| Tart cherry ACN 10% 6 | 20 g of diet powder/animal/day; approximately 10% below ad libitum intake, 3 weeks, p.o. | Cy3-glc-rut: 654.86 ± 3 2.18 fmoli/g tissue; Pn3-rut: 33.30 ± 2.95 fmoli/g tissue | LC/MS-MS | 18 male Wistar rats, 6 weeks old | [91] |
| Kenyan purple tea contained: Cy, the most abundant (1755.60 μg/m), Pn, Pg, De, Mv | 200 mg/kg bw/day, 14 days, p.o. | Identified: De, Cy, Pg, Pn, Mv | HPLC | 15 healthy Swiss white mice, 8-week-old female adult | [15] |
| Pelargonidin (99.82 %), dissolved in 50% aqueous ethanol | 50 mg pelargonidin/kg bw (one dose), p.o. | 0.16 nmol pelargonidin/g tissue (2 h after administration), only compound detected | HPLC and LC-MS | 6 male Sprague-Dawley rats | [67] |
| Pure ACN mixture extracted from Vitis vinifera grapes (almost 50% Mv3-glc) | 8 mg/kg bw for 10 min, intragastrically administrated | Mv3-glc: 122.0 ± 54.72 pmoli/g; Mv3-(6-O-p-coumaroyl) glc: 196.54 ± 71.92 pmoli/g | HPLC-DAD-MS | 13 male Wistar rats (Harlan Teklad 2018) | [92] |
| Cy3-glc (in PBS) | 668 nmol (one dose), i.v. | Cy3-glc (pmol/g): 7.48±0.79 (2 min); 2.18 ± 0.58 (15 min); Pn3-glc (pmol/g): 2.07 ± 1.18 (2 min), 0.40 ± 0.38 (15 min); Pt3-glc (pmol/g): 1.15 ± 0.62 (2 min), 2.45 ± 0.22 (15 min) | UPLC/MS-MS | 22 male Wistar rats (Rattus Norvegicus, Harlan Italy S.r.l.), same age (15 weeks) | [93] |
| 2.7 μmol of polyphenol microbial metabolites (inclusive of ACN metabolites), dissolved in 30 μL methanol into 0.3 mL PBS (one dose) 7 | HBA, a metabolite of Pg B-ring: 347; VA, a metabolite of Pn B-ring: 174; PCA, a metabolite of Cy B-ring: 226; GA, a metabolite of De B-ring: 3125, i.v. 8 | HBA: 126.63 (control); 206.97 (2 min); 407.92 (15 min); VA: 99.71 (control); n.d. (2 min); 384.71 (15 min); PCA: n.d. (2 min), n.d. (15 min); GA: 612.72 (2 min); 610.82 (15 min) 9 | UPLC/MS-MS | 20 male Wistar rats (Rattus Norvegicus, Harlan Italy S.r.l.), same age (12 weeks) | [94] |
| Anthocyanin | Sources | Reference |
|---|---|---|
| Total ACN: 147.0 mg/mL (method not specified) | Purple sweet potato, Balinese cultivar Ipomoea batatas L. (aqueous extracts, 1:1, kg/L) | [185] |
| Not specified | Purple sweet potato, Balinese cultivar Ipomoea batatas L. (aqueous extracts, 1:1) | [181] |
| Pt3,5-diglc, Pn3-glc, Mv3-glc, De3-glc, De3-(6-O-coumaroyl) glc (identified) 1 | Purple sweet potato extracts, from Bali, Ipomoea batatas L. (extract anthocyanin, commercially available) | [180] |
| Cy3-glc (extracted and purified anthocyanin), purity 98.3% | Morus alba berries (mulberries) | [165] |
| Cy3-glc: 0–21.28% 2 Total ACN: 0–22.07% 3 | Myrica rubra berries (bayberry) eight cultivars (Boqi 1, Boqi 2, Tanmei, Shuijing, Dongqui, Dingdai, Wandao, Wild) from China (purified anthocyanin extracts) | [182] |
| (Pt3-(p-coumaroyl)-rut-5-glc), purity 98.3% | Fruits of Lycium ruthenicu (extracted and purified anthocyanin) | [82] |
| Not specified | Vaccinium angustifolium (fresh lowbush blueberries) | [13] |
| Total ACN: 3.1% 3 | Petals of Echium amoenum (total anthocyanin extract dried by lyophilization) | [184] |
| Flavonoids, ACN and phenolic acid compounds with antioxidant activity (no other data) | Dorema aucheri leaves (extract hidroalcoholic in ethanol 70%) | [190] |
| Total ACN: 34.7%, expressed as Cy3-glc | Fruits of Vaccinium myrtillus (lyophilized extract) | [191] |
| Medox-75 mg ACN/capsule | Concentrate from wild Scandinavian bilberries (Vaccinium sp.) and black currants (Ribes nigrum), commercially available | [183] |
| Provinols™ composition (in g/kg of dry powder): total ACN: 61; total ACND: 19; proanthocyanidins: 480; catechin: 38, hydroxycinnamic acids: 18, flavonols: 14, polymeric tannins: 3704 | Red wine with polyphenolic compounds, commercially available | [186] |
| Cy | - | [189] |
| Cy3-glc (extracted and purified anthocyanin) | Tart cherries (Prunus cerasus fruits) | [188] |
| Cy3-glc | - | [187] |
| Mv | - | [81] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Manolescu, B.N.; Oprea, E.; Mititelu, M.; Ruta, L.L.; Farcasanu, I.C. Dietary Anthocyanins and Stroke: A Review of Pharmacokinetic and Pharmacodynamic Studies. Nutrients 2019, 11, 1479. https://doi.org/10.3390/nu11071479
Manolescu BN, Oprea E, Mititelu M, Ruta LL, Farcasanu IC. Dietary Anthocyanins and Stroke: A Review of Pharmacokinetic and Pharmacodynamic Studies. Nutrients. 2019; 11(7):1479. https://doi.org/10.3390/nu11071479
Chicago/Turabian StyleManolescu, Bogdan Nicolae, Eliza Oprea, Magdalena Mititelu, Lavinia L. Ruta, and Ileana C. Farcasanu. 2019. "Dietary Anthocyanins and Stroke: A Review of Pharmacokinetic and Pharmacodynamic Studies" Nutrients 11, no. 7: 1479. https://doi.org/10.3390/nu11071479
APA StyleManolescu, B. N., Oprea, E., Mititelu, M., Ruta, L. L., & Farcasanu, I. C. (2019). Dietary Anthocyanins and Stroke: A Review of Pharmacokinetic and Pharmacodynamic Studies. Nutrients, 11(7), 1479. https://doi.org/10.3390/nu11071479







