Postprandial Lipemic Responses to Various Sources of Saturated and Monounsaturated Fat in Adults
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Overall Study Design
2.3. Initial Assessment
2.4. Meal Trials
2.5. Test Meals
2.6. Statistical Analyses
3. Results
3.1. Participant Characteristics and Premeal Physical Activity
3.2. Postprandial Metabolic Outcomes Were Similar Across Meal Trials
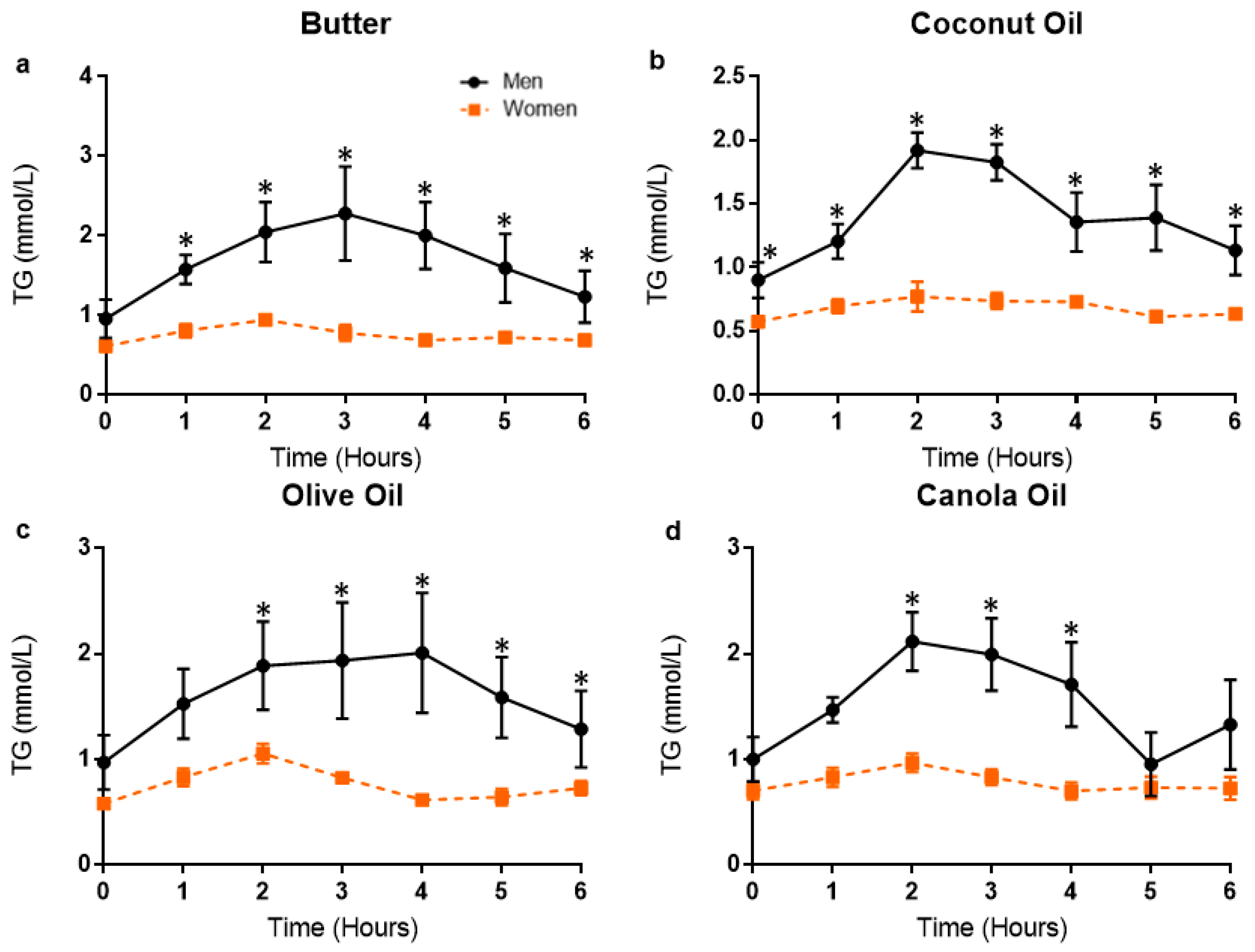
3.3. Postprandial Lipemic Responses Were Different between Men and Women
4. Discussion
4.1. Postprandial Responses in Triglycerides between Meals
4.2. Factors Influencing the Postprandial Lipemic Response
4.3. Strengths and Limitations
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Benjamin, E.J.; Muntner, P.; Alonso, A.; Bittencourt, M.S.; Callaway, C.W.; Carson, A.P.; Chamberlain, A.M.; Chang, A.R.; Cheng, S.; Das, S.R.; et al. Heart Disease and Stroke Statistics—2019 Update. A Report from the American Heart Association. Circulation 2019, 139, e56–e528. [Google Scholar] [CrossRef] [PubMed]
- Bansal, S.; Buring, J.E.; Rifai, N.; Mora, S.; Sacks, F.M.; Ridker, P.M. Fasting Compared with Nonfasting Triglycerides and Risk of Cardiovascular Events in Women. J. Am. Med. Assoc. 2007, 298, 309–315. [Google Scholar] [CrossRef]
- Hyson, D.; Rutledge, J.C.; Berglund, L. Postprandial Lipemia and Cardiovascular Disease. Curr. Atheroscler. Rep. 2003, 5, 437–444. [Google Scholar] [CrossRef]
- Herieka, M.; Erridge, C. High-fat meal induced postprandial inflammation. Mol. Nutr. Food Res. 2014, 58, 136–146. [Google Scholar] [CrossRef]
- Gower, R.M.; Wu, H.; Foster, G.A.; Devaraj, S.; Jialal, I.; Ballantyne, C.M.; Knowlton, A.A.; Simon, S.I. CD11c/CD18 expression is upregulated on blood monocytes during hypertriglyceridemia and enhances adhesion to vas- cular cell adhesion molecule-1. Arterioscler. Thromb. Vasc. Biol. 2011, 31, 160–166. [Google Scholar] [CrossRef]
- Kim, K.A.; Gu, W.; Lee, I.A.; Joh, E.H.; Kim, D.H. High fat diet-induced gut microbiota exacerbates inflammation and obesity in mice via the TLR4 signaling pathway. PLoS ONE 2012, 7, e47713. [Google Scholar] [CrossRef]
- Graner, M.; Kahri, J.; Nakano, T.; Sarna, S.J.; Nieminen, M.S.; Syvanne, M.; Taskinen, M.R. Impact of postprandial lipaemia on low-density lipoprotein (LDL) size and oxidized LDL in patients with coronary artery disease. Eur. J. Clin. Investig. 2006, 36, 764–770. [Google Scholar] [CrossRef]
- Cortés, B.; Núñez, I.; Cofán, M.; Gilabert, R.; Pérez-Heras, A.; Casals, E.; Deulofeu, R.; Ros, E. Acute effects of high-fat meals enriched with walnuts or olive oil on postprandial endothelial function. J. Am. Coll. Cardiol. 2006, 48, 1666–1671. [Google Scholar] [CrossRef]
- Tushuizen, M.E.; Nieuwland, R.; Scheffer, P.G.; Sturk, A.; Heine, R.J.; Diamant, M. Two consecutive high-fat meals affect endothelial-dependent vasodilation, oxidative stress and cellular microparticles in healthy men. J. Thromb. Haemost. 2006, 4, 1003–1010. [Google Scholar] [CrossRef]
- Lopez-Miranda, J.; Williams, C.; Lairon, D. Dietary, physiological, genetic and pathological influences on postprandial lipid metabolism. Br. J. Nutr. 2007, 98, 458–473. [Google Scholar] [CrossRef]
- Boren, J.; Matikainen, N.; Adiels, M.; Taskinen, M.R. Postprandial hypertriglyceridemia as a coronary risk factor. Clin. Chim. Acta 2014, 431, 131–142. [Google Scholar] [CrossRef]
- Jackson, K.G.; Poppitt, S.D.; Minihane, A.M. Postprandial lipemia and cardiovascular disease risk: Interrelationships between dietary, physiological and genetic determinants. Atherosclerosis 2012, 220, 22–33. [Google Scholar] [CrossRef]
- Meyer, E.; Westerveld, H.T.; de Ruyter-Meijstek, F.C.; van Greevenbroek, M.M.; Rienks, R.; van Rijn, H.J.; Erkelens, D.W.; de Bruin, T.W. Abnormal Postprandial Apolipoprotein B48 and TG responses in normolipidemic women with greater than 70% stenotic coronary artery disease. Atherosclerosis 1996, 124, 221–235. [Google Scholar] [CrossRef]
- Langsted, A.; Freiberg, J.J.; Tybjaerg-Hansen, A.; Schnohr, P.; Jensen, G.B.; Nordestgaard, B.G. Nonfasting cholesterol and triglycerides and association with risk of myocardial infarction and total mortality: The Copenhagen City Heart Study with 31 years of follow-up. J. Intern. Med. 2011, 270, 65–75. [Google Scholar] [CrossRef]
- Linton, M.F.; Fazio, S. Macrophages, inflammation, and atherosclerosis. Int. J. Obes. Relat. Metab. Disord. 2003, 27 (Suppl. 3), S35. [Google Scholar] [CrossRef]
- Chan, D.C.; Pang, J.; Romic, G.; Watts, G.F. Postprandial hypertriglyceridemia and cardiovascular disease: Current and future therapies. Curr. Atheroscler. Rep. 2013, 15, 309. [Google Scholar] [CrossRef]
- Masson, C.J.; Mensink, R.P. Exchanging saturated fatty acids for (n-6) polyunsaturated fatty acids in a mixed meal may decrease postprandial lipemia and markers of inflammation and endothelial activity in overweight men. J. Nutr. 2011, 141, 816–821. [Google Scholar] [CrossRef]
- Kruse, M.; von Loeffelholz, C.; Hoffmann, D.; Pohlmann, A.; Seltmann, A.C.; Osterhoff, M.; Hornemann, S.; Pivovarova, O.; Rohn, S.; Jahreis, G.; et al. Dietary rapeseed/canola-oil supplementation reduces serum lipids and liver enzymes and alters postprandial inflammatory responses in adipose tissue compared to olive-oil supplementation in obese men. Mol. Nutr. Food Res. 2015, 59, 507–519. [Google Scholar] [CrossRef]
- Jones, P.J.; Senanayake, V.K.; Pu, S.; Jenkins, D.J.; Connelly, P.W.; Lamarche, B.; Couture, P.; Charest, A.; Baril-Gravel, L.; West, S.G.; et al. DHA-enriched high-oleic acid canola oil improves lipid profile and lowers predicted cardiovascular disease risk in the canola oil multicenter randomized controlled trial. Am. J. Clin. Nutr. 2014, 100, 88–97. [Google Scholar] [CrossRef]
- Boniface, D.R.; Tefft, M.E. Dietary Fats and 16-year CHD mortality in a cohort of men and women in Great Britain. Eur. J. Clin. Nutr. 2002, 56, 786–792. [Google Scholar] [CrossRef]
- Mann, J.I.; Appleby, P.N.; Key, T.J.; Thorogood, M. Dietary Determinants of IHD in Health Conscious Individuals. Heart 1997, 78, 450–455. [Google Scholar] [CrossRef] [PubMed]
- Schwingshackl, L.; Hoffmann, G. Dietary fatty acids in the secondary prevention of coronary heart disease: A systematic review, meta-analysis and metaregression. BMJ Open 2014, 4, e004487. [Google Scholar] [CrossRef]
- de Oliveira Otto, M.C.; Mozaffarian, D.; Kromhout, D.; Bertoni, A.G.; Sibley, C.T.; Jacobs, D.R., Jr.; Nettleton, J.A. Dietary intake of saturated fat by food source and incident cardiovascular disease: The Multi-Ethnic Study of Atherosclerosis. Am. J. Clin. Nutr. 2012, 96, 397–404. [Google Scholar] [CrossRef]
- Teng, K.T.; Nagapan, G.; Cheng, H.M.; Nesaretnam, K. Palm olein and olive oil cause a higher increase in postprandial lipemia compared with lard but had no effect on plasma glucose, insulin and adipocytokines. Lipids 2011, 46, 381–388. [Google Scholar] [CrossRef] [PubMed]
- Panth, N.; Dias, C.B.; Wynne, K.; Singh, H.; Garg, M.L. Medium-chain fatty acids lower postprandial lipemia: A randomized crossover trial. Clin. Nutr. 2019, in press. [Google Scholar] [CrossRef]
- Mekki, N.; Charbonnier, M.; Borel, P.; Leonardi, J.; Juhel, C.; Portugal, H.; Lairon, D. Butter Differs from Olive Oil and Sunflower Oil in Its Effects on Postprandial Lipemia and Triacylglycerol-Rich Lipoproteins after Single Mixed Meals in Healthy Young Men. J. Nutr. 2002, 132, 3642–3649. [Google Scholar] [CrossRef]
- Thomsen, C.; Rasmussen, O.; Lousen, T.; Holst, J.J.; Fenselau, S.; Schrezenmeir, J.; Hermansen, K. Differential effects of saturated and monounsaturated fatty acids on postprandial lipemia and incretin responses in healthy subjects. Am. J. Clin. Nutr. 1999, 69, 1135–1143. [Google Scholar] [CrossRef] [PubMed]
- Lesser, M.; Mauldin, K.; Sawrey-Kubicek, L.; Gildengorin, V.; King, J. The Type of Dietary Fat in an Isocaloric Breakfast Meal Does Not Modify Postprandial Metabolism in Overweight/Obese Pregnant Women. Nutrients 2019, 11, 490. [Google Scholar] [CrossRef]
- Kristensen, M.; Savorani, F.; Christensen, S.; Engelsen, S.B.; Bügel, S.; Toubro, S.; Tetens, I.; Astrup, A. Flaxseed dietary fibers suppress postprandial lipemia and appetite sensation in young men. Nutr. Metab. Cardiovasc. Dis. 2013, 23, 136–143. [Google Scholar] [CrossRef]
- Sun, L.; Tan, K.W.; Lim, J.Z.; Magkos, F.; Henry, C.J. Dietary fat and carbohydrate quality have independent effects on postprandial glucose and lipid responses. Eur. J. Nutr. 2016, 57, 243–250. [Google Scholar] [CrossRef]
- Calabrese, C.; Myer, S.; Munson, S.; Turet, P.; Birdsall, T.C. A cross-over study of the effect of a single oral feeding of medium chain triglyceride oil vs. canola oil on post-ingestion plasma triglyceride in healthy men. Altern. Med. Rev. 1999, 4, 23–28. [Google Scholar] [PubMed]
- Lorenzen, J.K.; Nielsen, S.; Holst, J.J.; Tetens, I.; Rehfeld, J.F.; Astrup, A. Effect of dairy calcium or supplementary calcium intake on postprandial fat metabolism, appetite, and subsequent energy intake. Am. J. Clin. Nutr. 2007, 85, 678–687. [Google Scholar] [CrossRef] [PubMed]
- Hazim, J.; Hlais, S.; Ghattas, H.; Shatila, D.; Bassil, M.; Obeid, O. Phosphorus supplement alters postprandial lipemia of healthy male subjects: A pilot cross-over trial. Lipids Health Dis. 2014, 13, 109. [Google Scholar] [CrossRef] [PubMed]
- Kay, C.D.; Holub, B.J. The effect of wild blueberry (Vaccinium angustifolium) consumption on postprandial serum antioxidant status in human subjects. Br. J. Nutr. 2002, 88, 389. [Google Scholar] [CrossRef]
- Dubois, C.; Armand, M.; Azais-Braesco, V.; Portugal, H.; Pauli, A.M.; Bernard, P.M.; Latge, C.; Lafont, H.; Borel, P.; Lairon, D. Effects of moderate amounts of emulsified dietary fat on postprandial lipemia and lipoproteins in normolipidemic adults. Am. J. Clin. Nutr. 1994, 60, 374–382. [Google Scholar] [CrossRef]
- Dubois, C.; Beaumier, G.; Juhel, C.; Armand, M.; Portugal, H.; Pauli, A.M.; Borel, P.; Latgé, C.; Lairon, D. Effects of graded amounts of dietary fat on postprandial lipemia and lipoproteins and in normolipidemic adults. Am. J. Clin. Nutr. 1998, 67, 31–38. [Google Scholar] [CrossRef]
- Kasai, M.; Maki, H.; Nosaka, N.; Aoyama, T.; Ooyama, K.; Uto, H.; Okazaki, M.; Igarashi, O.; Kondo, K. Effect of Medium-chain Triglycerides on the Postprandial Triglyceride Concentration in Healthy Men. Biosci. Biotechnol. Biochem. 2003, 67, 46–53. [Google Scholar] [CrossRef]
- Hansson, P.; Holven, K.B.; Øyri, L.K.; Brekke, H.K.; Biong, A.S.; Gjevestad, G.O.; Raza, G.S.; Herzig, K.H.; Thoresen, M.; Ulven, S.M. Meals with Similar Fat Content from Different Dairy Products Induce Different Postprandial Triglyceride Responses in Healthy Adults: A Randomized Controlled Cross-Over Trial. J. Nutr. 2019, 149, 422–431. [Google Scholar] [CrossRef] [PubMed]
- Couillard, C.; Bergeron, N.; Prud’homme, D.; Bergeron, J.; Tremblay, A.; Bouchard, C.; Mauriege, P.; Després, J.P. Gender Difference in Postprandial Lipemia. Arterioscler. Thromb. Vasc. Biol. 1999, 19, 2448–2455. [Google Scholar] [CrossRef]
- Mekki, N. Influence of Obesity and Body Fat Distribution on Postprandial Lipemia and Triglyceride-Rich Lipoproteins in Adult Women. J. Clin. Endocrinol. Metab. 1999, 84, 184–191. [Google Scholar] [CrossRef]
- Blackburn, P.; Lamarche, B.; Couillard, C.; Pascot, A.; Tremblay, A.; Bergeron, J.; Lemieux, I.; Després, J.P. Contribution of Visceral Adiposity to the Exaggerated Postprandial Lipemia of Men with Impaired Glucose Tolerance. Diabetes Care 2003, 26, 3303–3309. [Google Scholar] [CrossRef] [PubMed][Green Version]

| Weight (g) | Energy (kcal) | Protein (g) | Fat (g) | CHO (g) | Fiber (g) | |
|---|---|---|---|---|---|---|
| Sauce | 257 | 577 | 5 | 52 | 25 | 3.3 |
| Bread | 28 | 78 | 3 | 1 | 16 | 1.5 |
| Pasta | 33 | 124 | 6 | 1 | 24 | 1.5 |
| Total | 318 | 780 | 14 | 54 | 65 | 6.3 |
| Total | Men | Women | p-Value | |
|---|---|---|---|---|
| Age | 23.8 ± 1.3 | 24.4 ± 1.5 * | 23.2 ± 0.8 | 0.03 |
| Weight (kg) | 76.52 ± 25.23 | 94.57 ± 23.09 * | 58.42 ± 8.79 | 0.01 |
| Height (cm) | 171.5 ± 10.1 | 176.0 ± 9.8 | 167.0 ± 9.1 | 0.10 |
| BMI (kg/m2) | 25.5 ± 7.2 | 30.3 ± 7.4 * | 20.7 ± 1.7 | 0.02 |
| Fasting TG (mmol/L) | 0.78 ± 0.41 | 0.96 ± 0.53 | 0.61 ± 0.10 | 0.21 |
| Fasting Glucose (mmol/L) | 4.85 ± 0.32 | 4.86 ± 0.29 | 4.84 ± 0.38 | 0.96 |
| Fasting TOTAL-C (mmol/L) | 4.76 ± 0.91 | 5.05 ± 1.09 | 4.47±0.68 | 0.44 |
| Fasting LDL-C (mmol/L) | 2.41 ± 0.79 | 2.95 ± 0.42 * | 1.87 ± 0.73 | 0.02 |
| Fasting HDL-C (mmol/L) | 1.62 ± 0.53 | 1.44 ± 0.68 | 1.79 ± 0.33 | 0.30 |
| MVPA (minutes) | 152.5 ± 17.3 | 166.9 ± 7.9 | 138 ± 24.4 | 0.51 |
| Steps/day | 7934.6 ± 747.7 | 7444.6 ± 1024.3 | 8424.6 ± 107.6 | 0.68 |
| Butter | Coconut Oil | Olive Oil | Canola Oil | p | |
|---|---|---|---|---|---|
| Triglycerides | |||||
| Peak (mmol/L) | 1.7 ± 1.1 | 1.4 ± 0.6 | 1.6 ± 0.9 | 1.6 ± 0.8 | 0.36 |
| Time to peak (hours) | 2.2 ± 0.8 | 3.0 ± 1.5 | 2.8 ± 1.1 | 2.7 ± 1.3 | 0.23 |
| tAUC (mmol/L × 6 h) | 7.6 ± 4.6 | 6.4 ± 2.8 | 7.4 ± 4.7 | 7.1 ± 2.8 | 0.33 |
| iAUC (mmol/L × 6 h) | 2.9 ± 2.5 | 2.0 ± 1.6 | 2.7 ± 2.4 | 1.9 ± 1.7 | 0.14 |
| Glucose | |||||
| Peak (mmol/L) | 5.4 ± 0.9 | 5.3 ± 0.7 | 5.4 ± 0.7 | 5.5 ± 0.8 | 0.76 |
| Time to peak (hours) | 3.2 ± 1.8 | 2.4 ± 1.9 | 2.5 ± 1.8 | 2.5 ± 2.3 | 0.48 |
| tAUC (mmol/L × 6 h) | 28.5 ± 3.7 | 27.5 ± 3.6 | 28.5 ± 2.2 | 28.0 ± 2.8 | 0.60 |
| iAUC (mmol/L × 6 h) | 0.3 ± 1.5 | −0.9 ± 2.3 | −1.1 ± 1.1 | 1.1 ± 2.6 | 0.26 |
| Metabolic Load Index | |||||
| Peak (mg/dL) | 241.0 ± 105.4 | 208.8 ± 60.5 | 230.4 ± 99.2 | 231.3 ± 231.3 | 0.24 |
| Time to peak (hours) | 2.7 ± 1.1 | 2.7 ± 1.4 | 2.9 ± 1.2 | 3.2 ± 1.9 | 0.64 |
| tAUC (mg/dL 6 h) | 1185.2 ± 447.7 | 1065.5 ± 302.4 | 1163.7 ± 448.9 | 1158.9 ± 367.8 | 0.12 |
| iAUC (mg/dL 6 h) | 260.7 ± 239.9 | 163.1 ± 151.6 | 219.1 ± 222.1 | 181.9 ± 228.6 | 0.08 |
| TOTAL-C | |||||
| Peak (mmol/L) | 4.6 ± 0.7 | 4.4 ± 1.2 | 5.1 ± 0.9 | 5.0 ± 0.9 | 0.12 |
| Time to peak (hours) | 3.2 ± 2.3 | 3.6 ± 2.4 | 4.3 ± 1.8 | 2.2 ± 2.3 | 0.09 |
| tAUC (mmol/L × 6 h) | 26.2 ± 4.2 | 24.7 ± 7.3 | 27.3 ± 5.0 | 28.7 ± 5.9 | 0.11 |
| iAUC (mmol/L × 6 h) | 0.4 ± 1.1 | 0.6 ± 0.9 | 1.1 ± 1.5 | 0.2 ± 1.6 | 0.37 |
| LDL-C | |||||
| Peak (mmol/L) | 2.7 ± 0.6 | 2.8 ± 0.8 | 2.8 ± 0.9 | 2.9 ± 0.8 | 0.66 |
| Time to peak (hours) | 2.4 ± 2.2 | 3.5 ± 2.8 | 3.3 ± 2.5 | 2.6 ± 2.1 | 0.59 |
| tAUC (mmol/L × 6 h) | 14.2 ± 3.4 | 14.3 ± 4.6 | 14.4 ± 4.6 | 15.5 ± 4.9 | 0.62 |
| iAUC (mmol/L × 6 h) | −0.3 ± 2.8 | 0.9 ± 2.7 | 1.1 ± 4.7 | 1.6 ± 5.1 | 0.72 |
| HDL-C | |||||
| Peak (mmol/L) | 1.5 ± 0.4 | 1.7 ± 0.5 | 1.6 ± 0.5 | 1.7 ± 0.6 | 0.19 |
| Time to peak (hours) | 3.4 ± 2.7 | 3.7 ± 2.0 | 3.5 ± 2.4 | 2.2 ± 2.4 | 0.52 |
| tAUC (mmol/L × 6 h) | 8.3 ± 2.1 | 8.9 ± 2.8 | 8.8 ± 2.5 | 9.2 ± 3.0 | 0.23 |
| iAUC (mmol/L × 6 h) | 0.1 ± 0.5 | 0.5 ± 0.9 | 0.1 ± 0.6 | −0.5 ± 1.2 | 0.16 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sciarrillo, C.M.; Koemel, N.A.; Tomko, P.M.; Bode, K.B.; Emerson, S.R. Postprandial Lipemic Responses to Various Sources of Saturated and Monounsaturated Fat in Adults. Nutrients 2019, 11, 1089. https://doi.org/10.3390/nu11051089
Sciarrillo CM, Koemel NA, Tomko PM, Bode KB, Emerson SR. Postprandial Lipemic Responses to Various Sources of Saturated and Monounsaturated Fat in Adults. Nutrients. 2019; 11(5):1089. https://doi.org/10.3390/nu11051089
Chicago/Turabian StyleSciarrillo, Christina M., Nicholas A. Koemel, Patrick M. Tomko, Katherine B. Bode, and Sam R. Emerson. 2019. "Postprandial Lipemic Responses to Various Sources of Saturated and Monounsaturated Fat in Adults" Nutrients 11, no. 5: 1089. https://doi.org/10.3390/nu11051089
APA StyleSciarrillo, C. M., Koemel, N. A., Tomko, P. M., Bode, K. B., & Emerson, S. R. (2019). Postprandial Lipemic Responses to Various Sources of Saturated and Monounsaturated Fat in Adults. Nutrients, 11(5), 1089. https://doi.org/10.3390/nu11051089






