Beer or Ethanol Effects on the Body Composition Response to High-Intensity Interval Training. The BEER-HIIT Study
Abstract
:1. Introduction
2. Materials and Methods
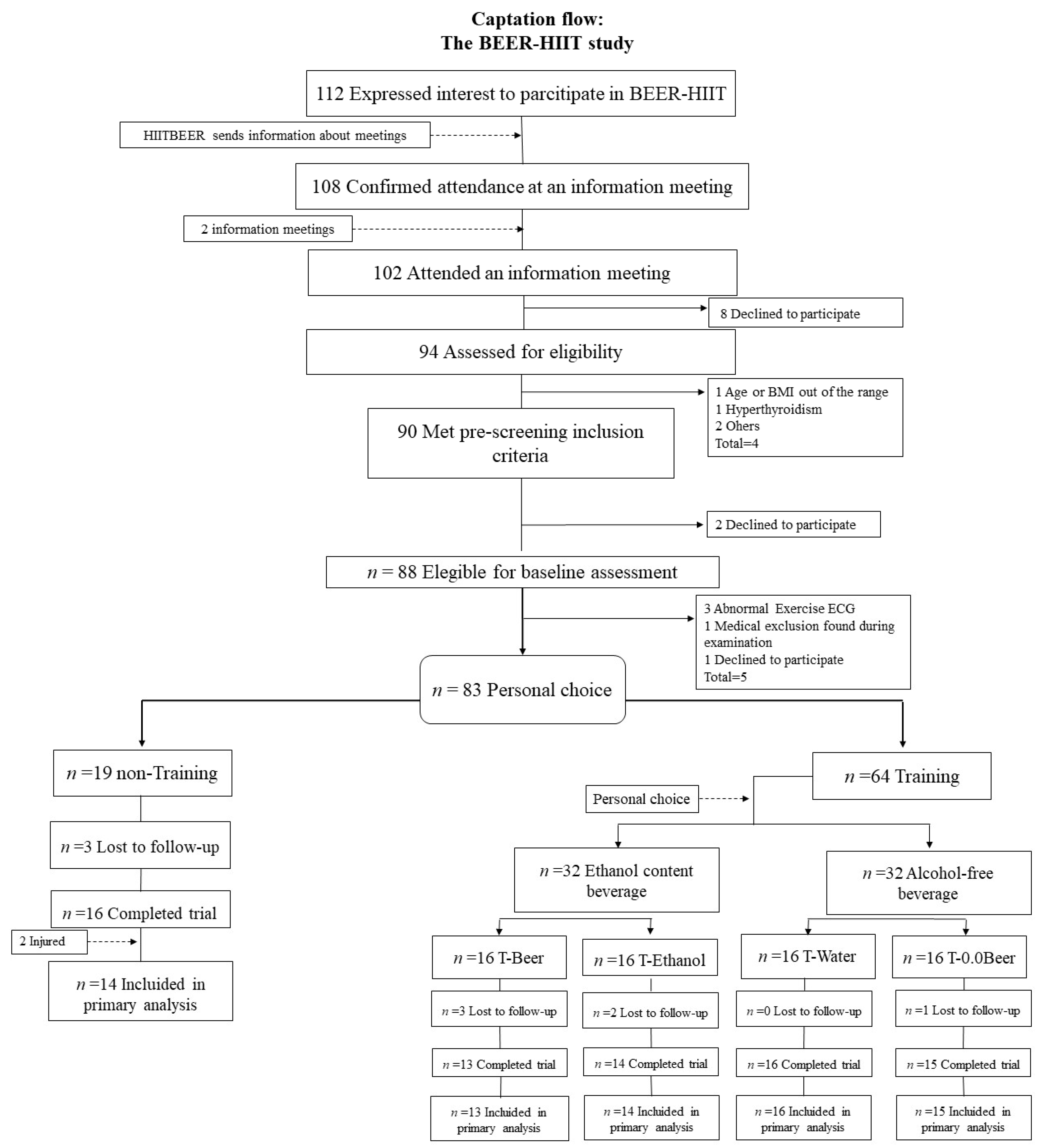
2.1. Study Design and Participants
2.2. Training Protocol
2.3. Beverage Intake Protocol
2.4. Anthropometric Parameters and Body Composition Assessment
2.5. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Keating, S.E.; Johnson, N.A.; Mielke, G.I.; Coombes, J.S. A systematic review and meta-analysis of interval training versus moderate-intensity continuous training on body adiposity. Obes. Rev. 2017, 18, 943–964. [Google Scholar] [CrossRef]
- World Health Organization (WHO). Global Recommendations on Physical Activity for Health; WHO: Geneva, Switzerland, 2010. [Google Scholar]
- Amaro Gahete, F.J.; De La O, A.; Jurado Fasoli, L.; Castillo, M.J.; Gutierrez, A. Fitness Assessment as an Anti-Aging Marker: A Narrative Review. J. Gerontol. Geriatr. Res. 2017, 6. [Google Scholar] [CrossRef]
- Trost, S.G.; Owen, N.; Bauman, A.E.; Sallis, J.F.; Brown, W. Correlates of adults’ participation in physical activity: Review and update. Med. Sci. Sports Exerc. 2002, 34, 1996–2001. [Google Scholar] [CrossRef] [PubMed]
- Wewege, M.; van den Berg, R.; Ward, R.E.; Keech, A. The effects of high-intensity interval training vs. moderate-intensity continuous training on body composition in overweight and obese adults: A systematic review and meta-analysis. Obes. Rev. 2017, 18, 635–646. [Google Scholar] [CrossRef] [PubMed]
- Brown, E.C.; Hew-Butler, T.; Marks, C.R.C.; Butcher, S.J.; Choi, M.D. The Impact of Different High-Intensity Interval Training Protocols on Body Composition and Physical Fitness in Healthy Young Adult Females. Biores. Open Access 2018, 7, 177–185. [Google Scholar] [CrossRef] [PubMed]
- Blue, M.N.M.; Smith-Ryan, A.E.; Trexler, E.T.; Hirsch, K.R. The effects of high intensity interval training on muscle size and quality in overweight and obese adults. J. Sci. Med. Sport 2018, 21, 207–212. [Google Scholar] [CrossRef]
- Maillard, F.; Pereira, B.; Boisseau, N. Effect of High-Intensity Interval Training on Total, Abdominal and Visceral Fat Mass: A Meta-Analysis. Sports Med. 2018, 48, 269–288. [Google Scholar] [CrossRef]
- Trapp, E.G.; Chisholm, D.J.; Freund, J.; Boutcher, S.H. The effects of high-intensity intermittent exercise training on fat loss and fasting insulin levels of young women. Int. J. Obes. 2008. [Google Scholar] [CrossRef]
- Smith-Ryan, A.E.; Melvin, M.N.; Wingfield, H.L. High-intensity interval training: Modulating interval duration in overweight/obese men. Phys. Sportsmed. 2015, 43, 107–113. [Google Scholar] [CrossRef]
- Heydari, M.; Freund, J.; Boutcher, S.H. The effect of high-intensity intermittent exercise on body composition of overweight young males. J. Obes. 2012, 2012. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Global Status Report on Alcohol and Health 2018. 2018. Available online: https://doi.org//entity/substance_abuse/publications/global_alcohol_report/en/index.html (accessed on 21 September 2018).
- Wijnen, A.H.C.; Steennis, J.; Catoire, M.; Wardenaar, F.C.; Mensink, M. Post-Exercise Rehydration: Effect of Consumption of Beer with Varying Alcohol Content on Fluid Balance after Mild Dehydration. Front. Nutr. 2016, 3, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Irwin, C.; Leveritt, M.; Desbrow, B. A hard earned thirst: Workplace hydration and attitudes regarding post-shift alcohol consumption. J. Health Saf. Environ. 2013, 29, 51–67. [Google Scholar]
- Jiménez-Pavón, D.; Cervantes-Borunda, M.S.; Díaz, L.E.; Marcos, A.; Castillo, M.J. Effects of a moderate intake of beer on markers of hydration after exercise in the heat: A crossover study. J. Int. Soc. Sports Nutr. 2015, 12, 1–8. [Google Scholar] [CrossRef]
- O’Brien, C.P.; Lyons, F. Alcohol and the Athlete. Sports Med. 2000, 29, 295–300. [Google Scholar] [CrossRef]
- Martens, M.P.; Dams-O’Connor, K.; Beck, N.C. A systematic review of college student-athlete drinking: Prevalence rates, sport-related factors, and interventions. J. Substain. Abuse Treat. 2006, 31, 305–316. [Google Scholar] [CrossRef]
- Desbrow, B.; Cecchin, D.; Jones, A.; Grant, G.; Irwin, C.; Leveritt, M. Manipulations to the Alcohol and Sodium Content of Beer for Postexercise Rehydration. Int. J. Sport Nutr. Exerc. Metab. 2015, 25, 262–270. [Google Scholar] [CrossRef]
- Brandhagen, M.; Forslund, H.B.; Lissner, L.; Winkvist, A.; Lindroos, A.K.; Carlsson, L.M.S.; Sjöström, L.; Larsson, I. Alcohol and macronutrient intake patterns are related to general and central adiposity. Eur. J. Clin. Nutr. 2012, 66, 305–313. [Google Scholar] [CrossRef] [PubMed]
- De Gaetano, G.; Costanzo, S.; Di Castelnuovo, A.; Badimon, L.; Bejko, D.; Alkerwi, A.; Chiva-Blanch, G.; Estruch, R.; La Vecchia, C.; Panico, S.; et al. Effects of moderate beer consumption on health and disease: A consensus document. Nutr. Metab. Cardiovasc. Dis. 2016, 26, 443–467. [Google Scholar] [CrossRef]
- Da Rocha, T.F.; Hasselmann, M.H.; Chaves Curioni, C.; Bezerra, F.F.; Faerstein, E. Alcohol consumption is associated with DXA measurement of adiposity: The Pró-Saúde Study, Brazil. Eur. J. Nutr. 2017, 56, 1983–1991. [Google Scholar] [CrossRef] [PubMed]
- Romeo, J.; González-Gross, M.; Wärnberg, J.; Díaz, L.E.; Marcos, A. Influye la cerveza en el aumento de peso?: Efectos de un consumo moderado de cerveza sobre la composición corporal. Nutrición Hospitalaria 2007, 22, 223–228. [Google Scholar]
- Poli, A.; Marangoni, F.; Avogaro, A.; Barba, G.; Bellentani, S.; Bucci, M.; Cambieri, R.; Catapano, A.L.; Costanzo, S.; Cricelli, C.; et al. Moderate alcohol use and health: A consensus document. Nutr. Metab. Cardiovasc. Dis. 2013, 23, 487–504. [Google Scholar] [CrossRef]
- Tolstrup, J.S.; Heitmann, B.L.; Tjønneland, A.M.; Overvad, O.K.; Sørensen, T.I.A.; Grønbæk, M.N.; Tolstrup, J. The relation between drinking pattern and body mass index and waist and hip circumference. Int. J. Obes. 2005, 29, 490–497. [Google Scholar] [CrossRef]
- Parr, E.B.; Camera, D.M.; Areta, J.L.; Burke, L.M.; Phillips, S.M.; Hawley, J.A.; Coffey, V.G. Alcohol Ingestion Impairs Maximal Post-Exercise Rates of Myofibrillar Protein Synthesis following a Single Bout of Concurrent Training. PLoS ONE 2014, 9, e88384. [Google Scholar] [CrossRef]
- Nissensohn, M.; López-Ufano, M.; Itandehui Castro-Quezada, L.S.-M. Valoración de la ingesta de bebidas y del estado de hidratación. Span. J. Community Nutr. 2002, 21, 58–65. [Google Scholar] [CrossRef]
- Buchheit, M.; Laursen, P.B. High-intensity interval training, solutions to the programming puzzle: Part I: Cardiopulmonary emphasis. Sports Med. 2013, 43, 313–338. [Google Scholar] [CrossRef]
- Gibala, M.J.; Little, J.P.; Macdonald, M.J.; Hawley, J.A. Physiological adaptations to low-volume, high-intensity interval training in health and disease. J. Physiol. 2012, 590, 1077–1084. [Google Scholar] [CrossRef]
- Lunt, H.; Draper, N.; Marshall, H.C.; Logan, F.J.; Hamlin, M.J.; Shearman, J.P.; Cotter, J.D.; Kimber, N.E.; Blackwell, G.; Frampton, C.M.A. High intensity interval training in a real world setting: A randomized controlled feasibility study in overweight inactive adults, measuring change in maximal oxygen uptake. PLoS ONE 2014, 9, e83256. [Google Scholar] [CrossRef]
- Amaro-Gahete, F.J.; De-La-O, A.; Jurado-Fasoli, L.; Espuch-Oliver, A.; Robles-Gonzalez, L.; Navarro-Lomas, G.; De Haro, T.; Femia, P.; Castillo, M.J.; Gutierrez, A. Exercise training as S-Klotho protein stimulator in sedentary healthy adults: Rationale, design, and methodology. Contemp. Clin. Trials Commun. 2018, 11, 10–19. [Google Scholar] [CrossRef]
- Samuelson, G. Global strategy on diet, physical activity and health. Scand. J. Nutr. 2004, 48, 57. [Google Scholar] [CrossRef]
- Sanchez-Delgado, G.; Martinez-Tellez, B.; Olza, J.; Aguilera, C.M.; Labayen, I.; Ortega, F.B.; Chillon, P.; Fernandez-Reguera, C.; Alcantara, J.M.; Martinez-Avila, W.D.; et al. Activating brown adipose tissue through exercise (ACTIBATE) in young adults: Rationale, design and methodology. Contemp. Clin. Trials 2015, 45, 416–425. [Google Scholar] [CrossRef]
- Hwang, C.-L.; Yoo, J.-K.; Kim, H.-K.; Hwang, M.-H.; Handberg, E.M.; Petersen, J.W.; Christou, D.D. Novel all-extremity high-intensity interval training improves aerobic fitness, cardiac function and insulin resistance in healthy older adults. Exp. Gerontol. 2016, 82, 112–119. [Google Scholar] [CrossRef]
- Thum, J.S.; Parsons, G.; Whittle, T.; Astorino, T.A. High-intensity interval training elicits higher enjoyment than moderate intensity continuous exercise. PLoS ONE 2017, 12, 1–11. [Google Scholar] [CrossRef]
- Borg, G.A. Psychophysical bases of perceived exertion. Med. Sci. Sports Exerc. 1982, 14, 377–381. [Google Scholar] [CrossRef]
- Gillach, M.C.; Sallis, J.F.; Buono, M.J.; Patterson, P.; Nader, P.R. The Relationship Between Perceived Exertion and Heart Rate in Children and Adults. Pediatr. Exerc. Sci. 1989, 1, 360–368. [Google Scholar] [CrossRef]
- Arena, S. Rate Perceived Exertion as a Measure of Exercise Intensity. Home Healthc. Now 2017, 35, 570. [Google Scholar] [CrossRef]
- Lamb, K.L. Children’s ratings of effort during cycle ergometry: An examination of the validity of two effort rating scales. Pediatr. Exerc. Sci. 1995, 7, 407–421. [Google Scholar] [CrossRef]
- Meister, K.A.; Whelan, E.M.; Kava, R. The Health Effects of Moderate Alcohol Intake in Humans: An Epidemiologic Review. Crit. Rev. Clin. Lab. Sci. 2000, 37, 261–296. [Google Scholar] [CrossRef]
- WHO. OMS|Obesidad y sobrepeso; WHO: Geneva, Switzerland, 2016. [Google Scholar]
- Marfell-Jones, M.J.; Stewart, A.D.; de Ridder, J.H. International Standards for Anthropometric Assessment; International Society for the Advancement of Kinanthropometry: Wellington, New Zealand, 2012. [Google Scholar]
- Shaffer, M.L.; Chinchilli, V.M. Including multiple imputation in a sensitivity analysis for clinical trials with treatment failures. Contemp. Clin. Trials 2007, 28, 130–137. [Google Scholar] [CrossRef]
- Amaro-Gahete, F.J.; De-la-O, A.; Jurado-Fasoli, L.; Ruiz, J.R.; Castillo, M.J.; Gutiérrez, Á. Effects of different exercise training programs on body composition: A randomized control trial. Scand. J. Med. Sci. Sports 2019. [Google Scholar] [CrossRef] [PubMed]
- Batacan, R.B.; Duncan, M.J.; Dalbo, V.J.; Tucker, P.S.; Fenning, A.S. Effects of high-intensity interval training on cardiometabolic health: A systematic review and meta-analysis of intervention studies. Br. J. Sports Med. 2017, 51, 494–503. [Google Scholar] [CrossRef]
- Verheggen, R.J.H.M.; Maessen, M.F.H.; Green, D.J.; Hermus, A.R.M.M.; Hopman, M.T.E.; Thijssen, D.H.T. A systematic review and meta-analysis on the effects of exercise training versus hypocaloric diet: Distinct effects on body weight and visceral adipose tissue. Obes. Rev. 2016, 17, 664–690. [Google Scholar] [CrossRef]
- Nybo, L.; Sundstrup, E.; Jakobsen, M.D.; Mohr, M.; Hornstrup, T.; Simonsen, L.; Bülow, J.; Randers, M.B.; Nielsen, J.J.; Aagaard, P.; et al. High-intensity training versus traditional exercise interventions for promoting health. Med. Sci. Sports Exerc. 2010, 42, 1951–1958. [Google Scholar] [CrossRef] [PubMed]
- Von Stengel, S.; Kemmler, W.; Lauber, D.; A Kalender, W.; Engelke, K. Differential effects of strength versus power training on bone mineral density in postmenopausal women: A 2-year longitudinal study. Br. J. Sports Med. 2007, 41, 649–655. [Google Scholar] [CrossRef]
- Harding, A.; Beck, B. Exercise, Osteoporosis, and Bone Geometry. Sports 2017, 5, 29. [Google Scholar] [CrossRef] [PubMed]
- Coulson, C.E.; Williams, L.J.; Brennan, S.L.; Berk, M.; Kotowicz, M.A.; Lubman, D.I.; Pasco, J.A. Alcohol consumption and body composition in a population-based sample of elderly Australian men. Aging Clin. Exp. Res. 2013, 183–192. [Google Scholar] [CrossRef]
- Kim, K.H.; Oh, S.-W.; Kwon, H.; Choi, H.; Park, J.-H.; Cho, B. Alcohol consumption and its relation to visceral and subcutaneous adipose tissues in healthy male Koreans. Ann. Nutr. Metab. 2012, 60, 52–61. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Lee, I.-M.; Manson, J.E.; Buring, J.E.; Sesso, H.D. Alcohol Consumption, Weight Gain, and Risk of Becoming Overweight in Middle-aged and Older Women. Arch. Intern. Med. 2010, 170, 453–461. [Google Scholar] [CrossRef]
- Schütze, M.; Schulz, M.; Steffen, A.; Bergmann, M.M.; Kroke, A.; Lissner, L.; Boeing, H.; Sch, M. Beer consumption and the ‘beer belly’: Scientific basis or common belief? Eur. J. Clin. Nutr. 2009, 63, 1143–1149. [Google Scholar] [CrossRef] [PubMed]
- Wannamethee, S.G.; Shaper, A.G.; Whincup, P.H. Alcohol and adiposity: Effects of quantity and type of drink and time relation with meals. Int. J. Obes. 2005, 29, 1436–1444. [Google Scholar] [CrossRef]
- Viana, R.B.; Naves, J.P.A.; Coswig, V.S.; De Lira, C.A.B.; Steele, J.; Fisher, J.P.; Gentil, P. Is interval training the magic bullet for fat loss? A systematic review and meta-analysis comparing moderate-intensity continuous training with high-intensity interval training (HIIT). Br. J. Sports Med. 2019. [Google Scholar] [CrossRef]
- Kukuljan, S.; Nowson, C.A.; Sanders, K.M.; Nicholson, G.C.; Seibel, M.J.; Salmon, J.; Daly, R.M. Independent and Combined Effects of Calcium-Vitamin D3 and Exercise on Bone Structure and Strength in Older Men: An 18-Month Factorial Design Randomized Controlled Trial. J. Clin. Endocrinol. Metab. 2011, 96, 955–963. [Google Scholar] [CrossRef] [PubMed]
- Sañudo, B.; Carrasco, L.; Tejero, S.; Firth, E.; De Hoyo, M.; Del Pozo-Cruz, J. A systematic review of the exercise effect on bone health. Menopause 2017, 24, 1208–1216. [Google Scholar] [CrossRef] [PubMed]
| Non-Training (n = 14) | T-Beer (n = 13) | T-0.0Beer (n = 15) | T-Water (n = 16) | T-Ethanol (n = 14) | |||||||||||||||||
| Age (years) | 20.1 (2.4) | 24.5 (5.6) | 24.5 (5.6) | 24.6 (6.6) | 26.1 (6.7) | ||||||||||||||||
| Sex (%) | |||||||||||||||||||||
| Men | 7 (50.0) | 6 (46.2) | 8 (53.3) | 9 (56.3) | 7 (50.0) | ||||||||||||||||
| Women | 7 (50.0) | 7 (53.8) | 7 (46.7) | 7 (43.8) | 7 (50.0) | ||||||||||||||||
| Beverage intake questionnaire (BVQ) | |||||||||||||||||||||
| Weekly Alcohol Intake | 5.9 (4.4) | 3.2 (3.5) | 9.3 (8.1) | 8.0 (8.2) | 5.3 (7.1) | ||||||||||||||||
| Test of Change over Time | Test of Treatment Effects | ||||||||||||||||||||
| PRE | POST | % | p | PRE | POST | % | p | PRE | POST | % | p | PRE | POST | % | p | PRE | POST | % | p | p Time*Group | |
| Anthropometric variables | |||||||||||||||||||||
| Body mass | 64.7 (10.8) | 65.0 (11.5) | 9.4 | 0.294 | 70.1 (15.6) | 70.9 (15.8) | 1.2 | 0.050 | 71.5 (16.9) | 71.9 (15.8) | 0.6 | 0.302 | 69.6 (10.1) | 70.5 (10.8) | 1.3 | 0.087 | 68.3 (14.4) | 68.8 (14.1) | 0.7 | 0.311 | 0.849 |
| BMI (kg/m2) | 22.1 (2.0) | 22.2 (2.1) | 0.5 | 0.335 | 24.0 (4.3) | 24.3 (4.3) | 1.2 | 0.048 | 24.99 (3.7) | 25.13 (3.8) | 0.6 | 0.279 | 24.86 (3.4) | 25.15 (3.5) | 1.2 | 0.088 | 24.22 (4.2) | 24.40 (4.0) | 0.7 | 0.298 | 0.842 |
| WC | 74.1 (0.1) | 75.6 (6.0) | 2.0 | 0.143 | 80.1 (11.9) | 80.5 (11.9) | 0.5 | 0.668 | 85.8 (11.5) | 80.5 (9.9) | −6.2 | 0.014 | 82.2 (10.1) | 81.8 (10.0) | −0.5 | 0.627 | 83.1 (10.1) | 81.8 (10.0) | −1.6 | 0.547 | 0.003 |
| HC | 95.4 (5.3) | 95.6 (6.0) | 0.2 | 0.846 | 97.0 (9.8) | 98.1 (9.5) | 1.1 | 0.338 | 98.8 (10.1) | 100.2 (7.4) | 1.4 | 0.444 | 99.4 (6.9) | 100.2 (6.1) | 1.4 | 0.373 | 98.6 (6.9) | 98.5 (6.8) | −0.1 | 0.945 | 0.900 |
| WHiR | 0.7 (0.1) | 0.8 (0.0) | 2.1 | 0.189 | 0.8 (0.1) | 0.7 (0.1) | −0.7 | 0.529 | 0.9 (0.2) | 0.8 (0.1) | −8.6 | 0.063 | 0.8 (0.1) | 0.7 (0.1) | −1.2 | 0.306 | 0.8 (0.11) | 0.7 (0.09) | −0.8 | 0.704 | 0.029 |
| Body composition variables | |||||||||||||||||||||
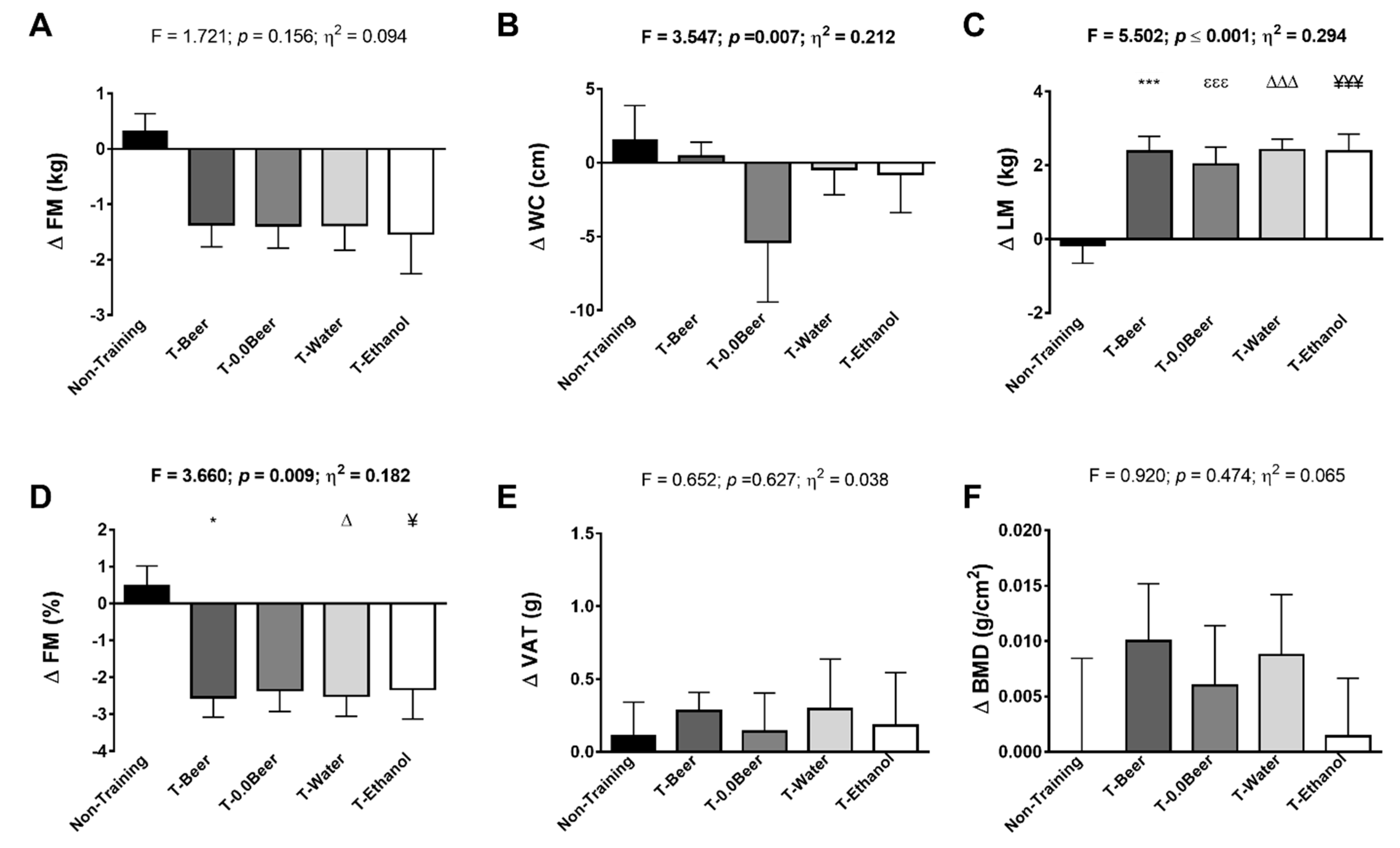
| FM (kg) | 17.0 (5.9) | 17.3 (5.7) | 1.9 | 0.327 | 20.0 (7.4) | 18.7 (7.3) | −6.8 | 0.005 | 22.4 (5.7) | 20.9 (5.9) | −6.2 | 0.004 | 22.1 (7.0) | 20.8 (6.7) | −6.2 | 0.009 | 19.9 (7.1) | 18.4 (5.5) | −7.7 | 0.056 | 0.048 |
| FM (%) | 26.93 (8.9) | 27.42 (8.7) | 1.8 | 0.378 | 29.0 (7.6) | 26.4 (7.2) | −8.8 | <0.001 | 32.2 (5.4) | 29.8 (5.6) | −7.3 | 0.001 | 32.3 (7.7) | 29.8 (6.9) | −7.7 | <0.001 | 29.7 (8.0) | 27.4 (7.2) | −7.8 | 0.014 | 0.003 |
| VAT (g) | 212.3 (69.6) | 213.5 (91.3) | 0.5 | 0.908 | 282.5 (161.2) | 254.5 (162.8) | −9.9 | 0.094 | 320.1 (154.4) | 280.8 (174.3) | −12.3 | 0.015 | 321.4 (170.5) | 283.2 (169.6) | −11.9 | 0.004 | 292.8 (189.5) | 262.8 (145.0) | −10.2 | 0.223 | 0.342 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Molina-Hidalgo, C.; De-la-O, A.; Jurado-Fasoli, L.; Amaro-Gahete, F.J.; Castillo, M.J. Beer or Ethanol Effects on the Body Composition Response to High-Intensity Interval Training. The BEER-HIIT Study. Nutrients 2019, 11, 909. https://doi.org/10.3390/nu11040909
Molina-Hidalgo C, De-la-O A, Jurado-Fasoli L, Amaro-Gahete FJ, Castillo MJ. Beer or Ethanol Effects on the Body Composition Response to High-Intensity Interval Training. The BEER-HIIT Study. Nutrients. 2019; 11(4):909. https://doi.org/10.3390/nu11040909
Chicago/Turabian StyleMolina-Hidalgo, Cristina, Alejandro De-la-O, Lucas Jurado-Fasoli, Francisco J. Amaro-Gahete, and Manuel J. Castillo. 2019. "Beer or Ethanol Effects on the Body Composition Response to High-Intensity Interval Training. The BEER-HIIT Study" Nutrients 11, no. 4: 909. https://doi.org/10.3390/nu11040909
APA StyleMolina-Hidalgo, C., De-la-O, A., Jurado-Fasoli, L., Amaro-Gahete, F. J., & Castillo, M. J. (2019). Beer or Ethanol Effects on the Body Composition Response to High-Intensity Interval Training. The BEER-HIIT Study. Nutrients, 11(4), 909. https://doi.org/10.3390/nu11040909







