Associations of Dietary Riboflavin, Niacin, and Retinol with Age-related Hearing Loss: An Analysis of Korean National Health and Nutrition Examination Survey Data
Abstract
1. Introduction
2. Materials and Methods
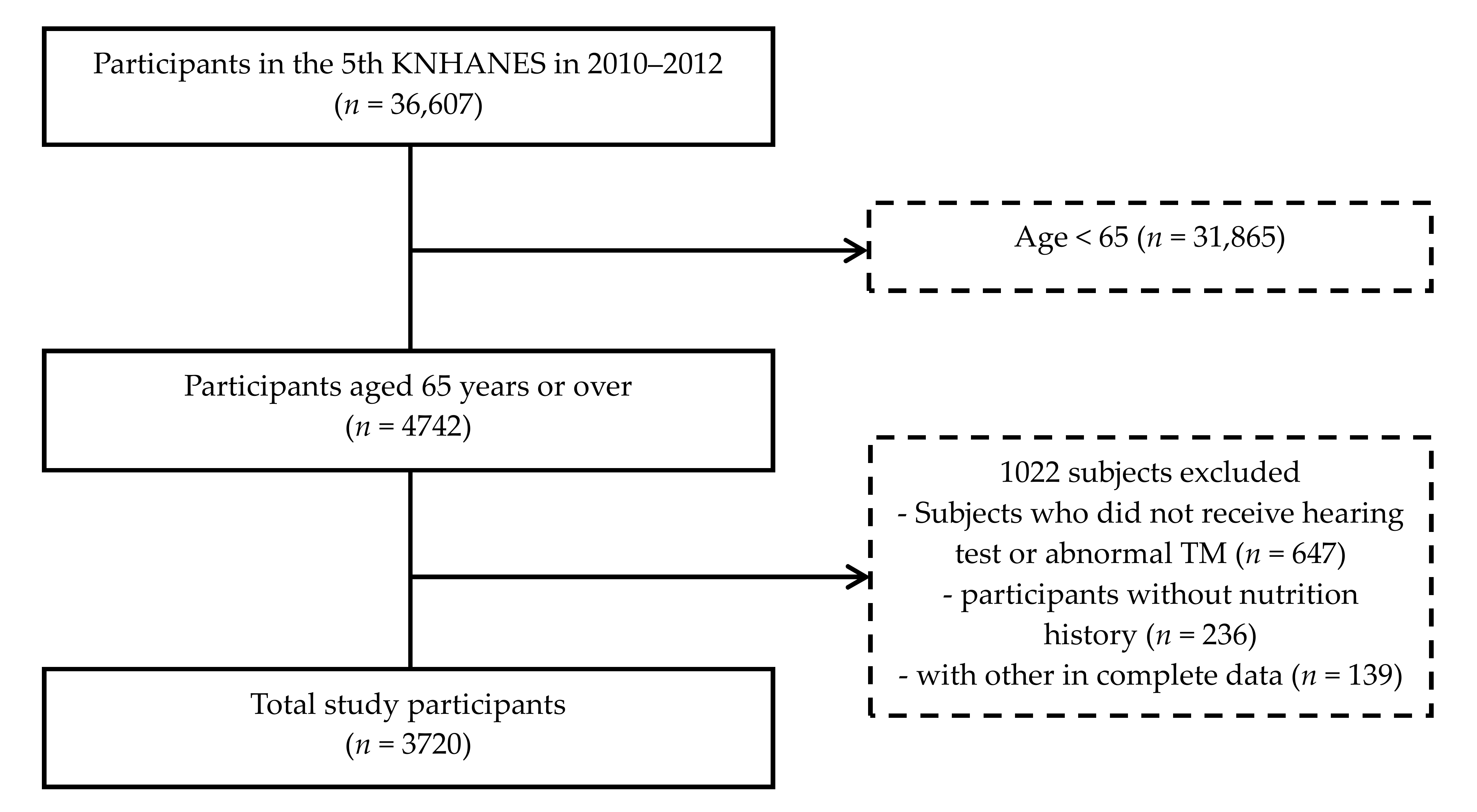
2.1. Study Participant
2.2. Otologic Examination and Audiologic Evaluation
2.3. Evaluation of Nutrition Intake
2.4. Statistical Analyses
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Seidman, M.D.; Ahmad, N.; Bai, U. Molecular mechanisms of age-related hearing loss. Ageing Res. Rev. 2002, 1, 331–343. [Google Scholar] [CrossRef]
- WHO. Global Estimates on Prevalence of Hearing Loss; World Health Organization: Geneva, Switzerland, 2012; Available online: http://www.who.int/pbd/deafness/WHO_GE_HL.pdf (accessed on 20 April 2019).
- Lin, F.R.; Yaffe, K.; Xia, J.; Xue, Q.L.; Harris, T.B.; Purchase-Helzner, E.; Satterfield, S.; Ayonayon, H.N.; Ferrucci, L.; Simonsick, E.M.; et al. Hearing loss and cognitive decline in older adults. JAMA Intern. Med. 2013, 173, 293–299. [Google Scholar] [CrossRef] [PubMed]
- Peracino, A. Hearing loss and dementia in the aging population. Audiol. Neurotol. 2014, 19 (Suppl. S1), 6–9. [Google Scholar] [CrossRef] [PubMed]
- Deal, J.A.; Sharrett, A.R.; Albert, M.S.; Coresh, J.; Mosley, T.H.; Knopman, D.; Wruck, L.M.; Lin, F.R. Hearing impairment and cognitive decline: A pilot study conducted within the atherosclerosis risk in communities neurocognitive study. Am. J. Epidemiol. 2015, 181, 680–690. [Google Scholar] [CrossRef] [PubMed]
- Gomaa, M.A.; Elmagd, M.H.; Elbadry, M.M.; Kader, R.M. Depression, anxiety and stress scale in patients with tinnitus and hearing loss. Eur. Arch. Otorhinolaryngol. 2014, 271, 2177–2184. [Google Scholar] [CrossRef] [PubMed]
- Brown, C.S.; Emmett, S.D.; Robler, S.K.; Tucci, D.L. Global hearing loss prevention. Otolaryngol. Clin. N. Am. 2018, 51, 575–592. [Google Scholar] [CrossRef] [PubMed]
- Jung, S.Y.; Kim, S.H.; Yeo, S.G. Association of nutritional factors with hearing loss. Nutrients 2019, 11, 307. [Google Scholar] [CrossRef] [PubMed]
- De Toro-Martin, J.; Arsenault, B.J.; Despres, J.P.; Vohl, M.C. Precision nutrition: A review of personalized nutritional approaches for the prevention and management of metabolic syndrome. Nutrients 2017, 9, 913. [Google Scholar] [CrossRef]
- Kim, T.S.; Kim, E.H.; Chung, J.W. The association between age-related hearing impairment and metabolic syndrome in Korean women: 5-year follow-up observational study. Metab. Syndr. Relat. Disord. 2017, 15, 240–245. [Google Scholar] [CrossRef] [PubMed]
- Kang, J.W.; Choi, H.S.; Kim, K.; Choi, J.Y. Dietary vitamin intake correlates with hearing thresholds in the older population: The Korean national health and nutrition examination survey. Am. J. Clin. Nutr. 2014, 99, 1407–1413. [Google Scholar] [CrossRef]
- Kim, S.Y.; Sim, S.; Kim, H.J.; Choi, H.G. Low-fat and low-protein diets are associated with hearing discomfort among the elderly of Korea. Br. J. Nutr. 2015, 114, 1711–1717. [Google Scholar] [CrossRef]
- Ries, P.W. Prevalence and characteristics of persons with hearing trouble: United States, 1990–1991. Vital Health Stat. Ser. 10 Data Natl. Health Surv. 1994, 188, 1–75. [Google Scholar]
- National Rural Resources Development Institute. Food composition table. Rural Res. Dev. Admin. 2006, 7, 1–453. [Google Scholar]
- Jung, K.I.; Kim, Y.C.; Park, C.K. Dietary niacin and open-angle glaucoma: The Korean national health and nutrition examination survey. Nutrients 2018, 10, 387. [Google Scholar] [CrossRef] [PubMed]
- Ministry of Health & Welfare. Dietary reference intakes for Koreans 2015. 2015, pp. 404–432. Available online: http://www.mohw.go.kr/react/modules/download.jsp?BOARD_ID=320&CONT_SEQ=337356&FILE_SEQ=199484 (accessed on 20 April 2019).
- Michikawa, T.; Nishiwaki, Y.; Kikuchi, Y.; Hosoda, K.; Mizutari, K.; Saito, H.; Asakura, K.; Milojevic, A.; Iwasawa, S.; Nakano, M.; et al. Serum levels of retinol and other antioxidants for hearing impairment among Japanese older adults. J. Gerontol. Ser. A Biol. Sci. Med Sci. 2009, 64, 910–915. [Google Scholar] [CrossRef]
- Gopinath, B.; Flood, V.M.; McMahon, C.M.; Burlutsky, G.; Spankovich, C.; Hood, L.J.; Mitchell, P. Dietary antioxidant intake is associated with the prevalence but not incidence of age-related hearing loss. J. Nutr. Health Aging 2011, 15, 896–900. [Google Scholar] [CrossRef]
- Peneau, S.; Jeandel, C.; Dejardin, P.; Andreeva, V.A.; Hercberg, S.; Galan, P.; Kesse-Guyot, E.; Group, S.V.M.R. Intake of specific nutrients and foods and hearing level measured 13 years later. Br. J. Nutr. 2013, 109, 2079–2088. [Google Scholar] [CrossRef]
- Shargorodsky, J.; Curhan, S.G.; Eavey, R.; Curhan, G.C. A prospective study of vitamin intake and the risk of hearing loss in men. Otolaryngol. Head Neck Surg. 2010, 142, 231–236. [Google Scholar] [CrossRef]
- Chole, R.A.; Quick, C.A. Estimate of vitamin a in the guinea pig cochlea. Ann. Otol. Rhinol. Laryngol. 1978, 87, 380–382. [Google Scholar] [CrossRef] [PubMed]
- Romand, R. The roles of retinoic acid during inner ear development. Curr. Top. Dev. Biol. 2003, 57, 261–291. [Google Scholar] [PubMed]
- Ahn, J.H.; Kang, H.H.; Kim, Y.J.; Chung, J.W. Anti-apoptotic role of retinoic acid in the inner ear of noise-exposed mice. Biochem. Biophys. Res. Commun. 2005, 335, 485–490. [Google Scholar] [CrossRef]
- Lefebvre, P.P.; Malgrange, B.; Staecker, H.; Moonen, G.; Van de Water, T.R. Retinoic acid stimulates regeneration of mammalian auditory hair cells. Science 1993, 260, 692–695. [Google Scholar] [CrossRef] [PubMed]
- Iwanaga, K.; Hasegawa, T.; Hultquist, D.E.; Harada, H.; Yoshikawa, Y.; Yanamadala, S.; Liao, H.; Visovatti, S.H.; Pinsky, D.J. Riboflavin-mediated reduction of oxidant injury, rejection, and vasculopathy after cardiac allotransplantation. Transplantation 2007, 83, 747–753. [Google Scholar] [CrossRef]
- Bjelakovic, G.; Nikolova, D.; Gluud, L.L.; Simonetti, R.G.; Gluud, C. Mortality in randomized trials of antioxidant supplements for primary and secondary prevention: Systematic review and meta-analysis. JAMA 2007, 297, 842–857. [Google Scholar] [CrossRef]
- Thakur, K.; Tomar, S.K.; Singh, A.K.; Mandal, S.; Arora, S. Riboflavin and health: A review of recent human research. Crit. Rev. Food Sci. Nutr. 2017, 57, 3650–3660. [Google Scholar] [CrossRef]
- Dringen, R.; Gutterer, J.M.; Hirrlinger, J. Glutathione metabolism in brain metabolic interaction between astrocytes and neurons in the defense against reactive oxygen species. Eur. J. Biochem. 2000, 267, 4912–4916. [Google Scholar] [CrossRef]
- Pompella, A.; Visvikis, A.; Paolicchi, A.; De Tata, V.; Casini, A.F. The changing faces of glutathione, a cellular protagonist. Biochem. Pharmacol. 2003, 66, 1499–1503. [Google Scholar] [CrossRef]
- Cui, X.; Chopp, M.; Zacharek, A.; Roberts, C.; Buller, B.; Ion, M.; Chen, J. Niacin treatment of stroke increases synaptic plasticity and axon growth in rats. Stroke 2010, 41, 2044–2049. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, N.; Schulze, J.; Warwas, D.P.; Ehlert, N.; Lenarz, T.; Warnecke, A.; Behrens, P. Long-term delivery of brain-derived neurotrophic factor (bdnf) from nanoporous silica nanoparticles improves the survival of spiral ganglion neurons in vitro. PLoS ONE 2018, 13, e0194778. [Google Scholar] [CrossRef] [PubMed]
- Kaplon, R.E.; Gano, L.B.; Seals, D.R. Vascular endothelial function and oxidative stress are related to dietary niacin intake among healthy middle-aged and older adults. J. Appl. Physiol. 2014, 116, 156–163. [Google Scholar] [CrossRef]
- Quaranta, N.; De Ceglie, V.; D’Elia, A. Endothelial dysfunction in idiopathic sudden sensorineural hearing loss: A review. Audiol. Res. 2016, 6, 151. [Google Scholar] [CrossRef] [PubMed]
- Spankovich, C.; Hood, L.J.; Silver, H.J.; Lambert, W.; Flood, V.M.; Mitchell, P. Associations between diet and both high and low pure tone averages and transient evoked otoacoustic emissions in an older adult population-based study. J. Am. Acad. Audiol. 2011, 22, 49–58. [Google Scholar] [CrossRef] [PubMed]
- Spankovich, C.; Le Prell, C.G. Healthy diets, healthy hearing: National health and nutrition examination survey, 1999–2002. Int. J. Audiol. 2013, 52, 369–376. [Google Scholar] [CrossRef]
- Lee, S.K.; Sobal, J. Socio-economic, dietary, activity, nutrition and body weight transitions in South Korea. Public Health Nutr 2003, 6, 665–674. [Google Scholar] [CrossRef] [PubMed]
- Choi, H.J.; Joung, H.; Lee, H.J.; Jang, H.B.; Kang, J.H.; Song, J. The influence of dietary patterns on the nutritional profile in a korean child cohort study. Osong Public Health Res. Perspect. 2011, 2, 59–64. [Google Scholar] [CrossRef] [PubMed]
| Characteristics | Bilateral Age-Related Hearing Loss (≥40 dB) | p Value | |
|---|---|---|---|
| No (n = 3055) | Yes (n = 665) | ||
| Age | 71.78 ± 4.83 | 75.40 ± 5.99 | <0.001 |
| Gender | <0.001 | ||
| Male | 1298 (42.5) | 337 (50.7) | |
| Female | 1757 (57.5) | 328 (49.3) | |
| BMI (kg/m²) | <0.001 | ||
| Underweight (<18.5) | 106 (3.5) | 41 (6.2) | |
| Normal (≥18.5, <23) | 1873 (61.3) | 458 (68.9) | |
| Over (≥23) | 1076 (35.2) | 166 (24.9) | |
| Smoking | 0.0023 | ||
| Never | 1869 (61.2) | 364 (54.7) | |
| Former | 843 (27.6) | 200 (30.1) | |
| Current | 343 (11.2) | 101 (15.2) | |
| Alcohol consumption | <0.001 | ||
| Non | 1523 (49.9) | 387 (58.2) | |
| in month<1 | 482 (15.8) | 70 (10.5) | |
| 1≤in month≤4 | 535 (17.5) | 103 (15.5) | |
| 2≤in week | 515 (16.8) | 105 (15.8) | |
| Diabetes | 0.87 | ||
| No | 2475 (81.0) | 537 (80.8) | |
| Yes | 580 (19.0) | 128 (19.2) | |
| Hypertension | 0.197 | ||
| No | 1441 (47.2) | 332 (49.9) | |
| Yes | 1614 (52.8) | 333 (50.1) | |
| Nutrient | Quartile 1 | Quartile 2 | Quartile 3 | Quartile 4 |
|---|---|---|---|---|
| Total energy intake (kcal) | <1239.64 | 1239.64–1599.27 | 1599.28–2028.59 | 2028.60≤ |
| Carbohydrate (g/day) | <229.03 | 229.03–296.03 | 296.04–367.13 | 367.14≤ |
| Fat (g/day) | <10.16 | 10.16–17.89 | 17.90–31.13 | 31.14≤ |
| Protein (g/day) | <35.03 | 35.03–48.96 | 48.97–68.07 | 68.08≤ |
| Fiber (g/day) | <3.81 | 3.81–5.76 | 5.77–8.69 | 8.70≤ |
| Ash (g/day) | <9.74 | 9.74–14.54 | 14.55–21.07 | 21.08≤ |
| Calcium (mg/day) | <217.14 | 217.14–350.01 | 350.02–543.39 | 543.40≤ |
| Iron (mg/day) | <6.85 | 6.85–10.58 | 10.59–16.71 | 16.72≤ |
| Potassium (mg/day) | <1601.38 | 1601.38–2289.28 | 2289.29–3224.92 | 3224.93≤ |
| Sodium (mg/day) | <2104.36 | 2104.36–3376.17 | 3376.18–5195.31 | 5195.32≤ |
| Phosphorus (mg/day) | <683.91 | 683.91–920.32 | 920.33–1217.36 | 1217.37≤ |
| Retinol (μg/day) | <3.22 | 3.22–15.51 | 15.52–60.55 | 60.56≤ |
| β-carotene (μg/day) | <1020.92 | 1020.92–2237.26 | 2237.27–4412.40 | 4412.41≤ |
| Vitamin A (μg Retinol Equivalents /day) | <208.76 | 208.76–426.34 | 426.35–816.56 | 816.57≤ |
| Thiamin (mg/day) | <0.64 | 0.64–0.90 | 0.91–1.29 | 1.30≤ |
| Riboflavin (mg/day) | <0.50 | 0.50–0.74 | 0.75–1.13 | 1.14≤ |
| Niacin (mg/day) | <8.26 | 8.26–11.59 | 11.60–16.35 | 16.36≤ |
| Vitamin C (mg/day) | <36.17 | 36.17–66.36 | 66.37–114.93 | 114.94≤ |
| Nutrient | Univariable Analysis | Multivariable Analysis | Nutrient | Univariable Analysis | Multivariable Analysis | ||||
|---|---|---|---|---|---|---|---|---|---|
| OR (95% CI) | p | OR (95% CI) | p | OR (95% CI) | p | OR (95% CI) | p | ||
| Total Energy | Retinol | ||||||||
| Q1 | Reference | Reference | Q1 | Reference | Reference | ||||
| Q2 | 0.77(0.61–0.98) | 0.030 * | 0.82(0.63–1.06) | 0.132 | Q2 | 0.97(0.78–1.22) | 0.819 | 1.05(0.82–1.34) | 0.700 |
| Q3 | 0.80(0.63–1.00) | 0.054 | 0.87(0.67–1.13) | 0.285 | Q3 | 0.72(0.59–0.91) | 0.006 * | 0.79(0.61–1.02) | 0.075 |
| Q4 | 0.66(0.52–0.84) | 0.001 * | 0.80(0.61–1.06) | 0.125 | Q4 | 0.61(0.48–0.78) | 0.001 * | 0.66(0.51–0.86) | 0.002 * |
| Carbohydrate | β-carotene | ||||||||
| Q1 | Reference | Reference | Q1 | Reference | Reference | ||||
| Q2 | 0.76(0.60–0.97) | 0.024 * | 0.88(0.68–1.14) | 0.321 | Q2 | 1.10(0.88–1.39) | 0.412 | 1.23(0.96–1.58) | 0.105 |
| Q3 | 0.86(0.68–1.08) | 0.198 | 0.92(0.71–1.19) | 0.527 | Q3 | 0.90(0.71–1.14) | 0.364 | 1.08(0.83–1.39) | 0.576 |
| Q4 | 0.73(0.58–0.93) | 0.010 * | 0.93(0.71–1.22) | 0.588 | Q4 | 0.80(0.63–1.01) | 0.065 | 1.00(0.77–1.31) | 0.981 |
| Fat | Vitamin A | ||||||||
| Q1 | Reference | Reference | Q1 | Reference | Reference | ||||
| Q2 | 0.78(0.62–0.99) | 0.037 * | 0.92(0.72–1.18) | 0.522 | Q2 | 0.95(0.76–1.20) | 0.683 | 1.03(0.80–1.32) | 0.828 |
| Q3 | 0.73(0.58–0.92) | 0.007 * | 0.81(0.62–1.04) | 0.102 | Q3 | 0.83(0.65–1.04) | 0.108 | 0.99(0.77–1.28) | 0.915 |
| Q4 | 0.62(0.49–0.79) | 0.001 * | 0.76(0.58–1.01) | 0.052 | Q4 | 0.72(0.56–0.91) | 0.006 * | 0.86(0.66–1.12) | 0.270 |
| Protein | Thiamin | ||||||||
| Q1 | Reference | Reference | Q1 | Reference | Reference | ||||
| Q2 | 0.86(0.68–1.08) | 0.185 | 0.99(0.77–1.27) | 0.919 | Q2 | 0.87(0.70–1.09) | 0.228 | 0.98(0.76–1.25) | 0.845 |
| Q3 | 0.71(0.56–0.90) | 0.005 * | 0.87(0.67–1.14) | 0.312 | Q3 | 0.66(0.53–0.84) | 0.001 * | 0.78(0.60–1.02) | 0.070 |
| Q4 | 0.63(0.49–0.80) | 0.001 * | 0.75(0.57–1.01) | 0.053 | Q4 | 0.64(0.51–0.82) | 0.001 * | 0.82(0.62–1.08) | 0.152 |
| Fiber | Riboflavin | ||||||||
| Q1 | Reference | Reference | Q1 | Reference | Reference | ||||
| Q2 | 0.85(0.68–1.07) | 0.175 | .97(0.75–1.25) | 0.798 | Q2 | 0.82(0.65–1.03) | 0.085 | 0.91(0.71–1.17) | 0.459 |
| Q3 | 0.91(0.72–1.14) | 0.413 | 1.11(0.86–1.43) | 0.418 | Q3 | 0.72(0.58–0.91) | 0.006 * | 0.86(0.67–1.11) | 0.258 |
| Q4 | 0.67(0.52–0.85) | 0.001 * | .89(0.68–1.17) | 0.404 | Q4 | 0.56(0.44–0.71) | 0.001 * | 0.71(0.54–0.94) | 0.016 * |
| ASH | Niacin | ||||||||
| Q1 | Reference | Reference | Q1 | Reference | Reference | ||||
| Q2 | 0.80(0.64–1.00) | 0.051 | 0.87(0.68–1.12) | 0.278 | Q2 | 1.03(0.83–1.30) | 0.775 | 1.19(0.93–1.42) | 0.174 |
| Q3 | 0.64(0.61–0.81) | 0.001 * | 0.76(0.58–0.01) | 0.054 | Q3 | 0.77(0.61–0.98) | 0.032 * | 0.95(0.73–1.23) | 0.675 |
| Q4 | 0.62(0.49–0.79) | 0.001 * | 0.77(0.59–1.01) | 0.062 | Q4 | 0.59(0.46–0.76) | 0.001 * | 0.72(0.54–0.96) | 0.025 * |
| Calcium | Vitamin C | ||||||||
| Q1 | Reference | Reference | Q1 | Reference | Reference | ||||
| Q2 | 0.74(0.59–0.94) | 0.012 * | 0.91(0.70–1.17) | 0.439 | Q2 | 0.83(0.66–1.05) | 0.115 | 0.97(0.75–1.24) | 0.790 |
| Q3 | 0.73(0.58–0.92) | 0.007 * | 0.95(0.73–1.23) | 0.679 | Q3 | 0.81(0.65–1.02) | 0.079 | 0.99(0.77–1.28) | 0.945 |
| Q4 | 0.68(0.54–0.86) | 0.001 * | 0.88(0.67–1.14) | 0.327 | Q4 | 0.63(0.50–0.80) | 0.001 * | 0.83(0.64–1.09) | 0.180 |
| Iron | Potassium | ||||||||
| Q1 | Reference | Reference | Q1 | Reference | Reference | ||||
| Q2 | 0.79(0.62–0.99) | 0.040 * | 0.88(0.68–1.13) | 0.314 | Q2 | 0.88(0.70–1.10) | 0.252 | 0.99(0.78–1.28) | 0.978 |
| Q3 | 0.74(0.59–0.93) | 0.011 * | 0.89(0.68–1.15) | 0.357 | Q3 | 0.72(0.57–0.90) | 0.005 * | 0.91(0.70–1.17) | 0.454 |
| Q4 | 0.71(0.56–0.89) | 0.004 * | 0.88(0.68–1.14) | 0.330 | Q4 | 0.57(0.45–0.72) | 0.001 * | 0.79(0.60–1.04) | 0.091 |
| Sodium | Phosphorus | ||||||||
| Q1 | Reference | Reference | Q1 | Reference | Reference | ||||
| Q2 | 0.88(0.70–1.10) | 0.263 | 0.96(0.75–1.24) | 0.754 | Q2 | 0.97(0.78–1.21) | 0.776 | 1.12(0.88–1.43) | 0.365 |
| Q3 | 0.86(0.69–1.09) | 0.216 | 0.96(0.74–1.24) | 0.730 | Q3 | 0.67(0.53–0.85) | 0.001 * | 0.77(0.59–1.01) | 0.055 |
| Q4 | 0.71(0.56–091) | 0.004 * | 0.85(0.65–1.12) | 0.251 | Q4 | 0.59(0.46–0.75) | 0.001 * | 0.76(0.57–1.01) | 0.055 |
| Nutrient | Univariable Analysis | Multivariable Analysis | Nutrient | Univariable Analysis | Multivariable Analysis | ||||
|---|---|---|---|---|---|---|---|---|---|
| OR (95% CI) | p | OR (95% CI) | p | OR (95% CI) | p | OR (95% CI) | p | ||
| Total Energy | Retinol | ||||||||
| Q1 | Reference | Reference | Q1 | Reference | Reference | ||||
| Q2 | 1.07(0.84–1.35) | 0.587 | 1.03(0.81–1.32) | 0.783 | Q2 | 0.96(0.77–1.21) | 0.726 | 0.96(0.76–1.21) | 0.741 |
| Q3 | 1.03(0.81–1.31) | 0.808 | 1.02(0.79–1.31) | 0.893 | Q3 | 0.74(0.59–0.95) | 0.015 * | 0.83(0.57–1.03) | 0.054 |
| Q4 | 1.06(0.84–1.34) | 0.629 | 1.02(0.78–1.33) | 0.895 | Q4 | 0.86(0.68–1.09) | 0.213 | 0.88(0.69–1.11) | 0.281 |
| Carbohydrate | β-carotene | ||||||||
| Q1 | Reference | Reference | Q1 | Reference | Reference | ||||
| Q2 | 1.21(0.95–1.54) | 0.116 | 1.20(0.94–1.54) | 0.145 | Q2 | 0.87(0.69–1.10) | 0.240 | 0.86(0.68–1.09) | 0.212 |
| Q3 | 1.13(0.89–1.43) | 0.327 | 1.12(0.87–1.44) | 0.364 | Q3 | 0.83(0.66–1.04) | 0.110 | 0.85(0.67–1.07) | 0.166 |
| Q4 | 1.15(0.91–1.46) | 0.246 | 1.15(0.89–1.49) | 0.284 | Q4 | 0.77(0.61–0.98) | 0.031 * | 0.79(0.62–1.01) | 0.059 |
| Fat | Vitamin A | ||||||||
| Q1 | Reference | Reference | Q1 | Reference | Reference | ||||
| Q2 | 0.96(0.76–1.21) | 0.721 | 0.92(0.73–1.17) | 0.510 | Q2 | 0.91(0.73–1.15) | 0.446 | 0.90(0.71–1.14) | 0.378 |
| Q3 | 0.88(0.69–1.11) | 0.278 | 0.85(0.67–1.09) | 0.206 | Q3 | 0.81(0.64–1.03) | 0.084 | 0.83(0.65–1.06) | 0.134 |
| Q4 | 0.92(0.73–1.17) | 0.511 | 0.90(0.70–1.16) | 0.432 | Q4 | 0.79(0.62–0.99) | 0.048 * | 0.80(0.63–1.03) | 0.080 |
| Protein | Thiamin | ||||||||
| Q1 | Reference | Reference | Q1 | Reference | Reference | ||||
| Q2 | 0.85(0.67–1.08) | 0.185 | 0.82(0.64–1.05) | 0.119 | Q2 | 1.19(0.94–1.50) | 0.152 | 1.20(0.94–1.53) | 0.143 |
| Q3 | 0.95(0.76–1.20) | 0.678 | 0.93(0.73–1.18) | 0.535 | Q3 | 1.05(0.83–1.34) | 0.670 | 1.05(0.82–1.35) | 0.693 |
| Q4 | 0.91(0.72–1.14) | 0.403 | 0.88(0.68–1.13) | 0.310 | Q4 | 1.02(0.80–1.29) | 0.902 | 1.01(0.78–1.31) | 0.936 |
| Fiber | Riboflavin | ||||||||
| Q1 | Reference | Reference | Q1 | Reference | Reference | ||||
| Q2 | 1.02(0.81–1.29) | 0.860 | 0.97(0.76–1.23) | 0.783 | Q2 | 0.88(0.70–1.11) | 0.289 | 0.88(0.70–1.12) | 0.309 |
| Q3 | 0.85(0.67–1.07) | 0.164 | 0.84(0.66–1.08) | 0.179 | Q3 | 0.78(0.62–0.99) | 0.042 * | 0.78(0.61–1.01) | 0.057 |
| Q4 | 0.87(0.69–1.10) | 0.253 | 1.04(0.81–1.33) | 0.783 | Q4 | 0.85(0.67–1.07) | 0.173 | 0.85(0.66–1.09) | 0.189 |
| ASH | Niacin | ||||||||
| Q1 | Reference | Reference | Q1 | Reference | Reference | ||||
| Q2 | 0.96(0.76–1.21) | 0.720 | 0.97(0.76–1.23) | 0.783 | Q2 | 0.96(0.76–1.21) | 0.718 | 0.92(0.72–1.18) | 0.505 |
| Q3 | 0.84(0.66–1.07) | 0.162 | 0.84(0.66–1.08) | 0.179 | Q3 | 1.01(0.80–1.28) | 0.905 | 1.03(0.80–1.31) | 0.839 |
| Q4 | 1.01(0.80–1.28) | 0.906 | 1.04(0.81–1.33) | 0.783 | Q4 | 0.95(0.75–1.20) | 0.673 | 0.92(0.71–1.19) | 0.510 |
| Calcium | Vitamin C | ||||||||
| Q1 | Reference | Reference | Q1 | Reference | Reference | ||||
| Q2 | 0.93(0.74–1.17) | 0.518 | 0.91(0.72–1.16) | 0.438 | Q2 | 0.83(0.66–1.04) | 0.110 | 0.82(0.65–1.05) | 0.109 |
| Q3 | 0.76(0.60–0.96) | 0.022 * | 0.75(0.58–1.03) | 0.054 | Q3 | 0.85(0.67–1.07) | 0.157 | 0.84(0.66–1.07) | 0.148 |
| Q4 | 0.88(0.70–1.11) | 0.287 | 0.89(0.69–1.13) | 0.334 | Q4 | 0.80(0.63–1.01) | 0.057 | 0.81(0.63–1.03) | 0.087 |
| Iron | Potassium | ||||||||
| Q1 | Reference | Reference | Q1 | Reference | Reference | ||||
| Q2 | 0.78(0.62–0.98) | 0.034 * | 0.86(0.60–1.04) | 0.065 | Q2 | 0.87(0.69–1.10) | 0.257 | 0.92(0.72–1.16) | 0.470 |
| Q3 | 0.70(0.56–0.89) | 0.003 * | 0.80(0.55–1.03) | 0.056 | Q3 | 0.89(0.70–1.12) | 0.312 | 0.88(0.69–1.13) | 0.304 |
| Q4 | 0.84(0.67–1.05) | 0.130 | 0.83(0.65–1.06) | 0.135 | Q4 | 0.87(0.69–1.10) | 0.257 | 0.89(0.69–1.15) | 0.364 |
| Sodium | Phosphorus | ||||||||
| Q1 | Reference | Reference | Q1 | Reference | Reference | ||||
| Q2 | 0.78(0.62–0.98) | 0.034 * | 1.02(0.80–1.30) | 0.903 | Q2 | 0.91(0.72–1.15) | 0.435 | 0.90(0.71–1.15) | 0.398 |
| Q3 | 0.70(0.56–0.89) | 0.003 * | 0.99(0.77–1.27) | 0.931 | Q3 | 0.98(0.78–1.24) | 0.859 | 0.97(0.77–1.24) | 0.787 |
| Q4 | 0.84(0.67–1.05) | 0.130 | 1.07(0.83–1.38) | 0.587 | Q4 | 0.90(0.71–1.14) | 0.367 | 0.88(0.68–1.14) | 0.320 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, T.S.; Chung, J.W. Associations of Dietary Riboflavin, Niacin, and Retinol with Age-related Hearing Loss: An Analysis of Korean National Health and Nutrition Examination Survey Data. Nutrients 2019, 11, 896. https://doi.org/10.3390/nu11040896
Kim TS, Chung JW. Associations of Dietary Riboflavin, Niacin, and Retinol with Age-related Hearing Loss: An Analysis of Korean National Health and Nutrition Examination Survey Data. Nutrients. 2019; 11(4):896. https://doi.org/10.3390/nu11040896
Chicago/Turabian StyleKim, Tae Su, and Jong Woo Chung. 2019. "Associations of Dietary Riboflavin, Niacin, and Retinol with Age-related Hearing Loss: An Analysis of Korean National Health and Nutrition Examination Survey Data" Nutrients 11, no. 4: 896. https://doi.org/10.3390/nu11040896
APA StyleKim, T. S., & Chung, J. W. (2019). Associations of Dietary Riboflavin, Niacin, and Retinol with Age-related Hearing Loss: An Analysis of Korean National Health and Nutrition Examination Survey Data. Nutrients, 11(4), 896. https://doi.org/10.3390/nu11040896





