Dietary Intake of Essential, Toxic, and Potentially Toxic Elements from Mussels (Mytilus spp.) in the Spanish Population: A Nutritional Assessment
Abstract
1. Introduction
2. Materials and Methods
2.1. Sampling and Collection
2.2. Standards and Elements
2.3. Analytical Procedure
2.4. Dietary Intake Estimates, Nutritional and Health Risk Assessment
2.5. Statistical Analysis
3. Results
3.1. Occurrence of Essential, Toxic, and Potentially Toxic Elements in Mussels
3.2. Estimated Daily Intake of Essential and Toxic Elements and Risk Assessment
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Oehlenschlager, J. Seafood: Nutritional benefits and risk aspects. Int. J. Vitam. Nutr. Res. 2012, 82, 168–176. [Google Scholar] [CrossRef] [PubMed]
- Arnich, N.; Sirot, V.; Rivière, G.; Jean, J.; Noël, L.; Guérin, T.; Leblanc, J.-C. Dietary exposure to trace elements and health risk assessment in the 2nd French Total Diet Study. Food Chem. Toxicol. 2012, 50, 2432–2449. [Google Scholar] [CrossRef] [PubMed]
- MAGRAMA (Ministerio de Agricultura, Alimentación y Medio Ambiente). El mercado del mejillón en España; MAGRAMA: Madrid, Spain, 2015; pp. 1–29.
- Chasapis, C.T.; Loutsidou, A.C.; Spiliopoulou, C.A.; Stefanidou, M.E. Zinc and human health: An update. Arch. Toxicol. 2012, 86, 521–534. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Hernández, Á.; Camacho, M.; Henríquez-Hernández, L.A.; Boada, L.D.; Valerón, P.F.; Zaccaroni, A.; Zumbado, M.; Almeida-González, M.; Rial-Berriel, C.; Luzardo, O.P. Comparative study of the intake of toxic persistent and semi persistent pollutants through the consumption of fish and seafood from two modes of production (wild-caught and farmed). Sci. Total Environ. 2017, 575, 919–931. [Google Scholar] [CrossRef] [PubMed]
- Miniero, R.; Abate, V.; Brambilla, G.; Davoli, E.; De Felip, E.; De Filippis, S.P.; Dellatte, E.; De Luca, S.; Fanelli, R.; Fattore, E.; et al. Persistent toxic substances in Mediterranean aquatic species. Sci. Total Environ. 2014, 494–495, 18–27. [Google Scholar] [CrossRef]
- Bogdanović, T.; Ujević, I.; Sedak, M.; Listeš, E.; Šimat, V.; Petričević, S.; Poljak, V. As, Cd, Hg and Pb in four edible shellfish species from breeding and harvesting areas along the eastern Adriatic Coast, Croatia. Food Chem. 2014, 146, 197–203. [Google Scholar] [CrossRef]
- MacMillan, G.A.; Chetelat, J.; Heath, J.P.; Mickpegak, R.; Amyot, M. Rare earth elements in freshwater, marine, and terrestrial ecosystems in the eastern Canadian Arctic. Environ. Sci. Process. Impacts 2017, 19, 1336–1345. [Google Scholar] [CrossRef]
- Perrat, E.; Parant, M.; Py, J.S.; Rosin, C.; Cossu-Leguille, C. Bioaccumulation of gadolinium in freshwater bivalves. Environ. Sci. Pollut. Res. 2017, 24, 12405–12415. [Google Scholar] [CrossRef]
- Beyer, J.; Green, N.W.; Brooks, S.; Allan, I.J.; Ruus, A.; Gomes, T.; Brate, I.L.N.; Schoyen, M. Blue mussels (Mytilus edulis spp.) as sentinel organisms in coastal pollution monitoring: A review. Mar. Environ. Res. 2017, 130, 338–365. [Google Scholar] [CrossRef]
- Pieniak, Z.; Verbeke, W.; Scholderer, J. Health-related beliefs and consumer knowledge as determinants of fish consumption. J. Hum. Nutr. Diet. 2010, 23, 480–488. [Google Scholar] [CrossRef]
- Mol, S.; Alakavuk, D.U. Heavy metals in mussels (Mytilus galloprovincialis) from Marmara sea, Turkey. Biol Trace Elem. Res. 2011, 141, 184–191. [Google Scholar] [CrossRef]
- González Laxe, F.; Martín Palmero, F. El mercado de mejillón en España. Distribución y Consumo 2014, 102, 1–10. [Google Scholar]
- MAGRAMA (Ministerio de Agricultura, Alimentación y Medio Ambiente). El mercado del mejillón en España; Ministry of Agriculture, Food and Environment: Madrid, Spain, 2017; pp. 1–48.
- Olmedo, P.; Hernandez, A.F.; Pla, A.; Femia, P.; Navas-Acien, A.; Gil, F. Determination of essential elements (copper, manganese, selenium and zinc) in fish and shellfish samples. Risk and nutritional assessment and mercury-selenium balance. Food Chem. Toxicol. 2013, 62, 299–307. [Google Scholar] [CrossRef] [PubMed]
- Olmedo, P.; Pla, A.; Hernandez, A.F.; Barbier, F.; Ayouni, L.; Gil, F. Determination of toxic elements (mercury, cadmium, lead, tin and arsenic) in fish and shellfish samples. Risk assessment for the consumers. Environ. Int. 2013, 59, 63–72. [Google Scholar] [CrossRef]
- Hussain, M.; Mumtaz, S. E-waste: Impacts, issues and management strategies. Rev. Environ. Health 2014, 29, 53–58. [Google Scholar] [CrossRef]
- Tansel, B. From electronic consumer products to e-wastes: Global outlook, waste quantities, recycling challenges. Environ. Int. 2017, 98, 35–45. [Google Scholar] [CrossRef] [PubMed]
- AECOSAN. ENIDE: National Survey of Dietary Intake (2009–2010); Results on Consumption Data; Agencia Española de Seguridad Alimentaria y Nutrición; Gobierno de España: Madrid, Spain, 2011. [Google Scholar]
- EFSA. Dietary Reference Value Finder. Available online: https://www.efsa.europa.eu/en/interactive-pages/drvs (accessed on 22 February 2019).
- European Food Safety Authority (EFSA). Scientific Committee on Food Scientific Panel on Dietetic Products, Nutrition and Allergies; Tolerable Upper Intake Levels for Vitamins and Minerals; European Food Safety Authority: Parma, Italy, 2006; pp. 1–482. [Google Scholar]
- WHO. Assessment of Prenatal Exposure to Mercury: Standard Operating Procedures; WHO: Geneva, Switzerland, 2018. [Google Scholar]
- EPA. Integrated Risk Information System. Available online: https://www.epa.gov/iris (accessed on 22 February 2019).
- Zhuang, M.; Wang, L.; Wu, G.; Wang, K.; Jiang, X.; Liu, T.; Xiao, P.; Yu, L.; Jiang, Y.; Song, J.; et al. Health risk assessment of rare earth elements in cereals from mining area in Shandong, China. Sci. Rep. 2017, 7, 9772. [Google Scholar] [CrossRef]
- Zhuang, M.; Zhao, J.; Li, S.; Liu, D.; Wang, K.; Xiao, P.; Yu, L.; Jiang, Y.; Song, J.; Zhou, J.; et al. Concentrations and health risk assessment of rare earth elements in vegetables from mining area in Shandong, China. Chemosphere 2017, 168, 578–582. [Google Scholar] [CrossRef] [PubMed]
- Commission Regulation (EU) No 488/2014 of 12 May 2014 Amending Regulation (EC) No 1881/2006 as Regards Maximum Levels of Cadmium in Foodstuffs. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/PDF/?uri=OJ:JOL_2014_138_R_0011&from=EN (accessed on 11 February 2019).
- Commission Regulation (EC) No 1881/2006 of 19 December 2006 Setting Maximum Levels for Certain Contaminants in Foodstuffs. Available online: https://eur-lex.europa.eu/legal-content/EN/ALL/?uri=CELEX%3A32006R1881 (accessed on 11 February 2019).
- Commission Regulation (EU) 2015/1005 of 25 June 2015 Amending Regulation (EC) No 1881/2006 as Regards Maximum Levels of Lead in Certain Foodstuffs. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/?uri=uriserv%3AOJ.L_.2015.161.01.0009.01.ENG (accessed on 11 February 2019).
- EFSA. Dietary exposure to inorganic arsenic in the European population. EFSA J. 2014, 12, 1–68. [Google Scholar]
- ATSDR. ATSDR’s Substance Priority List. Available online: https://www.atsdr.cdc.gov/spl/ (accessed on 4 April 2017).
- Robinson, C.D.; Webster, L.; Martínez-Gómez, C.; Burgeot, T.; Gubbins, M.J.; Thain, J.E.; Vethaak, A.D.; McIntosh, A.D.; Hylland, K. Assessment of contaminant concentrations in sediments, fish and mussels sampled from the North Atlantic and European regional seas within the ICON project. Mar. Environ. Res. 2017, 124, 21–31. [Google Scholar] [CrossRef]
- Besada, V.; Sericano, J.L.; Schultze, F. An assessment of two decades of trace metals monitoring in wild mussels from the Northwest Atlantic and Cantabrian coastal areas of Spain, 1991–2011. Environ. Int. 2014, 71, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Besada, V.; Andrade, J.M.; Schultze, F.; González, J.J. Comparison of the 2000 and 2005 spatial distributions of heavy metals in wild mussels from the North-Atlantic Spanish coast. Ecotoxicol. Environ. Saf. 2011, 74, 373–381. [Google Scholar] [CrossRef]
- Chiffoleau, J.-F.; Cossa, D.; Auger, D.; Truquet, I. Trace metal distribution, partition and fluxes in the Seine estuary (France) in low discharge regime. Mar. Chem. 1994, 47, 145–158. [Google Scholar] [CrossRef]
- Squadrone, S.; Brizio, P.; Stella, C.; Prearo, M.; Pastorino, P.; Serracca, L.; Ercolini, C.; Abete, M.C. Presence of trace metals in aquaculture marine ecosystems of the northwestern Mediterranean Sea (Italy). Environ. Pollut. 2016, 215, 77–83. [Google Scholar] [CrossRef] [PubMed]
- Jović, M.; Stanković, S. Human exposure to trace metals and possible public health risks via consumption of mussels Mytilus galloprovincialis from the Adriatic coastal area. Food Chem. Toxicol. 2014, 70, 241–251. [Google Scholar] [CrossRef]
- Falcó, G.; Llobet, J.M.; Bocio, A.; Domingo, J.L. Daily Intake of Arsenic, Cadmium, Mercury, and Lead by Consumption of Edible Marine Species. J. Agric. Food Chem. 2006, 54, 6106–6112. [Google Scholar] [CrossRef]
- Yap, C.K.; Cheng, W.H.; Karami, A.; Ismail, A. Health risk assessments of heavy metal exposure via consumption of marine mussels collected from anthropogenic sites. Sci. Total Environ. 2016, 553, 285–296. [Google Scholar] [CrossRef] [PubMed]
- COMMISSION REGULATION (EC) No 710/2009 Amending Regulation (EC) No 889/2008 Laying down Detailed Rules for the Implementation of Council Regulation (EC) No 834/2007, as Regards Laying down Detailed Rules on Organic Aquaculture Animal and Seaweed Production. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/?uri=uriserv:OJ.L_.2009.204.01.0015.01.ENG (accessed on 25 March 2019).
- EFSA NDA Panel (EFSA Panel on Dietetic Products, Nutrition and Allergies). Scientific Opinion on Dietary Reference Values for selenium. EFSA J. 2014, 12, 3846. [Google Scholar] [CrossRef]
- Leyssens, L.; Vinck, B.; Van Der Straeten, C.; Wuyts, F.; Maes, L. Cobalt toxicity in humans-A review of the potential sources and systemic health effects. Toxicology 2017, 387, 43–56. [Google Scholar] [CrossRef]
- Watanabe, F.; Katsura, H.; Takenaka, S.; Enomoto, T.; Miyamoto, E.; Nakatsuka, T.; Nakano, Y. Characterization of vitamin B12 compounds from edible shellfish, clam, oyster, and mussel. Int. J. Food Sci. Nutr. 2001, 52, 263–268. [Google Scholar] [CrossRef] [PubMed]
- EFSA NDA Panel (EFSA Panel on Dietetic Products, Nutrition and Allergies). Scientific Opinion on Dietary Reference Values for zinc. EFSA J. 2014, 12, 3844. [Google Scholar] [CrossRef]
- EFSA NDA Panel (EFSA Panel on Dietetic Products, Nutrition and Allergies). Scientific Opinion on Dietary Reference Values for molybdenum. EFSA J. 2013, 11, 3333. [Google Scholar] [CrossRef]
- Mertz, W. The essential trace elements. Science 1981, 213, 1332–1338. [Google Scholar] [CrossRef] [PubMed]
- Graci, S.; Collura, R.; Cammilleri, G.; Buscemi, M.D.; Giangrosso, G.; Principato, D.; Gervasi, T.; Cicero, N.; Ferrantelli, V. Mercury accumulation in Mediterranean Fish and Cephalopods Species of Sicilian coasts: Correlation between pollution and the presence of Anisakis parasites. Nat. Prod. Res. 2017, 31, 1156–1162. [Google Scholar] [CrossRef] [PubMed]
- Cammilleri, G.; Vazzana, M.; Arizza, V.; Giunta, F.; Vella, A.; Lo Dico, G.; Giaccone, V.; Giofre, S.V.; Giangrosso, G.; Cicero, N.; et al. Mercury in fish products: what’s the best for consumers between bluefin tuna and yellowfin tuna? Nat. Prod. Res. 2018, 32, 457–462. [Google Scholar] [CrossRef]
- EFSA. Scientific Opinion on the risk for public health related to the presence of mercury and methylmercury in food. EFSA J. 2012, 10, 2985–3226. [Google Scholar]
- Pagano, G.; Aliberti, F.; Guida, M.; Oral, R.; Siciliano, A.; Trifuoggi, M.; Tommasi, F. Rare earth elements in human and animal health: State of art and research priorities. Environ. Res. 2015, 142, 215–220. [Google Scholar] [CrossRef]
- Pagano, G.; Guida, M.; Tommasi, F.; Oral, R. Health effects and toxicity mechanisms of rare earth elements-Knowledge gaps and research prospects. Ecotoxicol. Environ. Saf. 2015, 115, 40–48. [Google Scholar] [CrossRef]
- Henriquez-Hernandez, L.A.; Boada, L.D.; Carranza, C.; Perez-Arellano, J.L.; Gonzalez-Antuna, A.; Camacho, M.; Almeida-Gonzalez, M.; Zumbado, M.; Luzardo, O.P. Blood levels of toxic metals and rare earth elements commonly found in e-waste may exert subtle effects on hemoglobin concentration in sub-Saharan immigrants. Environ. Int. 2017, 109, 20–28. [Google Scholar] [CrossRef]
- Mestre, N.C.; Sousa, V.S.; Rocha, T.L.; Bebianno, M.J. Ecotoxicity of rare earths in the marine mussel Mytilus galloprovincialis and a preliminary approach to assess environmental risk. Ecotoxicology 2019, 28, 294–301. [Google Scholar] [CrossRef]
| Galicia | Chile | New Zealand | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Type of Brand | Mode of Production | Type of Brand | Mode of Production | Type of Brand | Mode of Production | |||||||
| Name | Store | Conventional | Organic | Name | Store | Conventional | Organic | Name | Store | Conventional | Organic | |
| Preserved (n = 88) | 38 | 34 | 72 | - | 16 | - | 16 | - | - | - | - | |
| Frozen (n = 80) | 36 | - | 28 | 8 | 34 | - | 32 | - | 12 | - | 12 | |
| Fresh (n = 20) | 40 | - | 40 | - | - | - | - | - | - | - | - | - |
| Preserved Mussels (n = 88) | Frozen Mussels (n = 80) | Fresh Mussels (n = 20) | p Value a | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Element | Mean ± SD | Median | P25–P75 | Mean ± SD | Median | P25–P75 | Mean ± SD | Median | P25–P75 | Preserved vs. Frozen | Preserved vs. Fresh | Frozen vs. Fresh |
| Fe | 34,945.1 ± 11,496.8 | 33,376.0 | 25,848.3–41,727.6 | 45,965.2 ± 44,720.5 | 32,310.0 | 26,713.1–50,700.1 | 24,427.6 ± 5553.9 | 22,924.9 | 20,137.1–26,850.9 | n.s. | <0.01 | <0.01 |
| Zn | 46,011.4 ± 24,034.8 | 38,961.6 | 33,105.8–48,439.9 | 47,141.4 ± 17,788.6 | 46,231.2 | 37,356.5–58,914.3 | 56,624.1 ± 14,019.9 | 54,712.7 | 45,823.5–70,423.9 | <0.01 | <0.005 | <0.05 |
| Cu | 1332.5 ± 383.1 | 1268.8 | 1028.4–1492.1 | 1564.6 ± 340.9 | 1529.7 | 1349.5–1831.3 | 1235.9 ± 195.5 | 1313.7 | 1035.5–1405.7 | <0.05 | n.s. | <0.01 |
| Se | 646.0 ± 200.8 | 599.6 | 538.3–683.3 | 1075.9 ± 187.9 | 1085.6 | 949.0–1229.2 | 1223.8 ± 125.1 | 1274.6 | 1129.5–1315.2 | <0.005 | <0.005 | n.s. |
| Mn | 1251.0 ± 261.8 | 1227.7 | 1057.9–1407.0 | 1726.8 ± 812.5 | 1424.4 | 1170.0–2079.5 | 1479.8 ± 298.8 | 1569.1 | 1249.4–1702.4 | <0.05 | <0.01 | <0.05 |
| Mo | 271.4 ± 279.9 | 151.3 | 120.3–288.1 | 187.6 ± 113.6 | 142.4 | 106.7–208.5 | 187.4 ± 45.7 | 201.9 | 183.4–216.0 | n.s. | n.s. | n.s. |
| Co | 40.8 ± 11.0 | 40.6 | 33.6–46.5 | 66.4 ± 67.2 | 50.2 | 42.2–75.1 | 74.2 ± 16.5 | 74.1 | 58.9–91.0 | <0.01 | <0.005 | <0.005 |
| Preserved Mussels (n = 88) | Frozen Mussels (n = 80) | Fresh Mussels (n = 20) | p Value a | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Element | Mean ± SD | Median | P25–P75 | Mean ± SD | Median | P25–P75 | Mean ± SD | Median | P25–P75 | Preserved vs. Frozen | Preserved vs. Fresh | Frozen vs. Fresh |
| Major Toxic Elements | ||||||||||||
| As | 1869.8 ± 634.8 | 1641.9 | 1522.5–1973.1 | 4008.1 ± 59.2 | 3377.3 | 3017.5–4992.0 | 3696.5 ± 432.8 | 3785.0 | 3395.5–3994.6 | <0.005 | <0.005 | <0.05 |
| Cd | 241.9 ± 92.2 | 232.1 | 169.1–312.8 | 463.3 ± 306.4 | 353.3 | 252.1–606.6 | 281.0 ± 89.4 | 275.3 | 251.5–335.7 | <0.01 | <0.05 | <0.005 |
| Hg | 12.4 ± 9.2 | 11.2 | 6.4–14.4 | 14.9 ± 8.6 | 16.2 | 6.3–20.6 | 23.2 ± 3.6 | 22.5 | 20.2–26.6 | <0.01 | <0.001 | <0.01 |
| Pb | 167.5 ± 87.0 | 184.5 | 137.3–221.8 | 176.8 ± 181.0 | 111.5 | 21.1–271.9 | 249.4 ± 64.8 | 255.9 | 190.7–296.2 | <0.01 | <0.001 | <0.001 |
| Other Toxic or Potentially Toxic Elements | ||||||||||||
| Ag | 5.9 ± 2.9 | 5.4 | 3.8–7.4 | 7.6 ± 3.6 | 7.1 | 5.1–8.6 | 6.2 ± 2.3 | 5.8 | 4.6–7.5 | n.s. | n.s. | n.s. |
| Al | 49,278.1 ± 36,202.8 | 38,878.3 | 22,886.8–70,388.9 | 76,417.8 ± 11,1251.9 | 35,160.6 | 24,504.6–87,869.5 | 30,808.6 ± 31,245.7 | 21,122.7 | 7570.1–37,324.1 | <0.01 | <0.001 | <0.001 |
| Ba | 430.0 ± 303.2 | 318.4 | 202.8–618.4 | 708.2 ± 1259.2 | 353.9 | 190.0–659.4 | 238.6 ± 169.6 | 194.8 | 138.2–281.2 | <0.01 | <0.001 | <0.001 |
| Be | 3.0 ± 2.2 | 2.7 | 1.1–4.2 | 3.3 ± 3.9 | 2.4 | 0.8–4.3 | 3.1 ± 1.7 | 2.8 | 1.7–4.1 | n.s. | n.s. | n.s. |
| Cr | 110.4 ± 47.5 | 101.3 | 78.0–135.6 | 109.4 ± 113.6 | 91.0 | 61.7–136.4 | 123.7 ± 28.9 | 115.5 | 108.9–129.5 | <0.05 | <0.05 | <0.05 |
| Ni | 106.2 ± 29.4 | 103.2 | 86.4–114.6 | 140.1 ± 37.5 | 125.7 | 95.6–171.9 | 196.2 ± 59.8 | 181.1 | 164.2–235.8 | <0.01 | <0.001 | <0.005 |
| Pd | 0.07 ± 0.04 | 0.06 | 0.04–0.08 | 0.08 ± 0.02 | 0.08 | 0.07–0.1 | 0.1 ± 0.04 | 1.1 | 0.8–4.3 | n.s. | n.s. | n.s. |
| Sb | 7.3 ± 26.3 | 1.0 | 0.05–1.7 | 1.2 ± 0.5 | 1.1 | 0.08–2.1 | 3.8 ± 1.3 | 3.5 | 2.8–4.6 | n.s. | <0.01 | <0.005 |
| Sn | 26.2 ± 13.2 | 24.0 | 16.7–33.1 | 9.6 ± 9.3 | 6.0 | 1.5–18.0 | 11.7 ± 6.0 | 9.6 | 7.1–15.5 | <0.001 | <0.001 | <0.05 |
| Sr | 6403.4 ± 3618.4 | 5764.6 | 5006.4–6607.25 | 7169.8 ± 1352.8 | 6871.1 | 6293.6–7666.4 | 7903.9 ± 2478.6 | 8837.4 | 6395.1–9575.5 | <0.01 | <0.001 | <0.005 |
| Th | 7.5 ± 5.9 | 6.3 | 2.5–10.8 | 13.5 ± 24.2 | 5.5 | 2.0–13.7 | 8.3 ± 5.4 | 6.9 | 4.6–9.7 | <0.05 | n.s. | <0.01 |
| Tl | 1.4 ± 0.6 | 1.3 | 0.9–1.8 | 2.6 ± 2.4 | 1.8 | 1.2–2.5 | 1.3 ± 0.7 | 1.1 | 0.8–1.6 | n.s. | n.s. | n.s. |
| U | 34.5 ± 8.9 | 33.3 | 28.5–39.9 | 36.7 ± 12.3 | 35.4 | 29.2–41.8 | 34.3 ± 14.3 | 31.8 | 22.3–48.1 | n.s. | n.s. | n.s. |
| Sum REE b | 184.8 ± 133.9 | 144.9 | 102.1–247.1 | 330.3 ± 412.1 | 198.7 | 155.5–276.2 | 1913.2 ± 958.3 | 1570.6 | 1248.2–2485.42 | <0.01 | <0.001 | <0.001 |
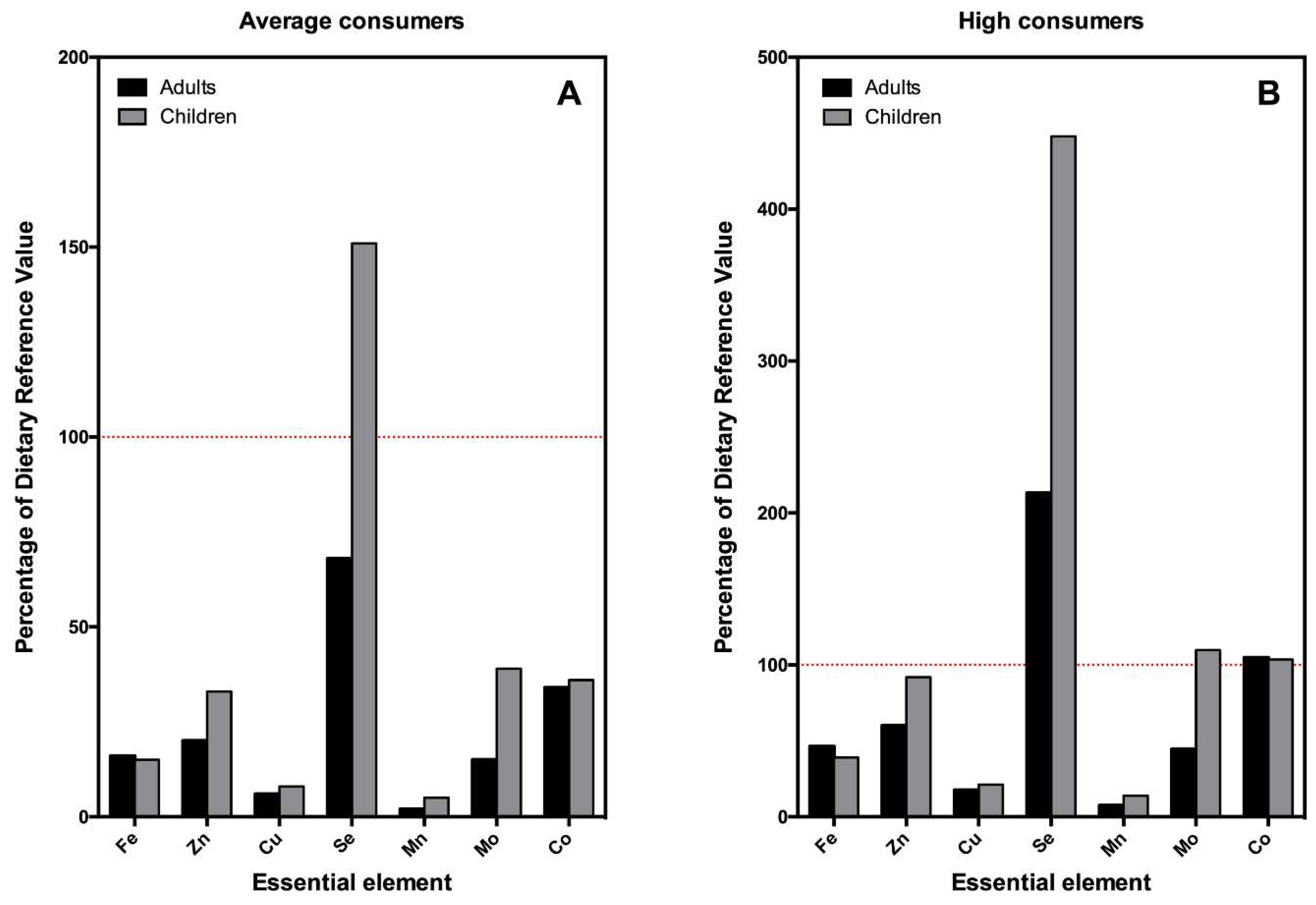
| Adults (>17 y.o.)—68.48 kg/bw—Both Genders | |||
| Essential Element | Dietary Reference Value a | EDI Average Consumer (µg/kg bw/day) b | EDI High Consumer (P97.5) (µg/kg bw/day) c |
| Fe | 160.63 d | 24.89 | 74.41 |
| Zn | 175.23 d | 34.45 | 105.54 |
| Cu | 18.98 e | 1.09 | 3.33 |
| Se | 1.02 e | 0.69 | 2.18 |
| Mn | 43.81 e | 1.06 | 3.25 |
| Mo | 0.95 e | 0.14 | 0.42 |
| Co | 0.12 d | 0.04 | 0.12 |
| Children (7–12 y.o.)—34.48 kg/bw—Both Genders | |||
| Essential Element | Dietary Reference Value a | EDI Average Consumer (µg/kg bw/day) b | EDI High Consumer (P97.5) (µg/kg bw/day) c |
| Fe | 160.63 d | 23.57 | 62.72 |
| Zn | 108.06 d | 35.34 | 99.31 |
| Cu | 14.60 e | 1.11 | 3.10 |
| Se | 0.51 e | 0.77 | 2.29 |
| Mn | 21.90 e | 1.09 | 3.05 |
| Mo | 0.37 e | 0.14 | 0.40 |
| Co | 0.12 d | 0.04 | 0.12 |
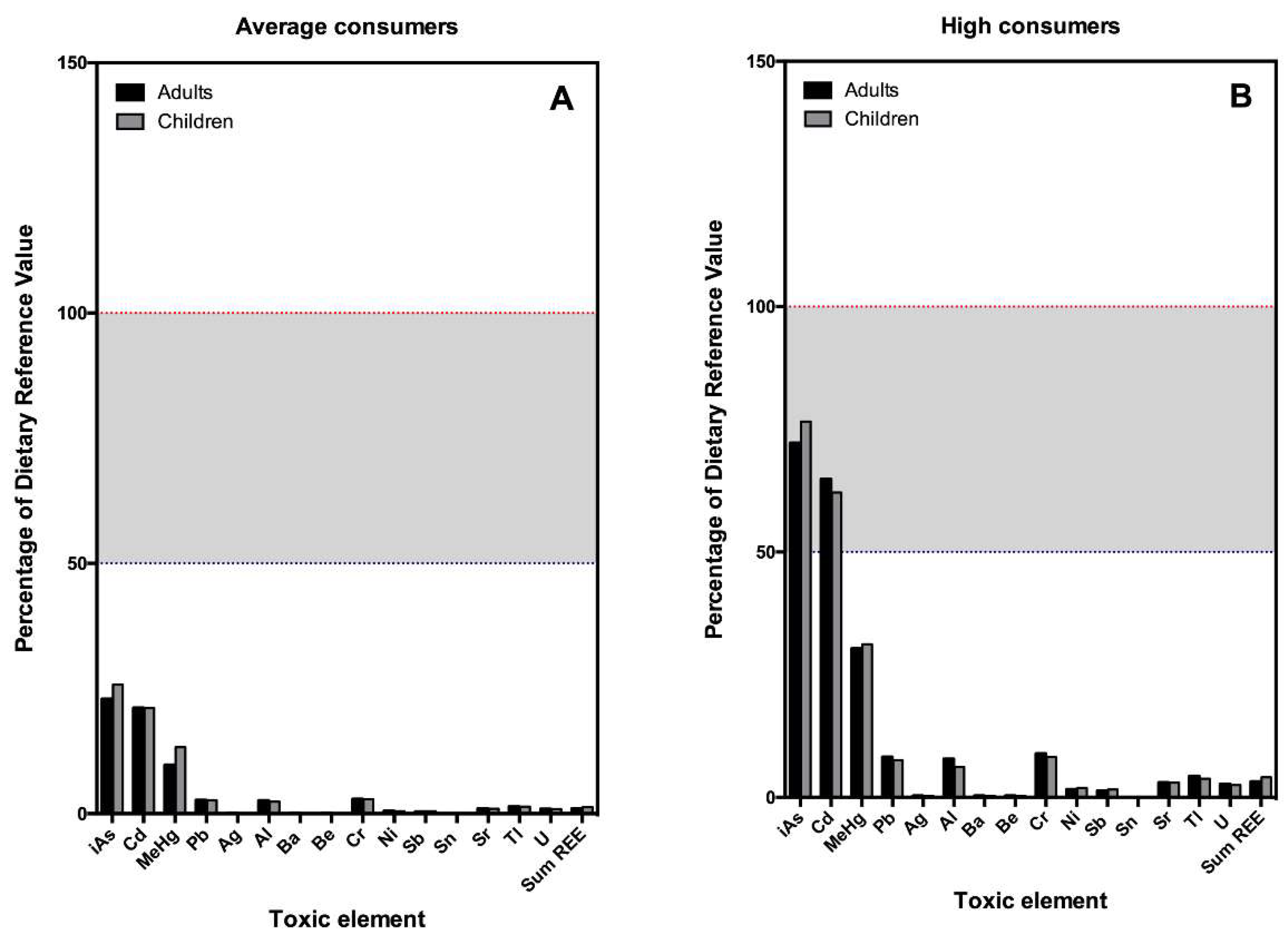
| Adults (>17 y.o.)—68.48 kg/bw—Both Genders | |||
| Toxic Element | Risk Reference Value a | EDI Average Consumer (µg/kg bw/day) b | EDI High Consumer (P97.5) (µg/kg bw/day) c |
| iAs d | 0.30 | 0.07 | 0.22 |
| Cd | 1.00 | 0.21 | 0.65 |
| MeHg e | 0.10 | 0.01 | 0.03 |
| Pb | 6.00 | 0.16 | 0.49 |
| Children (7–12 y.o.)—34.48 kg/bw—Both Genders | |||
| Toxic Element | Risk Reference Value a | EDI Average Consumer (µg/kg bw/day) b | EDI High Consumer (P97.5) (µg/kg bw/day) c |
| iAs d | 0.30 | 0.08 | 0.23 |
| Cd | 1.00 | 0.21 | 0.62 |
| MeHg e | 0.10 | 0.01 | 0.03 |
| Pb | 6.00 | 0.16 | 0.46 |
| Adults (>17 y.o.)—68.48 kg bw—Both Genders | |||
| Element | Risk Reference Value a | EDI Average Consumer (µg/kg bw/day) b | EDI High Consumer (P97.5) (µg/kg bw/day) c |
| Ag | 5 | 0.00 | 0.01 |
| Al | 1000 | 26.48 | 78.21 |
| Ba | 200 | 0.22 | 0.66 |
| Be | 2 | 0.00 | 0.01 |
| Cd | 3,00 | 0,09 | 0,27 |
| Ni | 20,00 | 0,10 | 0,32 |
| Pd | N.A. | 0.00 | 0.00 |
| Sb | 0.4 | 0.00 | 0.01 |
| Sn | 600 | 0.01 | 0.04 |
| Sr | 600 | 5.77 | 17.96 |
| Th | NA | 0.01 | 0.02 |
| Tl | 0.07 | 0.00 | 0.00 |
| U | 3 | 0.03 | 0.08 |
| Sum REE | 61 | 0.59 | 1.97 |
| Children (7–12 y.o.)—34.48 kg bw—Both Genders | |||
| Element | Risk Reference Value a | EDI Average Consumer (µg/kg bw/day) b | EDI High Consumer (P97.5) (µg/kg bw/day) c |
| Ag | 5 | 0.00 | 0.01 |
| Al | 1000 | 24.04 | 61.99 |
| Ba | 200 | 0.21 | 0.54 |
| Be | 2 | 0.00 | 0.01 |
| Cd | 3.0 | 0.09 | 0.25 |
| Ni | 20.00 | 0.11 | 0.31 |
| Pd | NA | 0.00 | 0.00 |
| Sb | 0.4 | 0.00 | 0.01 |
| Sn | 600 | 0.01 | 0.03 |
| Sr | 600 | 6.22 | 18.01 |
| Th | NA | 0.01 | 0.02 |
| Tl | 0.07 | 0.00 | 0.00 |
| U | 3 | 0.03 | 0.07 |
| Sum REE d | 61 | 0.78 | 2.51 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rodríguez-Hernández, Á.; Zumbado, M.; Henríquez-Hernández, L.A.; Boada, L.D.; Luzardo, O.P. Dietary Intake of Essential, Toxic, and Potentially Toxic Elements from Mussels (Mytilus spp.) in the Spanish Population: A Nutritional Assessment. Nutrients 2019, 11, 864. https://doi.org/10.3390/nu11040864
Rodríguez-Hernández Á, Zumbado M, Henríquez-Hernández LA, Boada LD, Luzardo OP. Dietary Intake of Essential, Toxic, and Potentially Toxic Elements from Mussels (Mytilus spp.) in the Spanish Population: A Nutritional Assessment. Nutrients. 2019; 11(4):864. https://doi.org/10.3390/nu11040864
Chicago/Turabian StyleRodríguez-Hernández, Ángel, Manuel Zumbado, Luis Alberto Henríquez-Hernández, Luis D. Boada, and Octavio P. Luzardo. 2019. "Dietary Intake of Essential, Toxic, and Potentially Toxic Elements from Mussels (Mytilus spp.) in the Spanish Population: A Nutritional Assessment" Nutrients 11, no. 4: 864. https://doi.org/10.3390/nu11040864
APA StyleRodríguez-Hernández, Á., Zumbado, M., Henríquez-Hernández, L. A., Boada, L. D., & Luzardo, O. P. (2019). Dietary Intake of Essential, Toxic, and Potentially Toxic Elements from Mussels (Mytilus spp.) in the Spanish Population: A Nutritional Assessment. Nutrients, 11(4), 864. https://doi.org/10.3390/nu11040864








